Abstract
Recently, dentin sialophosphoprotein (DSPP) was found to be expressed in the mandibular condylar cartilage (MCC), but the possible roles of this molecule in the formation, growth, and maintenance of the cartilage are largely unclear. To analyze such roles, we examined Dspp null mice compared with wild-type mice to assess the consequences of Dspp deletion on the morphology and structure of the MCC. Our data showed that DSPP is expressed in the prechondroblastic, chondroblastic, hypertrophic layers of the MCC. Dspp null mice exhibited decreases in the amount of MCC, with reduced formation of articular and prechondroblastic layers in which progenitor cell proliferation levels were distinctly affected. The expression of extracellular matrix molecules, including biglycan and collagen II, IX, and X, was remarkably altered. The findings in this study indicate that continuous DSPP action is required for the growth and/or maintenance of the MCC.
Introduction
The temporomandibular joint (TMJ) is a unique articulating joint that is essential for proper mastication and verbal communication (Embree et al. 2010). The mandibular condyle, whose surfaces are covered by fibrocartilage, is a component of the TMJ and is structurally distinct from both growth plate and articular cartilage in limbs (Serrano et al. 2011). Mandibular condylar cartilage (MCC) is often classified as a secondary cartilage and defined as cartilage arising from the periosteum of membrane bone after bone formation (Shibata and Yokohama-Tamaki 2008). MCC has unique characteristics with regards to its origin (Balic et al. 2009), cell differentiation, cell proliferation, and the synthesis of the extracellular matrix (ECM) (Shibata et al. 2002). This cartilage arises from alkaline phosphatase (ALP)-positive progenitor cells of the periosteum, which are derived from neural crest, continuous to the preliminarily formed ossifying mandible (Shibata et al. 2002).
Mandibular condylar cartilage consists of chondrocytes and an abundant ECM that is principally composed of water, collagen, and proteoglycans, with other non-collagenous proteins (NCP) and glycoproteins present in lesser amounts (Fox et al. 2009). Chondrocytes reside within the ECM and synthesize and maintain the ECM under normal conditions of low turnover (Goldring and Goldring 2007). The ECM plays a critical role in the preservation of cartilage tissue integrity (Embree et al. 2010).
The Small Integrin-Binding LIgand, N-linked Glycoprotein (SIBLING) family is one category of NCPs and includes dentin sialophosphoprotein (DSPP), dentin matrix protein 1 (DMP1), bone sialoprotein (BSP), osteopontin (OPN), and matrix extracellular phosphoglycoprotein (MEPE). Among the SIBLING family members, the former four are acidic proteins, whereas the latter one is a basic protein. Sun et al. (2010) analyzed the distribution of the four acidic members in the SIBLING family in the MCC of rat and observed the presence of significant amounts of DSPP in the condylar cartilage.
DSPP is a larger precursor protein first reported in 1997 (MacDougall et al. 1997); its specific cleavage products are dentin sialoprotein (DSP) and dentin phosphoprotein (DPP), discovered in 1981 (Butler et al. 1981) and in 1967 (Veis and Perry 1967), respectively. Initially thought to be unique for dentin formation during tooth development, DSPP has been shown to have a much broader expression pattern; it is also found in bone, cementum, and non-mineralized tissues (salivary glands, cartilage, liver, lung, kidney, and brain) (Qin et al. 2002; Baba et al. 2004; Alvares et al. 2006; Ogbureke and Fisher 2007; Sun et al. 2010; Prasad et al. 2011; Zhang et al. 2011). Notably, the expression level of DSPP is significantly higher in the MCC than in the long bone (Sun et al. 2010; Prasad et al. 2011). Dspp null mice develop tooth defects with enlarged pulp chambers, an increased width of the predentin zone, hypomineralization, and pulp exposure (Sreenath et al. 2003). As DSPP is expressed at a very low level in the long bone, its disruption in mice results in modest but significant changes in the material properties of long bones at certain time points (Verdelis et al. 2008). Although the levels of DSPP expression in cartilage are higher than in the long bone (Prasad et al. 2011), there have been no reports regarding the phenotypic alterations in the cartilage of Dspp null mice.
In this investigation, we examined the MCC of Dspp null mice, in comparison with wild-type (WT) mice, and evaluated its potential function in the postnatal development of condylar cartilage of the mouse mandible. The findings of this study showed that the proliferation of progenitor cells in MCC was affected, leading to disruption of the articular layer and prechondroblastic layer. Meanwhile, the ECM molecules, such as biglycan and collagen II, IX, and X, were also altered in the Dspp null mice.
Materials & Methods
Plain X-ray Radiography and Micro-computed Tomography (μ-CT)
Mandibles dissected from 2-month-old, 3-month-old, and 6-month-old WT and Dspp null mice were analyzed with the Faxitron MX-20DC12 Specimen Radiography System (Faxitron X-ray Corp.; Buffalo Grove, IL). Then, on the mandibles dissected from 3-month-old and 6-month-old mice, micro-computed tomography (μ-CT) analyses were performed using a Scanco micro-CT35 imaging system (Scanco Medical; Basserdorf, Switzerland) with a medium-resolution scan (7.0-µm slice increment). The images were reconstructed with the EVS Beam software using a global threshold at 240 Hounsfield units. The animal protocol was approved by the Baylor College of Dentistry Institutional Animal Use and Care Committee, Texas A&M Health Science Center (Dallas, TX).
