Abstract
Crohn’s disease (CD) is a gastrointestinal disorder of unknown etiology. CD-specific longitudinal ulcers show an association between disease pathogenesis and vasculature dysfunction. Granulomatous lymphangitis may also contribute to CD pathogenesis; meanwhile, vasculitis is the primary CD lesion. We investigated the association between granulomas and lymphatic and blood vessels to assess the role of vasculature in CD pathogenesis. Two small and large intestine specimens were obtained from four CD patients. From each specimen, 160 sequential sections were obtained and double immunohistochemical stained to label lymphatic and blood vessels in association with granulomas. We found that 289 of 342 granulomas (85%) were associated with a lymphatic vessel and 313 of 364 granulomas (86%) were associated with a blood vessel. Although intrablood vessel granulomas were not detected, intralymphatic vessel granulomas were. In the internal region of the granuloma, we found more blood vessels than lymphatic vessels. Hence, these results cumulatively demonstrate that CD epithelioid cell granulomas are differentially associated with lymphatic and blood vessels, suggesting both as essential for the formation and maintenance of these granulomas. Moreover, both lymphatic and blood vessels may participate in granulomatous inflammation in the primary CD lesions; however, additional studies with larger numbers of participants are required to validate our findings.
Introduction
Crohn’s disease (CD) is a chronic inflammatory disorder of the gastrointestinal tract of unknown etiology. Endoscopy in CD patients typically reveals a discontinuous distribution of longitudinal ulcers along the mesenteric aspect, wherein intestinal blood and lymphatic vessels assemble. Histologically, non-caseating epithelioid cell granulomas have been detected in numerous patients with CD. The association of blood vessels or lymphatic vessels with CD pathogenesis has been predicted based on the distribution of the ulcer region and several studies have examined the association between granulomas and blood or lymphatic vessels.1–5
Vasculitis has been reported as the primary lesion in CD as sequential pathogenetic events, including vascular injury, focal arteritis, fibrin deposition, and arterial occlusion occur exclusively during CD. 4 Other studies have reported the importance of examining granulomatous lymphangitis to fully understand the pathogenesis of CD.1,3 Previous studies on CD have reported lymphangiectasia, lymphatic obstruction, and the appearance of lymphatic vessels containing numerous lymphocytes and histiocytes. 2 As lymphatic endothelial markers have become recently available, studies have reevaluated the association between a granuloma and the lymphatic system in CD using these markers. 3
A study comparing granuloma-associated blood and lymphatic vessels demonstrated that a granuloma is more often associated with a lymphatic vessel than with a blood vessel. 6 However, the extent of the association between a granuloma and a blood or lymphatic vessel differs between studies,3,5,6 likely due to differences between the methods used. Specifically, these studies examined the association between a granuloma and the vasculature using one slide or a few sequential slides. Accurate examination of these structures is essential for assessing the importance of the association between a granuloma and the vasculature in CD pathogenesis.
In this study, we focused on the correlation between epithelioid cell granulomas and blood or lymphatic vessels in CD. We examined granulomas for which the whole structure presented a sequential double immunostaining for histiocytes and vasculature and investigated the association between granulomas and the vasculature.
Materials and Methods
Tissue Selection
This study was approved by the Ethics Committee of Tokyo Yamate Medical Center (J-001) and Tokyo Medical and Dental University Hospital (M2018-020). A total of 124 CD patients, who underwent surgical treatment between April 2017 and February 2018, were included. Two cases of small intestine (Case 1: ileum and Case 2: ileum) and two cases of large intestine (Case 3: sigmoid colon and Case 4: ascending colon), including a larger number of epithelioid cell granulomas, were selected. Patients did not have major comorbidities, nor had they undergone surgical treatment in the past. Patients’ characteristics and treatment histories are provided in Table 1.
Clinical Data.
