Abstract
Kidney fibrosis is the common histological end-point of progressive, chronic kidney diseases (CKDs) regardless of the underlying etiology. The hallmark of renal fibrosis, similar to all other organs, is pathological deposition of extracellular matrix (ECM). Renal ECM is a complex network of collagens, elastin, and several glycoproteins and proteoglycans forming basal membranes and interstitial space. Several ECM functions beyond providing a scaffold and organ stability are being increasingly recognized, for example, in inflammation. ECM composition is determined by the function of each of the histological compartments of the kidney, that is, glomeruli, tubulo-interstitium, and vessels. Renal ECM is a dynamic structure undergoing remodeling, particularly during fibrosis. From a clinical perspective, ECM proteins are directly involved in several rare renal diseases and indirectly in CKD progression during renal fibrosis. ECM proteins could serve as specific non-invasive biomarkers of fibrosis and scaffolds in regenerative medicine. The gold standard and currently only specific means to measure renal fibrosis is renal biopsy, but new diagnostic approaches are appearing. Here, we discuss the localization, function, and remodeling of major renal ECM components in healthy and diseased, fibrotic kidneys and the potential use of ECM in diagnostics of renal fibrosis and in tissue engineering.
Keywords
Introduction
Prevalence of chronic kidney disease (CKD) has reached epidemic proportions, with estimates of prevalence ranging between 11% and 13% of world population. 1 CKD is associated with a significantly higher mortality compared to the general population. 2 The common histological correlate and end-point of CKD is renal fibrosis, which is defined as an excessive, pathological accumulation of extracellular matrix (ECM). The kidney has a complex structure, with three histologically distinct compartments: glomeruli, tubulo-interstitium, and vasculature, each possessing different types of basal-membrane and non-basal membrane (interstitial) ECMs, all of which can be affected by fibrosis (Figs. 1–3).
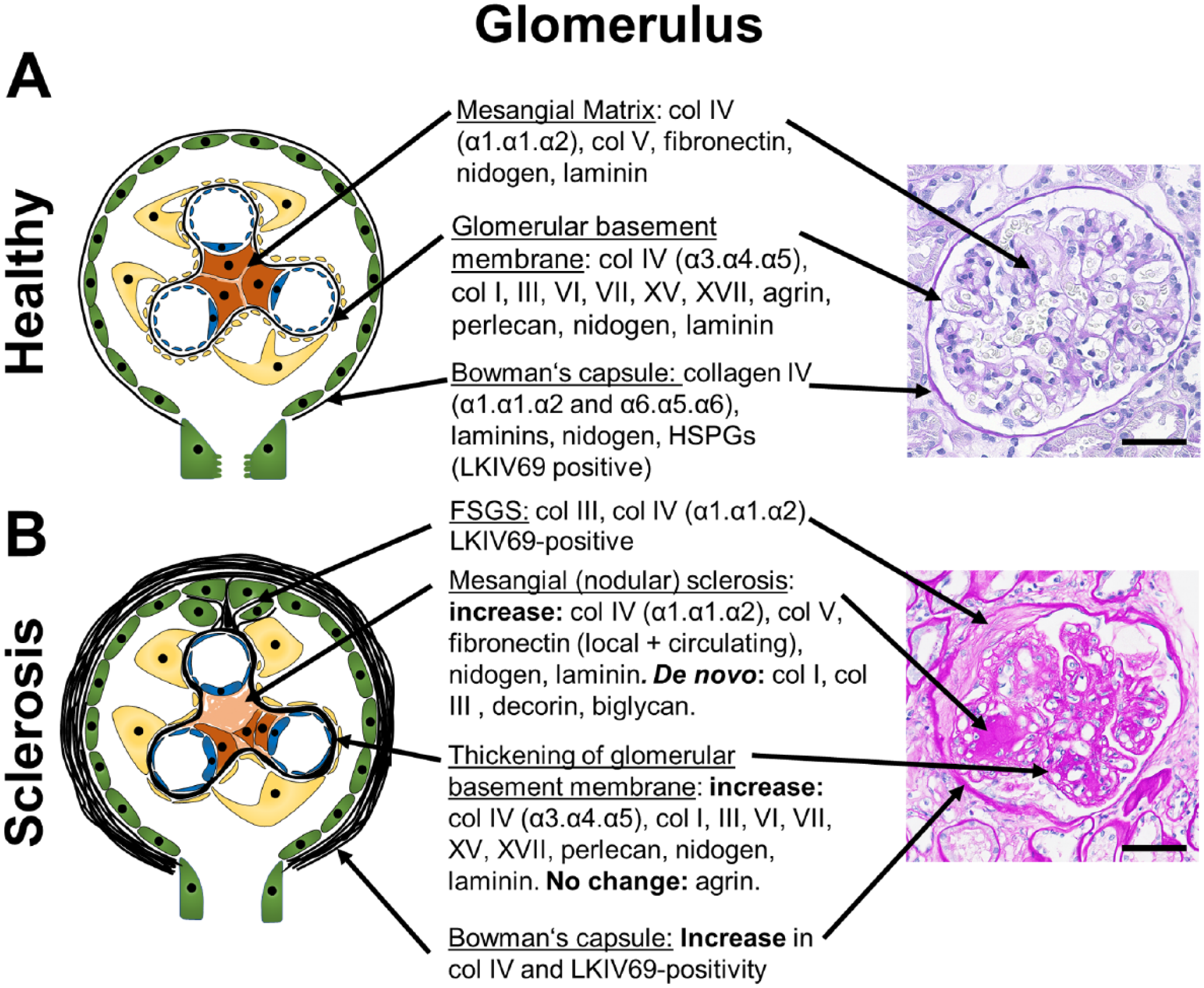
Glomerular ECM components and changes during glomerulosclerosis. A schematic of a healthy glomerulus (A) and a glomerulus with nodular or focal segmental glomerulosclerosis (B) with description of the major ECM-components and their alterations in fibrosis in the mesangial ECM, the glomerular basement membrane and the Bowman’s capsule are shown. Apart from increased Collagen IV- and LKIV69-positivity, little is known on the exact composition of the Bowman’s capsule and the ECM forming FSGS lesions, albeit the ECM of FSGS is likely similar to the ECM of Bowman’s capsule, as it is mainly produced by the parietal epithelial cells. The right panel shows representative PAS stained human sections. (B): PAS-Stain was provided by Dr. Cannata-Ortiz. Scale bars indicate 50 µm. Abbreviations: BC, Bowman’s capsule; PAS, periodic acid-Schiff; FSGS, focal segmental glomerulosclerosis; ECM, extracellular matrix; HSPGs, heparan sulfate proteoglycans.
