Abstract
Glioma stem cells (GSCs) live in a continuous process of stemness reprogramming to achieve specific cell commitment within the so-called GSC niches, specifically located in periarteriolar regions. In this review, we analyze the expression levels, cellular and subcellular location, and role of three scaffold proteins (IQGAP1, FKBP51, and AmotL2) in GSC niches. Scaffold proteins contribute to cell differentiation, migration, and angiogenesis in glioblastoma. It could be of diagnostic interest for establishing stages, for therapeutic targets, and for improving glioblastoma prognosis, which is still at the experimental level.
Keywords
Introduction
Glioma stem cells (GSCs) and astrocytoma cells are the predominant malignant cells in glioblastoma. The continuous stemness cell reprogramming of GSCs to achieve specific cell commitment is a complex process within the so-called GSC niches, initially defined as perivascular and hypoxic.1–4 Subsequent studies found them to be specifically located in periarteriolar regions2,4 similar to hematopoietic stem cell niches in the bone marrow.3,5 Recent studies indicate that the subventricular zone in glioblastoma patients is a major niche for neural stem cells in the brain, forming a niche protected against radiotherapy, chemotherapy, and resection and consequently may contribute to tumor recurrence after therapy. 6
Scaffold proteins are known to play major roles in cell fate, angiogenesis, migration, tumorigenesis, and tumor progression.7,8 However, very little is known about these proteins in the GSC niche. Scaffold proteins (scaffolins or scaffoldins) are a growing group of proteins of signal transduction pathways that share four common functions within a given node of the network: (1) they physically connect all proteins involved in that node; (2) they locate them in discrete subcellular compartments; (3) they coordinate the equilibrium between positive and negative stimuli; and (4) they insulate or encase all proteins in the node to protect them from inactivation and/or degradation originating outside the pathway. 9 A-kinase anchor proteins, within the protein kinase A signaling pathway, could be the paradigm of this group of proteins.9,10 Wide variations in their gene expression in neuropathy after chemotherapy are a common feature of three scaffold proteins, AmotL2 (angiomotin-like 2), IQGAPI, and FKBP51 (FK506-binding protein 51), currently under investigation by our group. 11
In this review, we will consider the expression and role of AmotL2, IQGAP1, and FKBP51 in stem cell niches.
AmotL2, IQGAP1, and FKBP51 Scaffoldins in GSC Niches
The protein AmotL2 is fundamental for maintaining apical-basal polarity in epithelial tissues by regulating cell-to-cell membrane interactions. 12 It plays a pivotal role in preventing endothelial detachment and promoting vascular tube formation.13–16 The regulation of AmotL2 by hypoxia and its effect on cellular migration make the study of this protein of particular interest in cancer progression and more specifically in tumor angiogenesis.17,18 Human AmotL2 occurs as two isoforms with a molecular mass of 100 and 60 kDa. 12 The 60-kDa isoform seems to be involved in promoting tumor growth and invasion by perturbing cellular polarity in a complex process, which is beyond the scope of this review. 15
The most frequent stencil for AmotL2 immunohistochemistry is strong AmotL2 costaining in Nestin+ GSCs (Fig. 1, panels A–C). Within the same stencil, scattered GFAP+ astrocytoma cells can be either AmotL2+ or AmotL2− in not yet well-defined proportions. However, the expression pattern for AmotL2 in the glomerular blood vessels shown is very different: a number of AmotL2+ cells in arterioles predominantly surround CD34+ endothelial cells, but, strikingly, cubic-shaped endothelial cells are not surrounded by AmotL2+ cells. 19 This fact might be indicative of a role for AmotL2+ GSCs in periarteriolar areas. AmotL2 is localized in cytoplasm and protrusions of differentiated GFAP+ GLIOBLASTOMA cells, as shown in the double immunofluorescence micrographs in Fig. 1, where most cells are either AmotL2+/GFAP− or AmotL2−/GFAP+. However, cells that were both AmotL2+/GFAP+ occurred less frequently (see Fig. 1, panels D–F).
