Abstract
Human breast milk (HBM) contains many bioactive components that protect infants from various microorganisms. Pattern recognition receptors on phagocytic cells recognize microbial pathogens and promote the innate immune system. This study aimed to evaluate the effect of HBM on the expression of pattern recognition receptors and innate cytokines in the monocytic cell line THP-1 and the phagocytic activity of RAW264.7 macrophages. Expression levels of specific mRNAs in THP-1 cells were quantitated using reverse transcription-polymerase chain reaction. Phagocytic activity was measured by fluorescence microscopy to detect the uptake of fluorescent dye-labeled carboxylate-modified polystyrene latex beads in RAW264.7 macrophages. HBM stimulated the phagocytic activity of RAW264.7 macrophages. HBM increased mRNA expression of pattern recognition receptors, including the cluster of differentiation 14 and toll-like receptor 2 and 4, and various innate cytokines, including tumor necrosis factor α, interleukin-1β, C-X-C motif chemokine 8, and C-C motif chemokine ligand 2, in THP-1 monocytic cells. Furthermore, milk oligosaccharides in HBM, such as lacto-N-fucopentaose I, enhanced the expression of pattern recognition receptors and various innate cytokines. HBM is able to modulate the innate immune response by upregulating the expression of pattern recognition receptors and various innate cytokines in monocytes/macrophages.
Keywords
Introduction
Pattern recognition receptors, such as the cluster of differentiation 14 (CD14), toll-like receptors (TLRs), β2-integrins, complement receptors (CD35), and C-type lectins, recognize distinct pathogen-associated molecular patterns (PAMPs), including lipopolysaccharide (LPS), lipopeptide, and microbial nucleic acids, and activate the innate immune system. 1 Of the pattern recognition receptors that initiate innate immunity, TLRs are the best-characterized.1–3
To date, at least ten TLRs (TLR1–TLR10) have been found in humans. 3 Different TLRs can recognize distinct PAMPs: TLR1/2, bacterial peptidoglycan; TLR3, double-stranded RNA; TLR4, bacterial LPS; TLR5, bacterial flagellin; TLR6, bacterial lipoprotein; TLR7/8, single-stranded RNA; TLR9, CpG DNA; and TLR10, triacylated lipopeptides. 3 Upon recognizing PAMPs, TLRs activate intracellular signaling pathways and initiate innate immune responses, including the secretion of inflammatory cytokines. CD14 is a glycosylphosphatidylinositol-anchored protein predominantly expressed on various immune cells, particularly on monocyte/macrophage cells. CD14 is responsible for the first recognition of gram-negative bacterial endotoxin, such as lipopolysaccharide (LPS), and gram-positive cell wall peptidoglycan4–6 and functions as a co-receptor that physically interacts with TLR2 or TLR4. 7 Since CD14 delivers the bacterial pathogenic ligands to TLR2 or TLR48,9 and activates innate immune responses, 10 the proper expression of CD14 and its co-receptors TLR2 and TLR4 in phagocytic cells is critical for the proper modulation of the innate immune responses.
Human breast milk (HBM) provides functional nutrients to infants. Accumulating evidence demonstrates that HBM contains many bioactive anti-inflammatory factors that function to protect infants and children from various microorganisms, including IgA, lactoferrin, lysozyme, defensins, and oligosaccharides.11–13 Monocytes, a component of the bone marrow cell lineage in blood, can infiltrate peripheral tissues and differentiate into macrophages. Thus, the recruitment of monocytes is essential for the effective removal of pathogenic microorganisms and the modulation of innate immune responses. However, limited information is currently available on the modulation of monocyte-mediated innate immune responses by HBM.
This study aimed to evaluate the effect of HBM on the expression of pattern recognition receptors in the monocytic cell-line THP-1, particularly CD14, TLR2, and TLR4. This work revealed a novel role of HBM in ameliorating the response of the innate immune system of infants.
Materials and methods
Cells and cell culture
The human monocytic leukemia cell line THP-1 cells (accession number: ATCC, TIB-202; RRID, CVCL_0006) and mouse macrophage RAW264.7 cells (accession number: ATCC, TIB-71; RRID, CVCL_0493) were obtained from the American Type Culture Collection (ATCC, Manassas, VA, MD, USA). THP-1 cells were cultured in Roswell Park Memorial Institute medium, and RAW264.7 cells were cultured in Dulbecco’s modified Eagle’s medium supplemented with 10% heat-inactivated fetal bovine serum (CellGro/Corning, Manassas, VA, USA) and 2 mM l-glutamine (Gibco BRL/Thermo Fisher Scientific, Waltham, MA, USA) in a humidified 5% CO2 atmosphere at 37°C.
