Abstract
Dentin sialophosphoprotein (DSPP), which expresses and synthesizes in odontoblasts of dental pulp, is a critical protein for normal teeth mineralization. Originally, DSPP was identified as a dentin-specific protein. In 2010, DSPP was also found in femoral head cartilage, and it is still unclear what roles DSPP play in femoral head cartilage formation, growth, and maintenance. To reveal biological functions of DSPP in the femoral head cartilage, we examined Dspp null mice compared with wild-type (WT) mice to observe DSPP expression as well as localization in WT mice and to uncover differences of femoral head cartilage, bone morphology, and structure between these two kinds of mice. Expression data demonstrated that DSPP had heterogeneous fragments, expressed in each layer of femoral head cartilage and subchondral bone of WT mice. Dspp null mice exhibited a significant reduction in the thickness of femoral head cartilage, with decreases in the amount of proliferating cartilage cells and increases in apoptotic cells. In addition, the subchondral bone mineralization decreased, and the expressions of vessel markers (vascular endothelial growth factor [VEGF] and CD31), osteoblast markers (Osterix and dentin matrix protein 1 [DMP1]), osteocyte marker (sclerostin [SOST]), and osteoclast marker (tartrate-resistant acid phosphatase [TRAP]) were remarkably altered. These indicate that DSPP deletion can affect the proliferation of cartilage cells in the femoral head cartilage and endochondral ossification in subchondral bone. Our data clearly demonstrate that DSPP plays essential roles in the femoral head cartilage growth and maintenance and subchondral biomineralization.
Introduction
Cartilage constitutes a single cell type, the chondrocyte, with an abundant extracellular matrix (ECM).1,2 Interestingly, chondrocytes synthesize and maintain healthy ECM. 3 The ECM plays a critical role in preservation of cartilage tissue integrity. 4 Many human skeletal disorders result from mutations in cartilage ECM components that compromise both ECM architecture and chondrocyte function. 2 In addition, many inherited human chondrodysplasias involve disruption of cartilage matrix assembly or cell–matrix interactions, resulting in abnormal skeletal development or early onset of cartilage degeneration in some cases. 5
Collagen types II, IX, and XI are the major structural constituents in the cartilage ECM fibrils.6–9 Recently, proteoglycan–glycosaminoglycan networks of aggrecan, hyaluronan, and some non-collagen protein such as Small Integrin-binding Ligand N-linked Glycoprotein (SIBLING) family are also found in cartilage.6–9 Dentin sialophosphoprotein (DSPP) is a precursor protein and a member of SIBLING family of proteins, which includes osteopontin (OPN), bone sialoprotein (BSP), dentin matrix protein 1 (DMP1), and matrix extracellular phosphoglycoprotein (MEPE). 10 The DSPP is produced by odontoblast of dental pulp inside the teeth. Its coding gene, Dspp, was discovered using a mouse odontoblast cDNA library in 1997. During the tooth development, the precursor DSPP can be cleaved into three different proteins, dentin sialoprotein (DSP), dentin glycoprotein (DGP), and dentin phosphoprotein (DPP). 11 Interestingly, DPP and DSP were found much early than DSPP. DPP was discovered in 1967 12 while DSP was discovered in 1981. 13 Then, it was recognized that the cleavage fragments, DPP and DSP, are the main forms in the dentin while full-length DSPP is hardly detected in general. 7 Therefore, DSPP, DSP, and DPP have been initially recognized to be unique for dentine formation during tooth development.
Actually, DSPP, DSP, and DPP can express in bone, salivary glands, cartilage, liver, lung, kidney, brain, cementum, alveolar bone, and so on.6,7,14–16 The DSPP plays an important role in biomineralization of dentinogenesis, osteogenesis, and cementogenesis.17–20 We, first, reported that DSPP plays an essential role in postnatal development and maintenance of mouse mandibular condylar cartilage in 2013. 21 Although DSPP exists in different categories of cartilage, the exact physical function of DSPP in other cartilage is still unclear.6,7,22 Comparisons of gene expression during the development of other joints with the temporomandibular joint (TMJ) reveal that there were some similarities between the hip joint and TMJ. 23 As an essential member of hip joint, femoral head consists of femoral head cartilage and subchondral bone. Differing from mandibular condylar fibrocartilage, femoral head cartilage is a hyaline cartilage. Histologically, the femoral head cartilage is permanent and provides articulating surfaces of hip joint, whereas the growth plate is transient and responsible for endochondral bone growth.
To understand DSPP functions in femoral head, we examined DSPP distribution and DSPP fragment pattern in wild-type (WT) mice compared with Dspp null mice, and evaluated DSPP functions during the development of femoral head cartilage and subchondral bone. Our data showed that the proliferation of cartilage cells in femoral head cartilage was abated and endochondral ossification was affected, leading to defection in each layer of femoral head cartilage and its subchondral bone.
Materials and Methods
Extraction of Non-collagenous Proteins (NCPs) From Femoral Head Cartilage
Dspp knockout mice were generated in National Institute of Dental and Craniofacial Research (NIDCR), National Institutes of Health (NIH), USA. 18 Animal experiments in this study were approved by the Institutional Animal Care and Use Committee of the Fourth Military Medical University. Three 2-month-old Dspp null mice and WT mice were used for the extraction of NCPs. The femoral head cartilage of Dspp null mice and WT mice was carefully separated at the cartilage bone interface under a dissecting microscope and was placed in 6 M urea/1 M NaCl solution plus proteinase inhibitors without EDTA (ethylenediaminetetraacetic acid) for 48 hr. Incisor and remnant femur NCPs from WT mice were extracted as previously described. 24 All NCPs extracts were analyzed by Western blot to evaluate DSPP.
Assay of Western Blot
Briefly, 10 μl of protein from each sample was used to run SDS-PAGE gels, and then transferred to polyvinylidene fluoride (PVDF) membrane. Blot was blocked, incubated with primary anti-DSP MAb (1:2000 dilution, anti-DSP-2G7.3, a gift from Dr. Chunlin Qin, Department of Biomedical Sciences and Center for Craniofacial Research and Diagnosis, Texas A&M University College of Dentistry, Dallas, TX). 25 Blot was washed three times with PBS containing 0.3% of tween 20, followed by incubation with secondary alkaline phosphatase-conjugated anti-mouse IgG (1:5000 dilution, Sigma Aldrich; St. Louis, MO), and then incubated with chemiluminescent substrate CDP-Star (Ambion; Austin, TX) for 5 min and exposed to X-ray films.
X-ray Radiography Imaging
Twenty-four mice (2, 3, and 6 months old) were used. Femoral head from Dspp null mice and WT mice was dissected and analyzed with the Faxitron MX-20DC12 Specimen Radiography System (Faxitron X-ray Corp.; Buffalo Grove, IL).
Micro-computed Tomography (μCT) Analyses
Femurs from 12-month-old mice were dissected, fixed with 4% paraformaldehyde for 48 hr, and then transferred into 70% ethanol and scanned with μCT50 (Scanco Medical AG; Bassersdorf, Switzerland). Following parameters were used during scanning: voxel size 12 μm, 200 mA, 70 kVp, and exposure time 300 ms. After reconstructed, data were analyzed with the 3D-Creator software supplied with the instrument. Morphometric parameters such as bone volume (BV), tissue volume (TV), bone volume fraction (BV/TV), porosity (1 – BV/TV), bone mineral density (BMD), and tissue mineral density (TMD) were calculated.
Histopathological Examinations
Under anesthesia, mice were perfused, through ascending aorta, with 4% paraformaldehyde in 0.1 M phosphate buffer. Femoral head was dissected and fixed in 4% paraformaldehyde overnight, and then decalcified in 8% EDTA (pH 7.4) at 4C for 2 weeks. Afterward, tissues were embedded in paraffin and cut serial 5 μm sections for hematoxylin & eosin (H&E), toluidine blue, tartrate-resistant acid phosphatase (TRAP), or immunohistochemical (IHC) staining. For IHC staining, samples were stained with primary rat anti-DSP polyclonal antibody (used at a dilution of 1:500) and rabbit anti-DMP1-N 859 polyclonal antibody (used at a dilution of 1:500) (a gift from Dr. Chunlin Qin, Department of Biomedical Sciences and Center for Craniofacial Research and Diagnosis, Texas A&M University College of Dentistry, Dallas, TX), 14 primary goat anti-biglycan polyclonal antibody (1:1000 dilution, a gift from Dr. Larry Fisher, Craniofacial and Skeletal Diseases Branch, NIDCR, NIH, Bethesda, MD), primary rabbit anti-CD31 monoclonal antibody (1:100 dilution, Cell Signaling Technology, Beverly, MA), primary rabbit anti-Osterix monoclonal antibody (1:1000 dilution), and rabbit anti-sclerostin polyclonal antibody (1:100 dilution, Abcam, Cambridge, MA) following color development with ABC kit, M.O.M. kit, or DAB kit (Vector Laboratories; Burlingame, CA) with methyl green for counterstaining according to manufacturer’s manuals.
Bromodeoxyuridine (BrdU) Cell Proliferation Assay
The BrdU (0.1 ml/kg, Life Technologies; Carlsbad, CA) was intraperitoneally administrated to 1-month-old Dspp null mice and WT mice. Two hours after injection, mice were sacrificed, femoral head was dissected and processed to embedded paraffin, and 5 µm section was cut and stained with Zymed BrdU staining kit (Life Technologies). BrdU-positive cells were counted from four serial sections of each mouse (four mice/group). Data were analyzed statistically and displayed as the average number of positive cells/mouse ± SD.
