Abstract
Deleted in malignant brain tumors 1 (DMBT1) is part of the innate immune system and is expressed on mucosal surfaces in various tissues throughout the human body. However, to date, the localization of DMBT1 has not been investigated systematically and comprehensively in normal human tissues. In this study, we analyzed the mRNA expression of DMBT1 in human tissue by quantitative real-time PCR and examined its localization and distribution in the tissue by immunohistochemical staining using the monoclonal DMBT1 antibody HYB213-6. Anti-ovalbumin was used as an isotype control. The highest level of mRNA expression of DMBT1 was found in the small intestine, and the expression level was high throughout the luminal digestive tract. The expression of DMBT1 was especially high in the luminal digestive tract and salivary glands. The lowest expression level was found in the spleen. Immunohistochemical staining showed a high expression level of DMBT1 on mucosal surfaces throughout the body. There was a clear correlation between the mRNA expression and immunohistochemical expression of DMBT1 in the tissue. DMBT1 is strongly expressed on mucosal surfaces and in salivary glands
Keywords
Introduction
Deleted in malignant brain tumors 1 (DMBT1), also known as gp340 and salivary scavenger and agglutinin (SALSA), is a secreted scavenger protein with functions in mucosal innate immunity and in epithelial differentiation and regeneration.1 –3 DMBT1 plays an important role as an innate immune defense molecule and inhibitor of infections and binds to lipopolysaccharides (LPS), lipoteichoic acid (LTA), and microorganisms (such as Helicobacter pylori, Pseudomonas aeruginosa, human immunodeficiency virus type 1, and influenza A virus), and interacts with innate immunoproteins, such as surfactant proteins A and D.4–11 DMBT1 promotes the activation of the complement pathway when bound to a surface and inhibits the lectin pathway of complement activation when free in the fluid.1,12,13 Furthermore, DMBT1 is upregulated in intestinal epithelial cells during inflammation and in the development of dysplasia in both humans and mice.14,15 DMBT1 is downregulated in skin cancer, and lung carcinogenesis changes the expression and location of DMBT1 from the lumen to the extracellular matrix.16,17 A recent case report found that mutations in the DMBT1-encoding gene were associated with increased susceptibility to mediastinal neuroendocrine tumors. 18 Loss of DMBT1 is associated with an increased risk of Crohn’s disease, metastasis, and cancer-associated death.19–21 High expression of DMBT1 promotes apoptosis of cancer cells in vitro and inhibits tumor progression in vivo. 22
DMBT1 has previously been detected in various tissues, such as the small intestine, lungs, vagina, placenta, mammillary ducts, and ocular surfaces.11,23–27 However, to date, the localization and distribution of DMBT1 has not been systematically studied in healthy human tissues. Therefore, we aimed to systematically investigate the localization and distribution of DMBT1 in different healthy human organ samples. We examined the expression of DMBT1 in 20 different human tissue samples by quantitative real-time PCR and investigated the distribution of DMBT1 in healthy tissue specimens from 30 human organs using immunohistochemical staining with the monoclonal DMBT1 antibody HYB213-6. 14
Materials and Methods
DMBT1-transfected Cells
Human breast adenocarcinoma (MCF-7) cells were transfected with DMBT1 using an Frt-site as described previously. 28 In brief, single copy insertion of the Frt-site carrying plasmid was confirmed by Southern blot analysis. The Frt site enabled Flp recombinase-mediated site-specific insertion of expression plasmids, and accordingly, the cells were named acceptor cells (Acc). The cells were cultivated as recommended by American Type Culture Collection (ATCC) but with the addition of 1000 µg/ml G418 (Life Technologies, Bleiswijk, Netherlands). Isogenic cell lines were generated by cotransfecting pOG44 (Invitrogen, Carlsbad, CA) for Flp recombinase with plasmids carrying the respective constructs in addition to the Frt-site at a ratio of 1:5. Cells were transfected with GeneJammer (Agilent Technologies, La Jolla, CA) according to the instructions of the supplier. Hygromycin B (120 µg/ml, Sigma Aldrich, Darmstadt, Germany) was used for selection of recombinant cells, and 50 ng/ml doxycycline was used to induce expression.
Recombinant cells carrying either the blank control insert, the DMBT1 6 kb gene, or the DMBT1 8 kb gene were seeded in T175 flasks (Thermo Fisher, Carlsbad, CA) with Dulbecco’s modified Eagle’s medium (Sigma Aldrich) with penicillin (100 U/ml), streptomycin (0.1 mg/ml), insulin (10 µg/mL), 10% fetal bovine serum (FBS), and 50 ng/ml doxycycline (all from Thermo Fisher) at 37C in humidified 5% CO2 for 72 hr. Approximately 3 x 108 cells of each cell line were harvested and spun down, and the resulting pellets were fixed in 4% normal-buffered formalin (NBF) for 48 hr at room temperature. The pellet was subsequently embedded in paraffin, cut into 4-µm-thick sections and stained using the same protocol as that used for staining the human tissue specimens (described below). DMBT1-transfected MCF-7 cells and blank MCF-7 cells were used to determine the specificity and titration of the antibody.
Quantitative Real-time PCR (qRT-PCR) of Human Tissue
RNA from 20 different human tissues was purchased (Clontech Laboratories, Inc., Mountain View, CA). The RNA had been extracted from tissue that was obtained postmortem where cause of death was noted as “sudden death” with the exception of the fetal samples which were obtained from spontaneous abortions. Complementary DNA (cDNA) was synthesized by the M-MLV Reverse Transcriptase kit (Sigma-Aldrich, Taufkirchen, Germany) according to the manufacturer’s instructions. cDNA was utilized as a template for RT-PCR analysis using the TaqMan Universal Mastermix II (no UNG) and TaqMan Gene Expression Assay (both from Thermo-Fisher). Reactions were performed using a StepOnePlus real-time PCR system. mRNA was measured relative to the housekeeping genes TATA box binding protein (TBP) and β-actin (ACTB) using the 2-ΔΔCt method (all from Applied Biosystems, Foster City, CA). The relative expression was analyzed using qBase Plus software (Biogazelle, Zwijnaarde, Belgium). Reactions were performed in technical triplicates. An overview of the number of samples for each organ is shown in Table 1.