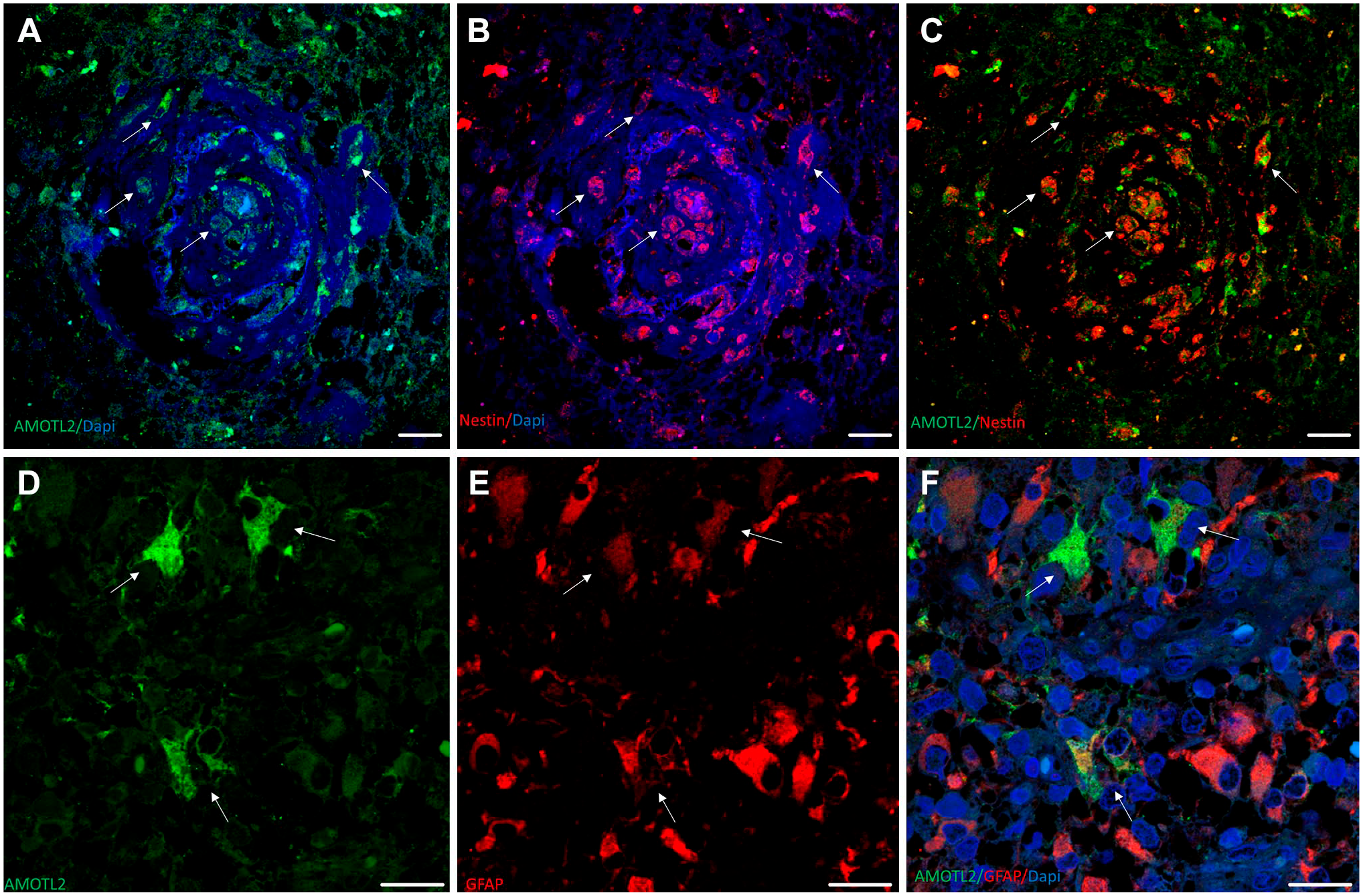
Immunolabeling of AmotL2, Nestin, CD34, and GFAP in human glioblastoma sections. (A–C) Vascular glomeruloid bodies. Several AMOTL2+/Nestin+ cells can be observed (arrows). (D–F) Double immunolabeling for AMOTL2 and GFAP. Arrows point to GB-differentiated cells GFAP+/AMOTL2+, where AMOTL2 is localized in cytoplasm and protrusions. Scale bar, 20 µm. Abbreviations: GFAP, glial fibrillary acidic protein; GB, glioblastoma.
