Abstract
Cathepsin L, a lysosomal cysteine proteinase, may have a key role in various biological and disease processes by intracellular and extracellular degradation of proteins. We examined the levels of cathepsin L and its intrinsic inhibitors in glomeruli of rats with puromycin aminonucleoside (PAN) nephrosis. In contrast to the weak levels of cathepsin L in normal glomeruli, on days 4 and 8, strong immunostaining was detected in almost all podocytes when proteinuria and pathological changes of the podocytes developed. Cathepsin L was reduced after day 28, but remained in a focal and segmental manner. Cystatin β, an intracellular inhibitor, was not detected in podocytes. However, cystatin C, an extracellular inhibitor, was detected in podocytes after day 4, coincident with cathepsin L. Cystatin C levels were gradually reduced but sustained in many podocytes on day 28, while cystatin C was not detected in podocytes sustained cathepsin L. These results demonstrated that cathepsin L levels are not always accompanied by the levels of its inhibitors in podocytes of PAN nephrosis, suggesting a potential role of cathepsin L in podocyte injury, which is a critical process for the development and progression of tuft adhesion and sclerosis.
Introduction
Podocytes constitute an essential part of filtration barrier to urinary protein loss. They are terminally differentiated epithelial cells incapable of replicating in glomeruli. Therefore, progressive podocyte loss is the most frequent cause of chronic kidney diseases. Reportedly, podocyte loss underlies the progression of glomerulosclerosis in several human glomerular diseases as well as in experimental animal models of glomerular diseases regardless of the basal diseases.1–4 The major cause of podocyte loss appears to be the detachment from the glomerular basement membrane (GBM), which leads to bare GBM and tuft adhesion to the Bowman’s capsule, leading to glomerulosclerosis. 5 The degradation of GBM by proteinases has been considered to be one of the molecular mechanisms for podocyte detachment. 6
Cathepsin L, a papain family cysteine proteinase, is a potent endopeptidase that is involved in the degradation of lysosomal proteins. Using various cultured cells, cathepsin L has been demonstrated to be translated initially as preprocathepsin L, which then is transferred through the Golgi apparatus as procathepsin L and stored in lysosome as mature cathepsin L.7–9 This enzyme is different from other members of cysteine proteinases in that procathepsin L is secreted abundantly into extracellular space after stimulation by various growth factors10,11 and activated by oncogenes,12,13 resulting in degradation of the extracellular matrix. 14 Through such intracellular and extracellular degradation of proteins, cathepsin L is implicated in diverse biological and disease processes, including bone turnover, 15 muscle degeneration, 16 atherosclerosis, 17 and cancer metastasis. 18 Server et al. 19 have demonstrated that cathepsin L is essential for proteinuria and that cathepsin L levels are significantly elevated in human proteinuric kidney diseases, ranging from minimal change disease (MCD) to diabetic nephropathy. In glomeruli, the activity of cysteine proteinase was detected using freshly isolated glomeruli, 6 and cathepsin L appeared to degrade GBM, 20 dynamin, 19 and synaptopodin 21 in experimental glomerulonephritis, resulting in podocyte actin reorganization and proteinuria. This argument is supported by the facts that isolated podocytes from cathepsin L-deficient mice are protected from cell puromycin aminonucleoside (PAN)-induced cell detachment 22 and cathepsin L-deficient mice do not suffer from albuminuria. 23 In addition, we found that cathepsin L is regulated in the glomeruli of rats with experimental nephrotic syndrome using differential display polymerase chain reactions, immunostaining and Western blotting. 22 Thus, it is highly likely that cathepsin L may participate in the disease processes of the glomeruli.
Proteinase inhibitors also have a pivotal role in biological and pathological processes by regulating proteinase activities intracellularly and extracellularly. Endogenous inhibitors of cathepsins are cystatins classified into three families as follows: intracellular cystatins A and B (family 1); extracellular cystatins C, D, S, SA, and SN (family 2); and intravascular kininogens (family 3). 24 Cystatin A expression is rather restricted, with especially high levels in skin and some blood cells, 25 whereas cystatin B (cystatin β in rats) expression is localized in the cytosol, mitochondria, and nucleus in various cells and protects cells from the detrimental release of the lysosomal cysteine cathepsins. In the nucleus, cystatin B interacts with nucleosomes, specifically with histones H2A.Z, H2B, and H3 and cathepsin L. 26 Cystatin C is expressed in virtually all organs of the body in contrast with the restricted expression of other family 2 cystatins in salivary glands. The ubiquitous distribution of cystatins B and C indicates that cathepsins are regulated intracellularly by cystatin B and extracellularly by cystatin C. We previously demonstrated that tumor growth factor β-1 enhances the secretion of cystatin C and the imbalance of the secretion of proteinases and their inhibitors after incubation of such growth factors. 27
Regarding PAN nephrosis, the intraperitoneal injection of PAN to rats is an experimental model characterized by massive proteinuria and by morphological changes in podocytes, including foot process effacement, focal adhesion with the Bowman’s capsules, and focal detachment form GBM.28,29 By the fourth week after PAN administration, the level of protein excretion and morphological changes of almost all podocytes were indistinguishable from those of control animals. 30 Thus, PAN nephrosis has been conventionally used as a similar model of MCD in humans. However, from a clinical point of view, MCD does not show such podocyte change without foot process effacement. Thus, we used this model as a podocyte injury model.
The present study evaluated the levels of cathepsin L and cystatins β and C in glomeruli of rats with PAN nephrosis to address whether cathepsin L and its inhibitors have an important role in disease processes of the glomeruli.
Material and Methods
Animals
Male Sprague-Dawley rats (weighing approximately 250 g) were obtained from Charles River Japan (Kanagawa, Japan). These rats were housed under specific pathogen-free conditions. All animal experiments were performed according to the guidelines of the Committee on Animal Experiments of Juntendo University, Tokyo, Japan.
