Abstract
The orderly development of the nervous system is characterized by phases of cell proliferation and differentiation, neural migration, axonal outgrowth and synapse formation, and stabilization. Each of these processes is a result of the modulation of genetic programs by extracellular cues. In particular, chondroitin sulfate proteoglycans (CSPGs) have been found to be involved in almost every aspect of this well-orchestrated yet delicate process. The evidence of their involvement is complex, often contradictory, and lacking in mechanistic clarity; however, it remains obvious that CSPGs are key cogs in building a functional brain. This review focuses on current knowledge of the role of CSPGs in each of the major stages of neural development with emphasis on areas requiring further investigation:
Keywords
Introduction
The development of the nervous system relies on a well-orchestrated series of steps that include cellular differentiation, neural migration, axonal and dendritic growth and finally synaptic formation and stabilization. Each of these processes is controlled by the modulation of the genetic program by signals from the cellular environment. Among these signals are proteoglycans, both trans-membrane and in the extracellular matrix, which are increasingly being recognized as a major influence on each of the steps in development. In this review, we will focus on the role of chondroitin sulfate proteoglycans (CSPGs) in these processes, acknowledging that other classes of proteoglycans, including heparan sulfate (HS) 1 and keratan sulfate (KS) 2 proteoglycans are also involved.
The glycosaminoglycan (GAG) chains of CS and its related GAG, dermatan sulfate (DS) are linear polysaccharides covalently attached to Ser residues in the various core proteins through a common linkage region of GlcA-Gal-Gal-Xyl. Additional disaccharide sugars glucuronic acid (GlcA) N-acetyl-galactosamine (GalNAc) are added to the GAG chains by six enzymes which are members of the chondroitin synthase family, including chondroitin synthases (ChSys),3–5 chondroitin-polymerizing factor (ChPF), 6 and CSGalNAcTs.7,8 The sugar backbone is then modified by the sulfation of hydroxyl groups at the C4 and C6 positions of GalNAc and C2 position of the glucuronic acid GlcA residues. Each disaccharide may have a different combination of sulfations, and the nomenclature of disaccharide units with various modifications were proposed by Sugahara and Mikami 9 as the A unit, consisting of GlcA-GalNAc(4-O-sulfate), the C unit, consisting of GlcA-GalNAc(6-O-sulfate), the E unit, consisting of GlcA-GalNAc(4,6-O-disulfate) and the D-unit, consisting of GlcA(2-O-sulfate)-GalNAc(6-O-sulfate). Some of the GlcA residues in a chondroitin backbone are enzymatically epimerized at the C-5 position by dermatan sulfate (DS) epimerases (DSE, DSEL) 10 to produce DS from CS. The disaccharide composition as well as the length of the GAG chain are not genetically determined, leading to a significant degree of diversity. In addition, GAG chains contain specific motifs which bind particular proteins and antibodies,11,12 and are associated with specific biological activities.
The main approaches used to reveal the functions of CSPGs in neuronal development include (1) identification of the spatial and temporal distribution patterns of CS core proteins and changes in sulfated GAGs at critical stages of development using antibodies, (2) degradation of CS GAG chains on the proteoglycans using enzymes such as chondroitinase ABC (ChABC), (3) genetic modification of synthetic enzymes or core proteins, and (4) addition of exogenous CS to interfere with the interaction with CS-binding molecules (Fig. 1). Over the past three decades, these techniques have illuminated the roles of CSPGs in neurogenesis, migration, axon and dendritic extension, axon pathfinding, and synaptic formation and stabilization in various regions of brain, spinal cord as well as peripheral nervous system. In this review, we present the data supporting a role for CSPGs in the mammalian central nervous system (CNS), with the understanding that many CSPGs have a role in more than one of these processes. Table 1 presents a summary of the putative roles of the core proteins in neural development.
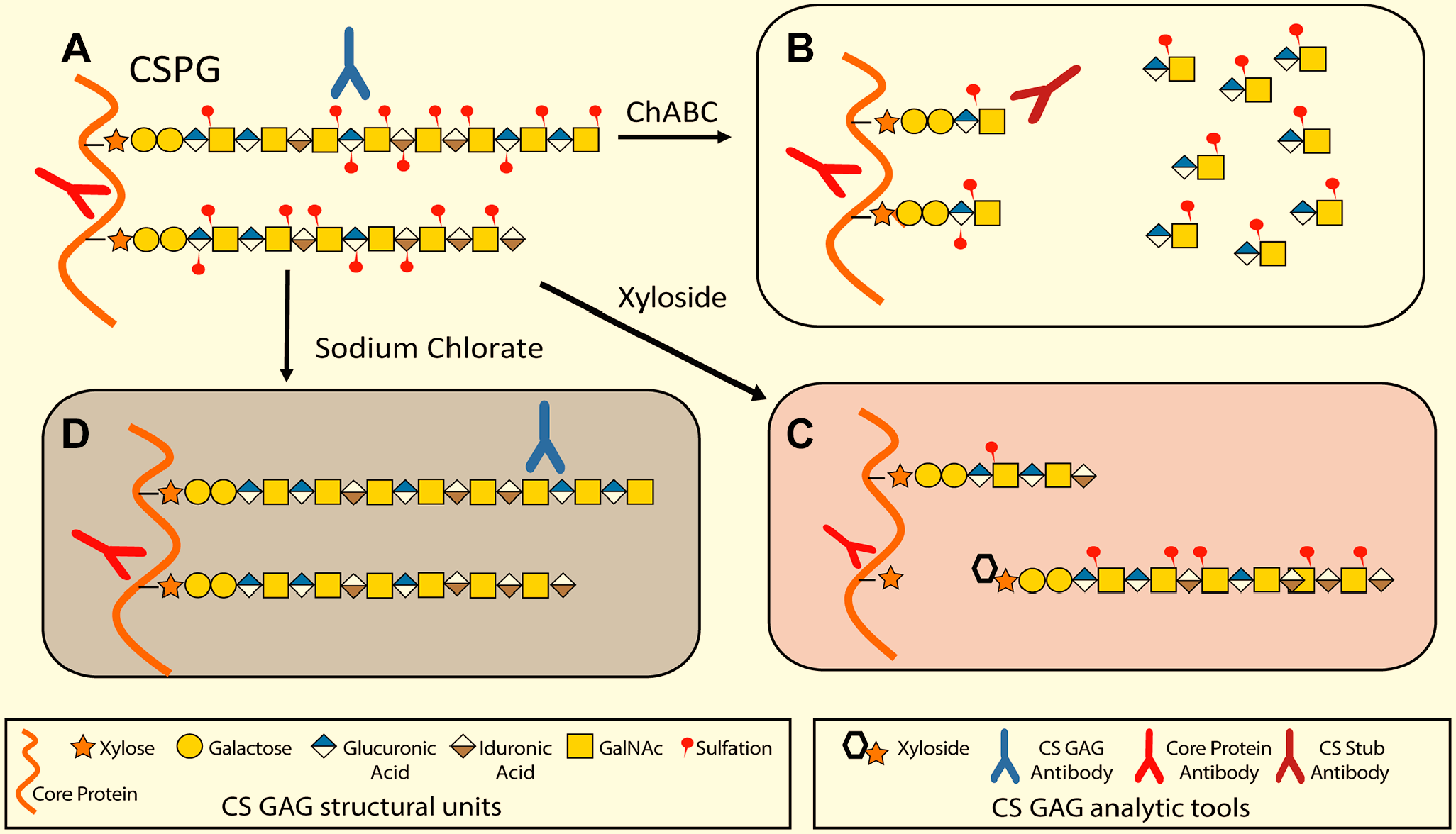
Common methods for GAG modification and CSPG identification. (A) A representative CSPG that contains some DS residues and varied sulfation patterns that can be identified using antibodies directed against epitopes in the GAG chain and core protein. (B) After complete enzymatic digestion by ChABC, GAG chains are reduced to short “stubs” that are recognized by antibodies directed against the core protein or remaining stub. (C) Xyloside treatment results in the formation of GAG chains that are not tethered to a core protein and CSPGs that lack GAG chains or have shortened GAG chains. Core protein antibodies are still effective. (D) Sodium chlorate treatment leads to GAG chains that lack sulfation. Core protein antibodies can still be used in identification, as can anti-CS-O antibodies. Abbreviations: GAG, glycosaminoglycan; CSPG, chondroitin sulfate proteoglycans; DS, dermatan sulfate; CS: chondroitin sulfate.
