Abstract
We investigated the expression of osteocalcin (OC), bone sialoprotein (BSP), osteonectin (ON), and alkaline phosphatase (ALP) during cell differentiation and bone nodule formation by fetal rat calvaria cells, using immunofluorescent and immunogold techniques at light and electron microscopic levels. Six hours after plating all proteins were expressed in calvaria cells. However, expression was not detected during the proliferation phase after plating. Cell morphological modifications were observed in osteoblastic cells expressing ALP, OC, and BSP, but not ON. During the matrix formation phase, all proteins were expressed with various intensities and OC was limited to differentiated osteoblastic cells. EM observations demonstrated that BSP was selectively associated with clusters of needle-like crystals, but not with collagen fibers, in mineralization foci and in the mineralized matrix. OC was localized intracellularly and in all the extracellular compartments, and was concentrated at the mineralization front. ON was distributed uniformly throughout the osteoid and mineralized matrix, which was intensely labeled. The results show that the expression of bone matrix proteins during differentiation of calvaria cells and nodule formation in vitro duplicate what is observed during osteogenesis in vivo.
Keywords
According to cell kinetics based on autoradiographic and microcinematographic studies (Nefussi et al. 1985,1989b,1993), four successive morphological steps of bone nodule formation can be distinguished: (a) cell proliferation with formation of multicellular layers; (b) cell surface morphological changes with cell differentiation; (c) cell activity with matrix formation and maturation and cell inclusion processes; and (d) woven bone matrix mineralization with formation of active bone surface and mature osteocytes. The biological relevance of the calvaria bone cell nodule system as a model for osteoblast differentiation is supported by ultrastructural (Nefussi et al. 1985,1989a, 1991; Barghava et al. 1988) and biochemical studies (Collin et al. 1992). In addition, the temporal expression of genes in vitro (Stein and Lian 1993; Lian et al. 1992; Aronow et al. 1990; Owen et al. 1990) is consistent with the expression of these genes by cells recognized to be at different stages of differentiation in vivo (Chen et al. 1992; Weinreb et al. 1990; Nomura et al. 1988) and during fetal calvarial development (Machwate et al. 1994). Therefore, bone nodule formation by rat calvaria cells appears to represent an appropriate model to study the sequential expression of the developing osteoblast phenotype during osteogenesis.
This study was undertaken to correlate the previously defined morphological steps with in situ localization of NCP synthesis and ALP activity and to identify the localization and variation of NCP expression during osteoblast differentiation in the calvaria nodule system. In this study, the expression of ALP, an early marker of the osteoblastic lineage, and of OC, BSP, and ON was determined during osteoblast differentiation in vitro. Osteocalcin was chosen because this protein is considered to be a marker for differentiated osteoblasts. We also studied the expression of BSP and ON, which are associated in the mineralization process, and are expressed by osteoblasts during bone formation (Roach 1994).
Materials and Methods
Bone Cell Isolation and Culture
Calvaria bone cells from 21-day-old fetal Sprague-Dawley rats (Ifa-Credo; Arbresle, France) were isolated according to the procedure described previously (Nefussi et al. 1985). Briefly, pregnant Sprague–Dawley rats were anesthesized and sacrificed by decapitation. The 21-day-old fetuses were removed, and their heads were cut off and immersed in PBS. Calvaria were then dissected out and processed for cell isolation and culture. Central parts of the parietal and frontal bones, with their endosteum and periosteum, were incubated for 2 hr at 37C in PBS with 0.25% collagenase (Sigma, St Louis, MO; Type I). Cells dissociated from the bone fragments were washed several times in PBS, counted, and seeded (2 × 104 cells/cm2) either on 20-mm glass coverslips or on 25-mm porous membrane of cell culture inserts in conjunction with six-well plates (Falcon). Whatever the support, the cells were cultured in Dulbecco's modified Eagle's medium (DMEM) (Gibco; Grand Island, NY) with 10% fetal calf serum (FCS) (Boehringer; Mannheim, Germany) supplemented with 50 U/ml streptomycin (Gibco) and 50 μg/ml penicillin (Gibco). Cell culture dishes were incubated in a humidified atmosphere of 5% CO2 in air at 37C. The medium was changed every 48 hr for 2 weeks. Cultures were stopped after 15 days and fixed for immunohistological examination.
