Abstract
Introduction
Pigeon breeder’s lung (PBL) is a kind of hypersensitivity pneumonitis (HP) that is induced by repeated inhalation of pigeon feathers, dander and other shed proteins by sensitive individuals. 1 The incidence of allergic alveolitis ranks third to fourth. 2 Exposure of susceptible people to known or unknown antigens can induce cellular and humoral immune interstitial lung diseases and lead to pulmonary inflammation. Due to the frequency, duration and intensity of pathogenic antigen exposure, the severity, clinical manifestations and natural course of HP are complex and diverse rather than a reflection of a single disease. In 2020, the ATS/JRS/ALAT issued diagnostic guidelines for adult HP. 3 Based on whether the patient’s radiology and/or histopathology shows fibrosis, HP is divided into fibrotic HP and nonfibrotic HP.
The pathogenesis of PBL depends on the inhalation of pigeon feathers, dander and other exfoliates. After inhalation, allergens are recognized and phagocytosed by pulmonary macrophages and combine with specific IgG to form immune complexes, which activate macrophages through the complement activation pathway. 4 Macrophages promote the transformation of CD4+ Th0 cells into Th1 cells through antigen presentation, and activated Th1 cells further activate macrophages by secreting interferon gamma (IFN-γ) and tumor necrosis factor (TNF)-α. 5 Macrophages are the key regulators of fibrosis and can secrete many fibrotic soluble mediators, chemokines and matrix metalloproteinases. Under different environmental stimuli, macrophages can be polarized into classically activated (M1) macrophages and alternatively activated (M2) macrophages. 6 The activation of M1 macrophages depends primarily on lipopolysaccharide and the Th1-related cytokines IFN-γ and C-C chemokine; M1 macrophages play a role in phagocytosis, bactericidal activity, antigen presentation and degradation of the extracellular matrix. In contrast, the activation of M2 macrophages depends mainly on glucocorticoid- or Th2-dependent cytokines (interleukin (IL)-4 and IL-13), and activated M2 macrophages can promote extracellular matrix reconstruction, cell proliferation, angiogenesis, and anti-inflammatory processes, as well as mediate the immune escape of pathogens and tumors. 7 In the early stage of pulmonary fibrosis, an increase in the proportion of M1 macrophages is conducive to the elimination of pathogenic microorganisms and promotes the progression of inflammation; however, in the late stage of pulmonary fibrosis, an increase in the number of M2 macrophages can inhibit the inflammatory response and promote fibrotic degradation.
The Notch signaling pathway is a highly conserved pathway that serves as the central hub for controlling genes, proteins and cells during infection. It also plays a role in the process of lung inflammation and in tissue regeneration after infection. 7 Blocking the Notch signaling pathway leads to loss of proinflammatory function. Therefore, Notch signaling directly or indirectly affects the proinflammatory function of macrophages and directly regulates the transcription of certain inflammatory factors. 8 The γ-secretase inhibitor DAPT ({(2S)-N-[(3,5-Difluorophenyl)acetyl]-L-alanyl-2-phenyl]glycine 1,1-dimethylethyl ester}) cleaves the specific sites of Notch signaling proteins in cells, which enables intracellular segments to detach from the inner membrane and enter the cell, thereby exerting downstream effects. Therefore, DAPT is commonly used to block Notch signals in macrophages in vitro. 9 In a previous study conducted by our team, 10 we found increased levels of IL-8 and macrophage inflammatory protein-1α (MIP-1α) in the serum of PBL patients, which suggested macrophage activation. Additionally, abnormal hypomethylation of Notch4 and its ligand delta-like ligand 1 (DLL1) was observed in PBLs, indicating that the Notch signaling pathway may be involved in pathogenesis. In the acute phase of PBL, activated Th1 cells participate in the cellular immune response by secreting IL-2 and IFN-γ. As the disease progresses to the chronic phase, Th2 factors are dominant when pulmonary fibrosis occurs in the lungs. Furthermore, we observed abnormal expression of Notch signaling pathway members at the fibrotic stage and found that the Notch signaling pathway is associated with the polarization of macrophages. Therefore, we speculate that the Notch signaling pathway may be related to the molecular mechanism that regulates the involvement of macrophages in PBL.
