Abstract
Lymphatic vessel networks can expand and regress, with consequences for interstitial fluid drainage and nutrient supply to tissues, inflammation, and tumor spread. A diet high in sodium stimulates hyperplasia of cutaneous lymphatic capillaries. We hypothesized that dietary sodium restriction would have the opposite effect, shrinking lymphatic capillaries in the tongue. Lingual lymphatic capillary density and size was significantly reduced in mice fed a low-sodium diet (0.03%) for 3 weeks compared with control-fed mice. Blood vessel density was unchanged. Despite lymphatic capillary shrinkage, lingual edema was not observed. The effect on lymphatic capillaries was reversible, as lymphatic density and size in the tongue were restored by 3 weeks on a control diet. Lymphatic hyperplasia induced by a high-sodium diet is dependent on infiltrating macrophages. However, lingual CD68+ macrophage density was unchanged by sodium deficiency, indicating that distinct mechanisms may mediate lymphatic regression. Further studies are needed to test whether dietary sodium restriction is an effective, non-invasive co-therapy for oral cancer.
Introduction
Lymphatic vascular networks drain the interstitial fluid that delivers nutrients and play a critical role in tissue immune surveillance.1–4 Like blood vessels, lymphatic capillaries can sprout and grow in response to tumors or inflammation.2,5 A balance between growth and regression is needed to prevent lymphedema, which can result from deficits in lymphatic vessel number or function. Lymphangiogenesis, or the formation of new lymphatic vessels, can be harmful and contribute to inflammation resulting from psoriasis, rheumatoid arthritis, and graft-versus-host disease. 1 Lymphatic vessels also provide a conduit for some types of tumor to metastasize.5,6 Compared with lymphangiogenesis, the conditions and mechanisms that mediate lymphatic vessel regression are poorly understood.1,3
Head and neck cancer spreads aggressively, in part, because of the extensive lymphatic network in oral tissues.7,8 Moreover, the tongue is drained by contralateral as well as ipsilateral lymphatic vessels that further promote tumor cell spread to distant sites even in early stage tumors. 7 Oral tongue squamous cell carcinoma is the most commonly diagnosed and metastatic of the oral cancers. 9 Current therapies for head and neck cancers include radiation, chemotherapy, and surgical resection. Yet, 5-year survival rates have remained stubbornly below 50–60% for decades. 7 Lymphatic vessel density is inversely predictive of survival in patients with oral squamous cell carcinoma.8,10 Thus, inhibiting lymphangiogenesis may limit the spread of oral cancers, as shown in mouse models. 11
Dietary sodium restriction is a potential non-invasive approach to block lymphangiogenesis or induce lymphatic vessel regression. A high-sodium diet promotes cutaneous lymphatic capillary growth and hyperplasia through the recruitment of macrophages that secrete the lymphangiogenic factor, VEGF-C. 12 We tested the effects of a low-sodium diet on lymphatic vessel density in the tongue because of the poor prognosis of oral squamous cell carcinomas in this tissue. Moreover, in our previous studies on inflammation in the tongue, we found that a sodium-deficient diet inhibits vascular cell adhesion molecule (VCAM)-1 expression and lingual macrophage responses to sterile injury.13–15 Macrophages are critical for the expansion of lymphatic networks,12,16 suggesting that sodium deficiency may also influence lymphatic vessel density. Thus, we hypothesized that dietary sodium restriction induces lymphatic vessel regression in the tongue. We demonstrate that dietary sodium deficiency reversibly decreases the density and size of lingual lymphatic capillaries.
Materials and Methods
Ethics Statement
The Animal Care and Use Committee at the Medical College of Georgia at Augusta University approved all procedures (AUP 2009-0178), which follow guidelines set by the National Institutes of Health Guide for the Care and Use of Laboratory Animals.