Overview of Tissue for DMBT1 Expression With Quantitative Real-time PCR.
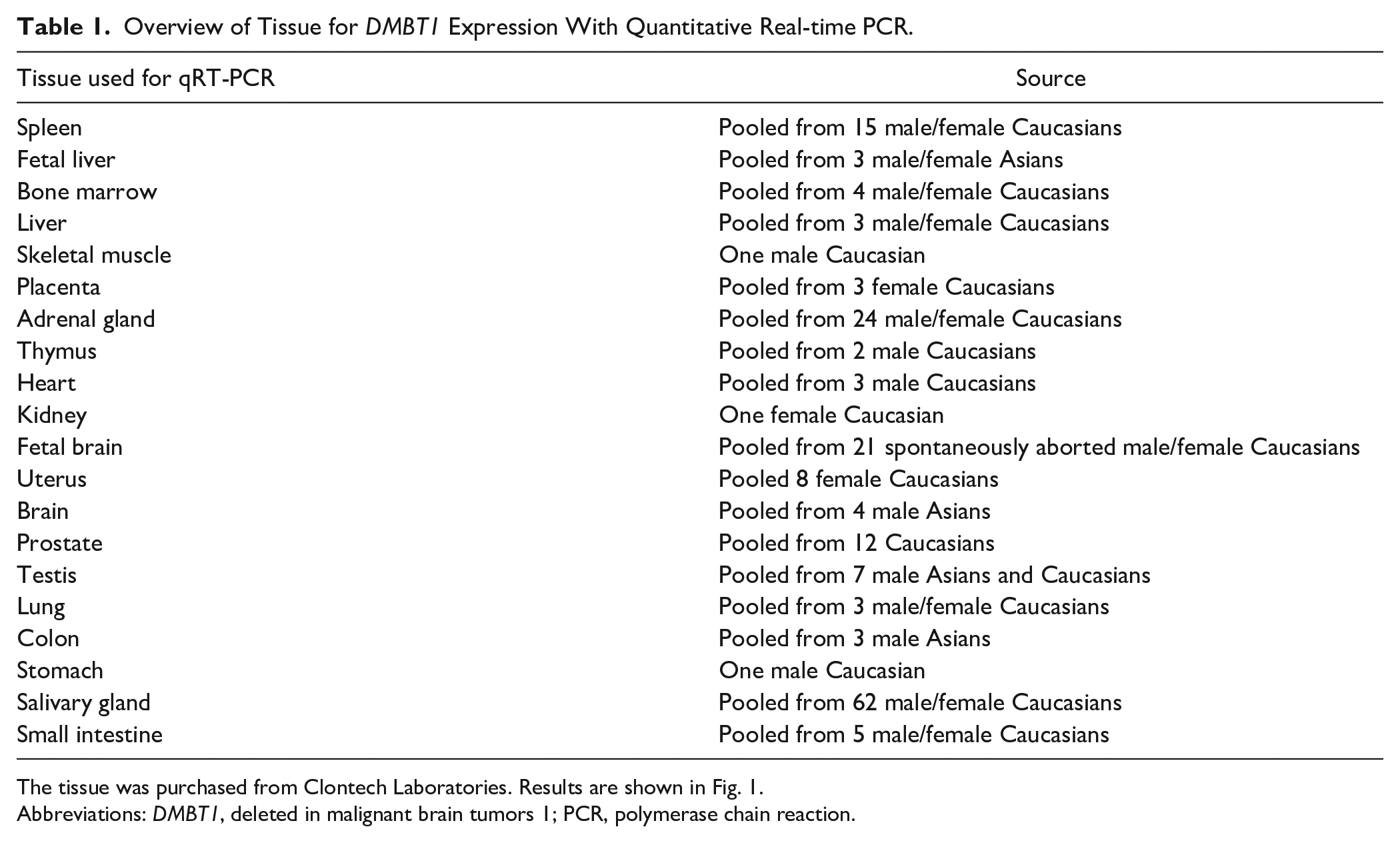
The tissue was purchased from Clontech Laboratories. Results are shown in Fig. 1.
Abbreviations: DMBT1, deleted in malignant brain tumors 1; PCR, polymerase chain reaction.
Immunohistochemical Staining of Human Tissue
Tissue samples from 30 different organs were obtained from the archives of the Department of Pathology, Odense University Hospital. The Regional Committees on Health Research Ethics for the Region of Southern Denmark approved the use of the human tissue samples (Ref. No. afS-VF-20050070). An overview of the number of samples for each organ can be seen in Table 2.
Overview of Tissue Stained for DMBT1.