This study aimed to investigate the possible effect of the Notch signaling pathway on the Th1/Th2 imbalance in pigeon shedding-induced fibrotic PBL by regulating the polarization of macrophages. This study will provide a scientific basis for the prevention and effective treatment of these diseases.
Materials and methods
Animals
Twenty-seven specific pathogen-free Sprague‒Dawley (SD) rats of either sex (Wt: 180-200g; age: 6–8 w) were purchased from Shanghai Sipur-Bikai Experimental Animal Co., Ltd (license number SCXK (Shanghai) 2018-0006). The feeding environment was as follows: temperature, 21°C–25°C; relative humidity, 50%–70%; air exchange times, 1–2 times per day; air flow speed, 0.1–0.2 cm/s; ambient noise, below 60 dB; and lighting, 15–20 lx. The animals were allowed to move freely and had free access to food and water. The health of the animals was monitored, and the animals were used in the experiments after 1 week of quarantine.
The present study followed international, national, and institutional guidelines for humane animal treatment and complied with relevant legislation and the procedures were approved by the Ethics Committee of the People’s Hospital of Xinjiang Uygur Autonomous Region (approval no., 2019030615).
Methods
Allergen preparation
According to the outcome of our previous study, 11 fresh pigeon exfoliates, such as feathers and dander, were dissolved in phosphate buffer (pH 7.4) at a ratio of 1:20 and then stirred for 24 h to prepare freeze-dried protein powder. 20 milligrams of freeze-dried protein powder was dissolved in 300 μL of normal saline to prepare a suspension, and PBL models were established (this allergen preparation method has been verified to be reliable according to our previous study). 11
Grouping and treatment
The animals were divided into the model (M) group (n = 9), the model plus γ-secretase inhibitor (M+DAPT) group (n = 9), and the control group (n = 9) equally.
In the M group, each animal received tracheal dripping of the freeze-dried powder suspension each week (300 μL each time) for four consecutive weeks. The animals in the M+DAPT group received the same treatment as the M group, as well as DAPT (HY13027; MedChemExpress, New Jersey, US) at 0.05 mg/kg, which was administered intraperitoneally twice a week for four consecutive weeks followed by once a week until the 20th week. The control group received 0.9% normal saline at the same dose as the PBL suspension through the airway once a week for four consecutive weeks.
At the 20th week, the animals were used for subsequent experiments. The treatment methods for the different groups are presented in Figure 1. Time schedules for establishing the three groups (9 rats in each group). Ctrl, control. M, model. M+DAPT, model+DAPT.
The purification and culture of alveolar macrophages
After modeling, a T-shaped incision approximately 2–3 cm long was made on the tracheal node, and a tracheal cannula was inserted. A straight-headed rat gavage needle was inserted into the trachea, and the lower trachea was ligated. Cold normal saline (5 mL) was infused into the right lung with a syringe and then slowly extracted. This procedure was repeated five times. A small amount of bronchoalveolar lavage fluid (BALF) was used to count the total number of cells, and cell survival was assessed using trypan blue staining.
12
According to the experimental requirements, the cell concentration was adjusted to 1×105 cells/mL. After aspiration and mixing, the cells were moved into a 24-well plate covered with round slides, cultured in a 5% CO2 incubator at 37°C for 2 h, and washed with D-hanks liquid (a balanced salt solution without calcium and magnesium ions; Boster Biological Technology Co. Ltd, Pleasanton, CA, US). RPMI 1640 medium (2 mL) was added to the wells of the adherent cells, and half of the medium was replaced every 2 days. After 7 days, adherent macrophages were obtained. The cells were removed from the incubator, the culture medium was removed, and the cells were washed twice with prechilled PBS. The specimens were fixed with 4% paraformaldehyde. An unconjugated anti-CD68 antibody (ab213363; Abcam, Cambridge, UK) was applied (CD68 is a macrophage marker protein) and the samples were incubated at 4°C away from light, followed by incubation with a fluorescence-conjugated secondary antibody. After staining, macrophages were identified under an inverted microscope (Ts2R; Nikon, Japan) equipped with a fluorescence module (Nikon, Japan). Our results showed that the purity of the isolated macrophages was greater than 95% (Figure 2), which satisfied the experimental standard (95%). Immunofluorescence of rat alveolar macrophages for identification (×400). (a): DAPI staining. (b): Merged staining. (c): Macrophages were green and isolated on the basis of CD68 surface staining.
