Abstract
Human labial glands secrete mucous and serous substances for maintaining oral health. The normal microbial flora of the oral cavity is regulated by the acquired and innate immune systems. The localization and distribution of proteins of the innate immune system were investigated in serous acinar cells and the ductal system by the method of immunohistochemistry. Numerous antimicrobial proteins could be detected in the labial glands: β-defensin-1, -2, -3; lysozyme; lactoferrin; and cathelicidin. Cytoskeletal components such as actin, myosin II, cytokeratins 7 and 19, α- and β-tubulin were predominantly observed in apical cell regions and may be involved in secretory activities.
Keywords
Introduction
The salivary glands of the lip (labial glands) belong to the minor salivary glands which are scattered in the oral submucosa, for example, palate, cheek, floor of the mouth, lingual mucosa. In the mucosal area of the lip, they are represented as mixed glands consisting of mucous, serous, and seromucous glandular cells. The ductal excretory system in minor salivary glands differs from that in major salivary glands. Modified intercalated ducts are the smallest ducts and are rarely found. Intralobular ducts normally do not provide the typical basal membrane infoldings and increased numbers of mitochondria like in striated type of ducts.1–3 Saliva produced by the sublingual, submandibular, and minor salivary glands contains, for example, mucins and amylase 4 and is crucial for digestion, tasting, pH-buffering, tooth remineralization, and immune defense. 5 The clinical importance of these small glands becomes evident in the possible development of a variety of tumors. Minor salivary gland tumors represent 10% to 15% of all salivary neoplasms located to 15% in the lips. 6 Benign tumors predominate on the upper lip, and malignant tumors on the lower, as a result of different embryonic development. The pleomorphic adenoma is the most common tumor of the major and minor salivary glands.7,8 Other lesions are the papillary cystadenoma 9 and the sialolipoma 10 ; salivary gland lesions comprise mainly the salivary duct cyst 11 and mucoceles. 12 The labial minor salivary glands are an ideal source for minimal invasive recruitment of potential stem/progenitor cells due to easy accessibility. 13 A biopsy of the minor salivary glands of the lip in context with further clinical criteria can be used to diagnose the Sjögren’s syndrome, a disease that is characterized by the loss of glandular function leading to dry eyes and dry mouth.14,15 The easy dissection of the labial glands is due to the lack of capsules and the superficial location. 3 Minor salivary gland neoplasm in infants involving the buccal mucosal salivary gland 16 is reported.
Antimicrobial peptides are components of the innate immune system showing activity against bacteria, yeasts, fungi, and viruses.17–19 Studies investi-gating antimicrobial proteins in this localization often deal with a general description of oral mucosa glands representing the minor salivary glands and the major salivary glands.17,20,21 Only few descriptions deal explicit with labial glands, studying glandular structures22,23 or glandular protein expression.3,20,24 Moreover, only a few studies on salivary glands in infants have been reported. Lee et al. 25 investi-gated the localization of lysozyme, lactoferrin, α1-antichymotrypsin, and α1-antitrypsin in major salivary glands of human fetuses. Host defense peptides of the entire lip vermilion mucosa were studied by RT-PCR in infants. 26
Proteins of the cytoskeleton are involved in processes of secretion in apocrine and eccrine secretion modes, for example, human ceruminous gland, axillary gland, submandibular gland, rat parotid gland, and pancreatic acinar cells.27–31 In lacrimal acinar cells, cytokeratin (CK)-based intermediate filaments were concentrated at the cell apex and periphery. 32 In the same cells, actin and myosin II located around secretory vesicles support the exocytosis process. 33 Microtubules facilitate the stimulated lacrimal secretion. 32 This study demonstrates for the first time distribution and localization of cytoskeletal proteins in labial salivary glands and discusses possible involvement in secretory processes.