Induction of PAN Nephrosis
PAN nephrosis was induced by a single intraperitoneal injection of PAN (Sigma, St. Louis, MO) dissolved in normal saline at a dose of 15 mg/100 g body weight. Control rats received a similar volume of normal saline. Six experimental groups with animals sacrificed 2, 4, 8, 28, and 56 days after the injection were studied. Control rats were sacrificed eight days after the injection of normal saline. Each group consisted of four rats. Before sacrifice, 24 hr urine was collected. Furthermore, blood was collected to determine serum creatinine. Urinary total protein and creatinine concentration were measured by the same methods as clinical examination.
Fixation of Kidneys
After the rats were anesthetized with pentobarbital (100 mg/kg; Dainippon Sumitomo Pharma, Osaka, Japan), the right kidneys were removed, and then blood samples were collected from the abdominal aorta. Finally, the left kidneys were perfused via the abdominal aorta for 5 min with 4% paraformaldehyde (PFA) in 0.1 mol/L phosphate buffer (PB). After perfusion with 4% PFA, a small amount of the inferior pole was sectioned for electron microscopy. The remaining left kidneys were perfused for 5 min with 18% sucrose in 0.01 mol/L sodium phosphate buffer (PBS).
Light and Electron Microscopy
For electron microscopy, slices of the left kidneys were immersed in fixative containing 2% glutaraldehyde in 0.1 mol/L PB (pH 7.4) overnight. Then, tissues were processed for epoxy resin embedding. For light microscopy, semithin sections (1 μm thick) stained with Periodic Acid-Schiff (PAS) were used. Ultrathin sections stained with uranyl acetate and lead citrate were observed in a Hitachi H7100 electron microscope (Hitachi, Tokyo, Japan).
Immunohistochemistry
Deparaffinized sections were used for double-immunostaining. Primary and secondary antibodies (Abs) are listed in Table 1. We checked that these secondary Abs did not stain without primary Abs (data not shown). After blocking with PBS containing 2% bovine serum albumin, 2% fetal calf serum, and 0.2% fish gelatin, sections were incubated with primary Abs at each appropriate condition, and secondary Abs in blocking solution were reacted. Images were acquired with an FV1000 confocal microscope (Olympus, Tokyo, Japan).
Primary Antibodies for Immunohistochemistry.

Glomerular Isolation and Western Blot
Rat glomeruli were isolated by progressive filtering using 212, 106, 75 μm sieves. For glomerular extraction, glomeruli were suspended in 3-[(3-cholamidopropyl)dimethylammonio]-1-propanesulfonate buffer and sonicated in short bursts in 1 min. After centrifugation, the supernatant was stored at −80C. Samples of glomerular extracts were analyzed by gradient gel (5–20% sodium dodecyl sulfate-polyacrylamide gel) and Western blotting under reducing condition.
As primary Abs, mouse monoclonal to cathepsin L (1:200; Santa Cruz Biotechnology, Inc., Dallas, TX), rabbit polyclonal to cystatin C (1:200; Merck Millipore, Darmstadt, Germany), and goat polyclonal to cystatin β (1:500; R&D systems Inc., Minneapolis, MN) were used. As second Abs, peroxidase conjugated affinity-purified goat antimouse IgG, peroxidase conjugated affinity-purified goat antirabbit IgG, and peroxidase conjugated affinity-purified donkey antigoat IgG were used (1:10,000; Jackson Immunoresearch, West Grove, PA). Equal protein load was confirmed by reproving the membrane with glyeraldehyde-3-phosphate dehydrogenase (GAPDH; 1:20,000; Sigma-Aldrich Corp., St. Louis, MO). A549 whole cell lysate (Novus Biologicals, LLC, CO) was used as a positive control of cystatin β.
Statistical Analysis
All values are given as mean ± SD. Statistical significance (defined as p<0.05) was evaluated using Prism 6.0 software (GraphPad Software, San Diego, CA) followed by Fisher’s paired least significant difference t-test.
Results
PAN Rats Showed Severe Proteinuria Compared With Control Rats
No elevation in urinary protein excretion was observed on day 2 after administration of PAN (7.57 ± 1.31 mg/day) with respect to the excretion in controls (3.73 ± 2.25 mg/day). Urinary protein excretion significantly increased on day 4 (401.0 ± 24.26 mg/day, p<0.0001 vs. control; Fig. 1A). Maximum proteinuria was observed on day 8 (521.0 ± 99.64 mg/day, p<0.001 vs. control; Fig. 1A). Thereafter, urinary protein excretion decreased on days 28 (75.97 ± 66.55 mg/day) and 56 (42.48 ± 21.42 mg/day; Fig. 1A).
