Abstract
Primary cilia have a pivotal role in bone development and the dysfunctions of primary cilia cause skeletal ciliopathies. Intraflagellar transport (IFT) proteins are conserved mediators of cilium signaling. IFT sub-complex A is known to regulate retrograde IFT in the cilium. As a core protein of IFT complex A, IFT140 has been shown to have a relationship with serious skeletal ciliopathies caused in humans. However, the effects and mechanisms of IFT140 in bone formation have not been systematically disclosed. To further investigate the potential role of IFT140 in osteogenesis, we established a mouse model by conditional deletion of IFT140 in pre-osteoblasts. The adult knock-out mice exhibited dwarf phenotypes, such as short bone length, less bone mass, and decreased bone mineral apposition rate. In addition, by IFT140 deletion, the expressions of several osteoblastic markers were decreased and loss of bone became severe with aging. These results suggest that cilia gene Ift140 is essential in bone development.
Introduction
Protruding from cell surface, primary cilia act as an antenna in most eukaryotic cells. 1 Recent studies indicate that primary cilia play a pivotal role in development and functioning of multiple organs by mediating signaling transduction and mechanosensation. 2 The dysfunction of primary cilia will cause a series of diseases called ciliopathies. 3 The maintenance and signaling of cilia require large protein particles, intraflagellar transport (IFT) complex, which is the bidirectional movement signal transport complex driven by motors within the cilium. There are two IFT complexes in cilium: IFT complex B which transports signals from the basal body to the ciliary tip by kinesin-2 motor, while retrograde IFT transports inversely through dynein motor and IFT complex A.4,5 The integrity of IFT-A is essential for cilia stability and proper cellular localization. IFT complex A consists of 6 subunits: IFT144, IFT140, IFT122, IFT139, IFT121, and IFT43. 6 Due to the regulation of ciliogenesis and retrograde transport, loss of IFT140, IFT144, or IFT121 could cause very short cilia or even loss of cilia entirely.7–9 Mutations in IFT-A members could cause a set of related symptoms with multiple organ defects accompanied by skeletal anomalies.10–12
Bone is a highly mineralized tissue that undergoes continuous remodeling during embryonic development, homeostasis, and wound healing. 13 All dysfunctions of bone formation occurring during the development and remodeling will cause severe bone diseases. Recent researches indicate that primary cilia have an essential role in bone development, and there is a close connection between IFT mutations and clinical skeletal features.14,15 It’s worth noting that, for members of IFT complex A, gene mutation always causes skeletal phenotypes. 16 Among them, the mutation of IFT140 has been newly identified in patients with severe skeletal disorders. These bone dysplasia disease include Mainzer–Saldino syndrome (MSS), 17 Jeune asphyxiating thoracic dystrophy (JATD), 18 or Opitz trigonocephaly C syndrome (OTCS) 19 which commonly lead to phalangeal cone-shaped epiphyses, mild abnormalities of the proximal femur, shortened ribs, and a narrow, bell-shaped chest. As a model of Jeune syndrome, the Ift140cauli/cauli mice with a recessive missense mutation in the Ift140 gene also exhibit several skeletal development defects. 7 All these evidences indicate that Ift140 has an essential role in skeleton development. However, the links between the IFT complex A and patients with ciliopathies have not been well explored. In addition, due to the mid-gestational lethality, available Ift140cauli/cauli model has limitations to study the role of IFT140 during bone development and remodeling.
In this study, we established mouse model with pre-osteoblast-specific deletion of IFT140 (Osx-Cre; Ift140flox/flox) to investigate functions of IFT140 in osteogenesis. The knock-out mice exhibited bone dysplasia with defects in bone formation. The detailed data presented here will further reveal the effects of osteogenesis regulated by IFT molecules in primary cilia.
Materials and Methods
Mouse Models
To investigate the role of IFT140 in bone formation, we generated conditional deleted Ift140 (Ift140-cKO) in pre-osteoblasts by crossing Ift140flox/flox mouse (Fig. 1A–C) with Osx-Cre mice. Detailed information remained the same as described previously. 20 Animals were maintained in a specific pathogen-free (SPF) facility under a 12/12h day/night illumination cycle. The mice were euthanized by cervical dislocation after inhalation anesthesia. The Animal Welfare Committee of Tongji University approved all animal experimental protocols used in this study.