Reagents
Endotoxin-free sterilized Pooled HBM Lyophilized was obtained from Innovative Research (Novo, MI, USA). LPS from Escherichia coli 0111:B4 was purchased from Sigma-Aldrich (St. Louis, MO, USA). Pathway signaling inhibitors U0126 (MEK inhibitor), SP600125 (JNK inhibitor), and SB202190 (p38 kinase inhibitor) were obtained from Calbiochem/Novagen (San Diego, CA, USA). Human breast milk oligosaccharides (2′-Fucosyllactoses, lacto-N-fucopentaose I, and lacto-N-tetraose) were from Carbosynth Ltd (Compton, UK). TRIzol RNA Extraction Kit was obtained from Invitrogen (Carlsbad, CA, USA). iScript cDNA Synthesis Kit was purchased from Bio-Rad (Hercules, CA, USA). Antibodies specific to phospho-ERK1/2 (Thr202/Tyr204), phospho-JNK1/2 (Thr183/Tyr185), and phospho-p38 (Thr180/Tyr182) were purchased from Cell Signaling Technology (Danvers, MA, USA). The glyceraldehyde 3-phosphate dehydrogenase (GAPDH) antibody was obtained from Santa Cruz Biotechnology (Santa Cruz, CA, USA). Enhanced chemiluminescence system kit was from GE Healthcare (Buckinghamshire, UK) and X-ray film was from Amersham Life Science (Buckinghamshire, UK).
Reverse transcription-polymerase chain reaction (RT-PCR)
Total RNA was isolated from the cells using a TRIzol RNA Extraction Kit, and the first-strand complementary DNA (cDNA) was synthesized using an iScript cDNA Synthesis Kit. Polymerase chain reaction (PCR) was performed as described previously. 14 The PCR thermal cycle conditions were as follows: initialization at 94°C for 5 min, denaturation at 94°C for 30 s, annealing at 55°C for 30 s, elongation at 72°C for 1 min, and a final extension at 72°C for 4 min. Gene-specific PCR primer sequences (Table 1) were obtained from Macrogen (Seoul, Republic of Korea). The PCR amplicon products were separated by 1% agarose gel electrophoresis. The PCR band intensities were quantified using ImageJ software (National Institutes of Health, Bethesda, MA, USA) and normalized to GAPDH mRNA levels. Expression levels in unstimulated controls were arbitrarily set to 1.
Gene-specific primer sequences.

Immunoblot analysis
The cells were lysed, and the total protein lysates (15 μg/sample) were separated by 10% SDS-polyacrylamide gel electrophoresis, transferred to a nitrocellulose membrane (Bio-Rad, Richmond, CA, USA), and then blocked with 5% fat-free skim milk in Tris-buffered saline containing 0.1% Tween-20 for 1 h. The membranes were incubated with antibodies specific to phospho-ERK1/2, phospho-JNK1/2, and phospho-p38 for 4 h followed by incubation with horseradish peroxidase-conjugated secondary antibodies for 1 h. Antibody-reacted protein bands were visualized using an enhanced chemiluminescence system and X-ray film as described previously. 15 The protein band intensities were quantified using the ImageJ software.
Phagocytosis assay
Phagocytosis was analyzed using 10% FBS-opsonized fluorescent dye-labeled carboxylate-modified polystyrene latex beads (2.0 μm mean particle size) according to the manufacturer’s instructions (Sigma-Aldrich, Saint Louis, MO, USA). RAW264.7 macrophages were exposed to vehicle (PBS), LPS (0.1 μg/mL), or HBM (25 μg/mL) for 24 h after which time the fluorescent dye-labeled latex beads (2 × 108 beads/mL) were added. After 2 h, the cells were washed to remove the extracellular beads, and the uptake of fluorescent latex beads was imaged using an EVOS FL Auto Cell Imaging System (Life Technologies, Carlsbad, CA, USA).
Statistical analysis
The data were statistically analyzed by one-way analysis of variance (ANOVA) followed by Dunnet’s multiple comparisons test using GraphPad Prism version 7.04 software (GraphPad Software Inc., La Jolla, CA, USA). Data are expressed as mean ± standard deviation (SD). A p-value of <0.05 was considered to be statistically significant.
Results
Effect of human breast milk (HBM) on macrophage phagocytosis
The physiologically relevant concentration of HBM is unclear because information on detailed analysis of the concentrations of bioactive components after breastfed is limited. A previous study used human milk at a 50 μg/mL concentration to study cytokine expression in THP-1 cells. 16 We tested the dose-response effect of HBM at concentrations of 1–100 μg/mL for CD14, a pattern recognition receptor, induction by RT-PCR, and observed that 20–50 μg/mL was the optimal sub-saturation range without detectable cytotoxicity. Based on these preliminary experiments, further experiments were performed using a 25 μg/mL concentration.
