Abstract
Although it is thought that there is a close relationship between Notch signal and preterm birth, the functioning of this mechanism in the cervix is unknown. The efficacy of surfactants and prostaglandin inhibitors in preterm labor is also still unclear. In this study, 48 female CD-1 mice were distributed to pregnant control (PC), Sham, PBS, indomethacin (2 mg/kg; intraperitoneally), lipopolysaccharides (LPS) (25 μg/100 μl; intrauterine), LPS + IND, and Surfactant Protein A Block (SP-A Block: SP-A B; the anti-SP-A antibody was applied 20 µg/100μl; intrauterine) groups. Tissues were examined by immunohistochemistry, immunofluorescence, and Western blot analysis. LPS administration increased the expression of N1 Dll-1 and Jagged-2 (Jag-2). Although Toll-like receptor (Tlr)-2 significantly increased in the LPS-treated and SP-A-blocked groups, Tlr-4 significantly increased only in the LPS-exposed groups. It was observed that Jag-2 is specifically expressed by mast cells. Overall, this experimental model shows that some protein responses increase throughout the uterus, starting at a specific point on the cervix epithelium. Surfactant Protein A, which we observed to be significantly reduced by LPS, may be associated with the regulation of the epithelial response, especially during preterm delivery due to infection. On the contrary, prostaglandin inhibitors can be considered an option to delay infection-related preterm labor with their dose-dependent effects. Finally, the link between mast cells and Jag-2 could potentially be a control switch for preterm birth:
Introduction
Premature birth is the onset of labor before 37 weeks and is divided into three subcategories; births <28 weeks are defined as extremely premature; between 28 and 32 weeks, very premature; and 32–37 weeks, premature. 1 About 15 million babies are born prematurely every year, and the preterm birth rate is around 11% worldwide. Premature birth is the primary cause of death, accounting for 18% of all deaths among children under 5 years of age and 35% of all deaths among newborns (<28 days). 2
Although advances in the newborn and child care fields and administration of prenatal steroids to women at risk improve the outcomes of premature babies, the incidence of preterm birth still rate is significantly high. 3 Intrauterine (IU) infections caused by bacteria are one of the important risk factors for preterm labor, 4 and the role of the infection in preterm delivery has not been fully determined. 5 The most common mechanism underlying infection-induced preterm labor is increased colonization of vaginal pathogens in the gestational cavity. 6 Lipopolysaccharide (LPS)-induced uterus and placenta studies in the literature have indicated a possible relationship between the Notch signaling pathway and preterm birth. 7
Notch receptors, which are unusual transmembrane receptors activated by their membrane-bound ligands, are thought to be preserved in all organisms from the simplest to the most advanced. 8 A Notch is a cell-surface receptor that transmits short-range signals to neighboring cells by interacting with transmembrane ligands. Notch ligands are characterized by an N-terminal DSL (Delta, Serrate, and LAG-2) domain required for interaction with Notch receptors.9,10 Ligands are classified as Delta (Delta-like in humans) and Serrate (Jagged in humans) depending on the presence or absence of cysteine-rich (CR) structures. Cells that initiate the signal express the Notch ligands belonging to the Delta-like (Dll-1, Dll-3, Dll-4) or Jagged (Jag-1, Jag-2) family. Cells that have received the Notch signal express Notch receptors. Notch signaling is activated by direct cell–cell contact involving ligand-receptor binding, which is followed by proteolytic cleavage of the intracellular domain of Notch (NICD).11,12
When the entire birth process is reviewed, the cervix is as crucial as the uterus. Today, theories put forward in the process of reshaping uterovaginal embryology suggest that the cervix should be evaluated not only as a part of the uterus but also as a gateway between two structures of different embryological origins. 13 The maturation of the cervix is a physiological process that includes biochemical and structural changes of connective tissue and is very important in the initiation and completion of delivery. 14
One of the agents frequently used for the maturation of the cervix is prostaglandins (PGs). 15 It is argued that decidual and myometrial PGs primarily trigger uterine contractions. 16 PGs increase the amount of submucosal water in cervix tissue, and collagen fibers dissolve. These changes are similar to those seen in pregnant women during normal birth and preterm birth. 17 Nonsteroidal anti-inflammatory agents such as indomethacin (IND) are prostaglandin biosynthesis inhibitors that are effective in the body by inhibiting the enzyme cyclooxygenase (Cox), and the cytokines and PGs produced during birth are similar to the ones produced during the inflammatory process. 18 IND is thought to delay premature birth by suppressing the production of PGs in different parts of the uterus. In most randomized controlled trials comparing the IND effect, IND has delayed premature birth for up to 48 hr.19–22
Different timing of Cox metabolite production by mast cells (MCs) in tissue may contribute to longer preservation of the inflammatory environment. It is thought that MC plays a role in the early phase response, and this early phase response can be blocked by the Cox-1 inhibitor. 23 It has been shown that MCs are able to initiate contractions in the myometrium of pregnant women. 24 Therefore, activation of MCs can be significant in the preterm labor process. 25 Inflammation is associated with preterm birth 26 and has shown that the number of macrophages in the cervix has increased in women giving birth early.27,28
When macrophages are induced with LPS or Th1 cytokines, M1 macrophage polarization occurs. They express surface receptors such as Toll-like receptor (Tlr)-2 and Tlr-4, 29 which are strongly associated with the pathogenesis of inflammatory diseases. 30 The extracellular domains of TLRs exhibit high structural diversity and have been shown to recognize a wide variety of pathogens, including bacteria. 31 Moreover, TLRs need to maintain a stable balance between activation and inhibition to avoid harmful inflammatory responses. Although the signaling cascade typically produces proinflammatory cytokines, it can also inhibit the proinflammatory response.32,33 Tlr-4 is a member of the TLR family and is activated by bacterial LPS, the main molecular component of the cell wall of gram-negative bacteria.34,35 Although the role of Tlr-2 and Tlr-4 in the cervix is unclear, few studies are showing increased Tlr-2 expression in the endocervix after infection 36 or Tlr-4 expression in cervical carcinoma. 37 However, these findings are not cervix findings during preterm delivery.
Some substances produced by the host may bind TLRs. Surfactant Protein A (SP-A) is an endogenous ligand for Tlr-2 and Tlr-4. 32 These TLRs are particularly related to preterm labor.38,39 Depending on the various conditions and the type of receptor affected, SP-A can act both proinflammatory and anti-inflammatory.40,41 SP-A is synthesized by fetal alveolar type II cells starting from the 28th week of pregnancy42,43 and is vital for optimal pulmonary function. 44 SP-A is a PGF2a inhibitor, 45 and SP-A mutant mice are prone to infection. 40
This study aims to analyze the molecular mechanism of SP-A and IND in mice in which we surgically model the infection-mediated preterm labor process caused by LPS and to determine the potential for use as a treating agent at preterm labor. In our previous study, N1, JAG-2, and DLL-1 were elevated in the human cervix in misoprostol-induced preterm labor 27 (a synthetic prostaglandin E1 analogue). 46 In this study, we wanted to investigate the status of Notch activity in the cervix after LPS induction. In addition, we questioned the existence of a Tlr-mediated interaction.
We hypothesized that Notch activity would increase in the cervix after LPS induction and that the potential treatment agents we investigated might act by suppressing this signal.
Materials and Methods
Experimental Design
This study used 48 female CD-1 mice that had not mated before (n=6 mice in each experimental group). The day when the vaginal plug was observed was counted as embryonic day 0.5 (E0.5). Intraperitoneal (IP) IND (Cox-1/Cox-2 inhibitor) and IU anti-SP-A antibody [SP-A blocked (SP-A B)] were administered on E14.5 day of pregnancy (as the placenta completed its formation during this period in mice). 47 At this time point, it was also considered that there is a possible transition from M1 to M2 macrophages. 48 In all, 25 µg/100 µl LPS 49 (LPS-Escherichia coli O111: B4; L2630, 10 mg; Sigma-Aldrich (U.S)) was administered to pregnant mice to induce preterm labor (IU). IND (I7378; Sigma-Aldrich (U.S)) was applied at a dose of 2 mg/kg/mouse IP for 2 days to IND control and IND + LPS groups (injection was performed twice with 24-hr interval). This dose was considered to be a less harmful dose in the literature. 50 The second day dose of IND was given 5 hr before LPS administration in the LPS + IND group. The mean half-life of IND is estimated to be about 4.5 hr, 51 and this period has been taken into account in practice. The total volume for each application is 100 µl in PBS (10010023, 1X, Ca++, Mg-free, pH 7.4; Gibco (U.S); Thermo-Fisher (U.S)). In this study, the Ca++-free buffer was preferred because the presence of Ca++ increases SP-A’s self-aggregation and similarly affects binding to LPS.52,53 Anti-SP-A of 20 µg/100 µl (ab115791; Abcam (U.S)) was administered (IU) only in one horn as the dose at which adequate response 54 could be observed. Mice were administered 0.016 ml/g of avertin in PBS proportional to their body weight (2.5% tribromoethyl alcohol, T48402; Sigma-Aldrich and 2.5% 2-methyl-2-butanol, 240486; Sigma-Aldrich (U.S)) for anesthesia. Before use, the working solution was filtered through a 0.22-μm sterile filter (SLGV033RS; Merck-Millipore (German)). All IU applications were implemented between two gestational sacs closest to the cervix (Fig. 9A and A′). 5.0 Vicryl (Ethicon) was used for surgical suture procedures. The half-life of LPS has been determined as approximately 12 hr. 55 The birth was observed within 12–24 hr after LPS administration. Subsequently, the cervix tissues were removed (E15.5) for experiments.