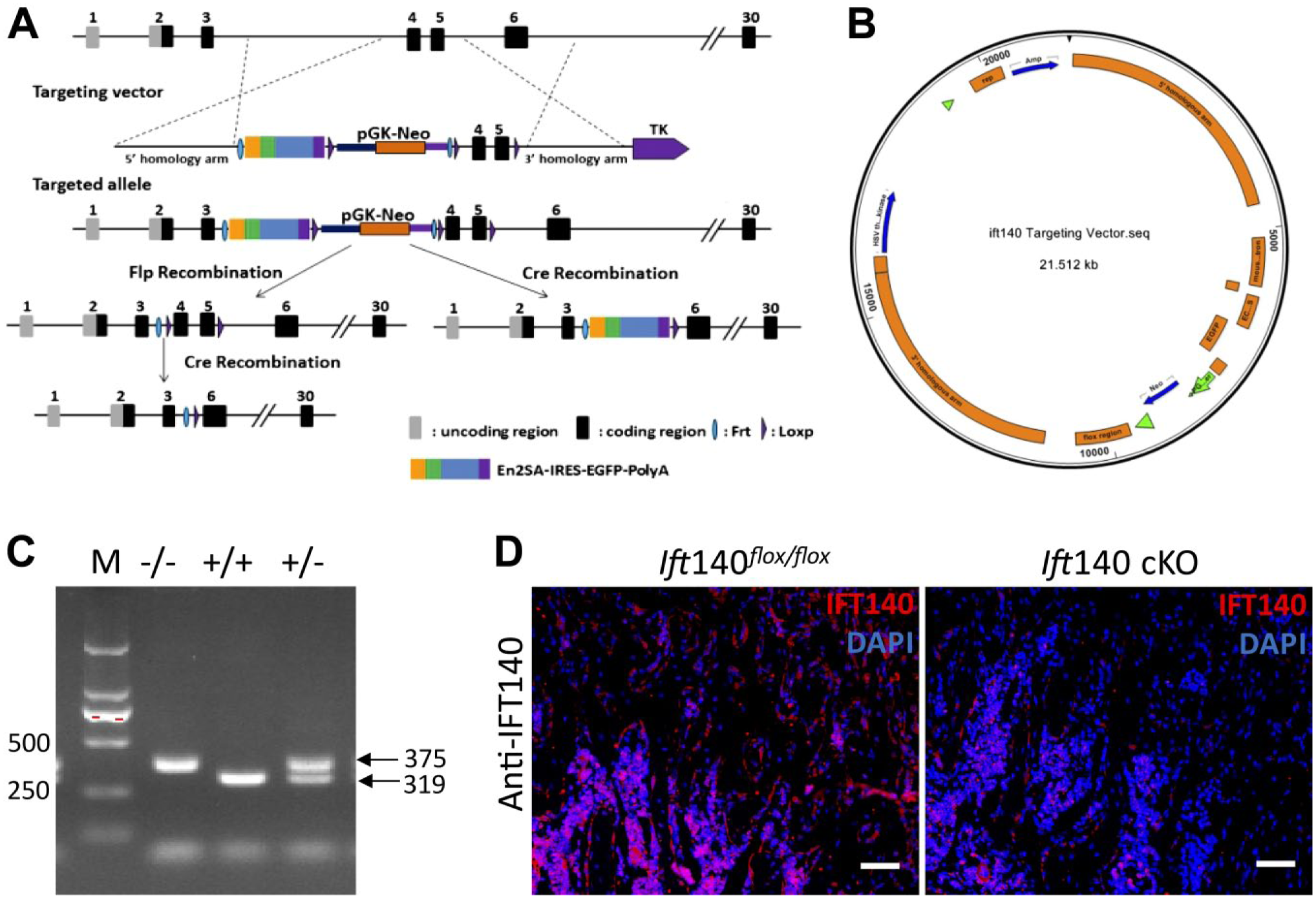
Deletion of IFT140 in pre-osteoblasts. (A) The design strategy of conditional deletion of Ift140 gene. (B) The targeting vector in embryonic stem (ES) cells based on C57BL/6J mice (Shanghai Biomodel Organism Science & Technology Development Co. Ltd, Shanghai, China). (C) Genotyping of Ift140flox/flox mice primers used for exons 4 and 5 conditional knock-out identification included the following: F-primer, 5’-ATCTTAATTTGTGTTGAAGGGGTT-3’, and R-primer, 5’-CTGCCAGGGGTACATGGTAGTAAG-3’. Product size of endogenous Ift140 was 319 bp, and product size of Ift140flox/flox was 375 bp. (D) Immunofluorescence staining of IFT140 (red) in trabecular bone of 8-week-old mice. Ift140 cKO mice showed remarkably less expression of IFT140 in trabecular bone. Scale bars: 50μm.
Micro–CT Analysis
All femurs dissected from eight 8-week-old and eight 6-month-old female Ift140 cKO mice and control littermates were fixed in 4% paraformaldehyde (PFA) for 48 hr. The femurs were subjected to analysis by micro–CT 50 (Scanco Medical, Zurich, Switzerland) at a scan resolution of a 10-μm slice increment with a voltage of 70 kV and a current of 200 μA. For quantifying trabecular bone parameters, 100 CT slices immediately proximal to the distal femoral growth plate were analyzed. The parameters of trabecular bone volume/total volume (BV/TV), trabecular number (Tb.N), trabecular thickness (Tb. Th), structure model index (SMI), connective density (Conn.D), and bone mineral density (BMD) were quantified according to the standard procedures. 21
Histological Analyses
For histological analyses, femurs from eight female Ift140 cKO and eight control mice were decalcified in 10% EDTA (pH 7.4) at 4C for 1 month. During dehydration through a graded ethanol series, specimens were embedded in paraffin and cut into 5-μm-thick sections. For morphological evaluation, the sections were stained with hematoxylin and eosin (H&E) (Sangon Biotech, Shanghai, China). To evaluate the content of collagen and proteoglycan, the sections were stained with Masson (Nanjing Jiancheng Bioengineering Institute, Nanjing, China) and alcian (Beijing Solarbio Science & Technology Co., Ltd. Beijing, China). For immunohistochemistry staining, sections were deparaffinized with xylene and rehydrated in a descending series of ethanol concentrations. Antigen retrieval was performed using hyaluronidase, then sections were treated with 3% hydrogen peroxide and goat serum blocking. Primary antibodies used were as follows: anti-DMP1-C-8G10.3 (1:300; gifts from Dr. Chunlin Qin, TA&M University, College of Dentistry) and anti-Osteocalcin (1:100; Santa Cruz Biotechnology, Dallas, TX). A DAB detection kit (Maxim Biotechnology, Fuzhou, China) was used to do color development. For immunofluorescence staining, specimens were embedded in OCT and sectioned at an 8-μm thickness after being decalcified. The sections were incubated with anti-acetylated α-tubulin (1:1000; Sigma-Aldrich, St Louis, MO) and by Alexa Fluor 488 IgG (1:1000; Invitrogen, Carlsbad, CA). Anti-IFT140 (1:50; Proteintech, Wuhan, China), anti-type II collagen (1:200; Boster Biological Technology, Wuhan, China) or anti-Ostreix (1:300; Abcam, Cambridge, UK) at 4C overnight and by Alexa Fluor 546 IgG (1: 1000; Invitrogen) and phalloidin at room temperature for 1 hr (50 μM; Sigma-Aldrich). Sections were subsequently stained with DAPI (Sigma-Aldrich). And Von Kossa staining was performed using 1% silver nitrate and 3% sodium thiosulfate solution counterstaining by nuclear fast red. For double fluorochrome labeling, Alizarin red (20 mg/kg; Sigma-Aldrich) was first injected into 8-week-old mice by intraperitoneal injection, followed by injection of calcein (5 mg/kg; Sigma-Aldrich) 7 days later. Meanwhile, we performed Van Gieson (Beijing Solarbio Science & Technology Co., Ltd. Beijing, China) staining of corresponding sections used in double fluorochrome labeling to display morphological image of bone cross section. The alkaline phosphatase (ALP) staining was performed using ALP assay kit and following the protocol (Sangon Biotech).
