Abstract
Two members of the N-acetylglucosamine-6-O-sulfotransferase (GlcNAc6ST) family, GlcNAc6ST-1 and GlcNAc6ST-2, function in the biosynthesis of 6-sulfo sialyl Lewis X–capped glycoproteins expressed on high endothelial venules (HEVs) in secondary lymphoid organs. Thus, both enzymes play a critical role in L-selectin-expressing lymphocyte homing. Human GlcNAc6ST-1 is encoded by a 1593-bp open reading frame exhibiting two 5′ in-frame methionine codons spaced 141 bp apart. Both resemble the consensus sequence for translation initiation. Thus, it has been hypothesized that both long and short forms of GlcNAc6ST-1 may be present, although endogenous expression of either form has not been confirmed in humans. Here, the authors developed an antibody recognizing amino acid residues between the first two human GlcNAc6ST-1 methionines. This antibody specifically recognizes the long form of the enzyme, a finding validated by Western blot analysis and immunofluorescence cytochemistry of HeLa cells misexpressing long and/or short forms of human GlcNAc6ST-1. Using this antibody, the authors carried out immunofluorescence histochemistry of human lymph node tissue sections and found endogenous expression of the long form of the enzyme in human tissue, predominantly in the trans-Golgi network of endothelial cells that form HEVs.
Circulating lymphocytes routinely home to secondary lymphoid organs such as lymph nodes, tonsils, and Peyer’s patches, where they recognize cognate antigens by interacting with antigen-presenting cells. Such homing is tightly regulated by sequential adhesive interactions. The initial step of the interaction, called “tethering and rolling,” is mediated by the carbohydrate-binding protein L-selectin expressed on lymphocytes and by its carbohydrate ligand peripheral lymph node addressin (PNAd), expressed on the luminal surface of high endothelial venules (HEVs). This step is a prerequisite for subsequent lymphocyte chemokine-dependent activation, integrin-mediated firm attachment to the endothelium, and transmigration across blood vessels (Springer 1994; Butcher and Picker 1996; von Andrian and Mempel 2003). PNAd is expressed not only on HEVs in secondary lymphoid organs but also on HEV-like vessels induced in various non-lymphoid organs under chronic inflammatory states (Michie et al. 1993; Salmi et al. 1994; Renkonen et al. 2002; Kobayashi et al. 2004; Aloisi and Pujol-Borrell 2006). Moreover, PNAd is also expressed in gastric mucosa-associated lymphoid tissue (MALT) lymphoma, a neoplastic lesion resulting from chronic Helicobacter pylori gastritis (Dogan et al. 1997; Kobayashi et al. 2011).
PNAd consists of a group of glycoproteins recognized by the MECA-79 monoclonal antibody (Streeter et al. 1988; Rosen 2004), which has an epitope that has been shown to be 6-sulfo N-acetyllactosamine (LacNAc) attached to extended core 1 O-glycans, Galβ1→4(sulfo→6)GlcNAcβ1 →3Galβ1→3GalNAcα1→Ser/Thr (Yeh et al. 2001). MECA-79 also recognizes the epitope’s sialylated and fucosylated form, 6-sulfo sialyl Lewis X, attached to extended core 1 O-glycans, sialic acid2→3Galβ1→4[Fuc α1→3(sulfo→6)]GlcNAcβ1→3Galβ1→3GalNAcα1→ Ser/Thr. N-acetylglucosamine (GlcNAc)-6-O-sulfation of the sialyl Lewis X tetrasaccharide, which is critical for L-selectin binding (Imai et al. 1993), is catalyzed by GlcNAc-6-O-sulfotransferases (GlcNAc6STs), which transfer sulfate from 3′-phosphoadenosine 5′-phosphosulfate (PAPS) to the 6-O position of GlcNAc residues (Fukuda et al. 2001; Grunwell and Bertozzi 2002). Thus far, five members of the GlcNAc6ST family have been cloned in humans, four of which have murine orthologues (Uchimura and Rosen 2006). Among them, GlcNAc6ST-1 (Uchimura et al. 1998; Li and Tedder 1999) and GlcNAc6ST-2 (Bistrup et al. 1999; Hiraoka et al. 1999) have been confirmed to be expressed in HEVs, and both play a critical role in L-selectin ligand biosynthesis (Kawashima et al. 2005; Uchimura et al. 2005). Relevant to human pathological states, we previously reported that the number of PNAd-expressing HEV-like vessels in the colonic lamina propria is increased in active ulcerative colitis (UC) compared with the number seen in remission phase UC and that such an increase is associated with increased levels of transcripts encoding GlcNAc6ST-1 (Suzawa et al. 2007; Kobayashi et al. 2009).
GlcNAc6ST-1 is a type II transmembrane protein composed of a short N-terminal cytoplasmic tail, a hydrophobic single-pass transmembrane domain, an intervening stem region, and a C-terminal catalytic domain that resides in the Golgi lumen (Grunwell and Bertozzi 2002). Human GlcNAc6ST-1 was cloned as a 1593-bp open reading frame showing two in-frame methionine codons at the 5′ end, spaced 141 bp apart from each other. Both potential start sites agreed with the consensus sequence for translation initiation (Kozak 1991) (Fig. 1). One of the authors of this study previously proposed that both long and short forms of GlcNAc6ST-1 are expressed (Uchimura et al. 1998). Thus far, in vitro studies employing cell culture and misexpression of human GlcNAc6ST-1 have characterized the biochemistry and function of the enzyme in detail (Uchimura et al. 1998, 2002; Tangemann et al. 1999; Bhakta et al. 2000; Li et al. 2001; Grunwell et al. 2002; Lee et al. 2003; de Graffenried and Bertozzi 2003, 2004; Desko et al. 2009); however, most have used expression vectors harboring cDNA encoding short and/or even shorter engineered soluble forms of the enzyme. In the only study using an expression vector harboring the long-form cDNA, the authors confirmed its mRNA expression by Northern blot analysis; however, expression of the protein was not evaluated (Li et al. 2001). More important, expression of native human GlcNAc6ST-1 protein, regardless of form, has not yet been confirmed.

Nucleotide and deduced amino acid sequences of the N-terminal region of human GlcNAc6ST-1 (long form). The first two methionines are boxed, and the sequence encoding the putative transmembrane domain is doubly underlined. The sequence of the antigenic peptide used to produce anti-GlcNAc6ST-1-N antibody is underlined in bold.