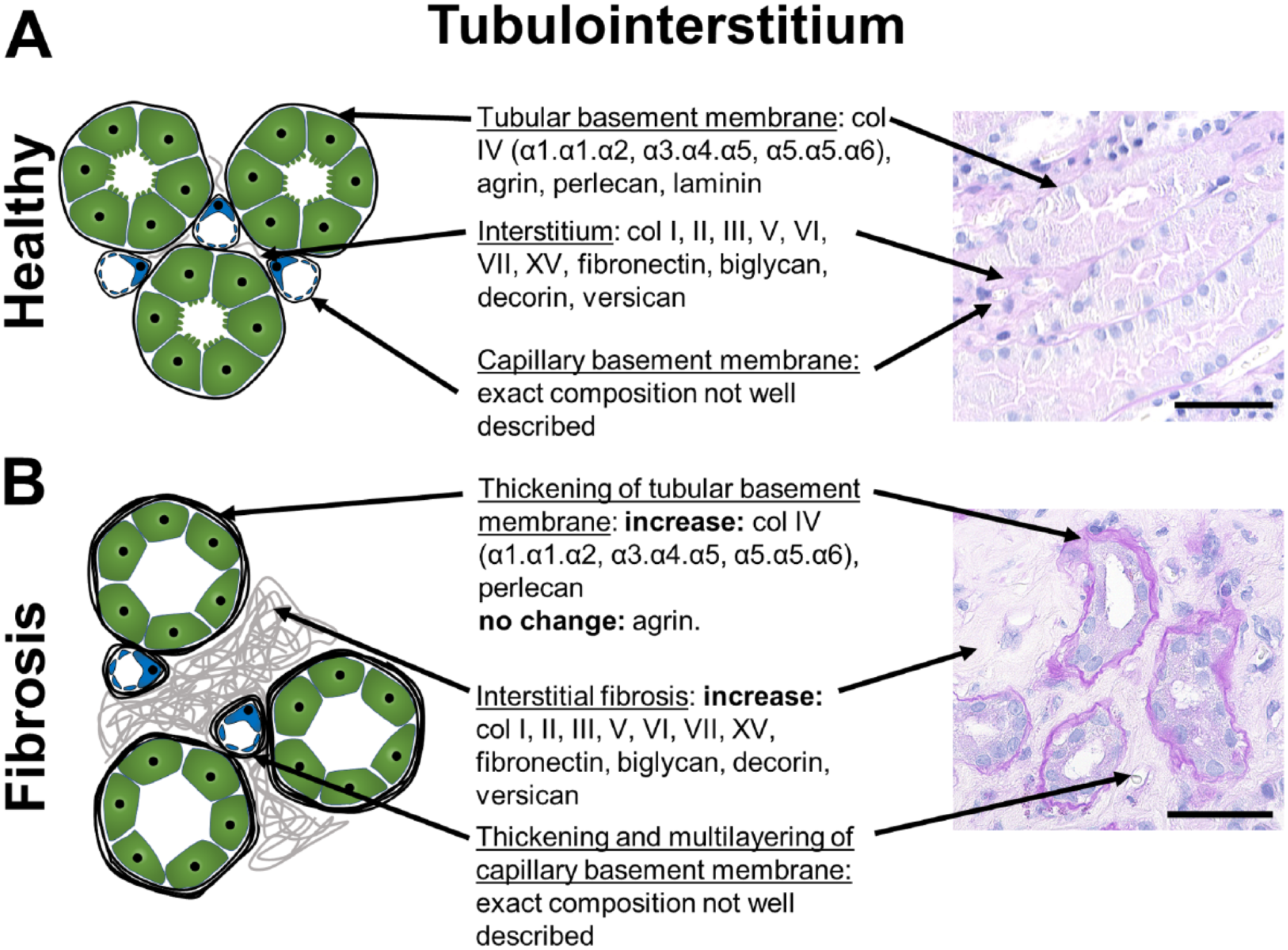
Tubulo-interstitial ECM components and changes during fibrosis. A schematic of a healthy (A) and fibrotic tubulo-interstitium (B) with description of the major ECM-components and their alterations in fibrosis in the interstitial ECM, the tubular and the peritubular capillary basement membranes are shown. The exact composition of the basement membrane of peritubular capillaries has not been investigated. Alterations of the basement membrane of peritubular capillaries are best appreciated by electron microscopy. The right panel shows representative PAS stained human sections. Scale bars indicate 50µm. Abbreviation: ECM, extracellular matrix; PAS, periodic acid-Schiff.
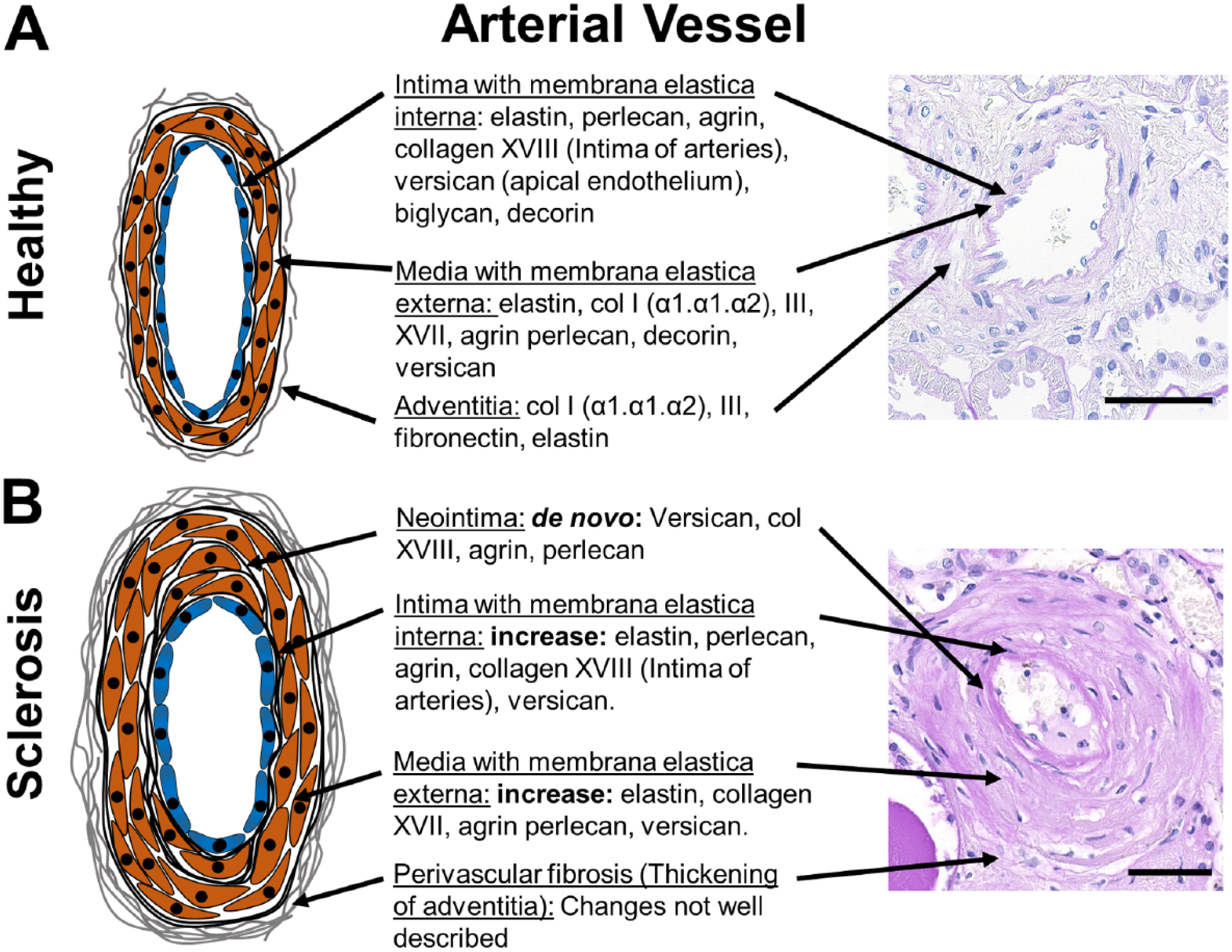
Vascular ECM components and changes during arteriosclerosis. A schematic of a healthy (A) and sclerotic renal arterial vessels (B) with description of the major ECM-components and their alterations in arteriosclerosis in the intima/neointima, media and adventitia are shown. Little is known on specific changes of adventitia in renal fibrosis. The right panel shows representative PAS stained human sections. Scale bars indicate 50µm. Abbreviation: ECM, extracellular matrix; PAS, periodic acid-Schiff.