Quantitative RT-PCR Analysis
Total RNA was isolated from 2-mm bony segment under the growth plate of eight 8-week-old female Ift140 cKO and eight control mice by using Trizol reagent (Invitrogen) according to the manufacturer’s protocol. cDNA was synthesized using a Transcriptor First Strand cDNA Synthesis Kit (Roche, Basel, Switzerland). A reaction mix was prepared using SYBR green master mix (Roche), following manufacturer instructions. The expression levels of target gene were evaluated by using Light Cycler 96 PCR system (Roche). Reactions of each sample needed to be run in triplicate. The primers used for osteogenic markers included the following: GAPDH-forward: 5’-TGTGTCCGTCGTGGATCTGA-3’, GAPDH-reverse: 5’-CCTGCTTCACCACCTTCTTGA-3’; Alp-forward: 5’-GATCATTCCCACGTTTTCACATT-3’, Alp-reverse: 5’-TTCACCGTCCACCACCTTGT-3’; Ocn-forward: 5’-GAGGACCATCTTTCTGCTCACTCT-3’, Ocn-reverse: 5’-TTATTGCCCTCCTGCTTGGA-3’; Opn-forward: 5’-GATCAGGACAACAACGGAAAGG-3’, Opn-reverse: 5’-GCTGGCTTTGGAACTTGCTT-3’; Bsp-forward: 5’-AGGACTGCCGAAAGGAAGGTTA-3’, Bsp-reverse: 5’-AGTAGCGTGGCCGGTACTTAAA-3’.
Statistical Analysis
All data were analyzed using Student’s t test for two-group comparison by SPSS 20.0. The p values less than 0.05 were considered statistically significant.
Results
Deletion of IFT140 Stunted Bone Development in Mice
In order to investigate the role of IFT140 in bone formation, Osx-Cre; Ift140flox/flox mice were generated to specifically delete the Ift140 in pre-osteoblasts. First, we performed immunofluorescence staining of IFT140. There were significant fewer IFT140 positive cells in trabecular bone area of Ift140 cKO mice (Fig. 1D). As observed, 8-week-old Ift140 cKO mice exhibited growth retardation and smaller body size than that of control (Fig. 2A). And the body weight of Ift140 cKO mice was obviously decreased (Fig. 2B).
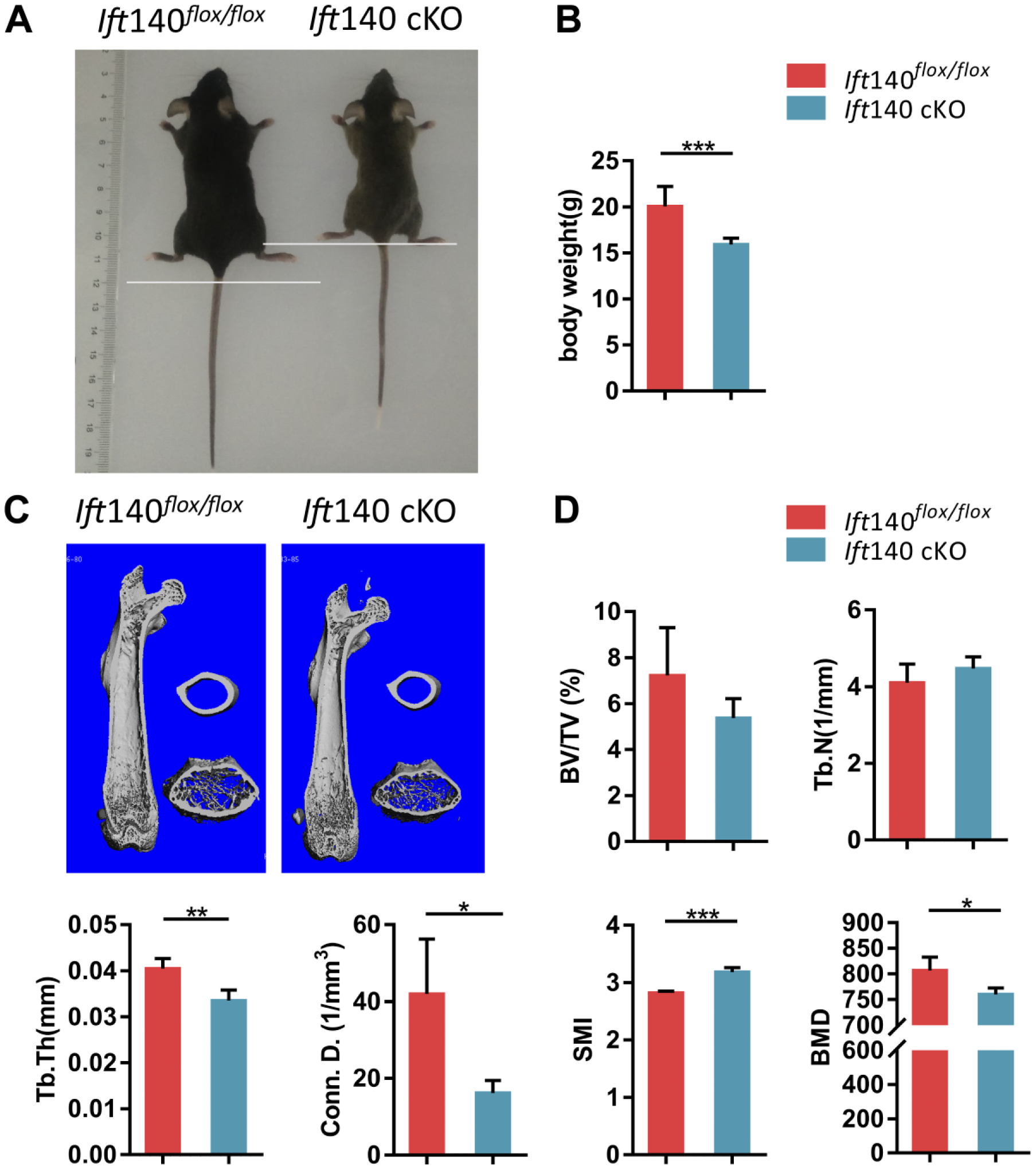