In the present study, we developed an antibody recognizing amino acid residues between the first two methionines (designated M#1 and M#2, respectively) of human GlcNAc6ST-1 and found that the long form of the enzyme is endogenously expressed in humans, predominantly in the trans-Golgi network (TGN) of endothelial cells that form HEVs. We also found that both forms of the enzyme show comparable subcellular localization and intracellular enzymatic activity.
Materials and Methods
Generation of an Antibody against the N-Terminal Site of Human Long-Form GlcNAc6ST-1
We generated a polyclonal antibody recognizing only the long form of human GlcNAc6ST-1 by directing it to residues between M#1 and M#2. An immunogenic peptide, NH2-C+LPQWPRRPGRRWPA-COOH, corresponding to amino acids 30 to 43 of human GlcNAc6ST-1 (Fig. 1, bold line), was chemically synthesized and conjugated with keyhole limpet hemocyanin (KLH). The KLH-conjugated peptide was mixed with Freund’s complete adjuvant and subcutaneously injected into a Japanese white rabbit, followed by four additional injections with Freund’s incomplete adjuvant at 2-week intervals. The antibody was purified by affinity chromatography on a column coupled to the immunogenic peptide. The purified antibody, designated anti-GlcNAc6ST-1-N, was used for subsequent experiments. The experimental protocol was approved by the Institutional Animal Care and Use Committee at Operon Biotechnologies (Tokyo, Japan).
Construction of Expression Vectors Encoding Long and Short Forms of Human GlcNAc6ST-1 with or without a FLAG Tag
pcDNA1-GlcNAc6ST-1 M#1 encoding the long form of human GlcNAc6ST-1 was constructed previously (Kobayashi et al. 2009). pcDNA1-GlcNAc6ST-1 M#1-FLAG, which encodes the long form with a FLAG epitope tag fused to its C-terminus, was similarly constructed using the polymerase chain reaction (PCR) by replacing the 3′-primer with 5′-AA
Expression of Human GlcNAc6ST-1 in HeLa Cells
HeLa cells were transiently transfected with one of the above expression vectors using Lipofectamine Plus (Invitrogen; Carlsbad, CA), according to the manufacturer’s instructions, and analyzed 24 to 48 hr after transfection. To express long and short forms of GlcNAc6ST-1 simultaneously, HeLa cells were cotransfected with pcDNA1-GlcNAc6ST-1 M#1 and pcDNA1-GlcNAc6ST-1 M#2-FLAG at a ratio of 1:1.
Western Blot Analysis
HeLa cell transfectants were harvested in phosphate-buffered saline (PBS) supplemented with Complete Mini protease inhibitor (Roche; Basel, Switzerland) with a cell scraper and subjected to three cycles of freeze/thawing to disrupt the plasma membrane. The membrane fraction was collected by centrifugation at 12,000 × g for 10 min and resuspended in 10 mM Tris/HCl and 1 mM EDTA (pH 8.0). Subsequently, 10% Triton X-100 was added to a final concentration of 1%, the mixture was gently rocked at 4C for 15 min, and the Triton X-100-soluble membrane fraction was obtained by centrifugation at 12,000 × g for 10 min. To remove N-glycans on the protein, an aliquot of each membrane fraction containing 5 mg protein was treated with 125 units of peptide N-glycosidase F (PNGase F) (New England Biolabs; Ipswich, MA) at 37C for 2 hr, lysed in sample buffer, and incubated at 95C for 5 min. Each sample was separated by sodium dodecyl sulfate–polyacrylamide gel electrophoresis (SDS-PAGE) using SuperSep Ace 7.5% (Wako Pure Chemical Industries; Osaka, Japan) and transferred onto a polyvinylidene difluoride (PVDF) membrane (Millipore; Billerica, MA). After blocking with Tris-buffered saline (TBS) (pH 7.6) supplemented with 5% skim milk and 0.1% Tween-20 for 60 min, the membrane was incubated with anti-GlcNAc6ST-1-N (1:1000) and anti-FLAG M2 (Sigma-Aldrich; St. Louis, MO) (1:5000) at 4C overnight, followed by incubation with horseradish peroxidase (HRP)–conjugated anti-rabbit IgG (Cell Signaling Technology; Danvers, MA) (1:2500) and anti-mouse IgG (Immuno-Biological Laboratories; Gunma, Japan) (1:5000), respectively, for 60 min. Immunoreactive bands were visualized using SuperSignal West Dura Extended Duration Substrate (Thermo Scientific; Rockford, IL) and a luminescent image analyzer LAS-3000 (Fuji Film; Tokyo, Japan).
Immunofluorescence Staining
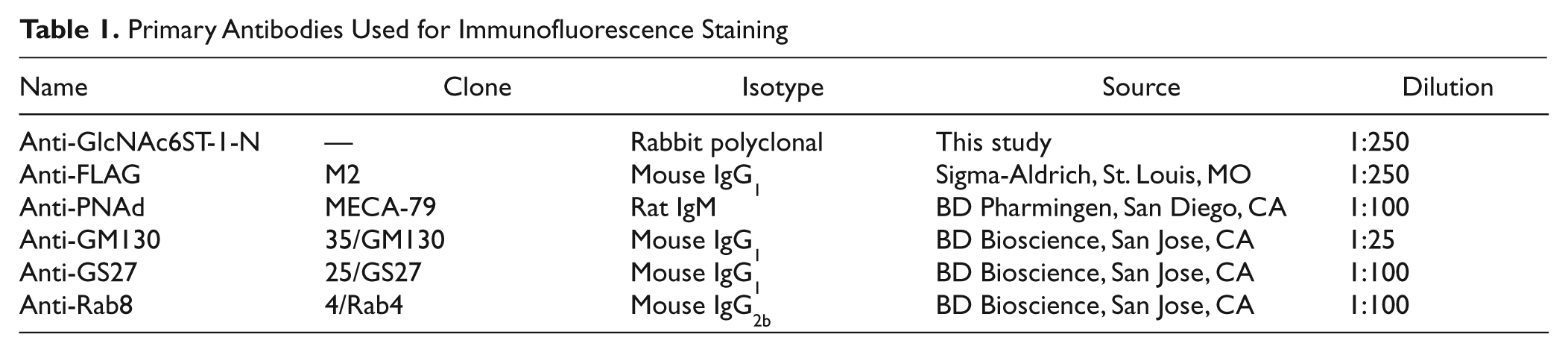
For immunofluorescence cytochemistry, HeLa cell transfectants grown on coverslips were fixed with 20% neutral-buffered formalin for 15 min, and cell membranes were permeabilized with 0.1% Triton X-100 in PBS for 15 min. For immunofluorescence histochemistry, fresh human lymph node tissues were embedded in Tissue-Tek OCT compound (Sakura Finetek; Tokyo, Japan) and frozen at −80C. Frozen tissues were sectioned at 6 mg, fixed with acetone for 5 min, and air-dried. Use of human lymph node tissues was approved by the Ethics Committee of Shinshu University School of Medicine. After blocking with 1% bovine serum albumin (BSA) (Sigma-Aldrich) in TBS for 15 min, samples were incubated with a cocktail of two primary antibodies for 15 min. After washing, samples were incubated for 15 min with a cocktail of two isotype-matched secondary antibodies differentially labeled with Alexa Fluor 488 and Alexa Fluor 555 (Invitrogen) (1:1000). Samples were mounted with Vectashield mounting medium (Vector Laboratories; Burlingame, CA) and observed under a fluorescence microscope AX-80 (Olympus; Tokyo, Japan). Primary antibodies used for immunofluorescence are listed in Table 1.
