Abstract
Inter-α-trypsin inhibitor (IαI) family members are ancient and unique molecules that have evolved over several hundred million years of vertebrate evolution. IαI is a complex containing the proteoglycan bikunin to which heavy chain proteins are covalently attached to the chondroitin sulfate chain. Besides its matrix protective activity through protease inhibitory action, IαI family members interact with extracellular matrix molecules and most notably hyaluronan, inhibit complement, and provide cell regulatory functions. Recent evidence for the diverse roles of the IαI family in both biology and pathology is reviewed and gives insight into their pivotal roles in tissue homeostasis. In addition, the clinical uses of these molecules are explored, such as in the treatment of inflammatory conditions including sepsis and Kawasaki disease, which has recently been associated with severe acute respiratory syndrome coronavirus 2 infection in children:
Keywords
Introduction
Since its discovery in the 1960s, the precise functional role of inter-α-trypsin inhibitor (IαI) has been a bit of a mystery as there are no known IαI deficiency syndromes and probably due to its critical role in female reproductive biology.1–3 Despite displaying serine protease inhibitory activity, 4 the need for this activity in serum was unclear given that serum contains an excess of serine protease inhibitory capacity, suggesting that IαI was a redundant serum protease inhibitor. More recent studies on IαI have revealed its localization in many tissues with diverse roles in cell regulation and matrix integrity (Table 1) in both health and disease.5,6 Although the liver is recognized as the main source of IαI,7–12 many other tissues synthesize IαI family components, including the kidney, 13 reproductive tissues, 14 lung, 15 connective tissues,16–18 and central nervous system19,20 (Table 2). Much research focus has been placed on the roles of IαI in pathology, and notably in inflammation, whereas its physiological roles, apart from those in ovulation, are less explored. Recent evidence for the diverse roles of IαI in both biology and pathology is reviewed and gives insight into the pivotal roles this unusual molecule plays in tissue homeostasis and the emerging roles in diagnostics and therapeutics.
Roles of Bikunin, HCs, and the IαI Family.
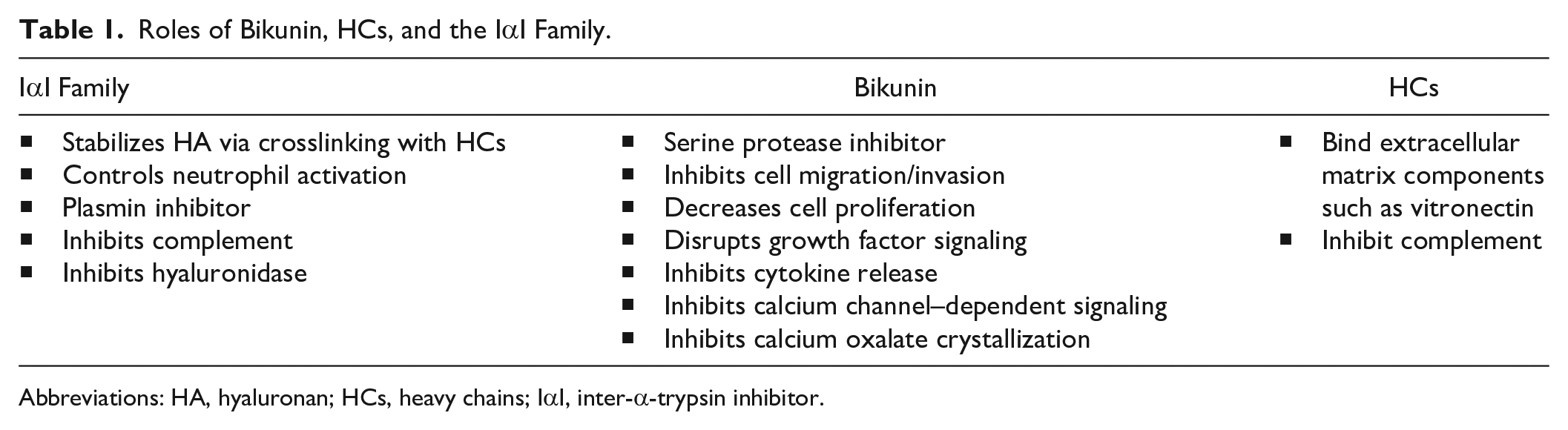
Abbreviations: HA, hyaluronan; HCs, heavy chains; IαI, inter-α-trypsin inhibitor.
Summary of Expression and Localization of IαI Family Components.
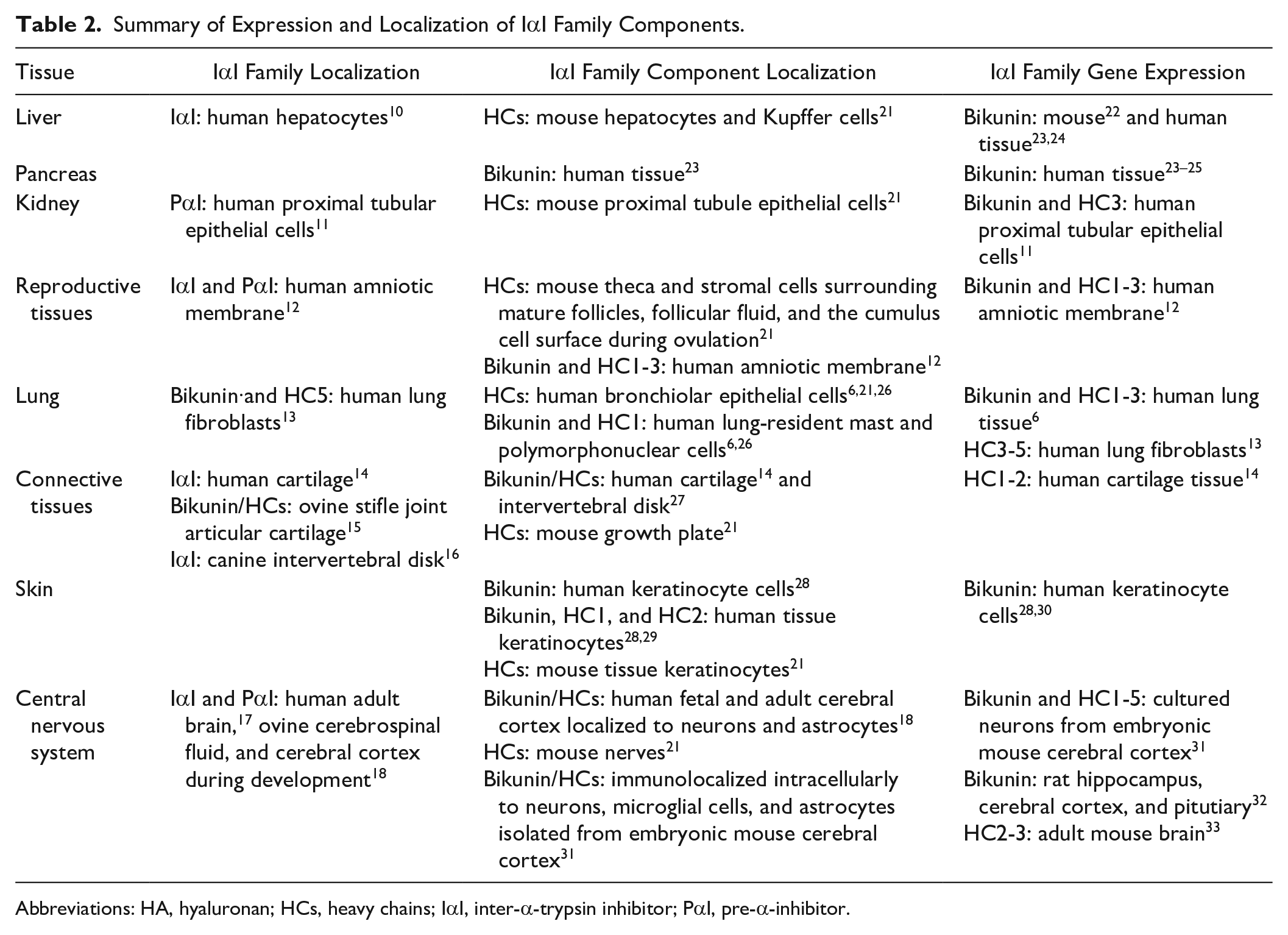
Abbreviations: HA, hyaluronan; HCs, heavy chains; IαI, inter-α-trypsin inhibitor; PαI, pre-α-inhibitor.