Terminal Deoxynucleotidyl Transferase–mediated dUTP Nick-end Labeling (TUNEL) Staining
The TUNEL staining was performed on paraffin sections from 1-month-old Dspp null mice and WT mice using TUNEL Apoptosis Assay Kit-FITC (7sae biotech; Shanghai, China). TUNEL-positive chondrocytes, labeled in orange, in femoral head cartilage were counted from four serial sections of each mouse (three mice/group). Data were analyzed statistically and displayed as the average number of positive cells/mouse ± SD.
Statistical Analysis
For Dspp KO and WT mice comparisons, data were analyzed with Student’s t-tests. Results are presented as the mean ± SD. The p value of <0.05 was considered statistically significant.
Results
DSPP Expression Profiles in the Femoral Head Cartilage of Mice
The presence of DSPP in tissues is usually reflected by anti-DSP Western immunoblotting or anti-DSP IHC staining because there is no available anti-DPP antibody. Data of Western blot demonstrated that the anti-DSP-2G7.3 antibody used herein could detect 53 kDa band, 70 kDa band, and two <40 kDa weaker bands (indicated in Fig. 1 by white arrowheads) in the femoral head cartilage (Fig. 1, Lane 2), which is similar to previous data. 6 Bands detected from subchondral bone were basically consistent with, but much weaker, bands from the femoral head cartilage, which indicated that the concentration of DSP in cartilage is higher than that in subchondral bone (Fig. 1, Lanes 2 and 3). The 78 kDa band from teeth (Fig. 1, Lane 5), which was considered to be 98 kDa protein devoid of any carbohydrate substituents,8,25 was not detected in the femoral head cartilage and subchondral bone of the femoral head. We, however, could also detect the 78 and 195 kDa band if we increased the loading amount of NCP extracts, which looked like the full length of DSPP from femoral head cartilage (data not shown). These suggest that DSPP is a heterogeneous protein.
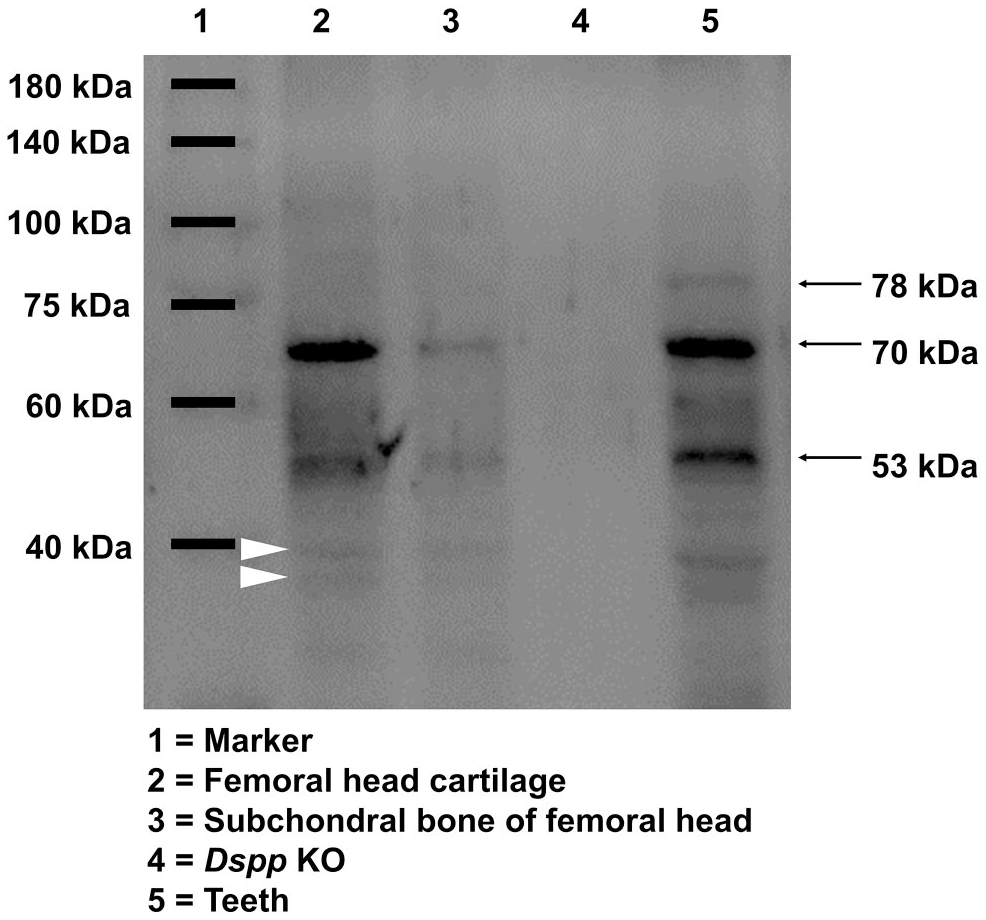
Western blot assay of DSP protein with anti-DSP-2G7.3 antibodies. Lane 1 is protein marker. Lane 2 is proteins extracted from femoral head cartilage of WT mice. Two major bands at 70 and 53 kDa, respectively, were recognized by the anti-DSP antibodies. Lane 3 is proteins extracted from subchondral bone of femoral head of WT mice. The bands recognized by anti-DSP antibody are similar to Lane 2, but much weaker. Lane 4 is proteins extracted from femoral head cartilage of Dspp null mice, used as negative control. Note that there was no DSP band. Lane 5 is proteins extracted from incisors of WT mice, used as positive control. Three major bands at 78, 70, and 53 kDa, respectively, were indicated by black arrows. Abbreviations: DSP, dentin sialoprotein; Dspp, dentin sialophosphoprotein; KO, knockout; WT, wild type.
IHC staining found that DSPP existed in the cells of all layers of femoral head cartilage of WT mice at 2 months, and also found in the growth plate and ossification area of WT mice (Fig. 2B and D). These findings indicate that DSPP expresses in both cartilage and bone, which is consistent with Western blot data. Dspp null mice at 2 months, however, liked a negative control and had no DSPP positive signal in femoral cartilage (Fig. 2A and C). We also observed a weak DSP positive signal in subchondral bone and secondary ossification center at 6 months, but hardly saw DSP immunoactivity in cartilage at the same time (Fig. 2F and H), indicating that the expression of DSPP in femoral head decreases with increasing age.
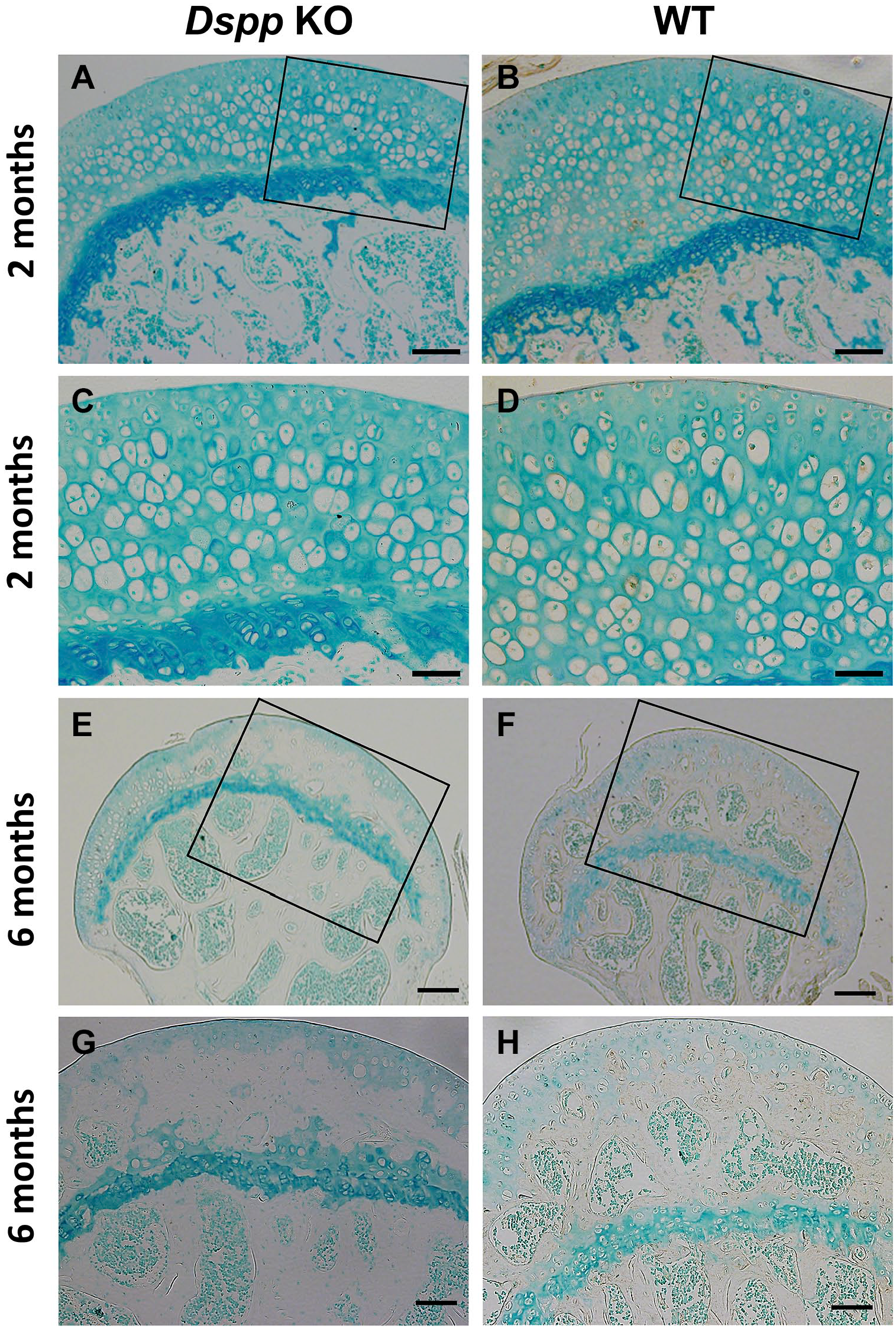
Immunohistochemical analyses of DSPP for femoral head cartilage using the polyclonal anti-DSP antibody. (A) Femoral head cartilage from Dspp null mice, 2 months old. Scale bars = 100 µm. (B) Femoral head cartilage from WT mice, 2 months old. Positive DSPP staining was observed in articular cartilage as well as subchondral bone matrix. Scale bars = 100 µm. (C) Higher magnification image for part of Fig. 2A. Scale bars = 50 µm. (D) Higher magnification image for part of Fig. 2B. Positive DSPP staining was mainly observed in the cytoplasm of cartilage cells while very weak positive DSPP staining was seen in the matrix of femoral head cartilage. Scale bars = 50 µm. (E) Femoral head cartilage from Dspp null mice, 6 months old. Scale bars = 200 µm. (F) Femoral head cartilage from WT mice, 6 months old. Scale bars = 200 µm. (G) Higher magnification image for part of Fig. 2E. Scale bars = 200 µm. (H) Higher magnification image for part of Fig. 2F. Scale bars = 200 µm. Noted that very weak positive DSPP signals were observed in subchondral bone matrix, but seldom be seen in cartilage (n=3). Methyl green was used for counterstaining. Abbreviations: Dspp, dentin sialophosphoprotein; DSP, dentin sialoprotein; KO, knockout; WT, wild type.
