Abstract
Grainyhead-like 2 (Grhl2) is a transcription factor regulating cell adhesion genes. Grhl2 acts as an epithelial–mesenchymal transition suppressor, and it is a proto-oncogene involved in estrogen-stimulated breast cancer proliferation. However, its expression during ovarian hormone–dependent mammary ductal development remains obscure. We here examined Grhl2 expression in the mammary gland of normal and steroid-replaced ovariectomized mice. Grhl2 protein signals were detected in both the mammary luminal epithelial and myoepithelial nuclei. The ratio and density of Grhl2-positive nuclei increased after the onset of puberty and progressed with age, whereas Grhl2-negative epithelial cells were detected in mature ducts. Claudin 3, claudin 4, claudin 7, and E-cadherin gene expression in the mammary gland was upregulated, and their expression was highly correlated with Grhl2 gene expression. Furthermore, Grhl2 mRNA expression and ductal lumen width were significantly increased by the combined treatment of estrogen and progesterone compared with estrogen alone. These results suggest that Grhl2 expressed in the luminal epithelial and myoepithelial cells from the early phase of ductal development, controlling the expression of cell adhesion molecules to establish functional ducts:
Keywords
Introduction
The mammary gland is a unique organ that primarily develops postnatally and forms in the ventral subcutaneous adipose tissue under the control of ovarian steroids. Newborn female mice possess quiescent rudimentary mammary ducts near their nipples that invade the surrounding stromal adipose tissue around 3–4 weeks of age and occupy the entire fat pad during puberty. The invading duct tips contain highly proliferative cell populations, and these structures are referred to as the terminal end buds (TEBs). They occasionally bifurcate into branch ducts to complete the ductal tree.1,2 Moreover, some side branches sprout laterally from the main ducts, and additional side branches of a different character begin to develop in the luteal phase. These additional side branches become the milk-secreting acini during pregnancy and after delivery. The acinar structure recedes by extensive apoptosis after weaning, whereas the primary ductal structures remain in their prepregnant state.3–5
Tight junctions (TJs) are composed of several integral membrane proteins, including occludin and claudin (Cldn), as well as ~30 cytosolic molecules. 6 In the mammary glands, TJs between adjacent secretory epithelial cells are formed during the period of lactogenesis and are instrumental in maintaining the difference between the basolateral side and the apical side of cells required for milk synthesis and secretion. 7 Milk stasis by discontinued suckling causes the alveolar size to increase and epithelial cells to flatten, followed by an increase in Cldn1 and Cldn3 protein expression and a decline in Cldn2 and Cldn16 protein expression. 8 Furthermore, a study on Cldn expression and localization in mammary gland development revealed that Cldn7 is present throughout development, Cldn3 is localized basolaterally to the TJ, and Cldn4 is found only in sparse puncta. 9 In addition to the TJ, cadherins contribute to cell–cell contact and communication. E-cadherin as an adhesion receptor is critical for normal epithelial architecture development and may act as a tumor suppressor because its downregulation in cancer cells induces phenotype alterations from epithelium to mesenchymal characteristics. This process is called the epithelial–mesenchymal transition (EMT) and leads to increase motility, invasiveness, and metastasis. 10
Grainyhead-like 2 (Grhl2) is one of the three genes in a family of mammalian transcription factors related to Drosophila Grainyhead 11 and activates gene expression encoding epithelial cell adhesion molecules, such as E-cadherin and Cldn4.12,13 Therefore, Grhl2 plays pivotal roles in epithelial barrier formation during kidney collecting duct development, 14 placental trophoblast branching morphogenesis, 15 and mucociliary airway epithelium establishment and maintenance. 16 In addition, Werner et al. 17 have demonstrated that Grhl2 has transforming activity as a proto-oncogene in breast cancer. Grhl2 can also act as an EMT suppressor molecule.13,17 Furthermore, Grhl2 is essential for estrogen-stimulated proliferation in estrogen receptor–positive (ER+) breast cancer cells. 18
In postnatal mammary gland development, both stromal and epithelial ERα-mediated signals are required for ductal morphogenesis. 19 Although Grhl2 is detected in the nucleus in normal breast epithelium adjacent to invasive ductal carcinoma, 17 its regulation in postnatal mammary ductal development remains obscure. In this study, we examined Grhl2 expression and function during mouse mammary ductal development and the possible effect of reproductive hormone on its expression.
Materials and Methods
Animal and Tissue Sampling
The experimental and animal care procedures were approved by the Animal Care and Use Committee of Hokkaido University (Hokkaido, Japan). All experiments using mice were performed in accordance with the guidelines of Hokkaido University Manual for Implementing Animal, in the animal facility approved by the Association for Assessment and Accreditation of Laboratory Animal Care (AAALAC) International. C57BL/6J female mice were purchased from Japan SLC Inc. (Shizuoka, Japan; n=42), housed in plastic cages in an air-conditioned room at 23C with a 12:12 hr light:dark cycle, and given free access to laboratory chow (Oriental Yeast; Tokyo, Japan). When applicable, the estrus cycle status was determined by vaginal smear analysis with Giemsa staining. Predominant detection of white cells in the smear was referred to as “time of estrus” and detection of the cornified epithelium as “time of non-estrus.”
To analyze Grhl2 expression during postnatal development, 3-week-old (n=6), 7-week-old (n=6), and 17-week-old (both estrus and non-estrus n=6 each) female mice were used (total n=24). The mice were then euthanized, and the thoracic, abdominal, and inguinal mammary glands were quickly removed. Tissue samples were divided and transferred into RNAlater storage solution (Thermo Fisher Scientific; Gaithersburg, MD) for quantitative PCR analysis or 4% paraformaldehyde-containing phosphate-buffered solution (PB) for histological examination.
To examine the effect of ovarian steroid on Grhl2 expression in the mammary gland, mice at 5 weeks of age were given an ovariectomy under anesthesia with a mixture of ketamine (75 mg/kg; Daiichi-Sankyo Propharma, Tokyo, Japan) and medetomidine (1 mg/kg; ZENOAQ, Fukushima, Japan). After the surgery, the mice received an intraperitoneal injection of antibiotics (ampicillin 5 mg/mouse; Meiji Seika, Tokyo, Japan) and a subcutaneous injection of atipamezole (1 mg/kg; ZENOAQ). After 1 week of the recovery period in terms of body weight, the ovariectomized mice received a daily single subcutaneous injection of corn oil as a control (50 µl, Co, n=4), estrogen (E, 1 µg/50 µl, n=4; Wako Pure Chemical, Osaka, Japan), progesterone (P, 1 mg/50 µl, n=5; Wako), or both estrogen and progesterone (EP, 1 µg E and 1 mg P/50 µl, n=5) for eight consecutive days (total n=18). Mammary glands were quickly excised 24 hr after the last injection and stored in RNAlater solution or 4% paraformaldehyde-containing PB.
Real-time PCR
Total RNA was extracted using the RNAiso reagent (Takara Bio; Shiga, Japan) according to the manufacturer’s protocol. Then, mRNA was purified with a Dynabeads mRNA Purification kit (Life Technologies; Carlsbad, CA) according to the protocol provided. The mRNA was reverse-transcribed using a 15-mer oligo(dT) adaptor primer and M-MLV reverse transcriptase (Promega; Madison, WI). Real-time PCR was performed on a fluorescence thermal cycler (LightCycler system; Roche Diagnostics, Mannheim, Germany) using FastStart Essential DNA Green Master Mix (Roche Diagnostics). Absolute expression levels were determined using a standard curve method with cDNA fragments as standards. The mRNA levels are expressed as relative values compared with β-actin mRNA levels. The primers used in this study are listed in Table 1.
Primer List Used in This Study.

Abbreviations: F, forward primer; R, reverse primer; Cldn, claudin.
Histology
Tissue specimens were fixed in 4% paraformaldehyde-containing PB, embedded in paraffin, and cut into 4-µm-thick sections that were stained with hematoxylin and eosin (H&E). The stained samples were examined under a light microscope (objective magnification ×4 or ×20, Olympus BX50F; Olympus Optical Co., Tokyo, Japan). After taking photographs, the epithelial cell height and maximal lumen width of 24 ducts in each experimental group were measured (see also Fig. 5A).
Immunohistochemical staining of Grhl2 protein was performed on an antigen retrieval section by heat treatment (105C, 15 min in 10 mM citrate buffer [pH 6.0]). The section was then sequentially incubated with 0.3% H2O2 for 20 min, with 10% normal goat sera (Histofine SAB-PO(R) kit; Nichirei, Tokyo, Japan) for 1 hr, and with the anti-Grhl2 rabbit polyclonal antibody overnight (500× dilution at 4C, HPA004820; Sigma-Aldrich Fine Chemical, St. Louis, MO). Control incubation was performed in the absence of the primary antibody. Afterward, the section was incubated with a biotin-labeled secondary antibody for 1 hr and peroxidase-conjugated streptavidin for 1 hr (Histofine SAB-PO(R) kit), with extensive washing between incubations. Finally, the section was incubated with 3′,3′-diaminobenzidine (DAB) solution for 10 min and hematoxylin for 10 sec. The stained samples were examined under a light microscope. The percentage of Grhl2-negative epithelial cells was assessed by counting all the Grhl2-stained and non-stained ductal epithelial cells of 19 mammary ducts from each experimental group.