Animals and Dietary Sodium Restriction
Female, specified pathogen-free C57/BL6J mice (Stock No. 000664, Jackson Laboratory, Bar Harbor, ME) were 8–11 weeks old at the beginning of experiments. Females were used in this initial study to minimize gender-related variability, and because less is known about lymphatic network remodeling in females. Mice were group-housed in polyurethane cages in a barrier facility with humidity, temperature, and light (12:12-hr light: dark with lights on at 6:00
IHC
Lymphatic vessels were identified with a well-characterized rabbit polyclonal antibody to lymphatic vessel endothelial hyaluronan receptor (LYVE)-1 (1:400, ab14917; Abcam, Cambridge, MA), which is strongly expressed by lymphatic endothelial cells forming capillaries.2,12,17,18 We used antiplatelet endothelial cell adhesion molecule (PECAM)-1 (CD31; 1:100, MCA2388; Bio-Rad, Hercules, CA) as a marker for blood vessels.2,19 Tissue macrophages were identified with CD68 (clone FA/11; 1:100, ab53444; Abcam, Cambridge, MA).12,20,21 We also performed CD68/LYVE-1 double immunoassays to confirm that these markers identify distinct, non-overlapping cellular populations in the tongue.
Coronal cryosections (8 µm) were collected from three regions of the anterior tongue between the tip and intermolar eminence, which is a distance of ~6 mm. We collected sections from the tongue tip (1.66 mm), mid (0.79 mm), and rear (0.79 mm) portions of anterior tongue, in similar proportion to our studies in rat.13,22,23 Sections were fixed, incubated in primary antibodies for 2 hr at room temperature or overnight at 4C. Secondary biotinylated antibodies (1:00; Jackson Immunoresearch, West Grove, PA) were applied to sections followed by avidin-biotin complex (1:100; Vector Labs, Burlingame, CA), and staining developed with diaminobenzidine (Thermo Fisher Scientific; Waltham, MA). We also used Alexa Fluor secondary antibodies to visualize co-labeling (1:1000; Molecular Probes/Thermo Fisher Scientific, Waltham, MA). Nonspecific staining resulting from incubation in species-specific non-immune sera was minimal for each primary antibody.
Image Analysis