Primary Antibodies Used for Immunofluorescence Staining
Cell Enzyme-Linked Immunosorbent Assay
HeLa cells were seeded into 96-well plates (BD Falcon; Franklin Lakes, NJ) at 2 × 104 cells/well and 24 hr later transiently transfected with one of the following expression vectors: pcDNA1-GlcNAc6ST-1 M#1, pcDNA1-GlcNAc6ST-1 M#2, pcDNA1-GlcNAc6ST-2, and pcDNA1 (mock). Forty-eight hours later, cells were fixed with 20% neutral-buffered formalin for 15 min. To quench endogenous peroxidase activity, cells were treated with 0.3% hydrogen peroxide in absolute methanol for 30 min and washed with TBS. After blocking with 1% BSA in TBS for 30 min, cells were incubated for 60 min with 0.5 µg/ml of S2 monoclonal antibody recognizing 6-sulfo sialyl LacNAc on N- and O-glycans (Hirakawa et al. 2010). After washing with TBS, cells were incubated with HRP-conjugated anti-mouse IgM (Jackson ImmunoResearch; West Grove, PA) (1:5000) for 60 min. After washing, 100 ml of 1-Step ABTS (Thermo Scientific) was applied to each well, and absorbance at 405 nm was read using a microplate reader (DS Pharma Biomedical; Osaka, Japan).
Fluorescence-Activated Cell Sorting Analysis
HeLa cell transfectants were dissociated into mono-dispersed cells in PBS containing 0.5 mM EDTA and fixed with 20% neutral-buffered formalin for 15 min. Cells were then permeabilized with PBS/0.1% Triton X-100 for 15 min and incubated with anti-FLAG M2 (1:250), followed by Alexa Fluor 488–conjugated anti-mouse IgG (1:1000). Stained cells were analyzed using FACSort (BD Biosciences; San Jose, CA) with FlowJo software (Tree Star; Ashland, OR).
Semi-Quantitative Reverse Transcriptase PCR
Semi-quantitative reverse transcriptase PCR (RT-PCR) was carried out essentially as described (Kobayashi 2006). Total RNA was extracted from HeLa cell transfectants using ISOGEN reagent (Nippon Gene; Tokyo, Japan) according to the manufacturer’s instructions. Single-stranded cDNA was synthesized as described (Suzawa et al. 2007). PCR was then carried out with primers for GlcNAc6ST-1, 5′-TCCTCCAAGCCTTTCGTGGTATCT-3′ (5′-primer) and 5′-TGGTAGCAAAACTCCTCCACCTGT-3′ (3′-primer), and glyceraldehyde-3-phosphate dehydrogenase (GAPDH), 5′-TGAGTACGTCGTGGAGTCCACT-3′ (5′-primer) and 5′-CAGAGATGATGACCCTTTTGGCTC-3′ (3′-primer). After initial denaturation at 94C for 2 min, 24 cycles of amplification with denaturation at 96C for 20 sec, annealing at 65C for 30 sec, and extension at 72C for 30 sec were performed, followed by a final extension at 72C for 2 min. PCR products were electrophoresed on 2% agarose gels containing 0.1 µg/ml ethidium bromide.
Statistical Analysis
Data are expressed as means ± SD. Differences among groups were statistically analyzed by one-way analysis of variance (ANOVA) with Bonferroni’s posttest, using InStat 3 software (GraphPad Software; San Diego, CA). p values less than 0.05 were considered significant.
Results
Anti-GlcNAc6ST-1-N Specifically Recognizes the Long Form of GlcNAc6ST-1
To determine whether anti-GlcNAc6ST-1-N selectively recognizes the long form of GlcNAc6ST-1, the membrane fraction of HeLa cell transfectants was subjected to Western blot analysis. As shown in Fig. 2 (left panel), immunoblotting with anti-FLAG of samples not treated with PNGase F showed multiple immunoreactive bands migrating at ~60 kDa for the long form and ~55 kDa for the short form. In addition, immunoreactive species migrating at >100 kDa, the molecular weight of enzyme homodimers, were also detected with both forms of the enzyme, as described previously (de Graffenried and Bertozzi 2004). The appearance of multiple bands is consistent with a previous study using HeLa cells transfected with wild-type or mutant forms of GlcNAc6ST-1, which demonstrated that at least three of four potential N-glycosylation sites were glycosylated (Desko et al. 2009). Indeed, PNGase F digestion converted the multiple bands into a single major band migrating at ~55 kDa for the long form and ~50 kDa for the short form. By contrast, immunoblots using anti-GlcNAc6ST-1-N (Fig. 2, right panel) revealed immunoreactive species only for the long form, and the migration patterns of these bands were similar to those detected by immunoblotting with anti-FLAG.