Effects of Dspp Deletion on Femoral Cartilage
Normal femoral head articular cartilage has three layers, superficial layer (S), middle layer (M), and deep layer (D), according to the orientation and the morphology of cells. 8 In general morphology, H&E staining and toluidine blue Staining revealed that femoral head of Dspp null mice exhibited no apparent abnormality compared with WT mice (Fig. 3). At 2 and 3 months, the articular cartilage was directly attached to the growth plate of both Dspp null mice and WT mice, whereas the articular cartilage of Dspp null mice became much thinner than that of WT mice (Fig. 3A and B). At 6 months, the articular cartilage and the growth plate were separated completely by the secondary ossification center, which is much thinner in Dspp null mice than that in WT mice (Fig. 3A and B). These indicate that the secondary ossification process is completed by 6 months in WT mice and Dspp null mice without difference, except thinner articular cartilage. Toluidine blue staining further confirmed these observations about the femoral head cartilage at 2-, 3-, and 6-month time points from WT and Dspp null mice (Fig. 3B). Especially from 6-month-old mice, the secondary ossification center was much thinner in the Dspp null mice than that in WT mice (Fig. 3A and B). One interesting observation, however, was that superficial layer and deep layer were much thicker in the Dspp null mice than that in WT mice (Fig. 3C–E). These suggest that there is a cartilage calcification defect in Dspp knockout mice at 6 months.
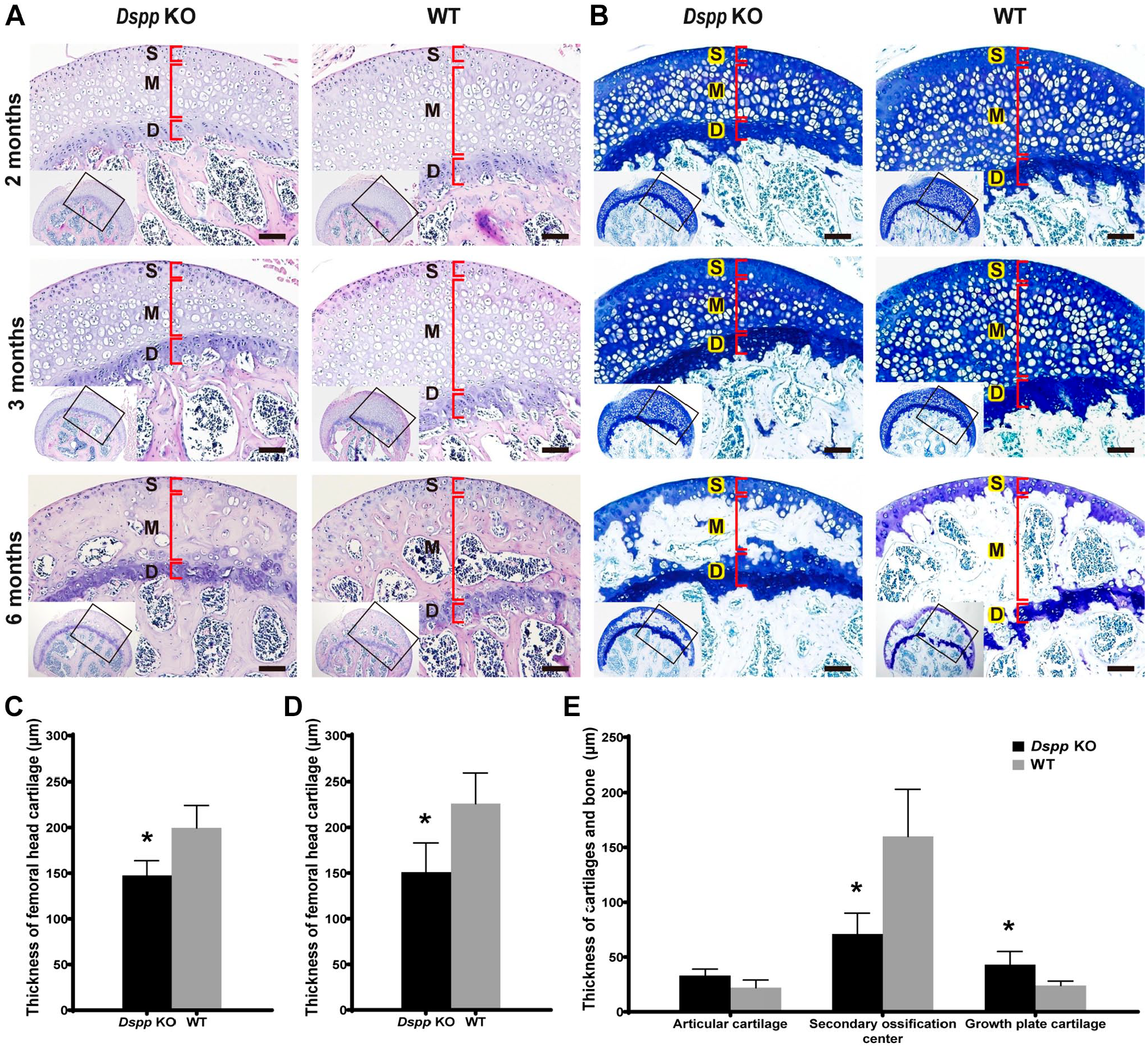
Femoral head stained with hematoxylin & eosin (H&E) and toluidine blue. (A) Images of H&E staining from Dspp null mice and WT mice at 2, 3, and 6 months. (B) Toluidine blue staining from Dspp null mice and WT mice at 2, 3, and 6 months. DSPP deletion decreased cartilage thickness in Dspp null mice compared with WT mice. Growth plate was wider in Dspp null mice than that in WT mice at 6 months. (C–E) Quantitative analyses of thickness of the femoral head cartilage at 2, 3, and 6 months, respectively. Data showed that the femoral head cartilage was thicker in WT mice than that in Dspp null mice. Data represent mean ± SD (n=5). Scale bars = 100 µm. Abbreviations: D, deep layer; Dspp, dentin sialophosphoprotein; KO, knockout; M, middle layer; S, superficial layer; WT, wild type. *p<0.05.
Importantly, as shown in plain X-ray, femoral head cartilage was homogenous gray, but not radiolucent (Fig. 4). H&E staining and toluidine blue analysis also confirmed that the homogenous gray areas are cartilage (Figs. 3 and 4). Phenotypic changes in the cartilage of femoral head of the Dspp null mice, however, were apparent by histological staining, but were not readily identified by plain X-ray analyses.
