Abstract
The chorioretinal junction comprises the retinal pigment epithelium, Bruch’s membrane (BM), and adjacent choroidal capillaries. Its significance lies in its ability to support the retina mechanically and metabolically. The aim of this cross-sectional study was to record the senescent changes affecting all the constituents of the chorioretinal junction in 40 histological specimens across the whole spectrum of the adult age range. This study included light microscopy, with hematoxylin and eosin and PAS stains, and fluorescent microscopy. Immunohistochemistry was done using antibodies against neurofilament, synaptophysin, S-100, and collagen IV. The descriptive microanatomy was corroborated by morphometry. The amount of melanin and lipofuscin granule and drusens were noted. The ratio of thickness of BM to capillary diameter reduced from 1:6 or less in the 2nd decade to 1:3 in the 10th decade. Complete hyalinization of intercapillary pillars was seen in the 10th decade. The accumulation of lipofuscin with age was documented with the diminution in the size of epithelial cells. The subepithelial accumulation of drusen was first noted in the specimen from the late 60s. We have described all senescent changes in the chorioretinal junction chronologically. Similar changes are found in a more pronounced form in age-related macular degeneration. These data might serve as a reference baseline for clinicians and pathologists.
Introduction
The junction of the choroid and the retina is an important zone that constitutes of the pigment epithelium of the retina, Bruch’s membrane (BM), and choroidal capillaries. The retinal pigment epithelium (RPE) is a simple cuboidal epithelium with apical processes. BM is a pentalaminar structure, formed by the fusion of basement membranes of the RPE and endothelium of abutting choroidal capillaries with inner collagen, elastic tissue, and outer collagen layers present between the two basement membranes. The choroidal capillaries provide vascular supply to the outer third of the retina, including the RPE, photoreceptor layer, and the outer nuclear layer. Many functions are attributed to the RPE and BM, including phagocytosis of membranous discs received from the photoreceptors, metabolic support for visual pigment resynthesis, transferring nutrients from the choriocapillaris to the neural retina, and absorption of light passing through the neural retina to prevent glare. It is an important component of the blood retinal barrier. 1 In addition, the RPE is responsible for maintaining the integrity of the choriocapillaris; surgical RPE removal or pharmacological RPE damage leads to secondary choriocapillaris atrophy. 2 The integrity of the RPE monolayer is maintained if there is proper attachment of the RPE cells to one another and to BM. 3 BM is under constant cyclical stress as the circulation pressure in the choroidal capillaries varies with cardiac rhythm. The mechanical properties of the BM, specifically its elastic property, enable it to sustain these potentially damaging changes. The chorioretinal junction shows various changes with increasing age. These changes disturb the delicate balance of the chorioretinal junction by alteration of physiological and mechanical properties. Most of these changes are cumulative and may in time lead to pathology. These changes have been studied before4–8 but very few studies deal with all the changes across the entire spectrum of the adult age range. We hypothesized that changes seen at the chorioretinal junction—thickening of Bowman’s membrane, RPE modifications, and decrease in the diameter of choriocapillary—have linear correlation with age. Here, we present sequential changes seen in the chorioretinal junction from 20 to 99 years of age using hematoxylin and eosin (H&E) stain, special stains, immunofluorescent microscopy, immunohistochemistry, and morphometry.
Materials and Methods
Forty adult retinal specimens obtained by the body/eye donation program were used. The age ranged from 20 to 99 years (Table 1). The eyeballs were removed within 2 to 4 hr of death and fixed in 10% buffered formalin for 48 hr. The eyeballs were sectioned parallel to the equator nearer to the posterior pole and processed for routine H&E and periodic acid schiff (PAS) staining. Analysis was limited in this study to the retinal section taken parallel to the equator, near the posterior pole.
Decade and Age-Wise Distribution of the Studied Specimens.