Histology & Immunohistochemistry
For the histology and immunohistochemistry (IHC) assay, under anesthesia, 1-month-old and 2-month-old mice (n=3 for each group) were perfused from the ascending aorta with 4% paraformaldehyde in 0.1 M phosphate-buffered saline (PBS). Half of the mouse head was dissected and further fixed in the same fixative overnight, followed by decalcification in 8% EDTA (pH 7.4) at 4C for approximately 2 weeks. Tissues were processed for paraffin embedding, and serial 5-μm sections were prepared.
The sections were stained with hematoxylin and eosin (HE) or toluidine blue or employed for IHC analyses. For immunohistochemical detection of DSPP/DSP, the anti-DSP monoclonal antibody, referred to as “anti-DSP-2C12.3” (Baba et al. 2004) was used at a dilution of 1:500. The anti-biglycan antibody (a gift from Dr. Larry Fisher of NIDCR, National Institutes of Health) was used at a dilution of 1:1000. The anti-collagen II antibody (Santa Cruz Biotechnology; Dallas, TX) was used at a dilution of 1:25. All IHC experiments were carried out using ABC M.O.M. kit and DAB kit (Vector Laboratories; Burlingame, CA) according to the manufacturer’s instructions. Methyl green was used for counterstaining.
HE sagittal sections corresponding to the middle portion of the MCC of each animal were captured using a digital camera. For each mouse, the condylar cartilage thickness was measured in a blinded, non-biased manner using the image pro plus 5.1 (Media Cybernetics, Inc.; Rockville, MD). Briefly, a 200× magnification image was captured at the central portion of the MCC. Then, the average thickness of the central portion in the MCC and the cartilage thickness of each layer were calculated along the perpendicular line through the midpoint of the intermediate zone of the articular disc. Furthermore, the cartilage cells in a 400× magnification image were counted and analyzed statistically. Measurements were performed on three sections from each mouse using three mice in each group. We employed Student’s t-test for statistical analyses between groups and took p<0.05 as statistically significant.
In Situ Hybridization
For the in situ hybridization (ISH) assay, samples were harvested as described above (n=3 for each group). The PBS and EDTA solutions were prepared using water pretreated with 0.1% diethylpyrocarbonate (DEPC). After fixation with 4% paraformaldehyde in PBS solution at 4C for 48 hr, the samples were decalcified in 15% EDTA (pH 7.4) at 4C for 7 or 10 days. Then, the tissues were processed for paraffin embedding, and serial 5-μm sections were prepared.
The RNA probes for DSPP and type X collagen were kindly provided by Xiaofang Wang (Texas A&M University Baylor College of Dentistry); the RNA probes for collagen type IX RNA were synthesized as described by Wang et al. (2012). Briefly, an 878-bp fragment from the full-length mouse collagen type IX cDNA was obtained by PCR using forward primer 5′-TGCC TCTCCCCACC CCTTTCCT-3′ and reverse primer 5′-GCGGATGGCTTTGCAGTGCT-3′. The PCR product was then subcloned into the pCRII-TOPO vector (Invitrogen; Carlsbad, CA), which was linearized with NotI to synthesize the antisense RNA probes using the Sp6 RNA polymerase or with HindIII to synthesize the sense RNA probes using the T7 RNA polymerase.
The probes were labeled with digoxigenin (DIG) using an RNA Labeling Kit (Roche; Indianapolis, IN). DIG-labeled RNA probes were detected by an enzyme-linked immunoassay using a specific anti-DIG-ALP antibody conjugate (Roche) and an improved substrate, which produces a red color for positive signals (Vector Laboratories), according to the manufacturer’s instructions. The sense probe was used in place of the antisense probe in negative control experiments.
BrdU Incorporation
BrdU was administered to 1-month-old Dspp null mice and WT mice (n=3 for each group) at a dosage of 1 ml per 100 g body weight by intraperitoneal injection according to the manufacturer’s instructions (Invitrogen). Two hours after the injection, the mice were sacrificed. Half of the mouse head was dissected and processed for paraffin embedding, and 5-µm sections were prepared for BrdU detection using a Zymed BrdU staining kit (Invitrogen) following the manufacturer’s instructions. Four serial sections from each of four individual samples of Dspp null mice and WT mice were counted, and the data were analyzed statistically.
Results
Loss of Dspp Reduces the Thickness of the Mandibular Condyle
To analyze the precise functions of DSPP in MCC, we examined this tissue in Dspp knockout mice in which exons 2 to 5 spanning the entire coding region of Dspp were ablated (Sreenath et al. 2003), and used WT mice for comparison purposes.
Analysis of the Dspp knockout mouse using plain X-ray showed mild phenotypic changes in the mandibular condyles of 2-month-old Dspp null mice (Figs. 1A and 1B). At 3 and 6 months of age, the defects to the condyles became more apparent (Figs. 1C–1F). A three-dimensional morphometric analysis of the mandibular condyle in each mouse was performed using μ-CT. The μ-CT data of 3- and 6-month-old mice (Figs. 1G–1J) were consistent with the findings in plain X-ray analysis.