Immunohistochemical Analysis
Cervix tissues were fixed for 24 hr in 10% formalin (818708; Merck). Tissues were washed with tap water for 3 hr and then were dehydrated in a graded series of ethanol (70%, 80%, and 90%). Followed by incubation in 100% ethanol (459844; Sigma-Aldrich) for 3 hr, the tissues were incubated in xylene (534056; Sigma-Aldrich) for about 5 min. After embedding in paraffin, the tissue blocks were cut into serial sections with a thickness of 5 µm. A 1× PBS (P4417-100TAB; Sigma-Aldrich) solution was prepared for washing the sections. Antigen retrieval was carried out by microwaving in citrate buffer (100244; Merck) buffer. Three percent H2O2 solution (106009; Merck and 18312; Sigma) was used to eliminate endogenous peroxidase activity, followed by Ultra V Block for 7 min at room temperature (TA-125-UB; Thermo Scientific). The following primary and secondary antibodies were used to determine the cellular localization of protein:
Anti-SP-A (115791, 1/150; Abcam), Tlr-2 (PA5-11592, 1/100; Thermo-Fisher), Tlr-4 (13556, 1/150; Abcam), N1 (8925, 1/150; Abcam), Dll-1 (10554, 1/150; Abcam), Jag-2 (109627, 1/150; Abcam), MC tryriptase (MCT; bios-2725R; 1/150), neutrophil/monocyte/macrophage marker CD-11b (133357, 1/100; Abcam), secondary antibody: goat anti-rabbit (vector, BA-1000; 1/500), and isotype control (vector, I-1000) were used. Diaminobenzidine tablet (D4168; Sigma) was used as chromogen, and the sections were counterstained with Mayer’s hemalum solution (109249; Merck). Sections were examined with an Olympus CX43 Microscope (Japan), and images were captured.
Immunofluorescence Analysis
After basic immunohistochemistry (IHC) staining steps, the sections washed with PBS were treated with fluorescent secondary antibodies (donkey anti-rabbit, Alexa Fluor 647, ab150075, red and goat anti-rabbit, Alexa Fluor 488, ab150077, green) for 45 min. Sections washed again with PBS were covered with DAPI (D8417; Sigma). Sections were examined under the Olympus BX61fluorescence microscope (Japan).
Protein Extraction and Western Blot (WB) Analysis
To prepare lysates of cervix tissues, tissues were homogenized with lysis buffer solution (Tris, 108387; Merck) and protease inhibitor cocktail (P8340; Sigma). The protein concentration of the lysates was measured with the BCA Protein Assay Kit (23225; Pierce). Each well was loaded with 50 µg of protein. SDS-polyacrylamide gel electrophoresis was performed using 4–12% Bolt Bis-Tris Plus (NW04120BOX; Invitrogen, 120 V, 60 min) gradient gels. The gels were then transferred onto nitrocellulose membrane using the iBlot Dry Transfer system (IB1001; Invitrogen). Membranes were blocked with 5% non-fat dry milk (Blotting-Grade Blocker, 1706404; Bio-Rad) at room temperature for 1 hr and then incubated with primary antibody overnight at 4C. Primers are as follows:
Anti-SP-A (115791, 1/500; Abcam), Tlr-2 (PA5-11592, 1/200; Thermo-Fisher), Tlr-4 (13556, 1/200; Abcam), N1 (8925, 1/500; Abcam), Dll-1 (10554, 1/500; Abcam), Jag-2 (109627, 1/500; Abcam) and beta-actin (13E5, 1/1000; Cell Signal).
After incubation, the membranes were washed with Tris-buffered saline containing 0.1% Tween 20 (TBS-T; 1% Tween-20, 822184; Merck). After washes, the membranes were incubated for 1 hr at room temperature with a horseradish peroxidase–labeled anti-rabbit (1/2000) secondary antibody (PI-1000; Vector). After rewashing, the membrane was incubated with Super Signal Chemiluminescence (CL-HRP, 34580; Thermo Scientific) for 5 min. Afterward, membranes were transferred to the hyper film (Amersham Hyperfilm ECL, 45-001-504; Fisher Scientific) in the darkroom.
Statistical Analysis
Immunohistochemical staining intensities were analyzed with Image J software (1.52 R; National Institutes of Health) on three randomly selected photographs from each of the experimental groups. Similarly, the WB band intensity value was determined by measuring each band with Image J. Differences in expression between groups were calculated using ANOVA and Sidak’s test (for comparison of multiple variables). Differences between groups were accepted to be statistically significant when the p value was <0.05. GraphPad Prism 8 was used for all statistical analyses. The significant difference between groups is indicated by different letters on the column. The differences between the group mean bearing the same letter were not significant. In the H-score analysis, the regions were evaluated by giving numerical values between 0 and 3 [0: (–), 1: weak, 2: medium, 3: strong] according to the staining intensity. The average for the whole tissue density was given graphically.
Bio Render drawing program was used to create (Fig. 10) drawing, and the general structure of the cervix is summarized in Supplemental Fig. 1. Supplemental Fig. 1 is a hematoxylin and eosin staining diagram created by magnifying 360° directly on the staining slide using Olympus BX 43 microscope (Japan) and Kameram 5 Argenit software (Turkey).
Results
SP-A Is Expressed by the Cervix Epithelium Starting From a Certain Point During the Pregnancy Period, and LPS Reduces SP-A Expression
When SP-A expression was evaluated in all groups, there was almost no expression except in the ectocervical surface epithelium in the non-pregnant group. There was a similar situation in the uterine and glandular epithelium (Fig. 6A). In pregnant groups, SP-A expression was started after a certain point in the cervix, and increased expression was observed, especially in the cervical-uterine transformation zone (Fig. 1C–U, expression beginning point; orange arrowheads). When the transformation zone epithelium is examined closely, they were not only very heavily stained cells with SP-A antibody (Fig. 1J, P, and V, blue arrows) but also unstained cells (Fig. 1J, P, and V, red arrows). These cells seem morphologically different, even in negative control cervix tissues whose primary antibody has not been added during the staining (Fig. 1#, red arrows). The properties or exact functions of these cells are not understood. SP-A staining intensity was lower in the LPS group (Figs. 1K and L, and 6A). There was weak immunostaining on the surface of the epithelium in the uterus neck (Fig. 1T, pink arrowhead) where SP-A was blocked (Fig. 1T, red star). However, in the non-blocked horn [Fig. 1T (small image), blue star], epithelial expression was still present [Fig. 1T (small image), brown arrowhead]. It was observed that the epithelial folds were flattened in the SP-A-blocked uterus horn (Fig. 1T, pink arrowhead).
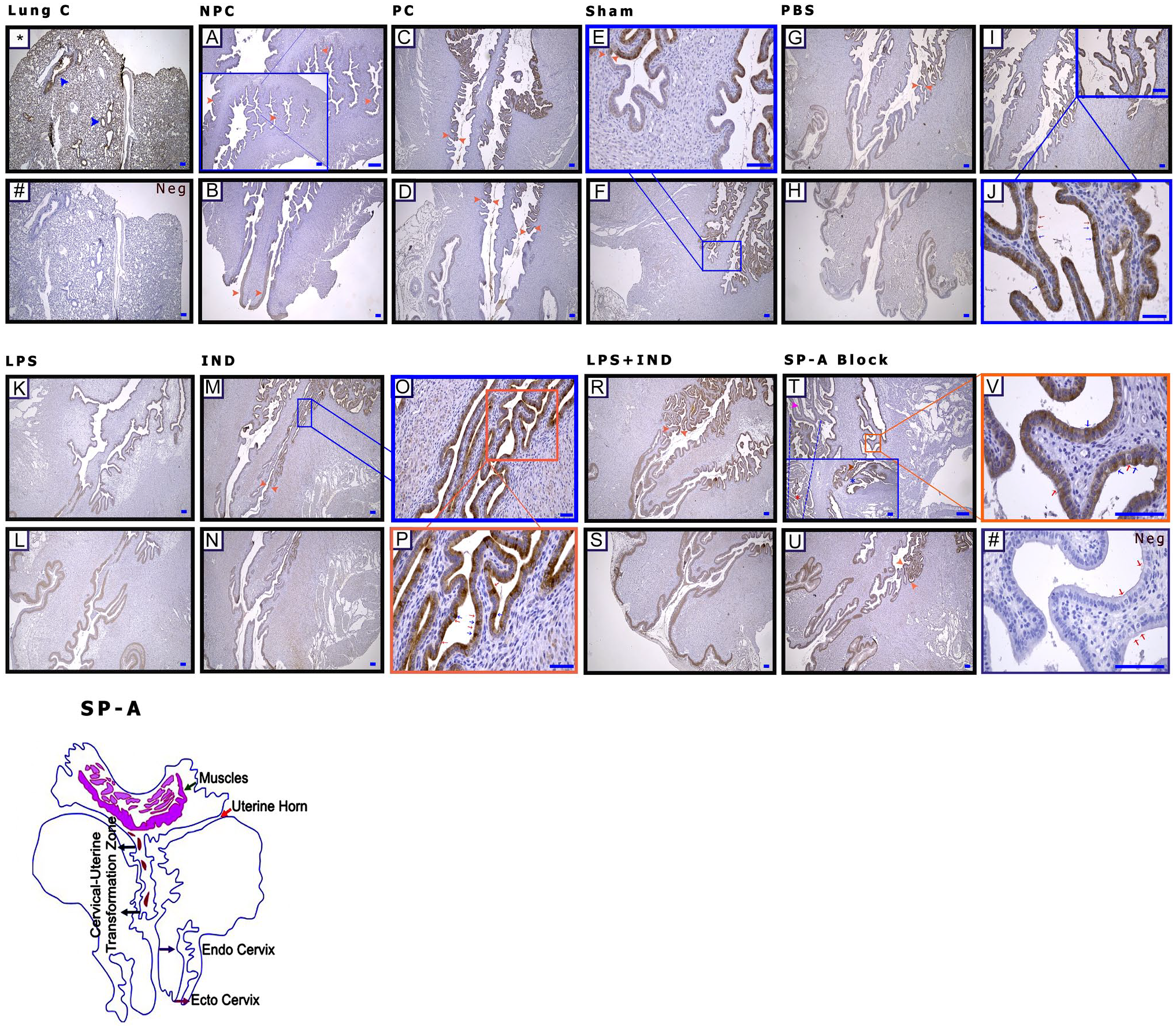
Surfactant Protein A (SP-A) is expressed by the cervix epithelium starting from a certain point during the pregnancy period, and lipopolysaccharide (LPS) reduces SP-A expression. (A and B) Non-pregnant control (NPC), (C and D) pregnant control (PC), (E and F) Sham, (G–J) phosphate-buffered saline (PBS; Ca++-free), (K and L) LPS, (M–P) indomethacin (IND), (R and S) LPS + IND, (T and V) SP-A Block, 4X. *Lung (positive) control; blue arrowheads: bronchioles; Neg (#): negative control; orange arrowheads (C–U): transition zone; (J, P, V) expression starting point; blue arrows: expressing positive cells; (T) red arrows: expressing negative cells, red star: uterine horn blocked with anti-SP-A antibody, blue star: unblocked uterine horn, pink arrowhead: loss of expression in the epithelium, and brown arrowhead: cells with positive expression in the epithelium. Scale bar: 50 µm.