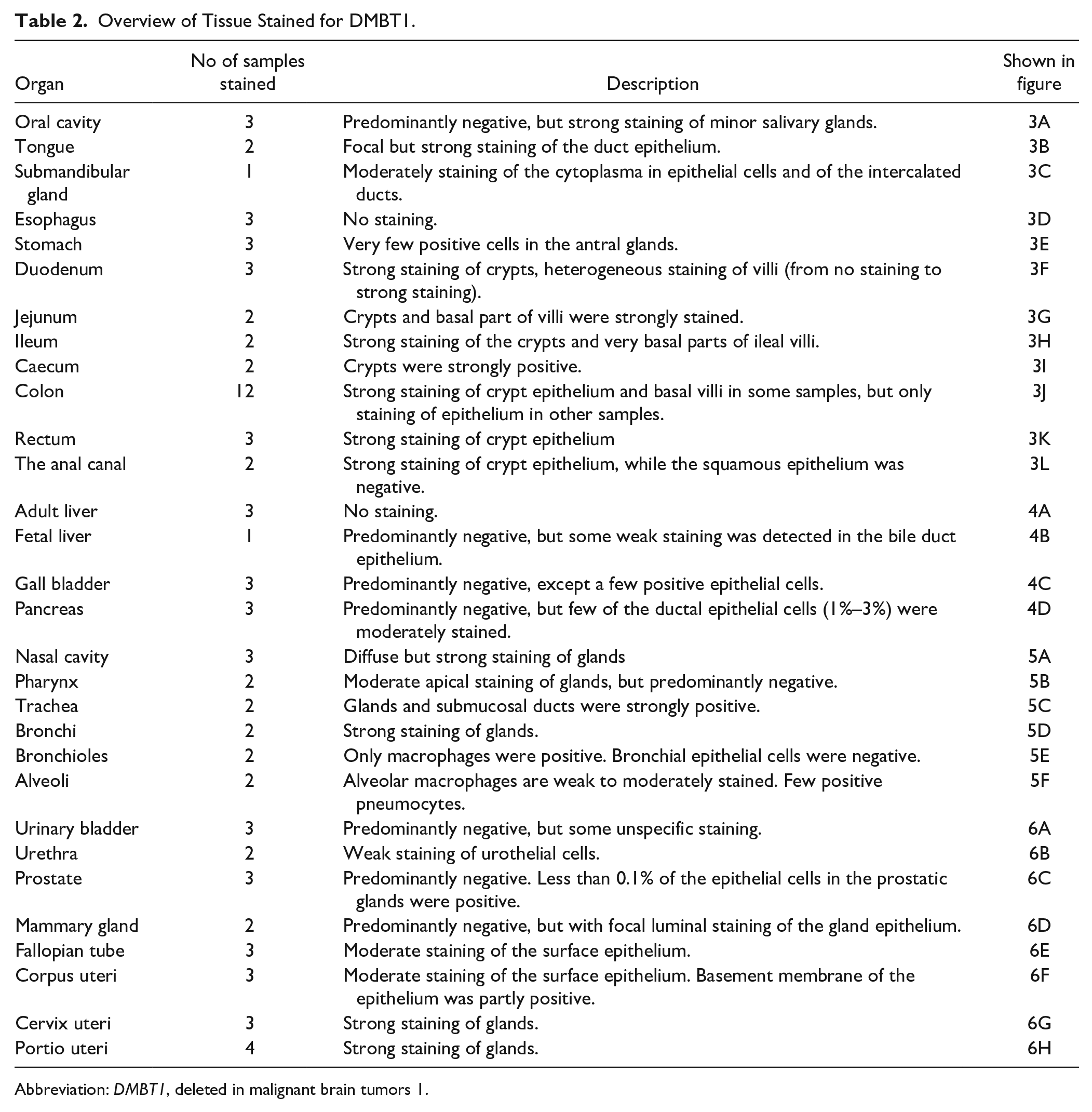
Abbreviation: DMBT1, deleted in malignant brain tumors 1.
Samples from nonmalignant and noninflammatory tissue were fixed in 4% NBF for 12 to 96 hr, conventionally dehydrated and subsequently embedded in paraffin. Antigen retrieval and detection were performed as previously described. 29 In brief, 4-µm sections of formalin-fixed paraffin embedded (FFPE) tissue blocks were cut on a microtome and placed on FLEX IHC slides. The endogenous activity was blocked by 1.5% hydrogen peroxide in TBS for 10 min. The GP340 HYB 213-6 (1 mg/ml) antibody produced as described in Holmskov et al.7,26 was diluted (1:400) in Antibody Diluent (Agilent Technologies, Glostrup, Denmark). The tissue was incubated with the antibody for 60 min at room temperature. After staining, the tissue was demasked for 15 min using AutostainerPlus and detected by the EnVision + horseradish peroxidase (HRP) (mono) K4001 system. Immunostaining was followed by nuclear counterstaining for 2 min in Mayer’s hematoxylin (Fagron Nordic, Copenhagen, Denmark).
Image Acquisition
Histology slides were scanned using a NanoZoomer-XR (Hamamatsu Photnoics, Hamamatsu City, Japan). NDP.view2 software (NanoZoomer Digital Pathology, Hamamatsu Photonics) was used to acquire the images, and Adobe Photoshop CC2017 (San Jose, CA) was used to automatically adjust for contrast of the entire image. All images are shown at 20X magnification. No other changes have been made to the images.
Results
DMBT1 mRNA Expression in Human Tissue
The expression of DMBT1 relative to the spleen was investigated in RNA isolated from 20 different human organs (Fig. 1). The cycle threshold (Ct), the number of cycles required for the fluorescent signal to exceed the background level, ranged from 17 to 37 cycles. The highest expression was found in the small intestine (Ct 17) followed by the salivary gland, stomach and colon (Ct 20-23). We detected moderate expression in the uterus, brain, and prostate (Ct 27-28). The lowest expression was found in the spleen, liver (both fetal and adult), and bone marrow (Ct 36-37). Ct-values for the endogenous controls ranged from 15 to 20 with a mean of 17.5 (ACTB) and from 21 to 27 with a mean of 24.9 (TBP). Which indicates that variation of the analyzed samples was acceptable.