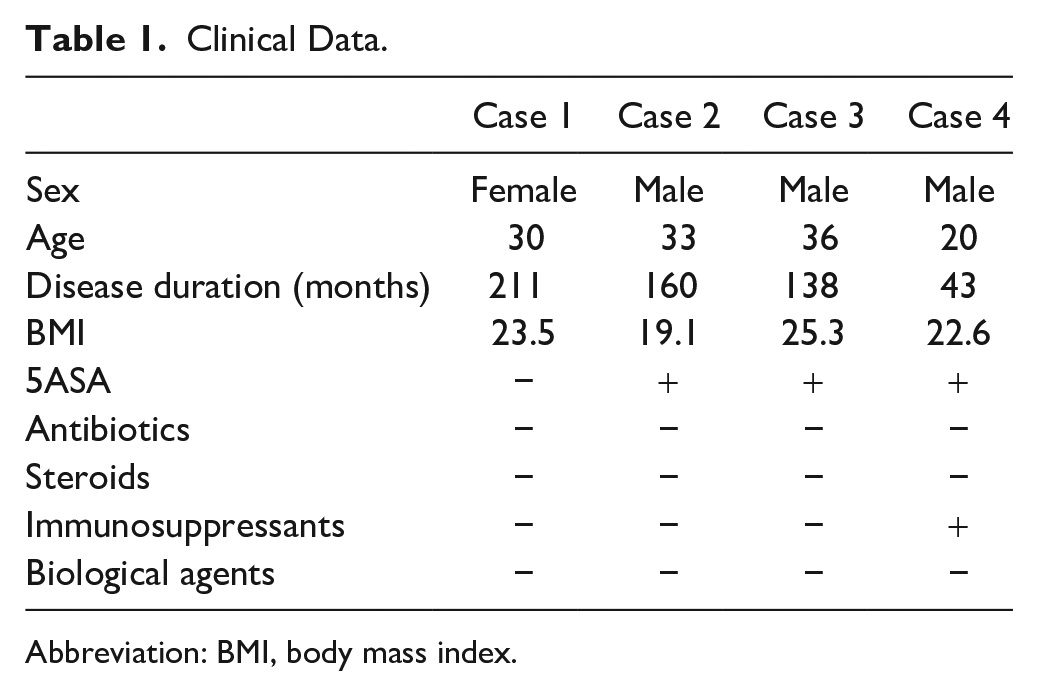
Abbreviation: BMI, body mass index.
Sequential Double Immunostaining
A total of 160 sequential sections (5 µm thick) were obtained from formalin-fixed paraffin-embedded tissue blocks; these sections were deparaffinized in xylene and rehydrated in alcohol. Antigen retrieval was performed using citric acid buffer (0.01 M, pH 7.0) at 96C for 40 min. The avidin–biotin complex immunoperoxidase method was used with the commercial Vectastain R.T.U Universal Elite ABC HRP kit (Vector Laboratories, Burlingame, CA) for the first step of immunostaining. All slides were incubated with 3% hydrogen peroxide to block endogenous peroxidase activity.
Regarding primary antibodies, anti-Von Willebrand factor antibody (Dako, Glostrup, Denmark) was applied to the first half of the 160 sections for blood vessel detection, and anti-podoplanin antibody (Dako, Carpinteria, CA) was applied to the second half for lymphatic vessel detection. All sections were incubated for 60 min at room temperature, washed with phosphate-buffered saline and 0.005% Tween 20, and incubated with R.T.U Universal Elite ABC HRP kit reagents A and B (Vector Laboratories) for 30 min. Sections were incubated for 10 min in 0.1 M Tris-HCl containing 0.05% 3,3′-diaminobenzidine, 0.04% nickel chloride, and 0.0075% H2O2. The first set of antibody reagents was inactivated by adding phosphate-buffered salts followed by incubation at 96C for 10 min. A Vectastain ABC-AP Staining kit (Vector Laboratories) was used for the second step of immunostaining. To detect histiocyte aggregation, all sections were incubated with primary anti-CD68 antibody (Dako) at room temperature for 60 min. A horse anti-mouse biotinylated secondary antibody (Vector Laboratories) was applied for 30 min. The Vectastain ABC-AP Staining kit reagent (Vector Laboratories) was used following the manufacturer’s instructions, and the sections were incubated for 30 min. After washing, visualization was performed using the Vector Blue Alkaline Phosphatase Substrate Kit III (Vector Laboratories). Cell nuclei were not counterstained. Sequential double-immunostained specimens were evaluated as follows. Granuloma was defined as the foci of tightly packed aggregated CD68-positive cells. Only entire granulomas within 80 sequential sections were evaluated (Fig. 1). The association between a granuloma and the vasculature was examined on each slide. The size of the granuloma and the distance between the granuloma and vasculature was measured using Olympus DP2-BSW software (Olympus, Tokyo, Japan). A video was generated by stacking sequential images using ImageJ software (NIH, Bethesda, MD).