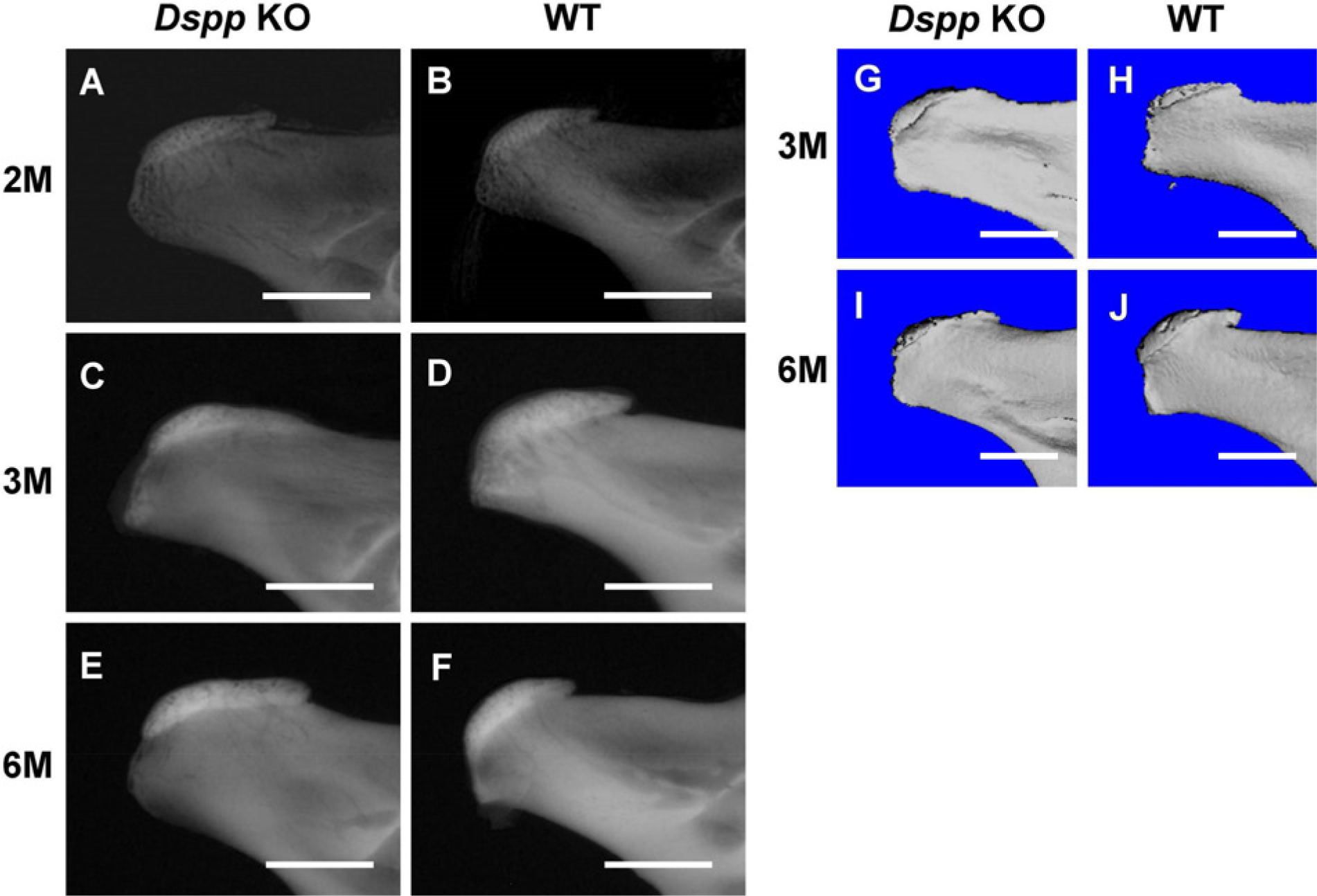
Plain X-ray and μ-CT analyses. (A–F) Plain X-ray radiographs of mandibular condyles from Dspp null (Dspp KO) and WT mice at the ages of 2, 3, and 6 months after birth. (G–J) μ-CT images of mandibular condyles from 3-month-old and 6-month-old Dspp null (Dspp KO) and WT mice. Dspp, dentin sialophosphoprotein; KO, knockout; M, month; WT, wild-type. Bars = 1 mm.
The mandibular condyle undergoes endochondral ossification and represents a major growth site in the mandible contributing to elongation of the mandibular ramus (Wang et al. 2011); in this role, MCC works as a growth cartilage. Given the abundance of DSPP in MCC, the phenotypic changes to the mandibular condyle observed by plain X-ray and μ-CT analyses indicate that Dspp deletion may influence MCC development and have an impact on the bone morphology of the mandibular condyle.
Loss of DSPP Alters the Articular and Prechondroblastic Layers of Mandibular Condylar Cartilage
As an important growth site for the mandible, the condylar cartilage is divided into five layers: articular, prechondroblastic, chondroblastic, hypertrophic, and cartilage–bone interface (Hinton and Carlson 2005).