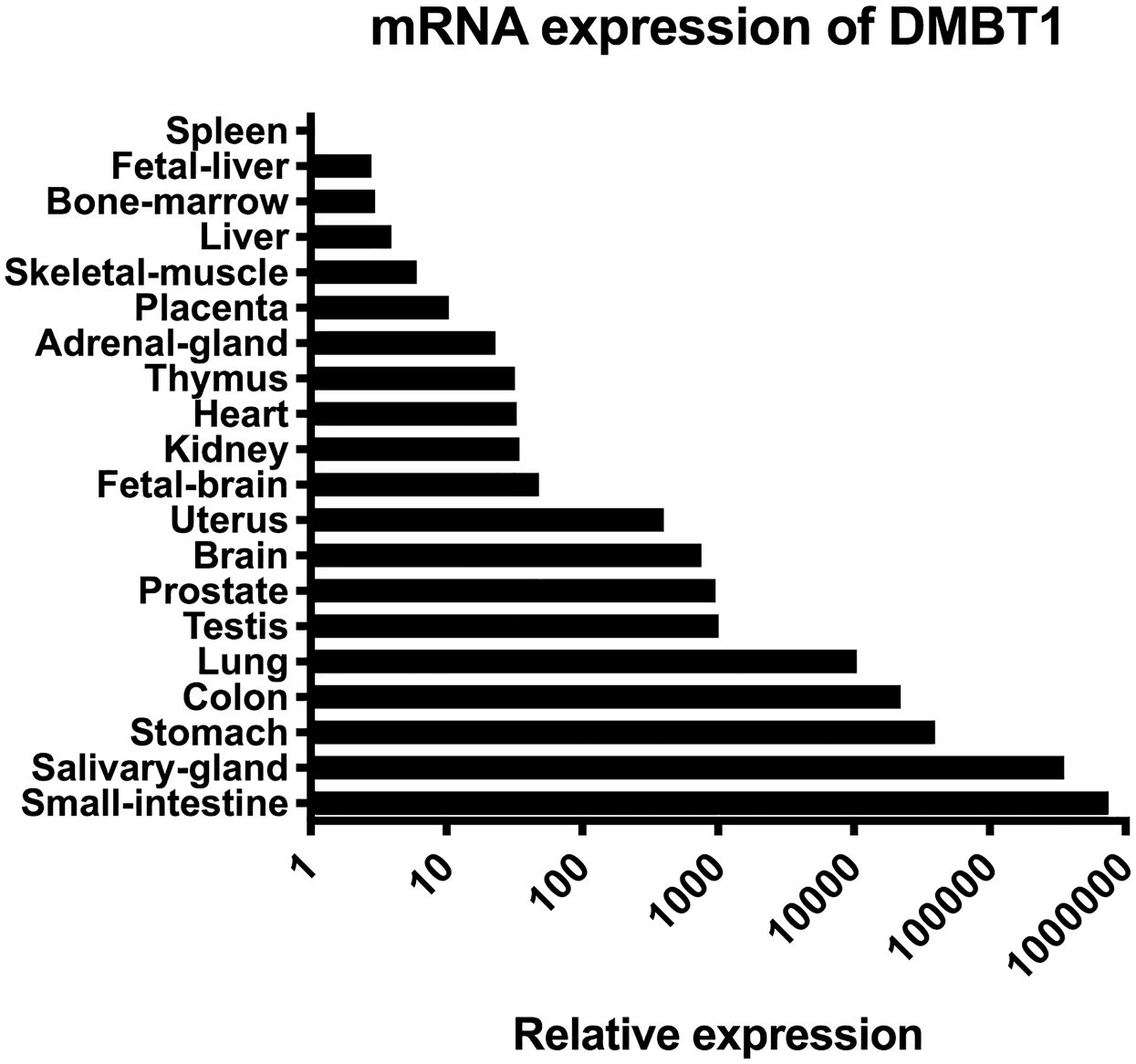
Relative mRNA expression of DMBT1 in 20 human organs measured by quantitative PCR. Expression is normalized to ACTB and TBP, setting the expression in the spleen to 1. Abbreviations: DMBT1, deleted in malignant brain tumors 1; PCR, polymerase chain reaction; ACTB, β-actin; TBP, TATA box binding protein.
Specificity of the Antibody
We performed staining of either MFC-7 cells transfected with DMBT1 (6-kb and 8-kb transcripts) or blank MFC7 cells at a 1:400 dilution (Fig. 2). The DMBT1-transfected cells showed intense staining (Fig. 2A and B), while the blank cells showed no reactivity (Fig. 2C). To determine the specificity of the antibody used, we used staining with anti-ovalbumin (anti-OVA) as an isotype control. We found no immunohistochemical reaction in the MFC7 cells stained with anti-OVA (Fig. 2D to F). Tissue samples used to investigate the distribution and localization of DMBT1 were stained with anti-OVA using the same protocol as DMBT1 staining and showed no reactivity (Supplementary Figs. 1 to 4).
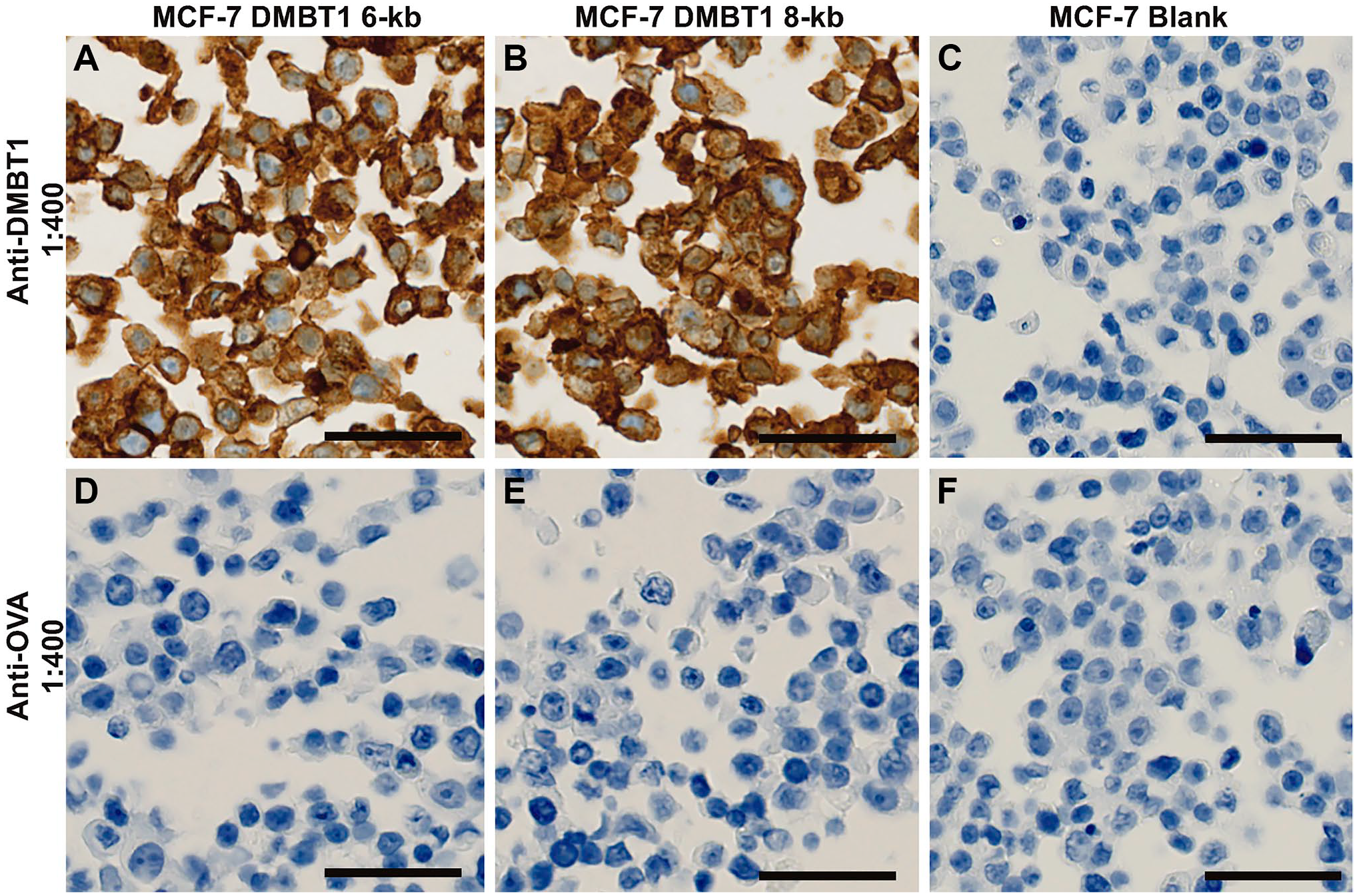
Determination of antibody specificity. MCF-7 cells stained with either anti-DMBT1 (A–C) or with anti-OVA (D–F) in 1:400. A–B and D–E are MCF-7 cells transfected with DMBT1 and C and F are blank MCF-7 cells. Scale bars indicate 50 µm. Abbreviations: MCF-7, human breast adenocarcinoma; DMBT1, deleted in malignant brain tumors 1; OVA, ovalbumin.