EM Immunohistochemical Procedure
Fifteen-day-old cell culture membrane inserts were fixed in situ in 3% paraformaldehyde for 1 hr and the membrane containing bone nodules was cut into small pieces, dehydrated, and embedded in LR White resin (Polyscience; Warrington, PA). Ultrathin sections were made with a diamond knife and collected on colloidion grids. Grid-mounted sections were floated for 10 min on PBS containing 5% bovine serum albumin (BSA; Sigma), then washed in 1% BSA in PBS and incubated separately for 1 hr at room temperature (RT) on a drop of the following polyclonal antibodies: rabbit anti-rat Type α1I collagen (LF-67), rabbit anti-rat BSP (LF-87), and rabbit anti-bovine osteonectin (LF-23) (Fischer et al. 1983,1987,1995) (1:60 dilution; a generous gift from Dr. L.W. Fischer (NIDR, NIH, Bethesda); rabbit anti-rat osteocalcin 1:200 dilution (Modrowski et al. 1992). After incubation, sections were rinsed three times with PBS–1% BSA for 10 min and incubated for 1 hr at RT with protein A–gold complex (dilution 1:30) (gold particles ~10 nm; Amersham, Poole, UK). Primary antibodies were omitted in controls. Tissue sections were then washed thoroughly with PBS–1% BSA, rinsed with distilled water, and stained with uranyl acetate and lead citrate.
In Situ Immunofluorescence Localization
In situ immunolabeling was performed by indirect immunofluorescence. Cells on glass coverslips were fixed at 6 hr, and at 4, 5, 6, 8, and 10 days for 10 min at RT in 3.7% formaldehyde, followed by 70% ethanol solution for 5 min. The cells were then incubated separately for 30 min at 37C with the different antibodies mentioned above and used at the same dilution. When mentioned, demineralization was performed by incubation overnight at 4C in 10% EDTA–PBS, pH 7; this procedure was used only in the mineralized nodule step and was processed before incubation with the first antibody. After extensive washing, fluorescein-conjugated goat anti-rabbit globulin diluted 1:30 was incubated at 37C for 40 min. After further washing, the coverslips were mounted and observed under a Leitz orthoplan fluorescence microscope.
Alkaline Phosphatase Activity
This activity was determined as described by Osbody and Caplan (1981). Cultured cells on coverslips were fixed at RT in 2% acetic acid solution for 20 sec. The cells were then exposed for 5 min to a solution containing naphthol AS-MX phosphate (Sigma) as substrate and Fast Violet B Salt (Sigma) as coupler. The coverslips were then observed without counterstain. In cultures with ALP and BrdU reactions, the ALP activity was always performed before BrdU labeling.