Graphically well described as a molecular puppeteer, 20 IQGAP1 is the IQ motif that contains Ras GTPase-activating protein 1. IQGAP1 is a highly versatile regulating protein involved in cellular processes, ranging from cytoskeleton organization to cell cycle. Ubiquitously expressed in cells and tissues,20,21 it determines the direction that should be taken in a given node in many signaling pathways,22–26 thereby contributing to processes such as cell migration, angiogenesis, and tumor progression.27–31 Establishing differences between the IQGAP1 function on healthy versus cancer cells and tissues is extraordinarily complex and, consequently, alluring for structural, molecular, and cancer and cell biology researchers. Rather than on structural variations, the changes in the function of the protein driving to pathological migration, cell cycle deregulation, and most oncogenic-related processes seem to be related to the amount of protein and the scaffolding groups of proteins nucleated by IQGAP1. An excellent example of IQGAP1 nucleation is with calmodulin, driving the activation of PI3Kα and Akt in KRAS-, HRAS-, and NRAS-driven cancers. 32 Thereby, it seems that a given protein not so frequently attached to IQGAP1 is mutated (or splices a deletion) and increases its affinity for this scaffoldin and nucleates a node that ultimately increases the activity of a pathway, deregulates a complex cell function, and initiates or cooperates in tumor progression.
In glioblastoma, the amount of IQGAP1 protein and subcellular localization are heterogeneous, located in cytosol, nucleus, and plasma membrane, but more prominent in podosome/invadopodia-like structures,19,30 filopodia, 33 and microvesicles ready to be released bound to its upstream effector cdc42. 24 Though ubiquitous, Nestin+ cells express low levels of IQGAP1 or no IQGAP1 at all (Fig. 2, panels A–B), as in GFAP+-differentiated astrocytoma cells, where IQGAP1 expression is variable. However, IQGAP1 is very abundant in the cell membranes and nuclei of endothelial cells of arterioles (Fig. 2, panels C–D), mainly in podosomes/invadosomes. 30 Moreover, IQGAP1 is also found in the soma of scarce neurons, but not in the axon. 30 Recently, specifically created stable IQGAP1 knockdown MA2 melanoma cells were used to confirm IQGAP1 as a crucial protein for metastasis through the extravasation regulation of these cells when injected in mice. 34 Furthermore, it has been recently reported that the co-occurrence of glioblastoma and melanoma is likely not coincidental and shows several molecular causes in common, 35 including IQGAP1expression and its interaction with members of the S100 family of Ca2+-binding proteins. 36