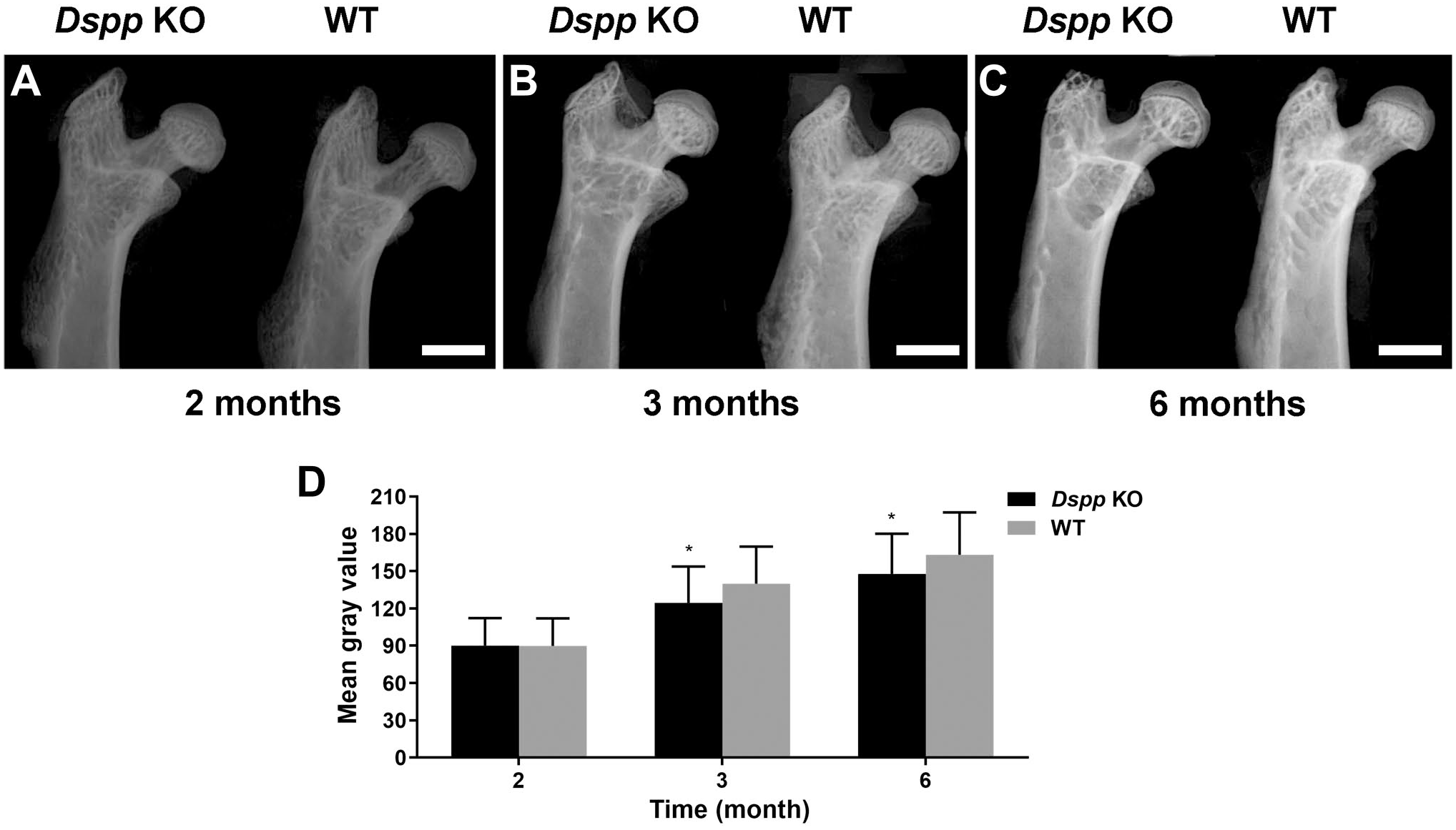
Plain X-ray analyses of femoral heads. (A–C) Plain X-ray radiographs of femoral head from Dspp null (Dspp KO) and WT mice at the ages of 2, 3, and 6 months. (D) Bar graphs for Fig. 4A to C. Data represent mean ± SD (n=4). Scale bars = 1.0 mm. Abbreviations: Dspp, dentin sialophosphoprotein; KO, knockout; WT, wild type. *p<0.05.
Effects of DSPP Deletion on Mineralization of Subchondral Bone
Data from plain X-ray images of femur demonstrated that mineralization was lower in Dspp null mice than that of WT mice, especially at 3 and 6 months (Fig. 4A–C), as evidenced by analyses of gray values (Fig. 4D).
To quantitatively measure subchondral bone mineralization of femoral head, μCT was used. Coronal sections of μCT image showed that there was less trabecular bone in femoral head of Dspp null mice at 12 months than that in the WT mice (Fig. 5A and B). In the 12-month-old mice, the secondary ossification center and growth plate in femoral head ossified and left articular cartilage persists in both WT mice and Dspp null mice. There were significant reductions in the BV (Fig. 5D), and BV/TV (Fig. 5E), TMD (Fig. 5G) and BMD (Fig. 5H) in Dspp null mice compared with WT mice while the BV/TV of subchondral bone porosity significantly increased in Dspp null mice compared with WT mice (Fig. 5F). There was no difference in the TV (Fig. 5C) between Dspp null mice and WT mice (p>0.05). These measurements suggest that loss of DSPP can lead to less bone mass and less mineralization of subchondral bone of femoral head although DSPP deletion cannot alter the femoral head volume.
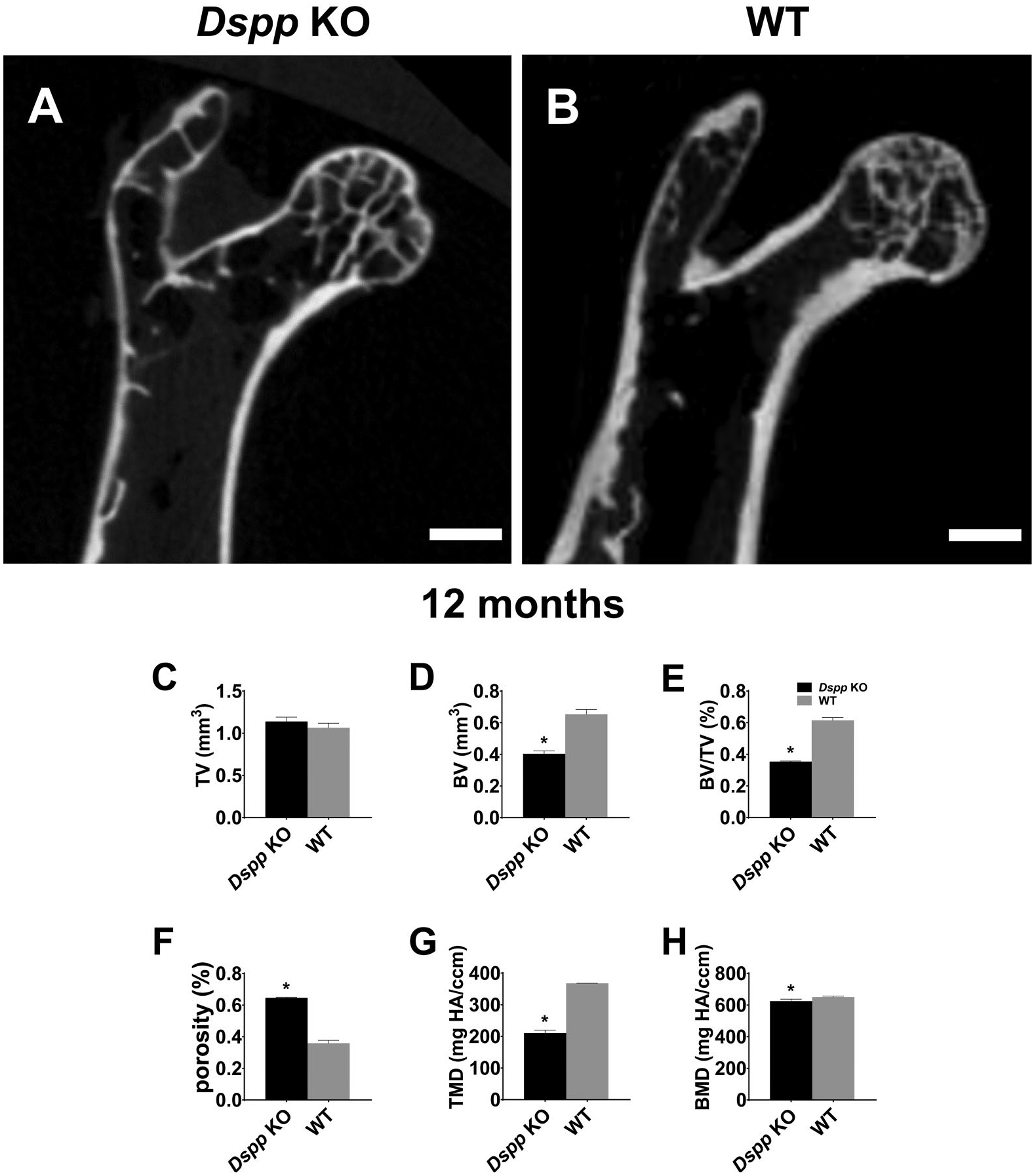
Micro-CT analyses for coronal sections of femoral heads at 12 months. (A) Micro-CT image of coronal sections of femoral head of Dspp null (Dspp KO) mice. (B) Micro-CT image of coronal sections of femoral head of WT mouse. (C–H) Quantification of the parameters concerning the subchondral bone by micro–CT. Data represent mean ± SD (n=3). Scale bars = 1.0 mm. Abbreviations: BMD, bone mineral density; BV, bone volume; BV/TV, bone volume fraction; CT, computed tomography; Dspp, dentin sialophosphoprotein; KO, knockout; Porosity, 1 – BV/TV; TMD, tissue mineral density; TV, tissue volume; WT, wild type. *p<0.05.