Bone phenotypes in Ift140 cKO mice. (A) Representative image of Ift140 cKO mice and control at 8-week-old. Body sizes of Ift140 cKO mice were shorter. (B) Quantification of body weight of Ift140 cKO mice and control at age 8 weeks, n=7. (C) Micro–CT analysis of femurs obtained from Ift140 cKO and control mice. Trabecular bones of IFT140 cKO mice were less and cortical bone of Ift140 cKO mice was thinner. (D) Quantitative micro-CT analysis of femur trabecular bone. *p<0.05, **p<0.01, ***p<0.001, n=5. Abbreviations: BV/TV, trabecular bone volume; CT, computed tomography; Tb.N, trabecular number; Tb.Th, trabecular thickness; Conn.D, connective density; SMI, structure model index; BMD, bone mineral density.
Less Bone Formation in 8-week-old Ift140 cKO Mice
Micro-CT scanning was employed to evaluate bone morphology and mass changes. Compared with controls, a narrow midshaft of bone marrow cavity, thinner trabecular bone, and cortical bone around trabecular bone were found in Ift140 cKO mice (Fig. 2C). The micro-CT analyses of trabecular bone indicated less bone mass in Ift140 cKO mice (Fig. 2D). Further histological analyses by H&E staining revealed less trabecular bone in ossification center of 8-week-old Ift140 cKO mice (Fig. 3A). Following merging of fluorochromes xylenol orange and calcein, the mineral deposition rate in the mid-shaft region of the femurs was compared. There was less bone deposition at the endocortical surface in Ift140 cKO mice at age 8 weeks (Fig. 3C and E). However, there was no obvious difference of Masson staining for matrix collagen between the Ift140 cKO mice and control littermates at age 8 weeks (Fig. 3B).
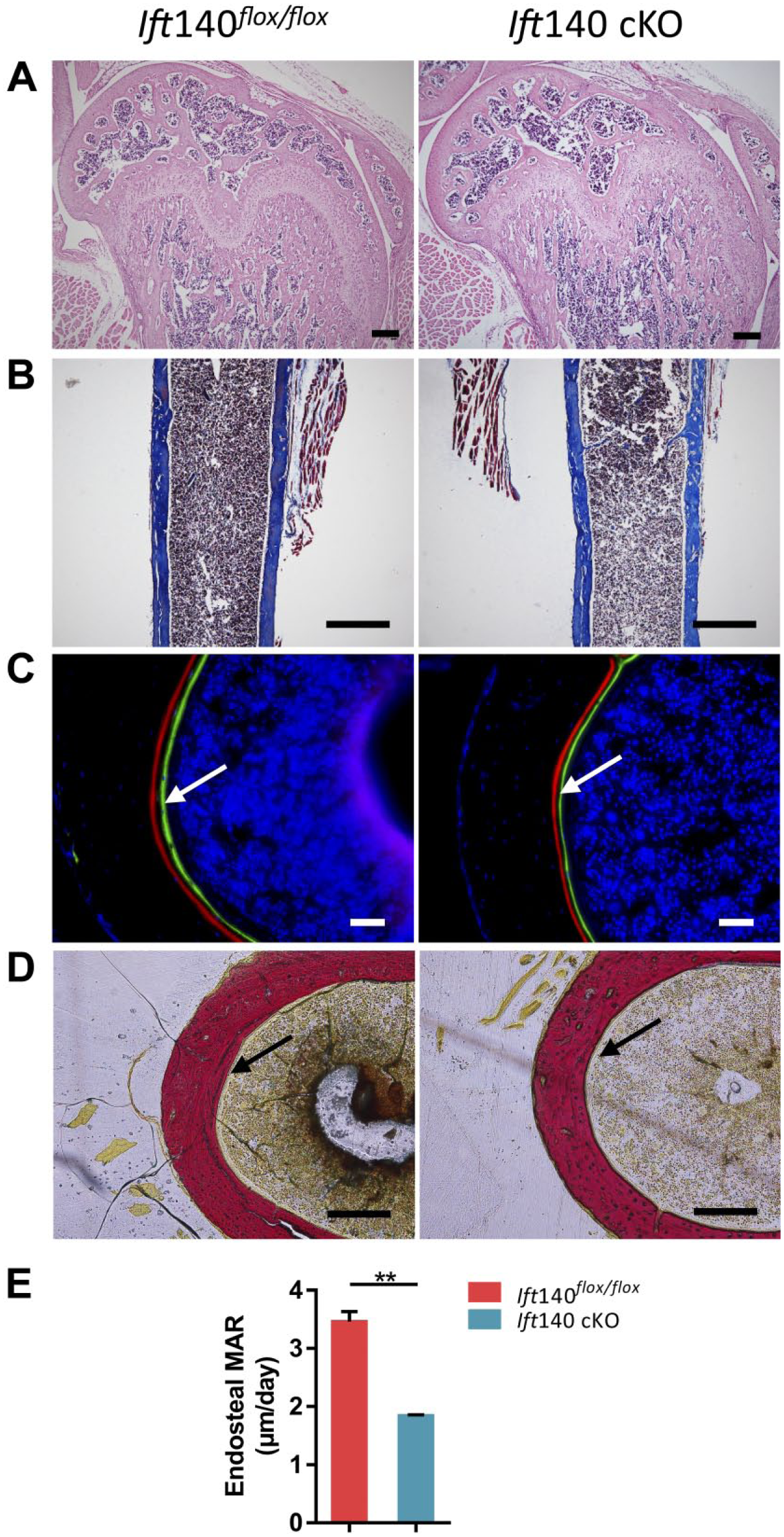
Reduced bone formation in Ift140 cKO mice at age 8 weeks. (A) Hematoxylin and eosin staining showed less and thinner trabecular bone in Ift140 cKO mice. Scale bars: 200 μm. (B) Masson staining showed no difference in collagen distribution in cortical bone. Scale bars: 100 μm. (C) Cross sections of femurs from control and Ift140 cKO mice. The distance between green (calcein) and red (Alizarin red) represented the mineral apposition rate of cortical bone in 7 d. The arrows showed the endocortical surface. Ift140 cKO mice had a slower mineral deposition rate compared with the control littermates. Scale bars: 50 μm. (D) Van Gieson staining of corresponding sections in 3C. The arrows showed the endocortical surface. Scale bars: 50 μm. (E) Quantitative measurements of the lower bone mineral apposition rate in Ift140 cKO mice. **p<0.01, n=4.