Overview of IαI
Structure of IαI
IαI is an approximately 225-kDa complex containing bikunin and two heavy chain (HC) proteins, designated HC1 and HC2 (Fig. 1). 34 IαI is present in many fluid compartments including blood, peritoneal, amniotic, cerebrospinal, and synovial fluids, whereas bikunin is also excreted in the urine.23,28,35 IαI is also present in many tissues including the liver, pancreas, kidneys, ovary, amniotic membrane, lung, connective tissues, skin, and brain, suggesting broad physiological roles.22,24,26,30,31,36–38
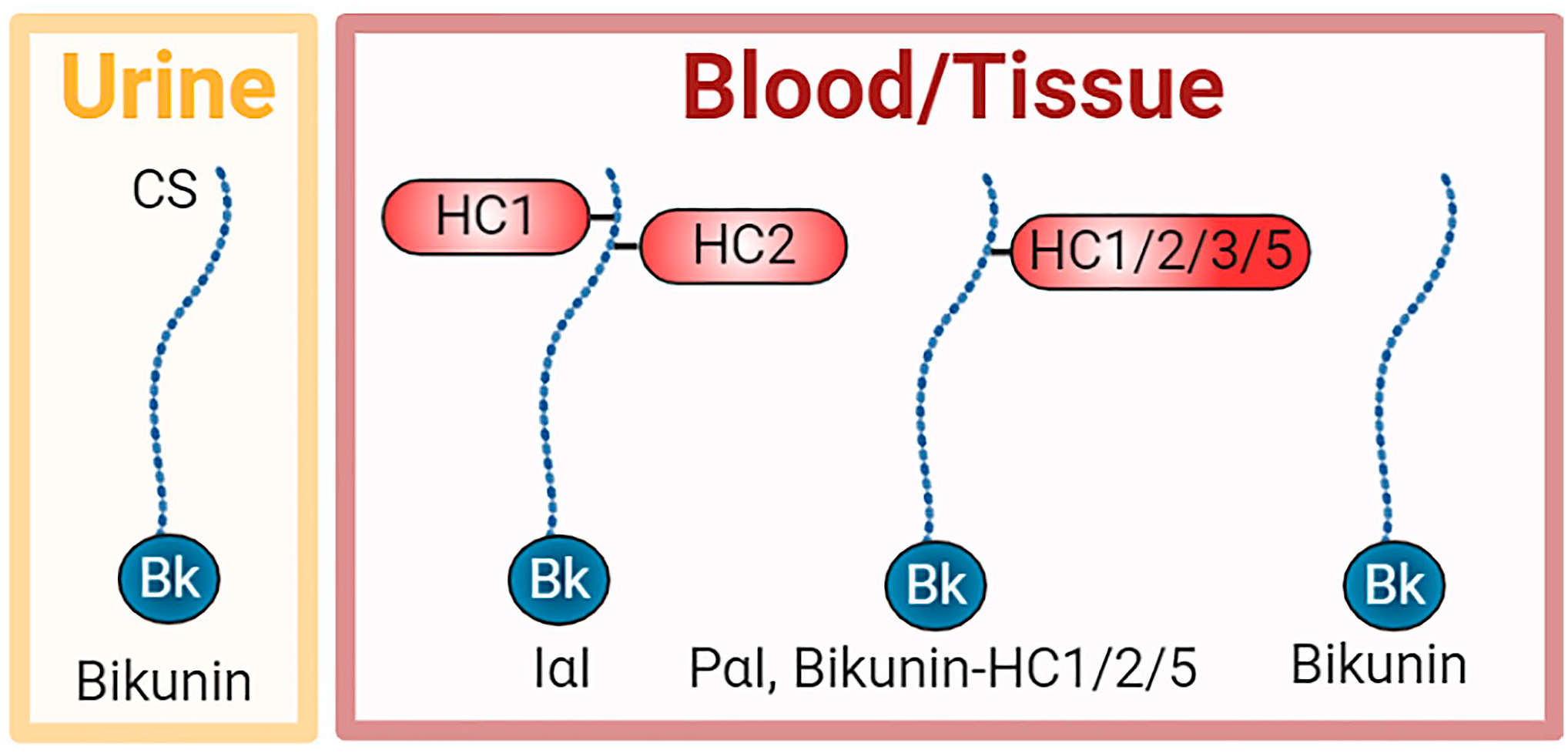
Schematic of members of the IαI family in urine, blood, and tissues. Bikunin is found alone in the urine, whereas it is also found in the blood and tissues either alone or complexed with one HC, including HC1, HC2, HC3, or HC5. HC3 covalently bound to bikunin is called pre-α-inhibitor (PαI). In addition, the predominant form is IαI, a complex of HC1 and HC2 covalently bound to the CS chain of bikunin. Abbreviations: CS, chondroitin sulfate; HC, heavy chain; IαI, inter-α-trypsin inhibitor.