When WB findings were examined, SP-A protein was almost absent in the non-pregnant group. The level of SP-A protein was the lowest in the LPS and LPS + IND groups (p<0.05) (Fig. 9B).
LPS Administration Causes Excessive N1 Expression in the Cervix
Notch 1 expression was increased, especially in the cervix uteri in pregnant groups compared with the non-pregnant group (Figs. 6B and 9C). The immunohistochemical staining intensity of Notch 1 was strong on the epithelial surface and in stroma in the groups that were LPS-applied (Figs. 2I–L, and 6B). It has also been observed that the staining intensity of some cells in the epithelial tissue lining differs compared with other cells in the same line (Fig. 2K, pink and blue arrowheads). In the uterus horn, which was blocked by anti-SP-A, the expression was decreased (Figs. 2R, red star, and 6B).
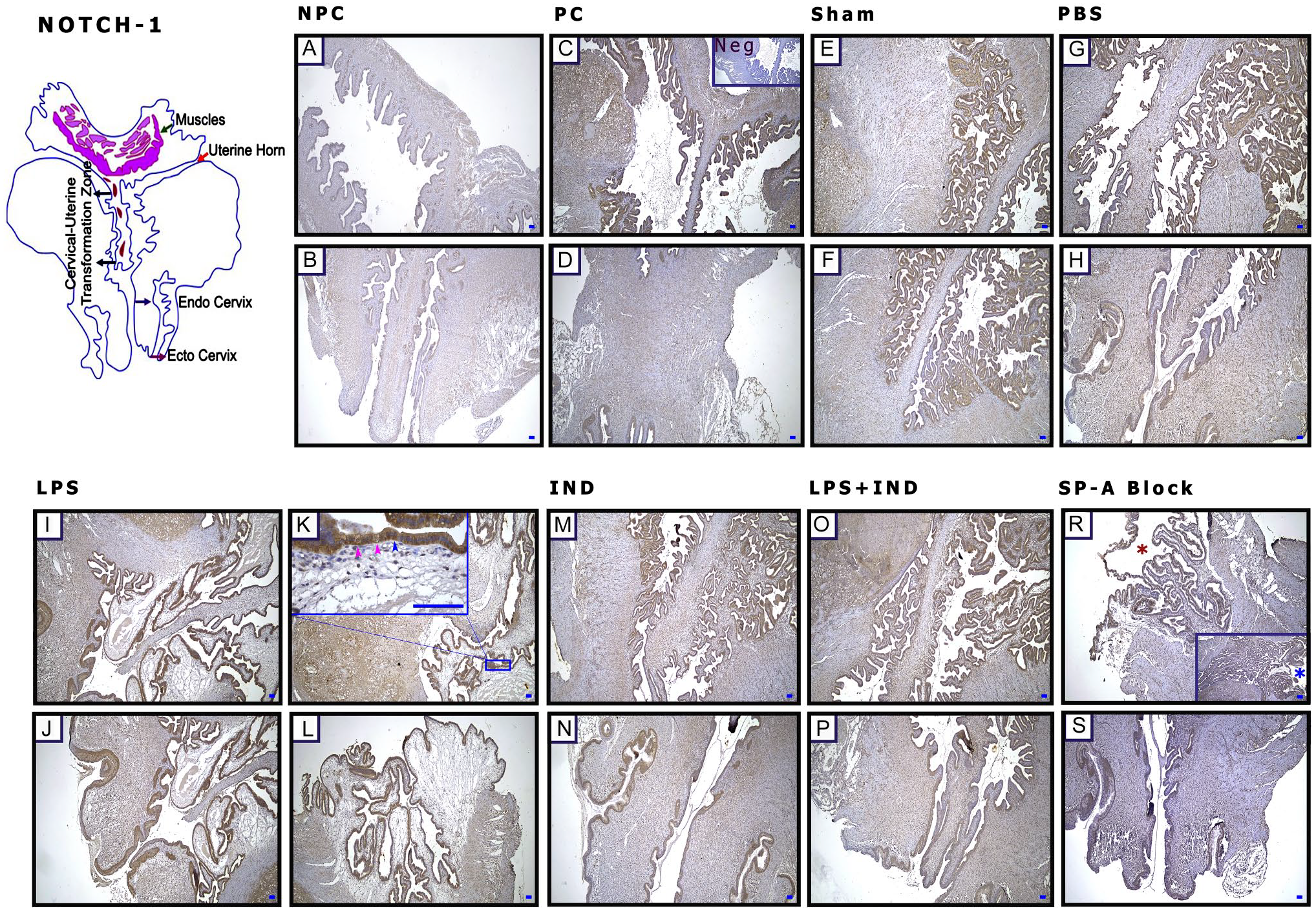
Lipopolysaccharide (LPS) administration causes excessive N1 expression in the cervix. (A and B) Non-pregnant control (NPC), (C and D) pregnant control (PC), (E and F) Sham, (G and H) phosphate-buffered saline (PBS; Ca++-free), (I–L) LPS, (M and N) indomethacin (IND), (O and P) LPS + IND, (R and S) Surfactant Protein-A Block (SP-A Block), and (C) negative control (Neg). (K) Pink arrowheads: negative expression cells in the epithelium; (R) blue arrowhead: expression of positive cells, blue star: unblocked uterine horn, and red star: uterine horn blocked with anti-SP-A antibody. Scale bar: 50 µm.
However, in WB, although N1 increased significantly in the LPS-applied groups, in the SP-A B group, the amount of N1 protein decreased significantly compared with other pregnant groups (p<0.05) (Fig. 9C).
Dll-1 Is Expressed Starting From a Specific Point in the Cervix Epithelium. LPS Increases Dll-1 Expression, Whereas SP-A Blocking Decreases Its Expression
In immunohistochemical staining, it was observed that Dll-1 was expressed by epithelial cells after a certain point in pregnant groups (Fig. 3C–P, red arrowheads). The most intense staining was observed in the stroma and epithelium in the cervix uteri of the LPS-applied group (Figs. 3I and J, and 6C). The LPS + IND group had positive [Fig. 3M (small image), blue arrowheads] and negative staining cells [Fig. 3M (small image), red arrowheads] in the same epithelium line. Similarly, in the epithelium, some cells in the SP-A B group exhibited higher nuclear staining intensity (Fig. 3S, orange arrowheads), whereas other cells did not show the same response (Fig. 3S, blue arrowheads). In this group, there were also positive staining cells in the stroma (Fig. 3S, pink arrows).