Proteoglycan Core Proteins in Neural Development.
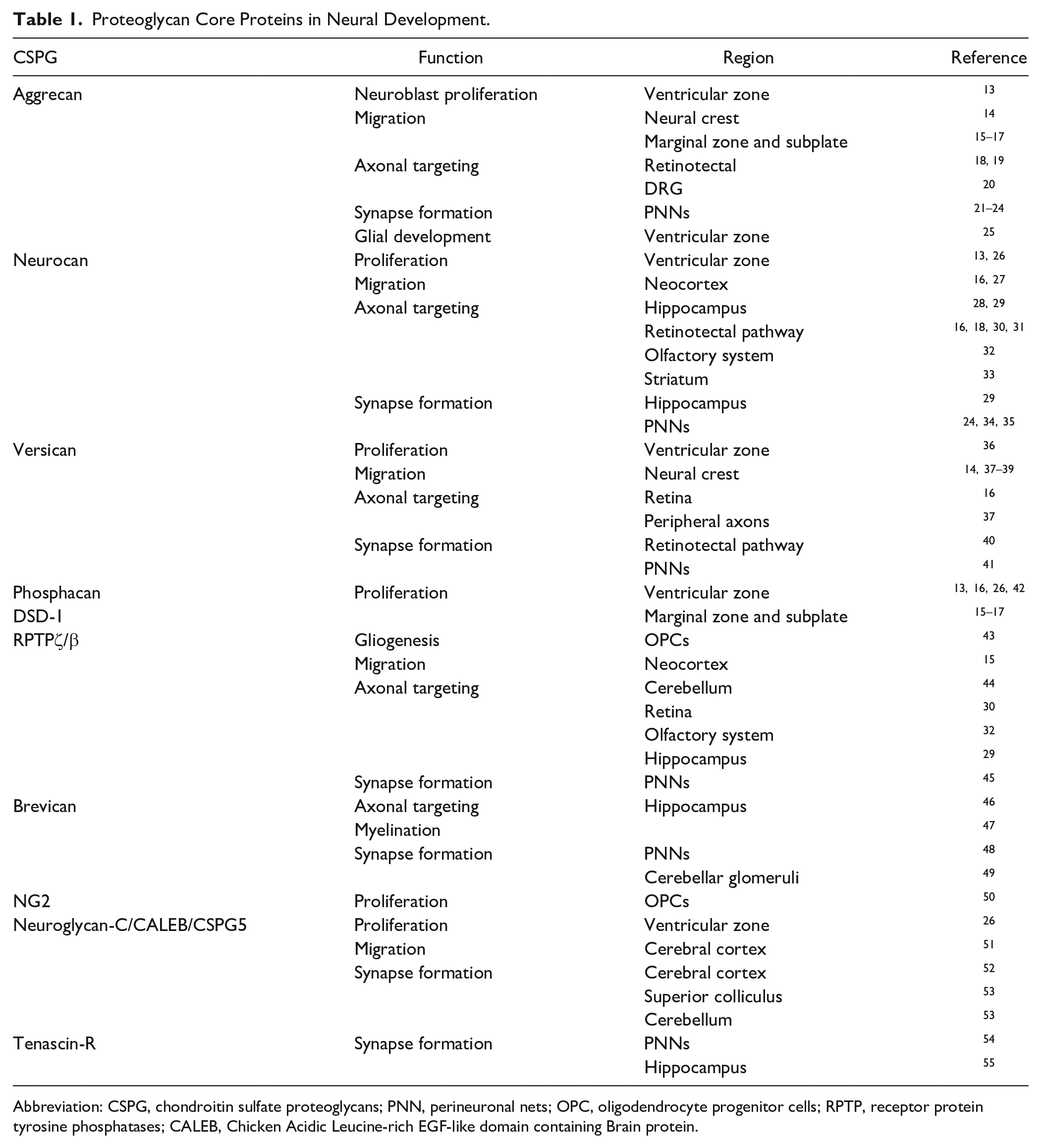
Abbreviation: CSPG, chondroitin sulfate proteoglycans; PNN, perineuronal nets; OPC, oligodendrocyte progenitor cells; RPTP, receptor protein tyrosine phosphatases; CALEB, Chicken Acidic Leucine-rich EGF-like domain containing Brain protein.
Many of the results summarized below which report detection of specific proteoglycan core proteins and sulfation patterns in GAGs depends upon the production and use of monospecific antibodies. Many antibodies have been produced to CSPG GAG chains (Table 2), which has provided insights into our understanding of the role of CSPGs in biological functions. Additionally, there are other anti-GAG chain antibodies that have been used in other biological systems (Supplementary Table 1), which have potential applicability to studies of CSPGs in the nervous system. However, the exact epitopes of many anti-GAG antibodies have not been mapped. Moreover, results with antibodies ostensibly directed against the same sulfation pattern often give different results, likely due to differences in fine structure of their epitopes in the GAG chain.12,56 In addition, many conclusions based on histology have not been verified with functional experiments. These caveats need to be recognized when comparing results from different studies.
Antibodies Shown to React Against Nervous System CS.