Immunofluorescence staining was performed using the anti-Grhl2 antibody (200× dilution) and anti-α-smooth muscle actin (α-SMA, a myoepithelium marker) mouse monoclonal antibody (200× dilution, A5228; Sigma) or Alexa Fluor 594 anti-CD324 (E-cadherin) rat monoclonal antibody (50× dilution; BioLegend, San Diego, CA) in combination with Alexa Fluor 488 anti-rabbit IgG and Alexa Fluor 594 anti-mouse IgG antibodies (A11008 and A11005; Invitrogen, Carlsbad, CA), respectively. The nuclei were stained by 5 μg/ml Hoechst 33258 solution (Wako) for 1 hr. Immunofluorescence staining was also performed by anti–Cldn3 rabbit polyclonal antibody (100× dilution, 16456-1-AP; Proteintech, Rosemont, IL) or anti–Cldn4 rabbit polyclonal antibody (100× dilution, 16195-1-AP; Proteintech) with Alexa Fluor 594 anti-CD324 (E-cadherin) rat monoclonal antibody (50× dilution). Control incubation was performed in the absence of the primary antibodies.
Proliferating cell nuclear antigen (PCNA) and Ki-67 protein immunohistochemical staining was performed on an antigen retrieval section by 0.1% Triton X-100 for 1 hr. The section was then sequentially incubated with 1% H2O2 for 15 min, with 10% normal goat sera (Histofine SAB-PO(R) kit) for 30 min, and with the anti-PCNA rabbit antibody (400× dilution at 4C, sc-7907; Santa Cruz Biotechnology, CA, USA) or the anti-Ki-67 rabbit monoclonal antibody (200× dilution at 4C, MA5-14520; Invitrogen) overnight. The section was then labeled using a biotin-labeled secondary antibody for 1 hr and peroxidase-conjugated streptavidin for 1 hr (Histofine SAB-PO(R) kit), with extensive washing between incubations. Finally, the section was incubated with DAB solution for 10 min. Control incubation was performed without primary antibodies.
Data Analysis
The results are expressed as mean ± standard error (SE). Statistical analysis was performed by JMP Pro (14.0.0; SAS Institute Inc., Cary, NC), using one-way analysis of variance (ANOVA) followed by a Tukey–Kramer post hoc test. The correlation between Grhl2 mRNA expression and claudin family or E-cadherin mRNA expression was evaluated by Pearson’s correlation coefficient.
Results
Grhl2 mRNA and protein expression was examined in 3-week-old (around the onset of puberty), 7-week-old (puberty), and 17-week-old (adulthood) mouse mammary glands at the time of either estrus or non-estrus. Mice increased their body weight and mammary gland weight as a function of age (Fig. 1A and B). Grhl2 mRNA expression was increased 3- to 4-fold from 3 to 17 weeks (Fig. 1C). Grhl2 protein signals at 3 weeks of age were found in the nuclei, but the staining intensity was relatively weak compared with other age stages (Fig. 2A). In addition, non-stained epithelial nuclei (Fig. 2A, arrowheads in e and i) were observed in ~25% of the whole ductal epithelium (Fig. 2A and B). Grhl2 protein signals were increased and intensified at 7 weeks of age and were more densely expressed at 17 weeks irrespective of the estrus cycle status. The number of non-stained epithelial nuclei decreased to ~5%. Immunofluorescence staining confirmed Grhl2 non-stained cells present in the duct luminal region and the TEB front region. α-SMA-positive myoepithelium also expressed Grhl2 (Fig. 2C).
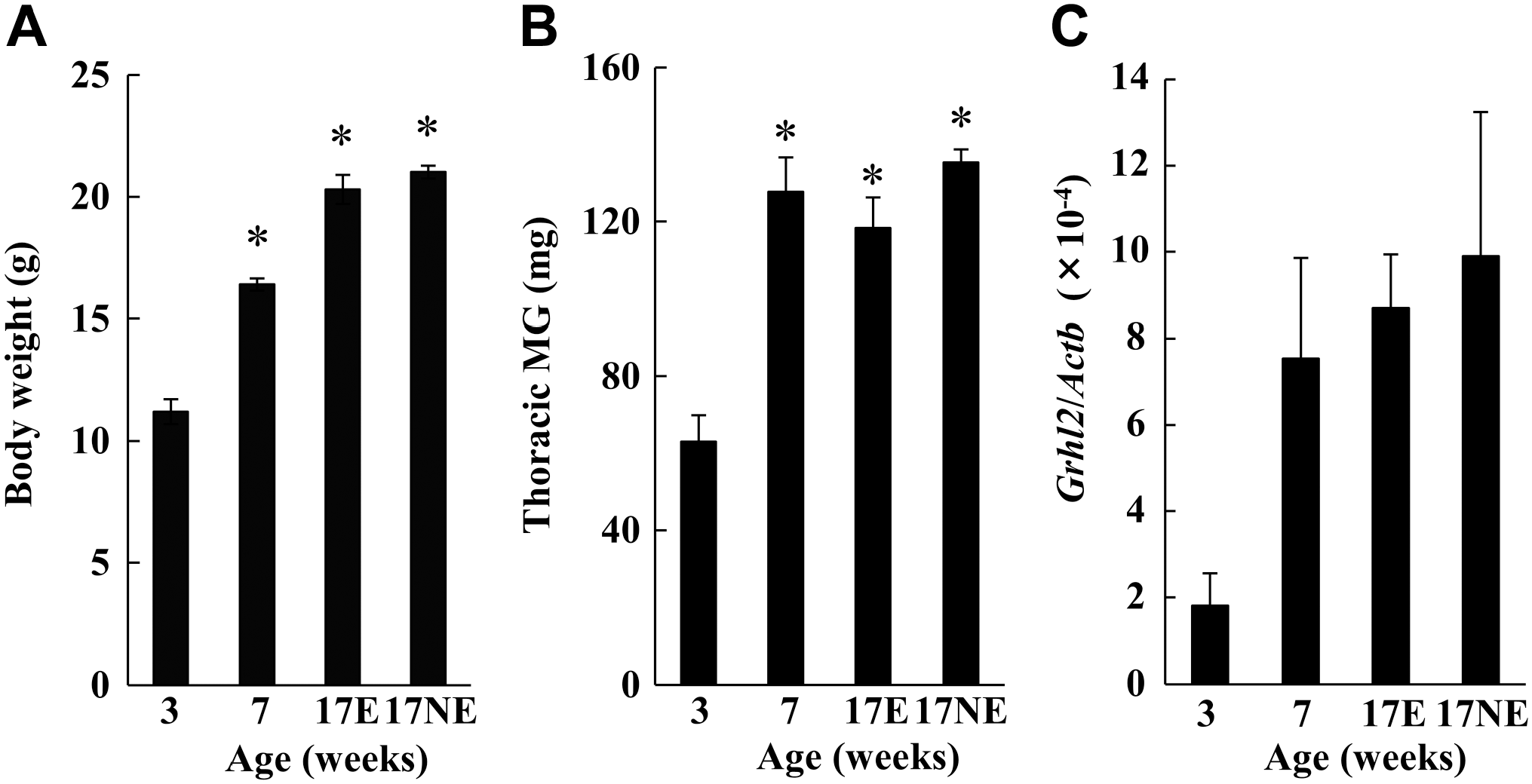
Changes in body weight, thoracic mammary gland weight, and Grhl2 mRNA expression during postnatal development in normal mice. Mice were sacrificed at 3 (n=6), 7 (n=6), and 17 weeks (n=12) of age. The body weight (A) and the thoracic mammary gland weight (B) were measured. Grhl2 gene expression in the abdominal mammary gland was determined by real-time PCR (C). Data for 17-week-old mice were analyzed depending on their estrus cycle status; 17E was in the time of estrus (n=6), whereas 17NE was in the time of non-estrus (n=6). *p<0.05 vs 3 weeks of age.
