Abstract
Diagnostic electron microscopy is a useful technique for the identification of viruses associated with human, animal, or plant diseases. The size of virus structures requires a high optical resolution (i.e., about 1 nm), which, for a long time, was only provided by transmission electron microscopes operated at 60 kV and above. During the last decade, low-voltage electron microscopy has been improved and potentially provides an alternative to the use of high-voltage electron microscopy for diagnostic electron microscopy of viruses. Therefore, we have compared the imaging capabilities of three low-voltage electron microscopes, a scanning electron microscope equipped with a scanning transmission detector and two low-voltage transmission electron microscopes, operated at 25 kV, with the imaging capabilities of a high-voltage transmission electron microscope using different viruses in samples prepared by negative staining and ultrathin sectioning. All of the microscopes provided sufficient optical resolution for a recognition of the viruses tested. In ultrathin sections, ultrastructural details of virus genesis could be revealed. Speed of imaging was fast enough to allow rapid screening of diagnostic samples at a reasonable throughput. In summary, the results suggest that low-voltage microscopes are a suitable alternative to high-voltage transmission electron microscopes for diagnostic electron microscopy of viruses.
Keywords
Introduction
Despite the pivotal use of molecular diagnostics for the identification of pathogens associated with infectious diseases, diagnostic electron microscopy (EM) of pathogenic microorganisms still is relevant. In veterinary medicine, for instance, it is so far not possible to provide molecular testing for all pathogens potentially infecting the various animals relevant to man. 1 Moreover, the amount of unassigned viral matter in nature seems to be large (less than 10% of the viral sequences possess homology with published sequences), and the potential to modify viruses steadily increases. 2 Screening of samples by light microscopy and EM is an appropriate method to spot pathogens causing a disease.3,4 In human medicine, diagnostic EM of microorganisms is an accessory technique in particular cases, such as outbreak investigations, diseases of unknown etiology, or biological threats caused by criminals.5–7 The generic “catch-all” approach and the speed of diagnostic EM are particularly useful to obtain a fast overview about any pathogen present in significant numbers in patients as well as in environmental samples. 8 Disadvantages of the method are the comparatively low diagnostic specificity (i.e., detection of morphological groups above species level) and the significant financial investment in the rather complex infrastructure for EM (i.e., the microscope, devices for sample preparation, skilled operators). 4
Diagnostic EM of viruses needs an optical resolution of about 1 nm, which is roughly 20 times better than the smallest mammalian viruses known (i.e., the
Possible alternatives to the use of high-resolution transmission EM for diagnostic EM are field-emission scanning electron microscopes. Due to improvements in the scanning transmission detectors and electronics, an optical resolution below 1 nm can be achieved at reasonable scanning speed. Because of their high versatility, scanning electron microscopes (SEMs) are widely used and therefore available, and can be shared for different purposes (e.g., material and life science samples). However, whether scanning transmission imaging in a field-emission SEM can be used for diagnostic EM of viruses or not has not been investigated so far. With the release of a low-voltage transmission electron microscope (LV TEM; available in two variants: LVEM25, Delong Instruments; Brno, Czech Republic; and MiniTEM, Vironova AB; Stockholm, Sweden) a couple of years ago, another possible imaging alternative for diagnostic EM became available. The microscope is operated at a maximum of 25 kV with a specified optical resolution of 1 nm and makes use of the same technical concept as the 5 kV desktop electron microscope already produced by Delong Instruments for more than two decades (see Frank et al. 1994 for details of the LVEM5). 10 The microscope has a small footprint and needs only standard room/lab conditions as well as standard power supply.
To evaluate the imaging capabilities of low-voltage electron microscopes for diagnostic EM of viruses, we compared imaging of diagnostic reference samples using the LVEM25/MiniTEM and a field-emission SEM (equipped with a scanning transmission detector) with imaging by a high-voltage transmission electron microscope (HV TEM). Samples comprised virus suspensions prepared by negative staining and thin sections from plastic-embedded virus-infected cell cultures.
Materials and Methods
Virus Propagation and Cell Culture
Negative Staining of Viruses From Suspensions
Negative staining was performed according to a standard protocol previously described in detail. 11 Briefly, filmed EM grids were treated with alcian blue before virus suspensions were added and incubated for 10 min at room temperature. After washing with three drops of water, grids were placed on a drop of 0.5% phosphotungstic acid (PTA) or 0.5% uranyl acetate (UA) for a second and blotted with filter paper.
Thin Sectioning of Virus-infected Cells
Virus-infected cell cultures were fixed with 2.5% glutaraldehyde in 0.05 M Hepes buffer (pH: 7.2) and embedded in epon resin according to a standard procedure, 11 which included postfixation in osmium tetroxide, tannic acid treatment, and en bloc contrasting with UA. Ultrathin sections (60–70 nm) were produced using a Leica UC7 microtome, collected on 300 mesh copper grids, poststained with 2% UA and 1% lead citrate, and were stabilized with a thin carbon layer of about 10 nm, if not noted otherwise.
EM
Imaging with the HV TEM JEOL JEM 2100 was used as a reference. The microscope was operated at 200 kV, and images were recorded with an EMSIS Veleta CCD camera at 2048 × 2048 pixel. Low-voltage EM was performed using the following microscopes:
FEI/ThermoFisher Teneo Volume Scope SEM, equipped with a scanning transmission EM detector (STEM3+): The microscope was operated at 30 kV and 0.1 nA beam current using the bright-field mode of the detector. For screening of samples, a scanning resolution of 768 × 512 pixel with 200 ns dwell time per pixel was used. Images were recorded at 3072 × 2048 pixel with 3 µs dwell time per pixel.
Delong Instruments LVEM25 LV TEM: The microscope was operated at 25 kV in bright-field mode or at 15 kV in STEM mode. Images were recorded at 2560 × 2160 pixel (bright-field mode) and 2048 × 2048 pixel (STEM mode).
Vironova AB MiniTEM LV TEM: The microscope was operated at 25 kV, and images were recorded at 2048 × 2048 pixel.
For further details and special features of all microscopes used in this study, see Table 1.
Technical Specifications of the Electron Microscopes Used in This Study.