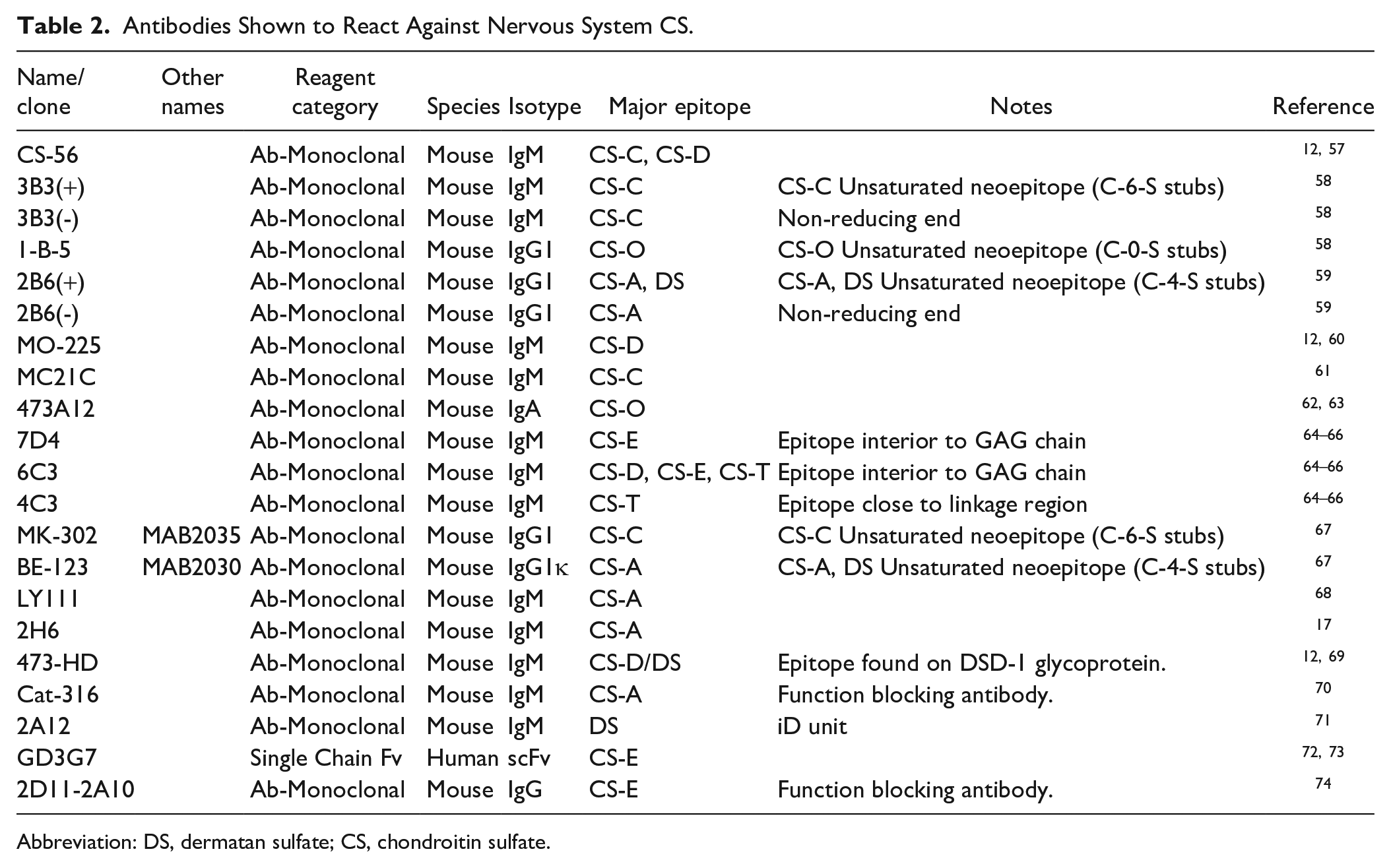
Abbreviation: DS, dermatan sulfate; CS, chondroitin sulfate.
CSPGs in Neurogenesis
Neurogenesis in the mammalian embryo takes place in the ventricular zone (VZ), where highly proliferative epithelial cells differentiate into radial glial cells which then give rise to neuroblasts. 75 CSPGs were first found in the VZ at embryonic day 11(E11) using anti-CS antibody 473HD, which recognizes the DSD-1 epitope on glycosaminoglycan chains of RPTPζ (also known as RPTPβ) as well as phosphacan, which is the spliced extracellular domain of RPTPζ. 13 Others have confirmed the presence of CS GAG chains carrying the DSD-1 epitope using 473HD immunoreactivity and their localization to a mitotically active radial glial population. 76 In culture, these cells could form neurospheres and differentiate into βIII-tubulin-positive neurons. Other studies confirmed the presence of mRNA for CS synthesizing enzymes and core proteins aggrecan, phosphacan, versican, and neurocan in neurosphere cultures.13,26,77–79 Antibodies directed against CS GAG chains and core proteins detected both in neurospheres26,36 as well as in the embryonic VZ. 26
The role of CS GAG chains in controlling the proliferation of neurospheres was investigated by the addition of ChABC. While some reported a reduction in proliferation after ChABC, 80 others did not. 36 Direct injection of ChABC into the VZ at E13 reduced neurosphere formation, and addition of ChABC into neurosphere cultures reduced proliferation in response to fibroblast growth factor (FGF)-2, confirming a role for CSPGs in vivo. 81 This suggests that CS GAGs may be acting indirectly by serving as co-receptors for the growth factors that promote proliferation (Fig. 2).
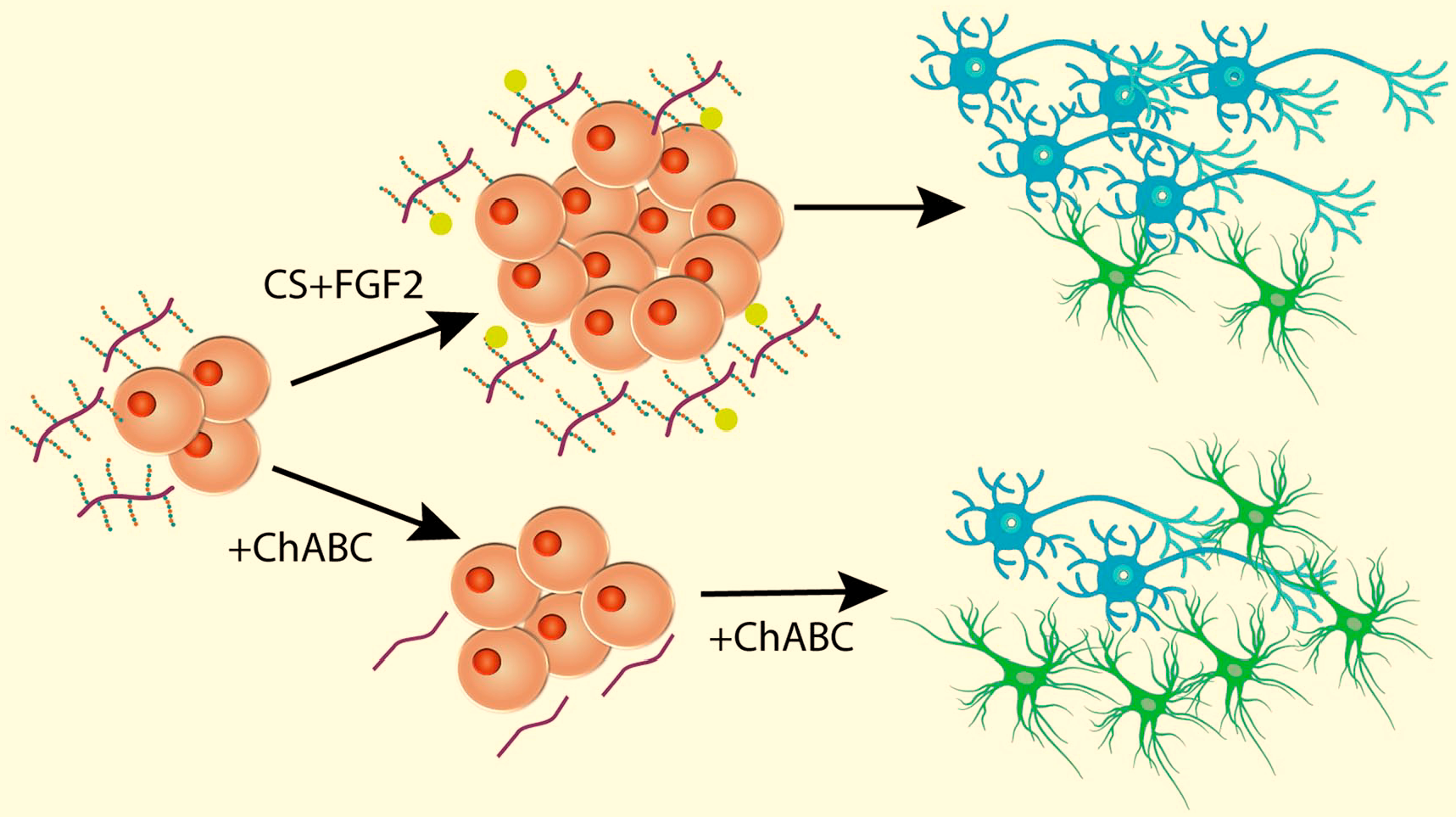
CSPGs affects proliferation and differentiation of neural stem cells. Adding CSPG along with FGF2 causes an increase in proliferation of neuronal stem cells that differentiate into a higher percentage of neurons vs. glia. Removal of CS using ChABC results in reduced proliferation and a higher percentage of glia vs neurons. After Ida et al. 26 and Sirko et al. 82 Abbreviation: CSPG, chondroitin sulfate proteoglycans; FGF, fibroblast growth factor.