Materials and Methods
Tissue
We obtained samples of lip tissue from infants who were born with cleft lips. Excess tissue of the upper lip was collected during surgery procedures for lip repair. Twenty-three patients, 13 male and 10 female, were investigated. The age range comprises from 2 to 9 months; one patient was 3 years old. The material was fixed immediately in buffered formalin (4.5%). The study was performed according to the guidelines of the Ethics Committee.
HE and Immunohistochemical Staining
Paraffin-embedded tissue was cut into 5-µm-thick paraffin sections. For histological orientation, hematoxylin and eosin (HE) staining was used; for polyanions Alcian blue staining (pH 2.5) and for neutral carbohydrates, periodic acid–Schiff reaction was performed according to Romeis. 34 Immunohistochemical analysis was carried out according to the avidin–biotin–horseradish peroxidase method with the following antibodies: actin (Abcam, Cambridge, UK), myosin IIA (Sigma, St. Louis, MO), cytokeratin 7 (CK7; Dako, Hamburg, Germany), cytokeratin 19 (CK19; Dako), α-tubulin (Novus Biologicals, Cambridge, UK), β-tubulin (Santa Cruz Biotechnology, Santa Cruz, TX), β-defensin-1 (Santa Cruz Biotechnology), β-defensin-2 (Santa Cruz Biotechnology), β-defensin-3 (Novus Biologicals, Littleton, CO), lysozyme (Thermo Fisher Scientific), lactoferrin (Abcam, Cambridge, UK), and cathelicidin (Thermo Fisher Scientific). For antigen retrieval, sections were pretreated with microwave irradiation in citrate buffer at pH 6.0 for 15 min. Endogenous peroxidase activity was quenched by incubation with 3% hydrogen peroxide for 10 min. Sections were blocked with swine serum, goat serum, or bovine serum albumin (BSA) for 30 min. The first antibody was applied to the tissue for 1 hr at room temperature and overnight at 4C. The following antibody dilution was used: anti-actin, 1:50; anti-myosin II, 1:2000; anti-CK7, 1:100; anti-CK19, 1:50; anti-α-tubulin, 1:200; anti-β-tubulin, 1:200; anti-β-defensin-1, -2, -3, 1:100; anti-lysozyme, 1:2000; anti-lactoferrin, 1:100; and anti-cathelicidin, 1:50. The second biotinylated antibody (Vector Laboratories, Inc., Burlingame, CA), diluted 1:200, was applied to the slides for 30 min. Subsequently, slides were incubated with peroxidase-labeled streptavidin (Vector Laboratories, Inc.) for 45 min, followed by diaminobenzidine for visualization. Control sections without primary antibody were treated identically. The specificity of immunostaining was established by using mouse or rabbit IgG, as appropriate, at the same concentration as the first antibody as the isotype control for each protein. The squamous epithelium of the lip served as the positive internal control for the cytoskeletal proteins, and serous glandular structures of the human sinonasal mucosa for the antimicrobial proteins. Counterstaining was performed with hematoxylin. Images were taken with a digital camera mounted on a microscope (Nikon, Düsseldorf, Germany).
Results
General Morphological Observations
Mucous and serous acinar glandular components of the minor salivary glands of the lip were located in the submucosa forming a huge globular structure (Fig. 1A). Intralobular ducts discharged into a large extralobular duct leading through the stratified squamous epithelium into the inner part of the lip. They were characterized by two to three cell layers. Intercalated ducts were rarely found. Mucous endpieces predominate in the tissue sections and are frequently capped with serous glandular cells (demilune cells; Fig. 1B–D). Serous glandular cells of the secretory unit form distal acini enclosed by myoepithelial cells (Fig. 1B inset).