LYVE-1 analyses were performed using MetaMorph software (MDS Analytical Technologies; Downington, PA) and a digital color camera (Cool Snap; Roper Scientific/Photometrics, Tuscon, AZ) as in previous studies.13,14,23 Images were captured at 25× from 4 regions of interest (ROI) placed in standard, non-overlapping, adjacent positions in the tip, mid, and rear tongue lamina propria and mucosa. The total standard area for images captured from these 3 lingual sampling regions was 587.01 mm2/mouse. LYVE-1 density values were similar between rostral-to-caudal sampling regions of tongue within treatment groups, and are presented as totals. We also analyzed standard-sized cutaneous ROIs of 195.67 mm2/mouse. To measure lymphatic density, we then thresholded immunopositive pixels to calculate the % LYVE-1+ pixels/total standard area. To control for potential staining variability between assays, we normalized LYVE-1 density by dividing each value by the mean control value from the same experiment and multiplying by 100.
We assessed vessel lymphatic vessel size by measuring their width across a transverse plane at 50×. The line region tool in MetaMorph was used to mark the width of lymphatic capillaries in single, straight segments bearing clearly defined abluminal borders, perpendicular to the longitudinal axis (Fig. 1A, red line). We measured 8 vessels in lingual regions placed in standardized regions of the dorsal mucosa in tip, mid, and rear of the tongue for a total of 24 vessels/animal. This method, which does not require dilated vascular lumens, has been used to measure the abluminal diameter of retinal capillaries and is suitable for assessing relative differences in vessel size. 24
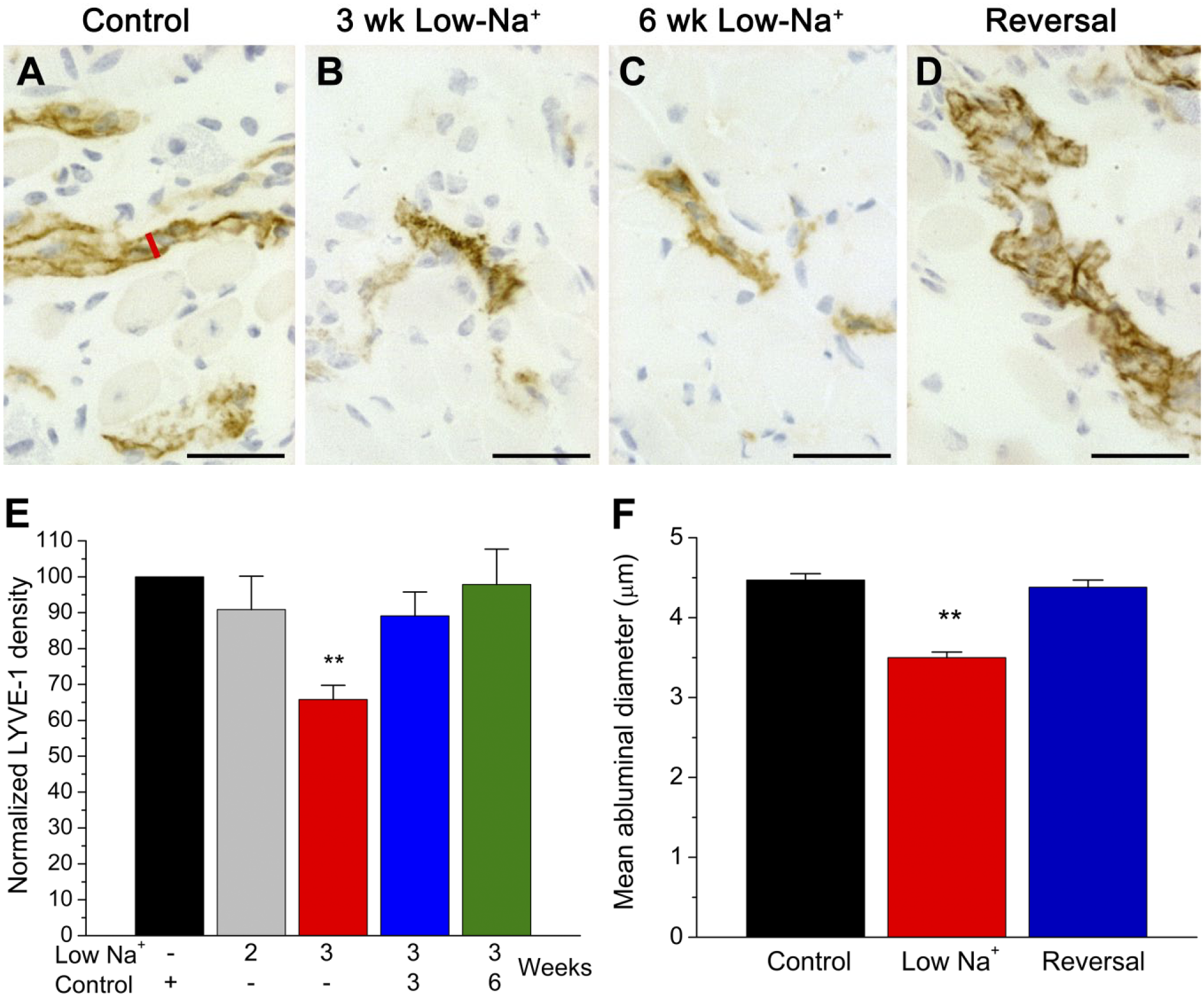
Dietary sodium restriction decreases LYVE-1+ lingual lymphatic vessel density and size. Images show brown, immunopositive lymphatic vessels in the lingual mucosa (A–D). Mice were maintained on a control diet, sodium restricted for 3 or 6 weeks, or returned to a control diet after sodium restriction (i.e., “Reversal”). The red line in (A) shows the transverse axis used to measure lymphatic vessel size. (E) Quantification of LYVE-1+ immunopositive pixels, which are normalized to within-experiment controls (black bar). LYVE-1 density was significantly decreased in mice fed a sodium-deficient diet for 3 weeks (H=24.18; **p<0.001). Reversal of the low-sodium diet restored lymphatic capillary density to control-like values. (F) The abluminal diameter of LYVE-1+ capillaries was significantly reduced after 3 weeks of dietary sodium restriction [F(2, 909) = 52.10, p=0.001]. Lymphatic capillary hypoplasia was reversed by 3 or 6 weeks of control feeding. Scale bars = 30 µm. Abbreviation: LYVE-1, lymphatic vessel endothelial hyaluronan receptor.