Certain sulfation patterns have been associated either with the inhibition 83 or promotion 84 of neural regeneration after injury; supporting the idea that specific patterns may interact with specific receptors to alter function. Sodium chlorate, which inhibits GAG chain sulfation by preventing the synthesis of the sulfate donor 3’-phosphoadenosine 5’-phosphosulfate (PAPS), 85 reduced the proliferation of neurospheres from the VZ, 78 and reduced proliferation and neuronal differentiation in spinal cord neurospheres. 86 This supports the importance of GAG chain sulfation in mediating these effects; however, as PAPS reduction affects sulfation of many molecules as well as GAGs besides CS, this does not conclusively link the effects to CS GAGs alone. Proliferation of neurospheres in response to FGF-2 was increased by the addition of highly sulfated CS GAGs CS-D and CS-E as well as DS along with FGF-2. 26 Moreover, neurospheres derived from mice with a deletion of the dermatan-4-sulfotransferase gene (Chst14) proliferated more slowly than wild type mice 87 providing further confirmation of the importance of specific CS sulfation motifs in controlling neurosphere proliferation.
While the VZ is the source of proliferating neuroblasts during development, the subventricular zone (SVZ) is one site identified by Allen 88 as having continued proliferative activity into the adult, later confirmed by several others using more modern techniques.89–91 The first hint that CSPGs might be involved in neuroblast proliferation in the SVZ was from a study by Gates, et al., 92 that showed a high intensity of CSPG staining in the adult SVZ. Further examination showed that this region also stained with 473HD. This region gives rise to the rostral migratory stream (RMS) from the SVZ to the olfactory bulb. CSPG immunoreactivity as well as BrdU uptake by neuroblasts has been demonstrated in the RMS, 93 implicating CSPGs in neuronal migration as well as proliferation. Once neuronal precursors reach the olfactory bulb, they migrate radially to their final position. This process continues in the adult. Interestingly, tenascin-R levels increase in the olfactory bulb with age, and tenascin-R knockout mice have an impairment of radial migration in the adult but not during embryogenesis. 94 Adult neurogenesis also takes place in the dentate gyrus (DG) of the hippocampus, 95 and newly generated neurons are thought to be involved in learning and memory. 96 A recent study demonstrated the presence of CS in the DG, and that levels of CS increased in animals exposed to an enriched environment. Moreover, ChABC treatment reduced the density of neural progenitor cells in the DG and the enriched environment hastened the recovery of CS and neurogenesis. A further requirement for CS was demonstrated by a reduced neurogenesis and a reduction in cognitive memory in animals with a deletion of chondroitin sulfate N-acetylgalactosaminyltransferase 1 (CSGalNAcT1), which is essential for CS synthesis. 97
The mechanisms by which CS GAGs increase neurosphere proliferation and survival are not clear. Some early data suggested that CS GAGs act through the potentiation of growth factor actions,26,81,98 as has been reported for HS GAGs. 99 However, later experiments revealed that there are two major classes of receptors for CS GAGs: the type IIa receptor protein tyrosine phosphatases (RPTPs), leukocyte antigen related (LAR) also known as RPTPF, RPTPσ, and RPTPδ 100 and the Nogo receptors (NgRs) NgR1 and NgR3. 101 Both RPTPσ and LAR have been identified in the developing nervous system, including the SVZ and VZ,102–104 as has NgR1. 105 Adding the NgR ligand Nogo66 increased neurosphere proliferation in wt animals but not NgR1-/- animals and adding either the RPTPσ or NgR blocking ectodomains reduced proliferation. 105 However, because these blocking experiments were done in the absence of exogenous ligands, it is not clear which receptor is mediating the actions of CSPGs.
CSPG in Neuronal Migration
Following neurogenesis, neurons migrate from the ventricular and SVZs to their destination within the CNS. 106 Projection neurons are born in the VZ and demonstrate a multipolar morphology and move in an indiscriminate manner into the intermediate zone and the SVZ. Once the multipolar neurons arrive at the subplate they convert to a bipolar shape and adhere to radial glial fibers before radially moving to the cortical plate into the marginal zone. 107 In contrast, interneurons move from the SVZ to the medial ganglionic eminence (MGE) and then migrate tangentially in the marginal zone before migrating radially into the cortical plate. The movement patterns of both excitatory and inhibitory neurons indicate that each cortical layer contains important factors which control neuronal migration.
The change in morphology of projection neurons in the subplate from multipolarity to bipolarity is an essential feature of proper migration. 108 The involvement of CSPGs in this process was first suggested by studying GAG localization in E12-E14 wild type (wt) and reeler mutant mice. 109 These studies showed that both the marginal zone and the subplate of a developing wt mouse cerebral cortex are rich in GAG chains, whereas reeler mutant mice, which have an inversion of their cortical layers, 110 demonstrated high levels of GAG chains in the subplate. Biochemical analysis did not reveal any difference in the composition of the CS GAGs between wt and reeler animals. 111 The presence of CS GAGs in the subplate was later confirmed using antibodies directed against CS. 112 Later studies demonstrated high levels of CSPG core proteins—for example, neurocan and phosphacan are highly expressed in the marginal zone and subplate.15–17 Additionally, versican has also been found in these regions. 18 While CS GAGs form boundaries to growing axons (see below), a direct effect on migration through the subplate was excluded because neurons in the subplate were not impeded in their radial migration. Direct tests of the role of these CSPGs in migration are necessary to supplement the anatomical observations.
CSPGs, such as phosphacan, neurocan and versican interact with a diverse variety of proteins including neural cell adhesion molecules (NCAM), fibronectin, laminin and tenascin-C,113–116 as well as growth factors including pleiotrophin and the related protein midkine.117–121 These interactions have been implicated in supporting the migration of immature projection neurons to their final cortical layer along radial glia. A prominent role in migration has been found for pleiotrophin, which is expressed by radial glial fibers, which binds to the transmembrane proteoglycan RPTPζ (as well as phosphacan).122,123 RPTPζ is found on migrating pyramidal neurons.15,124,125 In vitro cell migration assays 126 showed that pleiotrophin binding, which results in the suspension of RPTPζ’s tyrosine phosphatase activity,127,128 prompts the migration of cortical neurons. 125 Moreover, pleiotrophin-induced cortical cell migration was suppressed by RPTPζ inhibitors and sodium vanadate, a tyrosine phosphatase inhibitor, as well as by phosphacan (and other free chondroitin sulfate). 125 Pleiotrophin also binds to the HS syndecan3 (SDC3), 129 and deletion of SDC3 impairs radial migration. 130 Similarly, pleiotrophin, as well as midkine, bind to neuroglycan-C (NGC),126,131 a part-time proteoglycan also known as CSPG5 132 and CALEB (Chicken Acidic Leucine-rich epidermal growth factor [EGF]-like domain containing Brain protein), 133 and knockdown of NGC produces a neuronal migration defect. 51 Because the binding of pleiotrophin to NGC is both through the GAG chains and protein core, 131 whether migration defects are due to interactions with pleiotrophin, and whether they depend upon CS GAG chains, is an open question.