Localization of DMBT1 in the Digestive Tract
We found strong DMBT1 expression throughout the digestive tract (Fig. 3). The oral cavity was predominantly negative, but we detected strong expression of DMBT1 in the minor salivary glands (Fig. 3A). In the tongue, we found strong focal staining of cylindric epithelium in the submucosal glands, but the stratified squamous epithelium was negative (Fig. 3B). In the submandibular gland, we detected moderate expression of the apical surfaces and intercalated ducts (Fig. 3C). The squamous epithelium of the esophagus was predominantly negative (data not shown), but the esophageal submucosal glands showed moderate expression (Fig. 3D). In the stomach, a few cells in the antral glands were positive (Fig. 3E). In the duodenum, we found a high expression level of DMBT1 in the crypts in all samples (Fig. 3F), while the staining of the villi was heterogeneous and ranged from nonexistent to strong staining in different samples (data not shown). In the jejunum and ileum, we detected strong DMBT1 expression in the crypts and the basal part of the villi (Fig. 3G and H). In the cecum, the crypts were strongly stained (Fig. 3I). In the more distal colon, we detected heterogeneous expression, where some samples showed strong staining of both surface epithelium and basal crypts, while other samples only showed weak staining of the crypts (Fig. 3J). In the rectum and anal canal, the crypt epithelium was strongly stained, while DMBT1 expression was lacking in the squamous epithelium of the anal canal (Fig. 3K and L).
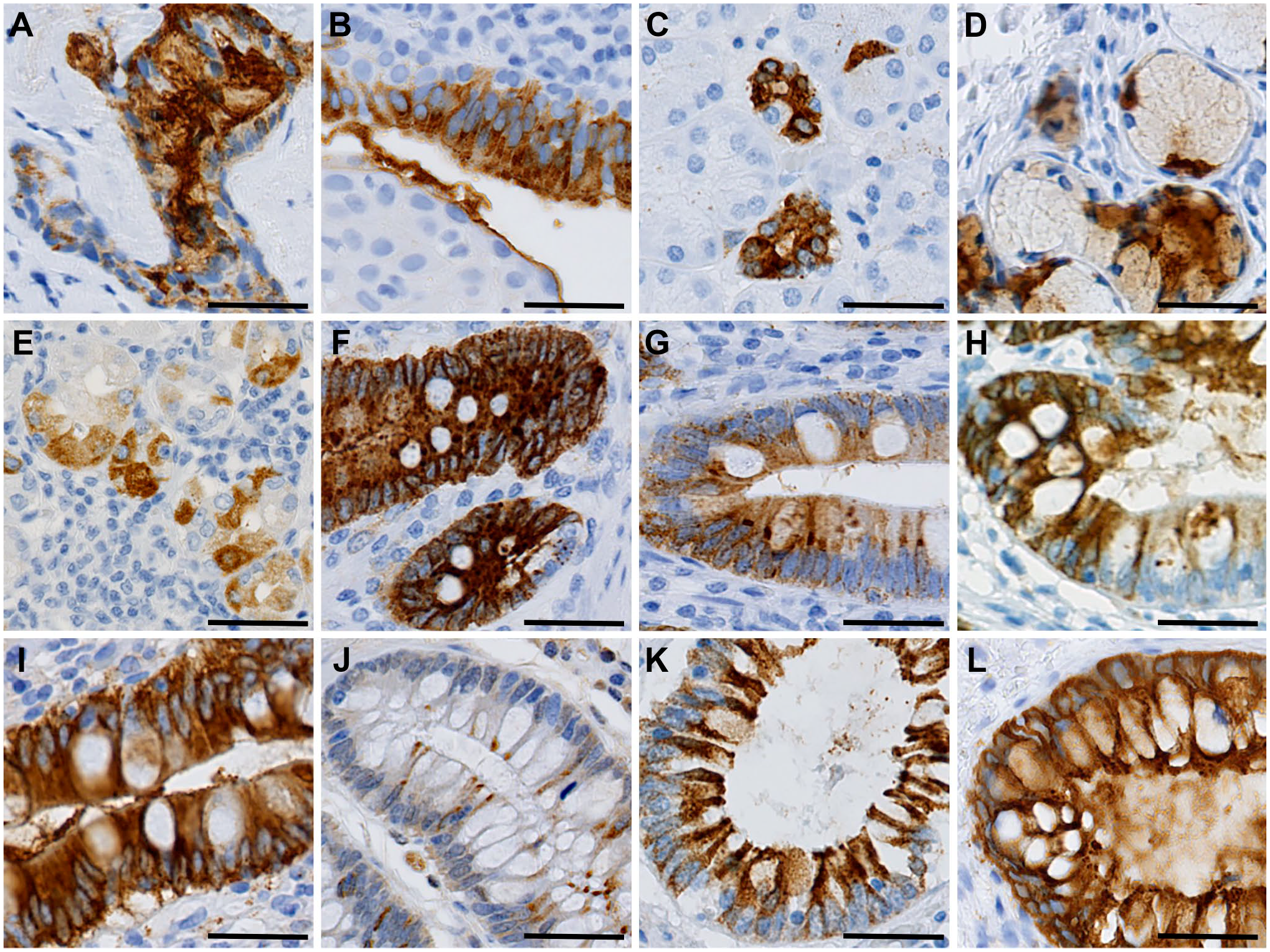
Immunohistochemical expression of DMBT1 in the human digestive tract. (A) oral cavity, (B) tongue, (C) submandibular gland, (D) esophagus, (E) stomach, (F) duodenum, (G) jejunum, (H) ileum, (I) caecum, (J) colon, (K) rectum, and (L) anal canal. Scale bars indicate 50 µm. Abbreviations: DMBT1, deleted in malignant brain tumors 1.