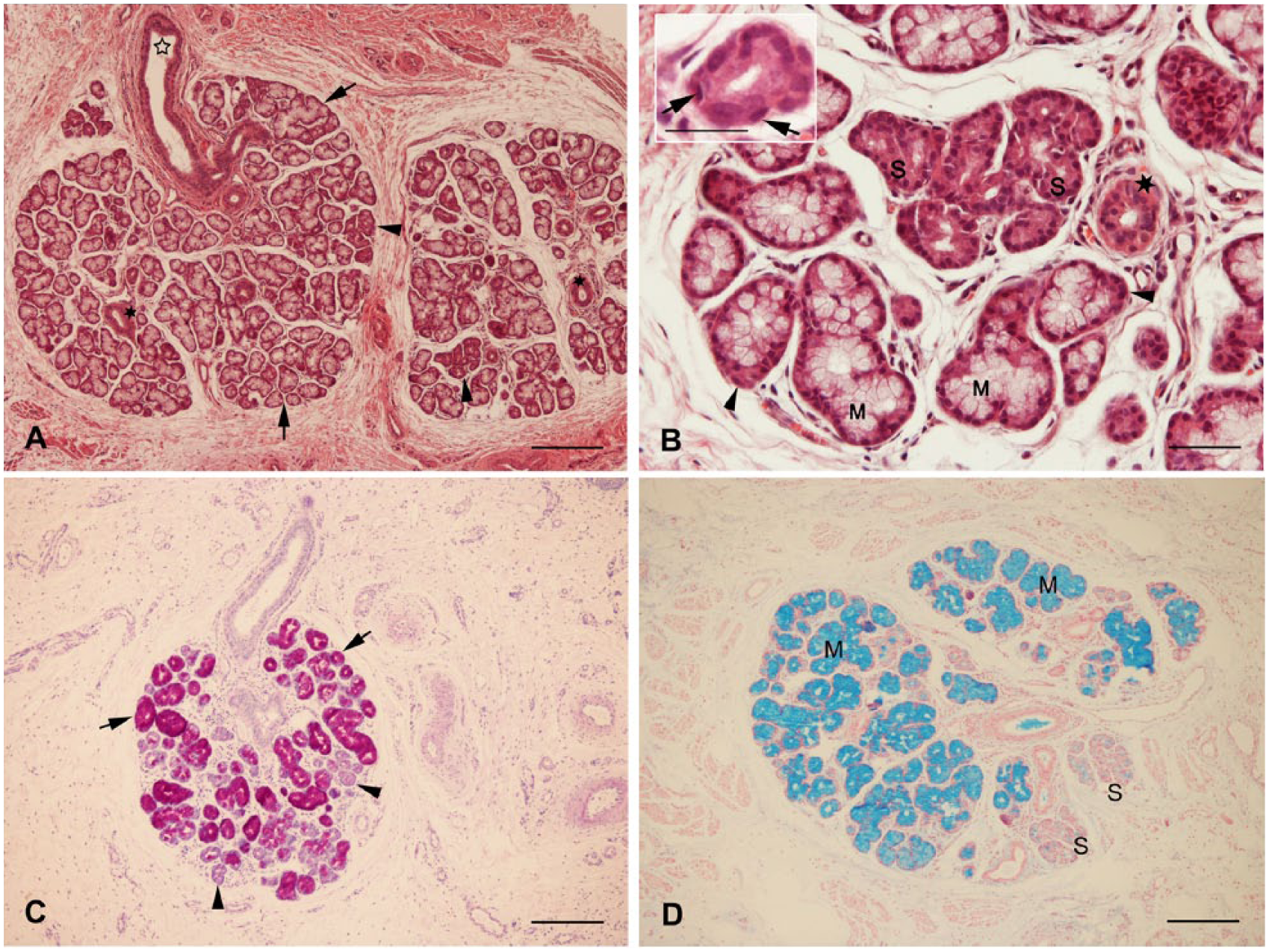
(A) Labial gland with serous acinar (arrowheads) and mucous (arrows) portions, intralobular duct (dark asterisks), and extralobular duct (clear asterisk). Hematoxylin and eosin (HE) stain. (B) Higher magnification of serous acinar (S) and mucous (M) glandular cells, serous demilunes (arrowheads), and intralobular duct (asterisk). Myoepithelial cells (inset) build a layer beneath the acinar cells and showed flattened nuclei (arrows). HE stain. (C) Periodic acid–Schiff stain, serous (arrowheads) and mucous (arrows) endpieces. (D) Alcian blue stain, serous cells (S), mucous cells (M). Scale bars: A, C, D = 250 µm; both B and inset = 50 µm.