Protein/GAG interactions are dependent upon the degree of sulfation of the GAG chain, with higher affinities correlating to greater degrees of sulfation.117,134 This is also true for GAG/pleiotrophin interactions.117,121 Moreover, deleting either uronyl 2-sulfotransferase and GalNAc4S-6ST in utero (which reduce the production of 2,6- and 4,6-sulfated chondroitin) resulted in impaired radial migration of cortical neurons. 135
As noted above, inhibitory cortical interneurons are generated in the ventral telencephalon, specifically in the MGE, caudal ganglionic eminence and preoptic area. They then migrate tangentially to the cortex. The interneurons migrating to the neopallium avoid the striatum, which expresses high levels of CSPGs along with the repulsive molecule Semaphorin3A (Sema3a), 136 by moving through the marginal zone, subplate, or lower intermediate/SVZs. Sema3a binds to CS-E, 137 and both blocking the Sema3a receptor Nrp1 or removing CS with ChABC promoted the entry of cortical interneurons into the striatum in brain slices. The combination did not increase migration, suggesting that Sema3a and CSPGs act in concert to control interneuronal migration. 136 CSPGs, likely aggrecan, are also found in apposition to migrating serotonergic neurons. 138
Additional chemorepulsive factors include Sema5a, Ephrin a3, Ephrin a5, and Slit 1; whereas glial cell line-derived neurotrophic factor (GDNF), NT-4, and neuregulin-1 act chemoattractive factors for cortical interneurons.121,139–145 These factors all bind to both CS and HS, which indicates that there is a likelihood that both CS and HS control their activity, and many co-localize with GAGs.136,145 However, further studies that provide direct evidence of their involvement are needed. Though interneurons avoid the CSPG-rich striatum they do migrate into the marginal zone and subplate, which both highly express CSPG. They are both rich in neurocan, phosphacan, and versican.15–17 However, cortical interneurons also move through the lower intermediate zone and SVZ—both of which express CSPG at a low rate.
CSPGs are also involved in the migration of neural crest cells which give rise to the peripheral nervous system (Fig. 3). As neural crest cells migrate laterally from the neural tube in avians, they avoid areas which are rich in CSPGs.146,147 In particular, in the chick, as they migrate in the ventral migratory pathway, crest cells avoid the posterior half of somites which are stained with antibodies against 6-sulfated GAGs148,149 and versican.37,150 Addition of exogenous CS or β-D-xyloside to prevent GAG chain elongation disrupted neural crest migration, 151 as did implantation of aggrecan. 14 Experiments in Xenopus laevis embryos demonstrate that the confining action of versican is required for neural crest cell migration, as migration was significantly reduced after versican knockdown. 38 A similar distribution of CS and migration pattern was found in the mouse, and treatment of mouse embryos with either sodium chlorate to prevent GAG chain sulfation or β-D-xyloside allowed migrating crest cells to enter the posterior sclerotome. 152 Overexpression of versican in the splotch mouse results in defective neural crest migration. 153 In culture, both CS-A and CS-C inhibit the migration of chick neural crest cells. 147 Thus, these inhibitory effects are due to the actions of the sulfated GAG chains.
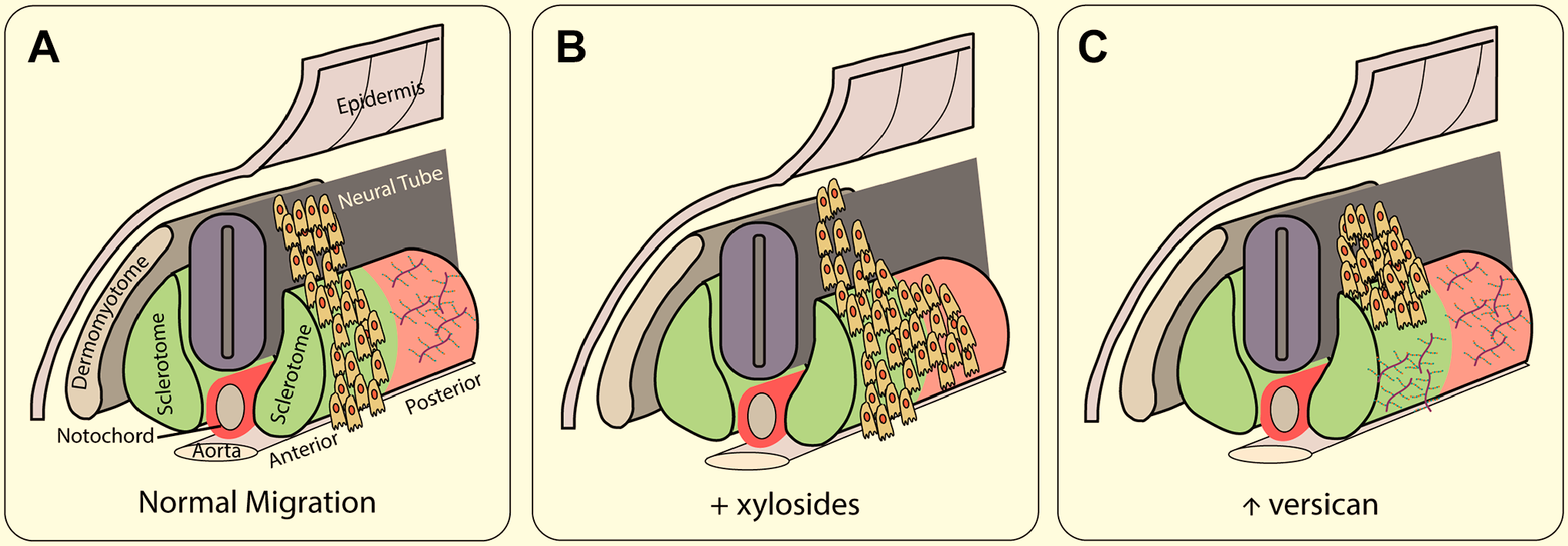
CSPGs help direct migration of neural crest cells. (A) In normal migration, cells migrate from the neural tube over the anterior portion of the sclerotome, seen in green, while avoiding the posterior, seen in red, which contains high levels of versican. (B) The removal of CS chains using ChABC leads to cells migrating in both the anterior and posterior sclerotome. (C) The addition of exogenous CSPG blocks neural crest cell migration. After Oakley and Tosney, 149 Landolt et al., 37 Moro Balbas et al., 151 Perissinotto et al. 14 Abbreviation: CSPG, chondroitin sulfate proteoglycans.