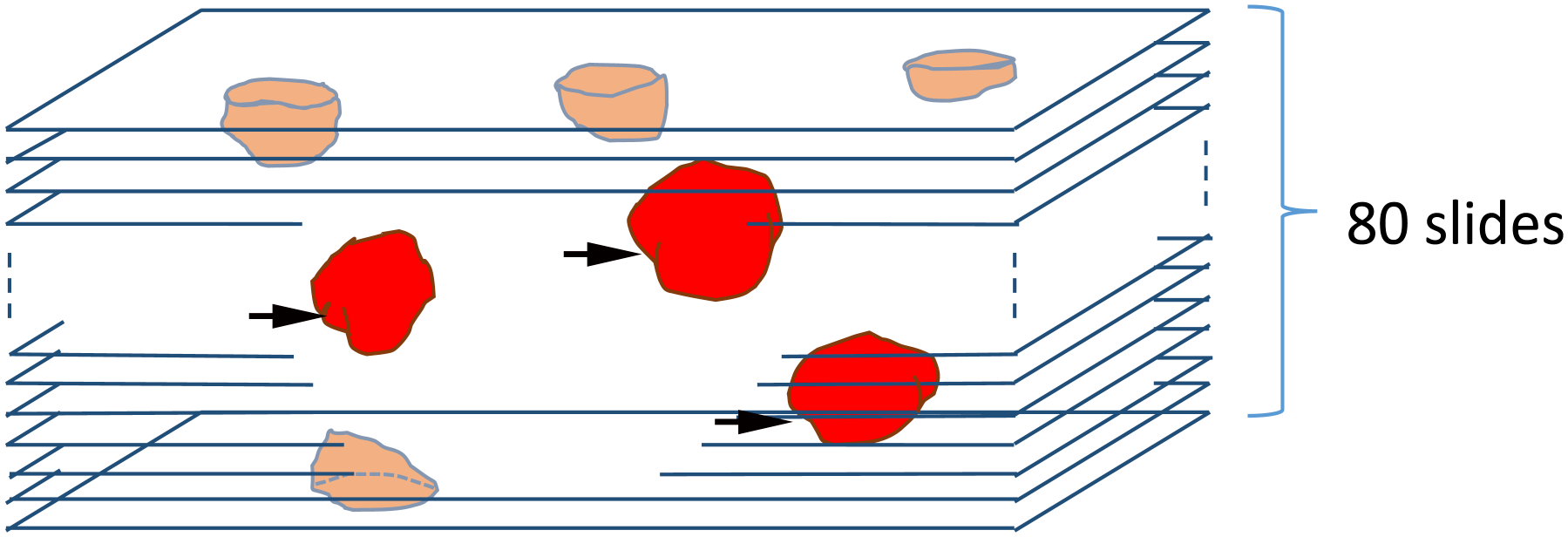
Only granulomas with the whole structure visible were included in the 80 consecutive sections (arrow) that were evaluated.
Statistical Analysis
All statistical analyses were two-tailed, and a p value > 0.05 was considered statistically significant. The chi-square exact test was performed to determine the associations between different variables. All statistical analyses and graphical representations were performed using GraphPad Prism 7 software (GraphPad Software, San Diego, CA).
Results
Number, Size, and Distribution of Epithelioid Cell Granulomas
A total of 706 granulomas were found; 364 granulomas were detected by double immunostaining for blood vessels and histiocytes, whereas 342 granulomas were detected by double immunostaining for lymphatic vessels and histiocytes. No significant differences in size or distribution were observed between granulomas associated with blood vessels and granulomas associated with lymphatic vessels. We detected 474 and 232 granulomas in the small and large intestine specimens, respectively, with a mean size of 119 μm. The size of granulomas did not differ significantly between the small and the large intestine (Fig. 2A) or between intestinal layers (Fig. 2B). The distribution of granulomas in each intestinal layer differed significantly between the small and the large intestine specimens (Fig. 2C, p < 0.0001) with more granulomas observed in the small intestinal mucosa (47/474; 10%) compared with the large intestinal mucosa (9/232; 4%), and fewer granulomas observed in the muscularis propria of the small intestine (101/474; 21%) compared with the muscularis propria of the large intestine (96/232; 40%). In the submucosa, 189 of 474 (40%) granulomas and 81 of 232 (35%) granulomas were detected in the small and large intestine, respectively. In the subserosa, 137 of 474 (29%) granulomas and 46 of 232 (20%) granulomas were detected in the small and large intestine, respectively.
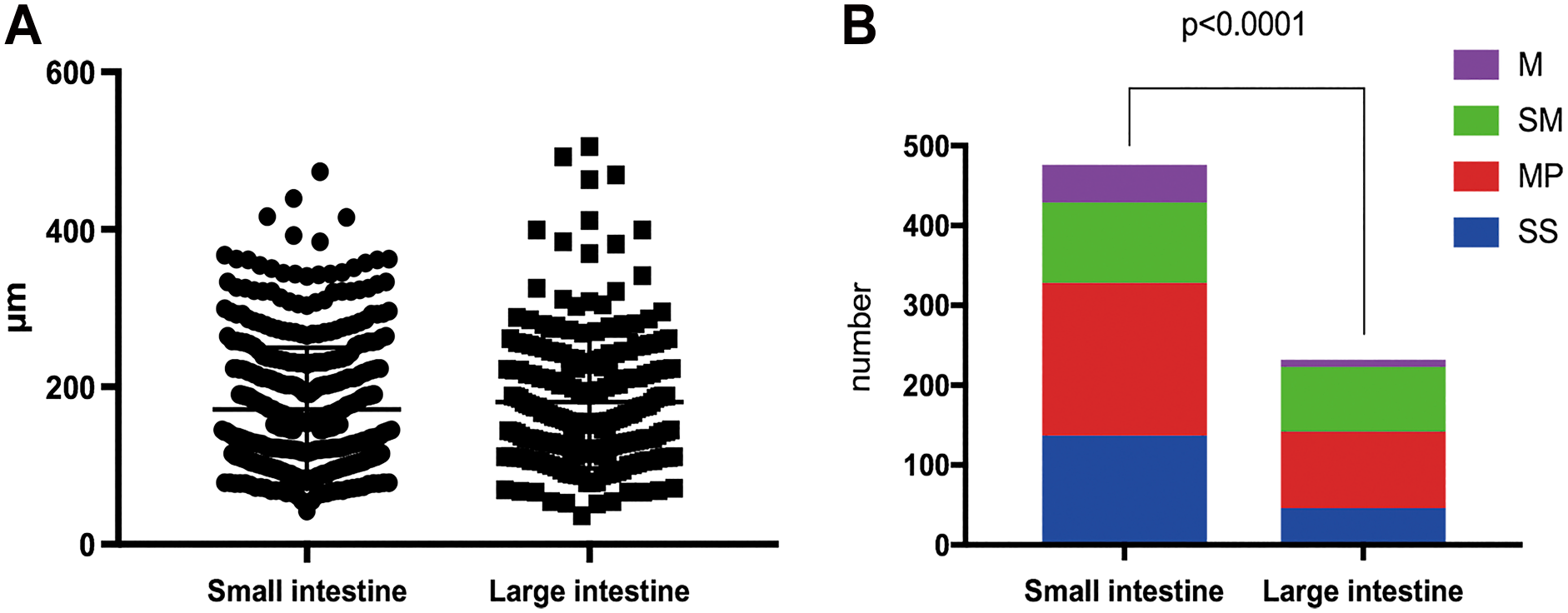
(A) The size of granulomas did not differ between the small and large intestine. (B) Granuloma distribution differed in each intestinal layer between the small intestine and the large intestine. Abbreviations: M, mucosa; SM, submucosa; MP, muscularis propria; SS, subserosa.
Association Between Epithelioid Cell Granulomas and Blood or Lymphatic Vessels