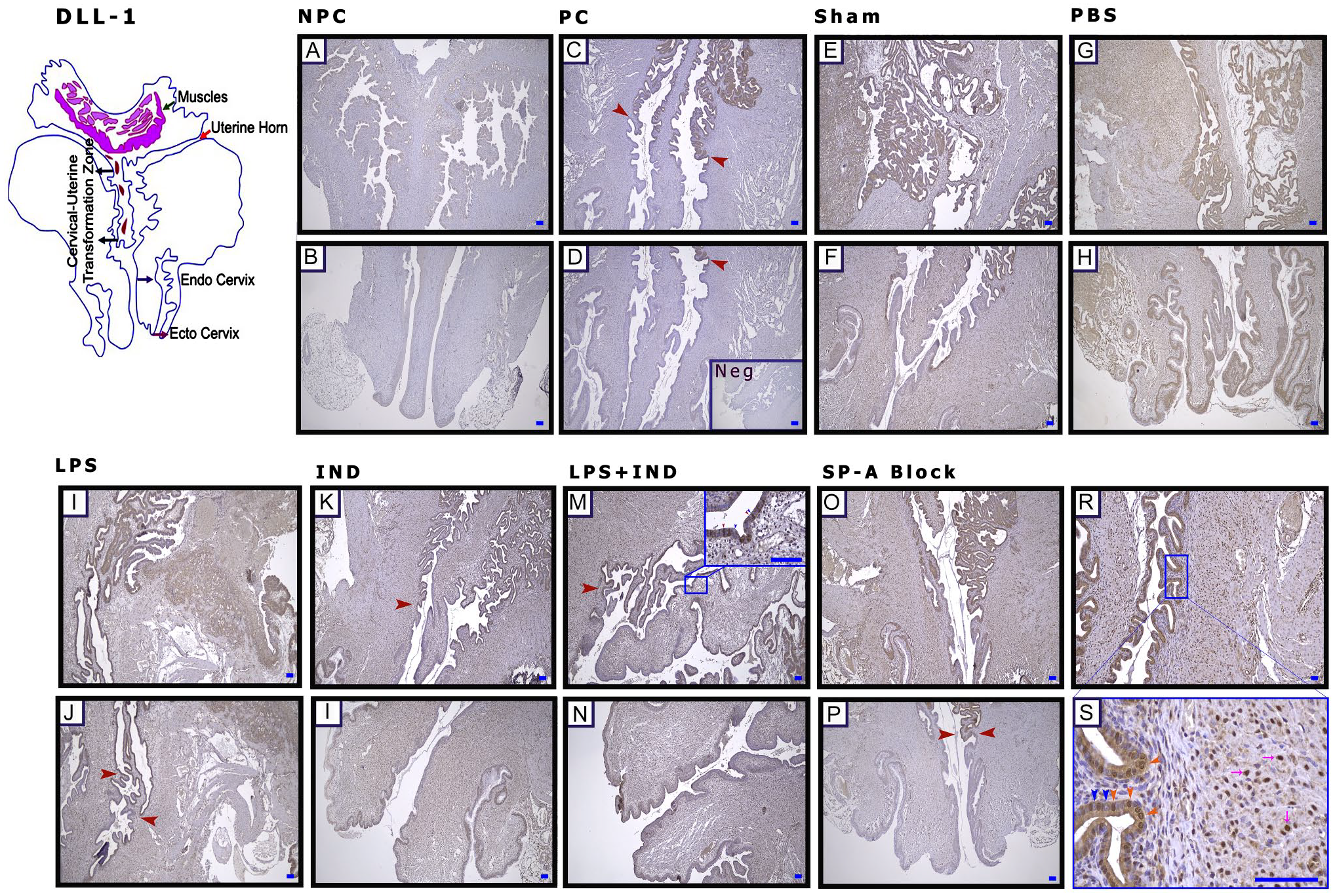
Delta-like ligand 1 (Dll-1) is expressed starting from a specific point in the cervix epithelium. Lipopolysaccharide (LPS) increases Dll-1 expression, whereas Surfactant Protein-A (SP-A) blocking decreases its expression. (A and B) Non-pregnant control (NPC), (C and D) pregnant control (PC), (E and F) Sham, (G and H) phosphate-buffered saline (PBS; Ca++-free), (I and J) LPS, (K and L) indomethacin (IND), (M and N) LPS + IND, (O–S) Surfactant Protein-A Block (SP-A Block), and (D) negative control (Neg). (C and P) Red arrowheads: transition zone, the starting point of expression in the epithelium; (S) orange arrowheads: nuclear expression of positive cells in the epithelium, blue arrowheads: nuclear expression of negative cells in the epithelium, and pink arrows: positive expression of cells in the stroma. Scale bar: 50 µm.
In WB, although there was a significant increase in the LPS group, the decrease in the level of Dll-1 protein was significant in the SP-A B group (p<0.05) (Fig. 9D).
In the Cervix, Jag-2 Is Expressed Specifically by Mast Cells
Jag-2 was expressed by specific cells (Fig. 4C–Y, orange arrowheads) in pregnant groups throughout the entire stroma, especially starting from the cervix. Cells were also seen in connective tissue areas in the stroma (Figs. 4F, orange arrowheads, and 6D). The same cells also were positively stained when stained with MC-specific tryptase (Fig. 4Y, *magnified area; MCT, orange arrow). In immunofluorescence staining, Jag-2 expression was observed, especially in areas where the MC marker was positive (Supplemental Fig. 2; not all images were included because groups were similar). The Jag-2 staining intensity obtained at the tissue level was not as pronounced (Fig. 6D) as the protein increase seen in WB in the LPS group (p<0.05) (Fig. 9E).
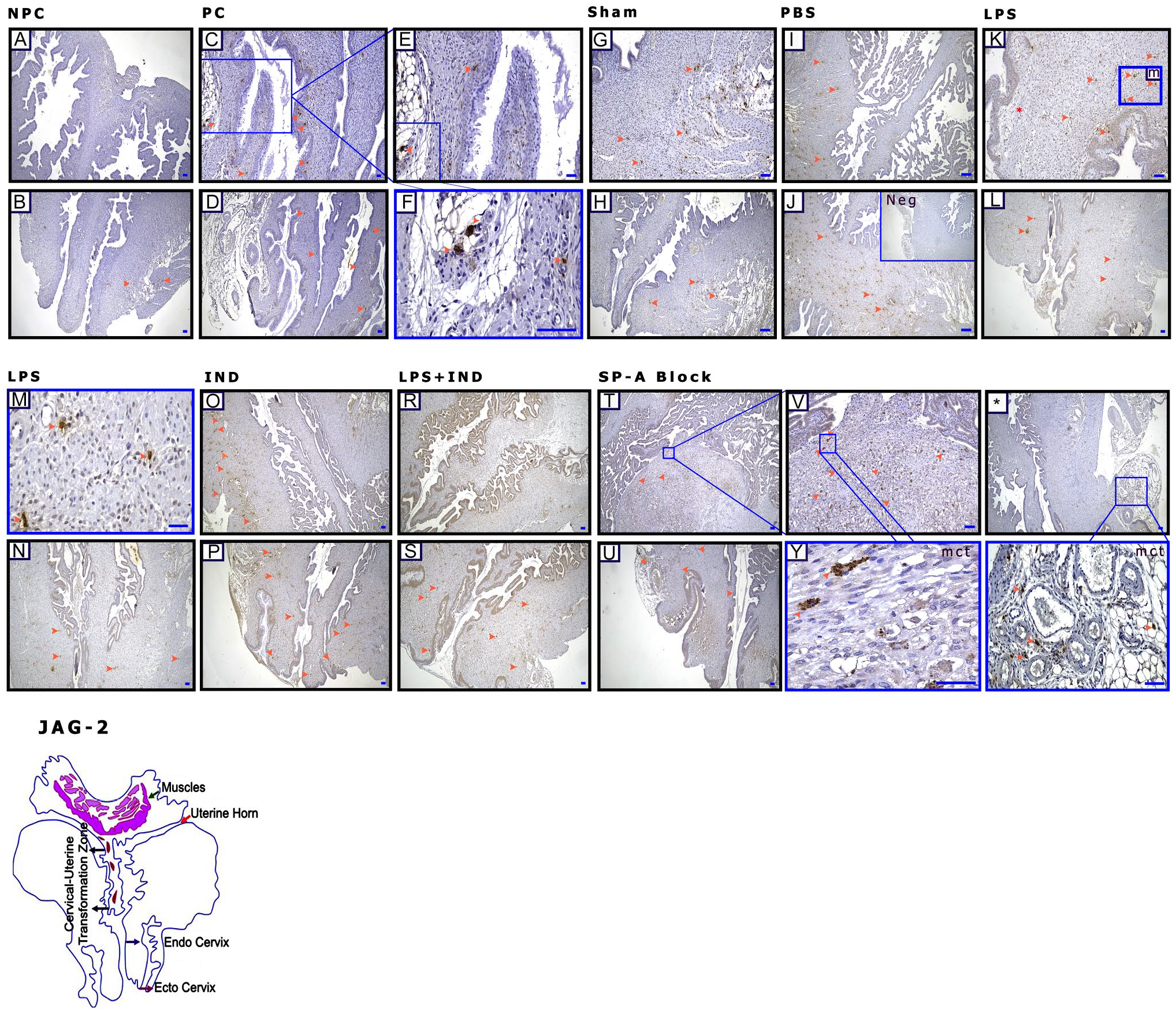
In the cervix, Jag-2 is expressed specifically by mast cells. (A and B) non-pregnant control (NPC), (C–F) pregnant control (PC), (G and H) Sham, (I and J) phosphate-buffered saline (PBS; Ca++-free), (K–N) lipopolysaccharide (LPS), (O and P) indomethacin (IND), (R and S) LPS + IND, (T to *) Surfactant Protein-A Block (SP-A Block), (J) negative control (Neg), and (B to *) orange arrowheads: mast cells; mct: mast cell tryptase–positive cells in the stroma. Scale bar: 50 µm. Abbreviation: JAG-2, Jagged 2.
LPS Administration and Blocking of the SP-A Increase Tlr-2 Expression in the Cervix
The Tlr-2 receptor staining intensity was increased, especially in LPS-treated and SP-A-blocked groups (Fig. 5I, J, and O–S). In these groups, it was also observed that the Tlr-2 staining intensity increased in the uterine stroma (Fig. 5I and R, blue arrows) and surface epithelium (Fig. 5I and O, red arrows). Although no staining was observed in the longitudinal muscles of the uterus (Fig. 5S, pink stars) in the SP-A-blocked group, the uterine apical circular muscles (Fig. 5S, pink arrowheads) stained weakly (Fig. 6E).