ECM in the glomerular basement membrane (GBM) mainly consists of collagen type IV (α3.α4.α5 heterotrimers), nidogen, laminins (mainly α5β2γ1), and heparan sulfate proteoglycans (HSPGs) (agrin, perlecan). Glomerular mesangial ECM mainly consists of collagen type IV (α1.α1.α2 heterotrimers), collagen type V, fibronectin, laminins, and HSPGs. The basement membrane of the Bowman’s capsule is composed of collagen IV (α1.α1.α2 and α5.α5.α6 heterotrimers), laminins, nidogen, and HSPGs 3 (Fig. 1). A proteomic study of glomerular ECM, that is, of both mesangium and GBM, revealed 144 structural components. 4 The most abundantly detected proteins were collagen IV with α1.α1.α2- and α3.α4.α5-heterotrimers, collagen I, laminin α5β2γ1, and HSPGs.
The ECM of the tubulo-interstitial compartment consists of basal membranes of peritubular capillaries and tubules, and of the interstitial space (Fig. 2). The latter is most commonly, and often exclusively, analyzed in studies of renal fibrosis. Interstitial ECM mainly consists of different collagens (types I, III, IV, V, VI, VII, and VIII), glycosaminoglycans (e.g., hyaluronan), polysaccharides, and glycoproteins (e.g., fibronectin, versican, biglycan, decorin). Renal medulla physiologically has a higher content of interstitial ECM compared to cortex. It is not yet clear whether there are major differences in the composition of cortical versus medullary interstitial ECM. Very little is known about the composition of the basement membrane of the peritubular capillaries.
Renal arteries consist of three compartments, the intima with subendothelial basal membrane and subendothelial interstitial ECM, media with smooth muscle cells and predominantly elastic fibers, and adventitia with interstitial type of collagen rich ECM. A comprehensive analysis of vascular ECM in the kidney is largely missing, although it is likely similar to vessels in other organs, reviewed elsewhere 5 (Fig. 3).
In renal pathology diagnostics, specific terms are used to describe fibrotic changes in each of the compartments, that is, glomerulosclerosis, which can be either nodular or focal segmental, in the glomeruli, interstitial fibrosis in the tubulo-interstitium and arteriosclerosis (and perivascular fibrosis) in the vessels. No specific terms exist for thickening and fibrosis of basement membranes, although they can be observed as an isolated change, for example, in diabetic nephropathy, where thickening of the GBM is one the earliest pathological signs of the disease. In the tubules, thickened basement membranes are linked with the term tubular atrophy rather than interstitial fibrosis, albeit both processes most often go hand-in-hand. Thickening and multi-layering of peritubular capillary basement membranes is less well described, most likely due to methodological issues requiring high-resolution or electron microscopy for its assessment. Such changes were recognized in chronic antibody-mediated rejection and in a lesser extent seem to be also a common finding in renal fibrosis in animals and humans. 6 Arteriosclerosis, that is, thickening of the intima is a common finding in renal fibrosis and is especially linked to hypertension. 7 However, it can also be found in healthy individuals without hypertension and is linked to aging and systemic atherosclerosis. 8 Little is known about perivascular (adventitial) fibrosis, although it is a relatively common morphological finding in renal fibrosis.
The main cells producing mesangial and interstitial ECM are mesangial cells in the glomeruli, and fibroblasts, myofibroblasts, and pericytes in the tubulo-interstitium. On the other hand, ECM of the basement membranes is mainly produced by the cells lining them, that is, podocytes and endothelial cells in the GBM, parietal epithelial cells in the Bowman’s capsule, tubular epithelial cells and endothelial cells in the tubular and peritubular capillary basement membranes, respectively. These respective cells are also the most important contributors to fibrosis of the respective ECM. For example, in focal segmental sclerosis (FSGS), parietal epithelial cells are the major ECM producing cells, and ECM of FSGS-lesions is similar to Bowman’s capsule.9–11 A detailed discussion of cellular and molecular mechanisms of renal fibrosis can be found elsewhere.12–14
Components of the Renal ECM
Collagens
Collagens are the most abundant components of the ECM, constituting approximately 30% of total body protein. 15 There are 54 collagen-genes in humans. 16 Each gene encodes a monomer collagen chain, termed alpha-chain, 17 which assemble into homo- or heterotrimers, forming 28 collagen types. For example, collagen type IV has six alpha-chains forming three different trimer combinations, that is, α1.α1.α2, α3.α4.α5 and α5.α5.α6.
The GBM is formed by the collagen type IV α1.α1.α2-heterotrimer during embryonic development, followed by a shift to the α3.α4.α5-heterotrimer in adult kidneys. 18 Collagen IV α3.α4.α5 heterotrimer is essential for glomerular function. This is well documented by rare diseases, such as Alport syndrome, a hereditary disorder characterized by mutations in the COL4A3, COL4A4 or COL4A5 genes19,20 and anti-GBM glomerulonephritis or Goodpasture Syndrome, caused by autoantibodies against collagen IV (Table 1).21,22 Proteomic analysis of the renal GBM identified the presence of collagens I, III, VI, XII, and XV. 4 Another collagen expressed in the GBM is collagen XVII, a transmembrane protein, which is considered important for glomerular maturation. 23
Renal Diseases With Primary Involvement of ECM Components.