Reduced Expressions of Osteogenic Markers in Long Bone of Ift140 cKO Mice
To further identify the impaired bone formation and mineralization in Ift140 cKO mice, we then performed immunohistochemical analyses by using anti-DMP1 and anti-osteocalcin antibodies, and found that expression of DMP1 or osteocalcin was lower in Ift140 cKO bone (Fig. 4A). The ALP staining also showed that ALP activity in mutant osteoblasts was significantly decreased (Fig. 4B). The expression of Osterix also reduced in Ift140 cKO mice (Fig. 4C). And the mRNA expression levels of osteogenic markers (Alp, Ocn, Opn, Bsp) were all reduced in Ift140 cKO mice (Fig. 4D).
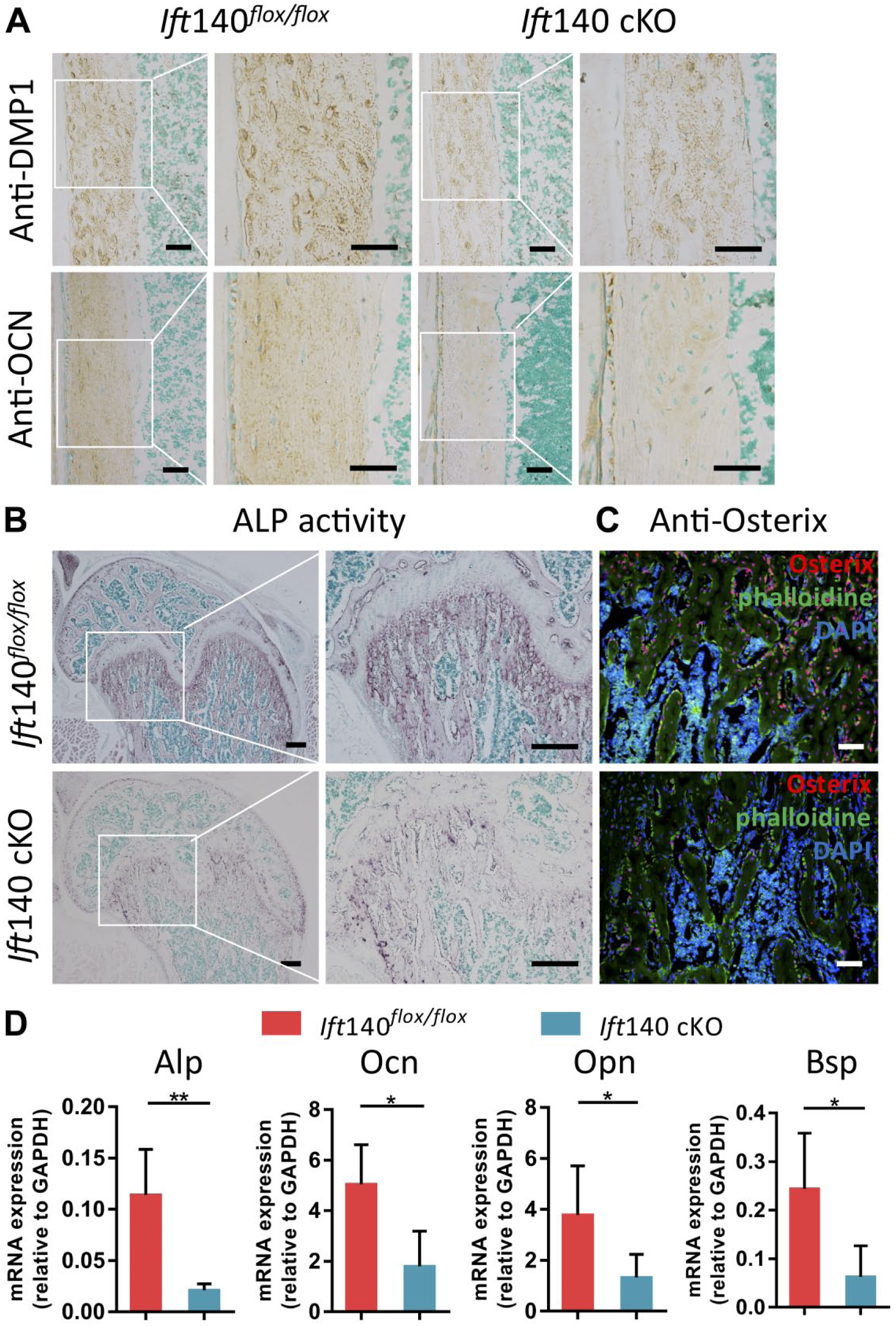
Disturbed osteogenic markers expressions in Ift140 cKO mice at age 8 weeks. (A) Immunohistochemistry (IHC) staining of DMP1 and OCN in cortical bone from 8-week-old mice. Ift140 cKO mice showed remarkably less expression of DMP1 and OCN in cortical bone matrix. Boxes in left panel were shown magnified in the right panel, respectively. Scale bars: 50 μm. (B) Alkaline phosphatase activity of trabecular bone. Ift140 cKO mice showed remarkably weakened staining. Boxes in up panel were shown magnified in the down panel, respectively. Scale bars: 200 μm. (C) Immunofluorescence staining of Osterix (red) in trabecular bone of 8-week-old mice. Ift140 cKO mice showed remarkably less expression of Osterix. Scale bars: 50 μm. (D) RT-qPCR quantification analysis of osteogenic markers. Ift140 cKO mice displayed down-regulated expression levels of Alp, Ocn, Opn and Bsp. *p<0.05, **p<0.01, n=5.