Histological observation
At 20 weeks, the animals were anesthetized with 10 mg/kg pentobarbital sodium and then sacrificed. The ribs were cut off with sterilized surgical scissors to expose the thorax, and the left lung tissue was immediately isolated. The tissue was submerged in 10% formalin solution for 24 h. A 1 mm3 piece of tissue was washed with PBS, fixed with 4% paraformaldehyde, dehydrated in ethanol and embedded in paraffin. Sections of 4 μm were made, and pathological changes were observed using hematoxylin-eosin (HE) (BASO, Zhuhai, China) staining. Masson staining was performed to assess collagen fiber deposition. The sections were imaged (DS-Ri2; Nikon), observed (H550S; Nikon) and then analyzed.
Western blotting analysis
Western blotting analysis was performed to determine the levels of key proteins in the Notch signaling pathway. Approximately 20–50 mg of the left lung tissue stored at −80°C was taken, and PBS buffer was added. The tissue was ground and then pressed through a 200-mesh sieve for homogenization. Enhanced RIPA lysis buffer (100 μL) was added, and cell proteins were extracted. Sodium dodecyl sulfate‒polyacrylamide gel electrophoresis (SDS‒PAGE) and membrane transfer were performed. The membrane was blocked with TBS+Tween (TBST) solution containing 5% skimmed milk (232,100; BD Biosciences, Franklin Lakes, NJ, USA) for 2 h and then washed three times (10 min each). Primary antibodies, including anti-DLL1 (ab10554; Abcam, UK), anti-DLL4 (NB600-892; Novus, San Francisco, CA, USA), anti-Jagged 1 (Jag1) (ab7771; Abcam, UK), anti-Jag2 (sc515725; Santa Cruz, CA, USA), anti-Notch1 (10,062-2-AP; Abcam, UK) and anti-Notch2 (ab118824; Abcam, UK), were applied and samples were incubated at 4°C overnight. The membrane was washed three times at room temperature. Secondary antibodies, including HRP-labeled anti-mouse (A0216; dilution, 1:5000; Beyotime, Shanghai, China), anti-rabbit (A0208; dilution, 1:5000; Beyotime, China) and anti-GAPDH (200,306-7E4; dilution, 1:5000; ZenBio, Chengdu, China) antibodies, were then added and samples were incubated at room temperature for 2 h. After three washes with TBST, the membrane was subjected to a chemiluminescence reaction (180–5001; Tanon, Shanghai, China). Images were obtained with a Tanon 5200 chemiluminescence imager (Tanon, China). ImageJ software was used to scan the grayscale values of the target bands, and the relative level of the target protein was calculated based on the ratio between its gray value and that of the internal reference protein.
Fluorescence quantitative polymerase chain reaction (PCR)
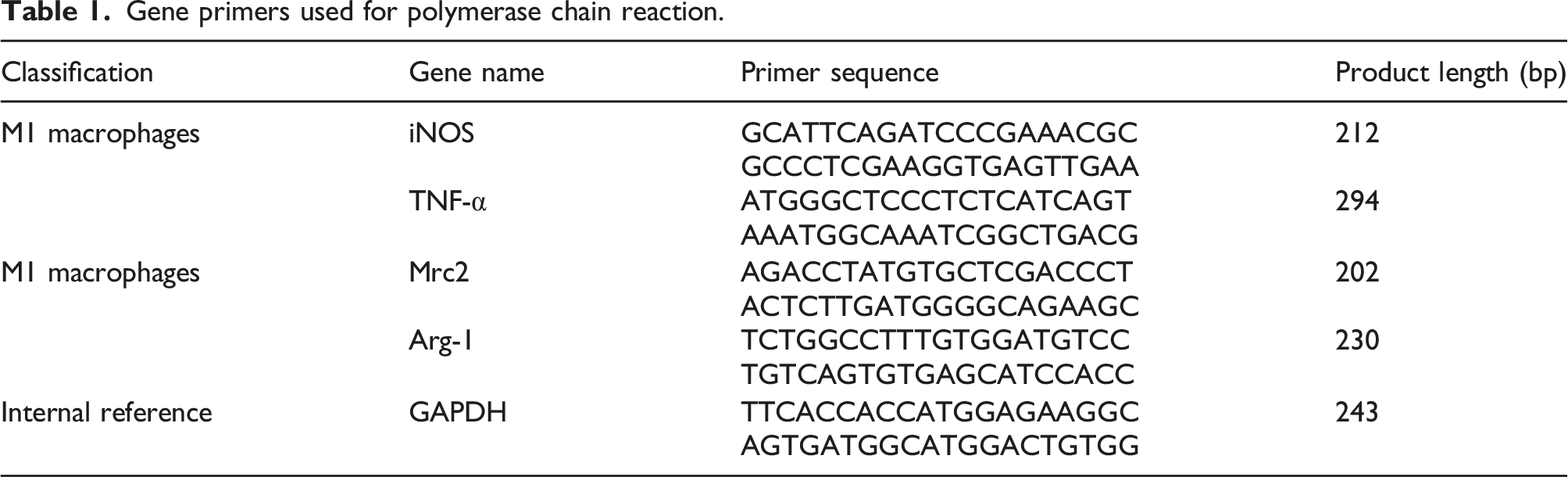
Gene primers used for polymerase chain reaction.