Immunohistochemistry
β-Defensin-1, -2, -3
β-Defensin-1 immunoreactivity showed a predominantly strong staining in serous acinar cells and serous cells at the distal end of a mucous glandular endpiece (demilune cells). A strong apical expression could be observed partially in serous acinar cells. Intralobular ducts were stained strongly (Fig. 2A). Antibody to β-defensin-2 resulted in a weak to medium staining intensity in serous and ductal structures (Fig. 2B). β-Defensin-3 exhibited a throughout strong staining similar to β-defensin-1 (Fig. 2C). Strikingly, in some sections, all β-defensin peptides were concentrated in ductal cells of the basal cell layer. Extralobular ductal cells were evenly stained.
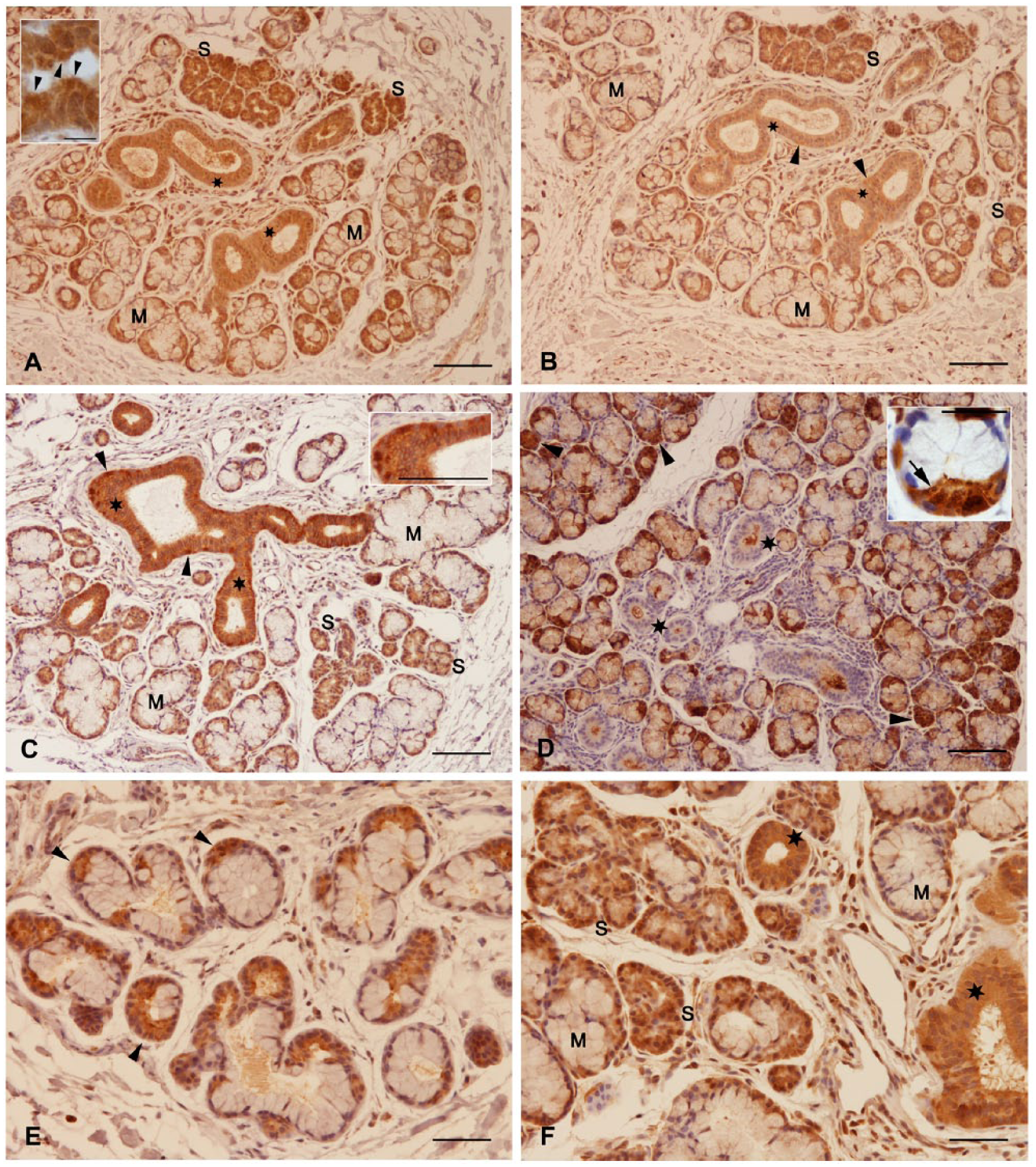
(A) β-Defensin-1, strong reaction of serous (S) acinar cells and intralobular ducts (asterisks). Mucous cells (M). Inset: Intense expression apically in serous acinar cells (arrowheads). (B) β-Defensin-2, positive staining in serous acinar cells (S) and intralobular ducts (asterisks). Mucous cells (M). (C) β-Defensin-3, protein expression in serous acinar cells (S) and intralobular ducts (asterisks). Mucous cells (M). Strong reaction in the basal part of ductal cells (arrowheads), see inset. (D) Lysozyme, granular staining reaction (see inset, arrow) was detected in serous acinar cells (arrowheads). Ductal cells remained unstained (asterisks). (E) Lactoferrin, positive staining in serous acinar and demilune cells (arrowheads). (F) Cathelicidin showed a strong expression in serous acinar cells (S) and intralobular ducts (asterisks). Mucous cells (M). Scale bars: A = 100 µm, inset = 10 µm; B = 100 µm; both C and inset = 100 µm; D = 100 µm, inset = 25 µm; E = 50 µm; F = 50 µm.
Lysozyme
Serous acinar cells and demilune cells stained strongly. Cytoplasmic secretory granules exhibited a positive reaction with the anti-lysozyme antibody. Intralobular and extralobular ducts remained unstained (Fig. 2D). Secreted material in the lumen of the duct showed an intensive staining.
Lactoferrin
A medium to strong staining could be detected in serous acinar and demilune cells with the antibody to lactoferrin (Fig. 2E). The duct system showed the same staining intensity. Partially apically stronger stained ductal cells occurred.
Cathelicidin
Acinar cells and serous cells at the distal end of a mucous glandular endpiece reacted by incubation with the cathelicidin antibody to a medium or strong extent (Fig. 2F). Intralobular and extralobular ducts showed the same staining pattern; the upper duct cells stained stronger apically.
Actin
A weak to medium reaction was observed with the antibody to actin in serous acinar cells and demilune cells (Fig. 3A). In serous endpieces, the basal part of the cell compartment was stronger stained than the rest of the cell. The apical and partially lateral cell membrane was strongly stained. Myoepithelial cells reacted positively. Intralobular ducts exhibited a weak to medium actin reaction with an apical accentuation. When the luminal duct cells showed protrusions, these were strongly marked with the actin antibody.