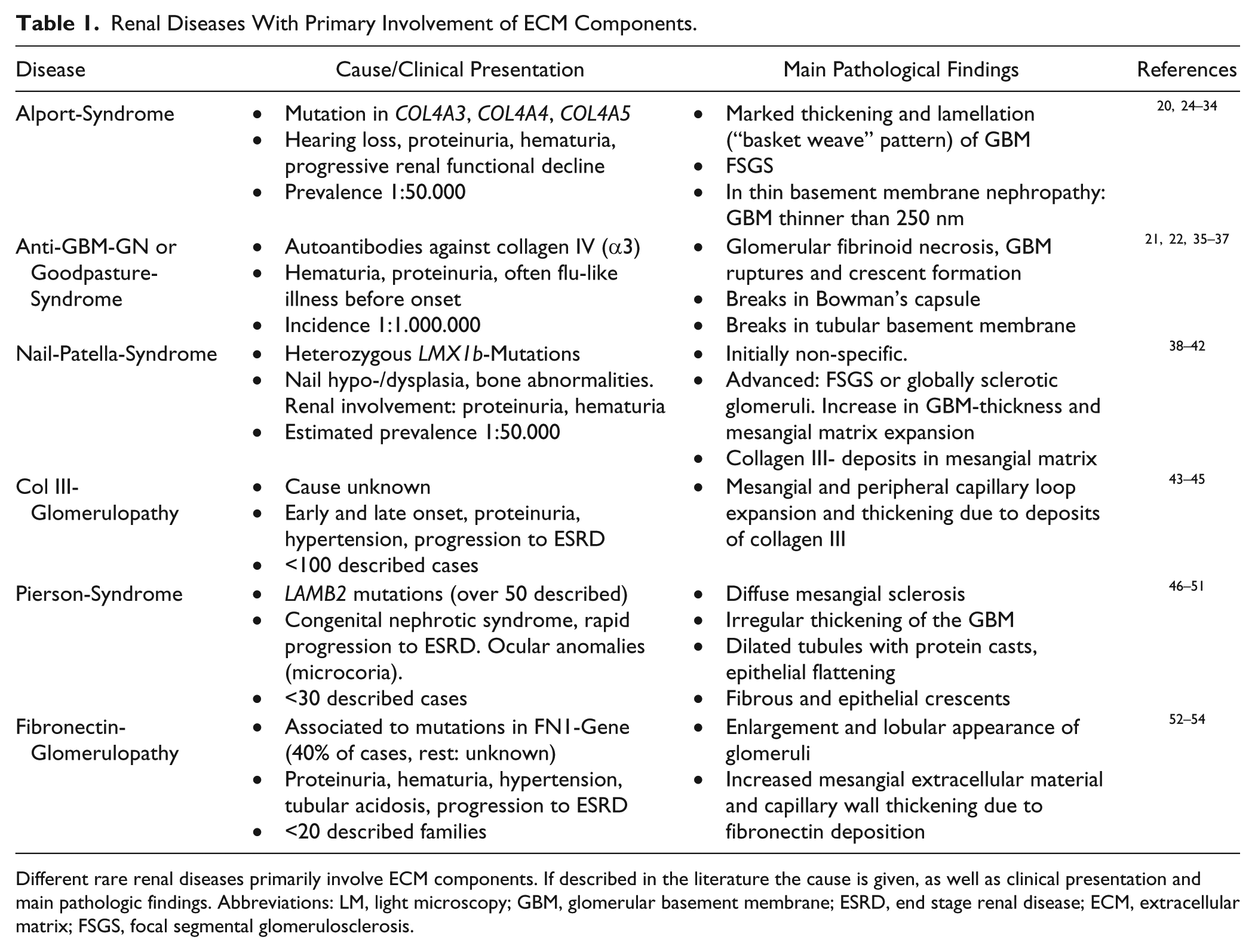
Different rare renal diseases primarily involve ECM components. If described in the literature the cause is given, as well as clinical presentation and main pathologic findings. Abbreviations: LM, light microscopy; GBM, glomerular basement membrane; ESRD, end stage renal disease; ECM, extracellular matrix; FSGS, focal segmental glomerulosclerosis.
Pathological deposition of collagen III is found in both mesangial nodular and focal segmental glomerulosclerosis,55,56 but also in specific rare diseases, that is, collagen type III glomerulopathy and Nail-Patella Syndrome38,57 (Table 1).
Collagens I, II, III, V, VI, VII, and XV are expressed in the renal interstitium and increased deposition was found during renal fibrosis. 58 Upregulation of collagen I and III is considered an early event in renal fibrosis.55,58,59 Collagen I is widely accepted as a major component of fibrotic tissues. A study using a cell-type specific knock-out of collagen type I provided first evidence for direct functional involvement of collagen in kidney diseases. The effects were dependent on disease context, that is, during reversion of early obstructive nephropathy, collagen I deficiency resulted in reduced renal functional recovery, whereas in a model of crystal-induced CKD and fibrosis, collagen I deposition was harmful and disease aggravating. 60
Glycoproteins and Proteoglycans
Glycoproteins are a large group of proteins with covalently attached oligosaccharide chains, and proteoglycans are glycosaminoglycans that are covalently bound to a protein core. 61
Fibronectin is an adhesive high molecular weight glycoprotein that plays a crucial role in wound-healing and ECM formation. 62 Although only one fibronectin-gene exists, there are multiple different fibronectin proteins generated through alternative splicing. 63 Fibronectin is found in the glomerular mesangium, Bowman’s capsule and tubulo-interstitium and its expression increased after injury during fibrosis64–66 (Figs. 1, 2 and 4). In addition to locally produced fibronectin, it was shown that circulating fibronectin can be incorporated into mesangial ECM during diabetes, thereby contributing to mesangial expansion. 67 Fibronectin is a major component of glomerular extracapillary proliferates (crescents). 68 Approximately 40% of patients with mutations in the FN1-gene develop fibronectin glomerulopathy, a very rare disease caused by pathological deposition of fibronectin in the mesangium and capillary walls of the glomeruli.52,53 Little is known about fibronectin in renal vessels: an increased expression in acute diffuse proliferative glomerulonephritis and malignant hypertension 65 and in renal cell carcinomas was described. 69
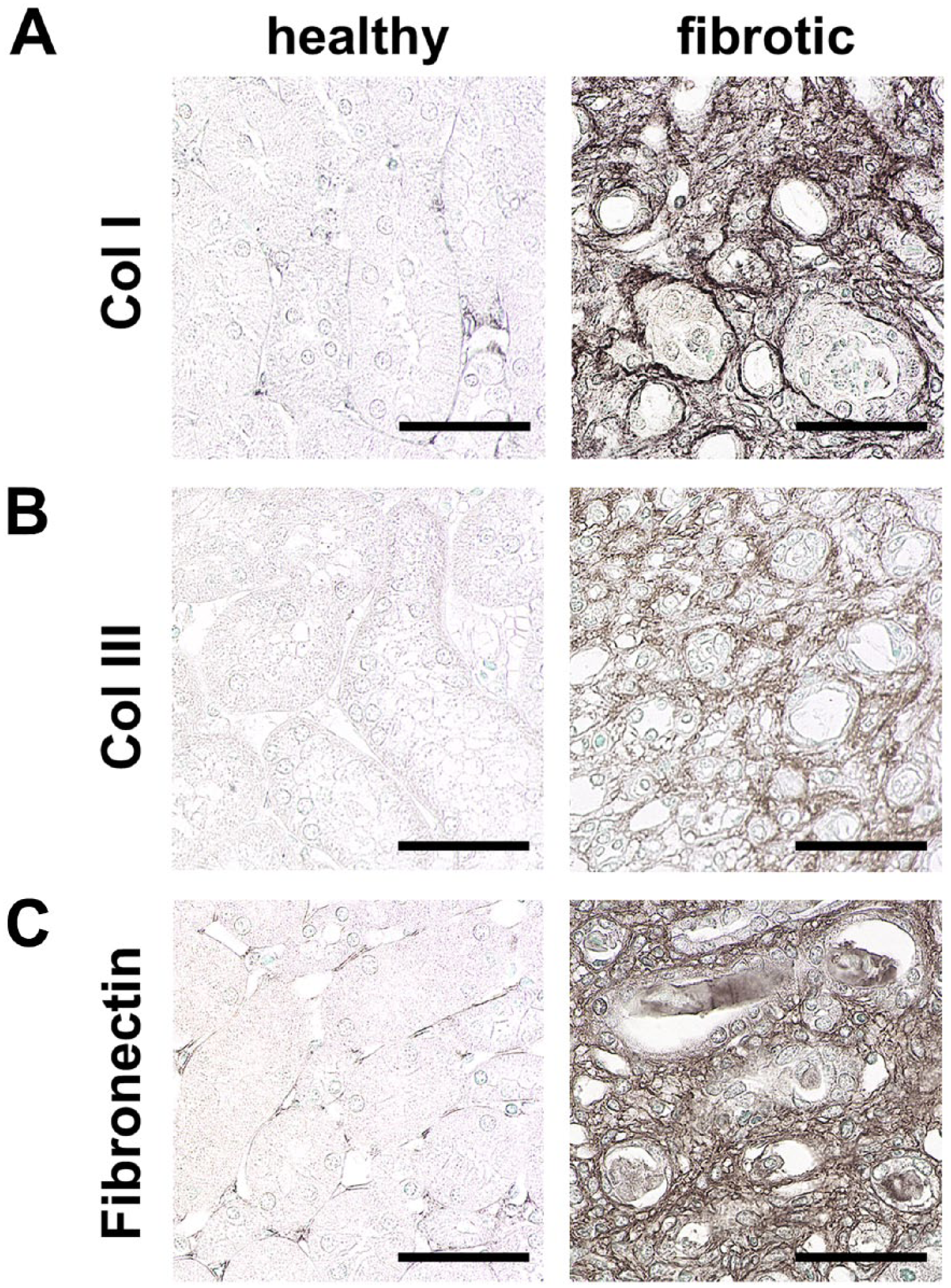
Hallmarks of renal interstitial fibrosis. Immunohistochemical staining of ECM components shows prominent upregulation of collagen I (A), collagen III (B), and fibronectin (C) in interstitial fibrosis compared to healthy mice. Scale bars indicate 50 µm. Abbreviation: ECM, extracellular matrix.
HSPGs are glycoproteins that contain one or more covalently attached heparan sulfate chains. 70 They can be divided into three different groups: membrane HSPG (e.g., glypicans), secreted ECM HSPGs (e.g., agrin, perlecan), and the secretory vesicle proteoglycan serglycin. In the healthy kidney, agrin is the major HSPG of the GBM, while perlecan is less abundant. In the mesangial matrix of healthy kidneys, both agrin and perlecan are expressed. 71 Both agrin and perlecan contribute to the electric charge of the GBM, but seem not to be important for glomerular filtration. 72 In a rat model of chronic transplant dysfunction, perlecan was markedly induced in the GBM and the mesangial matrix. 71 In IgA-nephropathy, perlecan was significantly upregulated and high expression correlated with a better outcome of patients. 73 Both perlecan and agrin are expressed in tubular basement membranes, and perlecan is also expressed in basement membranes of peritubular capillaries. In healthy and diseased kidneys, perlecan and agrin are not expressed in interstitium, but in vessel walls and neointima in arteriosclerosis. 71