Based on the association between granulomas and blood vessels or lymphatic vessels, four patterns were observed: Pattern A1, granuloma partly surrounded by vasculature; Pattern A2, granuloma containing vasculature; Pattern A3, granuloma protruded from, and striding over vasculature; Pattern A4, granuloma in the vasculature (Fig. 3A). We further defined an un-associated pattern wherein a granuloma is detached from the vasculature as pattern U. A granuloma displaying one of the patterns in at least one slide was considered a vasculature-associated granuloma, whereas a granuloma displaying only pattern U was considered a vasculature non-associated granuloma. Granulomas were highly associated with lymphatic and blood vessels; 313 of 364 granulomas (86%) were associated with a blood vessel, whereas 289 of 342 granulomas (85%) were associated with a lymphatic vessel, and differences between both were not significant (Fig. 3B). The differences in the distribution of blood vessel–associated granulomas between the different intestinal layers were significant in the small intestine (p < 0.0001), however, not in the large intestine (Fig. 3C). In the small intestine, the proportion of blood vessel non-associated granulomas was larger than that of blood vessel–associated granulomas in the mucosa. Alternatively, the proportion of blood vessel non-associated granulomas was markedly lower than that of blood vessel–associated granulomas in intestinal layers other than the mucosa. The distribution of lymphatic vessel–associated granulomas in each intestinal layer was not significantly different (Fig. 3D). In vasculature non-associated granulomas, no significant differences were observed in the distance between the granuloma and a blood vessel, or a lymphatic vessel (Fig. 3C). The mean distance between a granuloma and a blood vessel, or a lymphatic vessel, was 67 and 103 μm, respectively.
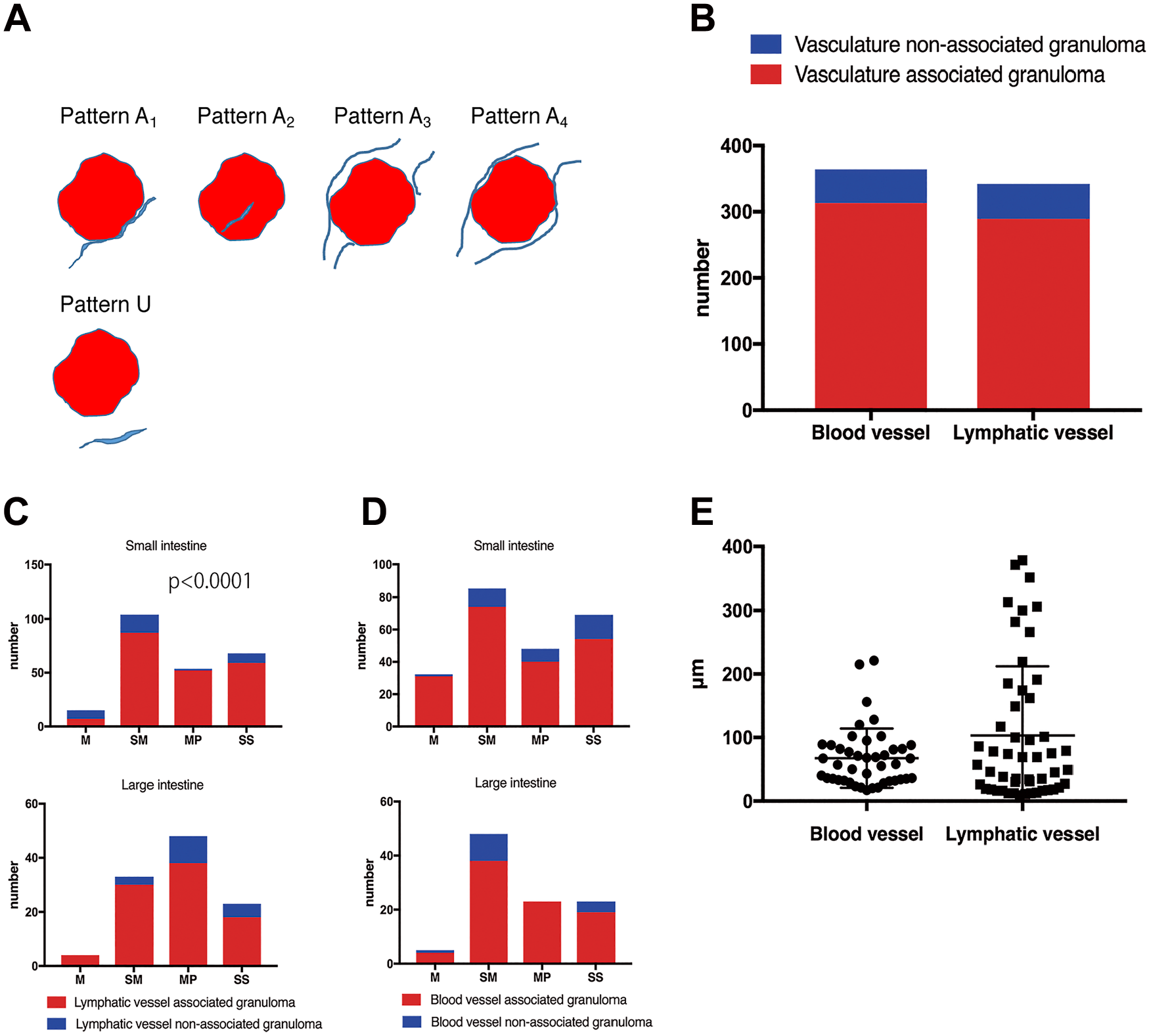
(A) Five different patterns were observed in the association between a granuloma and the vasculature. (B) Granulomas are associated with blood and lymphatic vessels. (C) The differences in the distribution of lymphatic vessel–associated granulomas between the different intestinal layers were significant in the small intestine but not in the large intestine. (D) The distribution of blood vessel–associated granulomas between the different intestinal layers remained unchanged in the small and large intestine. (E) No significant differences were observed in the distance between a granuloma and a blood vessel or between a granuloma and a lymphatic vessel. Abbreviations: M, mucosa; SM, submucosa; MP, muscularis propria; SS, subserosa.