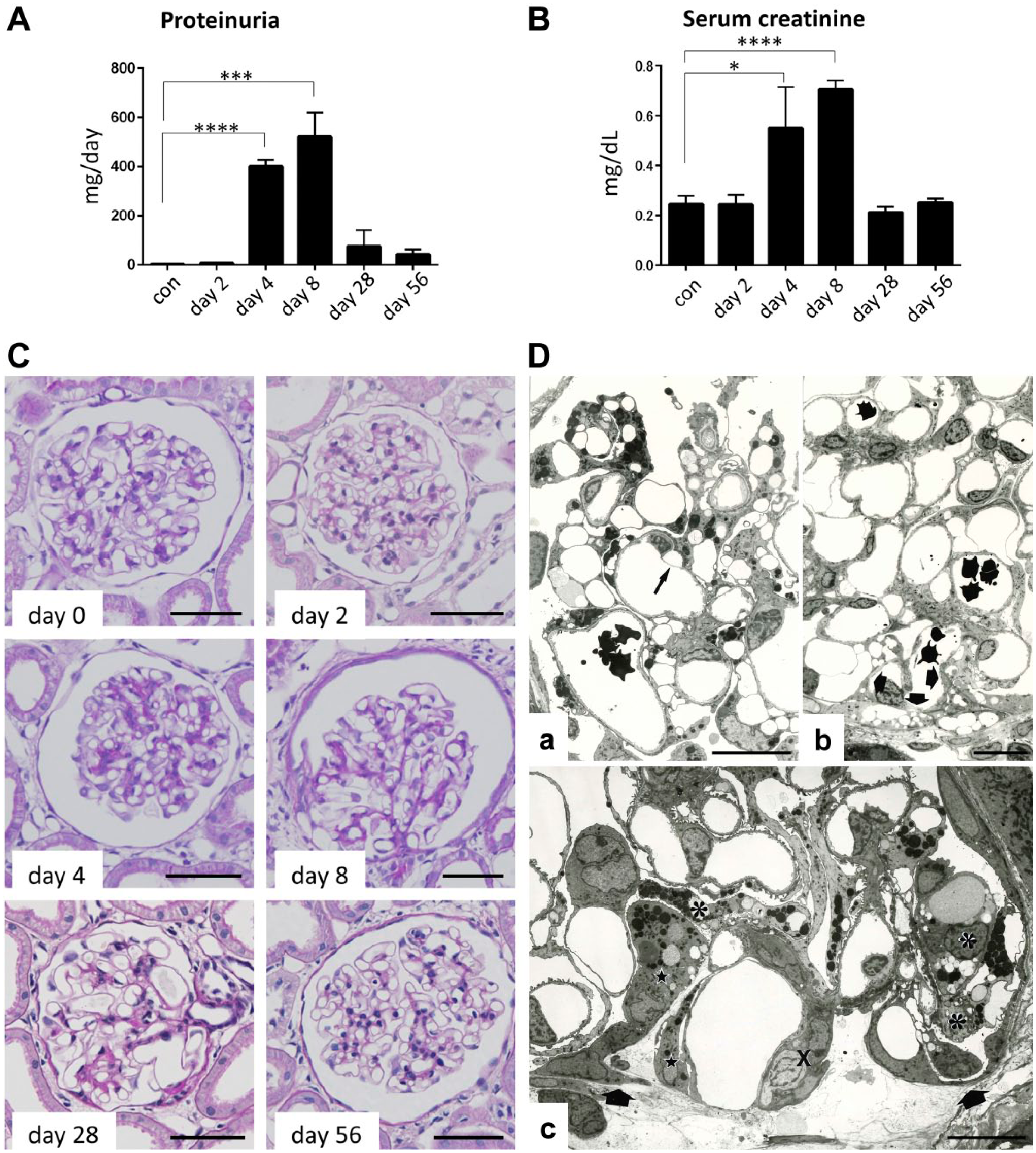
Proteinuria and serum creatinine levels in PAN rats and morphological analysis. After PAN administration, urinary protein excretion increased significantly on day 4 (401.0 ± 24.26 mg/day; ****p<0.0001) and increased to peak on day 8 (521.0 ± 99.64 mg/day; ***p<0.001) compared with controls (3.73 ± 2.25 mg/day; A). Serum creatinine level increased significantly similar to proteinuria on day 4 (0.55 ± 0.17 mg/dL; *p<0.05) and maximally on day 8 (0.71 ± 0.04 mg/dL; ****p<0.0001; B). On light microscopy, on days 8 and 28, glomerular adhesion was shown in glomeruli (C). On day 56, glomerular adhesion was reduced (C). On electron microscopy, in a glomerulus on day 4 of PAN nephrosis, podocytes are affected diffusely. Partial detachment from GBM is observed in one podocyte (Da: arrow). In a glomerulus on day 8 of PAN nephrosis, pseudocyst formation is prominent in podocytes (Db). Lysosome accumulation is hardly detectable in the podocytes. At the peripheral glomerular tuft, the denuded surface of GBM is covered by partial epithelial cells, forming an early stage of adhesion (Db: thick arrows). In a glomerulus on day 28 of PAN nephrosis, the expansion of adhesion (Dc: between arrows) is observed, associated with a collapsed capillary loop (Dc: X). Podocytes (Dc: asterisks) and parietal epithelial cells (Dc: stars) covering the flank of the adhesion contain many lysosomes. Scale bars: (C) 50 µm and (D) 10 µm. Abbreviations: PAN, puromycin aminonucleoside; GBM, glomerular basement membrane.
PAN Rats Showed Transient Renal Failure With Serum Creatinine Elevation
After administration of PAN, the serum creatinine level increased with the increase in urinary protein excretion. No elevation in serum creatinine levels was observed on day 2 (0.24 ± 0.04 mg/dL) with respect to the levels in controls (0.25 ± 0.03 mg/dL; Fig. 1B). Serum creatinine levels increased on day 4 (0.55 ± 0.17 mg/dL, p<0.05 vs. control; Fig. 1B). The highest creatinine level was observed on day 8 (0.71 ± 0.04 mg/dL, p<0.0001 vs. control; Fig. 1B). Thereafter, serum creatinine levels decreased on days 28 (0.21 ± 0.02 mg/dL) and 56 (0.25 ± 0.015 mg/dL; Fig. 1B).
Glomerular Injury Was Recognized in PAN Rats
Using light microscopy, focal and segmental glomerular adhesions were noted in glomeruli on days 8 and 28. On day 56, the glomerular adhesion had reduced. No cell proliferation was observed (Fig. 1C).