We first evaluated whether HBM affects the phagocytic capability of macrophages. LPS is an outer membrane component of Gram-negative bacteria. Since LPS is well characterized in triggering the immunostimulatory activity of macrophages, including phagocytosis and cytokine expression, 17 we used LPS as a positive control for activating the macrophages. RAW264.7 macrophages were treated with LPS in the presence or absence of 25 μg/mL HBM for 24 h, and then the latex bead uptake assay was performed. We found that the phagocytic uptake of fluorescent beads increased upon exposure to HBM and comparable to LPS treatment (Figure 1). These data suggested that HBM effectively stimulated the phagocytic activity of macrophages.
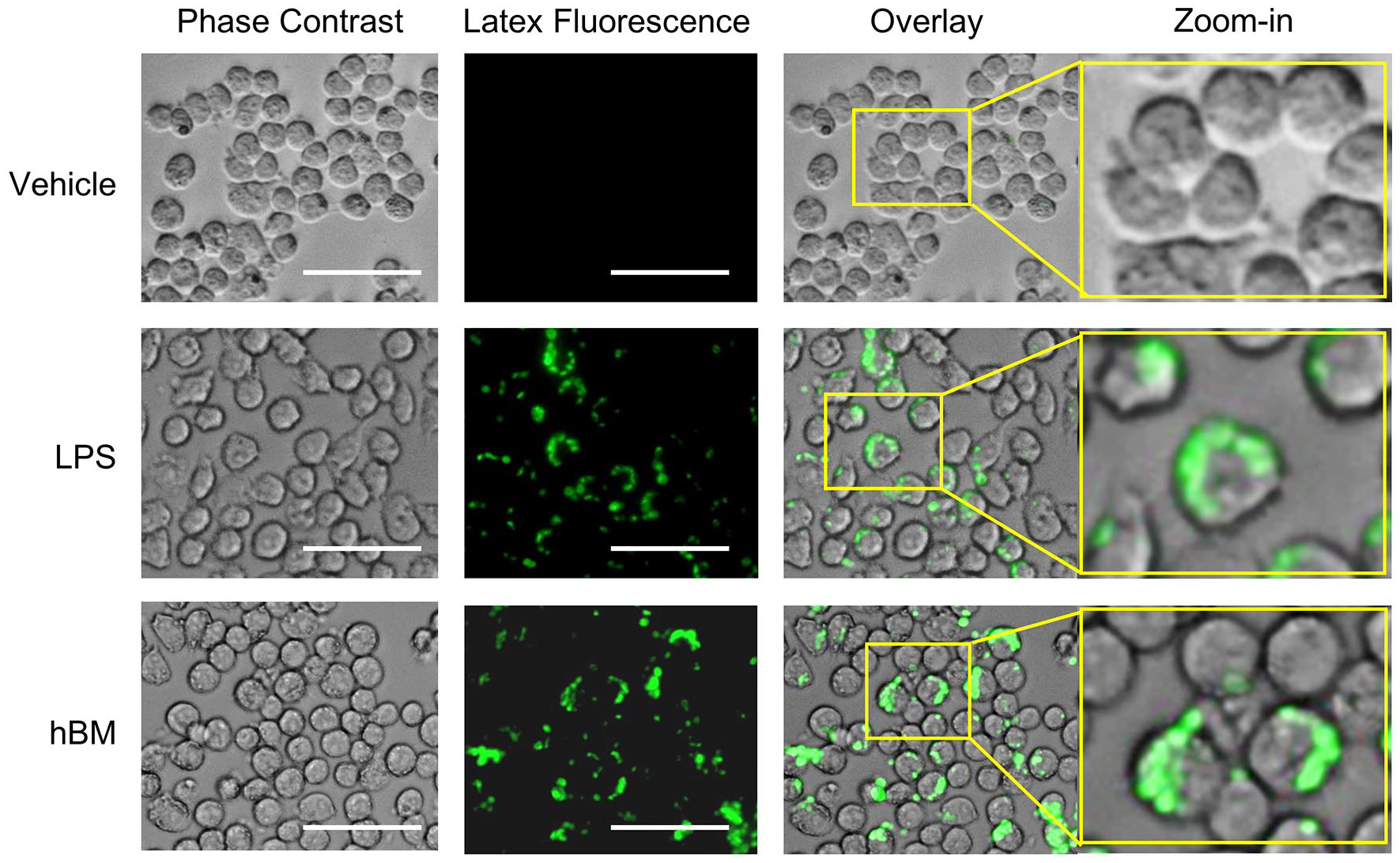
Effect of HBM on phagocytic activity of macrophages. RAW264.7 macrophages were incubated with the fluorescently labeled latex beads in the presence (HBM) or absence (Vehicle) of 25 μg/mL HBM for 2 h at 37°C. Lipopolysaccharide (LPS) was used as a positive control. The phagocytized latex beads were observed using a fluorescence microscope. Scale bar, 50 μm.