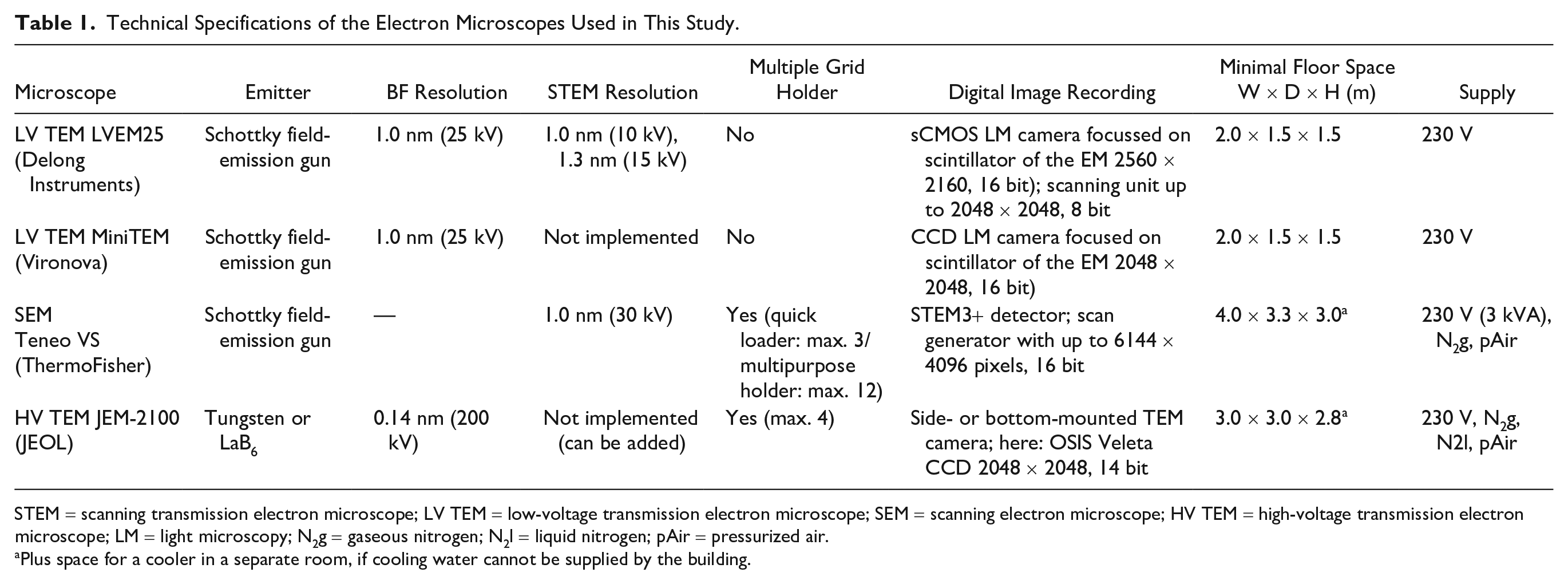
STEM = scanning transmission electron microscope; LV TEM = low-voltage transmission electron microscope; SEM = scanning electron microscope; HV TEM = high-voltage transmission electron microscope; LM = light microscopy; N2g = gaseous nitrogen; N2l = liquid nitrogen; pAir = pressurized air.
Plus space for a cooler in a separate room, if cooling water cannot be supplied by the building.
The small size of LVEM25 and MiniTEM is achieved by using a combination of electro- and magnetostatic lenses, which need no water cooling, in a compact column pumped by three ion getter pumping units. The image generated by the magnetostatic objective lens is projected onto a YAG (yttrium aluminum garnet) luminescent screen and is, thereby, converted into a visible image, which is further magnified with a light microscope objective and recorded by a light microscopy camera.
Images presented in the article were adjusted for brightness and contrast, and slightly filtered by a Gaussian filter (radius = 0.2–0.5 pixel), followed by a sharpening filter (unsharp mask with maximal 65% and a radius of 1 pixel) using Adobe Photoshop CS6, if not stated otherwise. The unprocessed (raw) image files are available at the data repository Zenodo (http://doi.org/10.5281/zenodo.3768447).
Results
Technical Specifications of the Microscopes
The electron microscopes used in this comparative study differed regarding their technical specifications. Table 1 gives an overview about the specifications which are relevant for diagnostic EM of viruses. Besides this information, a few other aspects are worth to mention. For testing, we operated the LV TEMs in a standard EM room at environmental conditions well within the specifications of a HV TEM and in a 3 m distance to the JEOL HV TEM. We never observed any interference between the microscopes. During high-resolution imaging with the LV TEMs, floor vibrations, such as induced by walking around the microscope, had to be avoided to prevent image distortions. Video-rate imaging was possible with all microscopes and specified cameras used in this study. To achieve a high image refresh rate at a sufficient image quality, image resolution was reduced by pixel binning (TEM bright-field mode) or by adjusting the resolution of the scan generator, and/or the scanning speed (SEM or TEM STEM mode).
Sample exchange could be performed within 3 min with all microscopes tested. However, with moist specimens, such as freshly prepared samples from negative staining or at humid air conditions, pumping time during specimen exchange of the LV TEMs occasionally increased (up to a maximum of 15 min), most probably because the vacuum in the airlock needed to be improved before connecting it with the column, which is directly connected with the field-emission gun (i.e., without separation by a valve).
All of the microscopes provided a graphical computer user interface for operation of the microscope. Operation of the LV TEMs was intuitive and quickly to learn for experienced TEM users. Most of the alignments were done by auto-functions, which worked out sufficiently if the microscope has been aligned properly by the field service. During work, operators could concentrate on screening the sample with few adjustments of the illumination. The HV TEM and the SEM possessed a more complex computer user interface (more menu entries and layers) than the LV TEMs, because they offered more functions and user interactions than the LV TEMs. The MiniTEM did not make use of manual controls by a separate panel as all of the other microscopes and relied only on computer mouse and keyboard controls. Automation of imaging tasks was possible with the SEM (using the MAPS software) and with the MiniTEM, which used an inbuilt macro generator for automated imaging and image analysis. However, the automated imaging capabilities were not evaluated in this study. The price of the different microscopes varies depending on their individual equipment and country of purchase. The field-emission SEM and the LaB6 HV TEM are usually more or less in the same price class, while the LV TEMs, especially the LVEM25, can be purchased at a significantly lower price.