Using the same methods and standard regions as for LYVE-1 analyses, we quantified macrophages and blood vessels by thresholding lingual CD68 or PECAM-1+ pixels. PECAM-1 and CD68 densities are expressed as immunopositive pixels/standard area rather than normalized, because staining was performed in a single large assay for each marker. Images were minimally adjusted for brightness and color balance applied to the entire image.
Statistical Analyses
Prism software (version 3.0; Graphpad Software, Inc., La Jolla, CA) was used for all statistical analyses. Group differences in lingual and cutaneous LYVE-1 density and tongue weights were determined with non-parametric Kruskal-Wallis tests followed by Dunn’s posttests. We report the mean normalized LYVE-1 values from mice (n=2) fed a low-sodium diet for 6 weeks below but did not include this group in statistical analyses because of the limited number. The effect of dietary treatment on CD68+ macrophages was determined with Mann-Whitney tests. Results from analyses of abluminal diameter and PECAM-1+ blood vessel density were normally distributed as determined by Kolmogorov-Smirnov tests. In this case, groups were compared with one-way ANOVAs followed by Newman-Keuls posttests tests (i.e., diameter) or Student’s t-tests (i.e., PECAM-1 density). The α level was p≤0.05 for all analyses.
Results
LYVE-1+ immunoreactivity was absent from the lingual epithelium and taste buds, but present in the mucosa, lamina propria, and within fungiform papillae regardless of dietary treatment. 25 Lymphatic vessels were prominent in the lingual lamina propria and mucosa of control-fed mice (Fig. 1A) across the rostral-caudal axis of the tongue. In comparison, there were fewer, smaller LYVE-1+ lymphatic vessels in mice fed a sodium-deficient diet. The reduction in LYVE-1+ density emerged after 3 weeks of dietary sodium restriction (Fig. 1B and E), and was maintained in additional mice (n=2) fed a low-sodium diet for 6 weeks (Fig. 1C; mean 70.0% ± 18.54 SEM). We then tested whether normal lymphatic density is restored in mice returned to a control diet after 3 weeks of dietary sodium restriction. Indeed, the density of lymphatic vessels in the tongue returned to control-like levels after 3 or 6 weeks on control chow (Fig. 1D and E). LYVE-1 density was statistically similar in reversal groups, which were combined for further analyses.
We also measured the abluminal, or transverse, width of LYVE-1+ lymphatic vessels to determine dietary effects on size (Fig. 1F). 24 Lingual lymphatic vessels were significantly smaller in sodium-deficient versus control-fed mice, but returned to normal size when the diet was reversed. Together, the size, position in the dorsal mucosa, and lamina propria, and strong LYVE-1 immunoreactivity of lymphatic vessels sensitive to dietary sodium identifies them as capillaries.12,18,26
Reducing lymphatic capillary size and density could prevent efficient drainage of interstitial fluid and lead to edema. Yet, mean ± SD wet tongue weights were similar in mice fed a sodium-deficient diet for 3 weeks (0.047 ± 0.005 mg), 6 weeks (0.048 ± 0.003 mg), and control-fed mice (0.046 ± 0.006 mg; p>0.05; not shown). These results demonstrate that a low-sodium diet reversibly decreases lymphatic vessel density and size in the tongue without causing lymphedema.
We next determined whether the dietary effects extend to lingual blood vessels. As shown in Fig. 2A–C, however, there were no significant differences in PECAM-1+ blood vessel density in sodium-deficient compared with control-fed mice. PECAM-1 and LYVE-1 identified different vascular populations, regardless of dietary treatment (Fig. 2D). Thus, blood vessels are distinct from lymphatic vessels in the tongue, as expected,17,27 and are maintained in sodium-deficient animals.