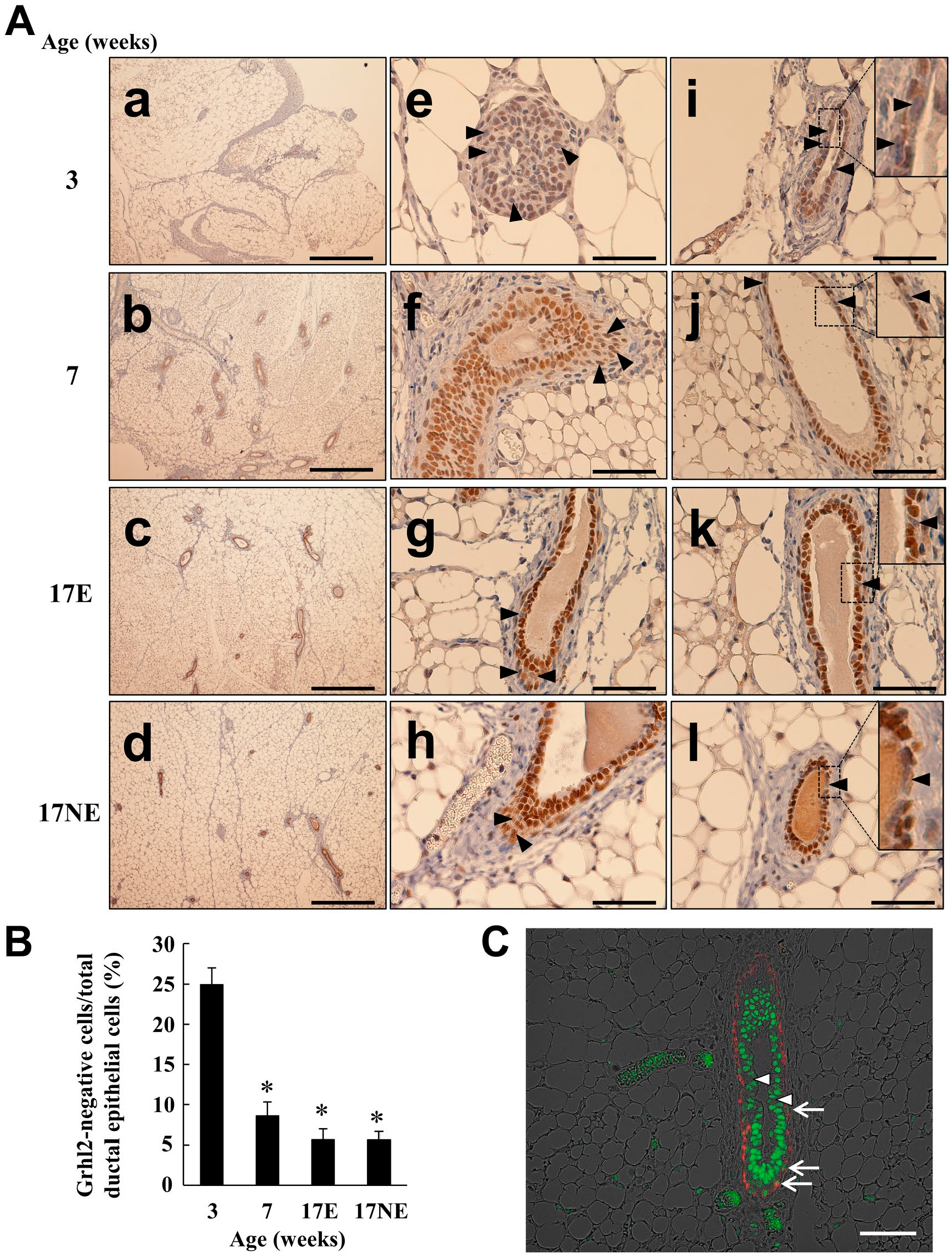
Grhl2 protein expression during postnatal development in normal mice. Mice were sacrificed as described in the Fig. 1 legend. In panel A, the first row (a, e, i) is from 3-week-old mice; the second row (b, f, j) is from 7-week-old mice; the third row (c, g, k) is from 17E; and the fourth row (d, h, l) is from 17NE. Arrowheads indicate the luminal epithelium not stained by the anti-Grhl2 antibody. In i to l, the insert shows the magnified image of the boxed region. In (B), the Grhl2-negative mammary epithelial cell percentage per total ductal epithelial cells was calculated by counting the cell number in 19 ducts from six mice in each age group. Tissue specimen from 7-week-old mice was also stained with both anti-Grhl2 (green) and anti-α-SMA (red, a marker of myoepithelium) in (C). Arrows indicate the myoepithelium Grhl2-expressing nuclei, whereas arrowheads show luminal epithelium not stained with anti-Grhl2. The scale bar in rows a to d in (A) indicates 500 µm, whereas the scale bar in rows e to l in (A) and (C) is 50 µm. *p<0.05 vs 3 weeks of age.
To determine the molecular mechanism of Grhl2 mRNA and protein expression in the mammary gland, the effect of ovarian steroid was examined in ovariectomized mice. Treatment of either estrogen or progesterone did not modify body weight or mammary gland weight, whereas combined treatment of both steroids slightly increased body weight (Fig. 3A and B). Grhl2 mRNA expression in the mammary glands decreased (Fig. 3C) and the number of Grhl2-negative cells increased in response to estrogen treatment but not to progesterone treatment (Fig. 4A and B). In addition, relatively dispersed nuclei were observed in estrogen-treated mice (Fig. 4A), accompanied by an increase in luminal epithelial cell height (Fig. 5) and cell proliferation signals, PCNA and Ki-67 (Fig. 6). Compared with estrogen-treated mammary glands, EP combined treatment significantly increased and decreased Grhl2 mRNA expression and the number of Grhl2-negative cells (Figs. 3C and 4). The ductal lumen width was also increased by the combined treatment compared with estrogen alone (Fig. 5B).
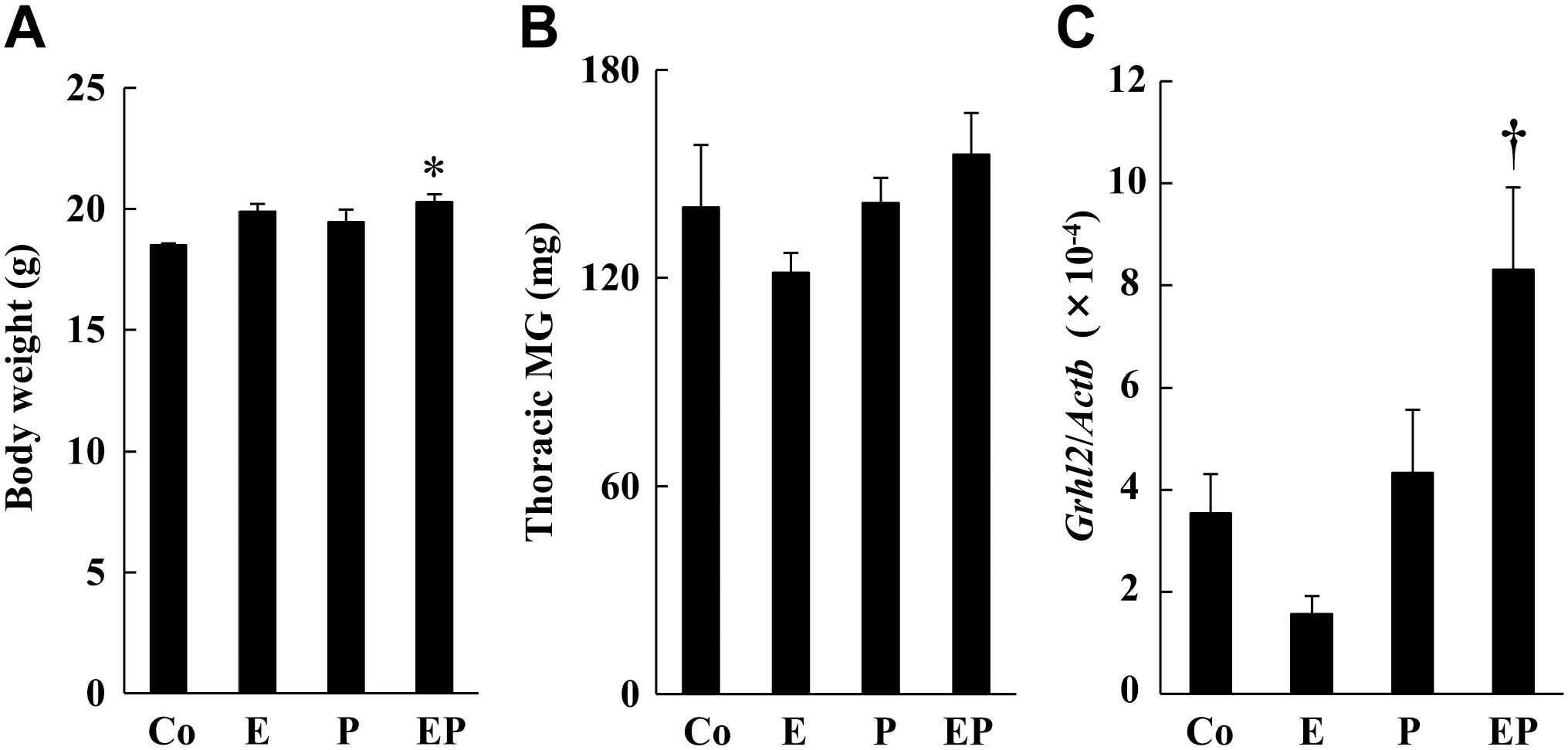
The effect of ovarian steroid on body weight, thoracic mammary gland weight, and Grhl2 mRNA expression in the mammary glands of ovariectomized mice. Ovariectomized mice at 6 weeks of age received single daily subcutaneous injections of corn oil as a control solvent (Co, n=4), estrogen (E, n=4), progesterone (P, n=5), or both estrogen and progesterone (EP, n=5) for eight consecutive days. Mice were sacrificed at 24 hr after the last injection. Body weight (A) and the thoracic mammary gland weight (B) were measured. Grhl2 mRNA expression in the abdominal mammary gland was quantified by real-time PCR (C). *p<0.05 vs control. †p<0.05 vs E injection.