Flow cytometry
Flow cytometry was performed to measure the Th1/Th2 ratio. Briefly, the cells in the BALF were collected, and 200 μL of FBS1640 suspension (2×105 cells) was transferred into a 12×75 mm flow cytometry tube. APC-CD3 antibody (dilution, 1:100; Invitrogen, Carlsbad, CA, USA) and Percp-cy5.5-CD4 antibody (dilution, 1:100; BioLegend, San Diego, CA, USA) were applied for a 20-min incubation at room temperature. The cells were washed with 1 mL of PBS and then centrifuged at 500 g for 5 min. The supernatant was removed. After the cell membranes were broken with a Transcription Factor Fixation/Permeabilization Kit (eBioscience, San Diego, CA, USA) according to the manufacturer’s instructions, a PE-IL-4 antibody (1:100 dilution; BioLegend, USA) and a FITC-IFN-γ antibody (1:100 dilution; BioLegend, USA) were applied; then, samples were subjected to a 20-min incubation at 4°C away from light. After washing, the cells were centrifuged at 500 g for 5 min, and the supernatant was removed. After two washes, the cells were resuspended in 400 μL of PBS. Flow cytometry (BD, FACSCantoII) was used to detect CD3 and CD4 double-positive cells on the FL3 and FL4 channels. The double-positive cells were selected to determine the proportion of IL-4- and IFN-γ-positive cells in the FL1 and FL2 channels, and the proportions and ratios of Th1, T and Th2 cells were calculated.
Statistical analysis
All the data were analyzed with SPSS 26.0 software. After assessing the homogeneity of variance, one-way analysis of variance was used to compare data between groups, and the least significant difference (LSD) method was used to test the significance of group differences. The test level was α = 0.05, and p < 0.05 was considered to indicate a significant difference.
Results
Pathological changes
At 20 weeks, the control group did not show inflammatory or fibrotic changes in the lung tissue. In contrast, the M group exhibited marked infiltration of interstitial inflammatory cells in the lungs and proliferation of fibrous tissue and smooth muscle tissue around the bronchioles and glands. Widened alveolar septa, granulomas and fibrosis in the centers of the granulomas were also observed. The infiltration of inflammatory cells and the severity of fibrosis were markedly reduced in the M+DAPT group compared with the M group, without clear granuloma formation in the M+DAPT group (Figure 3). Comparison of the collagen volume fractions (CVFs) of the three groups showed that the CVF of the M+DAPT group was much lower than that of the M group (Figure 3b). Typical microscopic pathological images of HE and Masson staining in the lung tissues of rats in each group (n = 9). (a) HE staining (×100). In the control group, pulmonary alveoli, bronchioles and blood vessels were normal, and no changes in inflammation or fibrosis were observed. In the