BrdU Labeling
Cells were labeled for 1 hr with a solution of BrdU (1:1000) and then fixed in a ethanol-acetic solution (95/5). After several washes the cells were exposed for 1 hr with BrdU antibody (Amersham) and then for 30 min with the second monoclonal antibody bound to peroxidase. The cells were then stained with 3,3′-diaminobenzidine (DAB) and observed.
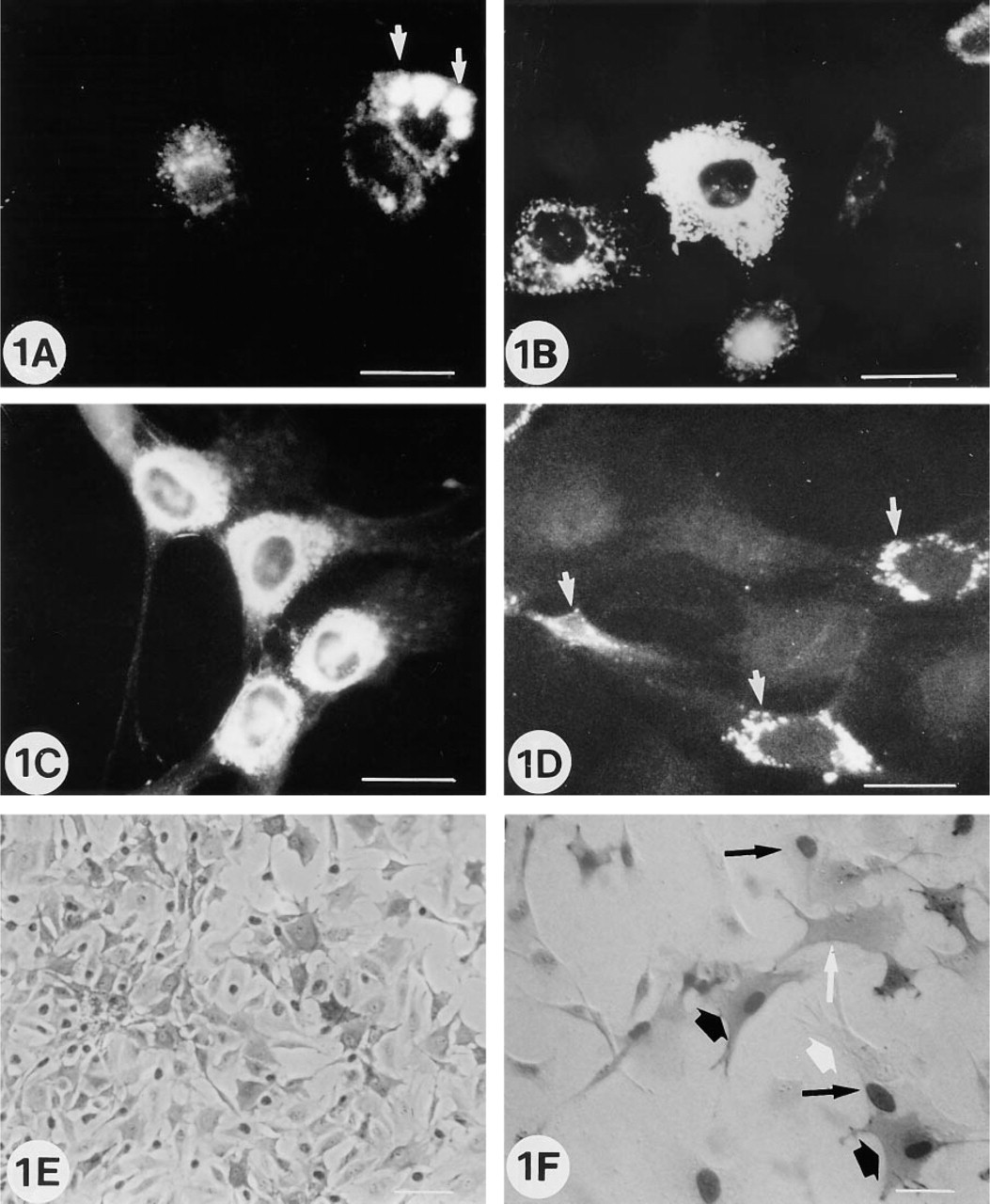
Collagen and non-collagen protein expression after 6 hr of culture
Results
In the first 6 hr after cell plating, isolated cells expressed OC, ON, BSP, Type I collagen proteins, and ALP activity (Figures 1A–1D). Diffuse intracytoplasmic immunofluorescent labeling was usually observed with OC antibody (Figure 1B), whereas individual immunofluorescent granular bodies were noted with ON and BSP antibodies (Figures 1C and 1D). Collagen labeling was usually expressed as large intracytoplasmic vacuoles (Figure 1A). Intense ALP proliferative (BrdU+) or nonproliferative (BrdU—) positive cells were also noted next to positive or negative ALP cells (Figures 1E and 1F). On Day 3 to 4 of culture, the cells were confluent and beginning to form multicellular layers, and most of the labeling observed in the isolated cells was no longer detectable (not shown).
On Day 5 to 6, individualized groups of ALP positive cells showed morphological cell modifications, which are the first visible expression of future bone nodule formation (Figures 2A and 2B). These cells were BSP- and OC-positive but ON-negative (Figures 2C and 2D). As previously mentioned, the BSP immunofluorescent labeling appeared as granular intracytoplasmic bodies, whereas OC labeling was diffuse.
During the following days (7–21 days), when the extracellular matrix is formed, immunofluorescent OC-and BSP-positive cells localized in the areas of the future bone nodule formation were more numerous, and some positive ON cells were observed. The OC labeling was expressed as an intracellular diffuse immunofluorescent labeling and was located only on one cell layer covering the matrix (Figures 3B and 3E). However, when nodules were demineralized with EDTA before staining, large immunofluorescent areas with positive dots corresponding to cells inside and around the nodule stained positive for OC (Figures 3C and 3D). Similar results were obtained with BSP and ON, but with a weaker intensity (data not shown).