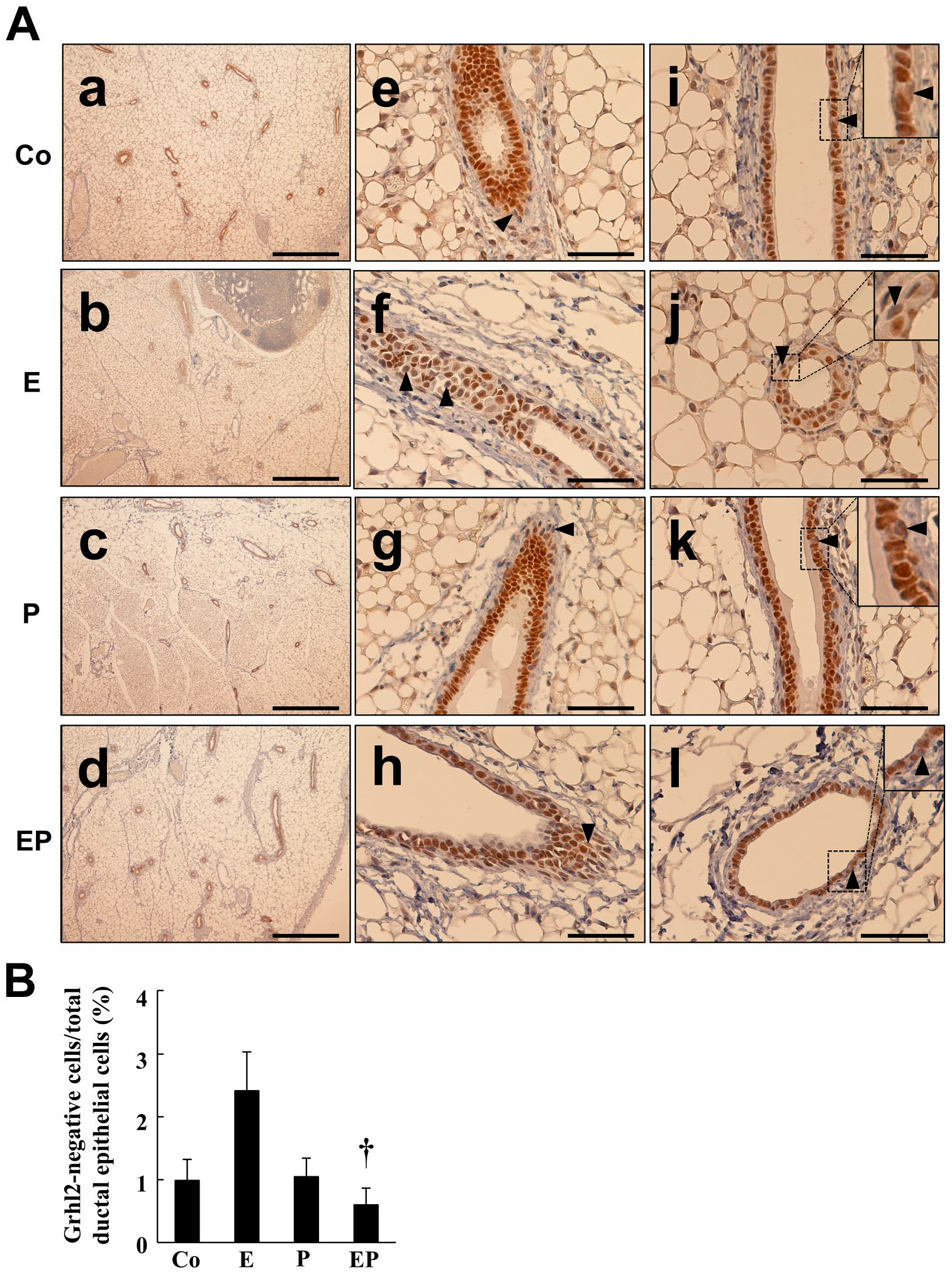
The effect of ovarian steroid on Grhl2 protein expression in the mammary glands of ovariectomized mice. Mice were treated as described in the Fig. 3 legend. In panel A, the first row (a, e, i) is corn oil (C, n=4); the second row (b, f, j) is estrogen (E, n=4); the third row (c, g, k) is progesterone (P, n=5); and the fourth row (d, h, l) is both estrogen and progesterone (EP, n=5). Arrowheads indicate the luminal epithelium not stained by the antibody. In i to l, the insert shows magnified images of the boxed region. In (B), the Grhl2-negative mammary epithelial cell percentage per total ductalepithelial cells was calculated by counting the cell number in 19 ducts of four to five mice in each experimental group. †p<0.05 vs E injection. The scale bar in a to d indicates 500 µm, whereas that in e to l is 50 µm.
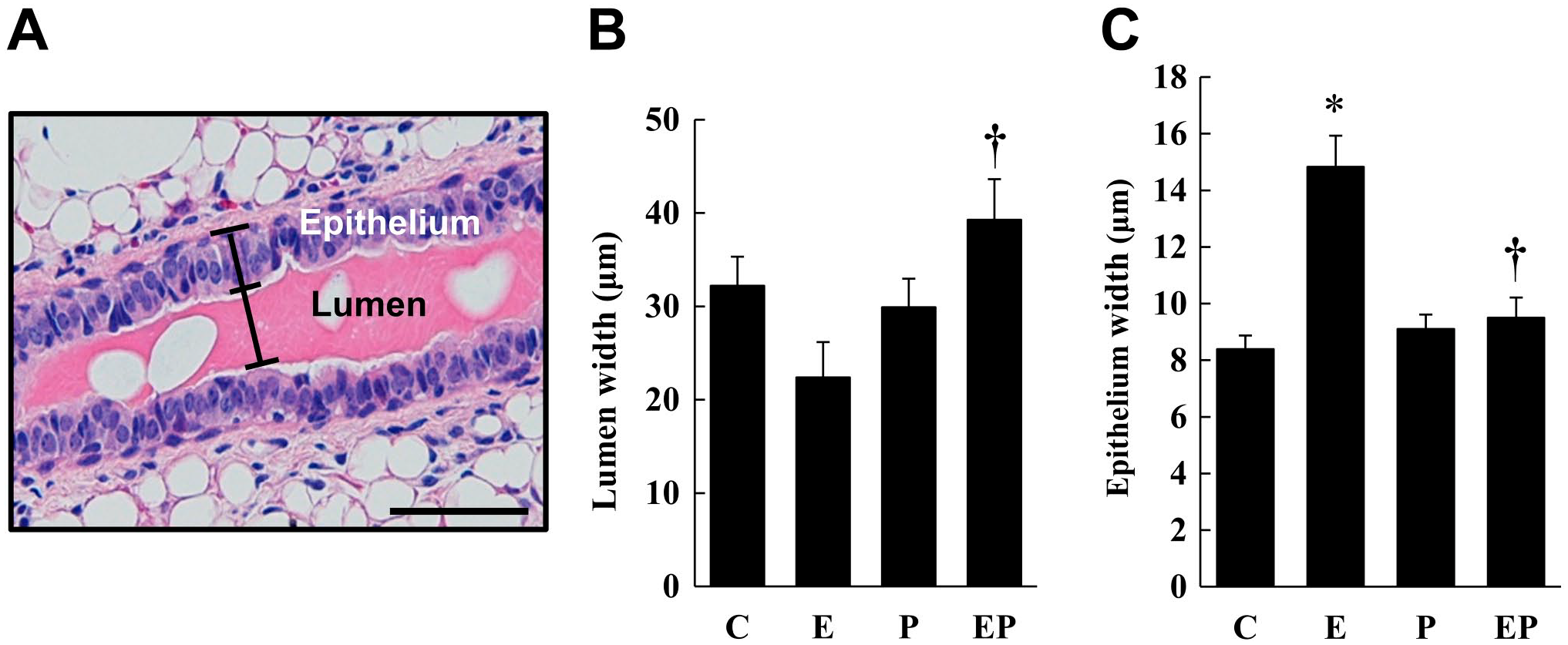
The effect of ovarian steroid on the ductal epithelial layer and ductal lumen width in the mammary glands of ovariectomized mice. Ovariectomized mice were treated with corn oil (Co), estrogen (E), progesterone (P), or both estrogen and progesterone (EP). H&E-stained mammary gland sections were prepared. The epithelial layer and lumen widths of 24 ducts from three mice in each group were measured as shown in (A). The lumen width and epithelial width (height) averages are summarized in (B) and (C), respectively. *p<0.05 vs Control (Co). †p<0.05 vs E injection. The scale bar indicates 50 μm.