Lack of IFT140 Induced Severe Bone Loss With Aging
Ift140 cKO mice displayed a severe bone loss phenotype with aging. Micro-CT scanning showed intumescentia in metaphysis of femur, shortened femur length, and less trabecular bone in femurs of mutant mice (Fig. 5A). Due to loss of growth plate structure in aged Ift140 cKO mice (Fig. 5A and B), we didn’t perform the bone histomorphometric analyses for secondary ossification center or trabecular bone area under growth plate. H&E and Von Kossa staining also exhibited significant bone loss (Fig. 5B). To further evaluate the quality of bone, Masson staining was performed and the results showed different collagen distributions and poor mineralization in cortical bone (Fig. 5D). Meanwhile, the expression levels of DMP1 and OCN were significantly reduced in Ift140 cKO mice by immunohistochemical analyses (Fig. 5E).
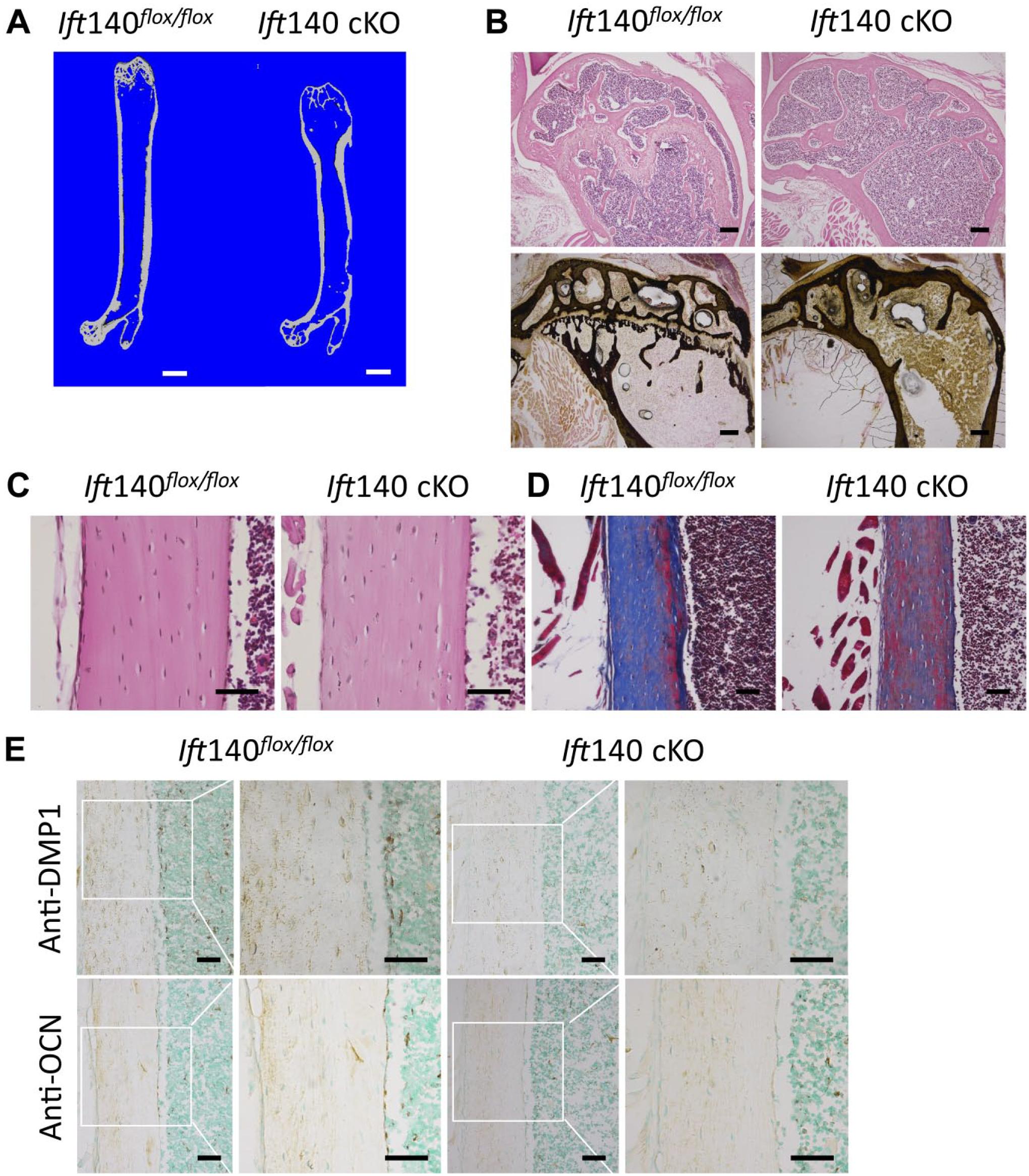
Severe bone abnormalities in Ift140 cKO mice at age 6 months. (A) Micro–computed tomography (CT) cross section scan of femur obtained from Ift140 cKO and control mice at age 6 months. Ift140 cKO mice showed severe morphological abnormality. Scale bars: 1 mm. (B) Hematoxylin and eosin staining and Von Kossa staining showed less trabecular bone and the disappearance of growth plate in 6-month-old Ift140 cKO mice. Scale bars: 200 μm. (C) Hematoxylin and eosin staining of cortical bone at age 6 months. Scale bars: 50 μm. (D) Masson staining of cortical bone exhibited abnormal collagen distributions and poor mineralization in Ift140 cKO mice at age 6 months. Scale bars: 50 μm. (E) Immunohistochemistry (IHC) staining of DMP1 and OCN of cortical bone from 6-month-old mice. Ift140 cKO mice showed remarkably less expression of DMP1 and OCN in cortical bone matrix. Boxes in left panel were shown magnified in the right panel, respectively. Scale bars: 50 μm.