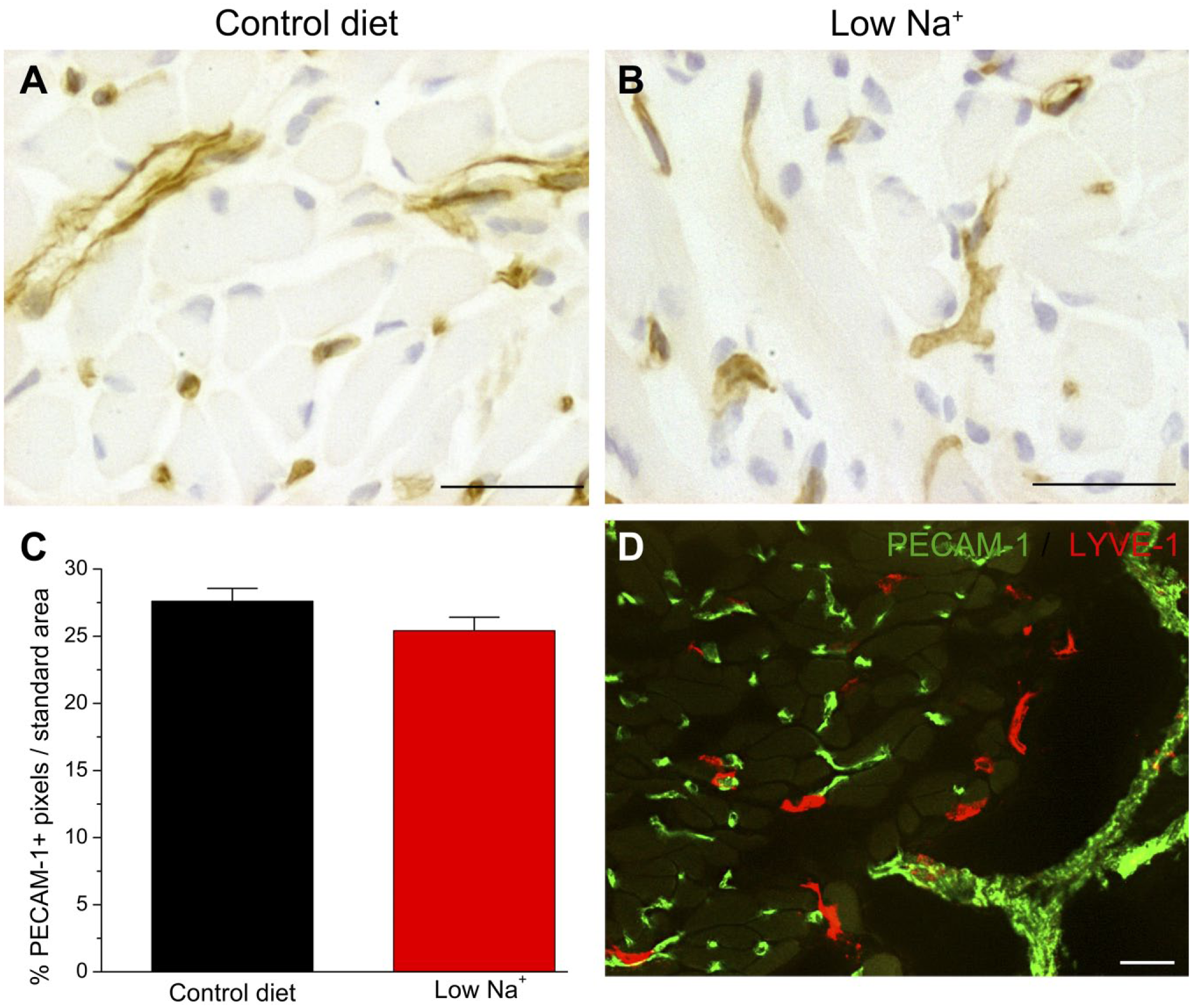
Dietary sodium deficiency did not alter lingual PECAM-1+ blood vessel density. Similar levels of PECAM-1 staining were observed in the tongues of (A) control-fed and (B) sodium-deficient mice. (C) The percentage of PECAM-1+ immunopositive pixels/standard area was not significantly different in mice on a control diet (n=10) compared with sodium-restricted mice (n=12; p>0.05). (D) PECAM-1+ and LYVE-1+ did not co-localize in the tongue in either dietary group, as shown in this mouse on the low-sodium diet for 3 weeks. Scale bars = 30 µm. Abbreviations: PECAM-1, anti-platelet endothelial cell adhesion molecule; LYVE-1+, lymphatic vessel endothelial hyaluronan receptor.