It is reported that the level of biglycan in calcified zone correlates with defective regions in mineralization. 17 To further evaluate the mineralization differences, we measured biglycan expression in the femoral head of both Dspp null mice and WT mice. Data demonstrated that the biglycan positive signal could be observed sporadically in the bone of femoral head of Dspp null mice at all time points of 2, 3, and 6 months while the biglycan positive signal could rarely be seen in WT mice (Fig. 6). These indicate that loss of DSPP can delay endochondral ossification in the femoral head cartilage of Dspp null mice.
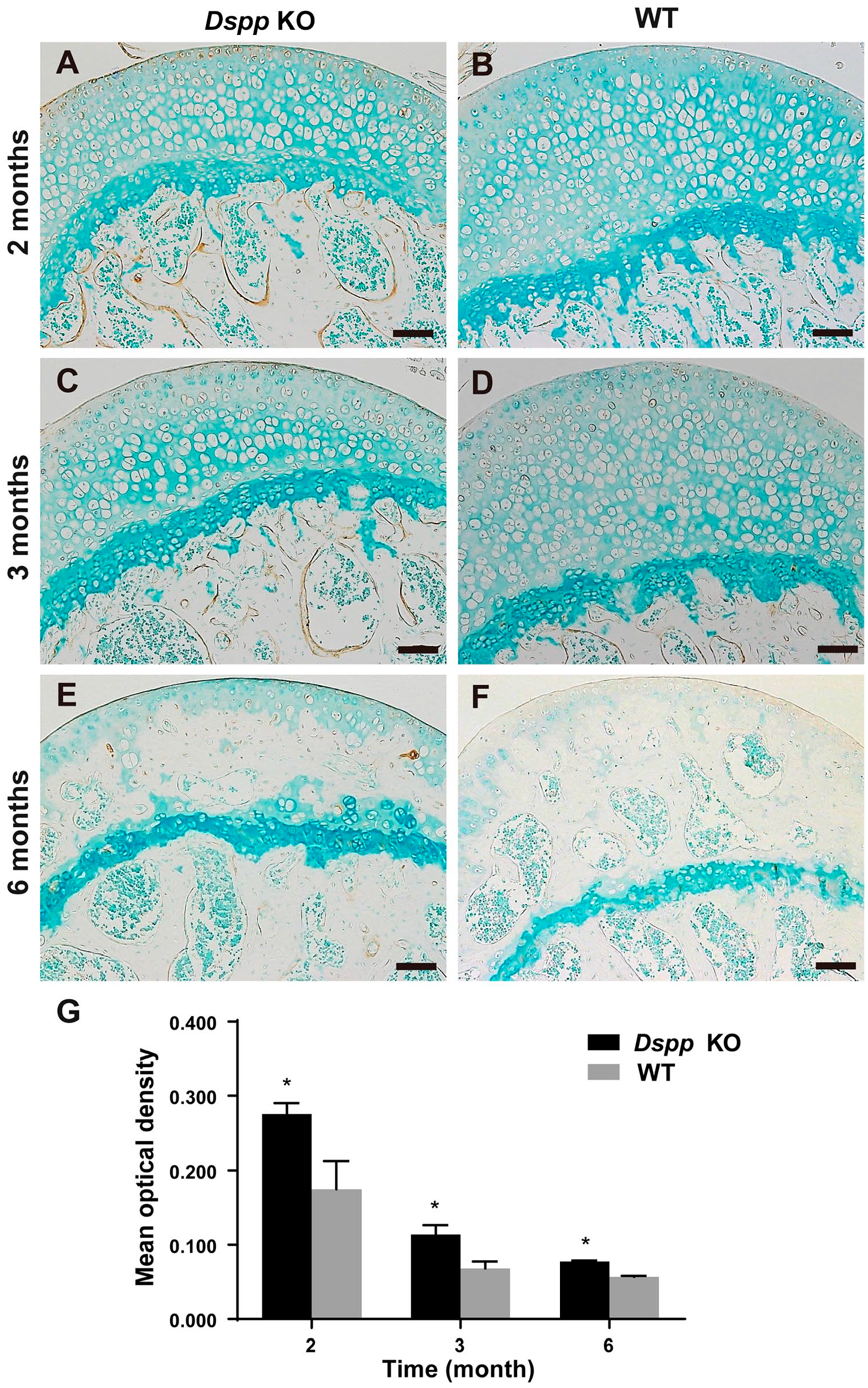
Immunohistochemical (IHC) analyses of biglycan. (A–F) IHC analyses of biglycan of femoral heads from Dspp null (Dspp KO) mice and wild-type (WT) mice at 2, 3, and 6 months. Biglycan in the femoral head was increased in Dspp null mice compared with WT mice. (G) Positive zone areas in the femoral head cartilage from the Dspp null mice and WT mice were measured with ImageJ software. Values are presented as mean ± SD (n=3). Methyl green was used for counterstaining. Scale bars = 100 µm. Abbreviations: Dspp, dentin sialophosphoprotein; KO, knockout; WT, wild type. *p<0.05.
Effects of DSPP Deletion on Endochondral Ossification in Femoral Head
Because blood vessel growth is associated with bone formation during endochondral ossification, 26 and the angiogenic protein vascular endothelial growth factor (VEGF) is believed to be a principal regulator of angiogenesis, 27 we examined its expression in femoral head using IHC. A remarked increase in positive signal of VEGF in cartilage and subchondral bone was observed in 1-month-old Dspp null mice compared with WT mice (Fig. 7A). Further quantitative analyses revealed a significant increase of VEGF-positive signal in cartilage, but there was no significant difference in subchondral bone (p=0.457) between Dspp null mice and WT controls (Fig. 7A). VEGF-positive cells in the growth plate, however, were significantly less in Dspp null mice than that in WT mice, indicating that DSPP deletion may affect the vascular invasion at the transition zone consisting of new endochondral bone formation adjacent to growth plate cartilage. To detect vessels in newly formed bone, we examined the expression of CD31, a marker for mature endothelial cells, in the femoral head from 1-month-old mice. 28 Data demonstrated that the CD31-positive signal was higher in the subchondral bone of femoral head of Dspp null mice than that of WT mice, but the vascular numbers in the regions of interest, that is, the transition zone, were less in Dspp null mice than that in WT mice (Fig. 7B). Data of CD31 expression are also consistent with the results of TRAP, which showed that TRAP-positive cell numbers decreased in Dspp null mice compared with that in WT mice (Fig. 8). The decreases of vessel and osteoclast invasion in the transition zone of subchondral bone indicated that loss of function of Dspp influences hypertrophic cartilage remodeling, ossification, and angiogenesis during the endochondral ossification of subchondral bone.
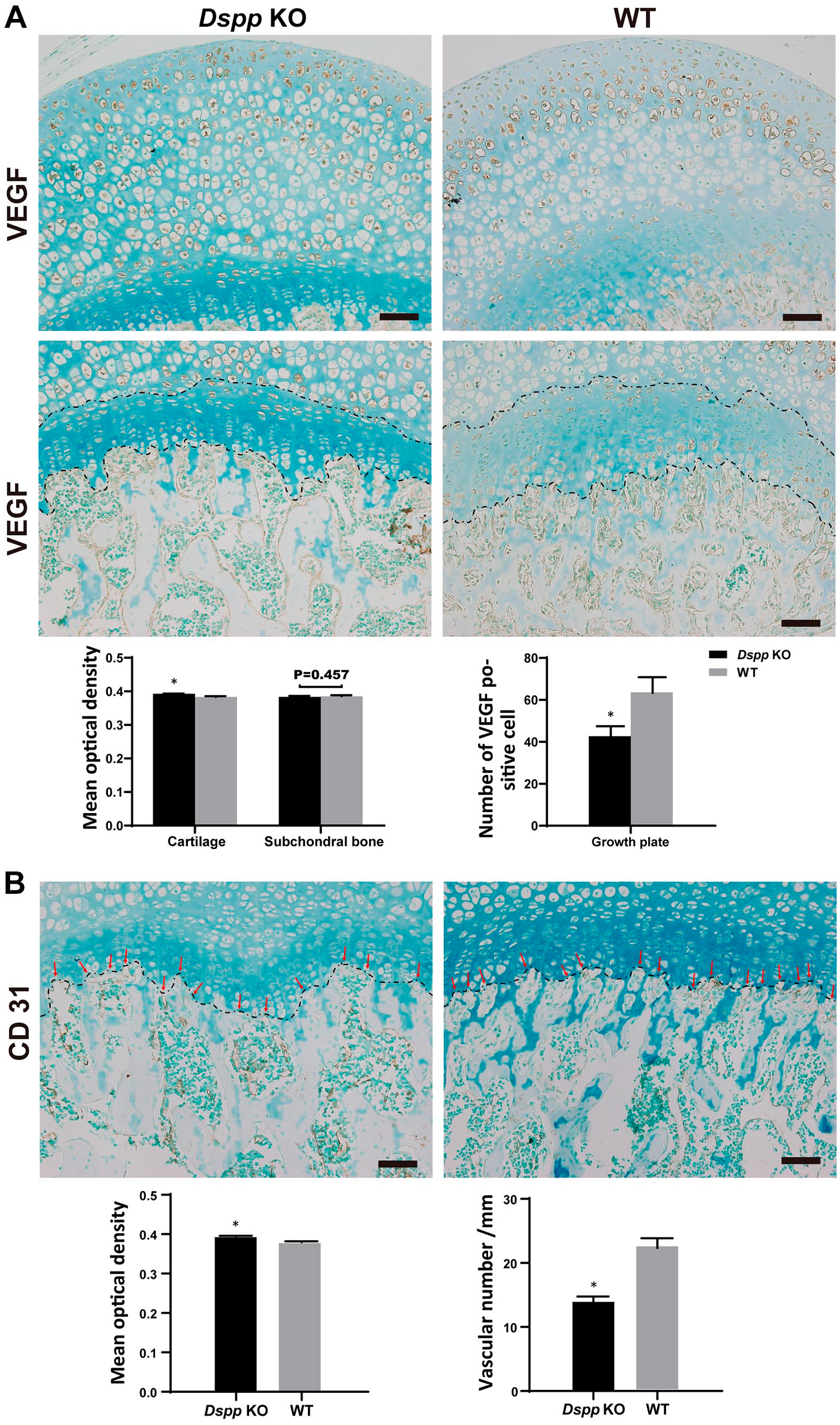
Immunohistochemical analyses of blood vessels in the subchondral bone of femoral head at 1 month. (A) VEGF staining of femoral heads from Dspp null mice (Dspp KO) and WT mice. Black lines indicate growth plate. (B) CD31 staining of femoral heads from Dspp null mice and WT mice. Black lines indicate the interface between growth plate and subchondral bone. Positive zone areas or numbers of positive cells were measured with ImageJ software. Values are presented as mean ± SD (n=3). Scale bars = 50 µm. Abbreviations: Dspp, dentin sialophosphoprotein; KO, knockout; VEGF, vascular endothelial growth factor; WT, wild type. *p<0.05.