M group, the infiltration of interstitial inflammatory cells in the lung centered on the airway was remarkable, with proliferated fibrous connective tissue and noticeable proliferated fibrous tissue and smooth muscle tissue around the bronchioles and glands. The alveolar septum was widened with occasional multinucleated giant cells (indicated by a black arrow), and loose granuloma was formed. The inflammatory cell infiltration and fibrous connective tissue proliferation along the bronchus in the M+DAPT group were less severe than those in the M group. (b) Comparison of the collagen volume fractions (CVFs) was analyzed at a magnification of 40x using ImageJ software. CVF is the percentage of the blue area of collagen occupied in the total tissue area, and a higher CVF value indicates more severe fibrosis. (c) Masson staining (×100). Normal thin layers of connective tissue were observed under the submucosa of the bronchioles in the Ctrl group. In the M group, fibrosis of the bronchus and surrounding interstitial tissue was noticeable, and fibrosis developed around the granuloma in some areas (indicated by a yellow arrow). Fibrosis in the M+DAPT group was less severe than that in the M group. Ctrl, control. M, model. M+DAPT, model+DAPT.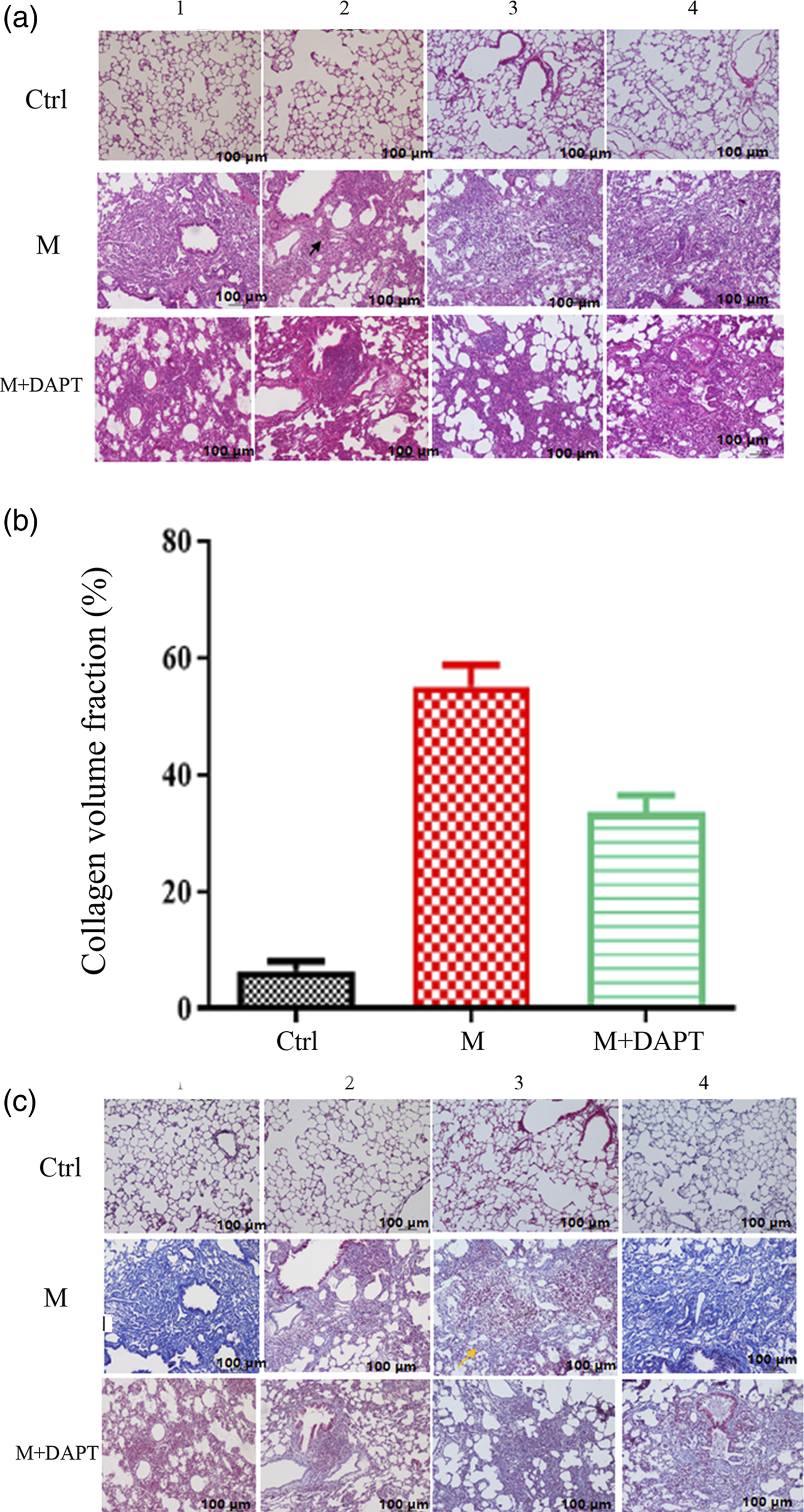
Levels of ligands and receptors in the Notch pathway
Western blotting analysis was performed to determine the levels of the key proteins of the Notch signaling pathway. The levels of the ligands DLL1, DLL4, JAG1, and JAG2 and the receptors Notch1 and Notch2 of the Notch signaling pathway in lung tissue were higher in the M group than in the control group. Among these molecules, the ligand DLL1 and the receptor Notch2 were significantly increased in the M group (p < 0.05). The levels of DLL1, JAG1, JAG2 and receptor Notch2 decreased after the Notch signaling pathway was inhibited by DAPT (p < 0.05). Although the protein levels of DLL4 and Notch1 were also decreased, the changes were not significant (p = 0.17 and p = 0.56, respectively) (Figure 4). These findings indicated that the Notch signaling pathway was activated in rats with fibrotic HP, but this response was blocked by DAPT. Western blotting analysis of the ligands and receptors in the Notch pathway in the three groups (9 rats in each group). (a): Analysis of key Notch pathway protein levels. (b)-(g): Levels of the receptor and ligand proteins of the Notch signaling pathway. The letter a indicates p < 0.05 for the M group versus the control group, and the letter b indicates p < 0.05 for the M+DAPT group versus the M group. Ctrl, control. M, model. M+DAPT, model+DAPT.
mRNA expression of inducible nitric oxide synthase (iNOS) and Arg-1