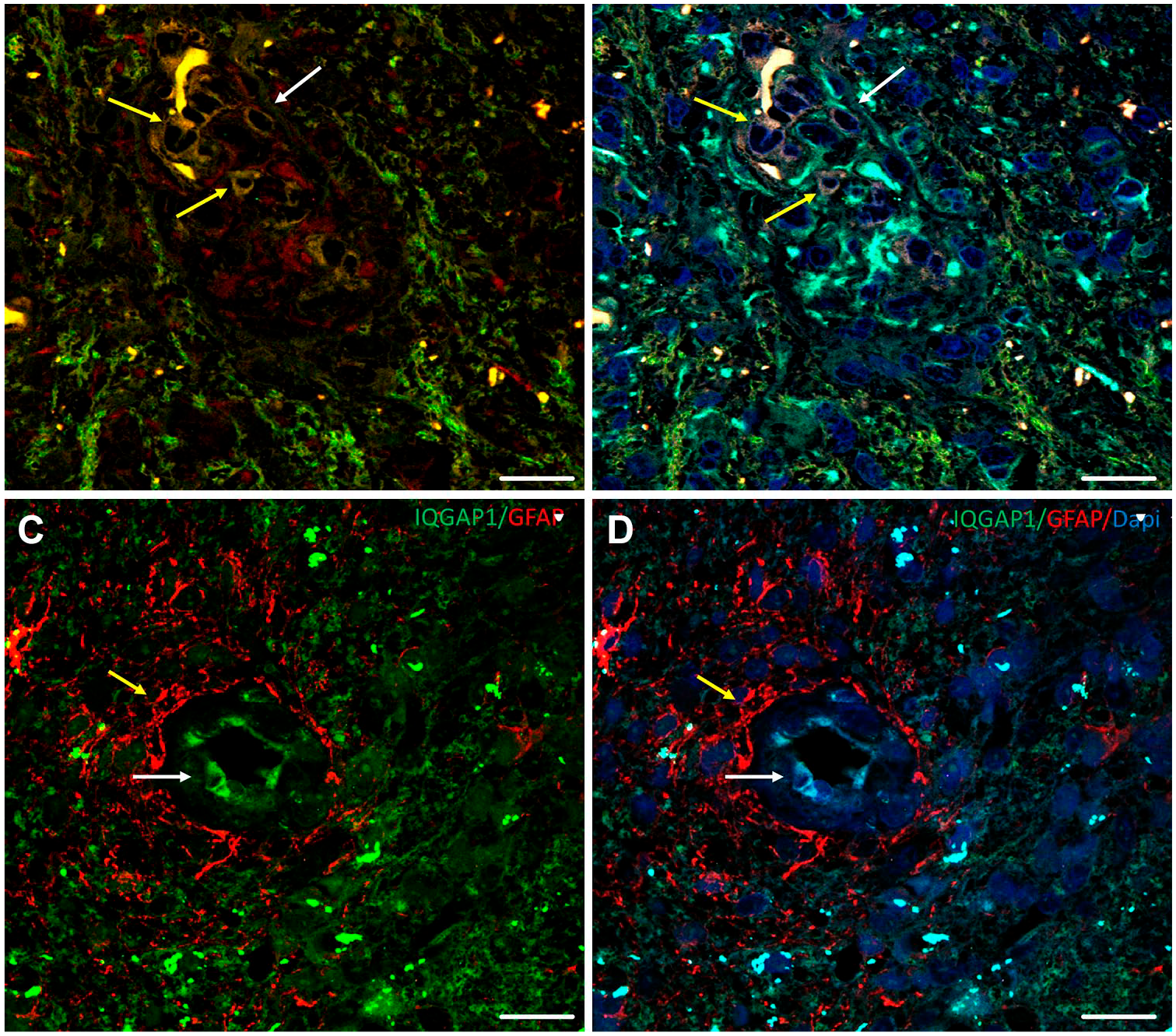
Immunolabeling of IQGAP1, Nestin, GFAP, and F-actin in human glioblastoma tissue sections. (A–B) IQGAP1+/Iba1+/Nestin+ (white arrow) and IQGAP1+/Iba1+/Nestin− (yellow arrows) cells are present in GB vascular glomeruloid body. (C–D) Tumor-associated arteriole: IQGAP1+ immunolabeling is mainly present in the cytoplasm and nuclei of endothelial cells (white arrow). Note GFAP+ protrusions enveloping the blood vessel. Scale bar, 20 µm. Abbreviations: GFAP, glial fibrillary acidic protein; GB, glioblastoma.
FKBP51 (gene: FKBP5) is an immunophilin protein, a peptidyl-prolyl isomerase, 37 and a cochaperone in the Hsp90 system in a concerted antagonist role with its homologue, FKBP52. 38 FKBP51 commutes among cytoplasm, nucleus, and mitochondria,39–42 interacting with many molecular partners in pivotal nodes in several signaling pathways.38,43 Altered expression levels of FKBP51 have been reported in a number of cancers and cancer models,43–49 including gliomas and glioblastoma,50–54 as well as in mental disorders. 55
Virtually all types of human glioblastoma tumor cells express FKBP51 in a different cellular and subcellular fashion. A subpopulation of Iba1+/CD31+ macrophages/microglia expresses FKBP51 in nuclei (Fig. 3, panel A, white arrows), whereas in other macrophages/microglia groups FKBP51 is found in the cytoplasm. 19 In GFAP+/FKBP51+ cells, FKBP51 is also intranuclear, although to a lesser extent. 19 FKBP51+/CD31+/Iba1+ cells are adjacent to the lumen of arterioles (Fig. 3, panels A–C, yellow arrow) but not around capillaries. To our knowledge, no specific association of FKBP51 expression levels with resting or active microglia has been reported in glioblastoma; however, it is interesting to mention an association of FKBP51 protein in the altered immune function and inflammation state of microglia and the development of psychological disorders, including anxiety and depression.56–59 Regarding FKBP51 protein expression in stem cells, it is common to find a high amount of this immunophilin, along with actin-rich core-like cellular structures in Nestin+ GSC cells (Fig. 3, panels D–F, white arrows).