CSPGs in Axon Guidance
The roles of CSPGs as being inhibitory to axon outgrowth has received considerable attention, primarily through the use of tissue culture models, the results of which are extensively reviewed elsewhere. 154 In vivo, the role of CSPGs on axonal elongation and pattern formation have been intensely studied in the visual system. At early developmental stages, there is a near uniform distribution of CS throughout the retina, with low levels of CS found near the dorsal fissure. This same area contains the axons from the earliest differentiated ganglion cells.155,156 With time, the expression of CSPGs in the rat retina diminishes from the center, remaining at the outer edge, with ganglion cell axons occupying the area free of CS. 155 Removal of CS in the retina using ChABC resulted in ectopic localization of retinal ganglion cells (RGCs) with their axons oriented randomly in all directions. 156 Addition of CS to cultured retinas also resulted in disrupted ganglion cell localization and axon outgrowth, 156 suggesting that CS plays an important role in RGC differentiation and axonal extension. A spatial and temporal expression of the CS epitope at the chiasm and in the optic tract was also demonstrated in mouse embryos using CS-56 staining, 157 and a similar distribution of CS, including CS-A and CS-C was found in the xenopus optic tract. 158 Removal of CS chains by ChABC digestion resulted in disrupted axon navigation crossing the midline, failure of turning into the ipsilateral optic tract for the normally uncrossed projections, and a loss of chronologic fiber organization in the optic tract.30,157
On their way to the tectum, retinal axons extend along the diencephalon-telencephalic boundary avoiding invading anteriorly to the telencephalon. 31 CSPGs in the telencephalic cells were found responsible for delimiting the anterior border of optic tract as removal of CS chains by ChABC led to an anterior enlargement of the optic tract, 159 but elimination of endogenous CS GAGs did not change the overall trajectory of retinal axons in the optic tract. 158 A restricted distribution of CS-D revealed by MO-225 antibody staining was found in the diencephalon-telencephalic boundary and the neuropil encircling the optic tract in chick embryos, suggesting that CS-D might be involved in delimiting the border of the optic tract and chronological sorting of the retinal axons. 160 A potential role of CSPGs in guiding retinal axons as they grow through the diencephalon toward the optic tectum was also reported in the developing visual system of Xenopus laevis. 158 CSPGs are expressed within the diencephalon of Xenopus embryos along the optic tract, and the addition of exogenous CS GAGs caused axon pathfinding errors evident by a wider optic tract and aberrant invasion of retinal axons into inappropriate regions of the diencephalon and telencephalon (Fig. 4). In the lateral geniculate nucleus (LGN), aggrecan appears to control the timing of entry of corticogeniculate (CG) axons: aggrecan levels are high in the dorsal LGN at birth, at a time when retinogeniculate axons enter the LGN, but CG axons do not. The entry of CG axons proceeds as aggrecan levels decrease. Moreover, genetic deletion of aggrecan is accompanied by premature entry of CG axons. 19
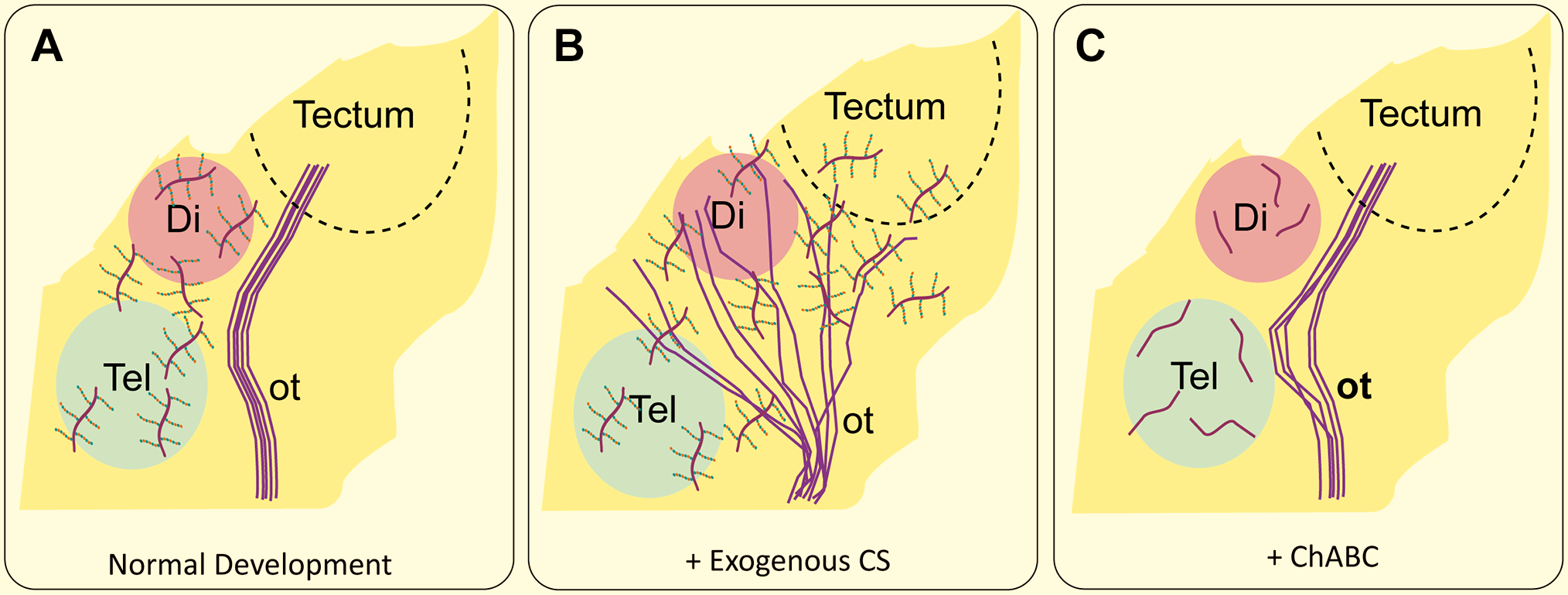
CSPGs serves as guidance cues for optic tract axons during development. (A) In normal development, the optic tract avoids the CSPG-rich areas of the telencephalon and the diencephalon and grows into the optic tectum. (B) When exogenous CS is added, retinal axon targeting is disrupted, and axons grow across the entire area. (C) When CS is removed using ChABC, the optic tract still terminates in the tectum, however it deviates slightly toward the telencephalon. After Walz et al., 158 Tuttle et al., 31 Ichijo and Kawabata. 159 Abbreviation: CSPG, chondroitin sulfate proteoglycans; ot, optic tract; Tel, telencephalon; Di, diencephalon.
In the newborn hamster, CS immunoreactivity was strongly concentrated in the midline of the tectum at the time that retinal axons are entering the tectum, which suggests that CSPGs act as a barrier to prevent optic axons from crossing the midline toward the contralateral tectum. Interestingly, the higher level of CS immunoreactivity in midline than in lateral tectum did not correspond to a higher level of CSPG core proteins, but an increased rate of synthesis or sulfation of CS chains. 161 These results may be species dependent, as growing retinal axons in the chick appear to pass through CS-rich areas in the retina, optic tract and tectum.162,163 Another possibility is that all these areas are rich in other extracellular matrix (ECM) molecules, some of which, like laminin, antagonize CS inhibition. 164
There are numerous other examples of CSPGs that form axon boundaries or barriers influencing axon navigation during development. In the developing spinal cord, Oakley and Tosney 149 found high levels of CS immunoreactivity and peanut agglutinin binding in several areas that are avoided by growing spinal motor axons, including the posterior sclerotome, the limb bud and roof plate of the spinal cord. Many of these same areas express high levels of versican. 37 In culture, explants of embryonic chick notochord repelled growing DRG axons, and this activity was eliminated by the addition of ChABC. 20 CSPGs in the notochord also form a barrier to motor neuron axons in zebrafish, as treatment with ChABC caused aberrations in spinal motor nerve trajectories. 165
CSPGs also appear to restrict olfactory axon projections. At the earliest, in both the chick, and rat and mouse, phosphacan and CS staining was found surrounding olfactory bulb glomeruli as they form, confining nascent axons.166–168 At later stages, phosphacan staining is found around the olfactory nerve as it extends, consistent with a role for phosphacan in shaping the guidance of the olfactory nerve.32,167,169 In contrast, neurocan is associated with growing olfactory axons and was reported to promote olfactory neurite outgrowth in culture. 32
The role of CSPGs in the developing cerebral cortex are more complex. Studies of the development of cortico-thalamic and thalamo-cortical neurons demonstrate that both sets of neurons pass through areas rich in CS-56, such as the preplate, suggesting that these early projections do not see CS as a barrier. 170 However, these CS-rich areas also contain other ECM molecules that could interact with CS GAG chains. 170 The expression of ECM, including CSPGs, along the anterior commissure follows a spatially and temporally regulated pattern correlating with the growth and guidance of commissural fibers in hamster embryos. CSPGs are distributed at the midline region before commissural axons approach and cross it. During the crossing stage, CSPGs formed a special tunnel surrounding the axon bundle through which the axons elongate. After anterior commissure axons cross the midline, the expression of CSPG declines gradually and disappears in the midline region, whereas it begins to distribute laterally in the tissue between the anterior and posterior limbs of anterior commissure defining their boundaries. 171 A similar pattern is found during the development of the corticospinal tract, where there are high levels of CS immunoreactivity appearing to form a boundary to growing axons. The level of CS drops temporally as axons reach their targets in the spinal cord, perhaps allowing for exit. 172