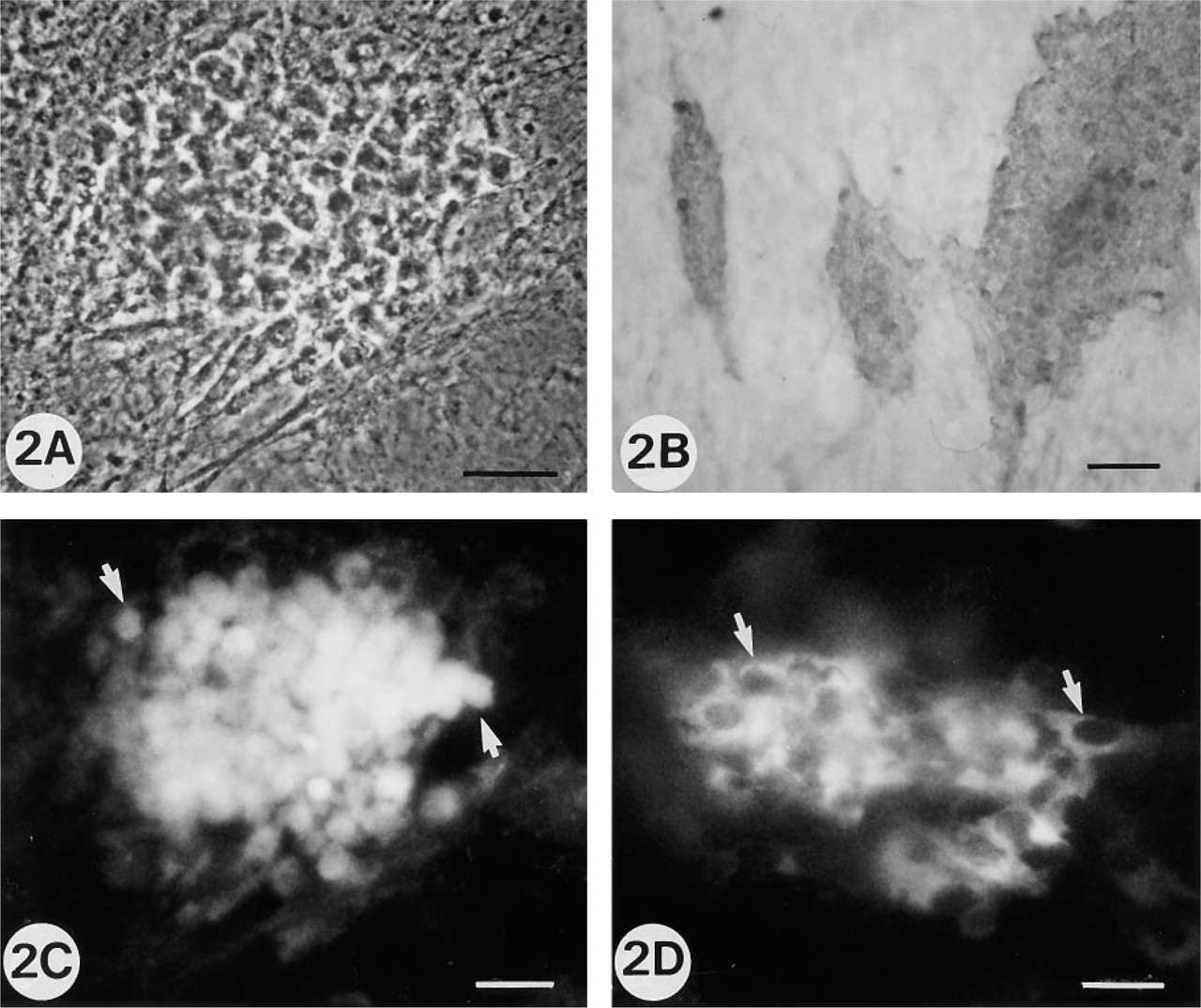
Rat calvaria cell cultures at 5 and 6 days, illustrating the cell surface morphological changes associated with onset of cell differentiation.
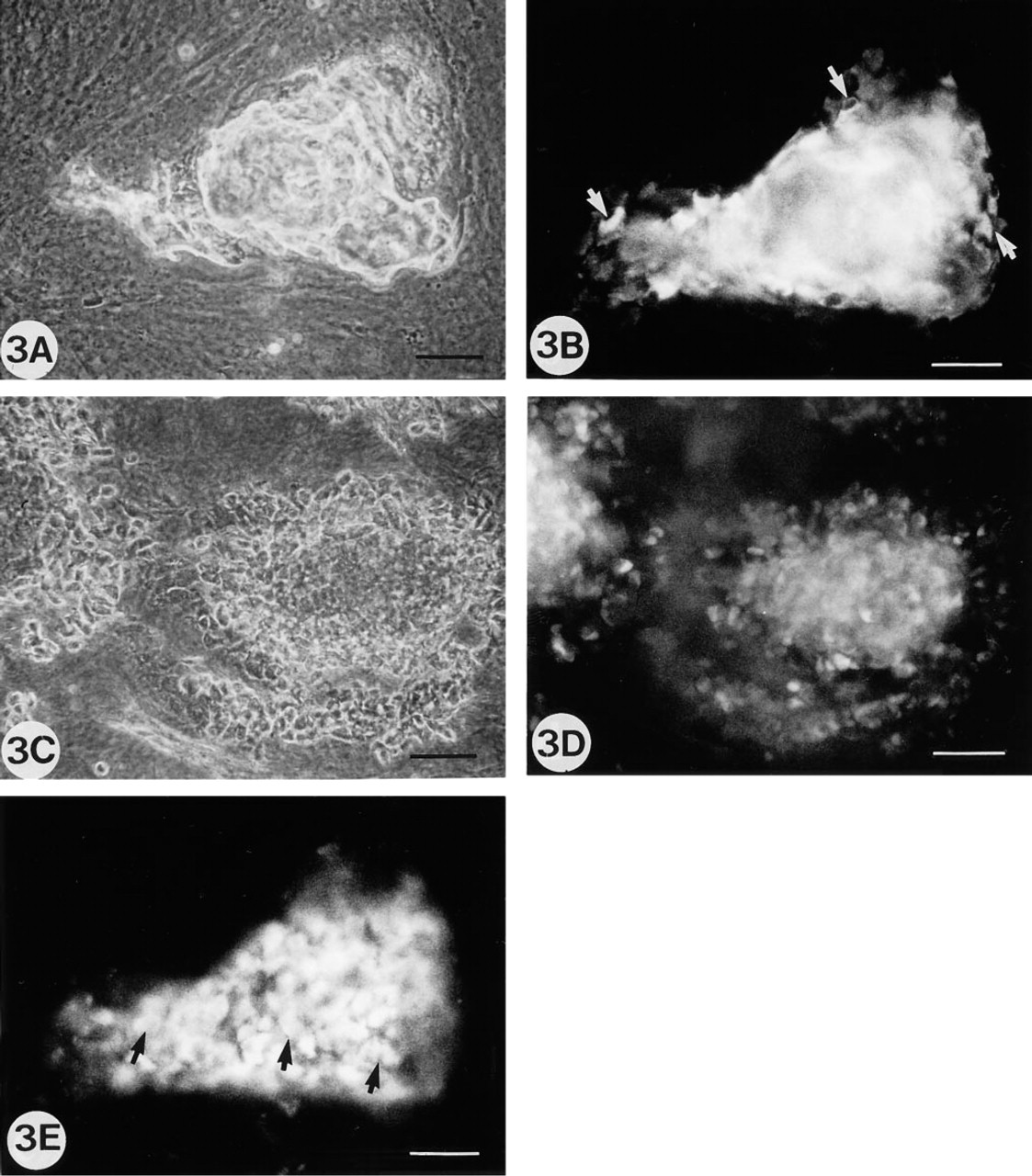
Rat calvaria cell cultures at Stages 3 and 4 (8–10 and 12–15 days of culture.
EM localization of NCPs was investigated in mature mineralized nodules. A very low background was observed in the control sections, with a few randomly distributed gold particles over the tissue section. In general, labeling of the intracellular compartment was low or even absent. Most of this intracellular labeling was observed with OC antibody and was restricted to vacuole structures (see below).
Bone sialoprotein detected by the immunogold antibody was present mostly in the mineralized tissue (Figure 4A). At higher magnification, the gold labeling appeared associated with the clusters of needle-like crystals but not with collagen fibers, as demonstrated by the lack of gold particles in unmineralized collagen fibers located in the matrix (Figure 4B). This observation was further strengthened by the presence of gold particles in mineralization foci where collagen fibers were absent (Figure 4C). The gold particles were usually associated with electron-dense granular and amorphous extrafibrillar networks. However, BSP was not observed on matrix vesicles (inset, Figure 4C). Finally, BSP labeling appeared quite homogeneously distributed in the electron-dense areas of the mineralized matrix, with the exception of some very limited areas in which patches of gold particles were observed (Figure 4D).

Osteonectin antibody was also mostly localized to the mineralized matrix. However, unlike the BSP distribution, the osteoid matrix was homogeneously and clearly labeled (Figures 5A and 5B). Osteoblasts and osteocytes were occasionally very weakly labeled.
Osteocalcin labeling was more diffuse and less intense than the reaction observed after incubation with BSP and ON antibodies. The heaviest labeling was located in the mineralized matrix, with more intense labeling of the mineralization front (Figure 5C). Some grains were also noted in the osteoid and in the intracellular compartment as singles or in patches of two or three particles associated with vesicles (Figure 5D). These results of the localization of NCPs at the OM and EM levels are summarized in Table 1.