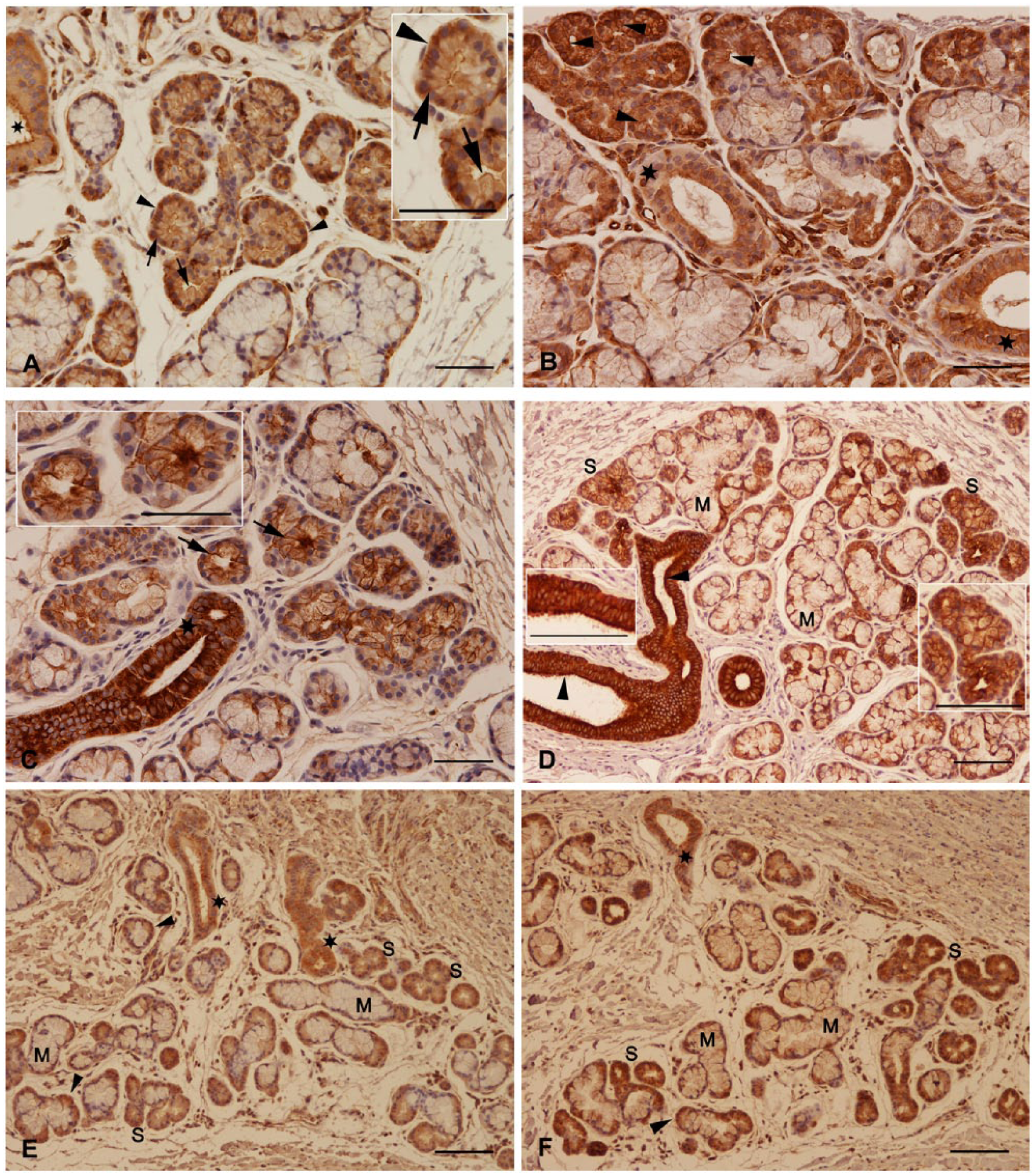
(A) Anti-actin immunostaining at the basal part of the cytoplasm (arrowheads) and at the apicolateral cell membrane (arrows) in serous acinar cells (inset). Positive staining of the apical membrane and protrusions of ductal cells (asterisk). (B) Myosin II was strongly expressed at the apicolateral cell membrane (arrowheads). Intralobular ducts (asterisks). (C) Strong cytokeratin 7 expression in apicolateral cell membrane (arrows) and intralobular ducts (asterisk). Inset with higher magnification. (D) Anti-cytokeratin 19 immunostaining, the intralobular duct system was strongly stained, especially the apical cell region (arrowheads). Serous gland (S), mucous gland (M), insets with higher magnification. (E) α-Tubulin showed a medium to strong staining reaction in serous acinar (S) and demilune (arrowheads) glandular cells and in ductal cells (asterisks). Mucous cells (M), ductal cells (asterisks). (F) β-Tubulin was localized in serous acinar (S) and demilune cells (arrowhead). Mucous cells (M), ductal cells (asterisk). Scale bars: both A and inset = 50 µm; B = 50 µm; C = 50 µm; both D and insets = 100 µm; E = 100 µm; F = 100 µm.