Morphological differences in MCC between Dspp null mice and WT mice were assessed in HE-stained (Figs. 2A–2D) tissue sections, which showed significant alterations in the articular and prechondroblastic layers in the Dspp null mice (Figs. 2S–2T). The defects in the Dspp-deficient mice were further confirmed by toluidine blue staining (Figs. 2E–2H). It is worth noting that, in the Dspp null mice, the whole layers of the MCC have metachromasia (blue or blue/purple staining), whereas in contrast, the articular and prechondroblastic layers of WT mice demonstrated almost no metachromasia (Figs. 2F and 2H). We also observed a thinner layer of cartilage in the mandibular condyle from the Dspp null mice compared with the WT mice at the ages of 1 month and 2 months. Our quantitative analyses revealed a significant decrease in the MCC thickness as a result of a decreased number of cells in 1-month-old and 2-month-old Dspp null mice, when compared with WT controls (Figs. 2Q–2R).
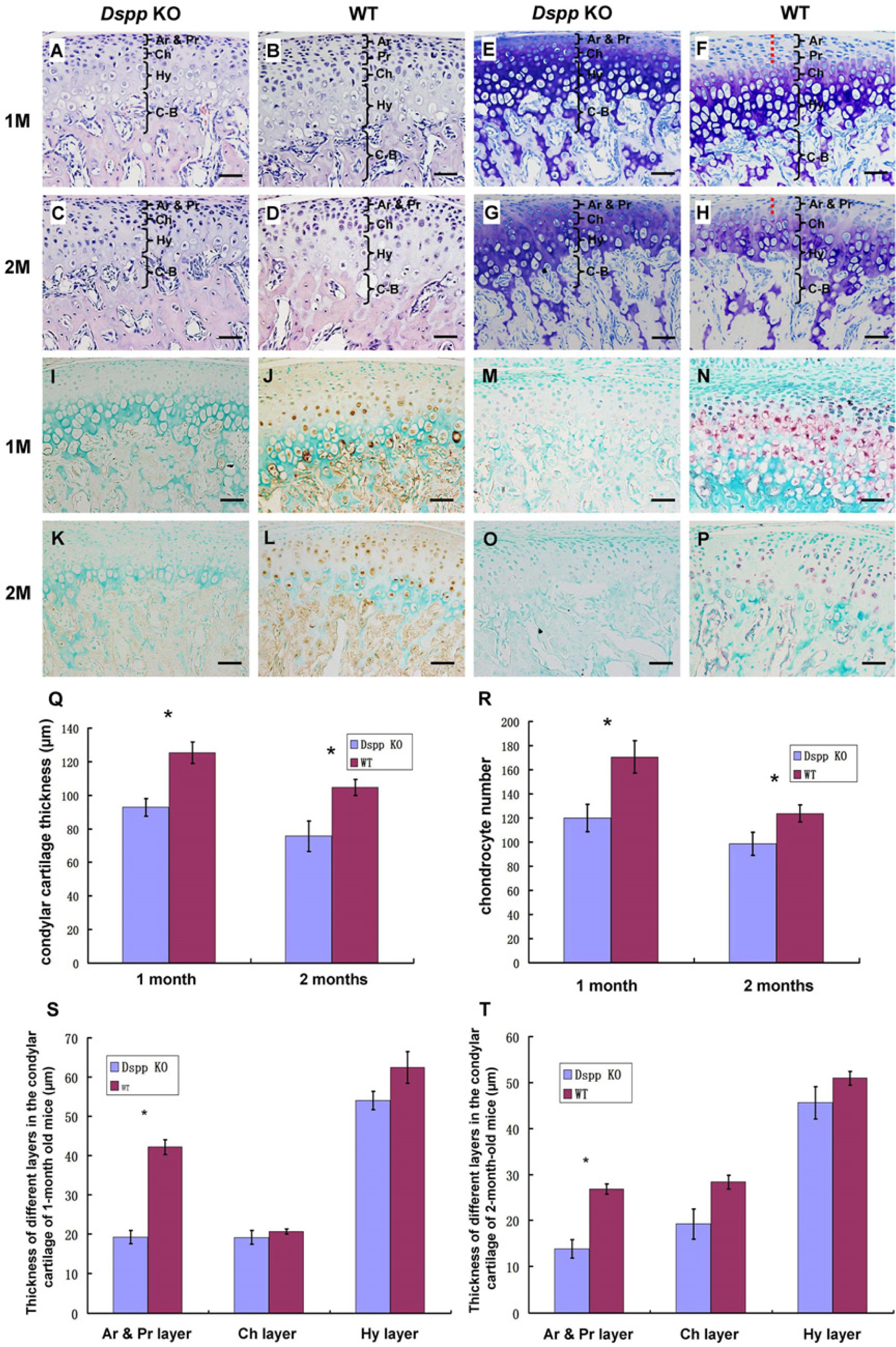
Hematoxylin and eosin (HE) staining, toluidine blue staining, immunohistochemistry (IHC), and in situ hybridization (ISH) analyses of dentin sialophosphoprotein (DSPP) in the mandibular condylar cartilage of 1- and 2-month-old mice. (A–D) Images from HE-stained sections. (E–H) Samples stained with toluidine blue. Note that the articular and prechondroblastic layers of WT mice had nearly no metachromasia (indicated by red dashed line in F and H). (I–L) Images from the sections immunostained with the anti-DSP antibody. The samples from Dspp null mice (Dspp KO) (I, K) were used as negative controls for the IHC analyses. (M–P) Images from the sections stained by ISH analyses with the anti-sense probe that was specific for DSPP mRNA. Samples from the Dspp null mice (Dspp KO) at the same ages were used as negative controls (M, O). (Q) Quantitative analysis for the whole thickness of condylar cartilage in the 1-month-old and 2-month-old Dspp null (Dspp KO) and WT mice. (R) Quantitative analysis for chondrocyte numbers in the condylar cartilage of 1-month-old and 2-month-old Dspp null (Dspp KO) and WT mice. We also did quantitative analyses on the changes in the thickness of different layers of mandibular condylar cartilage in 1-month-old and 2-month-old Dspp null (Dspp KO) and WT mice (S, T). Data in each graph represent mean ± SD (n=3). *p<0.05. Ar, articular layer; Pr, prechondroblastic layer; Ch, chondroblastic layer; Hy, hypertrophic layer; C–B, cartilage–bone interface; M, month; KO, knockout; WT, wild-type. Bars = 50 µm.