Comparison Between Patterns of Blood Vessel– and Lymphatic Vessel–Associated Granulomas
The frequency of each association pattern significantly differed between blood vessels– and lymphatic vessels–associated granulomas (Fig. 4A). Each pattern was counted only once per granuloma, even if a granuloma included the same pattern several times. For granulomas showing different patterns, each pattern was assessed individually. The distribution of each association pattern in each layer between a granuloma and a blood vessel is shown in Fig. 4B, and that between the granuloma and a lymphatic vessel is shown in Fig. 4C. Only Patterns A1 (303/434, 70%) and A2 (131/434, 30%), not Patterns A3 (0/434, 0%) or A4 (0/434, 0%), were observed in blood vessel–associated granulomas (Fig. 4A, B, and D), whereas Patterns A1 (358/437, 82%), A2 (33/437, 8%), A3 (5/437, 1%), and A4 (41/437, 9%) were observed in lymphatic vessel–associated granulomas (Fig. 4A, C, and E). The proportion of each pattern in each intestinal layer was significantly different in lymphatic vessel–associated granulomas of the small, but not the large intestine (Fig. 4C). Furthermore, the proportion of Pattern A4 in the mucosa of the small intestine was markedly greater than that of the other intestinal layers. Granulomas displaying Patterns A1, A2, or A3 tended to exclude Pattern A4, whereas most granulomas displaying Pattern A4 only showed that pattern (Supplementary Video 1). Along with epithelioid cell aggregation, isolated CD68-positive histiocytes were occasionally observed in lymphatic vessels (Fig. 4F); however, isolated histiocytes were also detected in blood vessels. The other distinct difference in the association patterns between blood vessels and lymphatic vessels was the proportion of Pattern A3, which was observed to be more frequently associated with a blood vessel than a lymphatic vessel (Fig. 4A). Therefore, more blood vessels than lymphatic vessels were detected in intragranuloma regions. Representative images of the associations between a granuloma and a blood, or a lymphatic vessel, are shown in Supplementary Videos 2 and 3, respectively.