Using electron microscopy, the main glomerular lesions of PAN nephrosis were observed in the podocytes. On day 4 when proteinuria developed, podocytes were diffusely affected, exhibiting loss of foot processes, formation of pseudocyst, accumulation of lysosome, and partial detachment of podocytes from GBM (Fig. 1Da). On day 8 when maximum proteinuria was achieved, pseudocyst formation, a prominent change, was observed in the podocytes (Fig. 1Db). Compared with day 4, accumulation of lysosomes was encountered less frequently but detachment of podocytes from GBM was observed much more frequently on day 8. Severe podocyte detachment led to complete bareness of GBM surface, a part of which was covered by parietal epithelial cells of the Bowman’s capsules (Fig. 1Db). The incidence of early adhesion in the semithin sections of 50 consecutive glomerular profiles ranged from 5% to 15% in individual rats. On days 28 and 56, progression of and recovery from podocyte damage were concurrently observed. In other words, glomerular adhesions had advanced to segmental sclerosis associated with progressive podocyte detachment. Podocytes and parietal epithelial cells covering the sclerotic lesions contained many lysosomes (Fig. 1Dc), and on the contrary, recovery of the foot process pattern was observed adjacent to the sclerotic areas even in the same glomeruli.
Cathepsin L Level Did Not Correspond With Cystatin β Level
In the glomeruli of control rats, virtually, cathepsin L and cystatin β were not observed (Fig. 2A). In the glomeruli of PAN rats, potent cathepsin L level was observed. On day 4, the distinct presence of cathepsin L was diffusely observed in the glomeruli as granules of various sizes, which resembled lysosomes. On day 8, intense granular staining was observed the most in the glomeruli (Fig. 2A). Cathepsin L and synaptopodin were merged, which indicated that cathepsin L emerged in podocytes (Fig. 3). On day 28, immunostaining for cathepsin L was weakly scattered along the glomerular capillary walls. However, cathepsin L was sustained in a focal and segmental manner on days 28 and 56 (Fig. 2B and D). Positive immunostaining for cathepsin L was detected in cells located on the glomerular tuft surface adhering to the Bowman’s capsules (Fig. 2B). The cells were regarded as podocytes based on their location, shape, and positive immunostaining for synaptopodin (Fig. 2B). Positive immunostaining for cathepsin L was also detected in the Bowman’s capsule (Fig. 2D), which were frequently attached to the glomerular tufts with their processes. These cells were negative for podocyte markers and thus regarded as parietal epithelial cells (Fig. 2D).
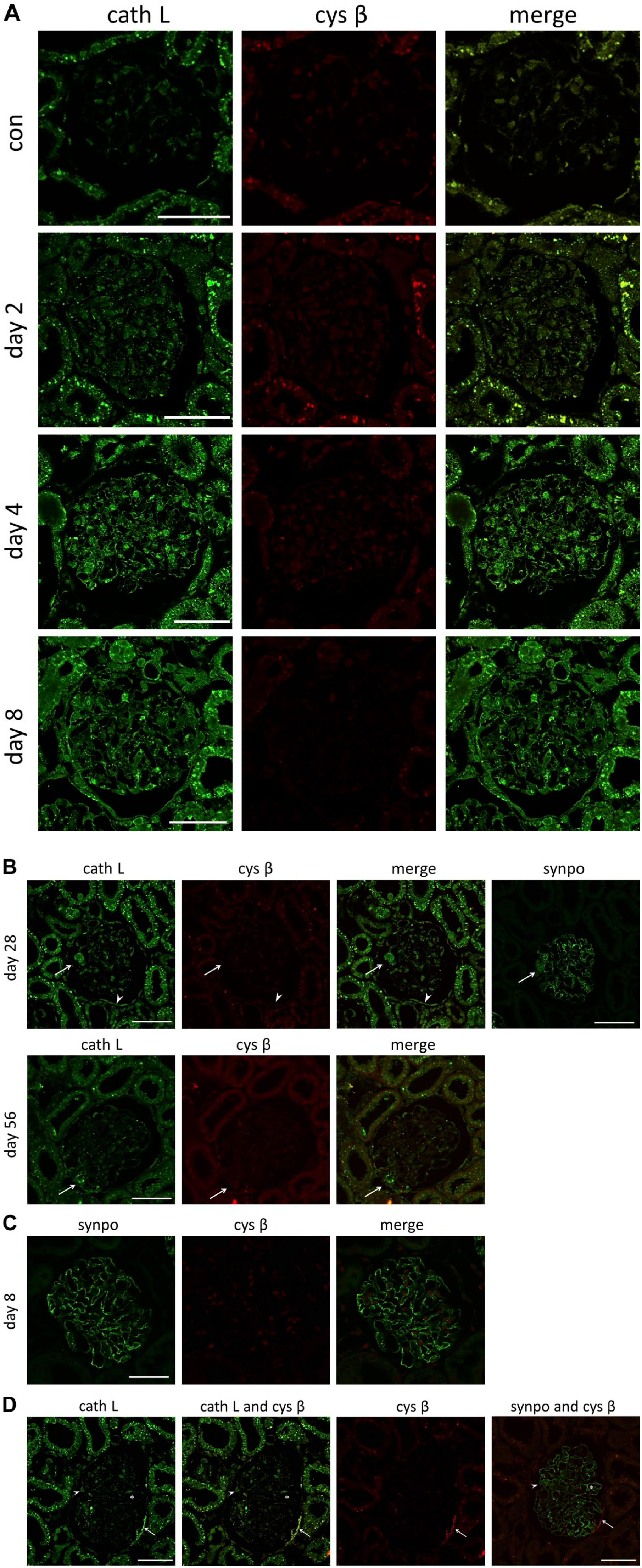
Immunohistochemical levels of cath L and cys β in PAN rats. Cys β was observed mainly in parietal cells that were positive for cath L in PAN rats (A–D). In a glomerulus on day 28 of PAN nephrosis, cath L is observed in podocytes (B: arrow) and parietal epithelial cells (B: arrowhead). Cys β is not observed in the podocytes (B: arrow), but it is positive in parietal epithelial cells (B: arrowhead). In a glomerulus on day 56 of PAN nephrosis, segmental level of cath L is observed. Intensive level is observed in cells along the capillary loop (B: arrow), and cys β is negative for cells along the capillary loop (B: arrow). Cys β-positive cells are not merged with synpo in immunofluorescent staining (C). Cells with sustained cath L and/or cys β levels identified using synpo as a podocyte marker in serially sectioned glomeruli on day 28 of PAN nephrosis are shown (D). Two types of cells can be identified by immunoreactivity for cath L and cys β (D). One type of cell (D: arrowhead and asterisk), which is positive only for cath L, is positive for synpo. Another type of cell (D: arrow), which shows positive immunoreactivity for cath L and cys β, is negative for synpo. Scale bars: (A–D) 50 µm. Abbreviations: cath L, cathepsin L; cys β, cystatin β; synpo, synaptopodin; PAN, puromycin aminonucleoside.