Localization of DMBT1 in the Liver, Pancreas, and Gallbladder
The expression of DMBT1 was notably weak in both the adult and fetal liver and limited to the bile duct epithelium (Fig. 4A and B). In the gallbladder, only a few of the epithelial cells expressed DMBT1 (Fig. 4C). In the pancreas, a few of the ductal epithelial cells showed moderate DMBT1 expression (Fig. 4D).
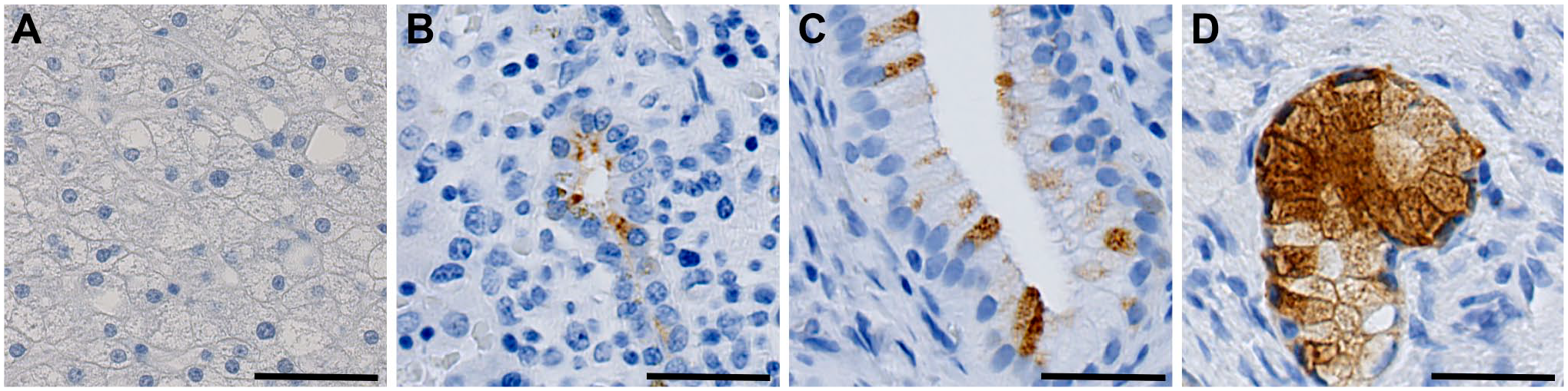
Immunohistochemical DMBT1 expression in the human liver, gallbladder and pancreas. (A) adult liver, (B) fetal liver, (C) gall bladder, and (D) pancreas. Scale bar indicates 50 µm. Abbreviations: DMBT1, deleted in malignant brain tumors 1.
Localization of DMBT1 in the Respiratory System
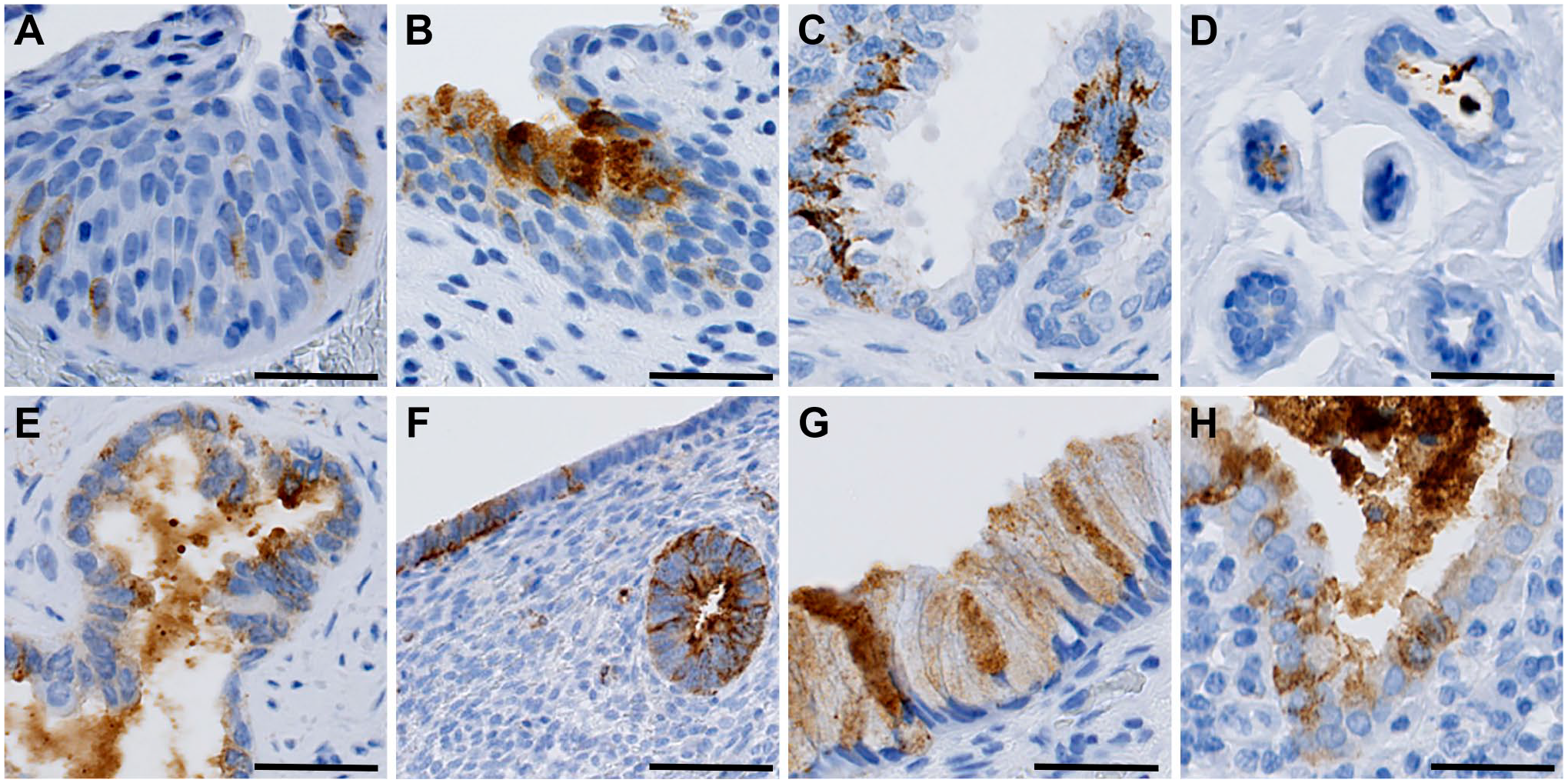
In the respiratory system (Fig. 5), we found strong DMBT1 expression in the mucosa and submucosa of the nasal cavity (Fig. 5A) around the serous glands, while the respiratory epithelium was predominantly negative. In the pharynx, the apical parts of the salivary gland ducts showed moderate staining, while the remaining tissue was predominantly negative (Fig. 5B). In the trachea and bronchial respiratory epithelium, DMBT1 expression was negative (not shown), while the serous glands and ducts were positive (Fig. 5C and D). In the bronchioles and the alveoli, the expression of DMBT1 in the epithelium was weak, with focal expression in the pneumocytes and only few positive macrophages (Fig. 5E and F).