Effect of HBM on the expression of bacterial pattern recognition receptors in THP-1 monocytic cells
CD14 is a pattern recognition receptor that recognizes gram-negative and gram-positive bacteria4–6 and activates innate immune responses. 10 CD14 is associated with TLR2 or TLR4.8,9 To investigate whether HBM may modulate the expression of CD14 and its co-receptors TLR2 and TLR4 in monocytes, we exposed THP-1 monocytic cells with 25 μg/mL HBM for different amounts of time (0–24 h). We found that compared to the unstimulated control cells, mRNA expression of CD14 increased significantly (p < 0.05) within 6 h after HBM exposure, reached a maximum at 12 h, and remained elevated for up to 24 h post-treatment (Figure 2).

Effect of human breast milk (HBM) on the mRNA expression of pattern recognition receptors. THP-1 cells were treated with 25 μg/mL HBM for different amounts of time (0–24 h). Total RNA was isolated, and RT-PCR was performed for CD14, TLR2, and TLR4. GAPDH was used as an internal control. Quantification of individual band intensities was measured using ImageJ software (right graphs). Values were normalized to GAPDH mRNA expression. The relative fold increases were compared with that of the vehicle-treated controls which were set to a value of 1. The data shown represent the mean ± SD (n = 3). NS (not significant) 1, p = 0.936; NS2, p = 0.422; NS3, p = 0.936; P1, p = 0.048; P2, p = 0.007; P3, p = 0.035, P4, p = 0.012; **, p < 0.001, as compared to untreated control group (0 h).
Effect of HBM on the expression of innate immune cytokines in THP-1 monocytic cells
Tumour necrosis factor alpha (TNFα) and interleukin-1 beta (IL-1β) are primary cytokines that promote innate immune responses with C-X-C motif chemokine 8 (CXCL8) and C-C motif chemokine ligand 2 (CCL2) playing essential roles in recruiting various immune cells.18–20 We, therefore, examined whether HBM stimulated the expression of innate cytokines. Upon HBM treatment, mRNA expression levels of TNFα, IL-1β, CXCL8, and CCL2 rapidly and significantly (p < 0.01) increased within 3 h compared to that of the control cells (Figure 3). These results suggested that in monocytes, HBM led to the sensitizing of endotoxin-mediated innate immune responses through the upregulation of bacterial pattern recognition receptors and multiple innate cytokines

Effect of human breast milk (HBM) on the mRNA expression of innate immunity-related cytokines. THP-1 cells were treated with 25 μg/mL HBM for different amounts of time (0–24 h). Total RNA was isolated, and RT-PCR was performed for TNFα, IL-1β, CXCL8, and CCL2. GAPDH was used as an internal control. Quantification of individual band intensities was measured using ImageJ software (right graphs). Values were normalized to GAPDH mRNA expression. The relative fold increases were compared with that of the vehicle-treated controls which were set to a value of 1. The data shown represent the mean ± SD (n = 3). NS (not significant) 1, p = 0.222; NS2, p = 0.146; NS3, p = 0.106; P1, p = 0.001; P2, p = 0.003; P3, p = 0.043; **, p < 0.001, as compared to untreated control group (0 h).
Effect of HBM on the activation of MAPK pathways in THP-1 monocytic cells
Next, we investigated the intracellular signaling pathways involved in HBM-induced innate immune responses. MAPKs play crucial roles in the regulation of innate immune responses, including the induction of pro-inflammatory cytokines and chemokines. 21 Extracellular signal-regulated kinase (ERK), c-Jun N-terminal kinases (JNKs) 1 and 2, and p38 kinase are major subfamilies of MAPK. To determine whether HBM modulated the MAPK pathways, we treated THP-1 cells with 25 μg/mL HBM for various amounts of time and assessed the phosphorylation status of MAPKs using phospho-specific antibodies. Immunoblot analysis showed that ERK1/2 phosphorylation at Thr202/Tyr204 increased within 30 min of HBM treatment and reached a broad plateau at 120 through 240 min, while the phosphorylation of JNK1/2 at Thr183/Tyr185 and p38 kinase at Thr180/Tyr182 peaked at 30 min and 15 min post-treatment, respectively (Figure 4). These data suggested that HBM rapidly stimulated three major MAPKs in THP-1 monocytic cells.

Effect of human breast milk (HBM) on the activation of mitogen-activated protein kinase (MAPK) signalings. THP-1 cells were treated with 25 μg/mL HBM for different amounts of time (0–240 min). Whole-cell lysates were prepared, and immunoblot analysis was performed using specific antibodies against phosphorylated ERK1/2 (P-ERK1/2), phosphorylated JNK1/2 (P-JNK1/2), or phosphorylated p38 (P-p38) MAPKs. Quantification of individual band intensities was measured using ImageJ software (right graphs). Values were normalized to GAPDH. The relative fold increases were compared with that of the vehicle-treated control, which was set to a value of 1. The data shown represent the mean ± SD (n = 3). NS (not significant) 1, p = 0.982; NS2, p = 0.140; NS3, p = 0.381; P1, p = 0.008; P2, p = 0.034; P3, p = 0.049; P4, p = 0.012; P5, p = 0.001; **, p < 0.001, as compared to untreated control group (0 min).