During hippocampal axon tract formation, neurocan and phosphacan showed distinct temporal and spatial expression patterns,28,29 with neurocan appearing earlier to confine the mossy fiber tract, while 6B4/phosphacan was higher in the adult hippocampus. 29 In the developing striatum, neurocan was found surrounding patches of intense dopaminergic innervation early in development. 33 Both neurocan and anti-CS-O antibodies showed a similar distribution, with disappearance at later times after innervation was complete. 173 In the cerebellum, the expression of phosphacan/6B4 proteoglycan and CS chains also showed a developmentally regulated pattern that closely followed the formation of the cerebellar mossy fiber system. 44 Similarly, 473HD staining of the DSD-1 proteoglycan is found in P7 cerebellum in all areas except the external granule layer. 174 Using antibody 3F8 to another region of phosphacan, it was demonstrated that phosphacan is also localized to the developing molecular layer and around adult parallel fibers, suggesting that it serves to prevent granule cell axons from entering the molecular layer, and also promoted fasciculation of parallel fibers. 175
In many cases, CSPGs appear to have an indirect role in axonal growth and pathfinding through organizing differential matrix-bound cues and modulating the interaction between axons and specific guidance. Embryonic thalamic neurons display layer-specific attachment and neurite outgrowth when plated onto cultured slices of mouse forebrain, which is correlated with the timing and pattern of thalamocortical innervation.176,177 The subplate/intermediate zone promotes thalamic neuron attachment and supports neurite outgrowth, while the cortical plate inhibits thalamic neuron attachment and repels their neurites. This layer specificity was abolished either by enzymatic removal of endogenous CS GAGs or by addition of soluble exogenous CS GAGs. These opposing inhibitory and stimulatory activities were attributed to distinct CS-binding proteins present at different layers, rather than the differential expression of CSPGs. 177 In an ex-vivo Xenopus brain preparation, addition of a mixture of CS-A and CS-C perturbed the normal trajectory of a subpopulation of embryonic forebrain NOC-2 axons within the tract of the optic commissure. 178 Instead of crossing the ventral midline, the axons stalled or grew inappropriately into the dorsal midbrain or continued extending longitudinally within the ventral longitudinal tract. Considering CSPGs do not display a specific distribution pattern along this pathway, it was postulated that CS interfered with navigation from specific guidance cues. 178 Another example is that sensory axons of the trigeminal nerve have a stereotypical growth pattern to get to their targets: they normally project to rhombomere 2 and are excluded from rhombomere 3. This behavior is reproduced in experiments where trigeminal neurons were grown on section of wild-type mouse heads; however trigeminal neurons entered rhombomere 3 on heads from mice with a deletion of ErbB4, a receptor for neuregulin 1. In addition, the aberrant behavior was reproduced after treatment of the preparation with ChABC, excess chondroitin 6-sulfate or hypertonic saline. Because ErbB4 did not directly inhibit axon growth, they concluded that the barrier by rhombomere 3 is due to an effect of CS-C on ErbB4. 179 In the development of the limbic system, axons in the fasciculus retroflexus project along the boundaries between prosomeres 1 and 2, both of which express Sema5a. Function blocking antibodies to Sema5a allowed altered axon trajectories, with axons inappropriately entering prosomere 2. However, in culture Sema5a alone was found to be an attractive guidance cue, while membranes from prosomere 2, which contain Sema5a, were repulsive. The attractive actions of Sema5a were abolished by heparanase, suggesting an involvement of HS. In contrast, CS was found to be high in prosomere 2, and the addition of ChABC allowed axons to enter prosomere 2. Furthermore, the addition of CS to the prosomere 1 membranes preparations changed them from attractive to repulsive. 145 Treatment with ChABC also alters the trajectory of commissural fibers from the vestibular nucleus. 180
CSPGs in Synapse Formation and Stabilization and Myelination
Synapse formation and maintenance are the final critical aspects of neuronal development. The induction, elimination, and stabilization of synapses is a complex process that involves multiple signaling cascades, cytoskeletal reorganization, and external stimulation. This cell–cell communication is reliant on both internal cell factors as well as interactions between the cells and the ECM. Proteoglycans, especially CSPGs and heparan sulfate proteoglycans (HSPGs), are one of many players involved in the formation and maintenance of these critical neural connections.
During cerebellar development, both PTPζ and pleiotrophin were found expressed abundantly in the Bergmann glia cells surrounding developing Purkinje cells, implying a role of Pleiotrophin-PTPζ signaling and Bergmann glia–Purkinje cell interaction in the morphogenesis of Purkinje cell dendrites. 181 Indeed, in organotypic slice cultures of rat cerebellum, disruption of the pleiotrophin-PTPζ interaction by adding antibodies against the extracellular domain of PTPζ, degradation of CS chains, or by addition of exogenous CS chains (CS-C, -D, and –E, but not CS-A), led to abnormal dendritic growth with multiple and disoriented primary dendrites. 181 The underlying mechanism might involve the glutamate-transporting activity of GLAST on the processes of Bergmann glia. Further studies using monoclonal antibodies against different CS epitopes showed that the MO-225 epitope, which recognizes CS-D, was deposited around Purkinje cell surfaces and was colocalized with phosphacan immunoreactivity. 182 Treatment of cerebellar slices with chondroitinase ABC or CS-D significantly reduced the amount of pleiotrophin. These data suggest that CS-D on the RPTPζ/phosphacan is responsible for mediating the interaction with pleiotrophin.