Our primary interest in this study was in the oral lymphatic vasculature, but we also measured dietary effects on lymphatic vessels in skin. As shown in Fig. 3A–B, cutaneous LYVE-1 density decreased in sodium-deficient mice, but was reversed by ≥3 weeks of control feeding (Fig. 3C and D). Thus, a low-sodium diet induced lymphatic vessel regression in the skin and tongue.
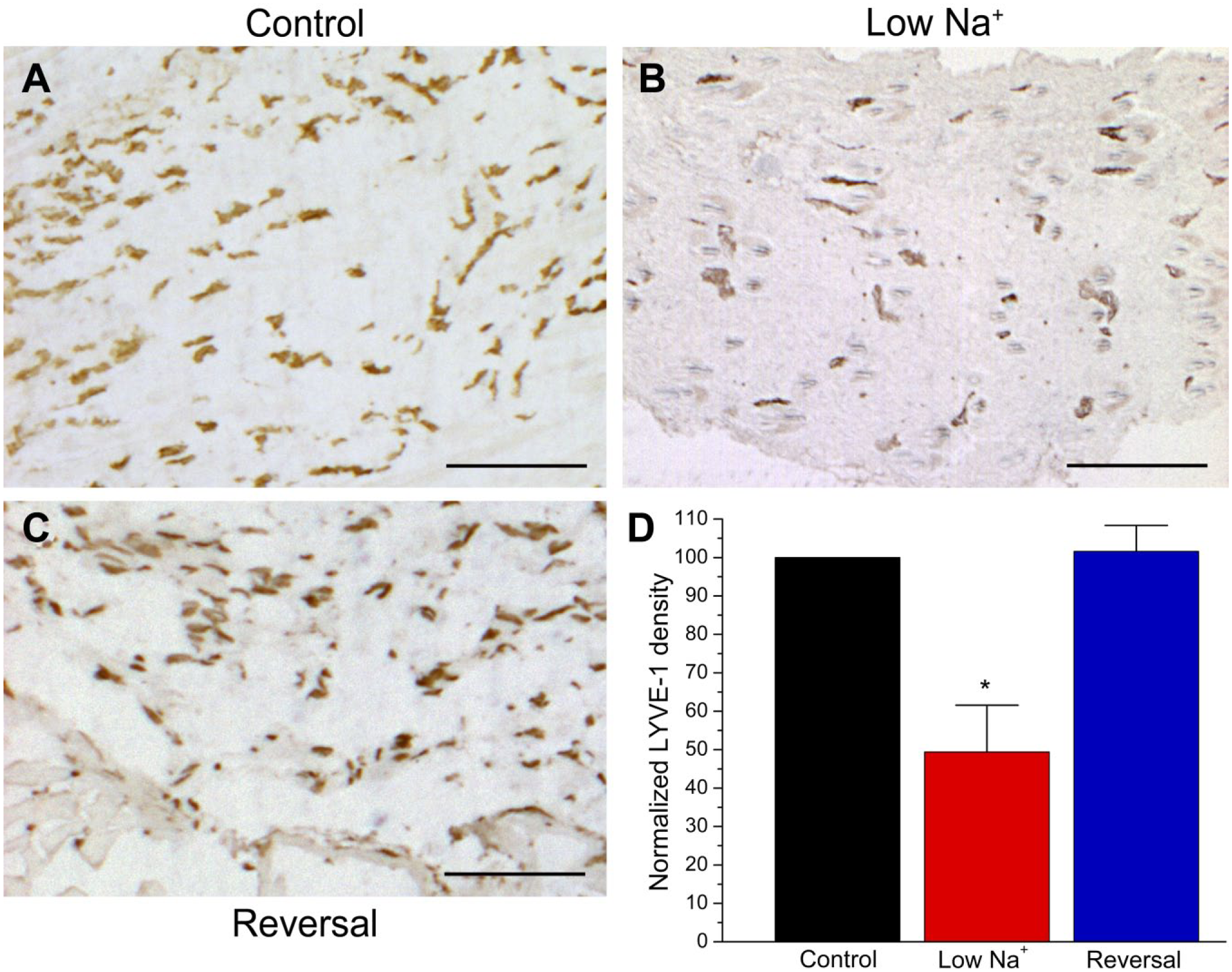
Decreased LYVE-1+ lymphatic vessel density in the skin of sodium-deficient mice. Cutaneous LYVE-1 staining following (A) control feeding, (B) 3 weeks on a low-sodium diet, and (C) 3 weeks of sodium restriction followed by 3 weeks on a control diet (i.e., “Reversal”). (D) Normalized LYVE-1 density was significantly reduced by 3 weeks on a low-sodium diet (n=4) versus a control diet (n=4), but restored by 3 or 6 weeks (n=4) of control feeding (H=7.43; *p=0.024). Scale bars = 200 µm. Abbreviation: LYVE-1, lymphatic vessel endothelial hyaluronan receptor.