Effect of mitogen-activated protein kinases (MAPKs) on the HBM-induced expression of the LPS receptor and innate immune cytokines in THP-1 monocytic cells
To address which MAPK pathway participated in HBM-induced innate cytokine expression, we used pharmacological inhibitors specific for different MAPKs (Figure 5). Pretreatment of THP-1 cells with the MAPK kinase (MEK) inhibitor U0126 inhibited HBM-induced expression of CD14, TNFα, IL-1β, and CCL2 but did not affect TLR4 expression. In contrast, the p38 kinase inhibitor SB203580 blocked HBM-induced TLR4 expression but did not affect CD14, TNFα, IL-1β, CXCL8, or CCL2 expression. The JNK inhibitor SP600125 prevented the HBM-induced expression of TLR4, TNFα, IL-1β, CXCL8, and CCL2 but did not affect CD14 expression. These data suggested that all three major MAPKs were rapidly activated in response to HBM exposure and were differentially involved in the HBM-induced upregulation of innate immunity-related genes.
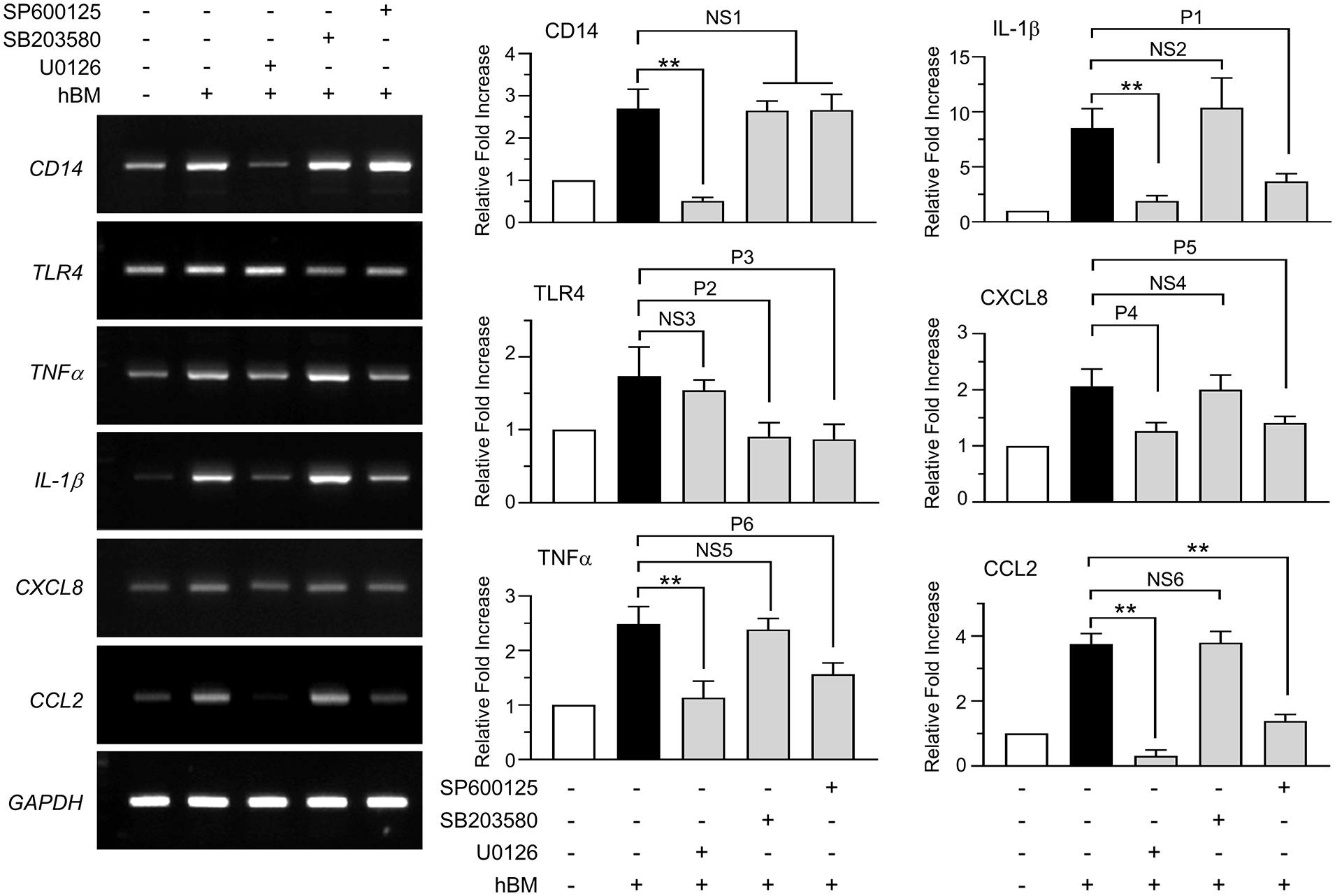
Effect of mitogen-activated protein kinases (MAPKs) on human breast milk (HBM)-induced expression of lipopolysaccharide (LPS) receptors and innate immune cytokines. THP-1 cells were either treated with MAPK inhibitors for 30 min or untreated, and then 25 μg/mL HBM was added. After 3 h of HBM treatment, total RNA was isolated, and RT-PCR was performed for pattern recognition receptors and innate immunity-related cytokines. GAPDH was used as an internal control. Individual band intensities were quantified using ImageJ software. Values were normalized to GAPDH mRNA (right graphs). The relative fold increases were compared with that of the vehicle-treated control, which was set to a value of 1. The data shown represent the mean ± SD (n = 3). NS (not significant) 1, p ⩾ 0.998; NS2, p = 0.414; NS3, p = 0.681; NS4, p = 0.981; NS5, p = 0.957; NS6, p = 0.999; P1, p = 0.009; P2, p = 0.004; P3, p = 0.003; P4, p = 0.002; P5, p = 0.008; P6, p = 0.003; **, p < 0.001, as compared to untreated control group.