Bikunin is a proteoglycan with a chondroitin sulfate (CS) chain attached to the protein core of approximately 20 kDa. HC1 and HC2 are similar in size at approximately 85 kDa. Unusually, these HCs are covalently attached to the CS chain. HC1 and HC2 are located close together on the CS chain with HC2 positioned closer to the bikunin core protein. 39 Bikunin·HC1 and bikunin·HC2 conjugates have also been detected in the circulation and tissues, whereas bikunin alone has been detected in both the circulation and urine.40,41 A related structure is pre-α-inhibitor (PαI) which is a complex of bikunin with HC3 (~90 kDa) covalently attached to the CS chain in a similar way as HC1 and HC2. More recently, bikunin complexed with HC5 has also been reported (Fig. 1). 15
The biosynthesis of IαI is unusual with regulation at many levels, including protein synthesis of three discrete proproteins, CS chain biosynthesis, proprotein cleavage, and assembly of the complex. The AMBP gene encodes both α1-microglobulin and bikunin. The α1-microglobulin/bikunin proprotein is posttranslationally modified on serine-10 of the bikunin protein core with a CS chain via well-established biosynthetic pathways.42,43 This is followed by the covalent attachment of HC1 and HC2. These HCs are transcribed by the ITIH1 and ITIH2 genes and their C-terminal prodomains cleaved by furin before covalent attachment to the bikunin CS chain via ester bonds between the C-terminal aspartic acid residues in the HCs and C-6 hydroxyls of internal N-acetyl galactosamine (GalNAc) residues in the CS chain in the trans Golgi.42,44–49 However, the mechanisms controlling the covalent attachment of both the number and type of HCs to the CS chain remain to be elucidated. The last step before secretion involves cleavage between α1-microglobulin and bikunin by an unknown enzyme, but hypothesized to be a furin-like protease.50,51 Although the transcription of the α1-microglobulin/bikunin proprotein is unusual, there is evidence that the coexpression of α1-microglobulin provides a chaperone role in the correct folding of bikunin. 52
Little is known about the factors that promote the expression of IαI components; however, they are thought to possess tissue-specific regulation pathways. 53 Regulation of ambp gene expression in the liver involves the transcription factor binding to the 5′ flanking region of the gene 54 ; however, such regulation has not been explored in other tissues. In addition, ambp gene expression in the liver can be regulated by glutamine and cell swelling, 55 as well as interleukin (IL)-6, leukemia inhibitory factor, and retinoic acid, and potentiated by dexamethasone in rat hepatocytes. 56 Furthermore, IL-6 can increase bikunin expression in macrophages. 57 The gene expression of IαI components in acute inflammatory states is not regulated in a coordinated way, as only the HC3 gene is upregulated, whereas the bikunin and HC2 genes are downregulated and the HC1 gene remains unaffected. 58
Early structural analysis of the CS chain indicated that it contained both unsulfated and 4-sulfated CS disaccharides, with the sulfated disaccharides concentrated at the reducing end.39,59 More recently, with the advent of different purification protocols and sources of bikunin, it was discovered that the CS chain is variable in both chain length and degree of sulfation and can contain unsulfated, monosulfated, and disulfated disaccharides.39,60–62 In addition, the linkage region can be variably sulfated.59,62–69
Roles of IαI
The most widely acknowledged role of the IαI family members is the transesterification of HCs from IαI to hyaluronan (HA) in the presence of tumor necrosis factor–stimulated gene-6 (TSG-6), a process that results in the covalent modification of HA with HCs (HA·HC)70–72 and recently reviewed in detail. 73 This process is conserved across mammals, birds, and reptiles. 74 It requires that HCs are present as part of the IαI family members for the transfer to HA, because HCs in the bikunin knockout mouse are present in their precursor form, but are unable to complex with HA. 2 It has been suggested that the energy for this reaction is contained within the ester bond between the HC and CS formed during biosynthesis. 71 During the transesterification process, the HCs become covalently attached to HA via an ester bond from their C-termini to the C-6 hydroxylated GlcNAc residues in HA, and thus analogous to the linkage to CS in IαI.41,75,76 The transesterification process requires the presence of TSG-6 and divalent cations.71,73,77–80 Self-association of HCs bound to HA likely enables crosslinking via a cation-dependent mechanism.81,82 This crosslinking is reported to be dynamic with TSG-6 able to reversibly transfer HCs to bikunin and between HA chains when present in serum whereby high-molecular-weight HA can act as both an acceptor and a donor of HCs.83–85 In addition, HA oligosaccharides can accept HCs,86,87 and there is also evidence that they do not donate them. 85 Although the function of HA oligosaccharide·HC complexes has not been reported, high-molecular-weight HA·HC complexes provide anti-inflammatory properties, including polarization of macrophages to the anti-inflammatory M2 phenotype. 88 When the HA·HC complexes were synthesized either in vivo or in vitro by cells in culture, they could support leucocyte adhesion89,90; however, when these complexes were synthesized in vitro using isolated components, they did not support the adhesion of CD44+ cells, 91 suggesting that the formation of these HA·HC complexes in more complex biological fluids involved additional components that together mediated cell adhesion. In addition, pentraxin-3 (PTX3) is essential for the formation of a crosslinked HA network in the cumulus cell oocyte complex92–94 where it crosslinks HA·HC complexes through its interactions with the HCs. 95 This suggests that distinct tissue environments may contain HA·HC complexes with different associated molecular components and possibly even distinct functions.
The CS chain attached to bikunin is essential for the transesterification of its pendant HCs to HA in the formation of HA·HC.46,80 In addition, the structure of the CS chain modulates the extent of HA·HC formation, with more highly sulfated CS chains supporting a greater extent of HA·HC formation than lower sulfated CS, 61 suggesting a mechanism for control of this process.
Other lesser explored roles of IαI include its ability to bind multiple extracellular matrix components, support stem cell expansion, control neutrophil activation, inhibit complement, and impart protease and hyaluronidase inhibitory activity. For example, transglutaminases can catalyze the covalent attachment of IαI onto fibrinogen and also crosslink proteins in a plasma clot. 96 IαI interacts with molecules that act as integrin ligands such as fibronectin and vitronectin, and the recent crystal structure of HC1 has revealed that HCs resemble integrin β-chains containing a von Willebrand Factor A domain with a metal ion–dependent adhesion site (MIDAS) motif and an associated hybrid domain. 81 However, in the case of HC1, surprisingly the interaction with fibronectin and vitronectin is not mediated by the von Willebrand Factor A domain, is not metal-dependent, and does not involve the Arg-Gly-Asp (RGD) motifs of these ligands. 81 IαI binding to vitronectin promotes epithelial adhesion, migration, and proliferation. 97 In addition, IαI supplementation of the defined medium supports the expansion and long-term maintenance of pluripotent stem cells, circumventing the need for the use of ill-defined preparations such as Matrigel or immobilization of cell adhesive proteins/peptides such as vitronectin. 98 IαI also controls neutrophil activation via a reduction in both reactive oxygen species production and adhesion to vascular endothelial cells. 99 IαI inhibits activation of the complement system, including the classical, lectin, and alternative complement pathways and specifically the processing of factor B that forms part of the alternative pathway C3 convertase.100,101 Whereas bikunin is known to possess protease inhibitory activity, IαI can inhibit plasmin and this effect can be potentiated by binding to TSG-6, which is further enhanced by the interaction of TSG-6 with heparin and heparan sulfate.102,103 In addition, IαI family members, in particular PαI, may have hyaluronidase inhibitory activity. 104
Roles of Bikunin
Bikunin is involved in a wide variety of processes as demonstrated by gene analysis of the bikunin knockout mice which revealed that in the absence of bikunin there was a dysregulation of genes associated with stress, apoptosis, proteases, signaling molecules, aging, cytokines, HA metabolism, and female fertility.2,3,105 Mice deficient in bikunin exhibit reduced female fertility,2,3 although the applicability of these findings to humans has not been established. In addition, these mice exhibit a higher frequency of spontaneous lung metastasis, 106 which correlates with the low level of bikunin in the urine of patients with some cancers.107,108 Mice deficient in bikunin also display an increased anxiety-like behavior. 109 Interestingly, overexpression of bikunin has not been reported or associated with human pathology.