PCR was performed to determine the expression of the macrophage marker genes iNOS and Arg-1 (Figure 5). The mRNA expression of the M1 marker genes TNF-α and iNOS in macrophages was significantly lower in the M group than in the control group, whereas the mRNA expression of the M2 marker genes Mrc2 and Arg-1 was significantly higher in the M group (p < 0.05). After DAPT was applied, the relative mRNA expression of TNF-α and iNOS increased, while that of Mrc2 and Arg-1 decreased (p < 0.05). These findings indicated that inhibition of macrophage M1 polarization reduced lung injury caused by related inflammatory mediators, while inhibition of M2 polarization effectively delayed the occurrence of pulmonary fibrosis. The expression of the M1 macrophage marker genes TNF-α and iNOS and the M2 macrophage marker genes Arg-1 and Mrc2 in the three groups (9 rats in each group) according to PCR. The letter a indicates p < 0.05 for the M group versus the control group, and the letter b indicates p < 0.05 for the M+DAPT group versus the M group. Ctrl, control. M, model. M+DAPT, model+DAPT.
Th1/Th2 ratio
The ratio of Th1 to Th2 cells in the lung tissue of each group was determined by flow cytometry (Figure 6). Compared with the control group, the M group had a significantly decreased Th1/Th2 ratio (p < 0.05). Compared with the M group, the M+DAPT group had a significantly increased Th1/Th2 ratio (p < 0.05). These findings indicated that the Th1/Th2 ratio was reduced in rats with fibrotic PBLs and that inhibiting the Notch signaling pathway could alleviate this reduction. Th1/Th2 ratio analysis of lung tissue in the three groups (9 rats in each) according to flow cytometry. The letter a indicates p < 0.05 for the M group versus the control group, and the letter b indicates p < 0.05 for the M+DAPT group versus the M group. Ctrl, control. M, model. M+DAPT, model+DAPT.
Discussion
PBL is an allergic disease mediated by Th1 cells. 14 After proteins from pigeon feathers, dander, and other exfoliated proteins are inhaled, they are engulfed by the pulmonary macrophages of the host to induce Th0-to-Th1 activation. Activated Th1 cells further activate macrophages by secreting IFN-γ and TNF-α. Persistent antigen exposure may result in the development of interstitial lung disease and pulmonary fibrosis. As the most abundant immune cells in the lungs (approximately 70% of immune cells), macrophages play an important role in airway remodeling in pulmonary fibrosis diseases. 15 The Notch signaling pathway is the key regulator of macrophage biological function. 16 Under different pathological conditions, the Notch pathway can determine the M1/M2 activation of macrophages, and application of the Notch signaling pathway blocker DAPT can delay the progression of pulmonary fibrosis. 17 This study aimed to establish a rat model of PBL and to study the role of the Notch signaling pathway in the Th1/Th2 imbalance during pulmonary fibrosis in PBL by regulating the macrophage polarization.