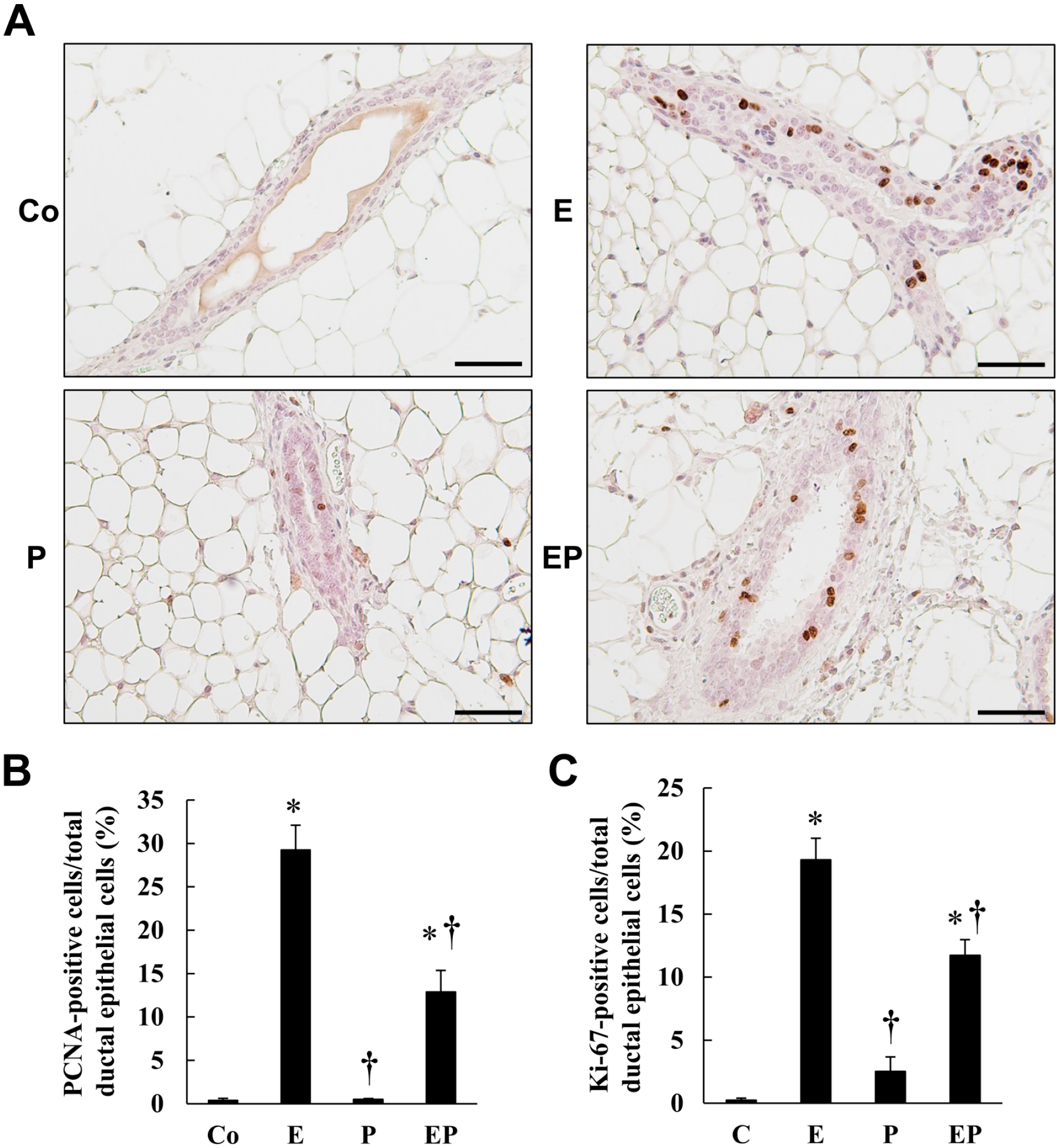
The effect of ovarian steroid on ductal epithelial cell proliferation in the mammary glands of ovariectomized mice. Ovariectomized mice were treated with corn oil (Co), estrogen (E), progesterone (P), or both (EP), as described in the Fig. 3 legend. PCNA and Ki-67 expression, cell proliferation markers, was determined by immunohistochemistry. Typical Ki-67 staining of ductal cells is shown in (A). The ratio of PCNA-positive (B) and Ki-67-positive (C) mammary epithelial cell to total ductal epithelial cells was calculated by the number of epithelial cells of 20–24 ducts from three mice for PCNA and 30 ducts from three mice for Ki-67. *p<0.05 vs Control (Co), †p<0.05 vs E injection. The scale bar indicates 50 µm. Abbreviation: PCNA, proliferating cell nuclear antigen.
To reveal the relationship between Grhl2 expression and gene expression of cell adhesion molecules in mammary glands, Cldn1, Cldn2, Cldn3, Cldn4, Cldn7, Cldn9, and E-cadherin gene expression was examined. Among these, the expression of Cldn2 and Cldn9 was relatively lower than the other Cldn tested. As Cldn2 mRNA levels in almost half the samples were less than the detection limit (data not shown), we excluded data of Cldn2 from the analysis. The expression of Cldn3, Cldn4, Cldn7, and E-cadherin genes increased age-dependently like Grhl2 expression, whereas the expression of Cldn1 and Cldn9 decreased with age (Figs. 1C and 7). In addition, the expression of Cldn family genes and E-cadherin was examined in ovariectomized mice. The expression of Cldn1 was significantly decreased by either estrogen or progesterone treatments, and similar decrease was observed by EP combined treatment (Fig. 8A). The other Cldn expression was not influenced by estrogen and progesterone alone, although EP combined treatment increased Cldn3, Cldn7, and Cldn9 expression significantly (Fig. 8B, D, and E). A similar trend was found with Cldn4 and E-cadherin expression (Fig. 8C and F). Furthermore, high positive correlations between Grhl2 mRNA expression and respective mRNA expression of Cldn3, Cldn4, Cldn7, and E-cadherin genes were observed, although the expression of Cldn1 and Cldn9 did not correlate with the expression of Grhl2 (Fig. 9 and Table 2). When the data of postnatal development and of steroid treatments in Cldn4, Cldn7, and E-cadherin separately were analyzed, the slopes of regression lines were almost identical between these two data. However, in the case of Cldn3, the steroid treatment has steep slope of linear regression, compared with postnatal development.