The earliest role of bikunin was ascribed to its two Künitz domains that possess protease inhibitory properties against several serine proteases, including leucocyte elastase and cathepsin G, pancreatic trypsin and chymotrypsin, plasmin, plasma and tissue kallikrein, and some of the coagulation cascade proteinases.110–113 This antiprotease activity is thought to support the growth of endothelial cells and fibroblasts.7,114 Interestingly, bikunin itself can be cleaved within the Künitz domains by mesotrypsin. 115
Bikunin is also recognized to exhibit a range of cell signaling functions mediated at the cell surface via interactions with cartilage link protein (HAPLN1), an HA binding protein, and an as-yet-unidentified protein named the bikunin receptor.5,116 Notably, bikunin inhibits cell invasion via inhibition of transforming growth factor (TGF)-β1 expression, which in turn inhibits selected mitogen-activated protein (MAP) kinase signaling pathways including Src-, MEK-, and ERK-dependent urokinase-type plasminogen activator (uPA) and uPA receptor expression.117–121 Bikunin can reduce cell proliferation via disrupting the heterodimerization of CD44 with growth factors, resulting in the suppression of receptor-mediated MAP kinase signaling. 122 In addition, bikunin can inhibit the dimerization of CD44 v9-containing isoforms, which in turn inhibits cell interactions with HA and consequently CD44/HA-mediated activation of MAP kinase signaling. 123 Bikunin can also inhibit cytokine release and nuclear translocation of nuclear factor-κB to inhibit apoptosis.124–127 Bikunin blocks calcium channels, inhibiting both the contraction of vascular smooth muscle cells and the calcium-dependent TGF-β1 signaling cascade.121,128,129 Although bikunin itself can be upregulated via IL-6 such as macrophages, it can inhibit the production of proinflammatory cytokines.48,57,130 In addition, bikunin can inhibit the formation of calcium oxalate crystals. 131
Roles of the Heavy Chains
Interestingly, six HCs have been reported with differential expression in tissue development and pathology.132,133 The roles of these HCs have not been established by mouse knockout studies; however, HC1-5 gene expression is reduced in multiple human solid tumors including breast, lung, and kidney, 25 suggesting a role for these proteins in tissue homeostasis.
HC1-3 is synthesized with C-terminal prodomains that are cleaved before their covalent attachment to the bikunin CS chain.34,44,46,47 HC4 does not contain the conserved cleavage site present in HC1-3, suggesting that HC4 attachment to either the bikunin CS chain or HA is unlikely.15,34 HC5 can covalently bind to the CS chain on bikunin, whereas HC6 has only been identified at the gene level.15,134
HCs can interact with molecules such as vitronectin and fibronectin, suggesting a more diverse role in extracellular matrix interactions beyond HA.81,97 HC1 binds to vitronectin in vitro, 81 and IaI potentiates cell adhesion to immobilized vitronectin.97,135 In addition, IaI supports vitronectin-mediated epithelial cell repair in an injury model involving cell adhesion, migration, and proliferation. 97 These findings suggest that HCs interact with proteins such as vitronectin and fibronectin in the extracellular space and may act as a linker to HA for broader interactions in the extracellular matrix to influence the biological activity of these molecules. However, whether these interactions functionally involve HCs in IαI family complexes, bound to HA or alone, remains to be elucidated. Recently, HC1 has been shown to bind directly to complement C3 via a MIDAS and inhibit the activity of the alternative pathway C3 convertase, which has a central role in the amplification of this component of innate immunity. 81
Roles of IαI in Biology
Components of the IαI family have been localized in many tissues such as the liver, kidney, pancreas, skin, lung, ovary, amniotic membrane, central nervous system, and connective tissues (Table 2), as well as multiple fluid compartments—most notably, blood.22–24,26,28,30,31,35–38 However, the presence of these components in IαI family complexes remains to be explored in some tissues as do their tissue-specific roles during homeostasis. HA matrix crosslinking roles for IαI family members have been explored during ovulation, in the amniotic membrane and gut asymmetry during intestinal morphogenesis.14,92,93,136 However, many other potential roles of these molecules in more broad extracellular matrix interactions, control of cell signaling, and matrix degradation (Table 1) remain to be explored and will likely reveal a myriad of roles of these ubiquitous molecules. This is particularly the case for tissues such as the pancreas, kidney, lung, connective tissues, skin, and central nervous system described below where IαI family member complexes and/or components have been reported without further exploration of their tissue-specific functions during homeostasis.
Liver, Pancreas, and Kidney
The liver constitutively produces IαI where it is passed into the circulation ready to infiltrate into tissues to exert its functions.7–11 HCs have been immunolocalized to both hepatocytes and Kupffer cells 21 (Fig. 2A), and the liver is found to predominantly secrete IαI at the protein level. 12 The high expression of the ambp gene in the liver during mouse development also suggests that the liver is the predominant source of IαI. 13 Both the human liver and pancreas express the bikunin gene, whereas bikunin at the protein level is also present in the human pancreas.25,30,31 In addition, isolated mouse β-islet cells express genes for bikunin as well as HC1 and HC2, and these IαI components have been colocalized with both HA and TSG-6. 137 TSG-6 is constitutively expressed by β-islet cells, suggesting it is possible that HA·HC forms a part of the normal islet matrix. The kidney, and specifically the proximal tubule epithelial cells, also constitutively expresses bikunin at both the gene and protein levels with predominantly bikunin·HC formed, and although the type of HC in this complex has not been determined, there is evidence that these cells express HC3 at the gene level.13,138,139 In addition, HCs have been immunolocalized to these cells (Fig. 2B). 21
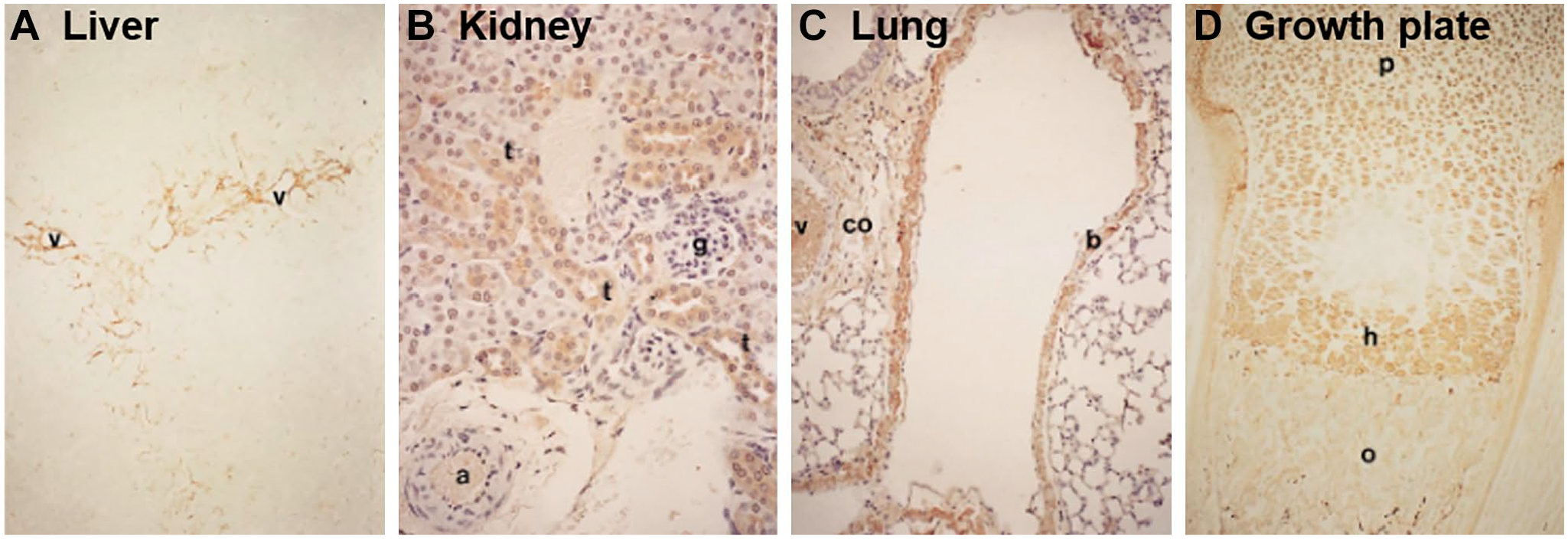