Immunohistochemical DMBT1 expression in the respiratory system. (A) nasal fossa, (B) pharynx, (C) trachea, (D) bronchi, (E) bronchioles, and (F) alveoli. Scale bars indicate 50 µm. Abbreviations: DMBT1, deleted in malignant brain tumors 1.
Localization of DMBT1 in the Urogenital System
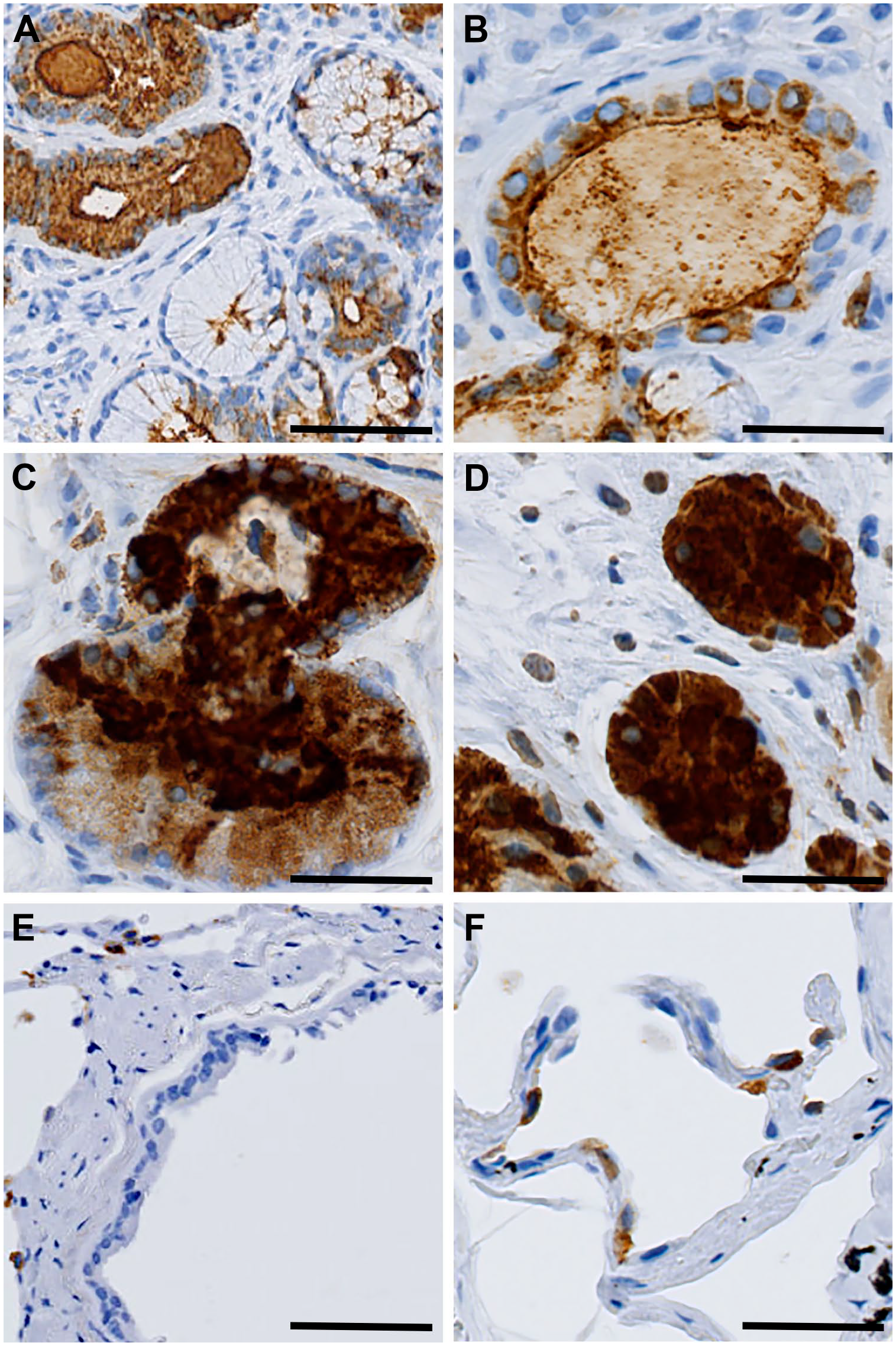
In the urogenital system, we observed varied DMBT1 staining intensity (Fig. 6). The expression was weak in the urinary bladder, with only a few focal urothelial cells being positive (Fig. 6A). In the urethra, we detected moderate expression of cells in the urothelium (Fig. 6B). The prostate was predominantly negative with only a few positive cells (less than 0.1%) in the glands (Fig. 6C). The mammary glands were predominantly negative, but we detected luminal expression in some glands (Fig. 6D). In the uterine tube, the basement membrane was partly positive (Fig. 6E). In the uterine body and cervix, there was moderate expression of the surface epithelium (Fig. 6F and G). In the uterine portio, the glands were strongly stained for DMBT1 (Fig. 6G and H). We detected no DMBT1 expression in the cerebrum, skin, lymph nodes, spleen, ovaries, or testes (not shown).