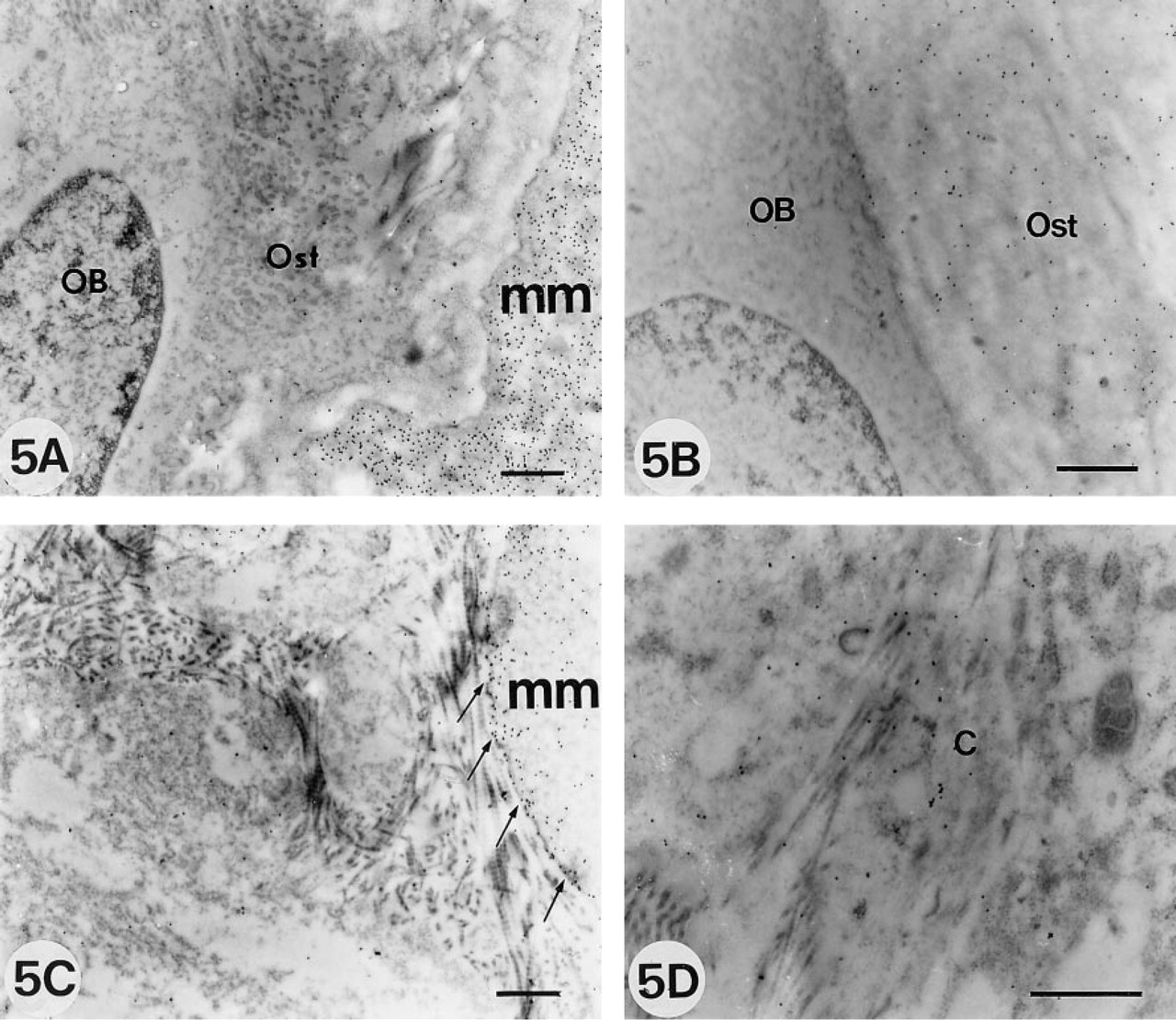
Electron micrographs showing immunolabeling with ON
Discussion
Expression of ALP, Type I collagen, and non-collagen proteins (NCPs), osteocalcin (OC), and osteonectin (ON), and bone sialoprotein (BSP) was investigated during bone nodule formation by differentiating calvaria cells. This calvaria nodule cell system is an ongoing process of osteoblast differentiation in which sequential morphological steps have been previously defined. However, these steps are repeated around the nodule as long as it is developing and occur concomi- tantly, but at different locations, during the bone nodule development (Nefussi et al. 1993). These steps therefore correspond to cellular sequences that occur before and during bone matrix formation but do not correlate with the actual lifetime development of a defined nodule.