An inhibitory role of CSPGs in synapse formation and maintenance is supported by data from both in vitro and in vivo experiments. Dendritic spines are the sites of synaptic input. In culture, brain-derived neurotrophic factor (BDNF) promotes spine formation on cortical neurons, and addition of CSPGs antagonizes the effect of BDNF. CSPGs also reduce the phosphorylation of the BDNF receptor TrkB, and the effects of CSPGs on spine formation and TrkB phosphorylation were eliminated in RPTPσ-knockout mice. 183 In vivo, neurons from RPTPσ-knockout mice show an increased dendritic spine density and length, 184 but whether CSPGs are involved was not investigated. Astrocyte-derived ECM, which includes CS 185 increases synaptic formation.186,187 In a co-culture system of astrocytes and hippocampal neurons, the number of excitatory, but not inhibitory, synaptic puncta was increased after inclusion of ChABC or hyaluronidase, both of which degrade CS, consistent with an inhibitory action of CS on synapse formation. 188 Dendritic spine dynamics are also regulated by CS, as treatment of organotypic hippocampal slice cultures with ChABC enhanced spine motility 189 In contrast, versican has been reported to promote presynaptic maturation of retinotectal synapses. RGCs growing on versican in vitro displayed increased presynaptic vesicle-rich varicosities as compared to axons growing on polylysine/laminin, and this effect was not found after ChABC treatment. In vivo, depletion of versican in the tectum via RNAi resulted in smaller vesicle rich puncta in RGC axons. 40
CSPGs are also involved with the stabilization of synapses, with a large portion of research focusing on their presence in perineuronal nets (PNNs) (Fig. 5). Formation of the PNN has been shown to be temporally regulated, appearing late in development, commonly associated with the ending of critical periods of neuronal plasticity.190,191 Enzymatic destruction of PNNs has been shown to reactivate limited plasticity and can shift spines into a state of enhanced filopodial motility that is associated with finding new synaptic partners.21,191 This increase in plasticity can alter ocular dominance, fear memory resilience, and enhance long-term recognition memory. 192
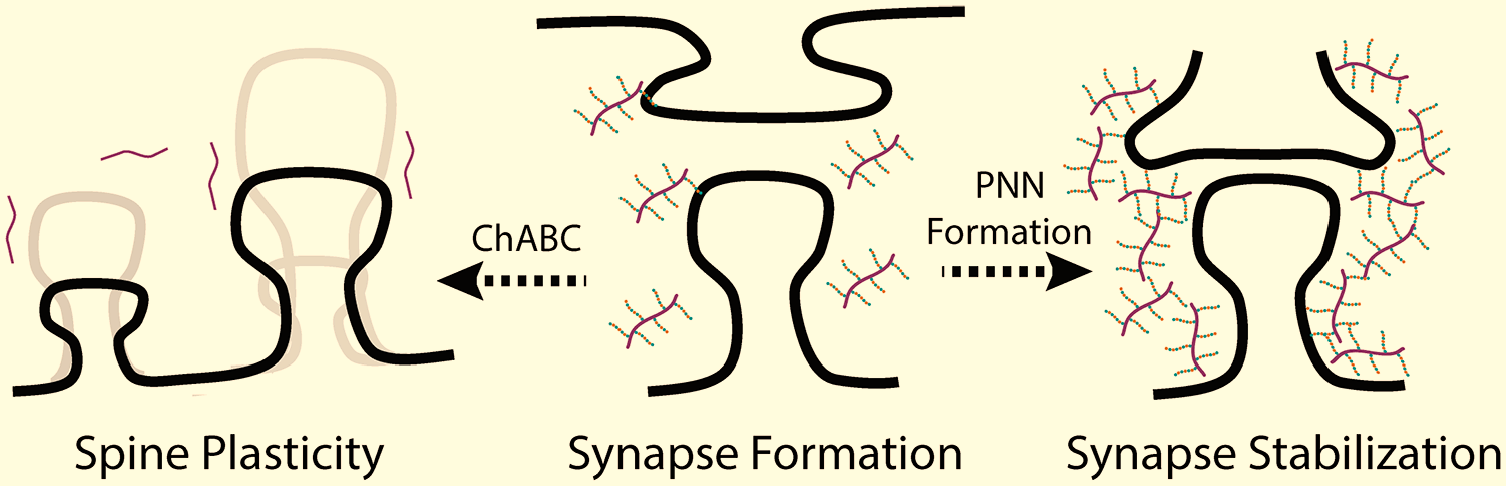
Increased chondroitin sulfate leads to reduced synaptic plasticity. Synapse formation and dendritic spines are sensitive to the presence of CSPGs. Enzymatic degradation of chondroitin sulfate by ChABC leads fluctuations in dendritic spine size and number which facilitates increased synapse plasticity. With development, there is an increased presence of CSPGs in perineuronal nets, which restrict synapse plasticity. Abbreviation: CSPG, chondroitin sulfate proteoglycans.
Components of PNNs include hyaluronic acid (HA), tenascin-R, link proteins, and CSPGs, including brevican, phosphacan, aggrecan, neurocan, and versican.15,21–23,34,41,45,48,54,193 HA binds to CSPGs through link proteins to help retain CSPGs and create the PNN. Treatment with ChABC or hyaluronidase destroys PNNs. 21 Mutants that lack PNN components such as tenascin-R, brevican and neurocan show a transient increase in synapses but result in overall compromised synapse formation and stabilization. 35 The composition of PNNs in rats appears to be similar in both the brain and spinal cord. 21 The formation of PNNs varies in a spatiotemporal manner with some brain nuclei forming PNNs earlier than others indicating possible structural variations and differing maturation speeds of brain regions. 194 This might be a function of neuronal input during PNN formation, which can affect overall PNN composition.23,195 CSPGs also play roles in synaptic stabilization other than through PNNs. In the cerebellum, brevican produced by astrocytes is enriched in glomeruli, 49 implying a role in synapse stabilization formation. Neuroglycan-C is found in perisynaptic membranes in the cerebral cortex, 52 while animals with a deletion of NGC show alterations in synapse formation and function in the colliculus 53 and cerebellum. 196 Interestingly, the GAG chain composition of NGC changes with aging, with a decrease in the percentage of 6-sulfated GAGs and an increase in the level of 4-sulfated GAGs, similar to what is observed in PNNs. 197
Myelination of axonal tracts is essential to provide for rapid transmission of signals. In the CNS, myelination is due to the wrapping of axons by processes of mature oligodendrocytes. 198 During development, oligodendrocytes develop from a population of oligodendrocyte progenitor cells (OPCs), characterized by their expression of the NG2 CSPG and PDGFRα. 199 Developing oligodendrocytes in the hippocampus transiently express NG2, also known as CSPG4 and melanoma chondroitin sulfate proteoglycan (MCSP), a transmembrane protein with a single GAG attachment site. 200 In culture, NG2 has an inhibitory role on neuronal process outgrowth, due to both the single GAG chain and the core protein.201,202 While NG2 has been used as a marker of OPCs, a functional role for NG2 in the nervous system has only been revealed in the NG2-knockout mouse. 203 In culture, knockout cells have a reduced sensitivity to PDGFα, 203 supporting a functional interaction. 50 In vivo, the number of OPCs and their rate of proliferation is significantly reduced. 204 However, there are no major anatomical differences from wild type mice: they mice have almost normal myelin, 204 no difference in hippocampal neurogenesis, 205 and there was no difference in the formation of somatosentory barrels in mice. 206 Developing oligodendrocytes in the hippocampus transiently express brevican, but a functional role for brevican has not been established. 46 The role of the CS GAG chain in any of these effects has yet to be explored.
In this review we explored the role of CSPGs in the development of the nervous system, with an emphasis on neurogenesis, migration, axon guidance and synapse formation. CSPGs are important for proliferation while also having been shown to slow differentiation in neurogenesis. During neural migration, they are known to affect changes in cell polarity and either induce or block migration. CSPGs in axon guidance are often inhibitory cues, serving as boundaries or barriers to growth. Finally, in regard to synapses, proteoglycans affect the formation, maturation and stability of neuron–neuron communication. The involvement of CSPGs in these aspects is wide-spread, complex and at times contradictory. While some aspects have been the subject of intense study, such as the proteins in PNNs, other avenues of research still require a more in-depth understanding. This especially pertains to the mechanisms of action behind many of the observed outcomes and the importance of the GAG chain and CS sulfation pattern in the spatio-temporally sensitive process known as neural development. This review serves to provide a snapshot of our current understanding of how CSPGs influence this process and serve as a springboard to try to further our understanding of the intricacies of proteoglycans in the brain.
Supplemental Material
2020-00130R1_Production_Supplemental_Data_online_supp – Supplemental material for The Role of Chondroitin Sulfate Proteoglycans in Nervous System Development
Supplemental material, 2020-00130R1_Production_Supplemental_Data_online_supp for The Role of Chondroitin Sulfate Proteoglycans in Nervous System Development by Caitlin P. Mencio, Rowan K. Hussein, Panpan Yu and Herbert M. Geller in Journal of Histochemistry & Cytochemistry
Footnotes
Author Contributions
All authors have contributed to this article as follows: planning (HG), manuscript drafting (CPM, RPH, PY, HMG), and all authors have read and approved the final manuscript.
Competing Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Division of Intramural Research of the National Heart, Lung, and Blood Institute, NIH.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