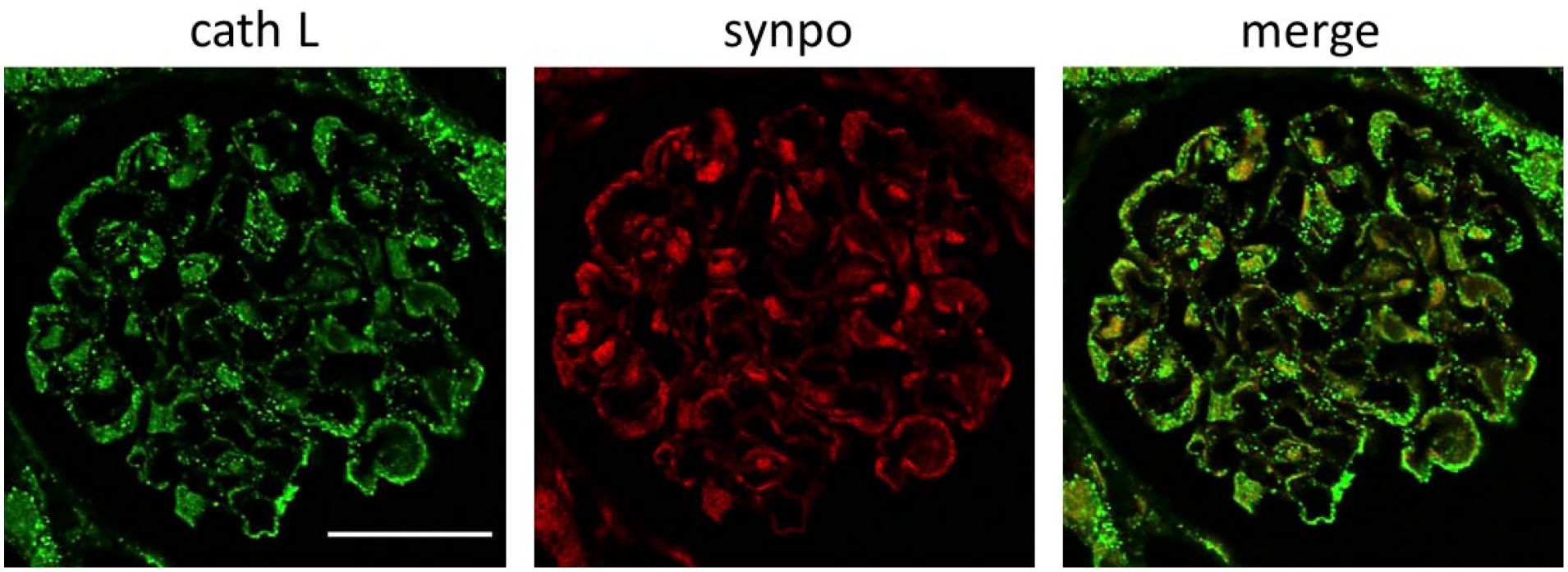
Immunohistochemical levels of cath L and synpo on day 8. To identify the cells that sustained cath L in the PAN rat model, we performed immunostaining with cath L (green) and synpo (red) on day 8. Cath L and synpo merged well, suggesting that podocytes sustained cath L in the PAN rat model. Scale bar: 50 µm. Abbreviations: cath L, cathepsin L; synpo, synaptopodin; PAN, puromycin aminonucleoside.
In contrast to cathepsin L, cystatin β was not detected in the podocytes in this study (Fig. 2A–C). On day 28, cystatin β was partially observed in some parietal epithelial cells on the Bowman’s capsule, which were positive for cathepsin L (Fig. 2D). These cells were negative for synaptopodin, indicating that they were not podocytes. Western blotting revealed slight cystatin β level in isolated glomeruli in every phase (Fig. 4F). Cystatin β level did not obviously correspond with cathepsin L level.