Lacto-N-fucopentaose I contributes to HBM-induced immunostimulation
Human milk oligosaccharides (HMOs) are the third most abundant component of HBM after lactose and lipids. 22 HMOs provide many health-promoting effects needed for infants. 23 Up to now, more than 15 structures of HMOs have been identified; however, their quantities and structural modifications are varied at different stages of lactation and genetic background of mothers. 24 Thus, we selected major HMOs, including fucosylated HMO, such as 2′-fucosyllactoses (2′-FL), and neutral N-containing HMOs, such as lacto-N-fucopentaose I (LNFP-I) and lacto-N-tetraose (LNT), which were commercially available. Their effects on the expression of pattern recognition receptors and innate cytokines/chemokines were examined in THP-1 cells. As shown in Figure 6, LNFP-I significantly (all p < 0.01) elevated expression of TLR2, TLR4, CD14, and innate cytokines/chemokines, including TNFα, IL-1β, CCL2, and CXCL8, in a dose-dependent manner. In contrast, 2′-FL and LNT had little effect compared to LNFP-I. These results suggested that certain HMOs, such as LNFP-I, may be involved in HBM-induced innate immune responses of THP-1 cells.
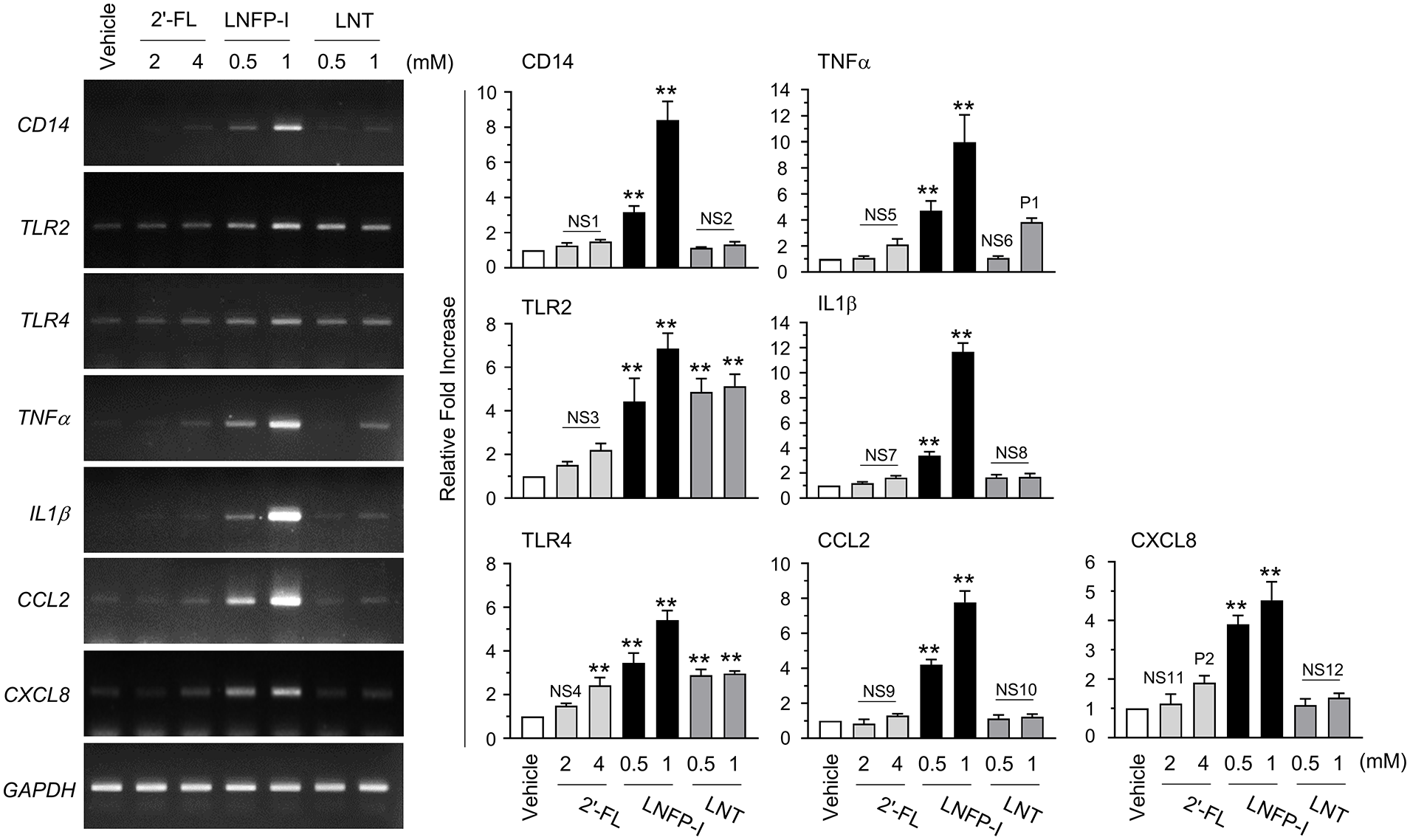
Effect of human milk oligosachharide (HMO) on the mRNA expression of pattern recognition receptors and innate immunity-related cytokines. THP-1 cells were treated with different concentrations of 2′-fucosyllactoses (2′-FL), lacto-N-fucopentaose I (LNF-I), and lacto-N-tetraose (LNT) for 12 h. Total RNAs were isolated, and RT-PCR was performed for pattern recognition receptors and innate immunity-related cytokines. GAPDH was used as an internal control. Individual band intensities were quantified using ImageJ software. Values were normalized to GAPDH mRNA (right graphs). The relative fold increases were compared with that of the vehicle-treated control, which was set to a value of 1. The data shown represent the mean ± SD (n = 3). NS (not-significant) 1, p ⩾ 0.553; NS2, p ⩾ 0.851; NS3, p ⩾ 0.102; NS4, p = 0.218; NS5, p ⩾ 0.469; NS6, p = 0.999; NS7, p ⩾ 0.125; NS8, p ⩾ 0.080; NS9, p ⩾ 0.691; NS10, p ⩾ 0.854; NS11, p = 0.021; NS12, p ⩾ 0.548; P1, p = 0.007; P2, p = 0.021; **, p < 0.001, as compared to vehicle-treated control group.