Myosin II
Serous acinar and demilune cells showed a medium to strong staining reaction with the antibody to myosin II. While partially an apical stronger staining could be detected in serous acinar cells, intralobular ducts were continuously stronger stained in the apical region showing a medium to strong cytoplasm staining in all cell layers (Fig. 3B).
Cytokeratins 7 and 19
The staining intensity and localization for CK7 and CK19 were identical (Fig. 3C and D). In serous acinar cells and in the serous demilune cells, CK7 and CK19 showed a weak to medium immunoreactivity; the apical and lateral cell membrane reacted stronger. In intralobular ductal cells, a strong staining could be detected. The luminal ductal cells showed a strong apical CK7 and CK19 expression. In some cases, ductal cells develop apical protrusions which seem to be pinched off and deposited in the lumen. These showed a strong CK7 and CK19 reactivity.
α- and β-Tubulin
α-Tubulin was expressed in a medium to strong intensity in serous acinar cells and demilune cells (Fig. 3E). Intralobular ductal cells were strongly stained. The apical part of ductal cells partially showed a stronger expression with the antibody to α-tubulin. The staining for β-tubulin (Fig. 3F) was identical to that for α-tubulin.
Discussion
Labial glands as a part of the minor salivary glands are important structures for the oral health in adults and children. The constant secretion of saliva by the labial glands prevents desiccation of the oral tissue. Especially, the localization of these glands at the oral cavity points to their important role in the first line of defense by proteins of the innate immune system. In the present study, we investigated the histological distribution of antimicrobial and cytoskeletal proteins of the less well-defined labial salivary glands in infants. The salivary secretion rate is identical in the labial mucosa of 3-year-old children, adolescents, and young adults. However, the numerical density of labial glands varied between the three groups showing a decreasing number of glands per surface unit at older ages. 35 Thus, the glandular labial structures in infants offer a promising area of the study of these glands.
Using the method of immunohistochemistry, β-defensin-1 was constitutively expressed in different glandular structures such as the human mammary gland, 36 ceruminous gland,27,37 and human glands of Moll. 38 Human β-defensin-1 mRNA was detected in minor salivary glands. 39 This is in accordance with the actual results in labial glands but not with the study by Sahasrabudhe et al. 40 in which human β-defensin-1 expression was restricted to the ductal cells and not to the serous acinar cells in minor salivary glands. We showed that β-defensin-2 and -3 were present in both ductal and acinar cells of the labial salivary glands. β-Defensin-2 is often induced by inflammation.41,42 But also constitutive expression was reported in literature.38,43 Moreover, β-defensin-3 is expressed either constitutively or induced by a variety of stimuli such as bacterial infection.44,45 Also in human minor salivary glands, 39 major salivary glands, 46 and oral mucosa, 47 determined by RT-PCR, β-defensin-2 and -3 were expressed in normal and inflamed tissue. Also the nature of bacteria whether they appear in encapsulated or unencapsulated form accounts for the susceptibility to antimicrobial peptides in the oral cavity. The sensitivity of capsulated Streptococcus mitis to human neutrophil peptides 1 to 3 is increased. However, a reduced sensitivity to human β-defensin-3 and cathelicidin was detected by capsule expression in S. mitis. 48 Interestingly, the transcription of human β-defensin-2 and -3 in the lip vermilion mucosa of infants is reduced compared with adults. 26 Defensin peptides can be produced in this tissue area from labial glands, the stratified squamous epithelium of the mucosa, or the epidermis of the lip. In our study, lactoferrin was expressed in serous acinar cells as well as in the duct system of the gland in the same intension. In prenatal salivary glands, lysozyme was present in serous acinar and demilune cells, whereas lactoferrin was low concentrated or nearly absent. 