As shown in Figs. 1 and 2, the phenotypic changes in the articular cartilage of mandibular condyle in the Dspp null mice were apparent using histology but were not readily identified by plain X-ray analyses.
In rats, others have shown that DSPP is observed across the whole area of the cartilage but is most pronounced within the cells in the articular layer and prechondroblastic layer at 2 weeks after birth. However, at 5 and 8 weeks, DSPP is more noticible in the chondroblastic layer and the upper hypertrophic layer than in the superficial layer (Sun et al. 2010). The information above indicated that, with increasing age, the primary expression of DSPP changes from superficial layers to deeper layers.
After observing the phenotypic changes in the MCC of Dspp null mice, we analyzed the expression pattern of Dspp mRNA and DSPP protein in the 1-month-old and 2-month-old WT mice while using the Dspp null mice as a negative control (Figs. 2I–2P). Data showed that Dspp mRNA and DSPP protein are present in the prechondroblastic, chondroblastic, and hypertrophic layers of the MCC at the ages of 1 and 2 months.
Loss of DSPP Alters the Proliferation of Mandibular Condylar Cartilage
To obtain further insight into the effects of DSPP inactivation on MCC, BrdU labeling was used to examine proliferating cells in the MCC. A significant decrease in proliferating cells was observed in the superficial layer of the MCC in the Dspp null mice, whereas the number of proliferating cells in the subchondral bone of the Dspp null mice did not differ from that observed in the WT mice (Fig. 3). These findings indicated that the loss of DSPP affects the proliferation of progenitor cells in the MCC.
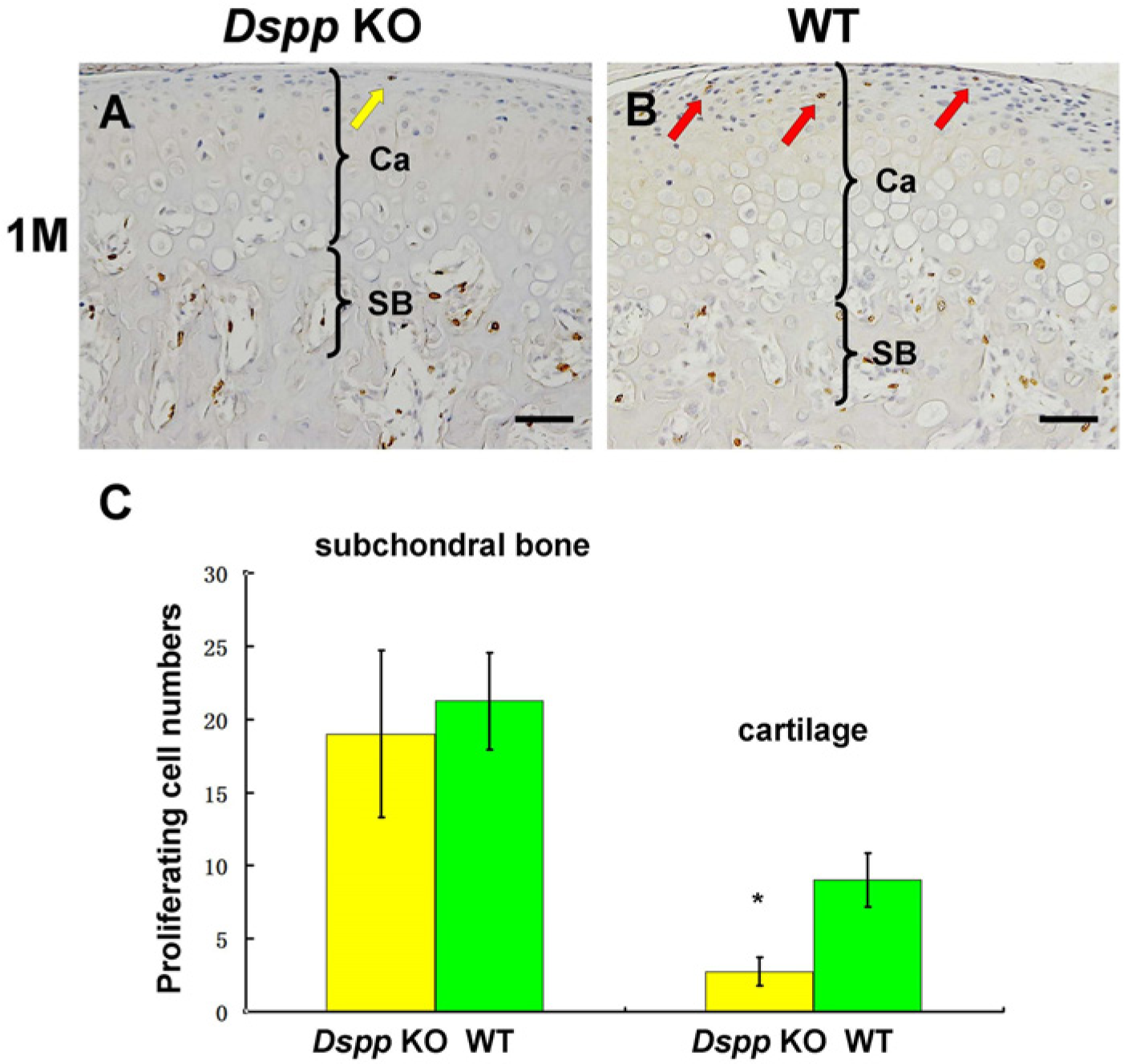
BrdU labeling of mandibular condylar cartilage (MCC) in Dspp null (Dspp KO) and WT mice at the age of postnatal 1 month. BrdU staining on sagittal sections of the temporomandibular joint (A, B) revealed fewer proliferating cells in the articular layer and prechondroblastic layer in Dspp null mice (yellow arrow in A) than in the WT mice (red arrows in B). (C) Quantitative analysis of BrdU-positive cells in the MCC of Dspp KO and WT mice. Data in C represent mean ± SD. *p<0.05. Ca, cartilage; SB, subchondral bone; Dspp, dentin sialophosphoprotein; KO, knockout; WT, wild-type; M, month. Bars = 50 µm.
Loss of DSPP Alters the Expression of ECM Molecules in the Mandibular Condylar Cartilage
Under physiological conditions, the cells in the chondroblastic layer are characterized by the expression of type II collagen, which is covalently bound by type IX collagen (Helminen et al. 2002), whereas the chondrocytes in the hypertrophic zone are characterized by the expression of type X collagen. Additionally, biglycan is abundant in the fibrocartilage, in which this proteoglycan plays a role in stabilizing the matrix (Chen et al. 2006).