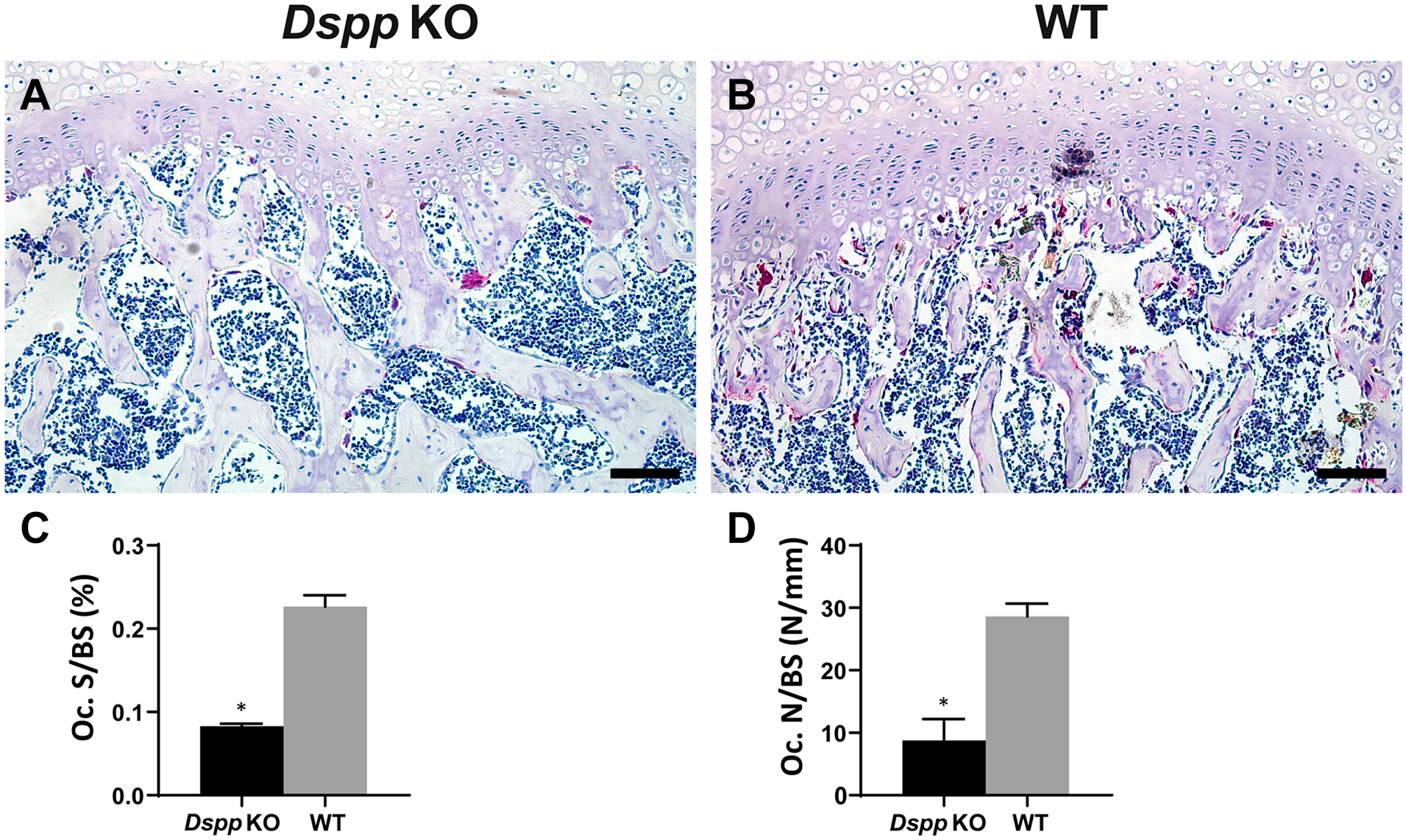
TRAP staining of osteoclasts in the subchondral bone of femoral head from 1-month-old mice. (A) Images of TRAP staining of the femoral head from Dspp null mice (Dspp KO) and WT mice. (B) Number of TRAP-positive cells with more than three nuclei. (C) Oc. S/BS. Values are presented as mean ± SD (n=3). Scale bars = 50 µm. Abbreviations: TRAP, tartrate-resistant acid phosphatase; Dspp, dentin sialophosphoprotein; KO, knockout; Oc. S/BS, osteoclast surface/bone surface of the subchondral bone; WT, wild type. *p<0.05.
In addition, to examine whether osteoblast and osteocyte were affected, we also detected Osterix (OSX), a marker of early differentiation stage of osteoblasts; DMP1, a marker of late differentiation stage of osteoblasts; and sclerostin (SOST), a marker of osteocytes in subchondral bone of the femoral head from 1-month-old mice. Data demonstrated that an increase in OSX expression and reduction in DMP1 expression in osteoblasts were observed in subchondral bone in Dspp null mice compared with WT mice (Fig. 9A–D). The SOST expression in osteocytes also decreased in Dspp null mice compared with WT mice (Fig. 9E and F). This implies that the mineralization of subchondral bone is delayed due to Dspp deletion compared with WT mice.
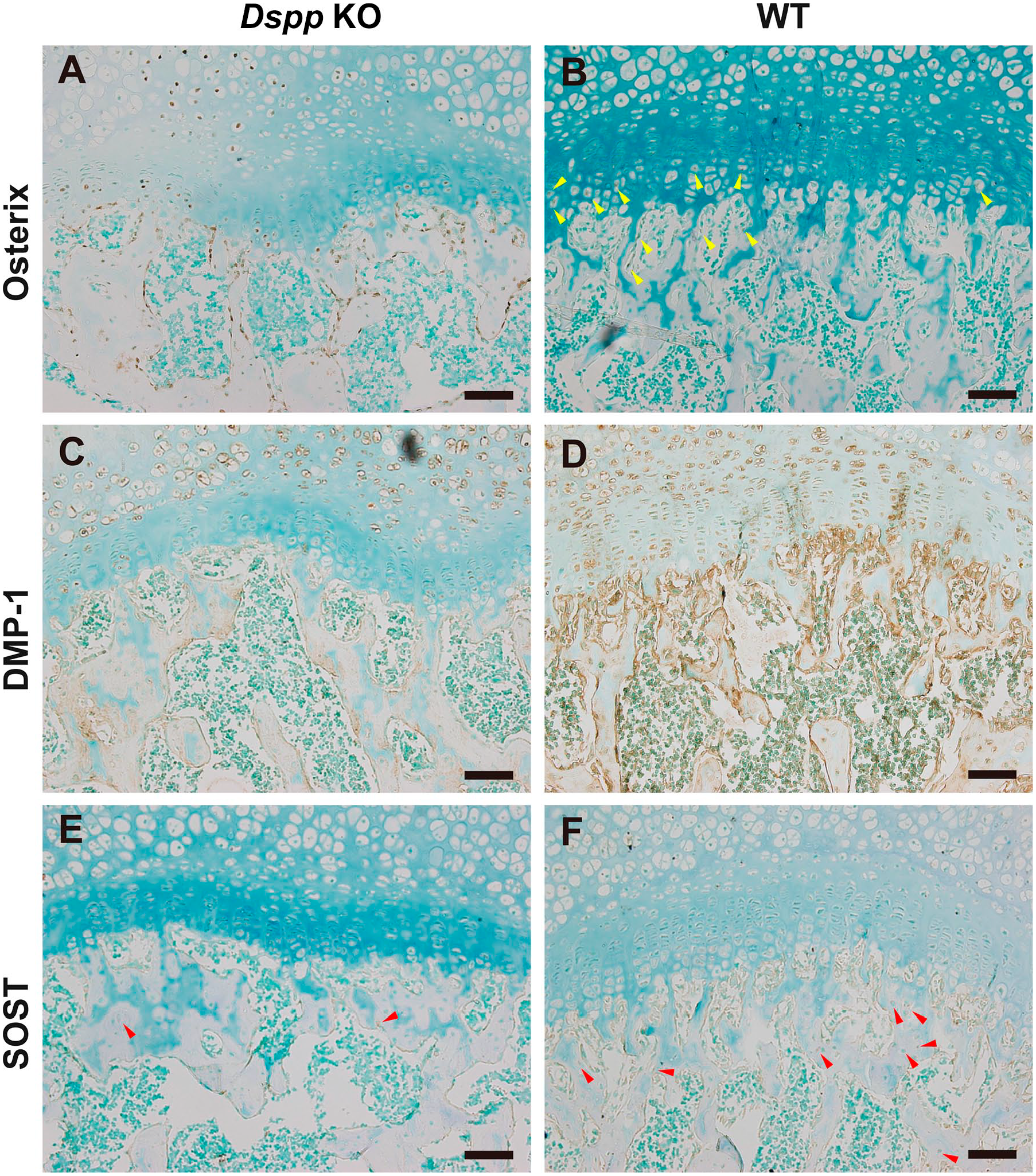
Immunohistochemical (IHC) analyses of osteoblasts and osteocytes in the subchondral bone of femoral head from 1-month-old mice. (A, B) Images from the sections immunostained with the anti-Osterix antibody of Dspp null mice (Dspp KO) and WT mice. Strong signals for Osterix were observed in the chondrocytes and osteoblasts in the subchondral bone of femoral head, while the signals of Osterix in the femoral head from WT mice were very weak. (C, D) IHC for DMP1. In contrary to Osterix, DMP1 signals were remarkably stronger in osteoblasts of femoral head of WT mice than that of Dspp null mice. (E, F) IHC for SOST. SOST-positive osteocytes were less in the subchondral bone of Dspp null mice than that of WT mice (red arrowhead) (n=4). Methyl green was used for counterstaining. Scale bars = 50 µm. Abbreviations: DMP1, dentin matrix protein 1; Dspp, dentin sialophosphoprotein; KO, knockout; SOST, sclerostin; WT, wild type.