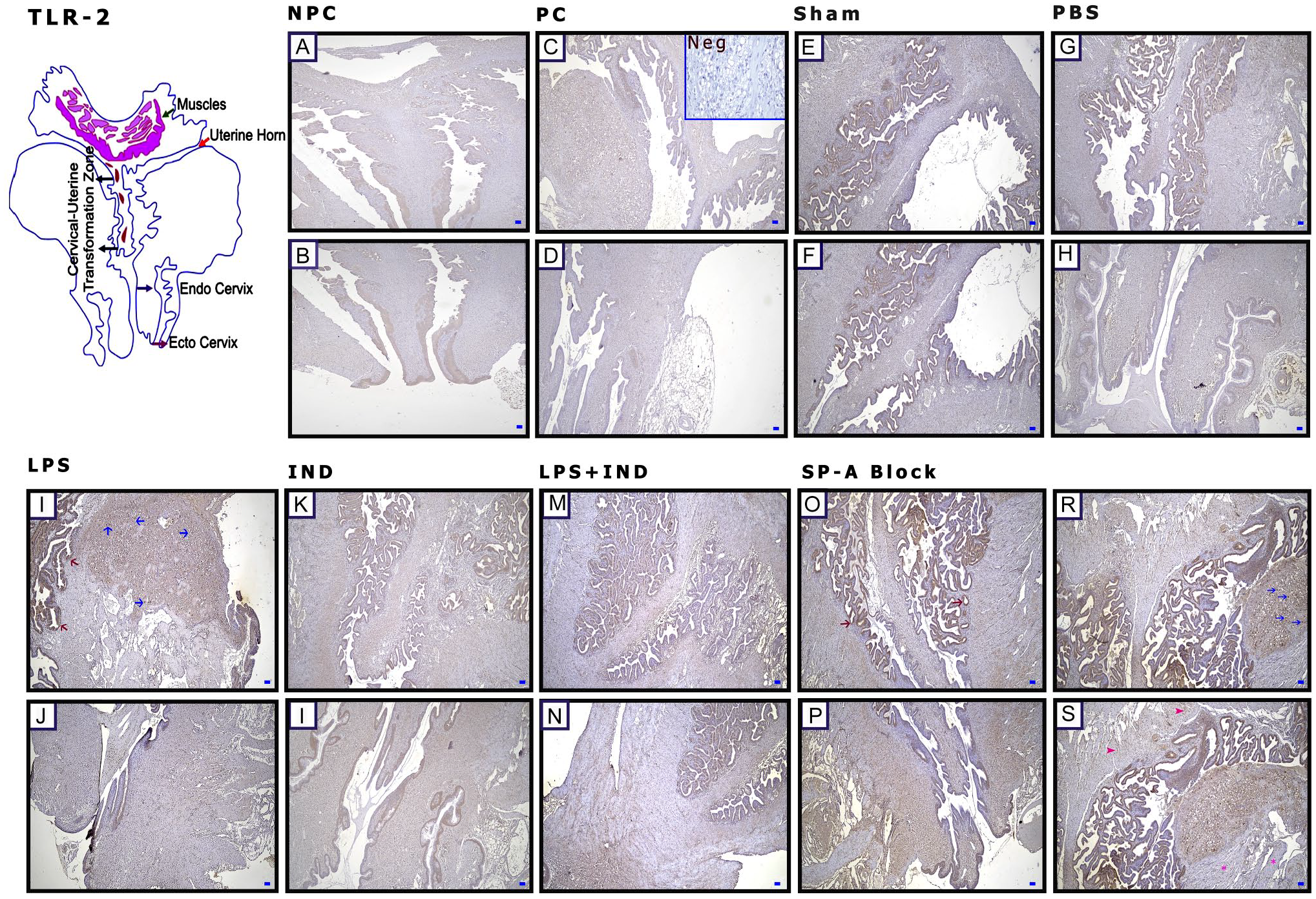
Lipopolysaccharide (LPS) administration and blocking of the SP-A increase Toll-Like Receptor (Tlr)-2 expression in the cervix. (A and B) Non-pregnant control (NPC), (C and D) pregnant control (PC), (E and F) Sham, (G and H) phosphate-buffered saline (PBS; Ca++-free), (I and J) LPS, (K and L) indomethacin (IND), (M and N) LPS + IND, (O–S) Surfactant Protein-A Block (SP-A Block), and (C) negative control (Neg). (I and O) Red arrows: positive expression of cells in the epithelium, blue arrows: positive expression of cells in the stroma; (S) pink arrowheads: poorly stained muscle cells with antibody, pink stars: negative muscle cells. Scale bar: 50 µm.
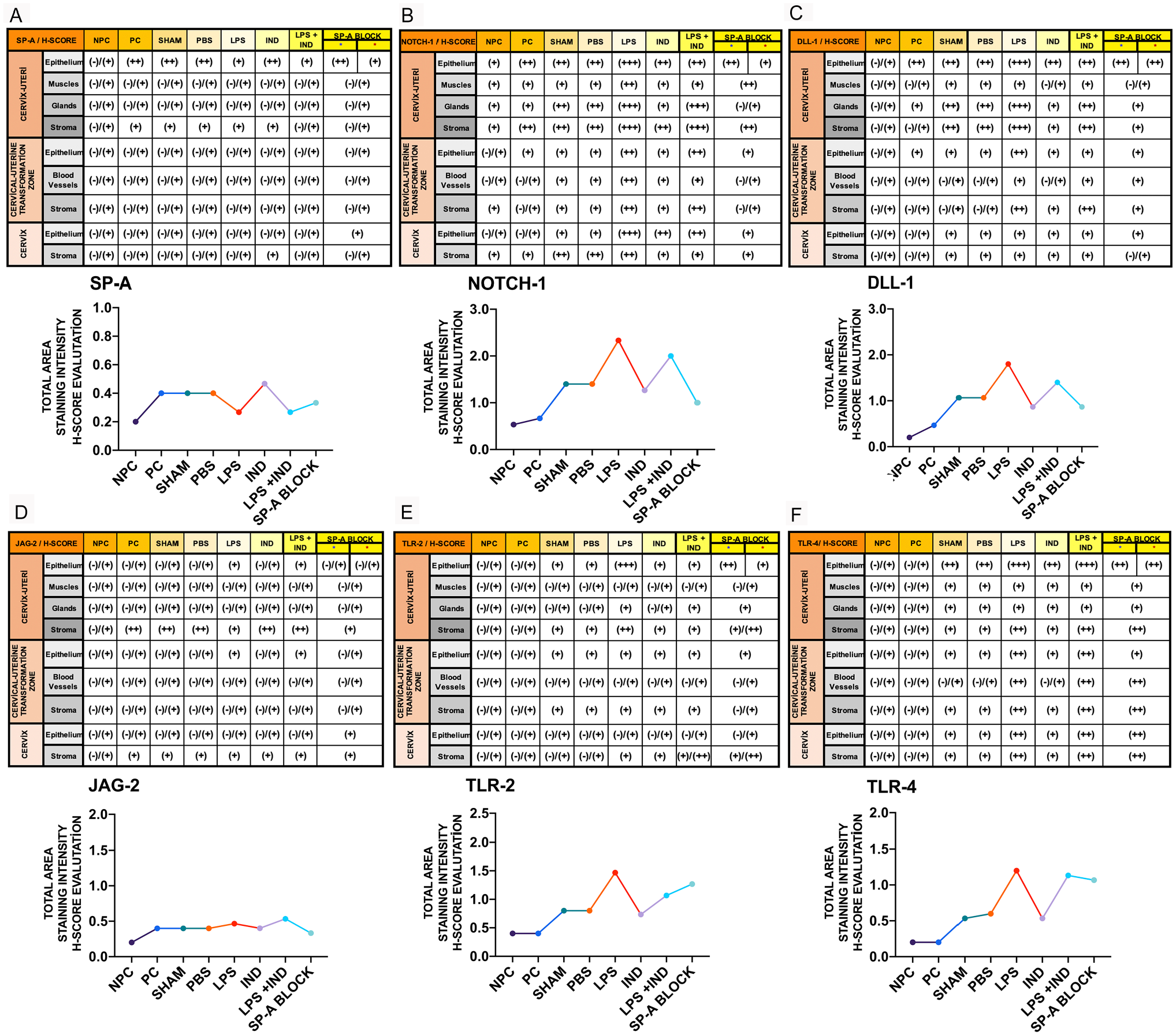
(A and F) H-score analysis of immunohistochemical staining intensity in different cervix regions of the groups. Non-pregnant control (NPC), pregnant control (PC), Sham, phosphate-buffered saline (PBS; Ca++-free), lipopolysaccharide (LPS), indomethacin (IND), lipopolysaccharide+indomethacin (LPS + IND), Surfactant Protein-A Block (SP-A Block).
WB findings and IHC findings were similar for Tlr-2 and were significantly increased in SP-A B and LPS groups (p<0.05). Also, the difference between LPS and LPS + IND groups was significant (Fig. 9F).
LPS Administration Increases Tlr-4 Expression on the Cervix Epithelial Surface
When immunohistochemical findings and WB were evaluated together, the Tlr-4 expression increased in LPS-applied groups (Figs. 6F and 7I–L). Furthermore, when the staining pattern of stroma (Fig. 7E, G, and L, red arrowheads) and muscle (Fig. 7K–S, pink arrowheads) was examined, staining was particularly intense on the apical surface of the epithelium (Figs. 6F and 7I and O, blue arrowheads).