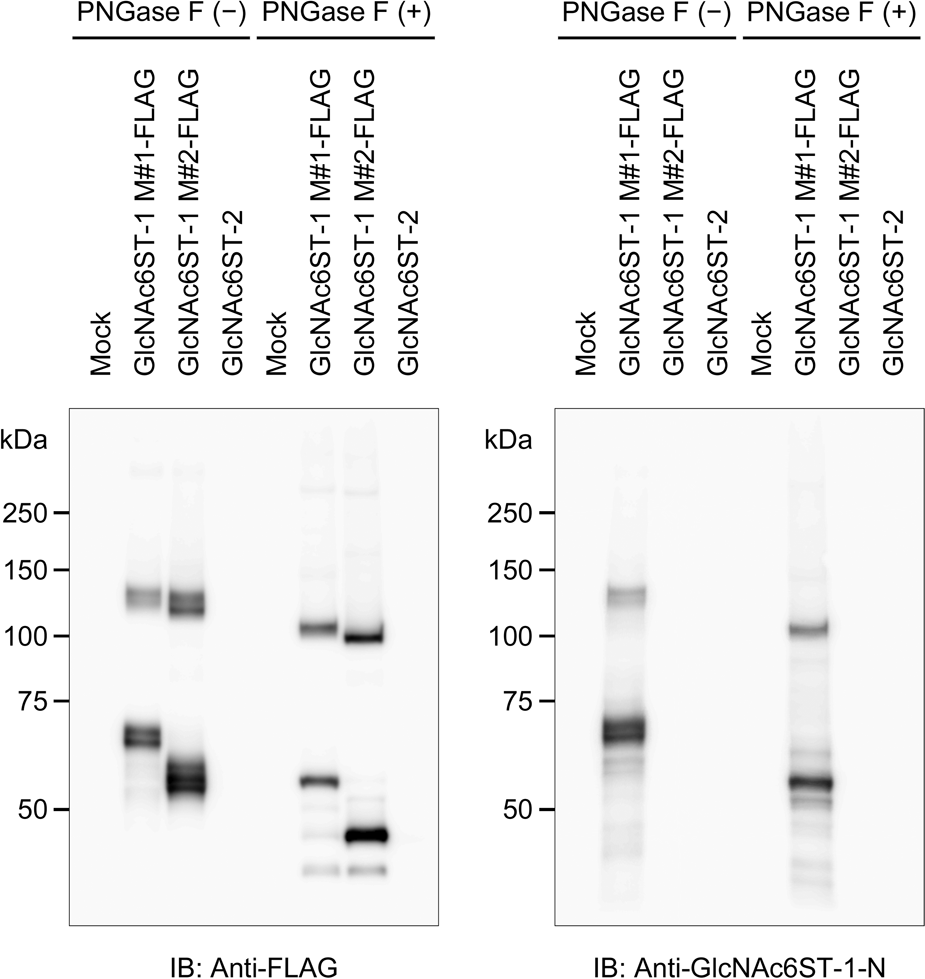
Western blot analysis of long and short forms of human GlcNAc6ST-1. The membrane fraction of HeLa cells transiently transfected with pcDNA1 (mock), pcDNA1-GlcNAc6ST-1 M#1-FLAG, pcDNA1-GlcNAc6ST-1 M#2-FLAG, or pcDNA1-GlcNAc6ST-2, with or without PNGase F digestion, was subjected to sodium dodecyl sulfate–polyacrylamide gel electrophoresis and probed with anti-FLAG (left panel) and anti-GlcNAc6ST-1-N (right panel) antibodies. Molecular weights are indicated to the left of each blot.
We then performed double immunofluorescence staining of HeLa cell transfectants using anti-FLAG and anti-GlcNAc6ST-1-N antibodies. Cells transfected with pcDNA1-GlcNAc6ST-1 M#1-FLAG and pcDNA1-GlcNAc6ST-1 M#2-FLAG exhibited FLAG signals chiefly in a perinuclear, punctate pattern (Fig. 3, left panels). By contrast, anti-GlcNAc6ST-1-N antibody stained only cells transfected with pcDNA1-GlcNAc6ST-1 M#1-FLAG and pcDNA1-GlcNAc6ST-1 M#1, that is, expression vectors harboring cDNA encoding the long form of GlcNAc6ST-1 (Fig. 3, middle panels). In the case of cells transfected with pcDNA1-GlcNAc6ST-1 M#1-FLAG (Fig. 3, second row), the staining pattern of anti-GlcNAc6ST-1-N was similar to that of anti-FLAG, as judged by yellow signals seen in the merged image (Fig. 3, right panels). Overall, these findings indicate that the anti-GlcNAc6ST-1-N antibody specifically recognizes the GlcNAc6ST-1 long form.