Immunolocalization of heavy chains (HCs) in adult mouse tissues: (A) liver, (B) kidney, (C) lung, and (D) growth plate. HCs were detected via immunohistochemistry using a polyclonal antibody raised against human serum inter-α-trypsin inhibitor and reactivity to mouse HCs, and specific staining is shown in brown. Annotations for tissue features include the following: a, artery; b, bronchiolar epithelial cells; co, connective tissue; g, glomerulus; h, hypertrophic zone; o, ossification; p, proliferative zone; t, proximal convoluted tubular cells; v, vessel. Images shown at 100× magnification. Reprinted with permission from Kobayashi et al. 21
Reproductive Tissues
IαI components are localized in the ovary, including in the theca and stromal cells surrounding mature follicles as well as the follicular fluid, and on the cumulus cell surface during ovulation, where IαI required for ovulation is derived from the circulation.3,21,140 Just before ovulation an HA-rich matrix forms around the oocyte, driving expansion of the cumulus cell oocyte complex; this “cumulus” matrix is stabilized by HA·HC complexes involving the transesterification of HCs from IαI and PαI onto HA catalyzed via TSG-6, where PTX3 is essential for the formation of a crosslinked HA network.92–94 Here, PTX3 directly crosslinks HA·HC complexes through its interactions with the HCs; however, the molecular details of how the formation of these higher order HA·HC/PTX3 complexes is regulated are not yet fully understood. 95 Weaker interactions between HCs are also likely to contribute to the stabilization of the cumulus matrix. 81 The deposition and stabilization of HA around oocytes are necessary for fertilization. This has been demonstrated by reduced fertility in female mice deficient in bikunin, PTX3, or TSG-6 and associated with defects in the formation of HA·HC complexes and crosslinking by PTX3.2,3,70,93,141 Furthermore, these defects were rescued by the exogenous addition of each of the molecules that were knocked out.2,70,93 In addition, this HA matrix is soft, but elastic, and thought to assist in both oocyte transport in the oviduct and sperm capture. 142
Bikunin and HC1-3 have been immunolocalized to the amniotic membrane epithelium and stromal cells/matrix and colocalized with both HA and TSG-6. 14 In addition, analysis of the tissue revealed the formation of IαI and PαI complexes, whereas gene-level analysis of isolated amniotic membrane epithelial and stromal cells revealed that these cells expressed the genes for bikunin and HC1-3, consistent with the local expression of IαI and PαI. 14 TSG-6 is also constitutively expressed in the amniotic membrane, suggesting that the HA·HC/PTX3 complexes found in this tissue 28 may be synthesized from endogenous components. Interestingly, only HC1 is reported to be present in the complexes, which is surprising given our current understanding of the biochemistry of HA·HC formation. 14 This matrix is reported to exert anti-inflammatory and anti-scarring properties via control of cell signaling processes. 143 Bikunin is also involved in the early phase of pregnancy in the conceptus attachment to the uterine luminal surface. 144
Lung
HCs have been immunolocalized to the bronchiolar epithelium (Fig. 2C).8,38,21 In addition, HC2, HC3, and bikunin have been localized to lung tissue–resident polymorphonuclear cells, whereas both HC1 and bikunin have been localized to lung tissue–resident mast cells.8,38 In addition, the human mast cell line, HMC-1, expresses the bikunin gene, suggesting that mast cells have the potential to secrete bikunin. 26 Recently, human primary lung fibroblasts were reported to express the genes for HC3-5 as well as both bikunin and HC5 at the protein level in a complex that was sensitive to chondroitinase ABC digestion consistent with HC5 covalently bound to the bikunin CS chain. 15 HC5·HA complexes were also found to be synthesized by these cells in the presence of TSG-6 following stimulation with TGFβ1 and were linked to the phenotypic change of fibroblasts to myofibroblasts. 15 These data suggest a role for IαI family members, and HA·HC complexes, in normal wound healing.
Connective Tissues
HCs have been localized to the lacunae of chondrocytes in the hypertrophic zone of the mouse growth plate with an identical localization pattern as HA (Fig. 2D). 21 This might suggest a matrix crosslinking role during endochondral ossification.126,145 In addition, HCs have also been immunolocalized to chondrocytes located between the superficial and middle zones in the mouse, whereas HC1 and HC2 were present in adult human cartilage and immunolocalized to the lacunae of chondrocytes in the superficial zone as well as the cartilage surface.16,21,27 HC1 and HC2 genes are expressed in human cartilage, whereas the expression of bikunin gene was absent. 16 IαI was detected at the protein level in these extracts, and it was suggested that bikunin and IαI present in the tissues were derived from the circulation and enter the tissue via the synovial fluid. 16 HCs/bikunin has been immunolocalized throughout the matrix of human intravertebral disk, 27 whereas canine intravertebral disk tissue extracts contain IαI. 18 More recently, bikunin has been identified in ovine stifle joint articular cartilage extracts in a complex of ~120 kDa consistent with the presence of bikunin·HC complexes, although the type of HC present was not reported. 17
Skin
Components of IαI have been localized to keratinocytes in the epidermis,21,24 with HC1, HC2, and bikunin shown to colocalize in this region (Fig. 3). 29 HC1, HC2, and bikunin have also been localized to the melanocytes and Langerhans cells in the epidermis, as well as fibroblasts, surrounding blood vessels, and perivascular lymphocytic cells in the dermis. 29 In addition, bikunin is expressed on the cell membrane of outer root sheath cells and hair bulb cells.24,29 Analysis of human keratinocytes also confirmed that these cells express bikunin at both the gene and protein levels.24,26 In addition, gene expression of HC5 has been identified in human dermal fibroblasts and the dermis. 133 The constitutive expression of TSG-6 in skin may suggest a role for these molecules in HA matrix crosslinking; however, the presence of IαI family complexes required to take part in the process remains to be established.29,146 HA·HC complexes are, however, likely present in normal skin, 146 supporting the presence of IαI family complexes.