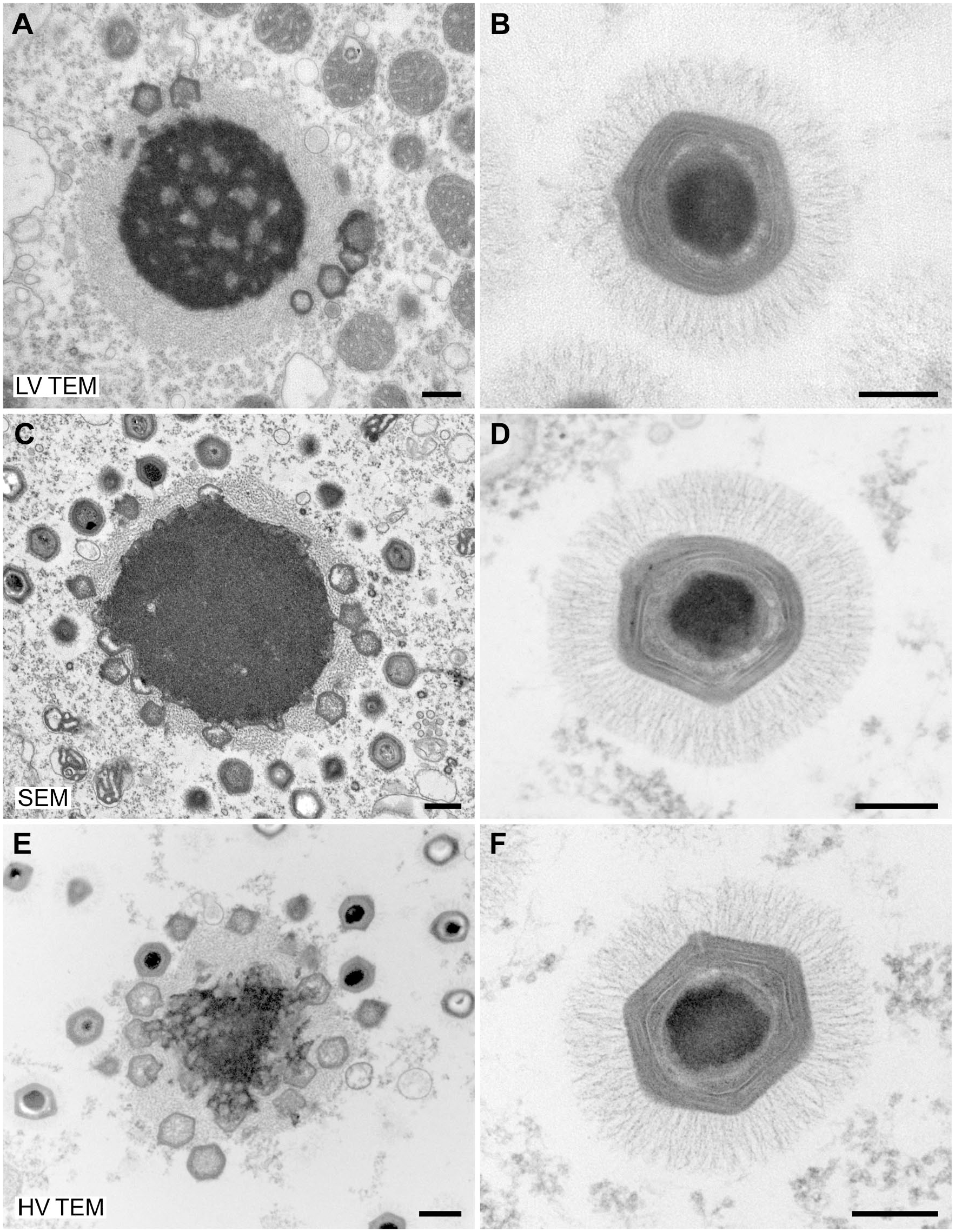
Negative Staining EM of Viruses
Negative staining EM is the fastest method for diagnostic EM of viruses.
6
For evaluation of the imaging capabilities of low-voltage electron microscopes, samples of
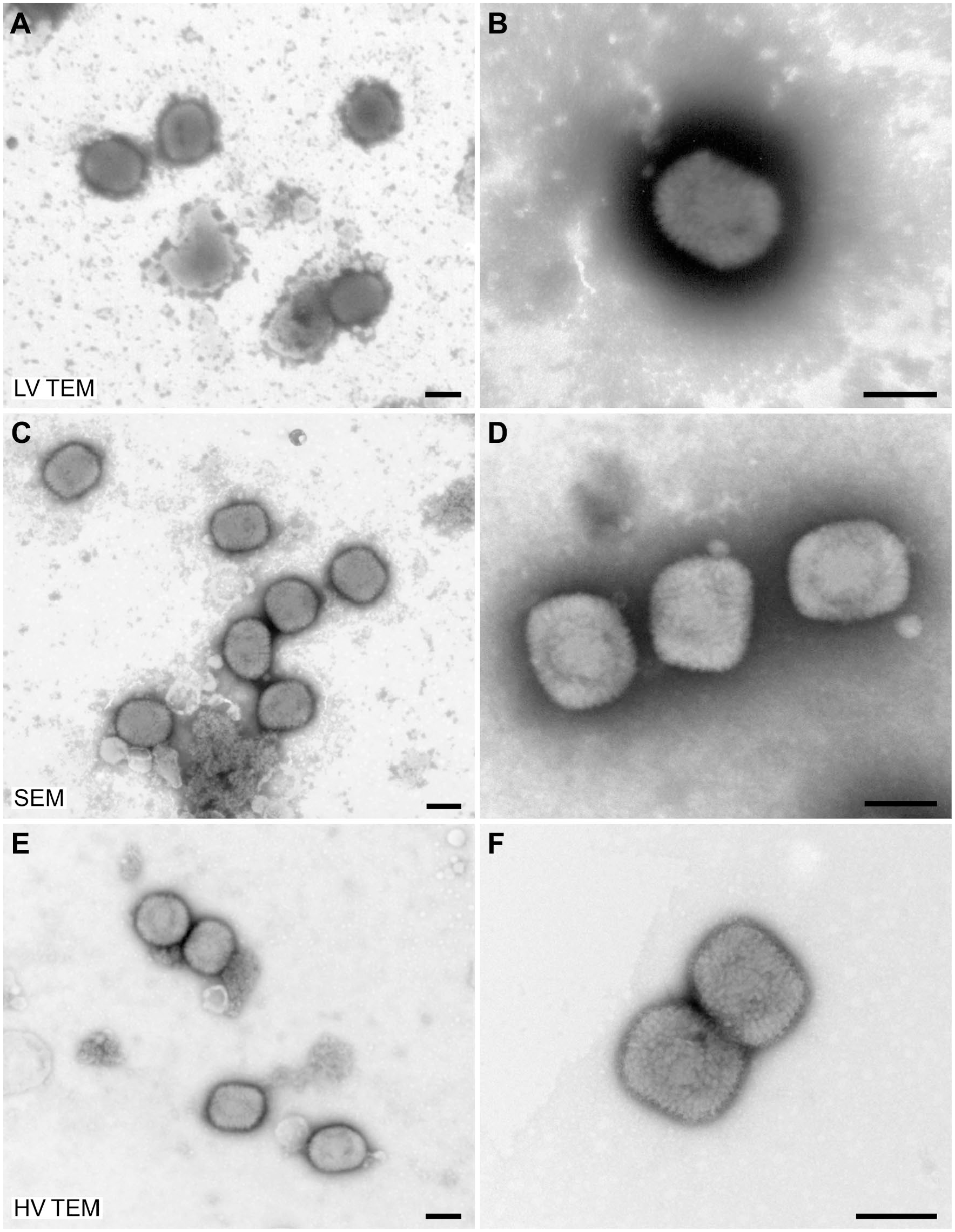
Negative staining of
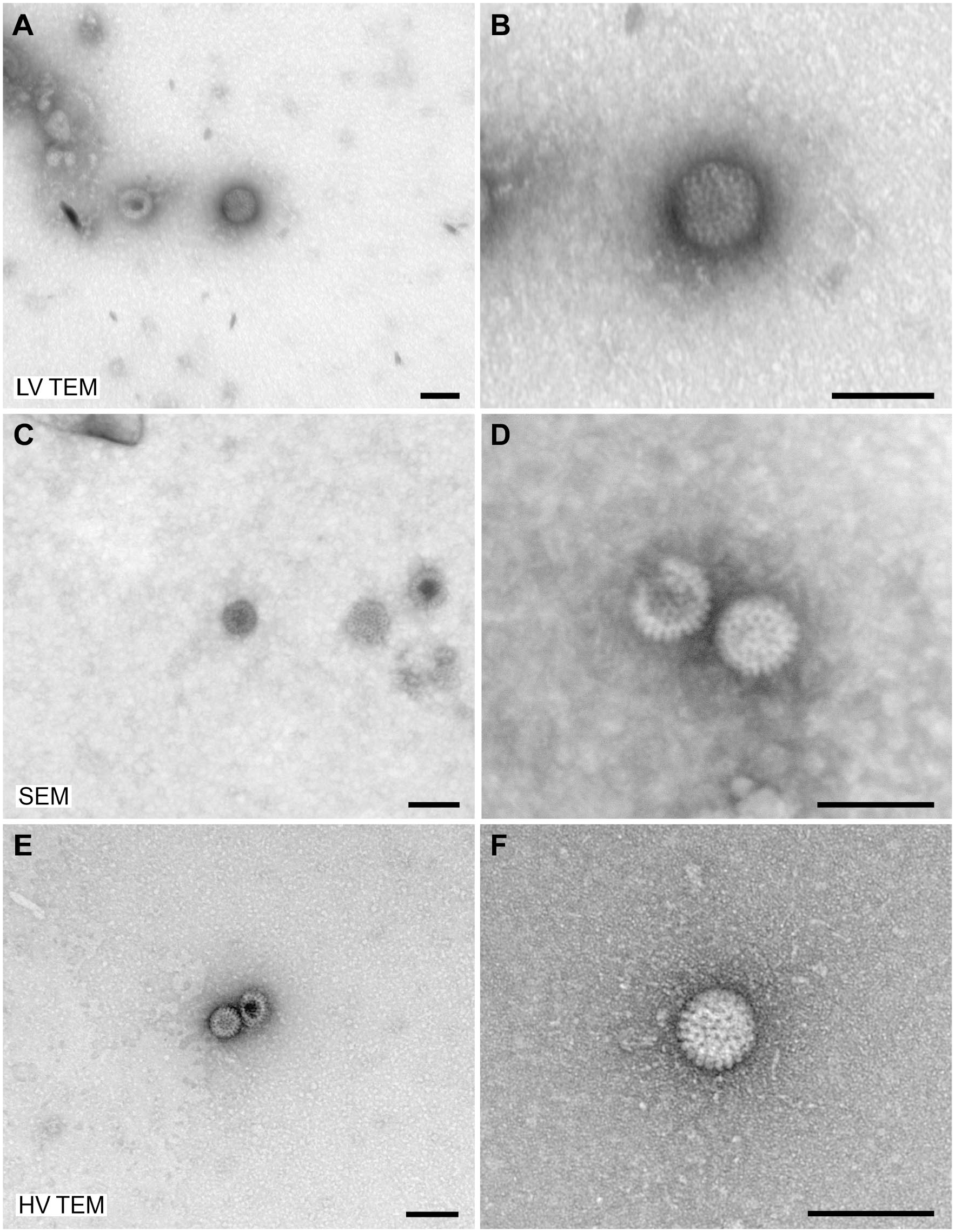
Negative staining of a
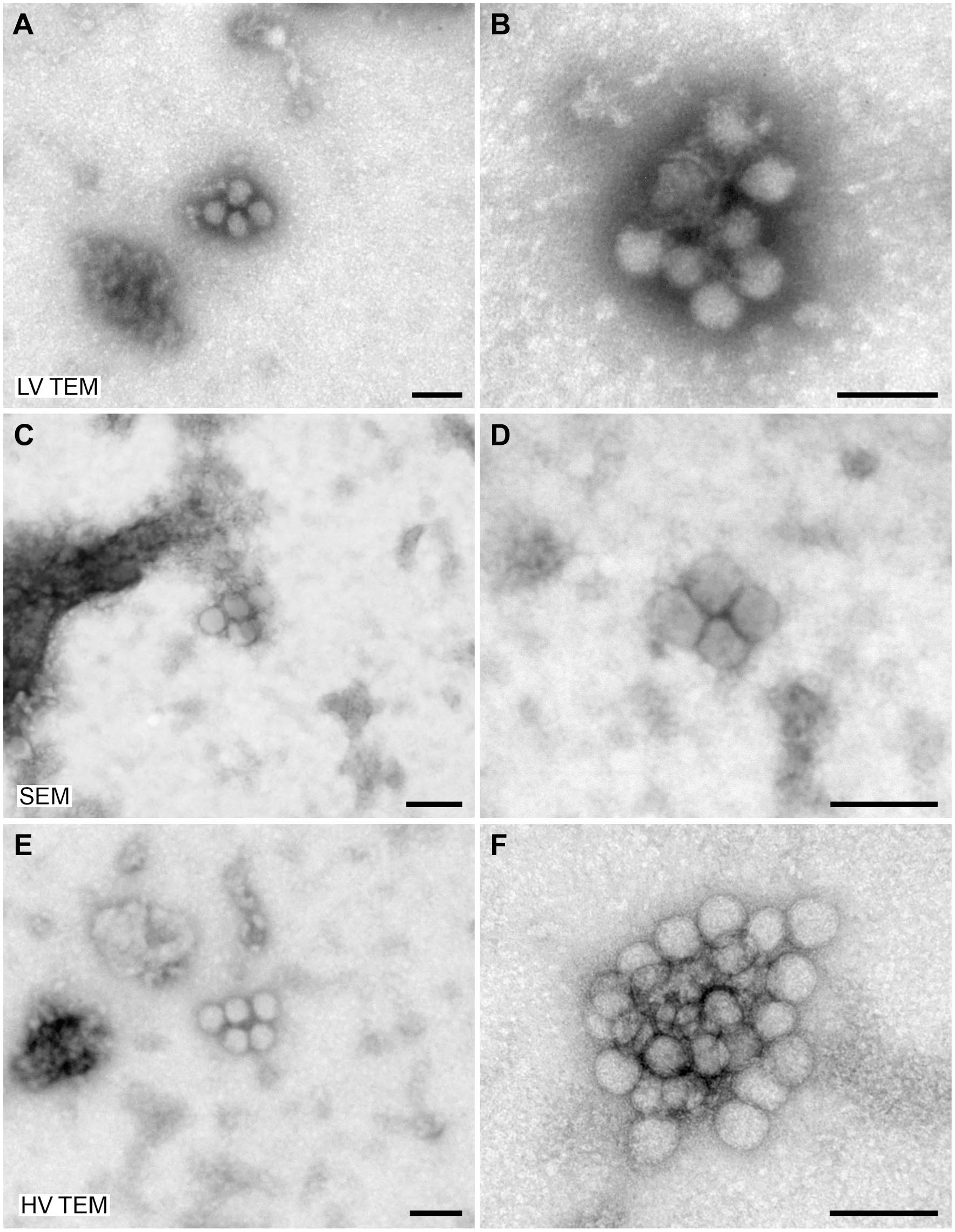
Negative staining of
To demonstrate the practical resolution of the different microscopes, we used TMV as a test organism. TMV provides two significant structural features which can be used as rulers: (1) the central channel with a width of maximally 4 nm, 12 and (2) the layer lines of the capsid which have a pitch of 2.3 nm.12,13 The central channel of the virus capsid was visible with all microscope tested while the layer lines of the capsid could only be detected convincingly with the HV TEM (Supplementary Fig. 1).