Laminins, which are a group of heterotrimeric glycoproteins, are one of the main constituents of basement membranes and are involved in cell and tissue differentiation, homeostasis, and survival. Laminins consist of an alpha, a beta, and a gamma chain. 74 There are five alpha, four beta, and three gamma chains. 75 In total, 16 different combinations of these chains have been identified in mammals, for example, laminin α1β1γ1. 76 Laminins are a major constituent of the GBM, 77 mesangial ECM 58 and tubular basement membranes. 78 Similar to the change in the collagen IV network, there is a shift in laminin isoform expression during renal development in the GBM. Laminin α5β2γ1 is expressed in the mature GBM, while Laminin α1β1γ1 is expressed during development. 79 Pierson syndrome, a rare genetic disorder is caused by a mutation in the LAMB2 gene encoding the laminin β2-chain 46 (Table 1). A podocyte-specific knockout of laminin β2 led to severe proteinuria in mice. 80 Introduction of a Lamb2 gene with C321R missense mutation leading to Pierson Syndrome in these Lamb2-knockout mice initially reduced proteinuria, but eventually led to progressive disease with development of end-stage renal disease (ESRD). 80 Increased deposition of laminins in the mesangial matrix and GBM, but not in the interstitium, has been shown in diabetic nephropathy. 81 There is no comprehensive description of laminins in tubular basement membranes in renal fibrosis.
Nidogens are monomeric glycoproteins expressed in basal membranes, important for normal development of some organs like heart or lung. 82 In the kidney, they are expressed in the GBM and tubular basement membranes but also in the mesangial ECM.58,83 The functional role of nidogen is not yet clear as global knockouts of nidogen 1 or nidogen 2 showed no kidney phenotype with normally developed GBM.84,85 Upregulation of nidogens in the mesangial matrix and the GBM was described in patients with glomerular diseases, that is, diabetic nephropathy, IgA-nephropathy, and lupus nephritis. 86
Versican is a large chondroitin sulfate proteoglycan considered an anti-adhesion molecule with multiple binding partners, for example, hyaluronan, collagen I, and fibronectin.87–89 In the healthy kidney, versican is expressed in the tubulo-interstitium and in intima and media of vessels, 90 but not in glomeruli. 71 In kidney diseases, both in animal models and patients, versican was found to be upregulated in interstitium during fibrosis 91 and it was found to be expressed de novo in glomerular crescents. 92 During arteriosclerosis, it is induced in the neointima. 92 High versican-expression is associated with worsening of CKD. 91
Decorin and biglycan belong to the large group of small leucine-rich proteoglycans that are either bound to the ECM or released as soluble mediators.93–96 In healthy kidneys, they are mainly expressed in the interstitium 97 but not in glomeruli. 98 Both biglycan and decorin showed de novo expression in glomeruli and elevated expression in the interstitium in various renal diseases and fibrosis in animal models and patients.92,98 In healthy renal arterial vessels, decorin and biglycan are expressed throughout all layers. 97 Both ECM-bound decorin and biglycan interact with collagens and regulate ECM-assembly. 95 After proteolytic cleavage, both biglycan and decorin can act as soluble mediators, 99 interact with toll-like-receptors and are considered ECM-derived promotors of inflammatory response. 100 Decorin was shown to exert antifibrotic functions in the kidney by blocking transforming growth factor β (TGF-β), a key protein in pro-fibrotic signaling. 101 Biglycan can also interact with TGF-β, 102 however, no effects of biglycan-treatment were found in pulmonary fibrosis. 103 Biglycan was recently found to induce autophagy in macrophages via TLR4 and CD44, thereby limiting inflammation and ischemia-reperfusion induced kidney injury in mice. 104
ECM Remodelling
All types of ECM undergo constant remodeling, with an equilibrium between degradation and production in healthy state. 105 In the kidney, the exact analyses of the half-life of the various ECM components is largely missing. In fibrosis, the equilibrium between ECM synthesis and breakdown is disturbed with increased production and reduced degradation leading to excessive ECM deposition. Degradation of ECM is mainly done by matrix-metalloproteinases (MMPs), which are counteracted by the tissue-inhibitors of metalloproteinases (TIMPs), both of which can be dysregulated in fibrosis.