Effects of DSPP Deletion on Cell Proliferation and Cell Apoptosis in Femoral Head
It is known that cell proliferation in growth plate cartilage undertakes primary roles for the growth of bone. 9 Therefore, we further evaluated whether DSPP deletion affected cell proliferation or apoptosis of growth plate. Data from BrdU incorporation assay showed that the femoral head cartilage of Dspp null mice had less BrdU-positive chondrocytes in both articular and growth plate cartilages compared with WT mice (p<0.05, Fig. 10). These reveal that DSPP deletion inhibits cell proliferation in femoral head. On the contrary, data from TUNEL assay showed that the femoral head cartilage of Dspp null mice had increased TUNEL-positive chondrocytes in both articular and growth plate cartilages compared with WT mice (p<0.05, Fig. 11). These reveal that DSPP deletion promotes cell apoptosis in femoral head. Taken together, DSPP deletion decreases cartilage cells through abating cell proliferation and increasing cell apoptosis.
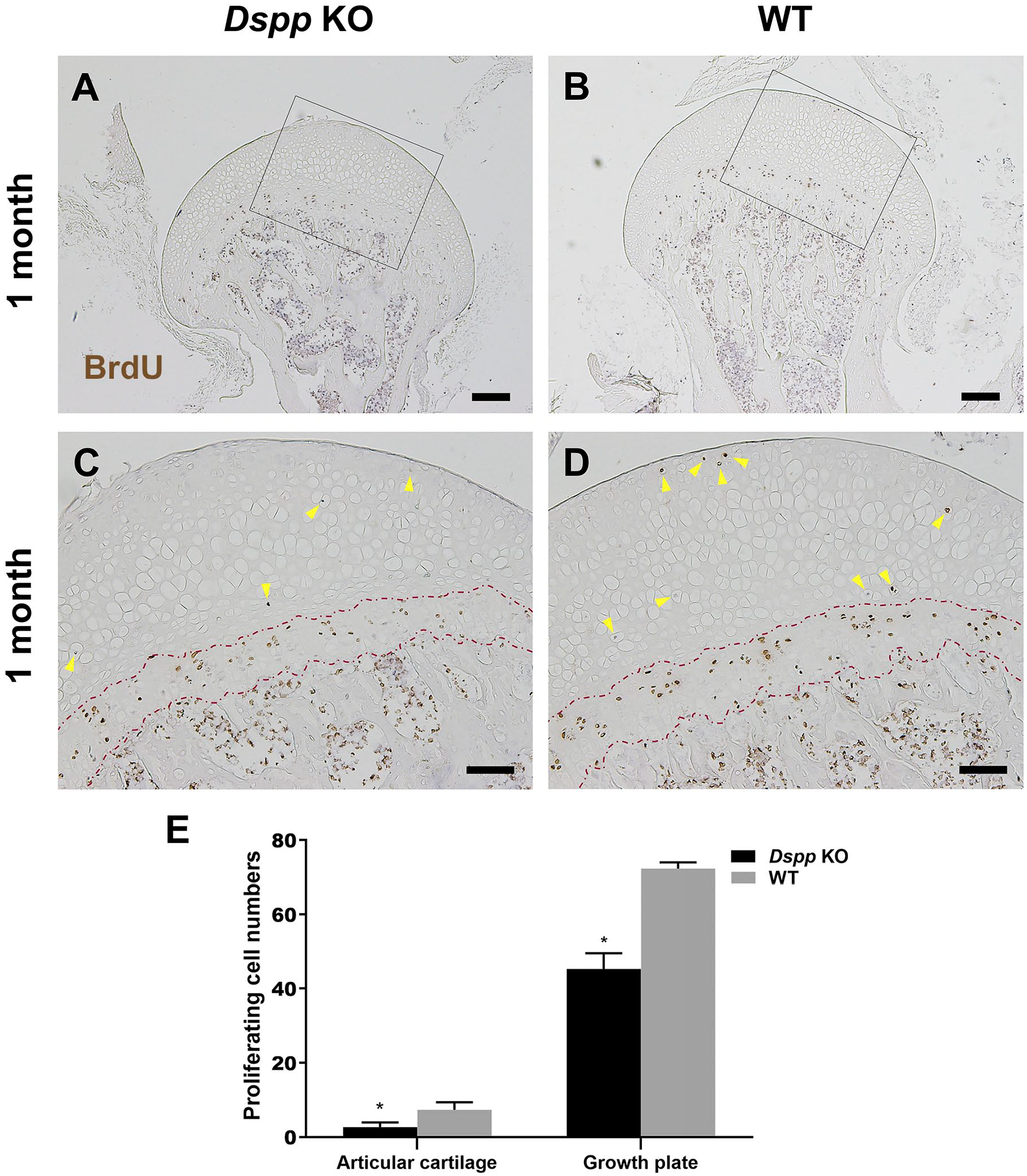
BrdU assay to evaluate the proliferation of the femoral head cartilage of Dspp null mice and WT mice at 1 month old. (A, B) Representative images of BrdU staining of the femoral head from Dspp null mice and WT mice. Scale bars = 200 µm. (C, D) Higher magnification images of Fig. 10A and B, respectively. Fewer proliferating cells in the articular cartilage and growth plate of Dspp null mice than that in the WT mice (yellow arrowheads). Red broken dash lines indicate growth plates in C and D. Scale bars = 100 µm. (E) Quantitative analysis of BrdU-positive cells of the femoral head cartilage, except growth plate cartilage and growth plate cartilage. Data represent mean ± SD (n=4). Abbreviations: BrdU, bromodeoxyuridine; Dspp, dentin sialophosphoprotein; KO, knockout; WT, wild type. *p<0.05.