Speed usually is an important factor in diagnostic EM of viruses. The bright-field transmission EM mode of the HV TEM and of the LV TEMs allowed rapid screening of the samples at the highest possible image quality. The STEM mode of the SEM required finding a compromise between image resolution and speed of image refresh for achieving a reasonable sample screening speed. In practice, it was necessary to switch between imaging settings for screening (high image refresh rate, low resolution) and for closer inspection of particles of interest (low image refresh rate, high resolution), which slowed down the screening of a sample. Moreover, at higher magnification, careful correction of astigmatism had to be applied, which needed additional time. However, with a little bit of training this time, loss could be reduced to a minimum (max. 20% more time needed than with TEM bright-field mode).
Thin-section EM of Virus-infected Cell Cultures
Thin-section EM is used to visualize interior structures of viruses or their life cycle within host cells. Both virus structure and their localization in the different cell compartments help to identify viruses.6,14 We used thin sections (60–70 nm, on naked grids, carbon coated) of cells infected with either a mimivirus or a flavivirus to compare imaging by low-voltage microscopes with imaging by high-voltage TEM. Imaging of sections was possible with all of the microscopes without significant beam-induced rupture of sections. Virus structures and ultrastructural details of the cells which were relevant to reveal virus genesis could be recognized (Figs. 4 and 5). Image quality (resolution, sharpness) obtained with the low-voltage microscopes suffered at high magnification without precluding the recognition of details that are relevant for the diagnosis. Regarding speed of section screening, similar results were obtained as for the imaging of negative staining samples (see above).
Ultrathin sections through an amebocyte of
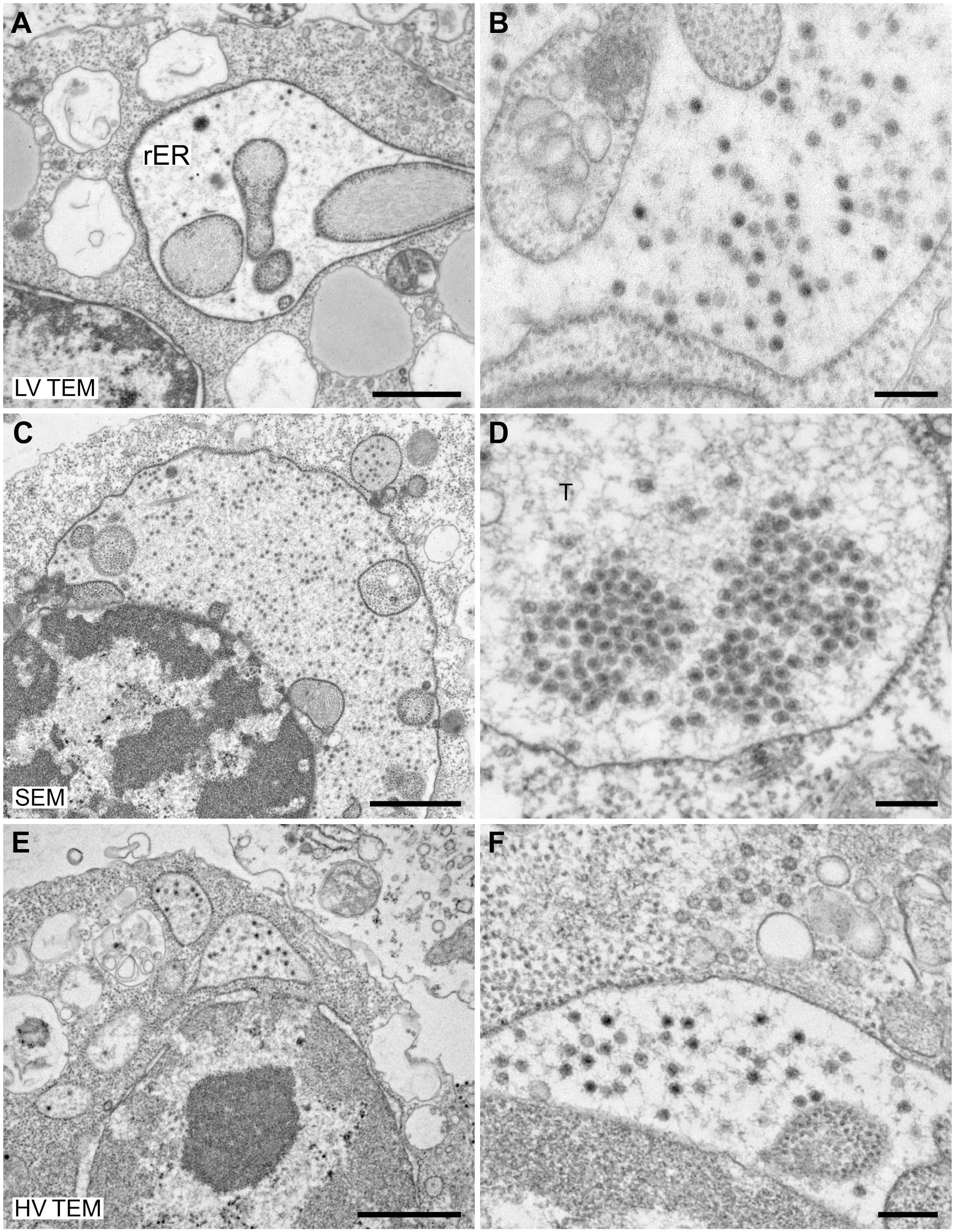
Ultrathin sections through C6/36 insect cells infected with