MMPs are a group of zinc-dependent endopeptidases that can degrade various ECM components. 105 They are considered as one of the main enzymes responsible for ECM remodeling.106,107 Of the 28 MMPs described in vertebrae, 23 are expressed in humans. 108 At least 10 MMPs are expressed in the kidney (MMP-1, -2, -3, -9, -13, -14, -24, -25, -27, -28).58,109 MMPs can be divided into different groups based on their substrates and structure: the collagenases (MMP-1, -8, -13, -18),108,110 the gelatinases (MMP-2, -9), the stromelysins (MMP-3, -10, -11), the matrilysins (MMP-7, -26), the membrane-type MMPs (MMP-14, -15, -16, -17, -24, -25), and other MMPs (MMP-12, -19-23, -27, -28). 108 Almost all of them cleave both collagen- and non-collagen-substrates. 108 Their ability to degrade ECM suggested a beneficial role in renal fibrosis. However, effects of MMPs are far more complex and different MMPs were shown to play divergent roles in fibrogenesis. For example, high MMP-2-expression in mesangial cells led to increased collagen production. 111 MMP-9 produced collagen fragments that are chemotactic for neutrophils and stimulate MMP9 production in these cells. 112 Conversely, MMP-7 knockout mice showed reduced fibrosis in unilateral ureteral obstruction (UUO). 113
Three of the four TIMPs, that is, TIMP-1, TIMP-2 and most strongly TIMP-3, are expressed in the kidney.109,114 Deficiency of TIMP-1 in mice undergoing UUO did not reduce tubulointerstitial fibrosis. 115 A knockout of TIMP-2 did not show increased collagen I and III deposition following renal injury, and these mice had reduced MMP-2 activity. 116 However, TIMP-3 knockout mice showed increased renal fibrosis and more collagen I and III deposition compared to wildtype controls in UUO. 114 MMP-2 activation was also elevated in TIMP-3 knockout mice. 116
Other proteases involved in ECM remodeling are the a-disintegrin-and-metalloproteinases with thrombospondin motifs (ADAMTS). ADAMTS have a variety of substrates including versican and fibrillar procollagens. 117 Little is known on the potential role of ADAMTS in renal fibrosis, despite their ability to degrade a variety of matrix proteins. Interestingly, ADAMTS-1-knockout mice developed dilated calyces and renal interstitial fibrosis after birth. 118 Recently, it was shown, that ADAMTS-1, -12 and -15 are increasingly expressed in the Adriamycin model of renal fibrosis. 119
During fibrosis, ECM is not only changed in a quantitative but also qualitative manner. For example, tissue-transglutaminase 2 can cross-link various proteins, including collagens and fibronectin. 120 This cross-linking of ECM-proteins leads to increased stability of ECM and resistance to proteolytic degradation. Tissue-transglutaminase 2 is upregulated in experimental models of renal fibrosis 121 and tissue-transglutaminase 2 knockout mice develop less interstitial fibrosis. 122 In diabetes, advanced glycation end-products were shown to increase stiffness of collagens by crosslinking, which also reduces collagen-digestibility and masks integrin-binding sites.123,124
Cell-ECM Interactions
Interactions between ECM and cells are mediated by ECM-binding transmembrane receptors, such as integrins. Integrins provide a mechanical link between the ECM and intracellular actin- and intermediate filaments of the cytoskeleton. 125 They were shown to modulate cell survival, migration, proliferation, and tissue homeostasis, providing a communication hub between the ECM and cells. 126 Integrins are transmembrane glycoproteins with 18 α- and 8 β-subunits, forming 24 heterodimers 127 and bind to a wide variety of ligands including ECM components like collagens, laminins, and fibronectin. Binding of extracellular ligands to integrins induces intracellular signaling via intracellular adaptor proteins, enabling cell-matrix signal transduction. 128 In the glomerulus, integrin α3β1, a laminin receptor, was shown to be expressed on podocytes 128 and integrin α1β1, a collagen receptor, on the mesangial cells. 129 In early stage diabetic nephropathy, α3β1-integrin was upregulated in podocytes, but was downregulated in later stages. 130 Mice with a podocyte-specific knockout of the integrin α3 subunit showed podocyte foot process effacement. 131 Knock-out of the α1β1-integrin in Alport mice led to reduced proteinuria and significantly prolonged life-span of these mice. 132
α6β1-integrin is expressed in renal tubular cells.133–135 Deletion of the α6-subunit in collecting ducts lead to a higher vulnerability of epithelial cells to obstructive nephropathy. 78 In a mouse model of polycystic kidney disease, knock-out of the β1-subunit reduced cyst formation. 136 Several integrin subunits were found in renal vascular endothelial cells, for example, α1, β2, β3. 137 A knockout of the α8-subunit, which is expressed in vascular smooth muscle cells and mesangial cells in the kidney, lead to a widening of the lumen of peritubular capillaries. 138 Integrins containing the αv-subunit, which act as receptors for fibronectin and laminin, were found to be key modulators in organ fibrosis.139,140 A knockout of the αv-integrin-subunit in myofibroblasts led to less fibrosis in lung, liver, and kidney and a specific inhibitor of αv-integrins effectively reduced fibrosis in liver and lung. 141 In a model of lipopolysaccharide-induced proteinuria in mice activation of the αvβ3-integrin by the urokinase receptor (uPAR) aggravated proteinuria. 142 Activation of β3-integrin via periostin was proposed as an important mechanism driving crescentic glomerulonephritis in mice via influencing podocyte foot process effacement, increased cell motility, and survival. 143
Syndecans are transmembrane HSPGs, attached to the actin cytoskeleton. 144 Both heparan sulfate and chondroitin sulfate chains can be bound to syndecans. As receptors, they can bind several ECM-proteins, including collagens and fibronectin. 145 In the kidney, syndecan-1, -2, and -4 were shown to be expressed. 146 A knockout of syndecan-4 reduced renal fibrosis by reducing transglutaminase 2 mediated ECM cross-linking. 147 Discoidin domain receptors (DDRs) are receptor tyrosine kinases that mainly bind to collagens and are generally considered profibrotic in glomerular and interstitial kidney diseases. In healthy kidneys, they are expressed mainly in the tubules. DDR1 is expressed de novo in glomerular injury in rats, 148 and DDR1-knockout mice showed less fibrosis and less proteinuria in a model of hypertension 149 and in Alport mice. 150 Dystroglycan is another transmembrane receptor connecting extracellular glycoproteins, for example, laminin and agrin, to the cytoskeleton. This complex is called the dystroglycan glycoprotein complex 151 and is expressed in podocytes and tubular epithelial cells in the kidney.152,153 However, both ubiquitous and a kidney cell-specific knockout of dystroglycan had no significant impact neither in healthy nor diseased kidneys. 154
Renal ECM Components—Biomarker Potential?