25 Moro et al. 24 who investigated the lower lip for lactoferrin expression in a fluorescence-based immunohistochemical study found only a weak signal in intercalated and intralobular ducts. We detected lactoferrin with a medium to strong staining intensity in serous acinar and ductal cells. This is in accordance with the investigation of Reitamo et al. 20 in which the peroxidase antiperoxidase method with diaminobenzidine as chromogen was used. Thus, staining differences might be due to methodical approaches. Immunoreactive lysozyme was localized in labial glands, serous acinar cells, and duct system concordant with our results.3,49 In our investigation, the presence of cathelicidin in human labial glands was described for the first time. So far, murine mRNA and protein of cathelicidin were detected in the salivary minor gland of the palate, palatal mucosa, and lingual epithelium. In addition, the human saliva 50 and the human gingiva 51 have been also tested positively for cathelicidin. The innate immunity represented by antimicrobial proteins and peptides is important predominantly in children because they are frequently exposed to germs and the acquired immune system is not fully developed. Especially in the first months of life, children explore their world by the tactile sense of the mouth. Studies reported that the immunoglobulin A (IgA) concentration is reduced in labial glands and in the saliva of children compared with adults. 52
The labial salivary glands secrete their products by exocytosis. Cytoskeletal proteins are significantly involved in the secretory machinery both in the cytoplasm and at the cell membrane. We found a strong actin staining at the apical membrane of serous acinar cells. F-actin is localized at secretory granules when the fusion with the apical membrane (opening of the fusion pore) occurs. 53 Furthermore, actin filaments separate secretory vesicles from the apical membrane. 54 Actin filaments are found in a region directly under the apical plasma membrane in parotid and pancreas serous acinar cells. 31 These actin bundles are distinct from the actin web under the lumen of the exocrine gland. 55 The actomyosin complex in regulated exocytosis is well studied in the salivary glands of rodents. 56 The collapse of the granules with the apical cell membrane is managed by the recruitment of F-actin and myosin II onto the surface of the granules. Interestingly, besides, the serous acinar cells, also ductal cells, show a strong actomyosin staining pointing to a probably extensive exocytosis event at this location. Microtubules composed of tubulin proteins are necessary for the transport of small secretory vesicles to the apical cell region during exocytosis.57,58 The secretory granules migrate thereby along the microtubule system in the direction of the apical membrane. 59 Luminal and cytoplasmic localization of tubulin proteins in the labial salivary glands point to their involvement in exocytotic processes. Stimulated secretion in lacrimal glands works with microtubules and/or microfilament pathways. 32 CK-based intermediate filaments were localized at the apical and peripheral cell membrane. This finding is in accordance with our results. Similar to actin and myosin II staining, the duct system shows also strong apical and lateral cell membrane staining with the CK antibodies. Exocytosis in duct cells of salivary glands has been studied, for example, in the intercalated duct of the parotid gland. 60
The human labial glands contribute to the innate immunity of the oral cavity by the production of antimicrobial proteins such as defensin-1, -2, -3; lysozyme; lactoferrin; and cathelicidin. The expressions of actin, myosin II, CK7 and CK19, and α- and β-tubulin as components of the cytoskeleton were located in the cytoplasm and were partially intensified in the apicolateral region. This might point to involvement in secretory activities and stabilization.
Footnotes
Acknowledgements
We thank Amela Klaus for excellent technical assistance.
Author Contributions
The authors contributed as follows: experiment design (MS, MRK), analysis and manuscript editing (MS), tissue preparation and clinical data (DJL, BO), experiment support (MS, DJL, CS, SK), and manuscript review (MS, DJL, BO, CS, SK, MRK).
Competing Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