We examined the distribution of biglycan and type II collagen proteins using IHC (Figs. 4A–4J). As the antibodies against collagen IX and X that we used had some cross reactions with other collagens and caused strong background, we used in situ hybridization to detect the expression of collagen IX and X mRNA in the cartilage (Figs. 4K–4P).
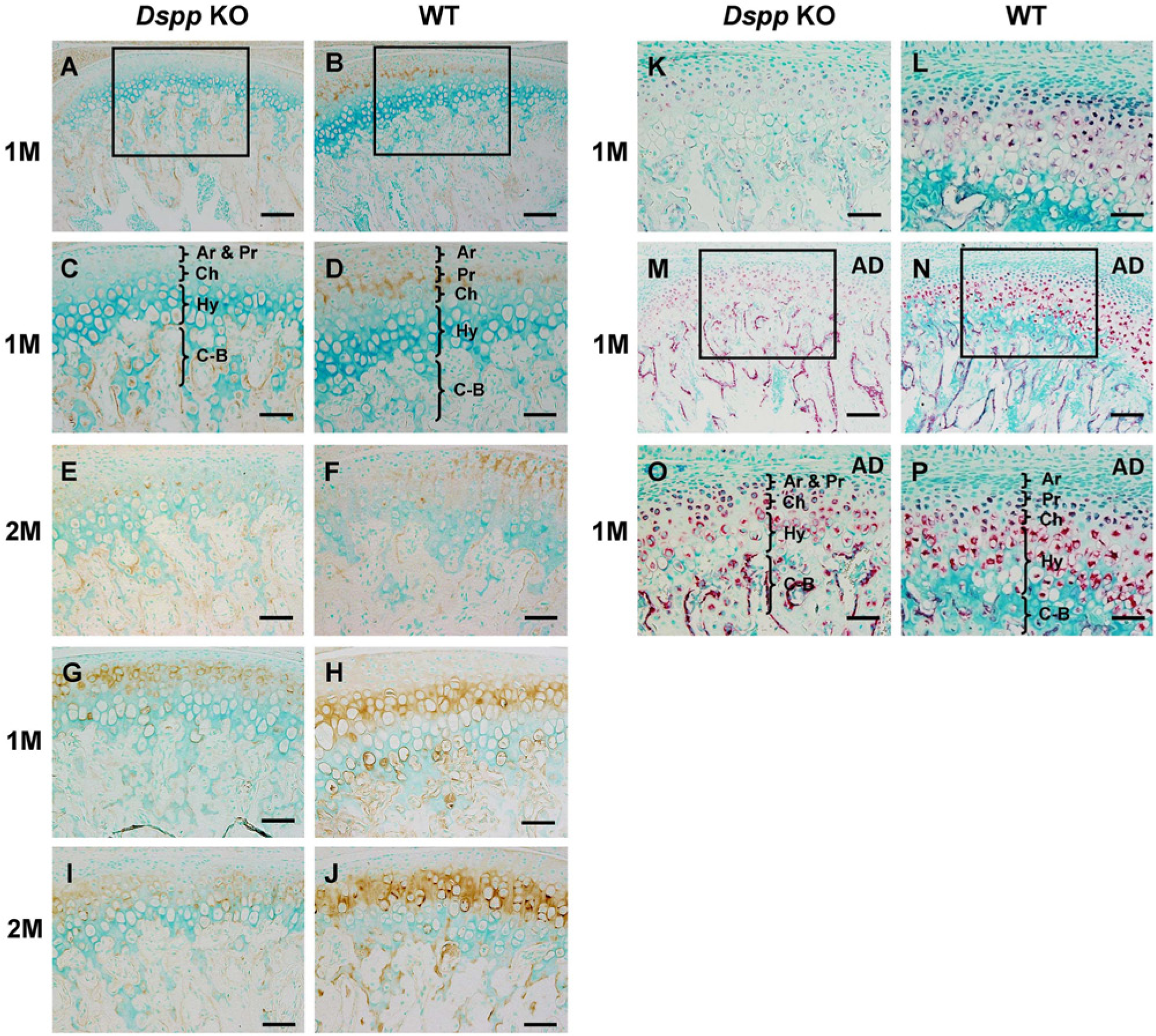
Immunohistochemistry (IHC) staining for biglycan and type II collagen, and in situ hybridization (ISH) staining for type IX collagen and type X collagen in the mandibular condylar cartilage (MCC) of 1- and/or 2-month-old mice. (A–D) IHC staining for biglycan in the MCC of 1-month-old mice. (C) Higher magnification view of the boxed area in A. (D) Higher magnification view of the boxed area in B. (E, F) IHC staining for biglycan in the MCC of 2-month-old mice. (G–J) IHC staining for type II collagen. (K, L) ISH staining for type IX collagen. (M–P) ISH staining for type X collagen. (O) Higher magnification view of the boxed area in M. (P) Higher magnification view of the boxed area in N. The pink/red color (in K–P) indicates positive ISH signal. AD, articular disc; Ar, articular layer; Pr, prechondroblastic layer; Ch, chondroblastic layer; Hy, hypertrophic layer; C–B, cartilage–bone interface; M, month; Dspp, dentin sialophosphoprotein; KO, knockout; WT, wild-type. Bars A, B, M, N = 100 µm; bars C–K, O, P = 50 µm.
In the WT mice, biglycan protein was predominantly present in the articular and prechondroblastic layers, whereas it was markedly reduced or rarely observed in any cartilage layers in the Dspp null mice at the age of 1 or 2 months. Type II collagen protein was mainly present in the ECM of the chondroblastic layer (Figs. 4H and 4J) in the WT mice. The protein level of type II collagen in the chondroblastic layer of the Dspp null mice (Figs. 4G and 4I) was lower than that in the WT mice.
Data from in situ hybridization demonstrated that Col 9 mRNA was highly expressed in prechondroblasts, chondroblasts, and hypertrophic chondrocytes in the MCC of the WT mice (Fig. 4L), whereas, in Dspp null mice (Fig. 4K), the Col 9 mRNA signal was confined to prechondroblasts and chondroblasts and was evidently weaker in the hypertrophic chondrocytes than in the same cells in WT mice at the age of 1 month. Col 10 mRNA was observed in the prechondroblasts, chondroblasts, and hypertrophic cells of the condylar cartilage and preosteoblasts of the subchondral bone of WT mice. In the Dspp null mice, Col 10 mRNA was seen in all cells of condylar cartilage, and its expression in the preosteoblasts was stronger than that in WT mice.