The extent of renal fibrosis is currently the best predictor of progressive functional decline of kidney diseases. 12 Increased deposition of ECM is the core hallmark and definition of fibrosis, and therefore ECM components can be viewed as the only truly specific fibrosis markers. Histological assessment of renal fibrosis on kidney biopsy is currently the only specific method for fibrosis quantification.
Renal interstitial fibrosis can appear in various morphological patterns. In diseases which affect renal parenchyma in a diffuse manner, a diffuse interstitial fibrosis is observed. In diseases with focal injury, for example, in many glomerular diseases, fibrosis develops in a patchy manner. A striped appearance of fibrosis was associated to calcineurin-inhibitor use 155 and broad fibrotic scars of interstitial fibrosis are observed after pyelonephritis or anemic infarction, similar to scars after myocardial infarction. 156 Given the focal nature of fibrosis, sampling bias could be a confounder when measuring fibrosis in biopsies. However, there are no studies that would systematically and comprehensively analyze such a bias. On the contrary, one study comparing two biopsy cores from various kidney locations showed no significant sampling error, suggesting that such a bias might not be as relevant as is often anticipated. 157
In renal biopsy diagnostic, the degree of interstitial fibrosis is assessed on routinely performed stains, preferentially on special stains for ECM, such as the Sirius red or Masson’s trichrome stain. Immunohistochemistry or immunofluorescence can be used for a more specific assessment of particular ECM components (Fig. 4), but this is currently not used in routine diagnostics. Given the predictive value of interstitial fibrosis, the degree of interstitial fibrosis is included as an integral part in the renal biopsy pathology report. The extent of interstitial fibrosis is most common assessed by renal pathologists in a semiquantitative manner using scoring systems or an estimate of the fibrotic area, which might have low reproducibility.158–160 Digitalization of pathological slides, that is, virtual microscopy, opens the possibility of automated, more exact and more reproducible quantification of fibrosis.161–163 Various alternative methods were shown to be useful for analysis of interstitial fibrosis. For example, a combination of Fluorescence Lifetime Imaging (FLIM) with Second Harmonic Generation (SHG) allowing a label-free, deep tissue imaging of a biopsy reproducibly detected fibrosis. 164 However, this technique requires sophisticated microscopy equipment, which is not freely available in routine pathology labs. Combining such novel microscopy techniques with tissue clearing, 165 opens exciting possibilities of 3D assessment of kidney morphology and fibrosis in research, 166 but is currently not useful or applicable for routine pathology diagnostics.
However, the invasive nature and the potential sampling bias are some of the disadvantages rendering renal biopsies not perfectly suitable for fibrosis monitoring. An ideal biomarker of renal fibrosis should be non-invasive and closely and specifically reflect kidney fibrosis. Several studies analyzed collagens and their production and degradation fragments as non-invasive biomarkers of renal fibrosis. N-terminal propeptide of type III collagen (PIIINP) is a fragment generated during production of collagen III. It was elevated in urine and serum of patients with different renal diseases and correlated with increased collagen III in biopsies. 55 The ratio of urinary PIIINP to creatinine showed a positive correlation with renal function parameters, increased with CKD stage and correlated with the extent of interstitial fibrosis. 167 Elevated urinary levels of PIIINP were associated with CKD-progression in elderly patients. 168
MMP-mediated collagen I and III degradation fragments, C1M and C3M, were shown to be a potential biomarker in animal models of fibrosis169,170 and acute kidney injury. 171 In two cohorts of IgA-nephropathy patients, collagen III-turnover suggested a profibrotic ECM remodeling with increasing CKD stage, with steadily increasing Pro-C3 as a marker of collagen III production and progressively decreasing C3M as a marker of collagen III degradation. 172 Similarly, renal transplant recipients with high CKD stage exhibited low urinary levels of C3M. 173 In a large study analyzing the urinary proteome of patients with and without CKD, fragments of collagen I and III were shown to have a strong positive correlation to progression in CKD. 174
High urinary collagen type IV was associated with lower renal function in a cohort of Japanese patients with type 2 diabetes. 175 In a long-term follow-up of patients with type 1 diabetes, it was possible to predict microalbuminuria by higher urinary collagen type IV. 176 In a Japanese non-diabetic population, a high urinary collagen IV to creatinine ratio was associated with an increased risk of 10% decline of eGFR per year. 177 Although collagen IV is increased in renal fibrosis, urinary and circulating collagen IV did not correlate with fibrosis in kidney biopsies in a cohort of patients with different diseases, that is, membranous glomerulonephritis, mesangioproliferative glomerulonephritis, tubulointerstitial nephritis, and ESRD. 55 However, high serum amounts of a fragment of collagen IV termed P4NP_7S were shown to be associated with worse survival in hemodialysis patients. 178
Endotrophin is a C-terminal fragment of collagen VI that is generated during production of collagen VI. 179 High serum endotrophin was associated with increased risk for developing ESRD and a higher mortality in a group of CKD patients. 180 Endotrophin was expressed de novo in interstitial fibrosis in patients and high urinary endotrophin was associated with a higher rate of one-year-progression to ESRD in CKD patients. 181
Increased serum and urinary LG1M, a specific neoepitope generated by cleavage of the laminin γ1 chain by MMP-9, was associated with a higher rate of development of end stage renal disease and with mortality. 182
Given the excretory function of the kidney and the fact that most, if not all ECM components are not kidney-specific, the specificity of urinary and circulating biomarkers in detecting and monitoring kidney fibrosis is limited. Non-invasive imaging of ECM could circumvent this problem and showed some promising results in other organs, particularly the liver. 183 A number of imaging techniques, in particular using magnetic resonance imaging (MRI), ultrasonography, and elastography, were evaluated in assessment of renal fibrosis with rather contradictory and not convincing results.14,183–185 Molecular imaging of ECM components might represent a more specific approach to image fibrosis. First promising data showed the feasibility of molecular fibrosis imaging using elastin-specific molecular probes. 186 The above discussed approaches to monitor fibrosis are summarized in Table 2.
Summary of Different Ways to Monitor Renal Fibrosis.