Sequential expression of non-collageneous proteins during in vitro differentiation and nodule formation by rat calvaria cells
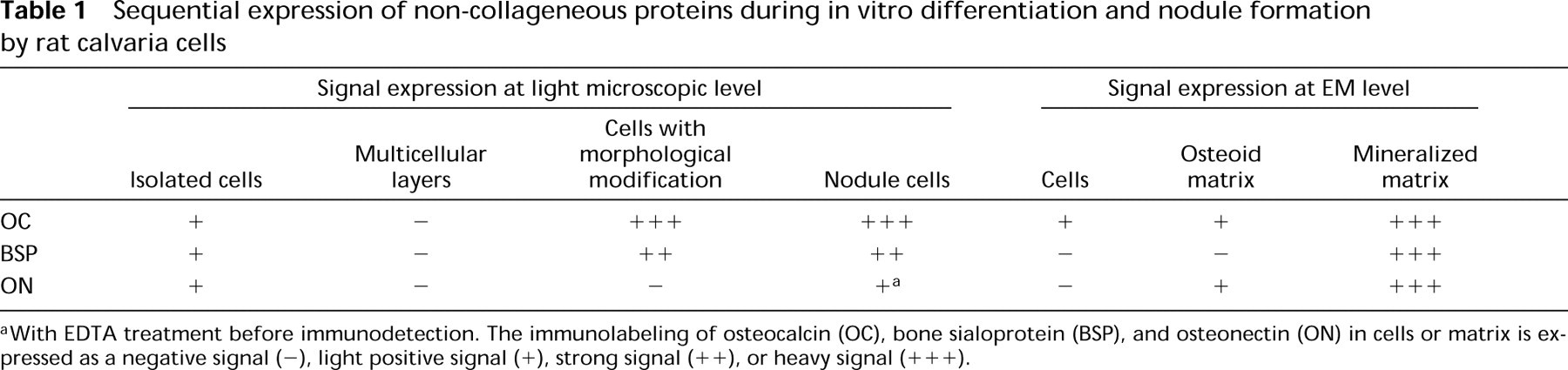
With EDTA treatment before immunodetection. The immunolabeling of osteocalcin (OC), bone sialoprotein (BSP), and osteonectin (ON) in cells or matrix is expressed as a negative signal (-), light positive signal (+), strong signal (++), or heavy signal (+++).
Expression of bone collagen and non-collagen proteins, as well as ALP activity, was observed in rat calvaria cells in the first 24 hr after cell plating. Various ALP activity was observed in proliferative (BrdU+) or nonproliferative (BrdU—) cells. This variable intensity of the reaction may be due to cell heterogeneity reflecting differences in cell maturation or activity (Heersche et al. 1985,1992; Turksen and Aubin 1991; Marks and Popoff 1988; Otawara and Price 1986). Because ALP expression appears critical for the “transition–restriction point” during the differentiation pathway in this in vitro system (Stein et al. 1990), the distribution of ALP-positive cells in the culture system may be of importance in determining the localization of future nodules.
During the next step, cell proliferation results in the formation of multicellular layers in which ALP and NCP were not expressed until morphological cell changes associated with osteoblast differentiation were noted. The presence of some ALP+-labeled cells in the first 24 hr and the disappearance of such labeling during this proliferation phase may be interpreted as a transient dedifferentiation state. During such a phase, cell activities would be more involved in metabolism related to proliferation to create a three-dimensional microenvironment, secondarily allowing cell differentiation expression. A few arguments are in favor of this interpretation: (a) Cell dedifferentiation after plating appears to be a common process in culture systems, as it has been reported in many other models before differentiation occurred (Sautier et al. 1993; Von der Mark et al. 1977). (b) Biochemical studies in a very similar system have demonstrated a time-dependent decrease in ON concentration in the medium in the first 24 hr of culture (Kasugai et al. 1992) and in confluent cells (Wrana et al. 1988). Similar observations have also been reported for other non-collagen proteins, such as osteopontin (Wrana et al. 1991; Gerstenfeld et al. 1990).
At the end of this proliferation stage when a permissive microenvironment was created by a three-dimensional cell organization, cells characterized by morphological modifications and ALP activity did express NCPs. Similar observations were previously reported for ALP and NCPs in total rat calvaria cultures (Pockwinse et al. 1992; Owen et al. 1991; Aronow et al. 1990; Stein et al. 1990; Bronkers et al. 1987), but no studies, to our knowledge, have reported the cellular localization and identification of the cells synthesizing these NCPs. The present results agree with in vivo studies showing the presence of BSP mRNA expression at this stage (Chen et al. 1992). Similar results showing NCP synthesis restricted to cells with morphological changes have been reported in rat stromal cells forming nodules (Malaval et al. 1994).