Discussion
In this study, we used the Dspp null mice to examine the roles of DSPP in the MCC. Plain X-ray and μ-CT analyses revealed an obviously flattened mandibular condyle in Dspp null mice compared with that in WT mice at the ages of 3 and 6 months, whereas histology analyses (HE and toluidine blue staining) revealed the occurrence of the cartilage defects at the ages of 1 and 2 months. The subchondral bone was affected by Dspp ablation, manifesting a lower mineralized subchondral area in plain X-ray and an increased proportion of osteoblasts, as determined using HE staining.
Moreover, we systematically evaluated the presence and distribution of DSPP in the MCC of mice using IHC and ISH approaches. Our data showed that DSPP mRNA as well as DSPP protein are mainly present in the prechondroblastic, chondroblastic, and hypertrophic layers in the 1- and 2-month-old mice; these findings are consistent with IHC results reported by Sun et al. (2010). We observed that, at the age of postnatal 2 weeks (Sun et al. 2010), the expression of DSPP was pronounced in the articular and prechondroblastic layers but, with increasing age, the distribution of DSPP shifted into deeper layers. Furthermore, Dspp was reported to be expressed at higher levels (1.6 times higher) in the perichondrium than in the cartilaginous portion of MCC of 2-day-old mice (Hinton et al. 2009). All these observations strongly suggest that DSPP expression may shift from the most superficial layer to deeper layers over time. Accordingly, the fact that the phenotypic changes in the Dspp null mice essentially happen at the articular and prechondroblastic layer at postnatal 1 and 2 months might be related to the temporal and spatial expression pattern of DSPP in the condylar cartilage. The condylar cartilage of the fetal mouse mandible is derived from periosteum, and the progenitor cells for condylar cartilage rapidly or directly differentiate into hypertrophic chondrocytes (Shibata et al. 1997). In neonatal mice (Hinton et al. 2009; Sun et al. 2010), DSPP is mainly expressed in the articular and prechondroblastic layers, whereas the deletion of Dspp dramatically affected the development of the two layers at the earlier stages. Along with the cell differentiation and development of MCC, the expression of DSPP in the WT mice shifts from the superficial layers into the deeper layers, whereas, in the Dspp null mice, the phenotypic changes remain at the articular and prechondroblastic layers.