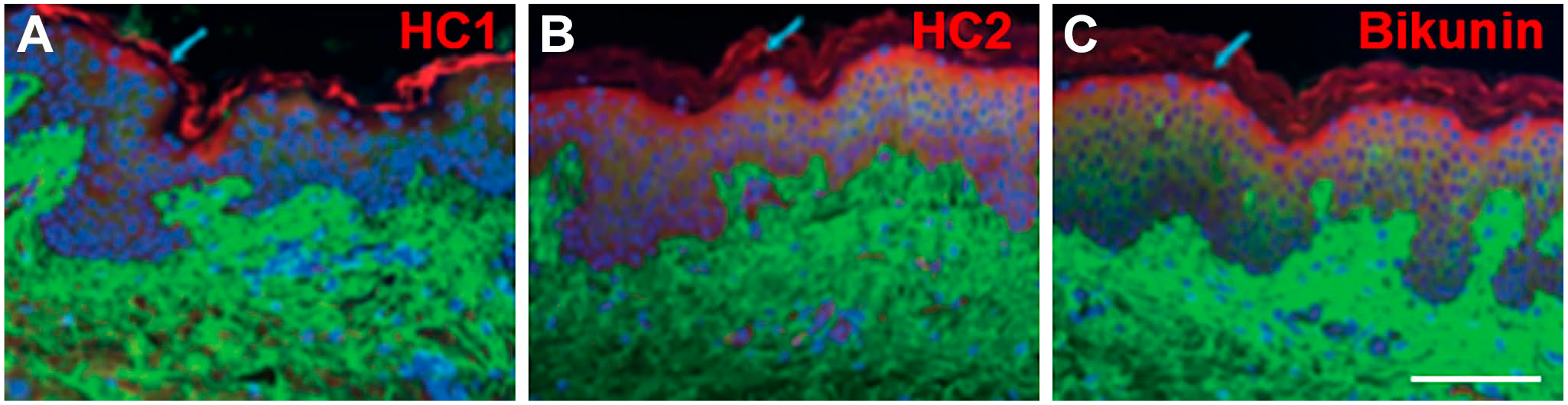
Immunofluorescent staining of normal human skin. Combined images are shown for hyaluronan (green; A-C), HC1 (red, A), HC2 (red, B), bikunin (red; D), and cell nuclei (blue; A-C). Scale bar, 100 µm. Reprinted with permission from Tan et al. 29 Abbreviation: HC, heavy chain.
Central Nervous System
IαI components have been identified in the human fetal and adult brain and localized to neurons and astrocytes, whereas this brain tissue was also found to contain IαI and PαI complexes. 19 In support of these findings, developing and adult ovine cerebral cortex and cerebrospinal fluid during development contain IαI and PαI complexes. 20 HCs have been immunolocalized to nerve fibers in the mouse brain along with the gene expression of HC2 and HC3.21,33 In addition, HC3 has been identified in the rat brain, as well as gene expression of bikunin has been identified in the hippocampus, cerebral cortex, and pituitary.32,147 Cultured neurons from embryonic mouse cerebral cortex express genes for bikunin, HC1-5 with bikunin/HCs immunolocalized intracellularly to isolated neurons, microglial cells, and astrocytes in culture. 22 These cultured neurons were found to contain IαI and PαI; however, their culture in the presence of serum cannot exclude this as the source of these complexes. Thus, the presence of IαI family members and their local expression indicates physiological roles such as brain development. There is evidence that neuroinflammation contributes to normal brain development, which may involve the formation of HA·HC complexes as has been reported for HC3 in the central nervous system and associated with an organized HA-rich pericellular matrix.148,149 In addition to its HA-interactive properties in the brain, IαI is an inhibitor of serine proteases which plays an important role in neuronal development and plasticity, suggesting that IαI modulates neuronal cell activities.150–152
Roles of IαI in Pathology
IαI is most often associated with inflammation where elevated levels of HA·HC complexes are detected in tissues and fluids.113,153,154 Indeed, the reported pathologies detailed below involving IαI family members exhibit inflammation, whether sterile or pathogenic forms, at some stage during the disease process. For example, islets in type 1 diabetic patients early in the disease exhibit increased amounts of IαI components associated with the peri-islet matrix containing increased levels of HA, whereas the expression of IαI components is decreased as the disease progresses and correlates with reduced HC1/HC2 expression in a mouse model of type 1 diabetes.155,156 The increased formation of HA·HC in many pathologies is correlated with increased expression of TSG-6, whereas the excretion of bikunin in the urine, in part as a by-product of HA·HC formation, is elevated in many inflammatory conditions.113,153 IαI itself is modified during inflammation where the CS chain that is biosynthesized is longer and less sulfated.63,157,158 This is hypothesized to facilitate the transfer of HCs from IαI to HA. 63 Lesser explored, however, are the roles of the IαI family in cell signaling, extracellular matrix interactions, and protease inhibition in the context of pathology.
Arthritis
HA·HC complexes are found in the synovial fluid and serum of patients with rheumatoid arthritis along with the expression of TSG-6 that is absent in normal synovial fluid.154,159,160 Osteoarthritis, although classically characterized as a non-inflammatory disease, is now recognized to involve inflammation with increased expression of TSG-6, but lower levels of HA·HC complexes in the synovial fluid than in rheumatoid arthritis.154,161–163 These complexes may have positive effects in the synovial fluid by preserving the hydrodynamic properties of the tissue that are compromised via the fragmentation of HA. In addition, the HA·HC complexes likely support infiltration of leucocytes into the synovial fluid 90 ; however it remains to be established whether this either contributes to the pathology or takes part in its resolution. In some contexts, HA·HC complexes are adhesive for naïve leucocytes,90,164–166 but not in others. 95 Recently, thrombin has been shown to ablate leucocyte adhesion to HA·HC matrices via cleavage of HC1 and is also likely to act in the same way on HC3-5. 167 Thrombin is elevated in the synovial fluid of patients with rheumatoid arthritis, 168 suggesting that thrombin-mediated modification of HA·HC complexes may modulate leucocyte adhesion. These cleaved HC fragments have been proposed to act as competitive inhibitors to HC–HC interactions between HA·HC complexes, 81 and thus thrombin could alter both the physical and biological properties of these matrices.
Although at low levels, HC1 was localized to the surface region of damaged cartilage in osteoarthritis as well as bikunin and HC2 with both interterritorial matrix and chondrocyte lacunae staining.16,163 It was recently shown that ADAMTS-5 and matrix metalloproteases can release HC2 from both IαI and HA·HC complexes, supporting the presence of truncated forms of HCs in osteoarthritic articular cartilage and synovial fluid.16,169
Fibrosis
Fibrosis is associated with myofibroblasts that fail to undergo apoptosis. 170 Although HA·HC complexes can support phenotypic modulation of fibroblasts to myofibroblasts in normal wound healing, 15 they may also support this phenotypic change during fibrosis. HA·HC complexes may also indirectly support fibrosis such as cystic fibrosis where these complexes support leucocyte attachment and are localized to the pulmonary vasculature and airway submucosa. 171 This tissue localization of HA·HC complexes results in a decrease in HA levels in sputum, likely affecting mucus hydration, viscoelasticity, and the clearance of pathogens. 171
Renal proximal tubular epithelial cells contribute to renal interstitial fibrosis via upregulating the secretion of the latent form of TGF-β1 with plasmin involved in its activation. IαI non-covalently binds TSG-6, and this complex exerts antiplasmin activity that controls TGF-β1 activation, which in turn modulates its expression. 13 These suggest a role for IαI family members in limiting inflammation during fibrosis, and recent work has shown that HC1 binds to both the small and large latent complexes of TFG-β1-3, thus perhaps having a role in controlling the activation and bioavailability of the mature growth factor. 81 Indeed, amniotic membranes composed of constitutively expressed HA·HC1/PTX3 complexes exert anti-inflammatory and antifibrotic effects via suppressing the proinflammatory activities of neutrophils and macrophages,88,172 as well as downregulating TFG-β1 expression and upregulating TGF-β3 (which counteracts TGF-β1 signaling) by fibroblasts and α-smooth muscle actin expression by myofibroblasts.28,143,173 In addition, in a mouse model of fibrotic lung injury, IαI was found to support angiogenesis and contribute to tissue repair, 174 whereas HA·HC complexes derived from the amniotic membrane are antiangiogenic. 175 These contrasting activities suggest that the composition of HA·HC complexes and the tissue context are important in determining the roles of IαI family members in fibrosis.