We successfully established an animal model of fibrotic HP by using pigeon exfoliates collected from the pigeon house of Xinjiang Xintou Pigeon Leaf Co., Ltd, in Kashgar, Xinjiang. 18 In this study, we divided the 18 PBL rats into the M group and the M+DAPT group and found that DAPT intervention greatly reduced the severity of inflammatory cell infiltration and fibrosis while preventing definitive granuloma formation.
The Notch signaling pathway is a highly conserved signal transduction pathway that participates in lung regeneration under healthy and diseased conditions. This pathway may therefore be a promising target for host-directed therapies to combat lung pathology. 19 Humans have four Notch receptors (Notch1-4). These receptors can be activated by the binding of ligands on the surfaces of adjacent cells, including Jag1 and Jag2 and Delta-like members (DLLs 1, 3, and 4). 20 Notch receptors and ligands are expressed by immune cells at different stages (e.g., monocytes, lymphocytes, macrophages and dendritic cells), and they can activate T cells and change the expression of related cytokines during the proinflammatory immune response. Although the expression spectra of Notch receptors in different cells of the respiratory tract remain unclear, Notch signaling is conserved during the development of the human respiratory system. 21 Notch1 is continuously expressed throughout fetal lung development and is expressed in alveolar epithelial and vascular endothelial cells after birth. The Notch ligands Jagged1 and Jagged2 are expressed in the pulmonary interstitium. DLL is a dendritic cell-derived Th1 differentiation signal that plays a role in promoting the differentiation of primitive CD4+ T cells into Th1 cells. A previous study has shown that the cross-binding of the Notch receptor and DLL1-Fc fusion protein can stimulate the Th1 cell response. 22 DLL4 can not only promote M1 macrophage differentiation but also inhibit M2-specific gene expression and apoptotic cell death to prevent M2 macrophage differentiation, thus promoting the Notch-dependent selection process. 23 In our study, we found that the expression levels of key proteins of the Notch signaling pathway, namely, DLL1, DLL4, JAG1, JAG2, and the receptors Notch1 and Notch2, were increased in the rat model of fibrotic HP. After DAPT was applied to inhibit the Notch signaling pathway, the expression of DLL1, JAG1, JAG2 and the receptor Notch2 decreased. It has been reported that the activation and differentiation of macrophages are often accompanied by increased expression of the Notch1 and Notch2 receptors. 24 To a certain extent, the Notch signaling pathway interacts with macrophages; activation of Notch signaling promotes the inflammatory response of macrophages, while macrophages release inflammatory mediators or activate their own Notch signaling pathways to increase the expression of Notch receptors and ligands. The results of this study are consistent with recent groundbreaking research showing the expression of Notch receptors and ligands in macrophages. 25 In that study, DLL4 expression was significantly increased in primary human macrophages in response to proinflammatory stimuli (lipopolysaccharide (LPS), IL-1β, and low-density lipoprotein). Our previous studies have highlighted the interaction between Notch signaling and endothelial/monocyte/macrophage inflammation and reported the activation of Notch in monocytes; this activation, triggered by endothelial DLL4 and IL-6, promotes the proinflammatory M1 phenotype of macrophages. Macrophages express Notch receptors (such as Notch1) and ligands, and activation of typical Notch signals contributes to M1 gene expression and cytokine production. DAPT, an inhibitor of Notch signaling, can inhibit the proliferation of CD4+ T lymphocytes in a time- and dose-dependent manner, block the epithelial–mesenchymal transformation (EMT) of alveolar epithelial cells, and delay the onset and development of pulmonary fibrosis. 26 Therefore, the Notch signaling pathway can promote the polarization of macrophages, which impacts pulmonary fibrosis in PBL patients.