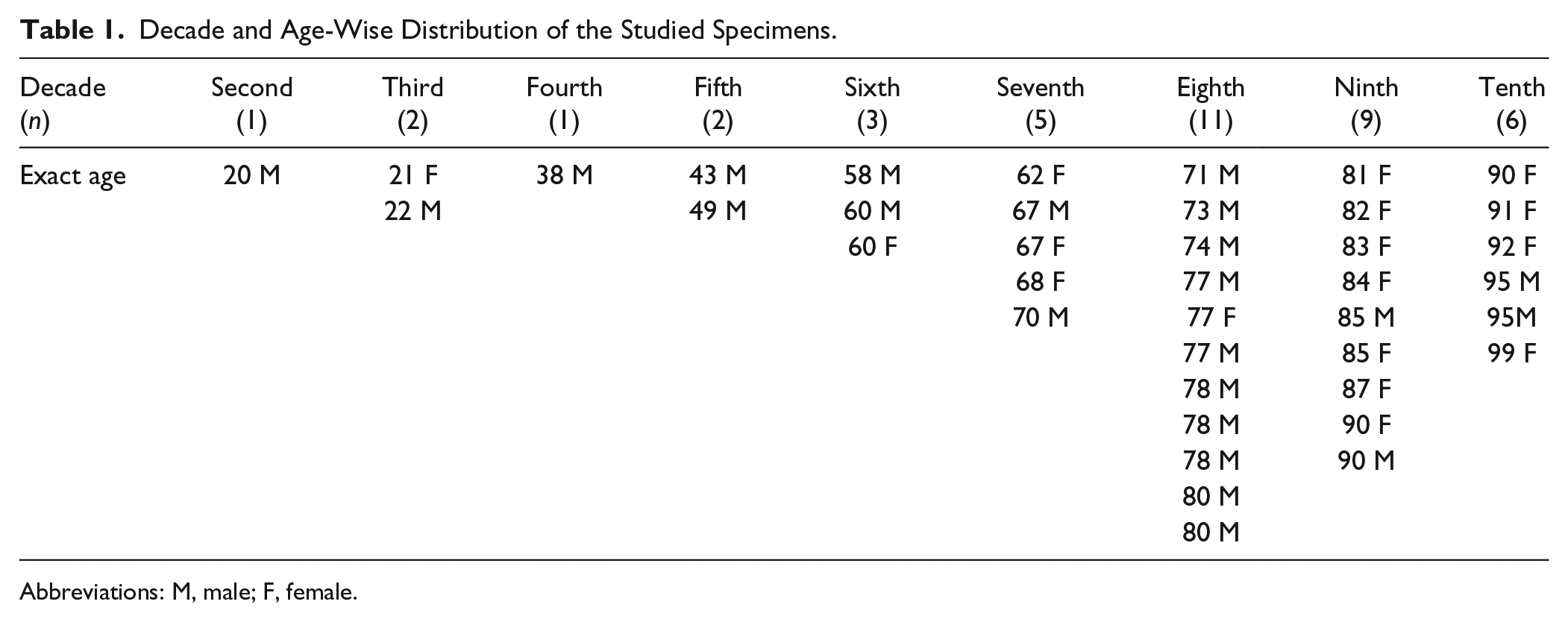
Abbreviations: M, male; F, female.
This was followed by paraffin embedding, block making, and sectioning. The sections for H&E were 5 µm thick whereas for the immunohistochemistry 4-µm-thick sections were taken on the poly-
Anti-neuro-filament protein (NFP) antibody (monoclonal; Dako)
Anti-synaptophysin (SP) antibodies (monoclonal; Dako)
Anti-S-100 antibodies (polyclonal; Dako)
Anti-collagen IV (monoclonal; Cell Marque, USA)
First three antibodies were preconstituted and ready to use with no need for dilution; anti-collagen IV antibody was used in 1:100 dilution.
The
Exclusion Criterion
Specimens with a history of ocular disease, ocular trauma, hypertension, or diabetes were excluded from the study.
Ethical clearance was taken from the institutional ethical committee.
Results
Retinal Pigment Epithelium
In the third decade, the RPE was a simple cuboidal epithelium with a rounded, vesicular, basally placed nucleus covered by melanin granules. The granules were multiple, discrete, with brown staining, and present in all parts of the cell but concentrated more on the retinal side. In the 38- and 43-year-old samples, the RPE cells were plump cuboidal as before, but melanin granules were reduced in number so that the basal nucleus with dispersed chromatin was clearly seen (Figs. 1 and 2). The granules were present in the retinal half of the cell extending either up till the nucleus or covering a small apical part of the nucleus. Only in a few cells the granules were present throughout the cell, covering the nucleus fully and reaching up to the basal part of the cells.