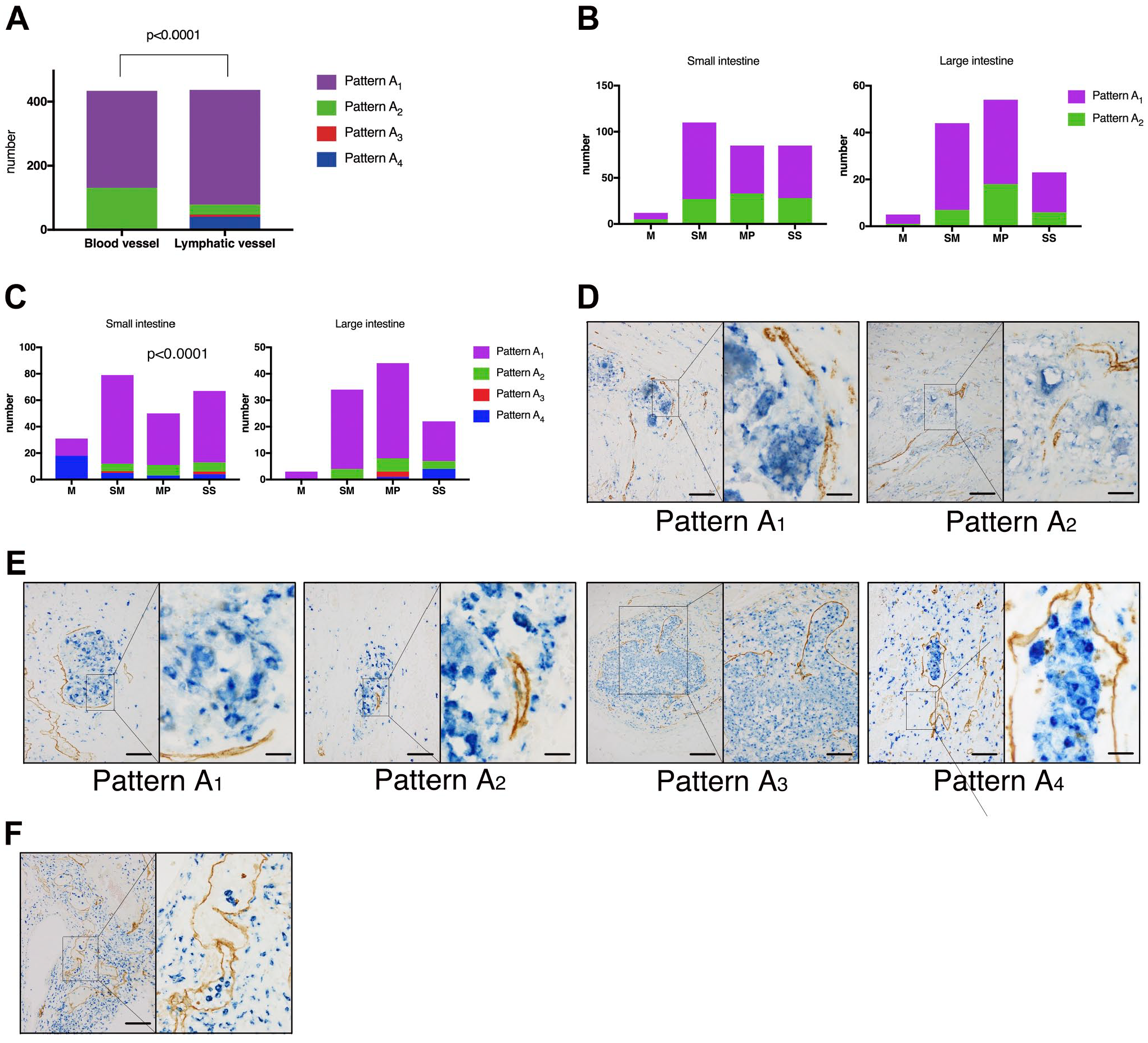
(A) Proportion of each association pattern differed between blood vessels and lymphatic vessels. (B) The distribution of each granuloma-blood vessel association pattern between the different intestinal layers. (C) The distribution of each granuloma-lymphatic vessel association pattern between the different intestinal layers. (D) Patterns A1 (Scale bar of the left panel = 100 μm, scale bar of the right panel = 20 μm) and A2 (Scale bar of the left panel = 200 μm, scale bar of the right panel = 50 μm) were observed in the association between granulomas (blue) and blood vessels (brown). (E) Patterns A1 (Scale bar of the left panel = 100 μm, scale bar of the right panel = 20 μm), A2 (Scale bar of the left panel = 100 μm, scale bar of the right panel = 20 μm), A3 (Scale bar of the left panel = 200 μm, scale bar of the right panel = 100 μm), and A4 (Scale bar of the left panel = 100 μm, scale bar of the right panel = 20 μm) were observed in the association between granulomas (blue) and lymphatic vessels (brown). (F) Intralymphatic isolated histiocytes with double immunostaining of a histiocyte (blue) and a lymphatic vessel (brown). (Scale bar of the left panel = 200 μm, scale bar of the right panel = 20 μm). Abbreviations: M, mucosa; SM, submucosa; MP, muscularis propria; SS, subserosa.