A remarkable advantage of the low-voltage TEMs was, that on-section contrasting (poststaining) with heavy metals (UA and lead citrate) was not necessary with the samples used in this study (Fig. 5A and B; Supplementary Fig. 2), which involved postfixation with osmium tetroxide and block contrasting by UA. Using HV TEM at 200 kV and STEM mode of the SEM at 30 kV for the imaging of sections without poststaining also allowed the recognition of ultrastructural details but at significantly lower contrast than imaging with poststaining (Supplementary Figs. 3 and 4).
Particular Aspects of Low-voltage EM
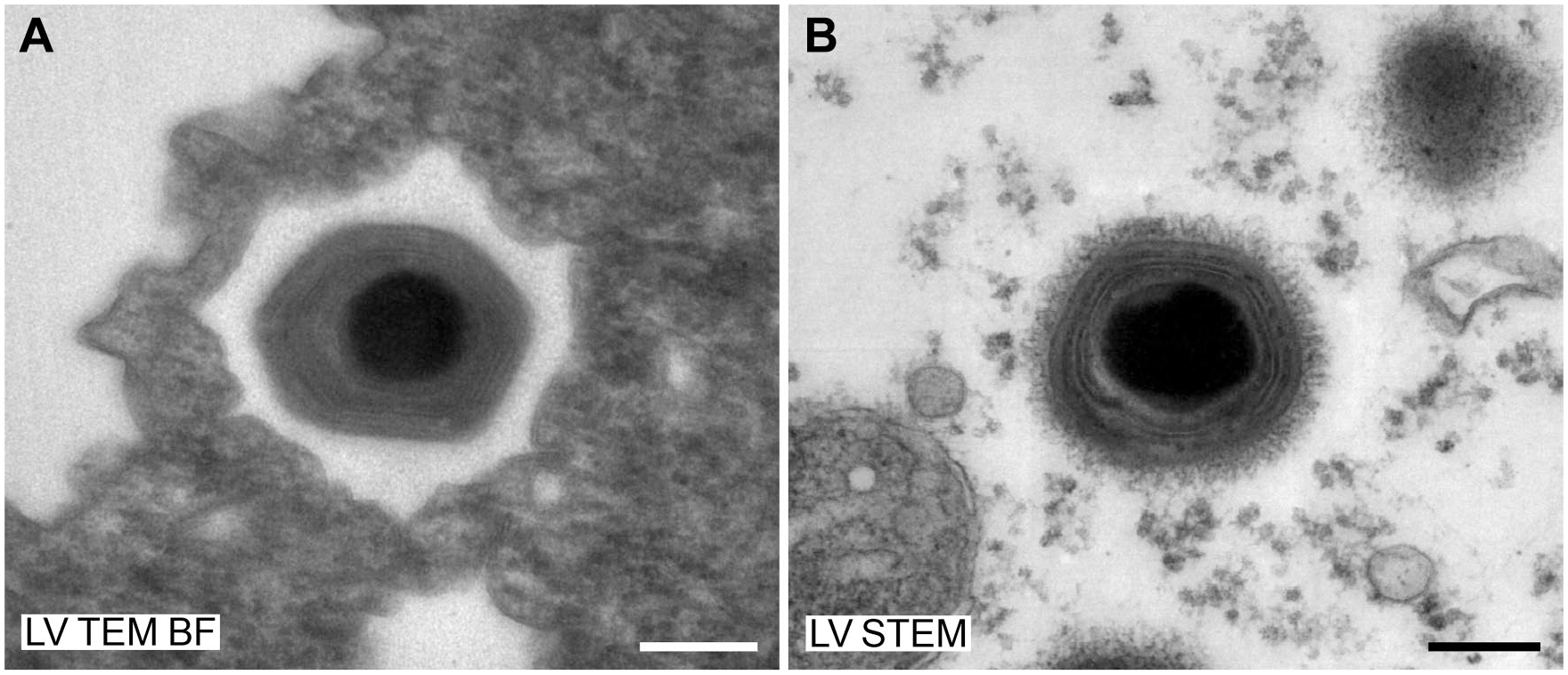
Imaging at a certain lens configuration (the lowest level of the high-magnification bright-field TEM mode) with the low-voltage TEMs sometimes suffered from image distortions visible at the periphery of the camera observation area (Fig. 6A). However, this effect was restricted to a particular magnification range and did not interfere with the recognition of the viruses.