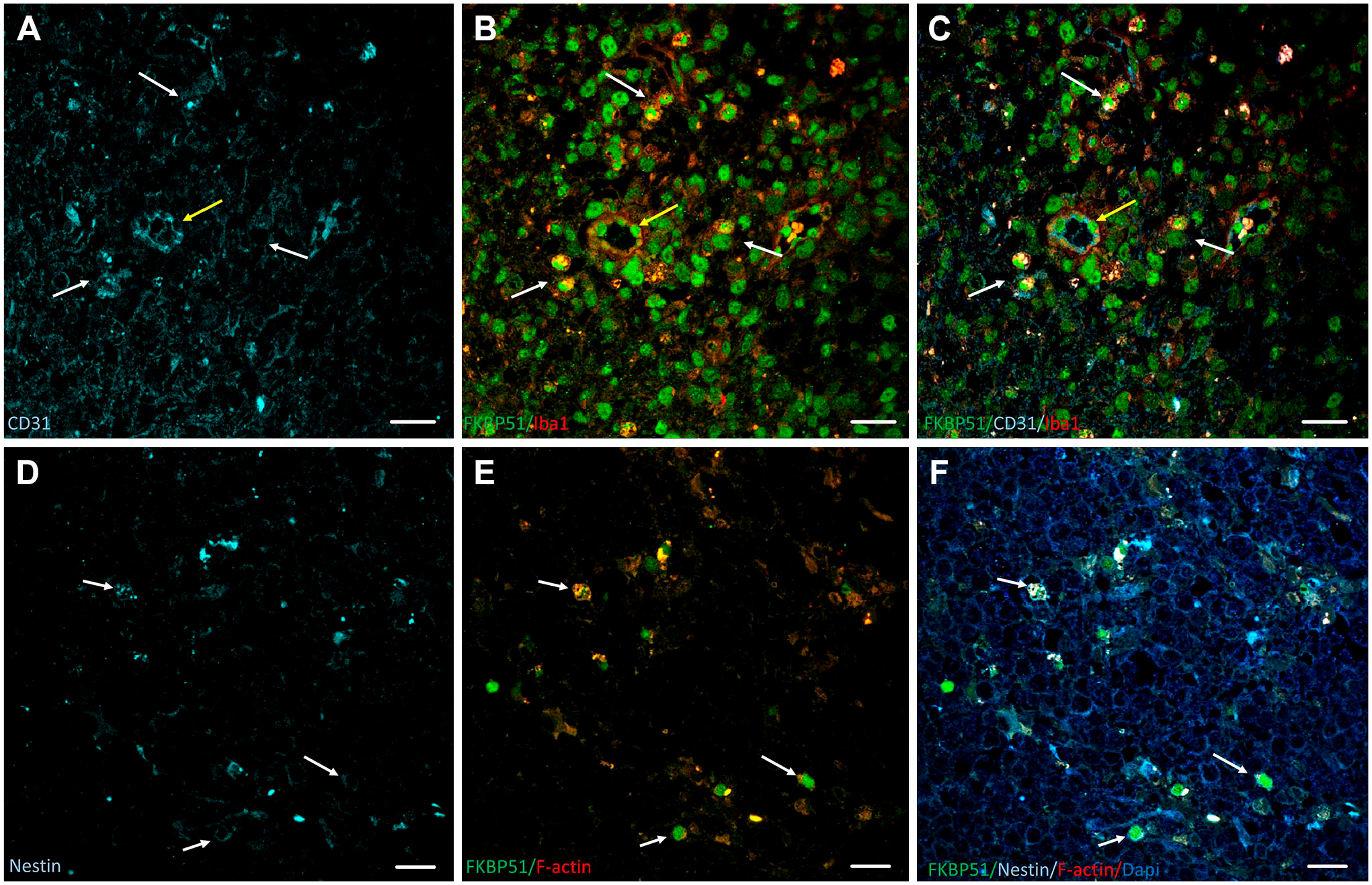
Immunolabeling of FKBP51, CD31, Nestin, Iba1, and F-actin in human glioblastoma tissue sections. (A–C) Iba1+/CD31+ macrophages/microglia express FKBP51 in nuclei (white arrows). Yellow arrow points to FKBP51+/CD31+/Iba1+ cells adjacent to the lumen of arterioles. (D–F) White arrows point to FKBP51+/Nestin+ cells, where FKBP51 protein localizes along with the actin-rich core-like cellular structures. Scale bar, 20 µm.