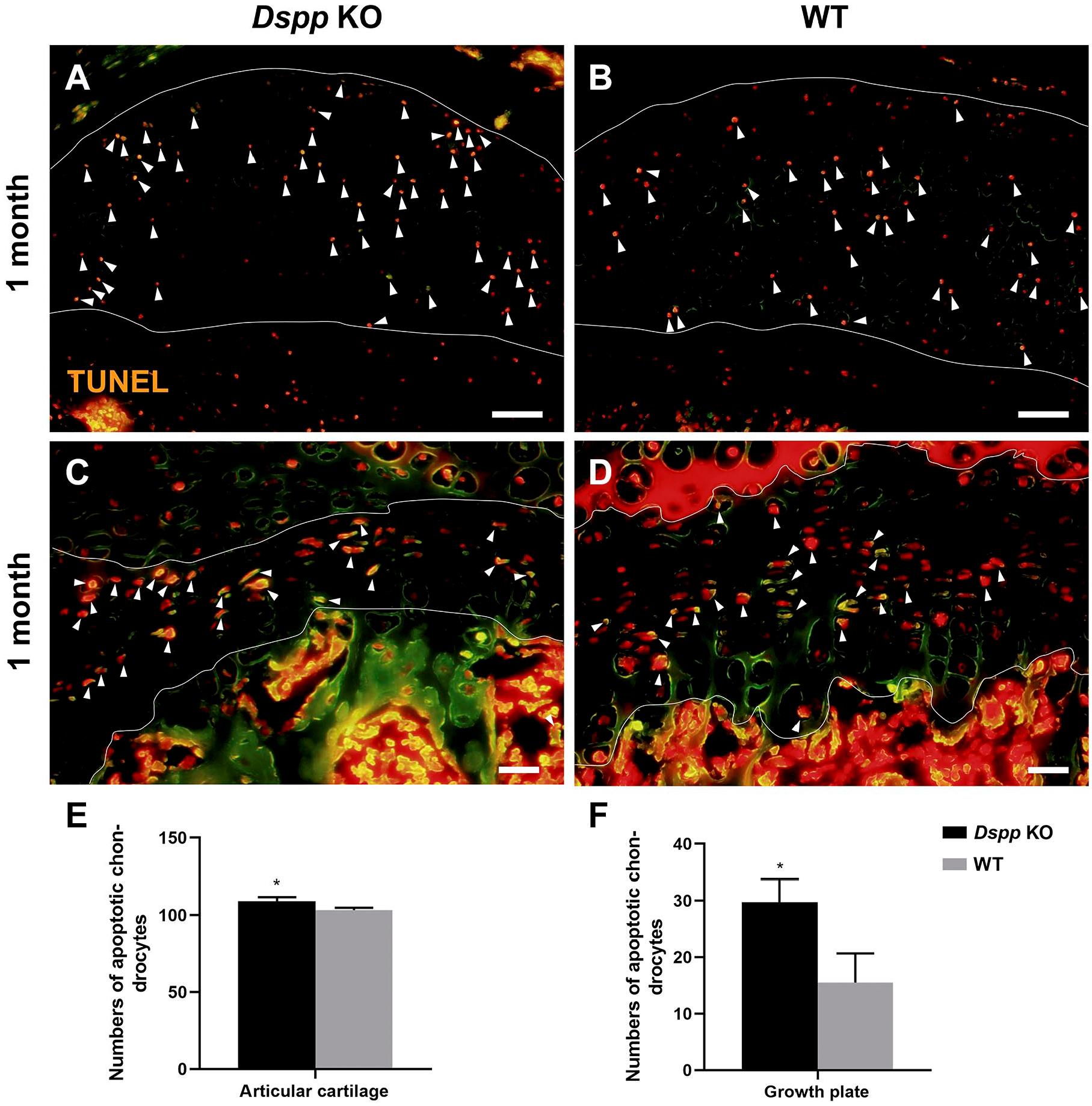
TUNEL assay of femoral head cartilage of Dspp null and WT mice at 1 month old. (A, B) Representative images of TUNEL staining of articular cartilage of the femoral head. White lines indicate articular cartilage. Scale bars = 50 µm. (C, D) Representative images of TUNEL staining of growth plate of the femoral head. White lines indicate growth plate. The orange color in A–D indicates TUNEL-positive cells. Scale bars = 20 µm. (E) Quantitative analyses of TUNEL-positive cells in the femoral head cartilage, except growth plate cartilage and growth plate cartilage. Data represent mean ± SD (n=4). Abbreviations: Dspp, dentin sialophosphoprotein; KO, knockout; TUNEL, Terminal Deoxynucleoitidyl Transferase–mediated Nick-end Labeling; WT, wild type. *p<0.05.
Discussion
DSPP, especially DSP and DPP, is an NCP that plays an important role in dentin mineralization with collagens. Our previous study was the first one to confirm DSPP’s role in the fibrocartilage of mandibular condyle. In current study, we further explore DSPP roles in the hyaline cartilage that constructs the articulating surfaces of long bones.
Generally speaking, the posttranslational modifications (PTMs) of DSPP/DSP are very extensive and extremely complex. DPP portion only has phosphorylation and has no glycosylation. While DSP has little or no phosphorylation, it has N-glycosylation, O-glycosylation, sialic acid modification, and glycosaminoglycan (GAG) chain modifications, which all increase the molecular weight and make the migration rates (Mrs) heterogeneous. The N-glycosylation, O-glycosylation, sialic acid modification/attachments, and GAG chain modifications themselves (all are carbohydrate branches) are very heterogeneous; the carbohydrate branches on different molecular species (same DSP amino acid sequences with various PTMs) vary in length—they vary in the same tissue and vary more greatly between different tissues.6–8,25
Our Western blot demonstrated that there were multiple sizes of DSP, including 70 kDa band, 53 kDa band, and two weak bands migrating below 40 kDa molecular marker, in the femoral head cartilage. These results are consistent with many previous reports6,8,29 and indicate that DSPP is a heterogeneous protein. In our current study, most DSP proteins were at 70 kDa and 53 kDa in the NCP extracts; the 78 kDa band was only detected in teeth, but was not detected in the cartilage and subchondral bone of femoral head. Interestingly, full-length DSPP is the major form in the mandibular condylar cartilage, 7 while we only found that full-length DSPP had trace amount in femoral head cartilage (data not shown). Dr. Qin’s team has noticed that Mrs of DSP/DSPP are different among the tissues of cartilage, salivary glands, brain, and dentin. 6 It is also a common phenomenon that a protein (with the same amino acid sequence) extracted from different tissues has different Mrs due to the variations in the amounts (lengths) of carbohydrates attached to the protein.6–8,25 Thus, the different Mrs of DSP/DSPP bands among different tissues are likely due to variations in degrees of N-glycosylation, O-glycosylation, sialic acid modification, or GAG chain modification between these tissues. 25 The only way to clarify these is to treat the samples with N-glycosidase, O-glycosidase, Sialidase, Chondroitinase ABC, and Chondroitinase AC, and then compare the Mrs between the enzyme-treated samples with the Mrs of samples not treated by the enzymes, which we did not further study.
Previous study found that DSPP expressed in all layers of femoral head cartilage of WT rats. 8 Similarly, our IHC staining also showed that DSPP was present in all layers of the cartilage and the subchondral bone in normal femoral head of WT mice, and decreased with increasing age. H&E and toluidine blue staining showed that Dspp deletion caused insufficient growth of articular cartilage of femoral head, and subsequently insufficient secondary ossification center formation during the growth process. However, Dspp deletion did not affect the morphology or the structural integrity of the femoral head in general. One interesting finding was that the femoral head cartilage thickness in Dspp null mice dramatically decreased. Therefore, these strongly indicate that DSPP plays a crucial role in the growth of femoral head cartilage, but not the histogenesis.
Cell proliferation and apoptosis assay showed that the cartilage cells in the articular cartilage and growth plate were affected by the deletion of Dspp before the occurrence of any detectable histological changes. These observations provide direct evidence that DSPP indeed play an interesting role in proliferation of the articular cartilage and growth plate, which is in coincidence with our previous report of DSPP functions in mandibular condylar cartilage. 21
Femoral head cartilage in the mammalian is one kind of tissue that possesses two distinct categories of cartilage, the epiphyseal cartilage of the growth plate and the hyaline cartilage at the weight-bearing surfaces that drives endochondral bone growth and facilitates joint articulation, respectively.9,30 During the development, femoral head cartilage is comprised of articular cartilage, subarticular epiphyseal cartilage, and endochondral ossification center. 31 Our data from 6 months showed that the secondary ossification center of Dspp null mice was much thinner than that of WT mice, while the region of epiphyseal cartilage was thicker. The biglycan-positive signal was more remarkable in both the secondary ossification center and the subchondral bone of Dspp null mice than that of WT mice. These findings indicate that the loss of function of DSPP can damage the endochondral ossification progress.
To further explore effects of DSPP deletion on endochondral ossification, we examined blood vessels, osteoblasts, osteocytes, and osteoclasts at the transition zone of subchondral bone and growth plate. We found that DSPP deletion abated vessel and osteoclast invasion at the transition zone, and delayed the maturation of osteoblasts and osteocytes in the subchondral bone during endochondral ossification. These observations indicate that DSPP may regulate ossification of calcified cartilage and bone maturation.
In addition, because the gross morphology of femoral head is intact, we speculate that Dspp plays roles in early events of cartilage cell proliferation, but not chondrocyte differentiation. Interestingly, we did find that DSPP deficiency reduced cell proliferation, leading to reduction of cartilage formation. The altered ill-mineralization, however, may be caused by indirect impact of DSPP deletion on cartilage and direct impact of DSPP deficiency on bone. These findings confirm a role of Dspp deletion in the decrease of the articular cartilage, which is consistent with our observation of the mineralization decrease in Dspp null mice.
In conclusion, DSPP plays important roles in the femoral head cartilage cell proliferation, cartilage development, and mineralization.
Footnotes
Author Contributions
QL contributed to conception, design, data acquisition, analysis, and interpretation, and drafted the manuscript. NM contributed to conception, design, data analysis, and critically revised the manuscript. QZ and HK contributed to mouse genotyping, sample collection. XD, HS, and DX contributed to data analysis and critically revised the manuscript. HS contributed to conception, design, and data analysis and critically revised the manuscript. Each author has read and approved the manuscript before submission.
Competing Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grant 81600890 from National Natural Science Foundation of China, 2015340 and 2015205 from Jilin University, 2016c044-3 from the Development and Reform Commission of Jilin Province, [2016]-484 and JJKH20180102KJ from the Education Department of Jilin Province, 2015Q014 from Jilin Provincial Department of Health, and 2018SCZWSZX-034, 2019SRCJ006, and JCSZ201978-10 from Jilin Provincial Department of Finance.