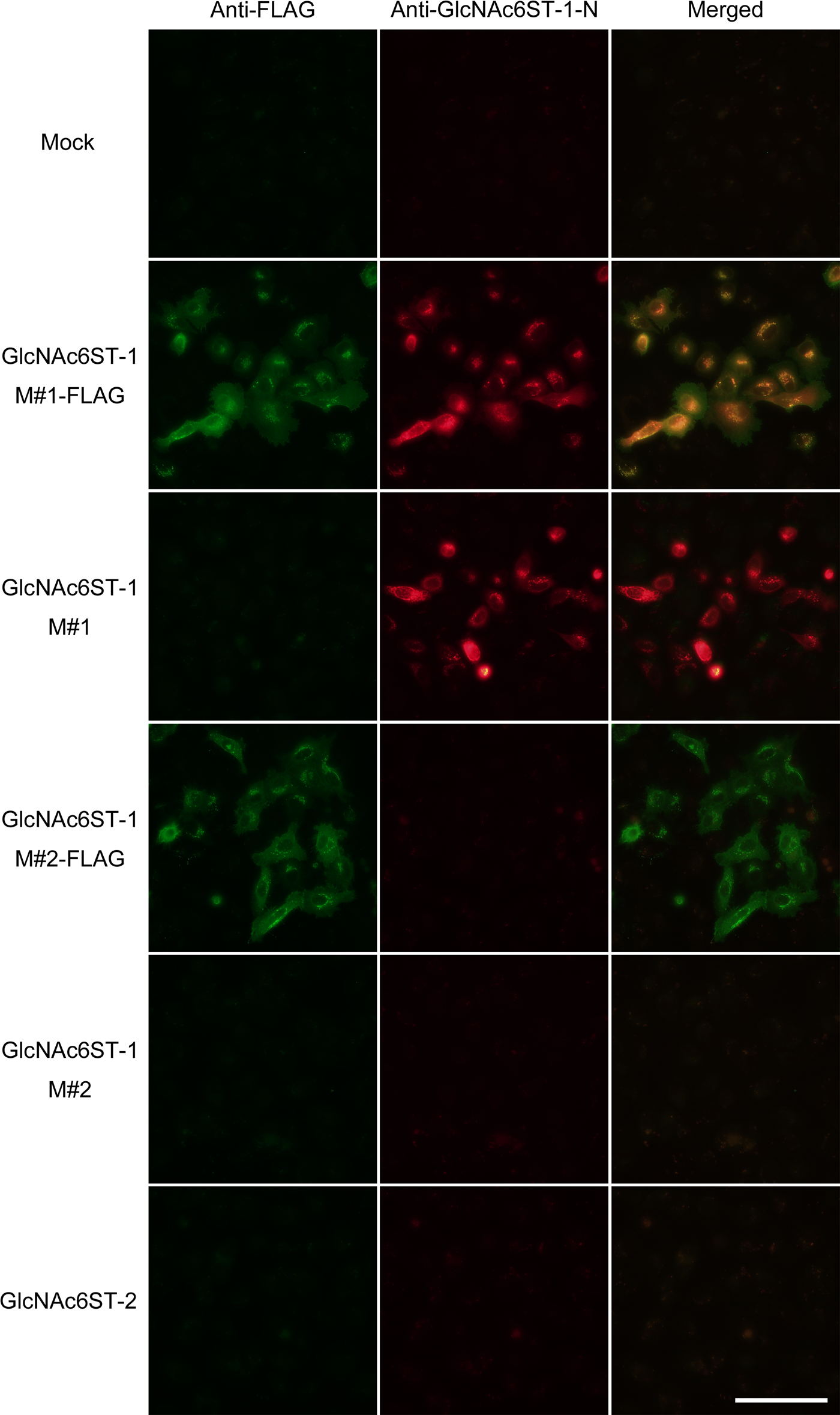
Specificity of anti-GlcNAc6ST-1-N antibody as assessed by immunofluorescence cytochemistry. HeLa cells were transiently transfected with expression vectors harboring cDNA encoding long (M#1) and short (M#2) forms of human GlcNAc6ST-1 with or without a FLAG epitope tag, as well as GlcNAc6ST-2 and mock (pcDNA1) as controls. Cells were doubly immunostained with anti-FLAG (green) and anti-GlcNAc6ST-1-N (red). Yellow signals in merged images indicate colocalization of the two antigens. Bar = 100 µm.
Comparable Subcellular Localization of the Long and Short Forms of GlcNAc6ST-1
To determine potential differences in subcellular localization of long and short GlcNAc6ST-1 forms, HeLa cells cotransfected with pcDNA1-GlcNAc6ST-1 M#1 and pcDNA1-GlcNAc6ST-1 M#2-FLAG were subjected to double immunofluorescence staining with anti-GlcNAc6ST-1-N (for the long form) and anti-FLAG (for the short form) (Fig. 4A). As shown in Fig. 4B, signals for the long form showed a perinuclear punctate pattern, indicative of the Golgi apparatus, and almost all signals colocalized with those for the short form, indicating that the subcellular localization of both forms does not differ substantially.
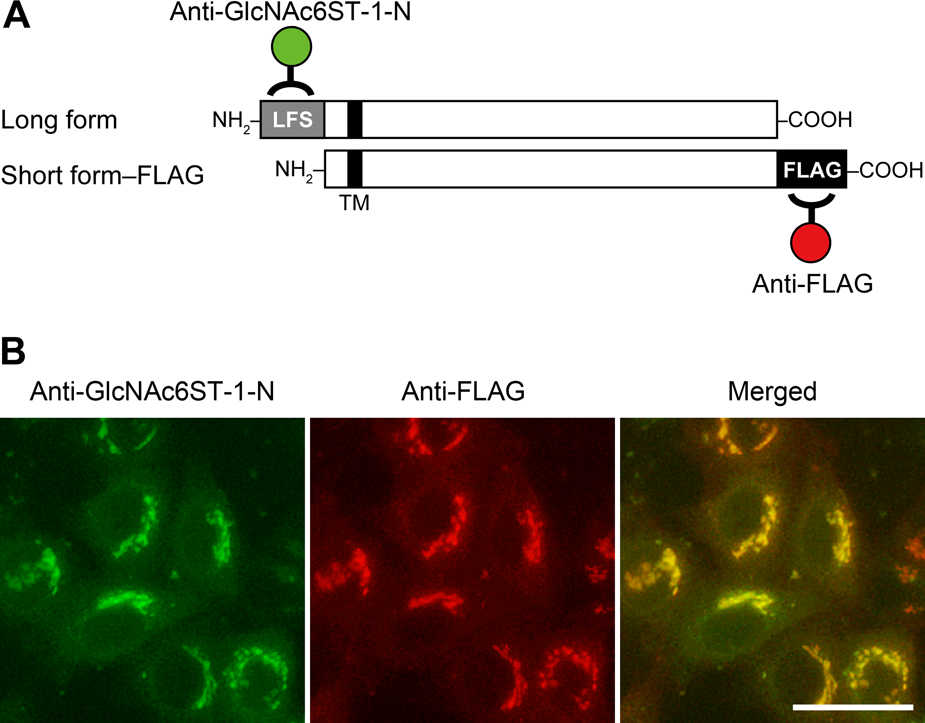
Subcellular localization of long and short forms of human GlcNAc6ST-1. (A) Schematic representation of long-form GlcNAc6ST-1 and short-form GlcNAc6ST-1 with a FLAG epitope tag. The antibody-binding site for anti-GlcNAc6ST-1-N (green) and anti-FLAG (red) is also indicated. LFS, long form–specific site. (B) HeLa cells doubly transfected with pcDNA1-GlcNAc6ST-1 M#1 and pcDNA1-GlcNAc6ST-1 M#2-FLAG were subjected to dual immunofluorescence staining for anti-GlcNAc6ST-1-N (green) and anti-FLAG (red). Yellow signals in merged images indicate colocalization of the two antigens. Bar = 50 µm.