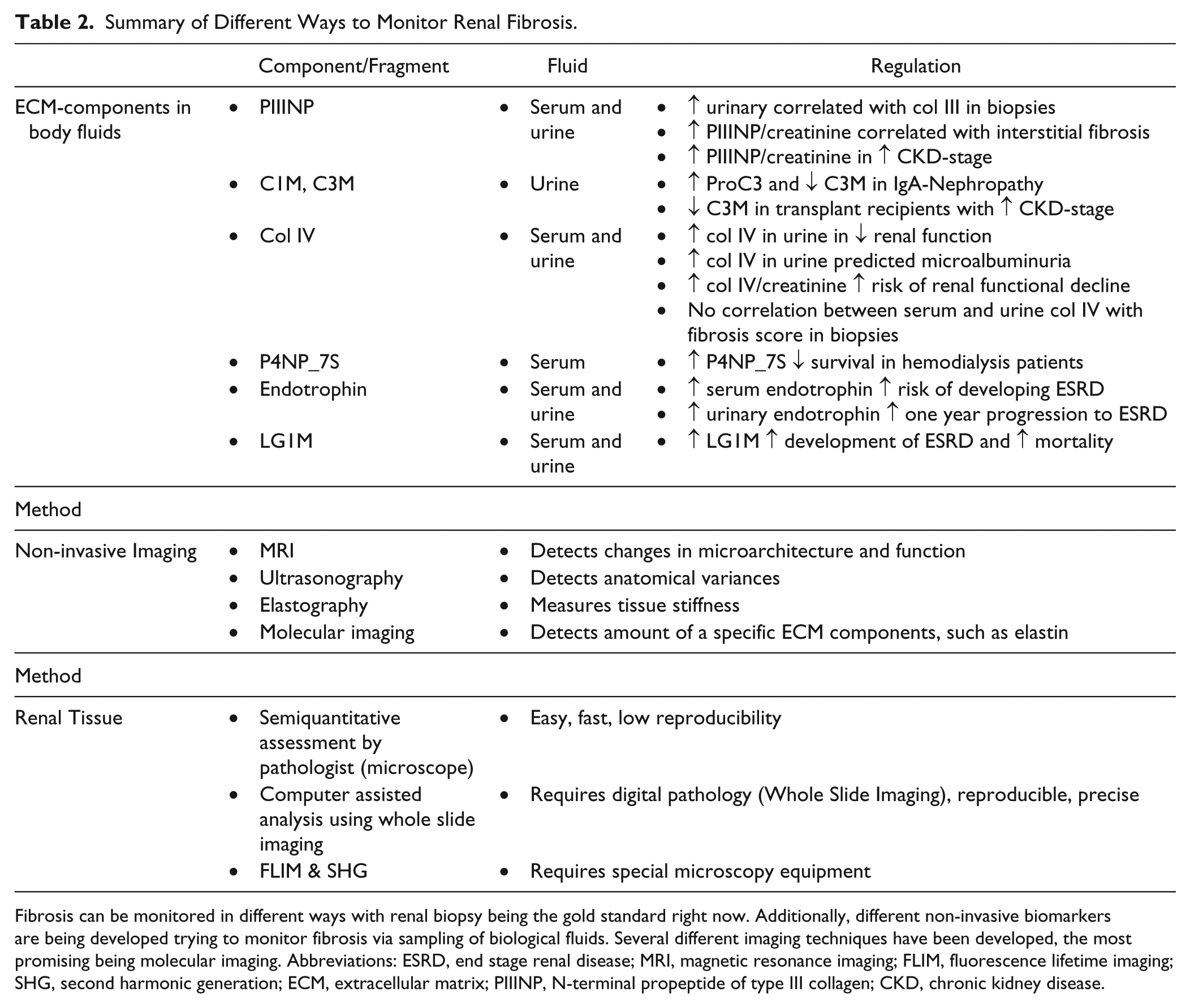
Fibrosis can be monitored in different ways with renal biopsy being the gold standard right now. Additionally, different non-invasive biomarkers are being developed trying to monitor fibrosis via sampling of biological fluids. Several different imaging techniques have been developed, the most promising being molecular imaging. Abbreviations: ESRD, end stage renal disease; MRI, magnetic resonance imaging; FLIM, fluorescence lifetime imaging; SHG, second harmonic generation; ECM, extracellular matrix; PIIINP, N-terminal propeptide of type III collagen; CKD, chronic kidney disease.
Renal ECM in Tissue Engineering
Tissue engineering provides a potential alternative for transplantation. Building up kidneys from patient-own cells is an exciting future perspective. As a first step in this direction, kidney organoids, that is, organ-like 3D structures of organ-specific cells, 187 were successfully generated in vitro using human embryonic stem cells or human-induced pluripotent stem cells (iPSCs).188,189 To date, it is only possible to recapitulate early embryonic development of the kidney, which is still far from generating organs suitable for transplantation. A possible alternative approach is to repopulate a kidney scaffold with iPSCs or more differentiated, kidney-committed precursors. 190 For this, renal ECM itself might be the best suitable scaffold, providing an exact functional and structural environment for repopulating cells.191,192 Decellularization, that is, organ perfusion with compounds destroying cell membranes and leading to cell death (e.g., Triton X-100 or sodium dodecyl sulfate), generates a cell-free ECM-scaffold that can be used for tissue reconstruction of a whole kidney.190,193 This is followed by repopulation of the ECM scaffolds with cells, potentially leading to generation of functional kidneys.193,194 Currently, also this approach is yet far from clinical applicability.
Another concept of tissue engineering is bioprinting, that is, 3D-organ printing from tissue components. 195 In this technique, cellular and non-cellular components are printed in parallel to form a replica of a tissue. Most biomaterials currently used for bioprinting do not replicate the complexity of the ECM. 196 However, decellularized ECM can be solubilized to be used as bio-ink. This solubilized ECM behaves as a pre-gel, being soluble at low temperatures allowing 3D-printing followed by incubation at 37C, generating a gel (a process termed gelation). 196 First examples of 3D-bioprinting of proximal tubules were shown to be suitable for drug testing. 197 However, the kidney is a very complex structure which might pose a substantial hurdle for whole organ 3D bioprinting.
In conclusion, the ECM of the kidney is a complex, dynamic structure essential for normal renal function. This is well documented by several rare diseases leading to progressive and currently non-treatable renal diseases. In addition, pathological ECM deposition is a hallmark of renal fibrosis and thereby involved in virtually all CKDs. Components of the ECM could be exploited as specific biomarkers of renal fibrosis, being potentially powerful progression markers. ECM scaffolds from decellularized kidneys might be crucial in tissue engineering of kidneys.
Footnotes
Acknowledgements
Competing Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
RDB drafted the manuscript and arranged figures and table, PB critically reviewed the manuscript, tables, and figures both for intellectual content and form. Both RDB and PB have read and approved the final version of the manuscript.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was financed by the German Research Foundation (DFG: SFB/TRR57 and SFB/TRR219, BO3755/3-1 and BO3755/6-1), the German Ministry of Education and Research (BMBF: STOP-FSGS-01GM1901A), and the RWTH Interdisciplinary Center for Clinical Research (IZKF: O3-7).