Discussion
We found that 86% and 85% of epithelioid cell granulomas in CD are associated with blood and lymphatic vessels, respectively. We evaluated the association between a granuloma and the vasculature (a lymphatic vessel or a blood vessel) by sequential double immunostaining of 80 sections, and only granulomas with the whole structure included in the sections were assessed. This method enables three-dimensional evaluation of the association between a granuloma and the vasculature. Moreover, immunohistochemical analysis was performed to distinguish a lymphatic vessel from a blood vessel. The association between these structures was examined in detail by sequential double immunostaining with a clearer evaluation than in previous studies, which only involved two-dimensional partial evaluations. Our results show that an epithelioid cell granuloma is highly associated with lymphatic and blood vessels with approximately the same frequency. In vasculature non-associated granulomas, the mean distance between the granuloma and a lymphatic or blood vessel was 103 and 67 μm, respectively. Considering that histiocytes are approximately 15–100 µm in diameter, 7 even in vasculature non-associated granulomas, most histiocytes were markedly close to a lymphatic or a blood vessel. The importance of blood and lymphatic vessels in the pathogenesis of CD has been reported separately in several studies. Our results suggest that granulomatous inflammation involving both lymphatic vessels and blood vessels is a primary lesion in CD.
Some granulomas were observed within a lymphatic vessel. Intralymphatic granulomas or intralymphatic epithelioid cell aggregations were also observed previously.1–3 One study reported an intrablood vessel granuloma 6 ; however, no intrablood vessel granulomas or epithelioid cell aggregations were observed in our study. Epithelial histiocyte aggregation and isolated histiocytes were occasionally detected in a lymphatic vessel but rarely in a blood vessel, suggesting that aggregated epithelioid cells circulate exclusively in the lymph and not in the blood. Histiocytes with certain epithelial characteristics are the primary constituent cells of an epithelioid cell granuloma. 8 However, the origin of these epithelioid histiocytes remains unclear. Lymphatic vessels facilitate the migration of histiocytes, such as dendritic cells and monocytes from the lamina propria to mesenteric lymph nodes.9,10 Epithelioid histiocytes comprising epithelioid cell granulomas are believed to be primarily derived from a lymphatic vessel, as they are associated with granulomas and their aggregation is observed only in lymphatic vessels and not in blood vessels. It remains unclear how epithelioid cells are supplied to a granuloma; however, we observed few granulomas protruding from a lymphatic vessel. These granulomas encompassed the internal and external region of lymphatic vessels; therefore, once formed, granulomas in lymphatic vessels may migrate to the external region of a lymphatic vessel. However, the proportion of the granuloma protruding from a lymphatic vessel was rather small compared with the proportion of intralymphatic granulomas, and granulomas in the external region of lymphatic vessels. Therefore, other possibilities may be considered. For example, we speculated that a histiocyte egressing from the lymphatic vessel with the previous state of granuloma can accumulate in the perilymphatic vessel region and gradually form an epithelioid cell granuloma.
Attachment patterns to a granuloma differed between blood and lymphatic vessels. Blood vessels tended to adhere to the edge and the internal region of the granuloma, whereas a lymphatic vessel was more commonly found adhered to the edge of the granuloma (Fig. 5). These differences in attachment are likely attributable to the function of the different vessels. For instance, the primary function of blood vessels is to transport oxygen and nutrients to the tissue and eliminate waste material from the tissue. Blood vessels adhering to a granuloma may supply materials that are essential for maintaining the functions and characteristics of the granuloma, which may account for why more blood vessels than lymphatic vessels were observed in the internal region of granulomas. Moreover, the increased pressure experienced by blood vessels compared with lymphatic vessels may cause blood vessels to penetrate the granuloma more readily.