The Notch signaling pathway is the key regulator of the biological function of macrophages, which can be polarized into classically activated (M1) macrophages and alternatively activated (M2) macrophages under different environmental stimuli. 27 For example, under different pathological conditions, the Notch pathway can determine the M1/M2 activation mode of macrophages and trigger the production of iNOS and a variety of cytokines, including IL-4, IL-13, and other inducers of the c-Jun N-terminal kinase (JNK) and extracellular regulated kinase (ERK) mitogen-activated protein (MAP) pathways, ultimately affecting downstream T-cell differentiation. TNF-α can also induce the expression of Notch1 and Notch4. 28 In this study, the mRNA expression of the M1 marker genes TNF-α and iNOS was lower in the pulmonary macrophages of rats with fibrotic HP than in those of normal control rats. The mRNA expression of the M2 marker genes Mrc2 and Arg-1 in macrophages increased (p < 0.05) after allergen stimulation, indicating that the M1 polarization of macrophages increased and the M2 polarization of macrophages decreased in this rat fibrotic PBL model. These findings suggest that macrophages in the PBL fibrotic phase can inhibit inflammatory reactions, promote fibrosis, and repair injury. This is consistent with the polarization of macrophages observed by Chen et al. 29 in acute lung injury. After inhibition of the Notch signaling pathway by DAPT, the relative mRNA expression of TNF-α and iNOS increased, while the relative mRNA expression of Mrc2 and Arg-1 decreased, indicating that M1 polarization decreased and suggesting that Notch may be involved in macrophage M1 polarization. M1 macrophages have proinflammatory properties, and Notch can promote the polarization of macrophages to the M1 phenotype. 29 We also found this feature in the rat model of pulmonary fibrosis in PBL. After administration of a Notch signaling pathway inhibitor, macrophages in PBL fibrotic rats polarized to the M2 phenotype. Previous research has shown that M2 macrophages can produce fibrogenic mediators and promote the proliferation of myofibroblasts. 30 In this study, the rats were in a state of pulmonary fibrosis and showed increased M2 polarization of macrophages, indicating that DAPT, an inhibitor of the Notch signaling pathway, affected macrophage M2 polarization in the rat model of pulmonary fibrosis in PBL.
In this study, Th1 cytokine levels decreased gradually with the progression of pulmonary fibrosis. 31 In contrast, Th2 cytokine levels increased gradually, and the immune response transformed into a Th2-dominated immune response in PBLs. Additionally, compared with the control group, the M group presented a reduced Th1/Th2 ratio, while after DAPT intervention, the ratio was significantly increased (p < 0.05). This finding indicates that inhibiting the Notch signaling pathway affects Th1/Th2 imbalance and can reduce the severity of pulmonary fibrosis in PBL rats.
This study had some limitations. First, the number of animals involved in this study was small, so we did not perform a power calculation of the final outcome. In the future, studies with larger sample sizes should be performed. Second, this study involved only animal experiments; thus, the findings should be validated in future human trials, especially considering the differences in the animal and human microenvironments.
Conclusions
In this study, we successfully established a rat PBL model with freeze-dried powder made of pigeon exfoliates. Our findings reveal that DAPT can attenuate fibrotic changes and that macrophage polarization plays an important role in regulating the development of pulmonary fibrosis. In addition, our results show that inhibiting the Notch signaling pathway can ameliorate the Th1/Th2 imbalance by regulating the macrophage polarization and alleviate the severity of pulmonary fibrosis in rats with fibrotic HP. The findings of this study may provide a new target for blocking the development of fibrosis in hypersensitivity pneumonitis.
Footnotes
Authors’ note
The present study followed international, national, and institutional guidelines for humane animal treatment and complied with relevant legislation.
Acknowledgments
This study was financially supported by the Autonomous Region Natural Fund Project (2020D01C099) and the Regional Fund Projects of the National Natural Science Foundation of China (81960005). The sponsor had no role in article preparation, study design, data analysis, statistical analysis, review of drafts, writing of the article, or identification of papers for inclusion, nor did the sponsor have any other form of input.
Author contributions
CW and XHY devised the study plan, WD and QPL collected the data, YZ was responsible for data analysis, ZP and YYS interpreted the data, WYW drafted the manuscript, and CW supervised the whole process and gave constructive advice. All authors have made contributions to the current work.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Regional Fund Projects of the National Natural Science Foundation of China (81960005), the Autonomous Region Natural Fund Project (2020D01C099).