Long and Short Forms of Human GlcNAc6ST-1 Show Comparable Intracellular GlcNAc-6-O-Sulfation Activity
To evaluate potential differences in intracellular GlcNAc-6-O-sulfation activity between long and short GlcNAc6ST-1 forms, cell enzyme-linked immunosorbent assays (ELISAs) for sulfation activity were carried out using HeLa cell transfectants. As shown in Fig. 5A, all GlcNAc6ST transfectants tested showed intracellular GlcNAc-6-O-sulfation activity, as assessed by reactivity of the S2 monoclonal antibody, which recognizes 6-sulfo sialyl LacNAc on N- and O-glycans (Hirakawa et al. 2010). The activity difference between both forms of GlcNAc6ST-1 was not statistically significant, but both GlcNAc6ST-1 forms demonstrated higher intracellular GlcNAc-6-O-sulfation activities than did GlcNAc6ST-2 (p<0.05).
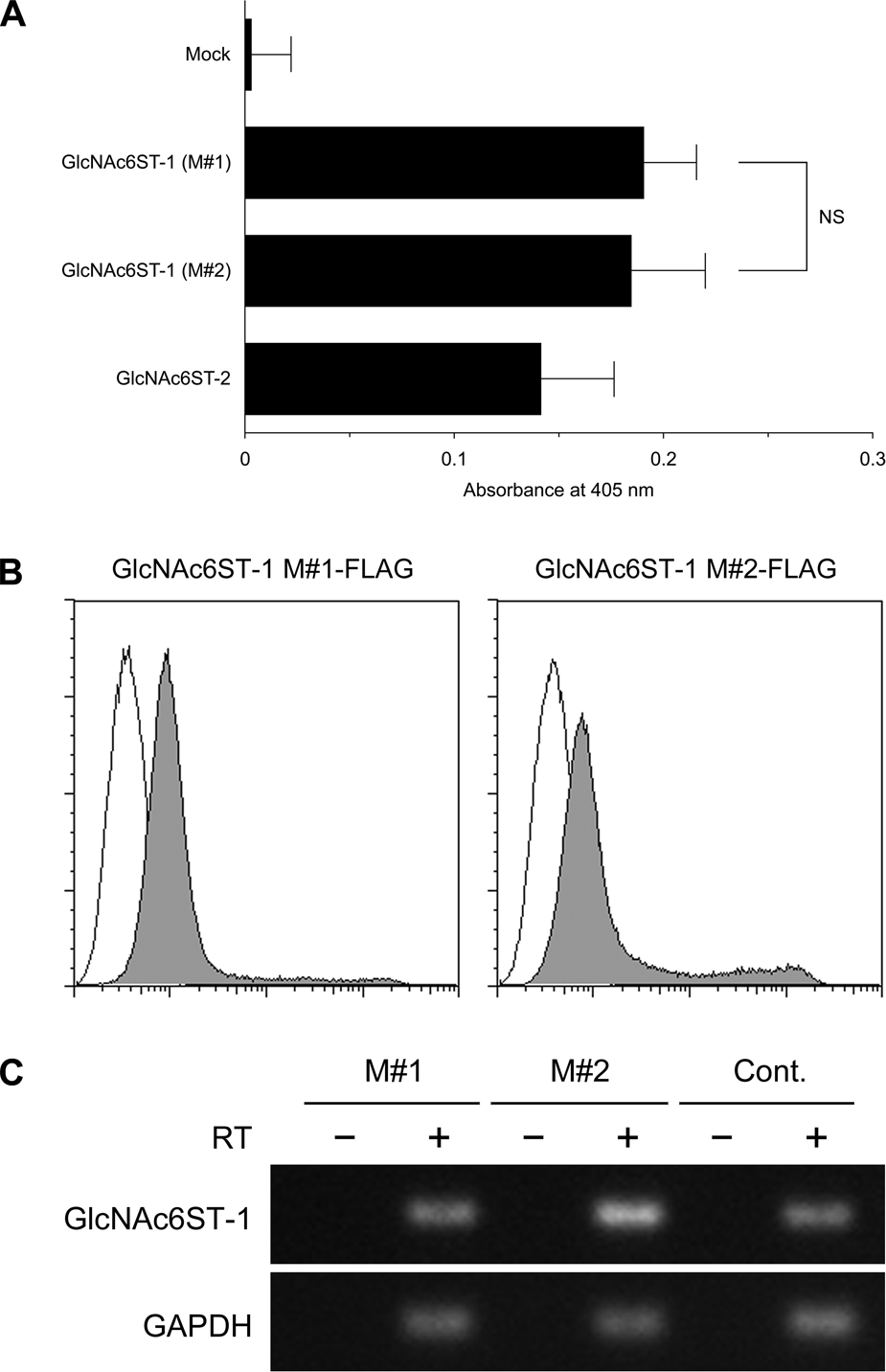
(A) Cell enzyme-linked immunosorbent assay (ELISA) showing intracellular GlcNAc-6-O-sulfation activity of GlcNAc6ST-1 M#1 and GlcNAc6ST-1 M#2. HeLa cells were transiently transfected with pcDNA1-GlcNAc6ST-1 M#1 and pcDNA1-GlcNAc6ST-1 M#2, as well as pcDNA1-GlcNAc6ST-2 and pcDNA1 (mock) as controls, and subjected to cell ELISA for S2 monoclonal antibody recognizing 6-sulfo sialyl LacNAc on N- and O-glycans. Data are expressed as means ± SD (n = 8 for each group). NS, not significant. (B) Intracellular expression levels of long (left panel) and short (right panel) forms of GlcNAc6ST-1 protein with a C-terminal FLAG epitope tag (gray histograms). Cells were stained for FLAG and subjected to fluorescence-activated cell sorting analysis. Open histograms represent negative control resulting from omitting the primary antibody. The x- and y-axes indicate fluorescence intensity and number of events, respectively. (C) Semi-quantitative RT-PCR showing mRNA expression levels of HeLa cells transfected with long (M#1) and short (M#2) forms of GlcNAc6ST-1 cDNA. Each RNA sample was treated with (+) or without (–) reverse transcriptase (RT). Cont., control amplification using distilled water (–) and plasmid harboring the target cDNA (+). GAPDH, glyceraldehyde-3-phosphate dehydrogenase.