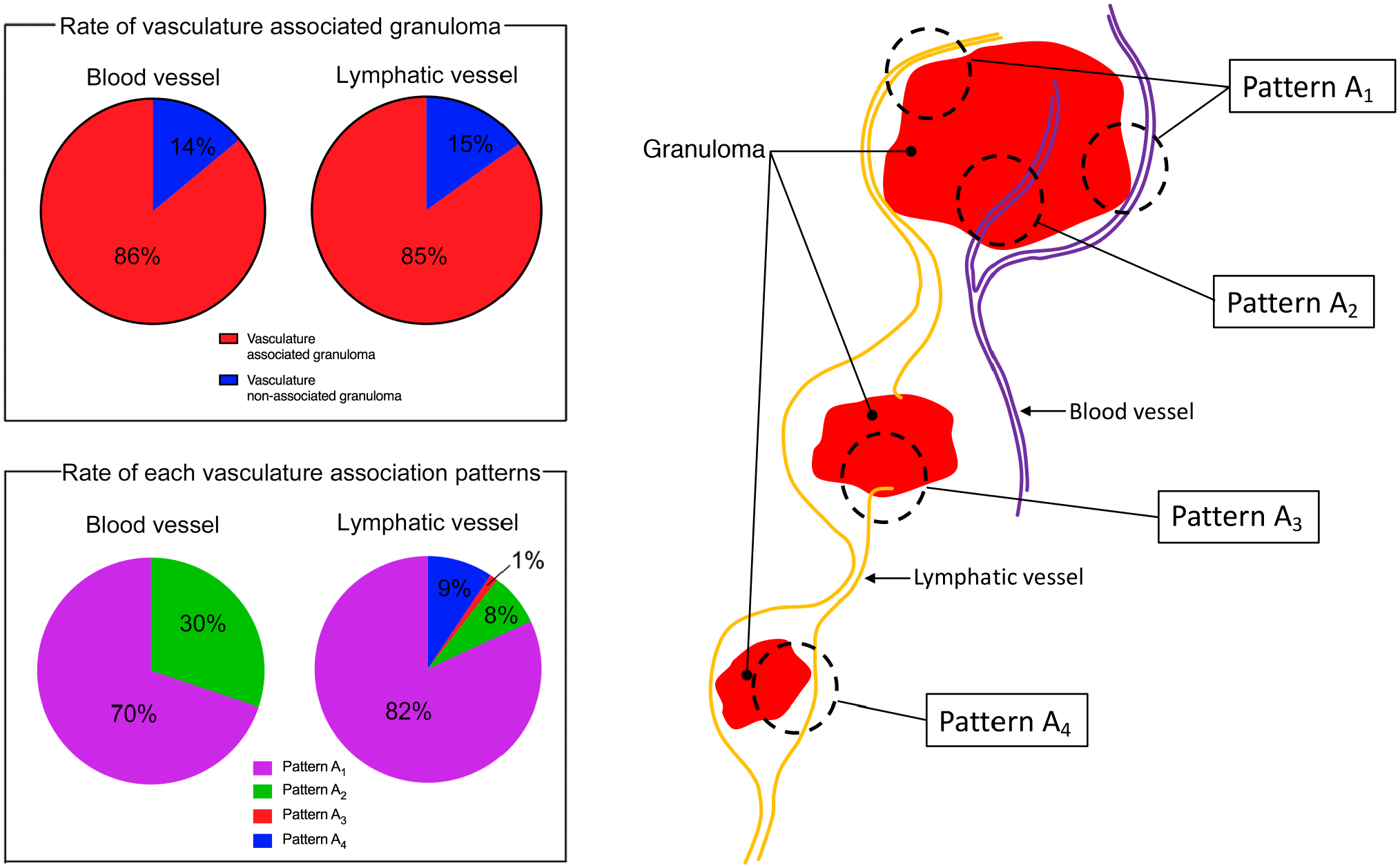
An epithelioid cell granuloma in Crohn’s disease is differentially associated with blood and lymphatic vessels.
The proportion of granulomas in the mucosa of the small intestine was larger than that in the mucosa of the large intestine. Moreover, intralymphatic granulomas were more frequent in the mucosa of the small intestine compared with that of the large intestine. The lymphatic distribution of the small intestine differs from that of the large intestine. Thus, lymphatic vessels known as lacteals are present in the small intestinal mucosa, but not in the large intestinal mucosa. 11 This difference in the distribution of lymphatic vessels may reflect the difference in the proportion of granulomas, in particular intralymphatic granulomas, in the mucosa between the small and large intestines. Regarding the association between a granuloma and blood vessel in the small intestine, the proportion of blood vessel non-associated granulomas to blood vessel–associated granulomas was larger in the mucosa than in other layers. This can be ascribed to the high levels of intralymphatic granulomas in the mucosa of the small intestine as the lymphatic system is separated from the blood system in peripheral tissue, 11 and a blood vessel cannot contact the intralymphatic granuloma.
It is important to note that the disease duration of the cases presented in this study was relatively long (average 138 months). However, no cases had a history of intensive treatment, including steroids or biological agents. Nevertheless, the findings reported here may have been influenced by other treatment regimens and may differ from what would be observed in patients with a shorter disease course. We, therefore, require additional cases to confirm the findings reported herein, as well as to better understand the influence of various treatment regimens.
In conclusion, this study demonstrates that an epithelioid cell granuloma in CD is differentially associated with blood and lymphatic vessels. These findings indicate that both blood and lymphatic vessels are crucial for the development and maintenance of epithelioid cell granulomas. In addition, granulomatous inflammation involving both lymphatic and blood vessels accounts for the primary lesion in CD.
Footnotes
Acknowledgements
We thank Yu Etoh for her assistance in performing the immunohistochemical examination and evaluation.
Competing Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
MK conceived and designed the research, carried out the immunohistochemistry, performed the data analysis and interpretation, and wrote the manuscript. DK and KA performed the data interpretation and helped with drafting the manuscript. RS, TY, SF, and SO helped with clinical data collection. TY and TT helped with planning the experiment. All authors have read and approved the final manuscript.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was financially supported by Tokyo Medical and Dental University.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