Immunohistochemical DMBT1 expression in the urogenital system. A) Urinary bladder, B) Urethra, C) Prostate, D) Mammary gland, E) Fallopian tube F) Uterine body, G) Uterine cervix, H) Uterine portio. Scale bars indicate 50 µm. Abbreviations: DMBT1, Deleted in malignant brain tumors 1.
Discussion
In this study, we investigated the localization and distribution of DMBT1 in healthy human tissue samples. We measured mRNA levels in samples from 20 different human organs and investigated the protein levels using immunohistochemical staining of DMBT1 in 30 different histological structures. DMBT1 was detected to varying degrees in tissue and glands from the digestive tract, abdominal organs, respiratory system, and urogenital system.
We found that the anti-DMBT1 antibody showed high specificity when used on human tissue, while the isotype control (anti-OVA) showed no reactivity. We detected no expression in the blank MCF-7 cells when staining with anti-DMBT1, while DMBT1-transfected MFC-7 cells showed strong expression, and we detected no staining of the cells when using the control anti-OVA antibody. Furthermore, a previous study 14 analyzed the specificity of the antibody with Western blot and found that the antibody recognized DMBT1 in both lung lavage and colonic tissue extract. Amino acid sequence of the derived peptides showed that the recognized bands were encoded by the DMBT1 gene. These results indicate that the antibody provided reliable and specific staining of DMBT1 in both cells and human tissue.
We found high mRNA expression in the small intestine and salivary gland; moderate mRNA expression in the brain, uterus, and prostate; and low mRNA expression in the spleen, bone marrow, fetal, and adult liver. We found a clear correlation between the immunohistochemical staining intensity and mRNA expression. For instance, the mRNA expression in the liver was low, and we found only weakly positive DMBT1 staining in adult and fetal liver tissue. Conversely, we found high mRNA expression in the small intestine and similar strong immunoreactivity in the duodenum, jejunum and ileum. However, although we detected moderate mRNA levels of DMBT1 in the testis and in the brain, we did not detect any immunohistochemical DMBT1 expression. This finding could be due to a mismatch between transcript and protein or due to variation in the expression of DMBT1 between individuals, as we also noticed some differences between specimens stained for DMBT1. In the case of the expression of DMBT1 in the brain the samples examined with qRT-PCR were all from Asian individuals while the samples used for IHC examination were from Caucasians which could also explain the observed difference.
In this study, we examined colonic samples from 12 healthy individuals, and while some specimens showed strong immunoexpression in both the surface and crypt epithelium, other specimens showed only very weak expression in the surface epithelium with only a few positive cells. This finding supports the notion that there is heterogeneous expression of colonic DMBT1 in the population, which may be of clinical significance. A previous study discovered that intestinal DMBT1 expression is induced by IL-22 and that DMBT1 levels are increased in the inflamed mucosa, 30 but we detected no signs of inflammation in colonic samples with high DMBT1 expression. However, we cannot fully exclude the possibility that some of the variation may be related to microscopically invisible changes, as the tissues derived from patients who underwent colon surgery for different conditions, such as tumors, even though the tissue was obtained as far away from the tumors as possible. To elucidate this, future studies should examine the DMBT1 expression in a higher number of colonic tissues.
Although we obtained all specimens from either autopsies or surgical procedures, the tissues included in this study had a normal morphology, as we aimed to describe the localization and distribution of DMBT1 in normal healthy tissue. Tissue samples showing signs of inflammation and dysplasia were excluded from this study. However, in previous studies based on tissue samples showing inflammation or dysplasia, we observed an increased expression of DMBT1, which was in keeping with previous observations.14–16,31 Based on these previously published studies, it may be hypothesized that DMBT1 could be a potential marker for inflammation and dysplasia and could be of use in a diagnostic setting.
It is possible that the autopsy specimens that we used for RNA analyses could have been exposed to a higher degree of ischemic damage and degradation of RNA and protein before fixation and that this could have affected the staining for DMBT1. All tissue used for immunohistochemical staining in this study appeared macroscopically and microscopically normal, and therefore the potential difference between autopsy specimens and surgically resected specimens are likely to be of only minor importance.
We demonstrate that DMBT1 is expressed in different mucosal surfaces and in salivary glands, both at the mRNA level and the protein level. Together with the known functions of DMBT1 as a pattern recognition receptor (PPR), this result indicates that DMBT1 plays an important part in mucosal defense.
In conclusion, we detected a strong DMBT1 expression on mucosal surfaces in the digestive tract (duodenum, cecum and anal canal) and respiratory system (trachea and bronchi), and in salivary glands at both RNA and protein levels.
Supplemental Material
2020-00010R1_Production_Supplemental_Data_online_supp – Supplemental material for Immunohistochemical Localization of Deleted in Malignant Brain Tumors 1 in Normal Human Tissues
Supplemental material, 2020-00010R1_Production_Supplemental_Data_online_supp for Immunohistochemical Localization of Deleted in Malignant Brain Tumors 1 in Normal Human Tissues by Anders Bathum Nexoe, Andreas Arnholdt Pedersen, Sebastian von Huth, Sönke Detlefsen, Pernille Lund Hansen and Uffe Holmskov in Journal of Histochemistry & Cytochemistry
Footnotes
Competing Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
UH conceptualized the study. SVH and AAP performed and optimized the immunohistochemistry and performed the qRT-PCR. PLH transfected the cells. ABN and SD investigated histological and immunohistochemically stained specimens. SVH, AAP, and ABN assembled all figures. ABN drafted the manuscript. All authors have contributed to the final version of the manuscript. All authors approved the final manuscript.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