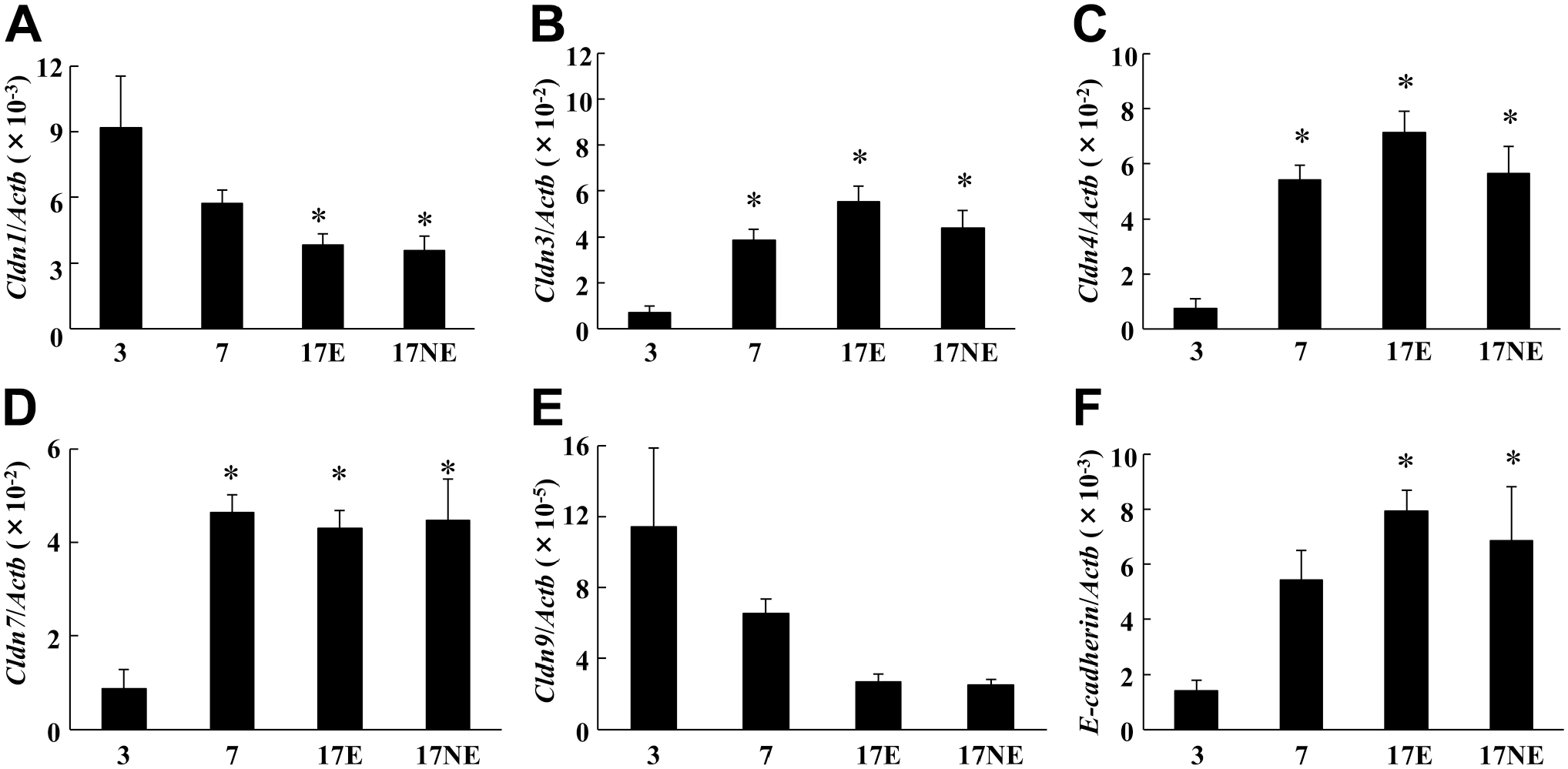
Claudin (Cldn) family and E-cadherin mRNA expression of mouse mammary glands during postnatal development. Cldn1 (A), Cldn3 (B), Cldn4 (C), Cldn7 (D), Cldn9 (E), and E-cadherin (F) mRNA expression was determined by real-time PCR on the same samples prepared in Fig. 1C (n=6 each). *p<0.05 vs 3 weeks of age.
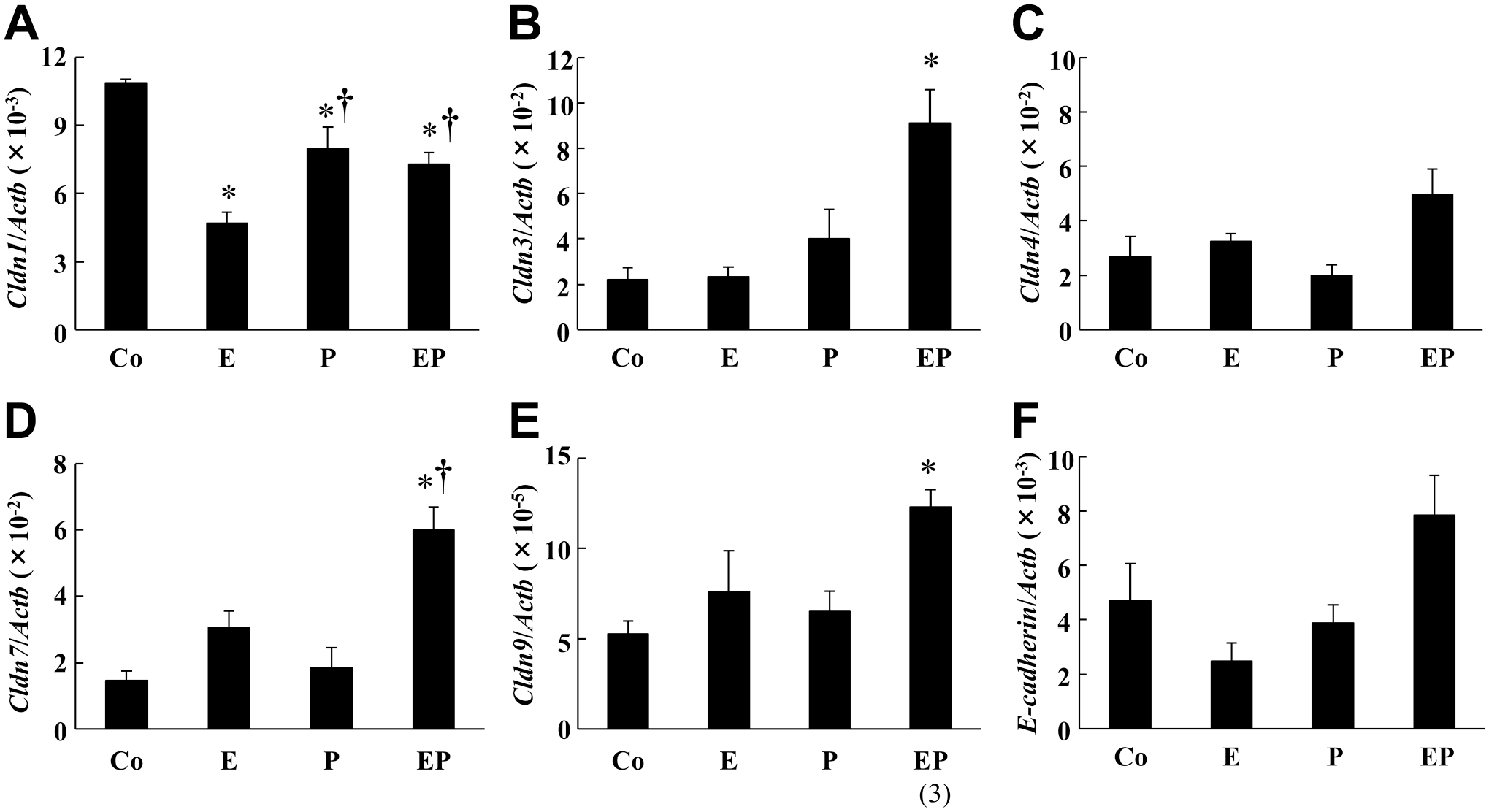
The effect of ovarian steroid on ovariectomized mice mammary gland, and Cldn family and E-cadherin mRNA expression. Cldn1 (A), Cldn3 (B), Cldn4 (C), Cldn7 (D), Cldn9 (E), and E-cadherin (F) mRNA expression was determined by real-time PCR on the same samples prepared in Fig. 3C (n=4 or 5). *p<0.05 vs Co, †p<0.05 vs E injection. Abbreviations: Cldn, claudin; Co, Control; E, estrogen; P, progesterone; EP, estrogen and progesterone.

Correlation between Grhl2 mRNA expression and mRNA expression of the cell adhesion molecules. Correlation between Grhl2 mRNA expression and Cldn1 (A), Cldn3 (B), Cldn4 (C), Cldn7 (D), Cldn9 (E), and E-cadherin (F) mRNA expression was analyzed. The closed circle indicates data from the postnatal development with a solid regression line (n=24). The cross symbol indicates data from the steroid treatment with a dotted regression line (n=16 or18). The respective correlation coefficients are summarized in Table 2. Abbreviation: Cldn, claudin.
Correlation Between Grhl2 mRNA Expression and mRNA Expression of Cell Adhesion Molecules.
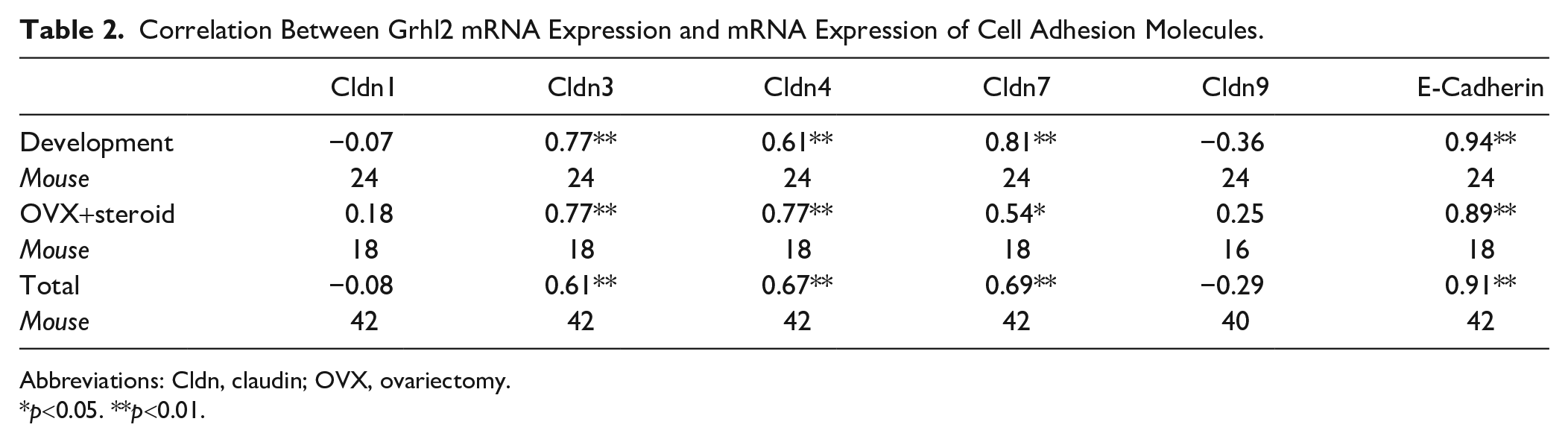
Abbreviations: Cldn, claudin; OVX, ovariectomy.
p<0.05. **p<0.01.