Discussion
The innate immune response acts as the first line of defense against extrinsic infectious agents. 25 The principal function of the innate immune system is to remove infectious agents, including pathogenic microbes, to recruit multiple immune cells to the infection site, and to activate the immune cells. 26 The pathogen-associated innate immune responses rely on phagocytic cells that recognize, uptake, and destroy invading microorganisms. However, the failure to rapidly remove dangerous pathogens could prolong inflammatory conditions that may cause immunosuppression and harmful side effects to host tissues. 27 Therefore, the immediate recognition and removal of pathogens are essential for providing the first defense line against foreign pathogens. However, infants have a weak or immature innate immune system, and their intestinal epithelium acts as a defensive barrier against pathogenic microbes.
TLR2 and TLR4 are the central pattern recognition receptors recognizing Gram-positive peptidoglycan and gram-negative LPS, respectively. 28 CD14 delivers the bacterial ligands to TLR2 or TLR4, thereby stimulating the innate immune responses.4–6 Indeed, transgenic mice overexpressing CD14 are susceptible to LPS-induced endotoxin shock. 29 On the contrary, CD14-deficient mice exhibit high resistance to endotoxin shock induced by LPS or gram-negative bacteria. 30 Therefore, CD14 plays an essential role in recognizing bacterial pathogens and the modulation of innate immune responses. Previous studies have demonstrated that HBM contains soluble forms of CD14 (sCD14) 31 and soluble TLR2 (sTLR2), 32 suggesting that the bioactive components modulating innate immunity are present in HBM. However, it is poorly understood whether HBM induces CD14 expression on the surface of monocytes/macrophages.