As mentioned above, the mitotic cells of the condylar cartilage are located within the perichondrium (specifically, the prechondroblastic layer). It is these undifferentiated cells, rather than the chondrocytes in deeper layers, that proliferate and mature to affect the growth of the MCC. In this study, BrdU labeling was used to compare cell proliferation in the MCC; the results revealed that proliferating cells in the prechondroblastic layer were rarely seen in the Dspp null mice, whereas, in WT mice, proliferating cells were sporadic in the prechondroblastic layer. However, there is no big difference in BrdU labeling between the subchondral bone of Dspp null and that of WT mice. These findings indicate that the loss of DSPP may influence the proliferation of progenitor cells in the MCC. We believe that the decrease in the proliferation of progenitor cells may be a contributing factor for the phenotypic changes in the MCC of Dspp null mice. It was reported that mechanical stress induced by mastication significantly affects the MCC (Enomoto et al. 2010). As the age of postnatal 21 days coincides with weaning and the initiation of mastication, from postnatal 21 days onward, the mandibular condyle and its associated cartilage undergoes remarkable changes in morphology. Thus, it is reasonable to speculate that the defective dentin mineralization in Dspp null mice may more or less affect MCC growth. However, comparison of 1-month-old transgenic mice expressing the Dspp transgene driven by a type I collagen promoter (Zhu et al. 2012) and WT mice demonstrated that DSPP accelerates mandibular cartilage proliferation (data not shown), further indicating a direct role of DSPP in the postnatal development of MCC.
Biglycan, a member of the small leucine-rich proteoglycan (SLRP) family, is considered to be a coordinator of matrix synthesis by proliferating cells, has a pericellular distribution, and its level is elevated in proliferating cartilage (Melrose et al. 2001). In our study, biglycan expression was observed in the superficial layers of condylar cartilage in WT mice, whereas, in the Dspp null condyle, its expression was strikingly decreased (nearly undetectable). The significant downregulation of biglycan in Dspp null mice suggests that DSPP is essential to the synthesis of biglycan in articular cartilage, and this change may be attributed to the altered proliferation of the progenitor cells in the superficial layers of articular cartilage.
Within the ECM, macromolecules interact with one another to build an architectural network. The key players for these interactions are collagens and proteoglycans that organize into various patterns with differing concentrations for individual components (Chen et al. 2006). The major form of collagen, type II collagen, has been used as a representative marker for cartilage. Our data revealed that the distribution pattern of type II collagen was altered in the Dspp null condyle. In addition, the articular and prechondroblastic layers over type II collagen-positive areas were remarkably thinner in Dspp null mice than in WT mice; this observation was consistent with the results from the HE and toluidine blue staining, providing additional evidence that Dspp ablation mainly affects the superficial layers, that is, the articular and prechondroblastic layers of MCC. It has been reported that biglycan interacts primarily with collagen II (Vynios et al. 2001) and that type IX collagen binds covalently to the surface of type II collagen fibrils and appears to anchor them to the surrounding matrix (Helminen et al. 2002). Thus, both biglycan and collagen IX interact with collagen II and may help stabilize the lattice-like architecture of articular cartilage (Chen et al. 2006). In this study, decreases in biglycan and collagen IX were observed in Dspp null mice, and the distribution pattern of collagen II was altered. Taken together, we speculate that the loss of DSPP may lead to changes in collagen fibrillogenesis, matrix assembly, and chondrocyte–ECM interactions.
Type X collagen, a marker for chondrocyte terminal differentiation, is transiently and developmentally regulated (Silbermann and Mark 1990). Type X collagen is presumably involved in the process of mineralization and endochondral ossification (Alini et al. 1994). Our data reveal the upregulation of type X collagen mRNA in the Dspp null mice. Previously, it was demonstrated that type X collagen was elevated in the chondrocytes of osteoarthritic articular cartilage (Walker et al. 1995). In our study, we did not observe any signs of osteoarthritis in the condylar cartilage of the Dspp null mice. It is clear that future studies are needed to explore the possible association between altered expression of DSPP and the onset of osteoarthritis.
In conclusion, the loss of DSPP alters the development of different layers of the MCC. During the development of the MCC, DSPP may be involved in the proliferation of progenitor cells and the regulation of ECM molecule synthesis or assembly in the cartilage. The findings in this study indicate that DSPP is essential for maintaining a healthy condylar cartilage in the mouse mandible.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grant DE005092 (to CQ) from the National Institutes of Health.