Macrophages infiltrate tissues undergoing lymphatic hyperplasia induced by high dietary sodium, inflammation, and tumor. 16 We tested whether the inverse also occurs, leading to reduced macrophage density in sodium-deficient mice. CD68+ macrophages were located in similar lingual tissue compartments as lymphatic vessels (i.e., mucosal and lamina propria rather than epithelial). As shown in Fig. 4, macrophage density was not significantly different in the tongues of sodium-deficient versus control mice. Therefore, the regression of lingual lymphatic capillaries under low-sodium conditions may depend on distinct, macrophage-independent mechanisms compared with high-sodium induced lymphatic hyperplasia. 12
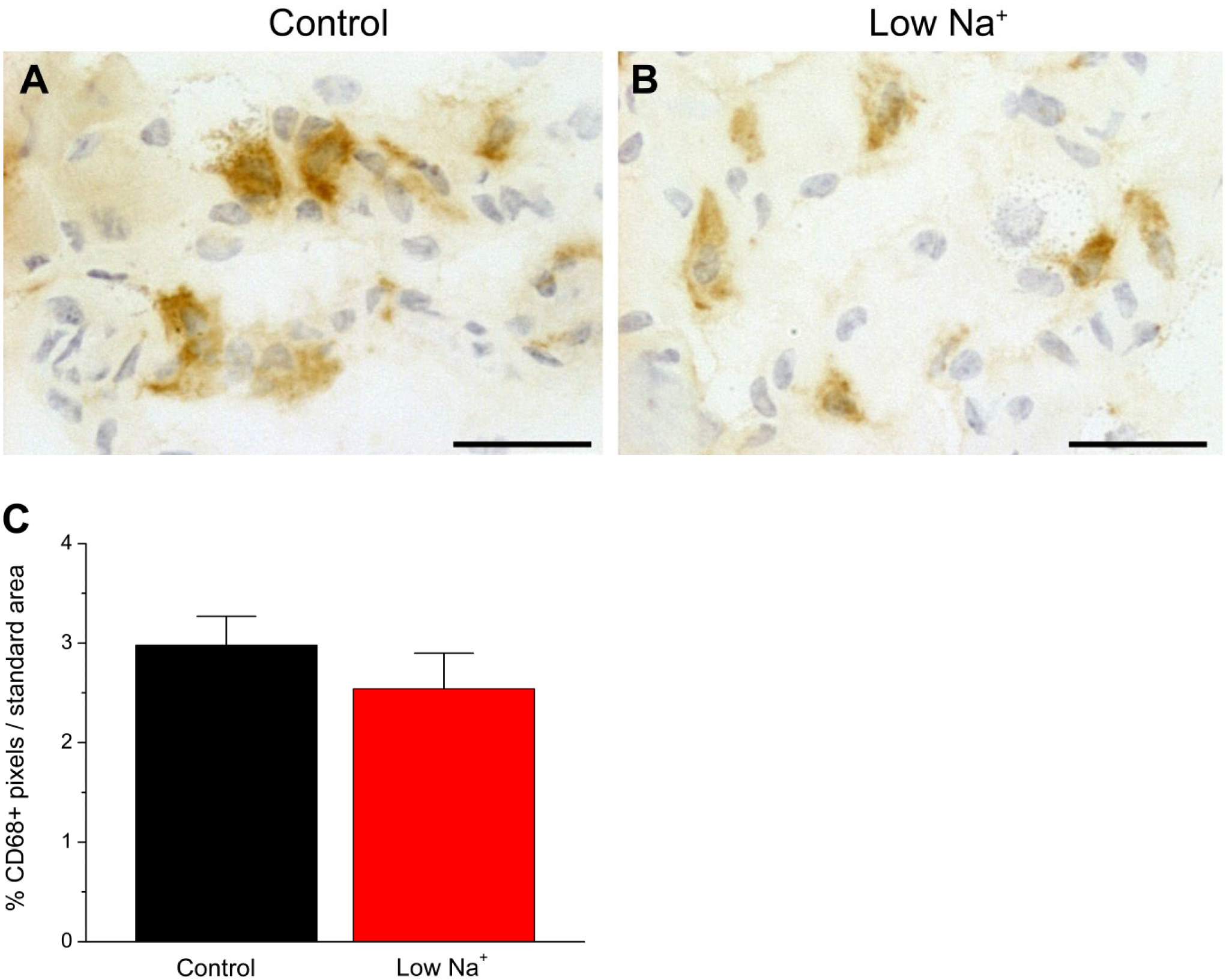
Dietary sodium restriction does not alter lingual macrophage density in the tongue. Brown, immunopositive CD68+ macrophages in mice fed a (A) control diet or (B) sodium restricted for 3 weeks. (C) CD68+ density was not significantly changed by dietary treatment (p>0.05). Scale bars = 30 µm.
Discussion
A relatively short period of dietary sodium restriction decreased the size and density of LYVE-1+ lymphatic capillaries in the tongue while sparing blood vessels. Moreover, the regression of lingual lymphatic capillaries is reversible upon restoration of normal dietary sodium levels. The shrinkage of lingual lymphatic vessel networks might, therefore, slow oral tumor metastases or treat oral inflammation. 11
Dietary sodium deprivation was shown decades ago to retard tumor growth and decrease whole-body extracellular fluid in rats implanted subcutaneously with B16 melanoma cells. 28 Our results suggest that lymphatic regression (or reduced tumor-induced lymphangiogenesis) might contribute to this effect of dietary sodium on tumor growth. The decreased volume of extracellular fluid in sodium-deprived animals may also explain why reducing lymphatic vessel size and density did not result in lingual edema in the current work.