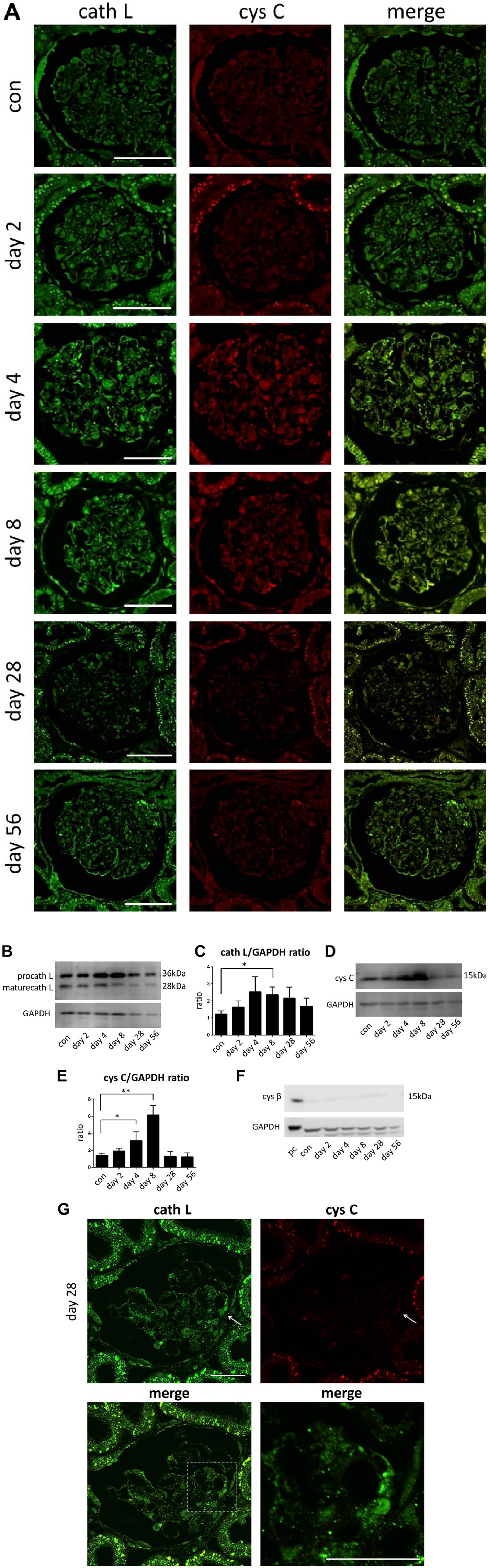
Immunohistochemical levels of cath L and cys C proteins in PAN rats. Immunohistochemical levels of cath L and cys C were analyzed in a control glomerulus. Cath L and cys C are weakly detected before PAN administration (A). In a PAN nephrosis glomerulus, cath L is observed diffusely on day 4 (A). In Western blotting analysis, the band of procath L was detected in 36 kDa (B). The band of mature cathepsin L was detected in 28 kDa (B). The levels were measured by comparison with the levels of GAPDH. On day 8, cath L was significantly increased (*p<0.05; C). In Western blotting analysis, the band of cys C was detected in 15 kDa (D). The levels were measured by comparison with the levels of GAPDH. On day 8, the level of cys C was significantly increased (**p<0.01; E). Cys β was observed at a very slight level in Western blotting analysis (F). We used A549 whole cell lysate as a positive control of cystatin β. Intense level of cystatin C is detected in several podocytes on day 28 of PAN nephrosis (G). However, cys C is not detected in podocytes around the site of adhesion, which sustained cath L (G: arrow). pc: positive control, Scale bar: (A) 50 µm and (G) 30 µm. Abbreviations: cath L, cathepsin L; cys C, cystatin C; procath L, procathepsin L; PAN, puromycin aminonucleoside; GAPDH, glyeraldehyde-3-phosphate dehydrogenase.
Cystatin C Level Corresponded With Cathepsin L Level in PAN Rats
On immunofluorescence staining on day 4 when cathepsin L was diffusely observed in the podocytes, cystatin C was weakly observed. On day 8 when the level of cathepsin L was maximal, cystatin C was also observed in almost all the podocytes (Fig. 4A). We collected isolated glomeruli and measured cathepsin L and cystatin C levels by Western blotting in each phase. Total cathepsin L level was significantly increased on day 8 (p<0.05 vs. control; Fig. 4B and C). Cystatin C level was also significantly increased on day 8 (p<0.01 vs. control Fig. 4D and E). In the chronic phase of PAN nephrosis (days 28 and 56), cystatin C was detected in fewer podocytes (Fig. 4A). However, small numbers of podocytes, which were positive for cathepsin L and situated on the surface of the glomerular tufts adhering to the Bowman’s capsules, were not positive for cystatin C (Fig. 4G).
Cystatin C and Cathepsin L Levels Were Observed in Proximal Tubules on Day 2 in PAN Rats
In control rats, cathepsin L, but not cystatin C, was observed in proximal tubules (Fig. 5A). However, on day 2, both cystatin C and cathepsin L were observed in proximal tubules. This level was expanded to distal tubules on day 8, and the conjugated proteins of cystatin C and cathepsin L were detected in the urinary space of the Bowman’s capsule and tubules (Fig. 5B).