Phenotypes of Growth Plate in Ift140 cKO Mice
During the bone development and remodeling, we found another noteworthy phenotype of growth plate. At age 8 weeks, the chondrocytes of growth plate became abnormal in Ift140 cKO mice. The column arrangements of chondrocytes were disordered and replaced by mineralized bone tissue gradually (Fig. 6A and A’), which also resulted in decreased proteoglycans and hyaluronic acid in bone matrix according to alcian staining (Fig. 6B and B’). The expression level of type II collagen was also reduced in Ift140 cKO mice (Fig. 6C and C’). And to further investigate the turbulence of growth plate, we performed staining of IFT140 (Fig. 6D and D’) and the axoneme to visualize primary cilia in chondrocytes (Fig. 6E and E’). The expression of IFT140 reduced in growth plate and there were fewer ciliated fluorescence positive cells in Ift140 cKO mice. Consequently, the growth plate began to disappear gradually and there was no growth plate at age of 6 months (Fig. 5B).
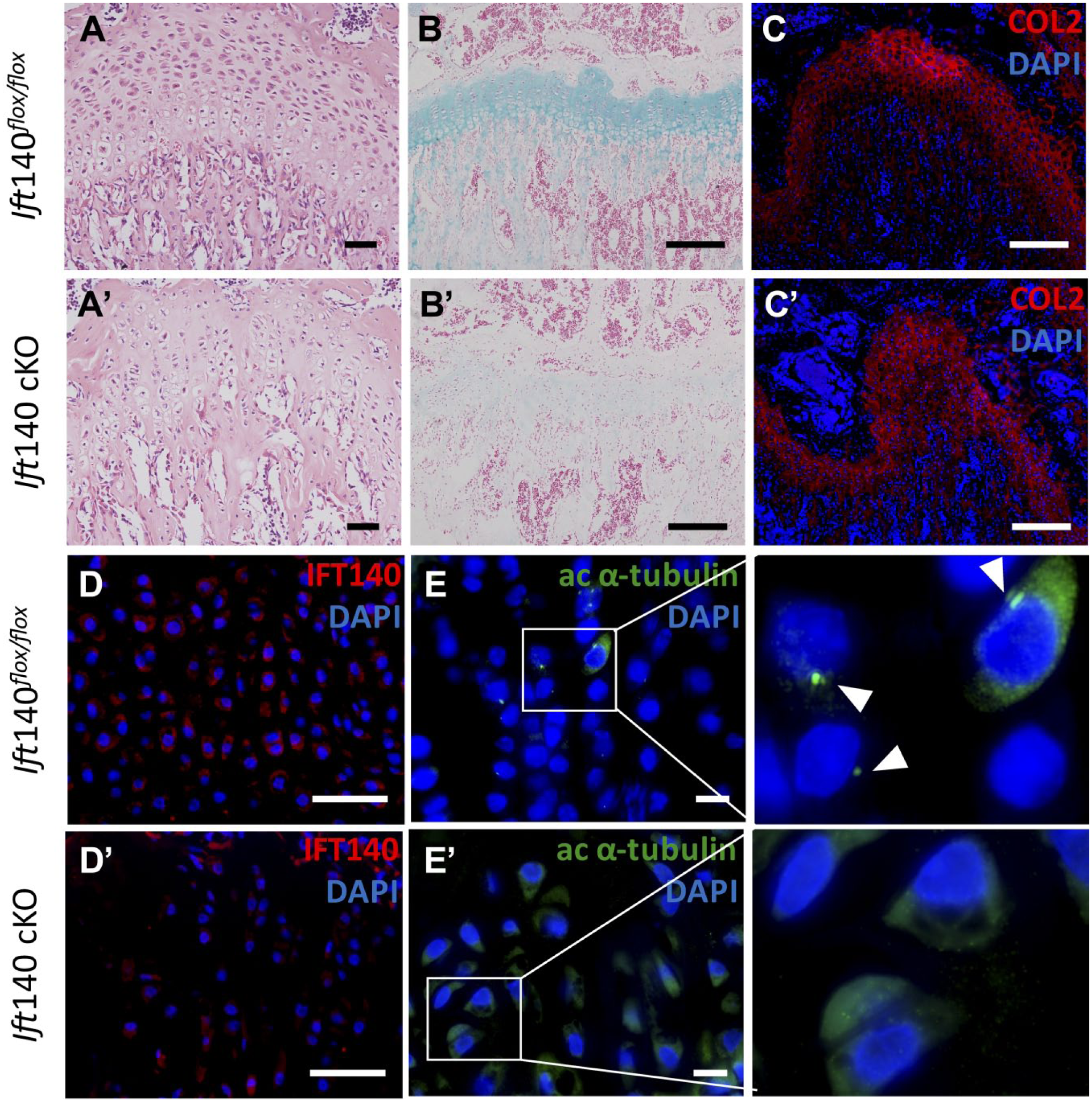
Defects of growth plate in Ift140 cKO mice. (A and A’) Hematoxylin and eosin staining of growth plate showed disordered column arrangements in Ift140 cKO at age 8 weeks. Scale bars: 50 μm. (B and B’) Alcian blue staining of growth plate at age 8 weeks. Scale bars: 200 μm. (C and C’) Type II collagen (red) immunofluorescence staining in growth plate at age 8 weeks. Scale bars: 200 μm. (D and D’) Immunofluorescence staining of IFT140 (red) in growth plate of 8-week-old mice. Ift140 cKO mice showed remarkably less expression of IFT140. Scale bars: 50 μm. (E and E’) Immunofluorescence staining of acetylated α-tubulin (green) in cilia in growth plate of 8-week-old mice. Boxes in left panel were shown magnified in the right panel, respectively. Arrowheads showed the primary cilia. Scale bars: 50 μm.