Finally, double staining was performed to confirm colocalization of Grhl2 and E-cadherin in the mouse mammary gland. As expected, mammary ductal epithelial cells expressed both E-cadherin and Grhl2 (Fig. 10). Moreover, Cldn3 signal was present on the plasma membrane of ductal cells where E-cadherin was expressed (Fig. 11). Cldn4 signal was found in the mammary duct at 4–8 weeks of age, but not in 15 weeks of age (Fig. 12).
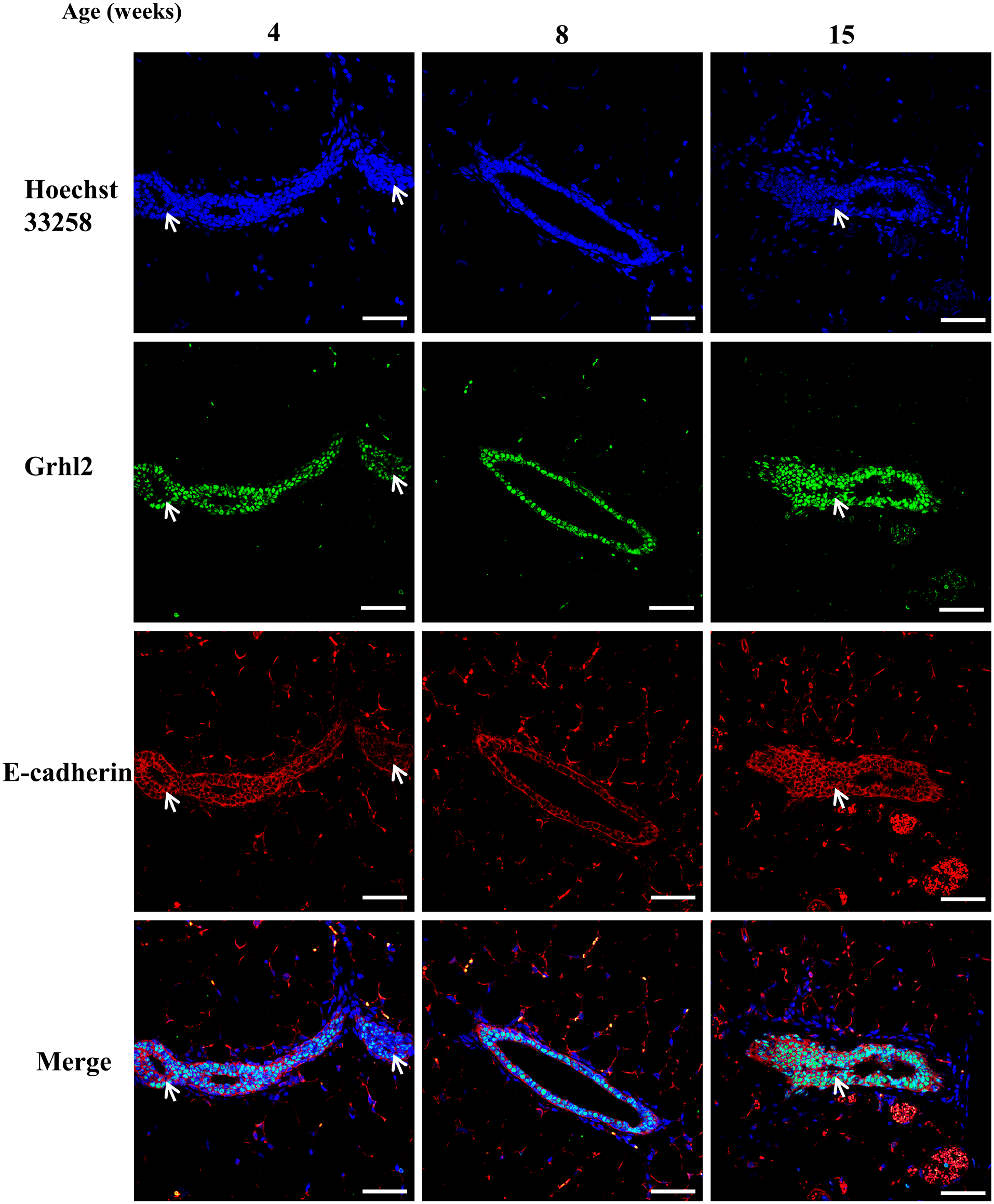
Expression of Grhl2 and E-cadherin protein during postnatal development in normal mice. Mice were sacrificed at 4, 8, and 15 weeks (n=2 each). Specimens were stained with both anti-Grhl2 (green) and anti-E-cadherin (red). Arrows indicate the Grhl2-negative cells. The scale bar indicates 50 µm.
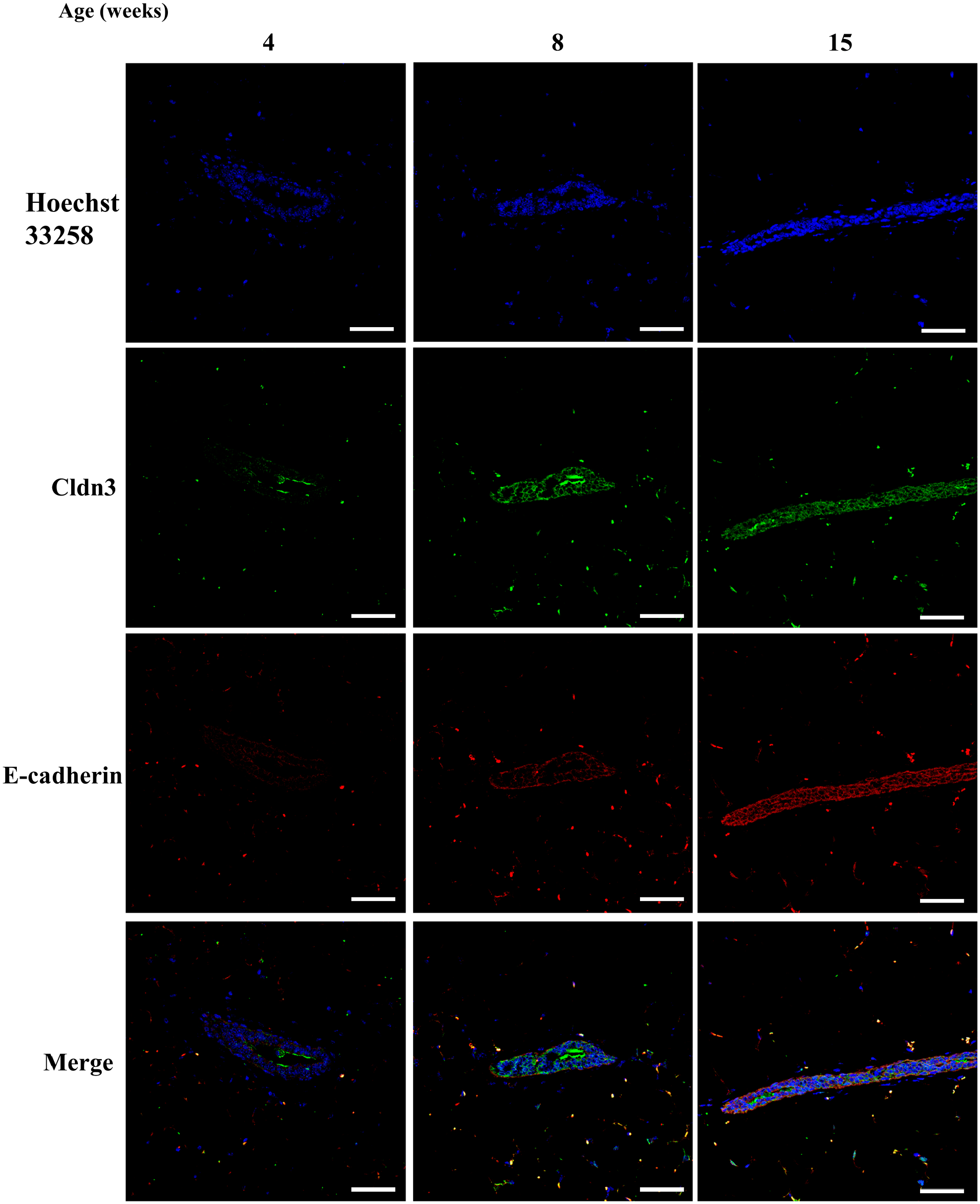
Expression of claudin 3 (cldn) protein along with E-cadherin during postnatal development in normal mice. Mice were sacrificed at 4, 8, and 15 weeks (n=2 each). Specimens were stained with anti-cldn3 (green), along with E-cadherin (red). The scale bar indicates 50 µm.