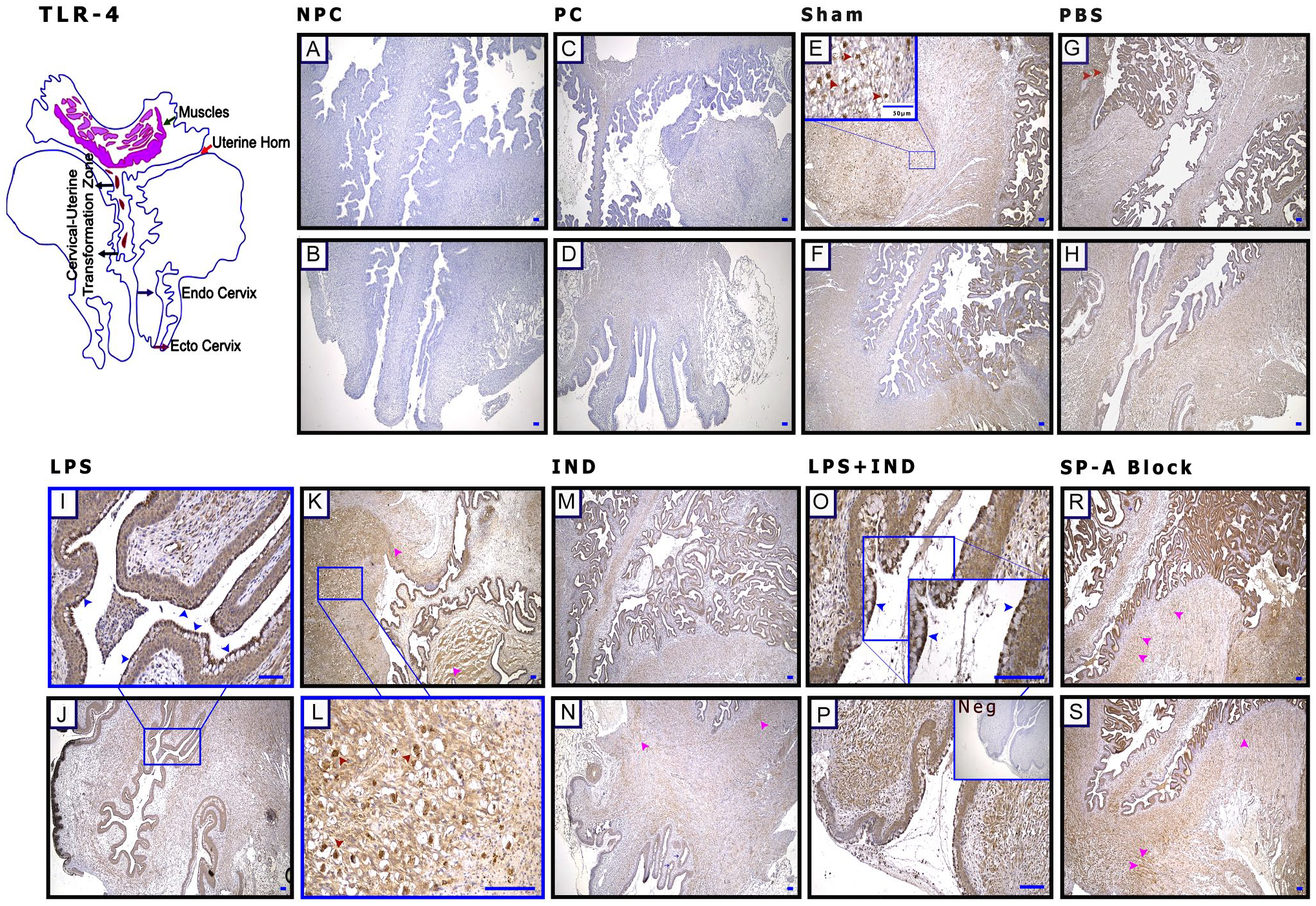
Lipopolysaccharide (LPS) administration increases Toll-Like Receptor (Tlr)-4 expression on the cervix epithelial surface. (A and B) Non-pregnant control (NPC), (C and D) pregnant control (PC), (E and F) Sham, (G and H) phosphate-buffered saline (PBS; Ca++-free), (I–L) lipopolysaccharide (LPS), (M and N) indomethacin (IND), (O and P) LPS + IND, (R and S) Surfactant Protein-A Block (SP-A Block), and (P) negative control (Neg). (E, G, L) Red arrowheads: positive cells in the stroma; (I and O) blue arrowheads: TLR-4-positive cells on the cervix epithelial surface; and (K–S) pink arrowheads: positively stained muscle cells. Scale bar: 50 µm.
In WB, the protein increases were significant when LPS-treated groups were compared with other pregnant groups (p<0.05), but no difference was found between LPS and LPS + IND groups (Fig. 9G).
LPS Implementation and Blocking of SP-A Increase CD11-b Expression in the Cervix
When neutrophil/monocyte/macrophage were evaluated in terms of pregnancy, it was seen that they were located in the external os and stroma before pregnancy (Fig. 8B and C, red arrowheads), whereas neutrophil/monocyte/macrophage were positively stained, especially in the stroma during pregnancy (Fig. 8D, red arrowheads). In addition to stronger staining of all these cells in SP-A B and LPS groups (Fig. 8A, H, and K, red arrowheads), muscle structures were also stained (Fig. 8H and K, blue arrowheads). In comparison, the leukocytes were slightly stained in pregnant groups that underwent any procedure (Fig. 8F, green arrowheads); the intensity of staining increased in LPS-applied and SP-A-blocked groups (p<0.05) (Fig. 8A and 8H–K, green arrowheads).
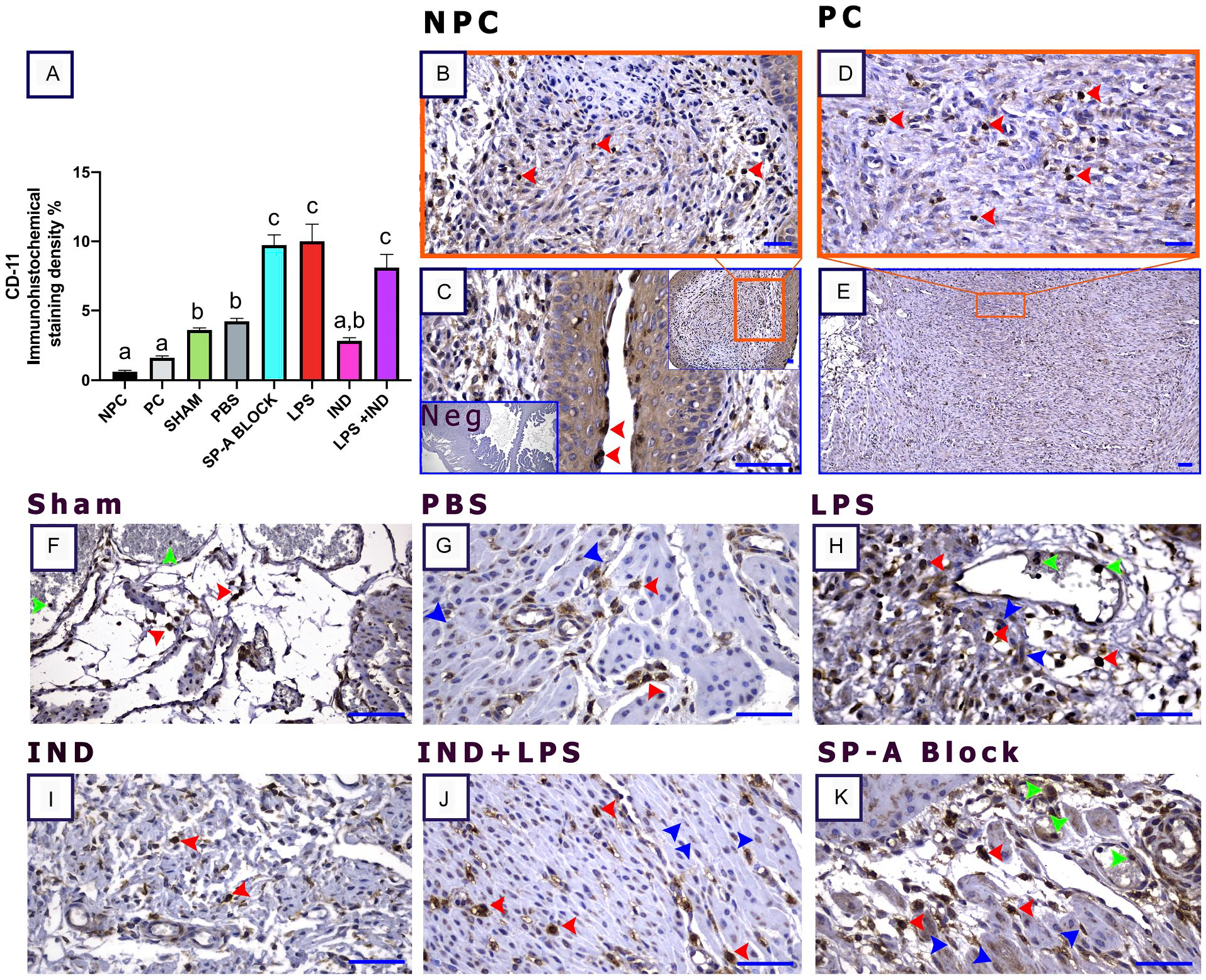
Lipopolysaccharide (LPS) implementation and blocking of Surfactant Protein-A (SP-A) increase CD11-b expression in the cervix. (A) CD-11 immunostaining density graphic, (B and C) non-pregnant control (NPC), (D and E) pregnant control (PC), (F) Sham, (G) phosphate-buffered saline (PBS; Ca++-free), (H) LPS, (I) indomethacin (IND), (J) LPS + IND, (K) SP-A, and (C) negative control (Neg). (B–K) Red arrowheads: positively stained cells in the stroma and epithelial surface; (F–K) green arrowheads: blood vessels and leukocytes, blue arrowheads: muscle cells, Scale bar: 50 µm.
Macroscopic Observation
Pregnant groups had an average of five or six pups in each horn (Fig. 9A and W, orange arrowheads). It was observed that the uterine horns were completely or at least 80% emptied in LPS groups (Fig. 9A; X: blue arrowheads). The offsprings were a fresh appearance at birth in the LPS + IND group (Fig. 9A; Y: pink arrowheads). In the group where SP-A was blocked, the horn structure was disrupted, and pups have died in the uterus or some stuck in the birth canal (Fig. 9A; Z: red arrowheads).