Discussion
IFT protein complex functions to transport various proteins within the cilia for participating transductions. Several IFT members have been proved to play key roles in regulating osteogenesis. Dysfunction of IFT molecules and primary cilia results in skeletal dysplasia.22,23 IFT88, a member of IFT complex B, works in keeping cilia formation and function. 24 Deletion of Ift88 in Prx1-positive cells results stunted limb growth due to disruptions in endochondral and intramembranous ossification. 25 In addition, deletion of Ift88 and subsequent depletion of primary cilia from chondrocytes (Col2a-Cre;Ift88flox/flox) results in disorganized columnar structure and early loss of growth plate. 26 And deletion of Ift80, another IFT members, by using Osx-Cre also causes a significant growth retardation and osteopenia. 27 Similar to Ift88 and Ift80, loss of Ift140 also exhibited the impaired bone formation and growth retardation in mice. There were significant differences in body length and weight between Ift140 cKO mice and control mice at age 8 weeks. Meanwhile, in Ift140 cKO mice, significant down-regulation of osteogenic markers began to be observed along with low mineral deposition rate. The osteodysplasia phenotypes of Ift140 cKO mice started at age 8 weeks and ultimately resulted in severe bone abnormalities at age 6 months. With aging, the Ift140 cKO mice displayed significant decrease in trabecular bone mass and mineralization in cortical bone. In addition, intumescent metaphysis area in mouse long bone is consistent with phalangeal cone-shaped epiphyses in MSS, a human disease with Ift140 mutation. 28 And the abnormalities in metaphysis area are not reported in the phenotypes of other IFT gene knockout.
It is also interesting to know that, in the process of osteoblast differentiation, the role of IFT140 has time and space specificity. Osterix is a pre-mature osteoblast marker, and Osx-Cre could delete Ift140 in early stage during osteoblast maturation. By crossing Ift140flox/flox mice with Osx-Cre line, the descendants exhibit morphological abnormality in bone development and remodeling. In addition, by crossing Ift140flox/flox mice with Dmp1-cre mice, which targets the mature osteoblasts and osteocytes, the bone phenotypes are the same as that of wild-type mice (data not shown). According to this, we believe that the role of IFT140 in osteogenesis is mainly in the early stage of osteoblast differentiation.
Interestingly, we also noticed the abnormal phenotype on growth plate in our mouse model. Osx-Cre; Ift140flox/flox mice (Ift140 cKO mice) exhibited narrow and disorganized growth plate. Recent studies showed that primary cilia could also mediate chondrocyte rotation to maintain columnar organization. 6 In addition to osteoblasts, the Osx-Cre transgenic mice has been shown to direct Cre recombinase activity in pre-hypertrophic and hypertrophic chondrocytes in the growth plate. 29 The growth plate degeneration of Ift140 cKO mice began at age 8 weeks. The growth plate showed disorganized columnar structure of chondrocytes and ectopic osteogenesis. And it also exhibited lower expression level of type II collagen in Ift140 cKO mice. Consequently, the growth plate completely disappeared by conditional knock out Ift140 in 6-month-old mice. Due to the responsibility of growth plate in the longitudinal growth of bones, 30 the advanced degeneration of growth plate resulted in postnatal dwarfism phenotypes in Ift140 cKO mice. Our results also suggest that IFT140 could be required to maintain the growth plate by functioning in pre-hypertrophic and hypertrophic chondrocytes. And it still needs systematic investigation by using different stages of chondrocyte-specific Cre lines to explore the function of IFT140 in growth plate and chondrocytes.
In conclusion, our study provides information of IFT140 in regulating the skeleton development. And it indicates that IFT140 is essential for bone development by regulating the activity of pre-osteoblasts and also maintaining growth plate.
Footnotes
Acknowledgements
We gratefully acknowledge the DMP1-C antibody from Dr. Chunlin Qin (College of Dentistry, Texas A&M University). And we also appreciate Ruilin Zhang, Mengqi Zhou, and Qiqi Fan (Department of Implantology, School & Hospital of Stomatology, Tongji University) for their help in sample preparation.
Competing Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
All authors have contributed to this article as follows: DT for acquisition of data and drafting the article, HX for sample processing and statistical analysis, CZ and GL for revising article, and YS for design and manuscript reviewing.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was founded by Key Project of Chinese National Programs for Research and Development (2016YFC1102705, Y. Sun), National Science Foundation of China (81822012, 81470715, and 81771043, Y. Sun), Shanghai Health System (2017 BR009, Y. Sun), and Basic scientific research operating expenses of central universities—Interdisciplinary research project of Tongji University (20173386).