In the current study, we investigated the effect of HBM on the expression of CD14 in monocytes. Our results showed that in THP-1 monocytes, HBM enhanced the expression of CD14 and its co-receptors TLR2 and TLR4, within 12 h of exposure. These results further supported the notion that HBM exhibits an immunostimulating potential. CD14+ monocytes are the putative precursors of inflammatory dendritic cells, which are the most potent antigen-presenting cells that function to activate T-lymphocytes and B-lymphocytes, which link the innate and adaptive immune systems. 33 Thus, in addition to intestinal immunity, it is also possible that HBM may trigger the differentiation of monocytes into CD14+ dendritic cells to strengthen the protective immunomodulatory mechanisms in infants.
Multiple pattern recognition receptors recognize distinct PAMPs. Although we have focused on the expression of bacterial pattern recognition receptors CD14, TLR2, and TLR4, we cannot rule out the possibility that HBM may also modulate the expression of other pattern recognition receptors. Also, it is unclear whether HBM may modulate the expression of pattern recognition receptors in immune cells in addition to monocytes, such as neutrophils and T-lymphocytes. Further studies are required to fully identify the pattern recognition receptors and target immune cells modulated by HBM in vivo.
TNFα is a pro-inflammatory cytokine implicated in dendritic cell differentiation and the initiation of innate immune defenses against bacterial infection. 34 Meanwhile, IL-1β enhances the expression of adhesion molecules in endothelial cells, enabling the transmigration of immune cells such as monocytes and lymphocytes from the blood to sites of infection. 35 The chemoattractant chemokine CCL2 functions to recruit innate immune cells such as dendritic cells, monocytes, and T-lymphocytes to inflammatory sites. 36 CXCL8, also known as IL-8, modulates macrophage phagocytosis and the chemotaxis of neutrophils to infection sites, leading to the activation of innate immune responses. 37 Interestingly, RT-PCR analysis revealed that HBM rapidly increased the expression of various innate cytokines within 3 h of treatment, including pro-inflammatory cytokines TNFα and IL-1β and chemokines CXCL8 and CCL2. Therefore, we suggest that HBM may help to stimulate immune responses by enhancing innate immune cytokines in unstimulated immune cells. This concept is further supported by the observation that HBM was able to stimulate latex bead-induced phagocytosis by RAW264.7 macrophages. These results support the notion that HBM may modulate innate immune responses through the priming of immune cells, particularly monocyte/macrophages (Figure 7). Therefore, HBM apparently exhibits strong immunostimulatory characteristics that may enhance the early recognition of invading pathogens.

Hypothetical mode of action of human breast milk (HBM) for stimulation of innate immune responses. CCL2, C-C motif chemokine ligand 2; CD14, cluster of differentiation 14; CXCL8, C-X-C motif chemokine 8; IL-1β, interleukin-1 beta; PAMP, pathogen-associated molecular pattern; TLR, toll-like receptor; TNFα, tumor necrosis factor alpha.
HBM contains a variety of nutrients and bioactive molecules such as proteins, carbohydrates, and HMOs. Most of the components of HBM are digested through the gastrointestinal tract, but HMOs, the third most abundant solid component of breast milk after lactose and lipids, 22 are resistant to the low pH environment of the stomach and the enzymatic hydrolysis of the upper gastrointestinal tract. 38 The majority of the HMOs that reach the colon are metabolized by the gut microbes; however, specific structures of HMOs can be absorbed intact through the intestinal epithelium and transported into the systemic circulation. 39 In this study, we observed that LNFP-I enhances the expression of innate immunity-related genes in THP-1 cells. This finding suggests that HMOs in the systemic circulation might be involved in modulating innate immune responses. However, the molecular mechanism underlying LNFL-I-induced upregulation of innate immunity-related genes remains to be evaluated.
There are several limitations of this study; (i) The innate immune-stimulating effect of HBM was analyzed at the cellular level using mouse monocyte/macrophage-like cell line RAW264.7 cells for analyzing phagocytosis and human monocyte-like cell line THP-1 cells for analyzing the expression of innate immunity-related genes. Results using the THP-1 cells in vitro may not be replicated in vivo. Further physiologically relevant in vivo studies are needed to provide a profound understanding of the potential of HBM as a functional immune stimulation supplement. (ii) Many components of HBM have been shown to modulate immune function. Of these, commercially available major HMOs, such as 2′-FL, LNFP-I, and LNT, were selected in this study to test the effect on the induction of innate immunity-related genes. The composition of hBM and the concentration of HMO may be driven by several maternal factors, such as genetic background, nutritional status, lifestyles, smoking/drug exposure, and lactation stage.24,40 To better understand the beneficial effect of HBM on the innate immune response, further extensive and controlled studies are needed to address the range and specificity of other HMOs in more detail.
Conclusion
HBM can ameliorate the innate immune system of infants/children by upregulating the expression of various innate cytokines and pattern recognition receptors such as CD14 and TLR2/4 in monocytes/macrophages.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Research Foundation of Korea (NRF) grant funded by the Korean government, MSIT (No. 2020R1A2C1005082). The data expressed in the submitted article are the author’s own and not an official position of the institution or funder.