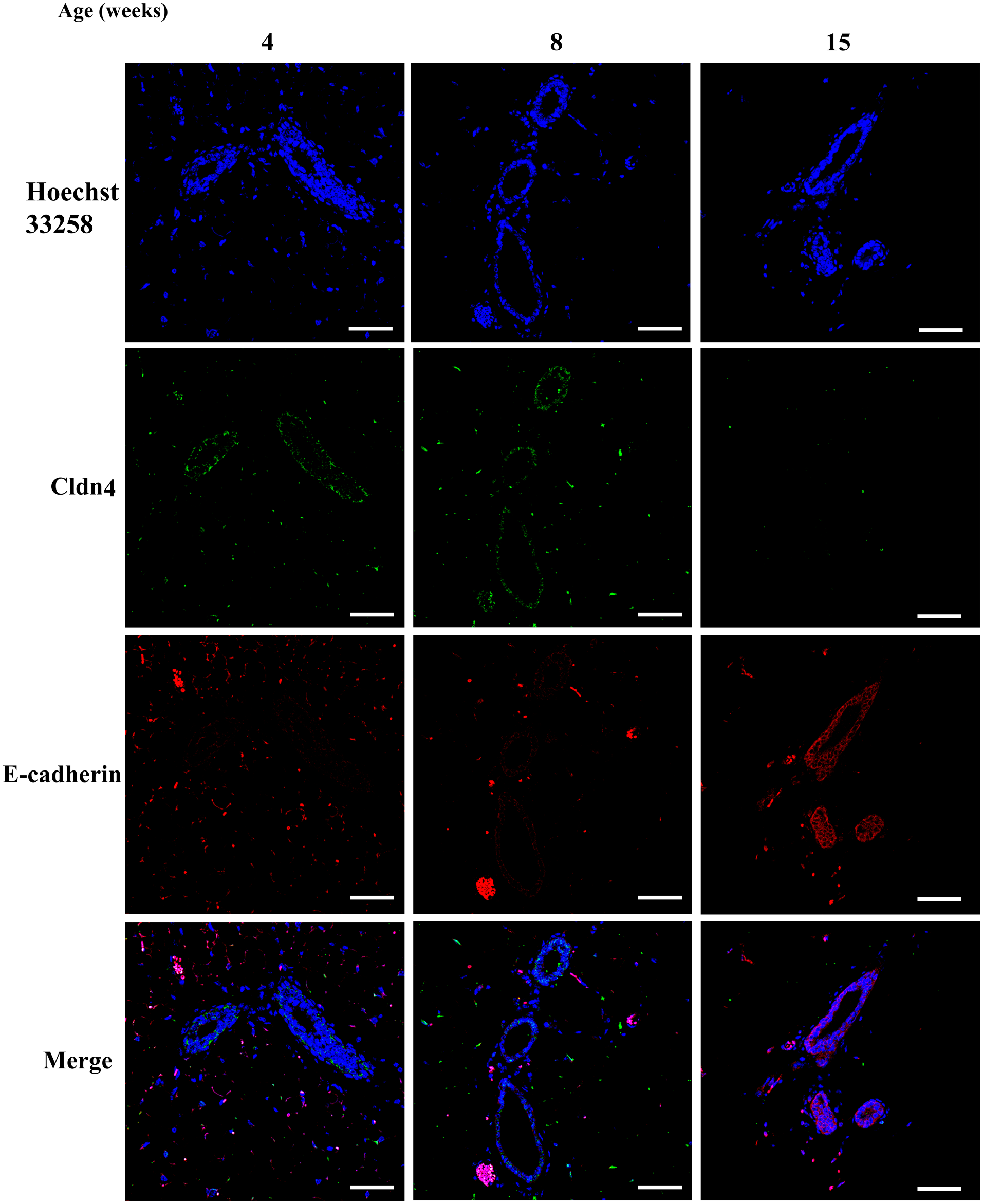
Expression of claudin 4 (cldn4) protein along with E-cadherin during postnatal development in normal mice. Mice were sacrificed at 4, 8, and 15 weeks (n=2 each). Specimens were stained with anti-cldn4 (green), along with E-cadherin (red). The scale bar indicates 50 µm.
Discussion
In this study, we have demonstrated that Grhl2 mRNA and protein expression in mouse mammary ductal epithelium is detected as early as 3 weeks of age, but signals in one-fourth of the ductal epithelium are negative. Furthermore, Grhl2 mRNA and protein expression increased at puberty and thereafter, suggesting that increased estrogen after the onset of puberty is involved in regulating Grhl2 gene expression. Although normal and tumor ER signals are distinct, Grhl2 acts as an ER-cooperating tumorigenic transcription factor. 18 Phosphorylated ER sites, unlike ER sites, are enriched in Grhl2 DNA-binding motifs, and estrogen treatment increases Grhl2 recruitment to former sites in MCF-7 cells. 20 However, estrogen treatment alone tended to decrease Grhl2 and its target gene expression in ovariectomized mice, indicating that estrogen is unlikely to induce Grhl2 transcription and activation in the normal ductal epithelial cells.
Estrogen caused cell proliferation, increased luminal cell width, and increased the Grhl2-negative cell number. They existed in the duct luminal region and the TEB front region. In the former, Grhl2-negative cells look like normal cells just after asymmetric division, which lose their polarity as described by Huebner and Ewald. 2 In addition, TEB is composed of internal low-polarity cells having a loose cell–cell contact without TJs. 2 Therefore, it is suggested that Grhl2 expression is inversely regulated to estrogen-dependent cell proliferation during ductal development.
It is reported that progesterone receptor signals are critical for mature mammary gland side branching and lobuloalveolar development.21,22 However, treatment of progesterone alone in ovariectomized mice did not alter any parameters tested, except decrease in Cldn1 expression. The combined treatment of EP increased Grhl2 expression when compared with estrogen treatment. This supports the view that estrogen signals in cooperation with progesterone signals govern mammary gland development in adult mice. 19 This increase in Grhl2 was accompanied by enhanced Cldn3 and Cldn7 expression and ductal lumen width enlargement. As the steroid doses used were expected to mimic physiological circulating concentrations of pregnant mice, 23 a duct with a functional TJ might be formed by these hormones, as was the case for late pregnancy and early lactational stages.
Grhl2 expression was proportionally expressed with Cldn3, Cldn4, Cldn7, and Cdh1 (E-cadherin). Cldn4 and E-cadherin are direct targets of the Grhl2 transcription factor12,13 and the Grhl2 gene is the most tightly correlated with E-cadherin expression in some cancer cells. 24 We also observed an extremely high correlation between Grhl2 and E-cadherin mRNA expression. As seen in Fig. 10, many of ductal epithelium expressing Grhl2 expressed E-cadherin, resulting in solid duct formation. The correlation analysis also revealed that Cldn3 expression was enhanced by the EP combination compared with normal ductal development, whereas Cldn4 and Cldn7 expression was regulated in a uniform way like E-cadherin. These results support the findings that each Cldn expression is regulated independently through mammary gland development, 9 although difference in the mechanism of postnatal and steroid-induced Grhl2-dependent Cldn3 expression needs to be clarified.
In summary, we have found that the EMT suppressor Grhl2 is expressed in the nuclei of luminal epithelium and myoepithelium as early as the onset of puberty. This causes the expression of EMT suppressor molecules, such as E-cadherin, Cldn3, Cldn4, and Cldn7. Steroid replacement studies in ovariectomized mice suggest that estrogen indirectly suppresses Grhl2 expression and increases cell proliferation. These data suggest that Grhl2 non-stained cells in the mammary duct in adult mice might express low levels of EMT suppressors and might be vulnerable to internal and external stimuli, leading to side branching and lobuloalveolar development after pregnancy or during tumorigenesis.
Footnotes
Competing Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
KK designed experiments. SM, HS, CK, and MK-T performed experiments. AK and YO-O provided experimental support and intellectual input. KK and SM wrote the manuscript. All authors have read and approved the final manuscript.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by KAKENHI grant (19H03116 to K.K.).