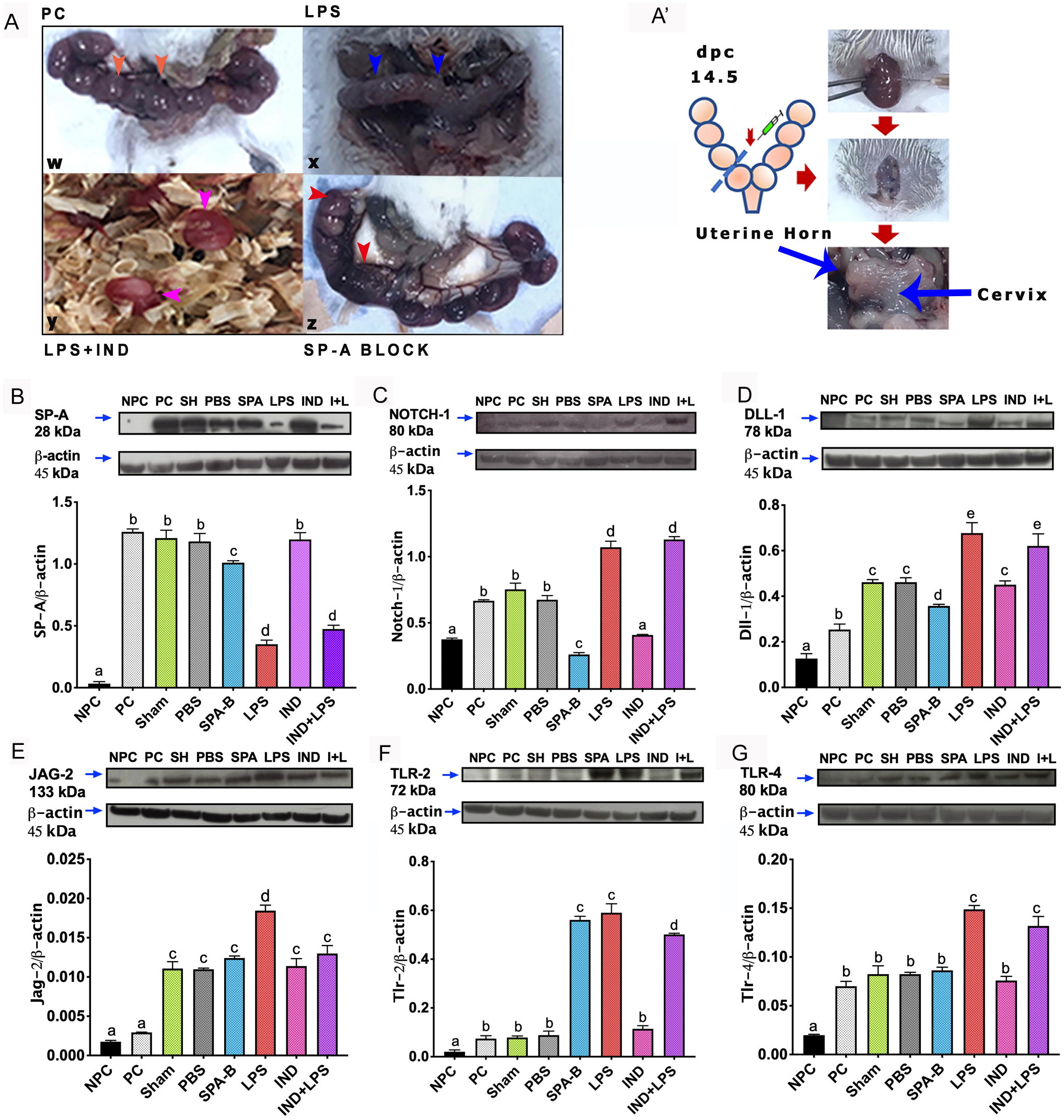
(B and G): Western blot analysis results of the experimental groups. (A and A′) The appearance of gestational sacs in pregnant control (PC), lipopolysaccharide (LPS), Surfactant Protein-A Block (SP-A Block), and LPS + IND groups after applications; orange arrowheads and blue arrowheads: uterine sacs, red arrowheads: morphological deterioration due to SP-A Block, pink arrowheads: fresh-looking fetus after preterm birth in the LPS + IND group. (W) (PC) group, (X) LPS group, (Y) LPS + IND group, and (Z) SP-A Block group.
Discussion
From a broad perspective, birth is an intense inflammatory process characterized by the influx of immune cells into gestational tissues, uterine contraction, cervix maturation, and the production of proinflammatory mediators that stimulate fetal membrane rupture.56,57 The etiology of preterm labor is still unknown, but it is thought to be associated with infection. 58 IU infusion of LPS can mimic the effects of a bacterial infection scenario responsible for 25–40% of all preterm births. 59 Besides the studies investigating the relationship between infection-mediated preterm birth and Notch signaling in the literature, the underlying mechanism has not been fully elucidated yet. Therefore, understanding how infection-mediated inflammation affects Notch signaling in the cervix environment will be interesting to investigate how it causes early tissue changes because molecular- and tissue-level studies focusing on preterm birth in terms of the cervix are scarce in the literature.
When 5 mg/ml of LPS is injected at E15.5, genes and proteases involved in the regulation of inflammatory activation are enriched in cervical tissues approximately 7–9 hr after administration. 58 Another study reports that preterm labor occurs within 48 hr after LPS administered between 14.5 and 19 gestational days. 60 In our study, preterm labor was observed within 12 and 24 hr after LPS injection at E14.5, and all fetuses were dead in the LPS group. These results are consistent with previous studies.
PGs are important mediators in LPS-mediated preterm birth, stimulating uterine contractions and enhancing cervical ripening. 61 IND (a PGE2 inhibitor) 62 inhibits both Cox-1 activities with higher selectivity and Cox-2. 63 When 14 µg/ml of IND was added to the drinking water after 10 µg of LPS injection in mice, fetal death was determined as 24%. When 35 µg/ml of IND was given subcutaneously, fetal death was 1%. 64 Among the systemic and functional damage caused by long-term use of IND, renal failure has been reported in rats using 4 mg/kg (IP) of IND for more than 8 days. 65 Although the use of maternal 4 mg/kg IND in the last 3 days of pregnancy caused neuronal necrosis in mice and rats, this finding was not found for 2 mg/kg. 50 When LPS + IND was administered together, 33% of this group gave birth in a similar time as the LPS group. As bleeding was observed in pregnant mice in this group within 5–7 hr (66% in total) when the observation was continued, the mean delay in preterm birth was accepted as 5 hr in the LPS + IND group compared with the LPS group. The observation time was limited as the mice were sacrificed at a similar time interval. The literature states that the response to IND can vary significantly in rats depending on the plasma P4 level. 66 Previous studies have shown that it completely inhibits LPS-induced endotoxemia-hyperthermia response when IND (2.5 mg/kg/OS) is given 30 min before LPS (1 mg E. coli LPS/kg/IP) administration in rats. It has also been stated that it can reduce gastrointestinal problems due to LPS. 67 Administration of 2 mg/kg of IND may have elicited these results in mice in the LPS + IND group in relation to hormonal changes caused by the presence of different numbers of offspring. However, delayed birth and the fresher appearance of the offspring suggest that IND may positively affect the placenta or circulation. The significant difference in Jag-2 and Tlr-2 levels, especially in LPS + IND groups, may be associated with these results.
Some studies, also including findings at the mRNA and protein level, address the relationship between N1 receptor and Dll-1, Hes-1 ligands of the Notch signaling pathway, and the onset of preterm labor due to inflammation.7,68 However, almost all of these studies were conducted on the uterus and placenta. Our previous study showed that in the human preterm delivery model induced by misoprostol, the receptor and ligand changes of the Notch signaling pathway in the cervix can affect the mechanism of preterm labor mediated by N1 DLL-1 and JAG-2. 27 In general, Notch signaling has been thought to support the inflammatory microenvironment, and macrophages drive proinflammatory responses in different inflammatory environments. 69 Also, MCs that produce a variety of inflammatory agents and have a functional role in cervical maturation are important. There are studies that associate cervical ripening and pregnancy loss with an increase in the activity or number of MCs.70,71 In our current study, the N1 receptor and Dll-1 ligand were significantly increased in LPS administration groups in the cervix. The findings are consistent with infection-mediated preterm labor findings reported in the literature. N1 was significantly decreased in the SP-A B group. Our results show that N1 and Dll-1 were expressed predominantly in the epithelium. These reasons suggested that the deteriorated epithelial structure in the SP-A B group decreased the basal expression level of N1 and Dll-1. As Jag-2 is especially expressed in MCs, the SP-A B group had a similar expression pattern with other intervention groups except the LPS-treated group. In addition, SP-A may have bound to receptors other than N1 in different situations to produce different responses. The basis of this view was discussed in the section that evaluates the results of SP-A blocking.
Although we could not find a study on the state of Jag-2 expression in the cervix during preterm birth, some studies say that mRNA levels of Jag 1 and Jag-2 were reduced significantly in the uterus and placenta during inflammation-induced preterm labor. 68 However, it has been shown in different studies that some pathogen-derived products upregulate Jag-2 mRNA expression. 72 For example, aqueous birch pollen extracts do not only mediate inhibition of LPS-induced Dll-1 and Dll-4 but also induce the Th2-instructing ligands Jag-2 and, to a lesser degree, Jag-1. 73 In these studies, Jag-2 has been associated with the induction of the Th2 response.72,73 MCs can release many important mediators that can cause inflammation. Th2 cytokines may be a signal to MC activation and proliferation. Loss of these signals can contribute to many diseases. 74 When we evaluated the expression of the Jag-2 ligand, it was observed that Jag-2 was specifically expressed by MCs. The increase in Jag-2 protein was significant in the LPS group. This increase was thought to be related to the modulation of the inflammatory process. However, this increase was not evident at the tissue level in the LPS group. It was thought that postsynthesis protein modifications might be related to this process. Moreover, it should be noted that because Jag-2 is intensely expressed by MCs, these cells can be found in different shapes and amounts in tissue sections, and this may affect the staining intensity. The association between MC and the expression of Jag-2 and the cervix may represent a crucial stage at the beginning of early labor. These findings are new to the literature.
Notch signaling pathways can activate in response to TLR ligands. 68 In the MC surface, Tlr-2 and Tlr-4 expression can be regulated by bacterial cell wall components and proinflammatory cytokines. 75 LPS associated with bacteria can upregulate the Tlr-4 ligand and increase its proliferation in epithelial cells. In addition, the severity of the inflammatory response can vary depending on different types of LPS and the TLR receptors they use. 76 It has been studied at the mRNA level that the anti-inflammatory effect of SP-A may require Tlr-2. 60 In our study, LPS administration increased Tlr-2 and Tlr-4 expression. However, Tlr-2 was especially high in the SP-A B and LPS-applied groups. Blocking SP-A increased the Tlr-2 response, similar to LPS. This finding was thought to be related to the effort of SP-A to increase epithelial cell proliferation and repair the impaired epithelial structure through the TLRs. The hypothetical thinking about SP-A is that SP-A changes are important in the pathogenesis of the lower genital tract. 77 SP-A in different tissues such as vaginal mucosa, 77 myometrium and uterus luminal epithelium, 78 gastrointestinal tract, 79 and central nervous system 80 has been demonstrated. Although its expression was shown in different regions, a detailed expression map was not found in the cervix in a model similar to our study.