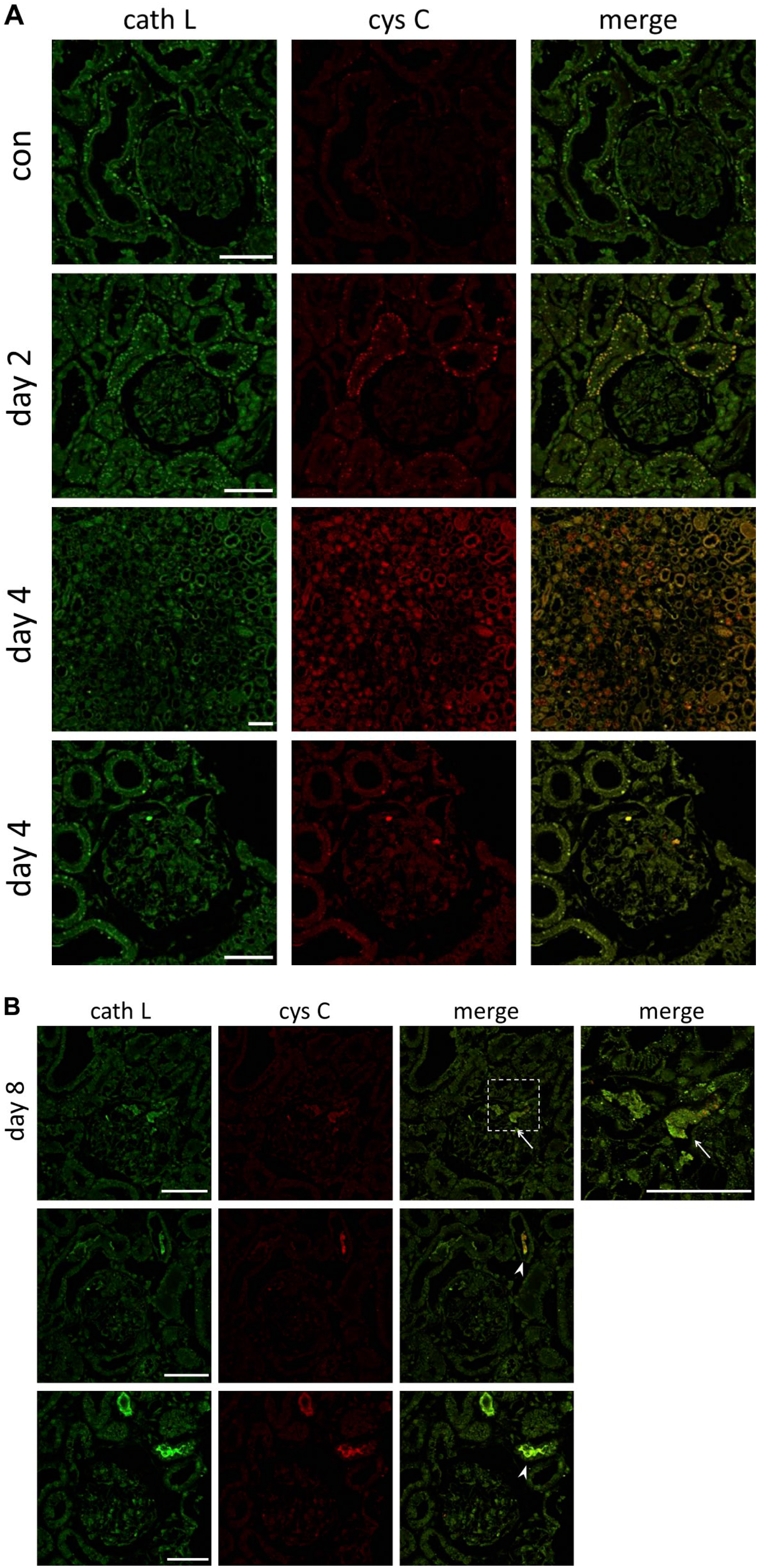
Cath L and cys C were observed in proximal tubules in PAN nephrosis rats. In proximal tubules, cath L was observed in control rats. However, cys C was not observed in control rats but after day 2 (A). It was expanded to the distal tubules on day 8, and they were conjugated with cath L in the urinary space in the Bowman’s capsule (B arrow) and tubules (B arrowhead). Scale bar: (A, B) 50 µm. Abbreviations: cath L, cathepsin L; cys C, cystatin C; PAN, puromycin aminonucleoside.
Discussion
The present study demonstrated the level of cathepsin L and its inhibitors, cystatins, in the glomeruli of rats with PAN nephrosis and revealed that the imbalance between cathepsin L and its inhibitors, cystatins, may accelerate podocyte damage, detachment from GBM and adhesion to the Bowman’s capsule, eventually leading to glomerular sclerosis.
Cathepsin L was observed diffusely in podocytes on day 8, coinciding with the development of proteinuria and podocyte damage including foot process effacement, pseudocyst formation, accumulation of lysosomes, and partial detachment from GBM. On days 28 and 56, cathepsin L was observed focally and segmentally in podocytes located around glomerular tufts adhering to the Bowman’s capsules. In a part of adhesion, podocyte detachment and bare GBM were observed on electron microscopy. The almost identical distribution between cathepsin L and damaged podocytes suggested the relevance of cathepsin L in cellular damage of podocytes, which was confirmed morphologically to be a critical step in the development and progression of glomerular tuft adhesion and sclerosis.
The relevance of cathepsin L to podocyte damage also was supported by the evidence from immunohistochemistry and Western blotting that intracellular inhibitors of cathepsin L were not detected and extracellular inhibitors were weakly detected compared with cathepsin L in podocytes. Considering these results, we hypothesized that the imbalance levels of cathepsin L and its inhibitors in podocytes contribute to the pathophysiological mechanisms of glomerular injuries of PAN nephrosis, including the cellular damage to podocytes.
Cathepsin L has been known as a lysosomal cysteine proteinase and has an amino acid sequence belonging to the papain family. This enzyme is different from other mammalian papain family cysteine proteinases in extracellular function.31,32 After induction of mRNA by various factors, high levels of procathepsin L were secreted into the extracellular space, where procathepsin L and its processed forms degrade extracellular matrix proteins in the presence of glycosaminoglycan. 14 By this extracellular function, cathepsin L contributes to remodeling of the extracellular matrix, which has an important role in cellular function and various disease processes. In vitro studies using isolated glomeruli demonstrated that endogenous cathepsin L effectively digests GBM and its component proteins, including type IV collagen and laminin.6,33,34 Thus, cathepsin L in podocytes may be responsible for degradation of GBM proteins, inducing increased permeability of GBM and impairment of the connection between podocytes and GBM, finally resulting in proteinuria and podocyte detachment. Furthermore, cathepsin L degrades the proteins that are important for normal podocyte architecture, such as the CD2-associated protein, 35 synaptopodin, 21 and dynamin. 19 In PAN nephrosis, cathepsin L was upregulated in podocytes both in vivo and in vitro. 22 This is consistent with our observation that increases in the amount of proteinuria and augmentation of podocyte detachment on day 8 were continued by diffuse expression of cathepsin L on day 4, coinciding with the protein level of cathepsin L. Progressive podocyte detachment from GBM around the site of adhesion on days 28 and 56 was associated with cathepsin L.