Concluding Remarks, Glioblastoma Staging, and Therapy Perspectives
Rather than filling moonlighting roles, scaffold proteins have multifunctional molecules that hide proteins to avoid contact with others and guide the signal in a specific direction. The final objective is to modulate complex cell functions such as migration, proliferation, or regulation of gene expression, all three on the basis of cancer. As reviewed here, AmotL2, IQGAP1, and FKBP51 are greatly involved in the angiogenesis and cell migration of glioblastoma. Although several authors have identified that AmotL2, 60 FKBP51, 59 or IQGAP153,61,62 protein expression levels exert a relevant role in glioma progression and aggressiveness, a scale for pathology staging or prognosis has not been stablished yet. Consequently, the potential use of these scaffoldins as milestones for staging or as references for prognosis is worth to be set and considered as targets for the treatment of glioblastoma. In this later line of research, successful in vitro experiments blocking any of these scaffoldins in several cancer cells have been reported.
AmotL2 is accepted as a tumor suppressor. 22 The expression of AMOTL2 can be increased through miR-135b and miR-135a inhibition, resulting in a remarkable decrease in cell viability, self-renewal, and stem cell marker expression in spheroids obtained from medulloblastoma. 63
IQGAP1 small interfering RNA significantly inhibited cell proliferation, adhesion, migration, and invasion in U251 and U373 glioma cell lines. 64 Direct repression of IQGAP1 by epigenetically silenced miR-124 in endometrial cancer cells can reverse epithelial-to-mesenchymal transition by inhibiting IQGAP1 gene expression. 65 The Pseudomonas aeruginosa N-acyl homoserine lactone targets IQGAP1 and modulates epithelial cell migration 65 in Caco-2 cells. However, this product does not affect proliferation or viability.
IQGAP1 nucleates MEK and its target ERK, and the increase and decrease in intracellular IQGAP1 concentrations result in impairment of stimulation of MEK and ERK mitogenic activity 23 ; consequently, designing drugs for blocking this node is an exciting topic for researchers in onco-pharmacology.
FKBP51 overexpression inhibits BT325 cell proliferation by inducing G0/G1 cell cycle arrest. This overexpression enhances the susceptibility of BT325 to carmustine, a standard adjuvant chemotherapy for glioma treatment. 66 In a search for depression and posttraumatic stress disorder treatments, through a screening of 1280 pharmacologically active compounds, three compounds rescued FKBP51-mediated suppression of glucocorticoid receptor (GR) and one disrupted the association of FKBP51 with GR/Hsp90. 67 Some of these compounds targeting the FKBP51/GR/Hsp90 complex may be a viable approach for treating glioblastoma because FKBP51 is expressed at high levels in all kinds of glioblastoma cells, including GSCs and vascular endothelial cells. 19
Evidence pointed out in this review shows a promising research field in physiopathology, personalized diagnosis, and subsequent scaffoldin-directed therapy of glioblastoma that, unquestionably, will improve bad prognosis currently.
Footnotes
Acknowledgements
The authors are grateful to Prof. Cornelis van Noorden, Dr. Stephen Hewitt, and Dr. Vashendriya Hira for inviting us to write this review and to Ms. Chanel George for editorial managing.
Competing Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
All authors conceptualized and discussed the manuscript. DR and JA edited the figures. PM-V coordinated the production.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The research group was supported by grants FIS PI11/00114 to P.M.-V. and grant FIS PI12/00729, Spain, to J.A.