High dietary sodium, tumors, and inflammation can stimulate lymphangiogenesis but conditions that cause lymphatic regression in adulthood are much less well-understood.1,2 The withdrawal or antagonism of growth signals such as vascular endothelial growth factor (VEGF)-C, which is important for blood and lymphatic vessel growth,11,29 or the engagement of negative signals, such as angiopoietin-2, are among the mechanisms hypothesized to shrink lymphatic networks. Macrophages play a critical role in lymphatic hyperplasia. 16 In animals on a high-sodium diet, the increased salt in cutaneous interstitial fluid recruits macrophages that secrete VEGF-C, in turn stimulating lymphatic vessel growth. 12 We demonstrate that tissue macrophage density in the tongue was unchanged by a low-sodium diet. 12 Low-sodium conditions may reduce VEGF-C release or trigger other functional changes in local macrophages that impact lymphatic capillaries without affecting their number.
Future preclinical studies are needed to determine the maximum level of dietary sodium effective in reducing lymphatic vessel density, mechanisms responsible for the regression of lymphatic networks, and whether sodium restriction slows the growth and spread of oral squamous cell carcinoma in animal models. Three-dimensional reconstructions of labeled or tracer-filled lingual lymphatic vessels might also reveal diet-induced changes in lymph capillary networks12,30–32 in addition to the relative changes in density and size reported here. This non-invasive dietary treatment may be useful as an adjuvant therapy for oral cancer or corneal transplant-induced lymphangiogenesis, which contributes to graft rejection. 33
Footnotes
Competing Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
LH performed experiments and image analyses and contributed technical information to the manuscript. LPM designed and performed experiments, analyzed results, and wrote the manuscript.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by National Institute on Deafness and Other Communication Disorders grant DC-005811 and by the Medical College of Georgia Intramural Grants Program.