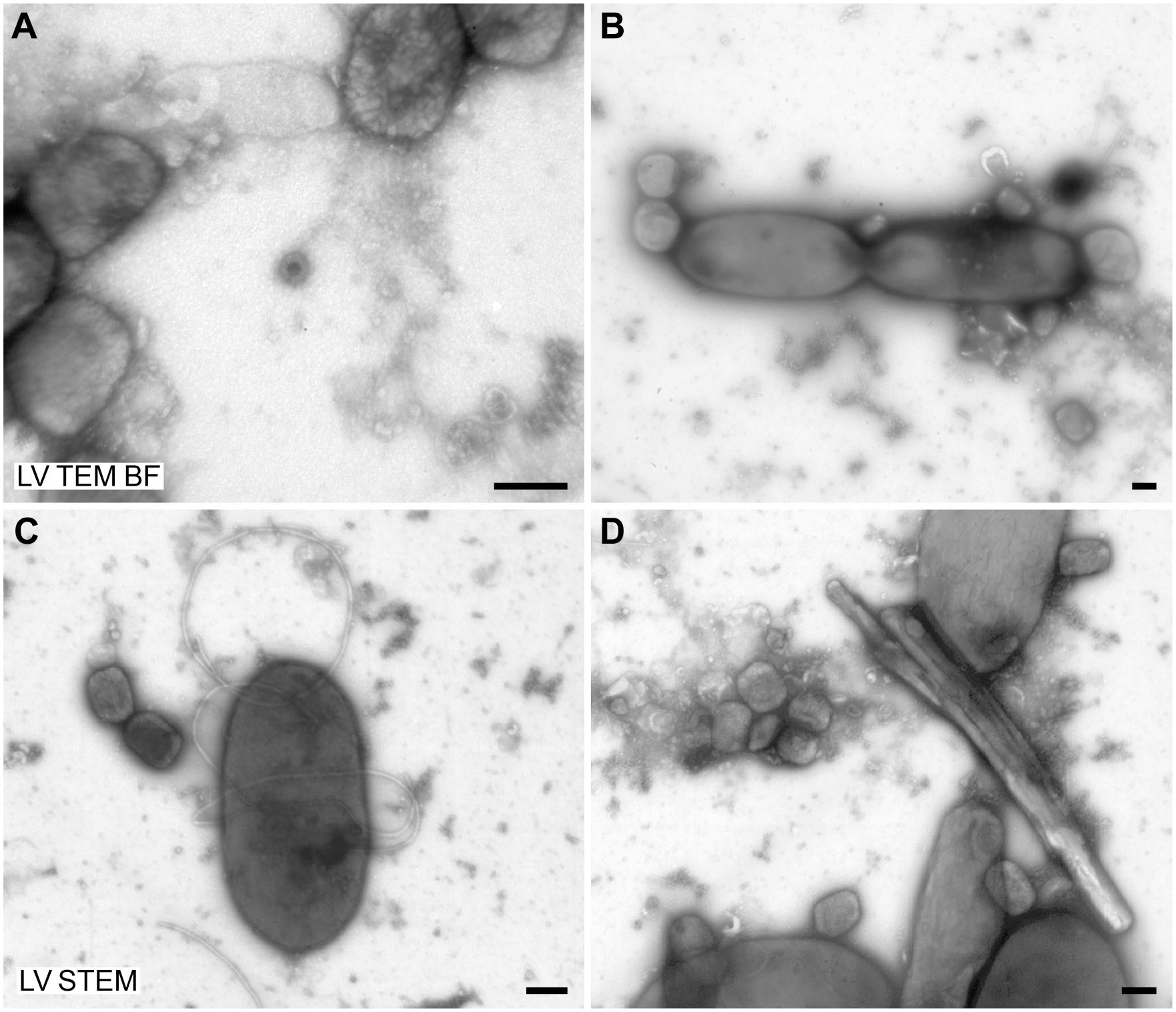
Special aspects of low-voltage TEM with the LVEM25/MiniTEM. Negative staining (0.5% UA) of a human stool sample, which was supplemented with
Another remarkable point is that bright-field TEM imaging of thicker/denser objects with the LV TEMs occasionally caused a slight image blurring. In negative staining EM, sometimes even
Ultrathin sections of
It is of note that the STEM imaging with the SEM was possible with all samples tested, regardless of their thickness or density. At higher magnification, adjusting the imaging parameters, including correction of astigmatism, usually was more laborious and time-consuming than at lower magnification to get sufficient image resolution, which extended the observation time needed for inspection of a sample (see above). One drawback of the STEM imaging with the SEM was, that scanning at high-resolution and low-speed left scan traces in the sample, usually in form of darkened imaging areas (scanning window) or dark lines (Supplementary Fig. 5). To a lesser extent, scan traces were also observed with the STEM mode of the LVEM25. As a further validation of the STEM mode imaging for diagnostic EM of viruses with the SEM, we successfully applied blinded testing of virus test samples (Supplementary Fig. 6) and used it for real diagnostic samples, without noticing any differences regarding to the diagnosis if compared with results obtained by HV TEM.
Discussion
The results of this study indicate that resolution, image quality, and speed of the tested low-voltage microscopes are sufficient for diagnostic EM of viruses in negative staining samples and in ultrathin sections, because small (e.g., flaviviruses) and large viruses (e.g., poxviruses) could be imaged and identified. However, neither the smallest mammalian viruses (i.e., circo- and parvoviruses) were investigated, nor a systematic blinded testing of samples or wider application on patient samples was done, and therefore, it is yet unclear whether this conclusion can be supported by diagnostic practice. Further tests should be expanded to a panel of diagnostic samples and parallel examination of the microscope performance using equally trained operators. At least, the appearance of structural details, such as rotavirus substructure, fine surface structure of flaviviruses, or the visualization of the central channel (4 nm wide) in TMV capsids, demonstrates that the optical resolution of the low-voltage microscopes is sufficient for revealing fine ultrastructural details well below 10 nm in size. Thus, it is likely that even the smallest features relevant to diagnostic EM of viruses should be resolved.
Image quality was superior with the HV TEM than with the low-voltage microscopes used in this study, especially at higher magnification, which can be related to their better optical resolution in comparison with the low-voltage EMs. At low or mid magnification, differences between high-voltage and low-voltage microscopes were less obvious and sometimes hard to detect at first glance. The LV TEMs occasionally revealed image distortions at the periphery of the field of view in the low range of the high-magnification bright-field TEM mode, which most probably resulted from a cumulative effect of chromatic aberration and high projection angles of the second projector lens (personal communication by Michal Drštička, Delong Instruments). However, this effect was restricted to the special lens configuration used in this particular magnification range if thicker/denser sample areas were inspected and did not interfere with the recognition of virus structure.