In addition, it was reported that administration of a cysteine proteinase inhibitor with a high degree of selectivity for cathepsin L resulted in a significant reduction of proteinuria in an experimental glomerulonephritis. 20 Faul et al. 21 also demonstrated that E64, a selective cathepsin inhibitor L, could prevent the protein kinase A inhibitor (H89) and/or the calcium-dependent protein kinase II inhibitor (KN62) induced loss of stress fibers and synaptopodin in podocytes. In another report, isolated podocytes from cathepsin L-deficient mice were protected from PAN-induced cell detachment. 22 Above all, cathepsin L-mediated proteolysis has a key role in the development of proteinuria and glomerular diseases because it causes podocyte injury.
Cystatin C is one of the most potent extracellular inhibitors of cysteine proteinases, and the insufficiency of the inhibitor indicates that the activity of cysteine proteinases is not regulated or inhibited properly in extracellular fluid. Western blotting revealed that in chronic phase (day 28 and 56), cystatin C level, but not cathepsin L level, decreased to the control level. It is thus postulated that the level of cathepsin L in the podocytes contributes to proteinuria and podocyte detachment by the degradation of GBM proteins in PAN nephrosis.
Another mechanism of cellular damage by cathepsin L was suggested by the absence of cystatin β in podocytes demonstrated in this study. Compared with the level of cathepsin L, cystatin β level was not obviously detected by immunostaining and Western blotting. Intracellular proteolysis by cathepsin is usually restricted within lysosomes to prevent self-digestion of cellular components. The lysosomal membrane separates enzymes from cytoplasm, and neutral pH of cytoplasm and intrinsic inhibitors.36,37 The stability of lysosomal membrane decreases in PAN nephrosis. 38 Accordingly, lysosomal enzymes with acidic fluid in the podocytes of PAN nephrosis may easily leak into the cytoplasm when lysosomal membrane is expanded by a large amount of endocytosis proteins as observed in our study. In vitro, PAN-treated podocytes showed a strong cathepsin L staining throughout the cytoplasm. 22 Server et al. 19 also reported that the presence of cathepsin L in the cytoplasm. However, they showed that this was not due to lysosomal leakage and that cytoplasmic specific “short cathepsin L (34 kDa)” was the key regulator in a lipopolysaccharide proteinuric mice model. In our present data, we could not find specific short cathepsin L; this may be due to the difference of species. Therefore, cathepsin L in the podocytes of PAN nephrosis may have the potential for uncontrolled proteolytic activity in the cytoplasm in the absence of intracellular inhibitors and subsequent intracellular damage of podocytes. This is consistent with the observation that parietal epithelial cells of the Bowman’s capsules around the adhesion, which were positive for cathepsin L and cystatin β, were not lost but proliferated to cover the flank of adhesion.
The mechanisms underlying the regulation of cathepsin L in the podocytes of rats with PAN nephrosis remain uncertain. Increased level on day 4 when proteinuria developed and on days 28 and 56 around the adhesion site where plasma leaked through areas of denuded GBM suggested that expression of cathepsin L in the podocytes of PAN nephrosis is secondary to proteinuria in the proteolysis of endocytosis proteins. Indeed, increased level of cathepsin L was detected on day 4, which reached the maximal level on day 8 when maximal proteinuria was reached. Based on these findings, it is conceivable that endocytosis and subsequent proteolysis in lysosomes, including the level of cathepsin L, may depend on the amount of protein leaking through GBM. However, some investigators reported that they are controlled by specific factors, such as growth factors in vitro studies.10–13 It is reported that the growth factor involved in proteinuria and podocyte damage is basic fibroblast growth factor (FGF).39–41 This growth factor was also reported to induce cathepsin L expression in podocytes, 27 cultured synovial fibroblast-like cells from patients with rheumatoid arthritis, 42 and skeletal muscle cells. 43 Further studies are thus required to examine whether growth factors, including basic FGF, cause podocyte damage and degradation of extracellular matrix by induction of cathepsin L to reveal the mechanisms of glomerular damage.
In summary, the level of cathepsin L and absence of its inhibitors in podocytes of rats with PAN nephrosis are suggested to be important factors in proteinuria and podocyte damage by extracellular and intracellular proteolysis. Podocyte damage was morphologically confirmed to be a critical step in the progression of glomerular tuft adhesion and sclerosis. Proteinuria and podocyte damage are observed in various human kidney diseases, such as MCD, membranous nephritis, diabetic nephropathy, chronic nephritis, which are strongly associated with the progression of chronic kidney diseases. The pharmacological blocking of the enzyme activity as well as the inhibition of induction of this enzyme may be a promising therapeutic intervention for treatment of proteinuria and podocyte damage.
Supplemental Material
DS_10.1369_0022155418791822 – Supplemental material for Expression of Cathepsin L and Its Intrinsic Inhibitors in Glomeruli of Rats With Puromycin Aminonucleoside Nephrosis
Supplemental material, DS_10.1369_0022155418791822 for Expression of Cathepsin L and Its Intrinsic Inhibitors in Glomeruli of Rats With Puromycin Aminonucleoside Nephrosis by Ayano Kubo, Isao Shirato, Teruo Hidaka, Miyuki Takagi, Yu Sasaki, Katsuhiko Asanuma, Kazumi Ishidoh and Yusuke Suzuki in Journal of Histochemistry & Cytochemistry
Footnotes
Acknowledgements
Competing Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
AK, IS, TH, KA, and YSuzuki designed the experiments. KI provided essential material. AK, IS, MT, and YSasaki executed the experiments and analyzed of the study. AK, IS, TH, and YSuzuki wrote the manuscript. All authors have read and approved the final manuscript.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