We next performed fluorescence-activated cell sorting (FACS) analysis of HeLa cell transfectants to evaluate expression levels of long and short forms of GlcNAc6ST-1 protein. As shown in Fig. 5B, expression of the short-form protein (right panel) was higher compared with that of the long form (left panel). This finding confirms findings from our Western blot analysis that demonstrated that the intensity of immunoreactive bands representing the short form of the protein was greater than that of the long form (Fig. 2, left panel). In addition, semi-quantitative RT-PCR demonstrated that mRNA expression of short-form GlcNAc6ST-1 was higher than that of the long form (Fig. 5C).
The Long Form of GlcNAc6ST-1 Is Expressed Endogenously in Human HEVs
Finally, to determine whether the GlcNAc6ST-1 long form is endogenously expressed in human tissues, we performed double immunofluorescence staining of human lymph node tissue sections using anti-GlcNAc6ST-1-N and the anti-PNAd antibody MECA-79. As shown in Fig. 6A, MECA-79 clearly stained HEVs, predominantly the cell membrane of endothelial cells. By contrast, anti-GlcNAc6ST-1-N signals were detected in HEVs in a cytoplasmic perinuclear punctate pattern, indicative of the Golgi apparatus. This anti-GlcNAc6ST-1-N staining was abolished by addition of the peptide used for immunization (Fig. 6B), confirming antibody specificity.
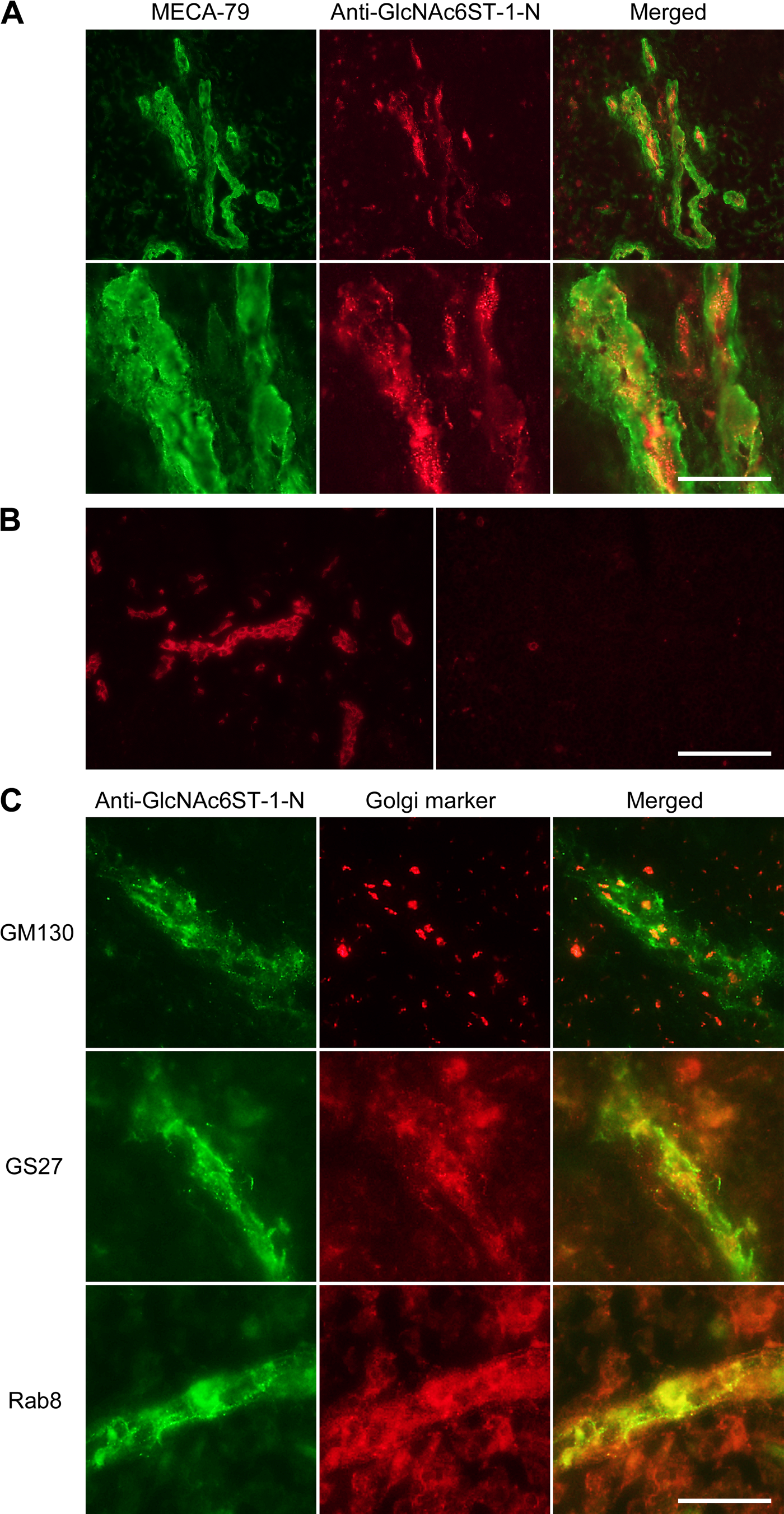
Endogenous expression of long-form GlcNAc6ST-1 protein as revealed by immunofluorescence histochemistry of human lymph node tissue sections. (A) Double immunofluorescence staining with MECA-79 (green) and anti-GlcN Ac6ST-1-N (red). Lower panels are enlarged images of the upper panels. Bar = 100 µm for upper panels and 25 µm for lower panels. (B) A competitive inhibition assay on tissue sections for anti-GlcNAc6ST-1-N binding in which antibody is preincubated at 4C overnight with 5 µg/ml of synthetic peptide used for immunization. Anti-GlcNAc6ST-1 staining on high endothelial venules (left panel) was abolished by this procedure (right panel). Bar = 100 µm. (C) Double immunofluorescence staining with anti-GlcNAc6ST-1-N (green) and one of the following Golgi markers (red): cis-Golgi marker GM130 (upper panels), medial-to-trans-Golgi marker GS27 (middle panels), and a trans-Golgi network marker to plasma membrane protein Rab8 (lower panels). Bar = 25 µm.