Information on the effectiveness mechanism of SP-A is complex. Based on its binding to inhibitory regulatory protein α or CD91/calreticulin receptors via the calcium-dependent carbohydrate recognition domain or collagen domain, it is thought to activate both anti- and proinflammatory signaling pathways. 41 In addition, SP-A can play a role in regulating inflammation, contributing to the innate defense. 81 Our findings showed that the expression of SP-A in cervico-uterine tissues starts from a certain point, and its expression increases with pregnancy. The SP-A protein level was significantly reduced in the LPS-applied groups. These mice often had an unsuccessful birth profile and uterine and placental structures in the obstructed and disrupted horn. The monocyte/macrophage staining pattern of LPS and SP-A B groups was similar. SP-A can bind to various bacterial and viral pathogens, and LPS. 82 Therefore, our interpretation is that the strong decrease in SP-A observed in the protein level in groups given LPS may be related to the binding of LPS and SP-A. Also, this situation may be an alternative response to the expression reductions of N1 and Dll-1 in the SP-A B group. Regarding the possible anti-inflammatory effect of SP-A, its depletion in the inflammatory environment may have been accelerated. These findings suggest that SP-A may be necessary for the preservation of epithelial integrity and for pregnancy. Findings regarding the possible effectiveness of SP-A and IND on the cervix in preterm birth are new in the literature.
Overall, in this experimental model, the epithelial response was seen to increase from a certain point of the cervix to the uterus. Taking into account the relationship between SP-A and Tlr-2 and the rapid depletion of SP-A against LPS, it suggests that the effectiveness of SP-A supplements in bacterial infections and its role in premature birth may be worth investigating. Again, PG inhibitors can also be important in delaying premature birth in relation to their dose-dependent effects. In addition, the role of surfactants in terms of maintaining a pregnancy and preventing preterm labor should be examined in more detail. And finally, the link between MCs and Jag-2 has the potential to be a control key for preterm birth. In light of the findings obtained from the cervix, the functioning of the possible preterm delivery mechanism and the cellular relationship we are trying are revealed in Fig. 10.
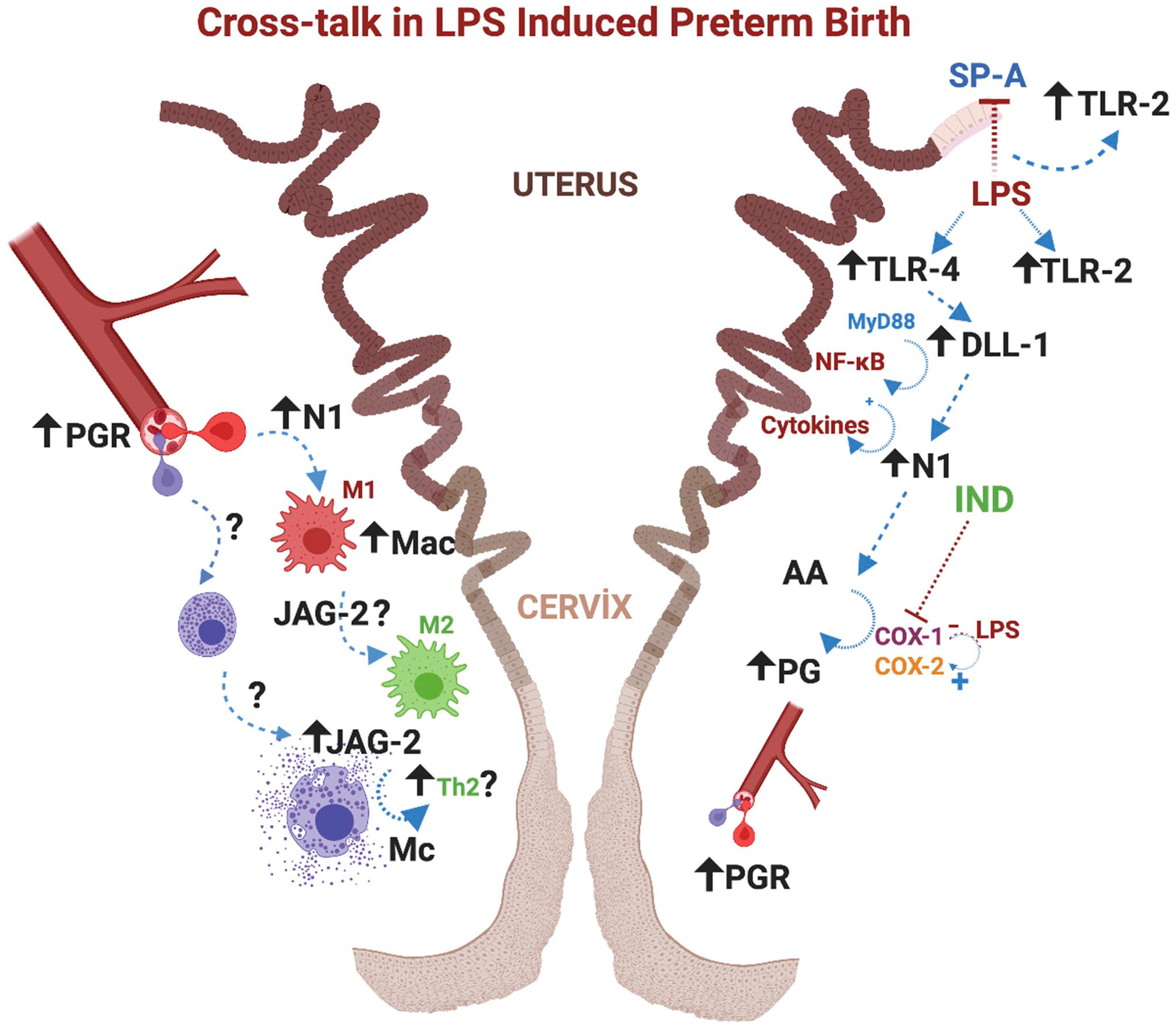
Crosstalk: Possible mechanism and diapedesis mediated by Notch signal in LPS-induced preterm birth; LPS causes Tlr-4 and Tlr-2 activation in the epithelium. With Tlr-4 increasing, epithelial cells function as cells that initiate this postactivation signal to produce the Dll-1 ligand. Stromal cells, which receive the signal, activate N1 receptors. As a result of this cellular interaction (possible MyD88 and NF-κB activation), PG and cytokine synthesis increase, and the inflammatory process is triggered. Interaction between PGR and blood vessels attracts the macrophages and related cells in the stroma to suppress the inflammatory process as a response to increased PG and cytokines. This process is known as diapedesis, and macrophages (possibly N1 and Cox-2 related M1 macrophages) and bone marrow–derived mast cells are diverted from blood to tissue. The cyclooxygenase enzyme (Cox) is responsible for the production of prostaglandins. After inflammatory processes and LPS stimulation, the production of Cox-2 increases instead of the constitutively expressed Cox-1. IND, a non-selective Cox inhibitor, reduces PG production depending on dosage and time. It is thought that Jag-2 activation, which is found to be positive in mast cells, may be related to the immunomodulatory roles of these cells. In addition, it is assumed that there is a relationship between Jag-2, which is known to increase bactericidal-phagocytic activity, and the Th-2 response. 83 Tlr-2 increases due to LPS implementation and block of SP-A. This increase is thought to be connected with the Th-2 response process that occurs to restore altered balance instead of the increased Th-1 response due to SP-A suppression and LPS stimulation. Abbreviations: LPS, lipopolysaccharide; TLR: Toll-Like Receptor; N1, Notch 1; DLL-1, Delta-like ligand 1; NF-κB: nuclear factor kappa B; MyD88, myeloid differentiation factor 88; PG, prostaglandin; PGR, prostaglandin receptor; COX, cyclooxygenase; IND, indomethacin; JAG-2, Jagged 2; Th-1, T-helper 1; Th-2, T-helper 2; SP-A, Surfactant Protein-A; Mac, macrophage; Mc, mast cell.
Supplemental Material
sj-pdf-1-jhc-10.1369_00221554211061615 – Supplemental material for Relationship of Notch Signal, Surfactant Protein A, and Indomethacin in Cervix During Preterm Birth: Mast Cell and Jagged-2 May Be Key in Understanding Infection-mediated Preterm Birth
Supplemental material, sj-pdf-1-jhc-10.1369_00221554211061615 for Relationship of Notch Signal, Surfactant Protein A, and Indomethacin in Cervix During Preterm Birth: Mast Cell and Jagged-2 May Be Key in Understanding Infection-mediated Preterm Birth by Sema Avci, Nilay Kuscu, Leyla Kilinc and Ismail Ustunel in Journal of Histochemistry & Cytochemistry
Footnotes
Competing Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
SA designed the study, performed the experimental and surgical procedures, analyzed the data, and drafted the article. NK and LK assisted SA in experiments, during surgical procedures, and in generating data. IU acted as a consultant during the execution of the thesis project.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was produced from Sema Avcı’s PhD thesis. The thesis was supported by the Scientific and Technological Research Council of Turkey (1059B141700505-2214/A-International Research Fellowship Programme) and the Akdeniz University Scientific Research Projects Coordination Unit (TDK-2018-3256).
Consent to Participate
All authors read and agreed with the content of the manuscript; all authors gave consent to participate in the study.
Consent for Publication
All authors gave consent for the publication of this research.
Ethics Approval
Approval was obtained from the ethics committee of Akdeniz University Animal Experiments Local Ethics Committee/702/2018.01.004. The procedures used in this study adhere to the tenets of the Declaration of Helsinki.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