Asthma and Allergen-induced Lung Disease
Asthma is characterized by an increase in airway remodeling involving the deposition of HA in the bronchial mucosa. In a mouse model of asthma prepared via ovalbumin sensitization and challenge, the formation of HA·HC complexes stabilized the HA-rich matrix and suppressed inflammation by inhibiting tumor necrosis factor-α (TNF-α) activity through interaction with TNF receptor 1 in wild-type mice, whereas bikunin knockout mice exhibited high numbers of infiltrating neutrophils. 176 Furthermore, TSG-6·HC complexes were present in bronchoalveolar lavage fluid from patients with asthma, indicating that HA·HC complexes are prevalent in asthma. 113 This formation of HA·HC complexes is facilitated by the increased expression of TSG-6 where it is induced by TNF-α and IL-1β.113,177 TSG-6 also potentiates the antiplasmin activity of bikunin and inhibits tissue kallikrein, a serine protease, that acts to resolve inflammation when released from IαI.102,103,113,178 Although HA·HC complexes may provide protective effects, prevention of the formation of these complexes in the TSG-6 knockout mouse was shown to prevent airway hyperresponsiveness, suggesting that they may contribute to the pathogenesis in some lung diseases.179,180
Sepsis
Sepsis involves a systemic inflammatory response mediated by the host immune system and is associated with the release of neutrophil-derived proteases including elastase. IαI is susceptible to cleavage by elastase, releasing bikunin to exert its inhibitory activity on serine proteases. 181 There is a reduction in circulating IαI in patients with sepsis which correlates with increased mortality rates, whereas administration of exogenous IαI reduced mortality.182–185 These findings correlated with bikunin knockout mice being more sensitive to lipopolysaccharide (LPS)-induced death and suggest the protective role of IαI family members.127,186 Systemic endotoxemia in a mouse model has recently been shown to induce the deposition of HCs that colocalized with intravascular HA and is thought to contribute to improved outcomes via retention of neutrophils in the liver sinusoids.187,188
Cutaneous Wound Healing and Lichen Sclerosus
The presence of HA·HC complexes in skin has been reported to be elevated after wounding and to correlate with increased TSG-6 expression, suggesting a role for IαI family members in wound healing. 146 Lichen sclerosus is a chronic inflammatory skin disorder characterized by the presence of a broad hyalinized zone in the upper dermis where an increase in HA is observed. 189 IαI is associated with lichen sclerosus accumulating in the superficial dermis compared with normal skin and colocalized with HA. 190
Central Nervous System Injury
Reduced expression of IαI and PαI was observed in the brain following ischemia–reperfusion injury in sheep fetuses. 191 IαI has been suggested to possess neuroprotective effects as its systemic administration immediately following neonatal cerebral hypoxic-ischemic injury in rats was found to attenuate infarct volume and reduce cell death in the cortex. 192 Administration of IαI in this model has also been able to show improved working memory in adulthood. 193 Bikunin is also reported to possess neuroprotective effects as administration reduced infarct volume and neutrophil infiltration in rat occlusion–reperfusion brain injury model. 194 These studies suggest that IαI family members may have roles in neuroplasticity. TSG-6 is constitutively expressed by astrocytes and upregulated following injury, providing a means to form HA·HC complexes in line with the colocalization of HCs and HA in the pericellular matrix of glial scars (Fig. 4). 147 The HCs themselves have been identified in disease, including HC3 being reported to be a Tau-interactive protein in human Alzheimer’s brain and the gene for HC6 identified in autism. 195 Although the implication of these findings has not been explored, 196 both HA and TSG-6 are increased in the brain during Alzheimer’s disease. 197 In addition, genetic variants of the HCs are related to psychiatric disorders including schizophrenia and bipolar disorders, suggesting roles for the HCs themselves. 198
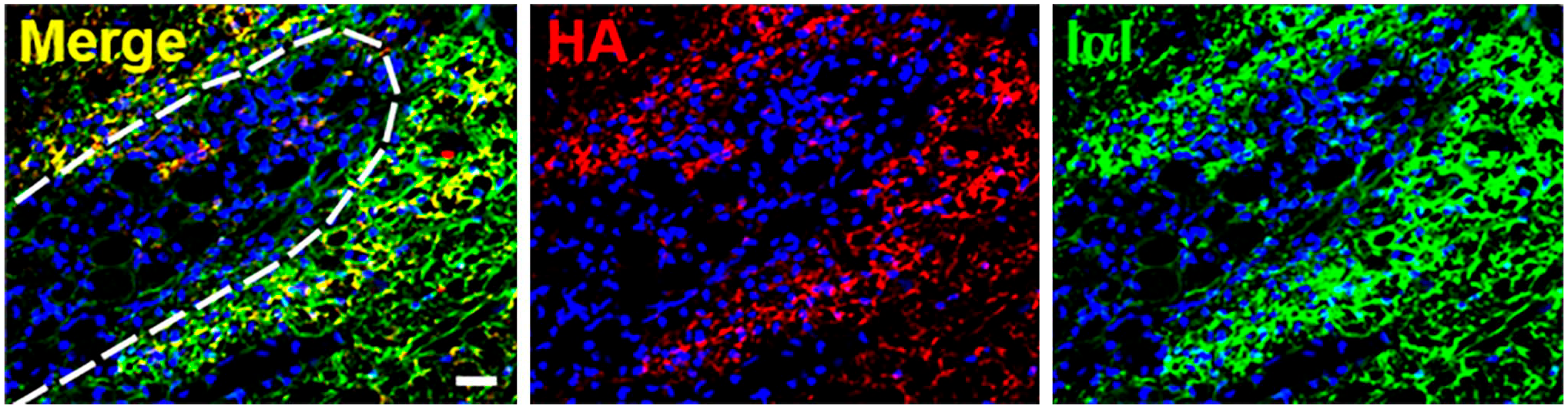
HA and IαI colocalize in the rat glial scar after injury. A spinal cord injury was created in 8- to 10-week-old female Sprague-Dawley rat using D-lysophosphatidylcholine administered to the dorsal and ventral funiculi which causes focal demyelination at the injection site. The glial scar was analyzed 15 days after injury via immunohistochemistry for the presence of HA (biotinylated HA binding protein; red), IαI (anti-IαI antibody [Dako, A0301]; green with known reactivity to HC1-3 and bikunin), and cell nuclei (DAPI; blue). Colocalization of HA and IαI immunoreactivity is shown surrounding the wound site. The dashed line indicates the wound boundary. Scale bar, 20 µm. This research was originally published in the Journal of Biological Chemistry. © The American Society for Biochemistry and Molecular Biology. Reprinted with permission from Coulson-Thomas et al. 147 Abbreviations: HA, hyaluronan; IαI, inter-α-trypsin inhibitor.