To determine the sub-Golgi localization of this staining, we undertook double immunofluorescence staining for anti-GlcNAc6ST-1-N and a battery of Golgi markers. As shown in Fig. 6C, anti-GlcNAc6ST-1-N signals colocalized with those of Rab8, a membrane marker of the TGN (Chen et al. 1993), and partially colocalized with the medial-to-trans-Golgi marker GS27 (Lowe et al. 1997). Anti-GlcNAc6ST-1-N signals did not colocalize with the cis-Golgi marker GM130 (Nakamura et al. 1995). These findings, taken together, indicate that the long form of GlcNAc6ST-1 is endogenously expressed in human endothelial cells of HEVs and localizes primarily to the TGN.
Discussion
In the present study, we developed an antibody specific for the long form of human GlcNAc6ST-1. Previously, one of the authors of this article reported that human GlcNAc6ST-1 cDNA potentially encodes long and short isoforms (Uchimura et al. 1998). However, most in vitro studies of human GlcNAc6ST-1 have employed expression vectors encoding the short and/or soluble forms of the enzyme, which lack a putative 47–amino acid N-terminal cytoplasmic tail. Moreover, expression of native human GlcNAc6ST-1 protein has not been confirmed. This prompted us to clarify whether the long form of the enzyme is endogenously expressed in humans. Employing the antibody developed here, we found that the long form of GlcNAc6ST-1 protein is endogenously expressed in human tissues, predominantly in endothelial cells that form HEVs.
Western blot analysis indicated that both forms of GlcNAc6ST-1 protein migrate as multiple bands of different molecular weights, consistent with previous reports (de Graffenried and Bertozzi 2003, 2004; Desko et al. 2009). Similar to other Golgi-resident proteins, GlcNAc6ST-1 displays four consensus sequences for N-glycosylation (Bause 1983). Desko et al. (2009) determined that three of those are indeed modified with N-glycans—namely, N196, N410, and N428 (N243, N457, and N475 for the long form). Consistently, pretreatment with PNGase F altered the size of immunoreactive species of both long and short forms of GlcNAc6ST-1 expressed in HeLa cells.
We also employed HeLa cell transfectants to compare subcellular localization and intracellular enzymatic activity of both forms. Double immunofluorescence staining of HeLa cells transfected with both GlcNAc6ST-1 forms revealed no gross difference in their subcellular localization. It is noteworthy that β1,4-galactosyltransferase 1 (β4GalT-1), a type II membrane-bound enzyme that transfers galactose (Gal) to acceptor sugars in a β1,4-linkage, also exists in long and short forms (Lopez et al. 1991). Both forms share an 11–amino acid cytoplasmic tail, but the long form exhibits an additional 13–amino acid sequence and is preferentially targeted to the plasma membrane, whereas the short form resides primarily within the Golgi compartment. In addition to β4GalT-1, differential subcellular localization of two enzyme isoforms has been recently reported for glucuronyltransferase P (GlcAT-P) and lactosylceramide α2,3-sialyltransferase (GM3 synthase) (Kizuka et al. 2009; Uemura et al. 2009). However, our study shows that, unlike these enzymes, the 47–amino acid sequence in the short cytoplasmic tail of long-form GlcNAc6ST-1 does not affect its subcellular localization.
Cell ELISAs carried out in the present study also showed that both long and short forms of GlcNAc6ST-1 demonstrate comparable intracellular GlcNAc-6-O-sulfation activity. Catalytic properties and substrate specificities of N-terminally truncated/protein A fusions of human GlcNAc6STs were previously reported (Uchimura et al. 2002). In that study, both N-terminally truncated, soluble GlcNAc6ST-1 and the full-length short form of transmembrane GlcNAc6ST-1 showed similar substrate specificities and enzymatic activity, which is consistent with the present finding. It should be noted that expression of the short-form GlcNAc6ST-1 protein exceeded that of the long form, as evaluated by FACS and Western blot analyses. It is possible that long-form GlcNAc6ST-1 may exhibit higher GlcNAc-6-O-sulfation activity. Alternatively, the short form might have difficulty accessing the substrate, leading to less product elaboration. Considering the results of semi-quantitative RT-PCR analysis, the higher expression of the short-form GlcNAc6ST-1 protein is likely due to higher mRNA expression level compared with that seen for the long form.
Among GlcNAc6ST family members, GlcNAc6ST-1 is unique in having an N-terminal long cytoplasmic tail (in the long form) and an intervening stem region between the transmembrane and catalytic domains. A previous study reported that GlcNAc6ST-1 sulfotransferase is confined to the TGN, whereas GlcNAc6ST-2 is distributed throughout the Golgi apparatus (de Graffenried and Bertozzi 2003). To clarify the mechanism underlying differential localization, those authors constructed a chimera comprising the localization domain (for the short form) and the stem region of GlcNAc6ST-1 fused to the GlcNAc6ST-2 catalytic domain and found that the chimeric protein was confined to the TGN and adopted GlcNAc6ST-1 substrate preference. Our result is consistent with these results because both long and short forms of GlcNAc6ST-1 expressed in HeLa cells contain the putative localization domain proposed by these authors.
Although short-form GlcNAc6ST-1 can be misexpressed in HeLa cells in vitro, it is not known whether the short form is indeed expressed endogenously in humans. It has been reported that a suboptimal context around the first AUG codon causes some 40S ribosomal subunits to bypass the first AUG and initiate at a second AUG codon (Kozak 1989, 1991). Given our observation of endogenous long-form GlcNAc6ST-1, future studies are required to determine whether the short form is also expressed in vivo.
Footnotes
Acknowledgements
The authors thank Drs. Tomoya Akama and Nagako Kawashima for useful discussion, Dr. Hiroto Kawashima for providing S2 monoclonal antibody, Mses. Kayo Suzuki and Yasuyo Shimojo for technical assistance, and Dr. Elise Lamar for critical reading of the manuscript. Part of the work was presented as a poster at the Annual Meeting of the Society for Glycobiology, held in Seattle, Washington, November 9–12, 2011.
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The authors disclosed receipt of the following financial support for research and/or authorship of this article: This work was supported by Grant-in-Aid for Young Scientists 20790278, 22790343 (both to MK), 23790426 (to HH), and 22790303 to (KU) from the Ministry of Education, Culture, Sports, Science and Technology of Japan; Grant-in-Aid for Scientific Research 21390104 from the Japan Society for the Promotion of Science (to JN); and Grant PO1 CA71932 from the National Institutes of Health (to MFukuda).