The next step after the osteoblast differentiation process is characterized by the active synthesis of matrix by the cells. Many sections through such areas of matrix formation showed that it was not the surface cells that were synthesizing the matrix but rather the cells underneath, allowing cell proliferation in the layers localized between the surface cells and the active osteoblasts cells secreting the bone matrix (Nefussi et al. 1993). We found that only the active osteoblastic cell layers were expressing OC. Such observations agree with in vivo studies (McKee et al. 1992; Bronkers et al. 1987; Groot et al. 1986) and with an in situ hybridization study of osteocalcin mRNA (Heersche et al. 1992) demonstrating that only functional osteoblasts synthetize osteocalcin. BSP and ON are also expressed during this step, but it is not clear from the present results which cells (i.e., preosteoblasts and/or osteoblasts) expressed these proteins. When EDTA was used, the matrix, which was previously negative, appeared positive. However, by light microscopy we could not determine whether the osteocytes were positively stained for these proteins.
EM investigations on the distribution of BSP, OC, and ON demonstrated a reproducible heavy labeling of these proteins in the mineralized tissue, whereas faint or absent labeling was found in the osteoid and in the cellular compartment. Most in vivo studies have reported similar localization of these NCPs (Sodek et al. 1992; Chen et al. 1991a,b; Boivin et al. 1990; Bianco et al. 1989; Schultz and Jundt 1989; Vermeulen et al. 1989; Bronkers et al. 1987; Camarda et al. 1987; Groot et al. 1986), with inconstant labeling of the mineralization front. Technical and biological reasons can be proposed for this heavy labeling of the mineralized tissue. First, the affinity of these proteins for the hydroxyapatite and the presence of the mineral as a stabilizing element of non-collagen proteins may account for these observations. Second, the osteoid matrix is likely to be more easily and completely penetrated by resin, and therefore linked to it, than the mineralized tissue (Bianco et al. 1985). It is also possible that the relative faint labeling in osteoid may be related to the fact that part of the proteins released by cells are degraded in the extracellular compartment unless they are stabilized in a defined conformation with other proteins or mineral elements in the calcified matrix.
The observation of BSP labeling in the osteoid is restricted to areas at which crystallites are present (Figure 4C), suggesting that when all the elements are present in adequate conformation, mineral crystal will form. Interestingly, either in the osteoid or in mineralized matrix, BSP labeling was associated with amorphous, electron-dense, granular extrafibrillar materials but never with collagen fibers. Such in vitro observations correlate well within vivo studies (Chen et al. 1994; Hultenby et al. 1994; McKee et al. 1993) and in vitro biochemical data (Nagata et al. 1991). BSP has been found previously to be concentrated on spherical aggregates corresponding to the sites of early mineralization (Bianco et al. 1993; Sodek et al. 1992). This confirmed previous observations performed in this model with histochemical dyes, which identified these extrafibrillar elements in bone as proteoglycan substances associated with lipids (Nefussi et al. 1989b,1992). Similar observations have been reported in other mineralized tissues (Chardin et al. 1990; Shepard and Mitchell 1986). Such elements associated with noncollagen proteins could be epitactive nucleation structures involved in the mineralization process, as suggested previously by Bonucci and colleagues (1987,1988).
The very low or absence of labeling for NCPs noted in osteoblastic cells may be explained by the rapid excretion of the proteins after synthesis. The labeling might be seen only in few protein synthetic organelles such as the Golgi apparatus, where the proteins accumulate before excretion. Similar observations have been reported in in vivo studies (Hultenby et al. 1994; Bianco et al. 1993; McKee et al. 1992) and this can be explained either by very low amounts of protein synthetized by the cells (as all NCPs are not major proteins in the osteoid matrix compared to the collagen element) and/or, as argued previously, a possible difference between the antigenic properties of the immature intracellular proteins compared to their mature extracellular forms. Finally, the differences noted between immunolabeling intensities with the different antibodies in osteoid and in mineralized matrix can be associated with the titers of the antibodies used, which were different and would therefore not directly reflect quantitative protein differences.
In summary, the present study shows that the sequential expression of NCPs during in vitro formation of nodules is similar to the known protein expression during in vivo osteogenesis. We found that OC and BSP were initially expressed during OB differentiation, whereas ON appeared to be associated only with the formation of the matrix. Although OC, BSP, and ON were associated with the mineralization process, various density distributions are observed between all these proteins. BSP is associated only with crystal needles, whereas ON and OC are found in both the osteoid and in the mineralized matrix. Finally, OC appears to be the only protein associated with the mineralization front.
Footnotes
Acknowledgements
This work was supported by INSERM Grant C143 and the Foundation Dentaire de France.
We would like to thank Dr L.W. Fischer (NIDR, NIH, Bethesda) for the generous gift of antibodies against bone matrix proteins, Dr P.J. Marie for helpful discussions and revision of the manuscript, and E. Marie–Rose for secretarial assistance.