Calcium Oxalate Nephrolithiasis
The formation of calcium oxalate kidney stones is triggered by reactive oxygen species which, when dysregulated, cause tissue injury and inflammation. 199 IαI family components are found in kidney stones including HC1-3, along with HA, and correlated with increased gene expression for bikunin with hyperoxaluria.138,200–202 Interestingly, HA inhibits calcium oxalate crystallization in vitro. 202 Renal epithelial cells increase the expression of bikunin upon exposure to oxalate and may be protective in response to oxalate-mediated nephrotoxicity via inhibiting stone formation and attachment to the epithelium.138,199,203
Cancer
IαI family members are downregulated at the gene level in multiple tumors, including breast, colon, and lung cancer, and renal cell and oral squamous cell carcinoma.25,204,205 HC1-5 gene expression is reduced in multiple solid tumors, including breast, lung, and kidney. 25 Specifically, the HC5 gene has been identified as a tumor suppressor gene in multiple types of cancer, including breast, bladder, pancreatic, and colon cancer and acute myeloid leukemia.206–210 It can suppress the proliferation and migration of breast, bladder, and colon cancer cells in vitro.206,207,210 HC5 gene expression is reduced in cancer due to the aberrant hypermethylation of the promoter.208,211 Expression of the HC5 gene predicts longer overall survival in gastric and breast cancer and lung adenocarcinoma.210,212,213 Bikunin has known roles in tumor suppression by inhibiting cell–cell interactions, cell invasion, and metastasis, as well as providing serine proteinase inhibitory activity.107,117,118,214–219 In support of these findings, bikunin knockout mice have an increased prevalence of lung metastasis. 106 These animal studies support human studies reporting low levels of bikunin in the urine of patients with bladder cancer and in high-grade glioma tissue.107,108 In addition, low bikunin gene expression is an independent predictive factor of death in cancer patients due to the metastatic advantage conferred by low bikunin levels and is correlated with lower 5-year survival rates.57,116
Diagnostic and Therapeutic Uses of IαI Family Members
The diagnostic value of IαI family members has been explored in cancer, with low gene and/or protein levels correlating with poorer health outcomes. 57,116 More recent proteomics approaches have reported increased levels of IαI family members in diseases, including ovarian, gastric, and non–small cell lung cancer.220–222 However, such a biomarker approach will likely miss the patients with poorer prognostic outcomes as low levels of bikunin have correlated with lower 5-year survival rates.57,116 Thus, interpretation of proteomic screens can be refined to provide prognostic markers of better health outcomes.
The diagnostic value of IαI is being explored in the context of pregnancy, with preterm delivery involving cervical ripening that can be inhibited by bikunin via inhibition of calcium channel signaling that prevents myometrial contraction. 223 The level of bikunin in amniotic fluid is positively correlated with preterm delivery and thus may be a useful diagnostic biomarker. 224 Preeclampsia is characterized by systemic inflammation in response to ischemic hypoxia and oxidative stress, and patients with preeclampsia display higher cerebrospinal fluid and serum levels of bikunin than normotensive pregnant patients.225–227 These differences may be of diagnostic value for early identification of patients at risk of preeclampsia.
Bikunin, also known as ulinastatin, is used clinically in the treatment of conditions including acute respiratory distress syndrome, sepsis, and pancreatitis.228–230 It is marketed for clinical use as Miraclid, Ulinase, U-Tryp, Ulistin, Ustatin, or Techpool Roan and sourced either from human urine or from recombinant expression technologies. Meta-analyses of randomized controlled trials showed that the administration of bikunin reduced mortality, reduced serum inflammatory markers, and reduced hospitalization time, among other parameters, in patients with acute respiratory distress syndrome or sepsis.228,229 Although the exact mechanisms by which bikunin exerts its therapeutic functions in these conditions remain to be elucidated, it is tempting to speculate that its protease inhibitory functions may limit damage caused by the excessive release of proteolytic enzymes, and particularly elastase, from neutrophils.231,232
Topically, autopsies of patients who died from severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) revealed that their lungs were filled with a clear jelly, and while not positively identified is most likely HA-associated with acute respiratory distress syndrome, suggesting that bikunin is a potential treatment option for these patients.233,234 Similarly, HA is greatly increased in the airway lavage fluid of mice infected with the H1N1 influenza A virus, with elevated levels persisting 10 weeks after infection along with the presence of HA·HC complexes; here, removal of HA with hyaluronidase restored lung function to normal, proving another potential option for treating the lung symptoms associated with COVID-19. 235 A retrospective analysis of patients diagnosed with pancreatitis and treated with bikunin had a lower mortality rate. 230 Patients with Kawasaki disease, a condition characterized by vasculitis in infants and recently linked to SARS-CoV-2 infection in children, 236 is usually treated intravenously with immunoglobulin together with aspirin to resolve inflammation; however, coronary artery lesions develop in some patients. A retrospective analysis of concurrent administration of bikunin was found to reduce the occurrence of coronary artery lesions, suggesting that it may be of clinical value. 237
IαI itself may also provide therapeutic benefit. Animal models suggest that IαI administration during sepsis reduced the mortality rate,185,238 administration after hypoxic-ischemic brain injury provided neuroprotective effects, 192 and coadministration with antimicrobial agents protected mice challenged with Bacillus anthracis from death. 239
To conclude, the literature provides a wealth of evidence for the roles of IαI family in both biology and pathology. IαI family members have roles in matrix organization, cell signaling, protease inhibition, and regulation of complement activation and are found in many tissues. IαI family members have a role in inflammation where they can either act to limit and resolve this process to restore tissue homeostasis or perpetuate pathology. These contrasting activities are likely due to the composition of IαI family members in their temporal tissue context. As such, IαI family members have been explored for both diagnostic and therapeutic applications and used clinically in some countries. Although our understanding of the roles of IαI family members is largely associated with its HA matrix organization properties, future research is needed to explore their diverse roles in both physiological and pathological processes for both fundamental knowledge and to progress treatments for improved health outcomes.
Footnotes
Competing Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: MSL and JMW are Directors of Glycos Pty Ltd, which is focused on the generation of bioengineered glycosaminoglycans as therapeutics. AJD is a Director of Link Biologics, which is focused on the use of a TSG-6-based biological drug for inflammatory conditions. The other author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
All authors contributed to the literature review and manuscript preparation.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: M.S.L. and J.M.W. were funded by the Australian Research Council under the Linkage Project (LP140101056) and Discovery Project (DP150104242) schemes, and A.J.D. was funded by Versus Arthritis (19489, 20895, 21946, and 22277).