A remarkable advantage of the low-voltage TEM is that ultrathin sections of samples prepared with osmium tetroxide and block contrast did not need any additional contrasting for imaging of structural detail. This gain of contrast is a well-known effect of imaging with low-voltage accelerated (i.e., slow) electrons, which are scattered at higher angles than faster electrons and, thereby, increasing the number of scattered electrons excluded from the detector (Egerton 2016, chapter 4). 15 A drawback of the imaging with slow electrons is that the probability of inelastic scattering is higher than in comparison with imaging with fast electrons, which causes heat accumulation in the sample and also image blurring due to increased chromatic aberration (Egerton 2016, chapters 2 & 4). 15 Both effects increase with the thickness of the sample (Egerton 2016, chapter 4). 15 However, heat accumulation was not critical in the experiments performed in this study (e.g., we did not observe significant drift or rupture of samples). One reason for this observation could be that we used stable carbon-reinforced films for negative staining and carbon coating of the ultrathin sections, which efficiently dissipates the introduced heat across the surface. However, our samples were thin and therefore the amount of elastic scattering, which generates heat in the sample, low. It is of note that ultrathin sections of about 200 nm on plastic film ruptured quickly by bright-field electron beam illumination in the LV TEMs.
Image blurring was observed with low-voltage TEM and the bright-field mode in particular cases related to sample thickness/density. One case is associated with a specific lens configuration and probably thick/dense sample regions, mentioned above, which did not interfere with the diagnosis. The other cases are related to the general thickness/density of the sample and were rather independent of the magnification. In negatively stained samples using standard grids (plastic filmed and coated with carbon), larger particles, such as thick viruses and bacteria, sometimes appeared slightly blurred and the same could be observed for ultrathin sections on filmed grids. The reason for this effect can be related to increased probability of inelastic scattering and chromatic aberration in comparison to imaging with faster electrons (see above). To circumvent this problem, grids with thin carbon films could be used for negative staining instead of the plastic-filmed, carbon-reinforced grids, and ultrathin sections can be placed on naked grids, as we did in our study. This would reduce the sample thickness and prevent image blurring for most relevant samples in diagnostic EM. Moreover, a thinner overall sample thickness could also be beneficial for the practical spatial resolution (see above) and therefore should be considered in further studies in more detail.
Our experiments allow the conclusion that STEM systems, which can be installed in most modern SEMs, are quick and sensitive enough for fast screening of diagnostic virus samples and are able to visualize smallest detail at sufficient resolution. While this could be already expected from reports in the literature,16,17 the performance of the STEM mode of the low-voltage TEM LVEM25 was surprising. It was operated at 15 kV and also provided sufficient speed and resolution for diagnostic purposes. Moreover, thicker samples could be visualized without image blurring, which can be related to fact that inelastic scattering in the STEM mode has no effect because electrons do not pass another lens after having crossed the specimen (Egerton 2016, p. 84 and 115), 15 in contrast to imaging in bright-field mode. Thus, the STEM mode is a valuable addition for low-voltage bright-field TEMs tested in this study.
The new low-voltage TEMs LVEM25/MiniTEM provide significant improvement of the imaging performance in comparison with the low-voltage desktop TEM LVEM5 which is operated at 5 kV. 10 The LVEM5 uses the same core technology than the LVEM25 and is much smaller than the LVEM25/MiniTEM, but penetration depth of the electron beam is limited to very thin samples such as nanoparticles, 18 small viruses (Michael Laue, unpublished observations), or very thin sections,19,20 which prevents its routine usage in diagnostic EM of viruses. Although the LVEM25/MiniTEM can be used for diagnostic EM of viruses, a few features should be optimized. The airlock time of very wet samples or in case of high air humidity is very long (i.e., > 3 min, up to 15 min) and should be shortened. A multisample holder could somehow compensate for the rather long airlock time. In addition, the mechanical dampening of the system could be improved. One interesting feature, which we did not investigate in this study, was the automation module of the MiniTEM. This module allows automation of image recording and automated image analysis, including pattern recognition. Preliminary testing showed that setting up an automated task and conducting it takes some time and seems to be useful for the automated analysis of multiple repetitions of similar samples but not for diagnostic EM. However, as the field of image analysis and automation is rapidly developing, further improvement is expected in near future. Recently, Vironova AB demonstrated virus recognition based on machine learning for projects of quality assurance. 21
In summary, we could show that low-voltage EM using a field-emission SEM with an STEM detector and two new LV TEMs is suitable for diagnostic EM of viruses, which expands the number of useful microscope types for application in this particular field. Moreover, the promising results should encourage other fields of diagnostic EM, such as ultrastructural pathology, to validate low-voltage EM for their purposes, because it is very likely that the performance of the low-voltage EMs tested in this study will be sufficient.
Supplemental Material
2020-00018R1_Production_Supplemental_Data_online_supp – Supplemental material for Diagnostic Electron Microscopy of Viruses With Low-voltage Electron Microscopes
Supplemental material, 2020-00018R1_Production_Supplemental_Data_online_supp for Diagnostic Electron Microscopy of Viruses With Low-voltage Electron Microscopes by Lars Möller, Gudrun Holland and Michael Laue in Journal of Histochemistry & Cytochemistry
Footnotes
Acknowledgements
This work was only possible with the support of Delong Instruments (Brno, Czech Republic) and Vironova AB (Stockholm, Sweden), which allowed us to test the low-voltage microscopes LVEM25 and MiniTEM in our lab environment for some time. We are especially grateful to Eva Coufalova, Michal Drštička, and Jiří Zelinka from Delong Instruments and Ida-Maria Sintorn, Gustaf Kylberg and Lars Haag from Vironova AB for their support. The viruses were propagated by our colleagues at the Robert Koch Institute. We would like to thank Andreas Nitsche and Silvia Muschter (
Competing Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The authors prepared this work within the scope of their employment with the German government.
Author Contributions
LM, GH, and ML performed the microscopy, analyzed, and interpreted the results. LM and ML designed the study. ML wrote the paper. All authors have read and approved the final manuscript.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was conducted as a regular research project on diagnostic electron microscopy by the Robert Koch Institute which is funded by the German government.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