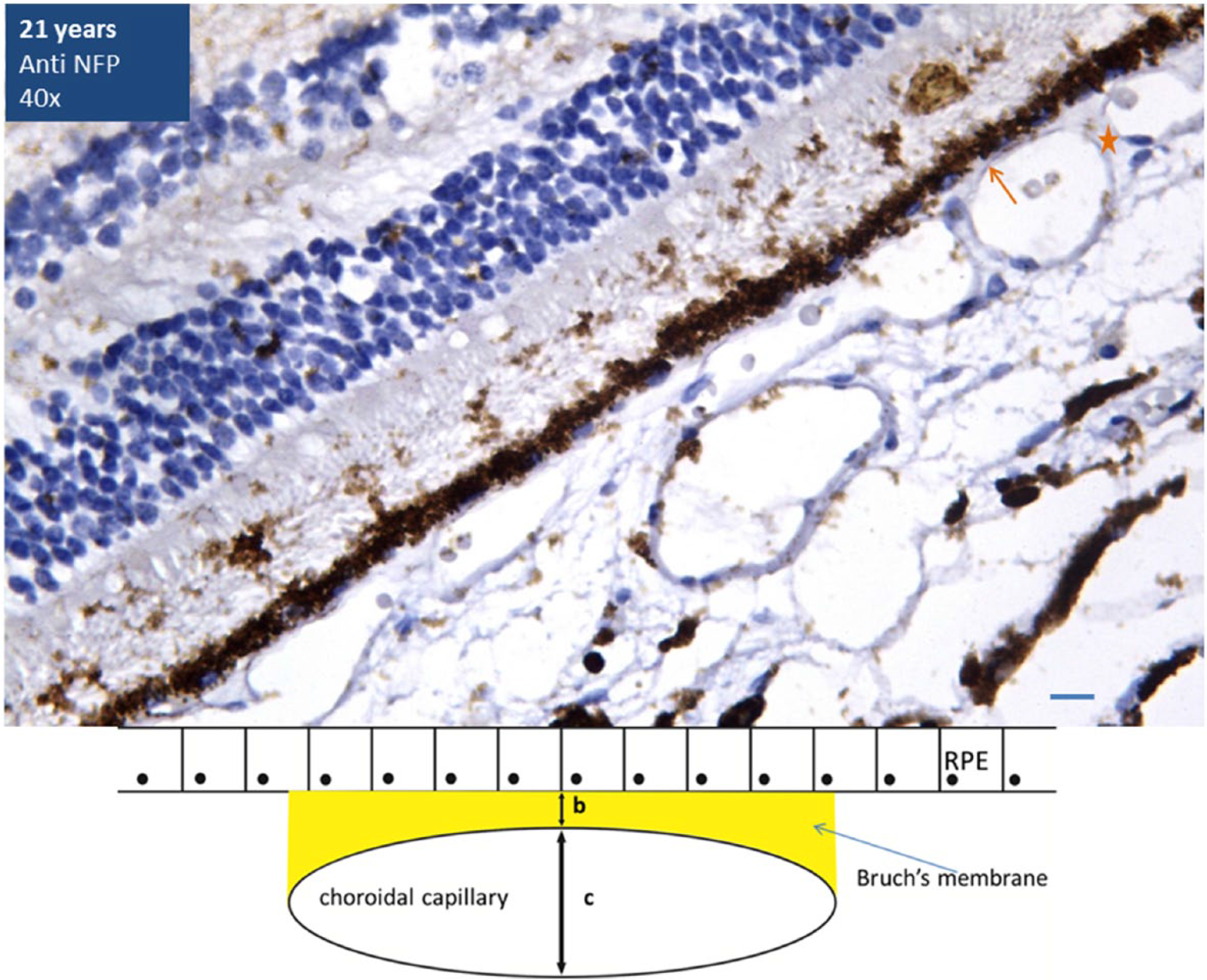
Twenty-two-year-old retina immunostained with NFP antibodies (40×). The RPE cells are fully filled with melanin granules that also cover most of the basally located nucleus. Thin Bruch’s membrane is seen. The ratio of (b) the Bruch’s membrane thickness to (c) the diameter of the abutting choroidal capillary (b:c) is more than 1:6. Line diagram is depicting the chorioretinal junction. Scale bar, 5 µm. Abbreviations: NFP, neurofilamentary protein; RPE, retinal pigment epithelium.
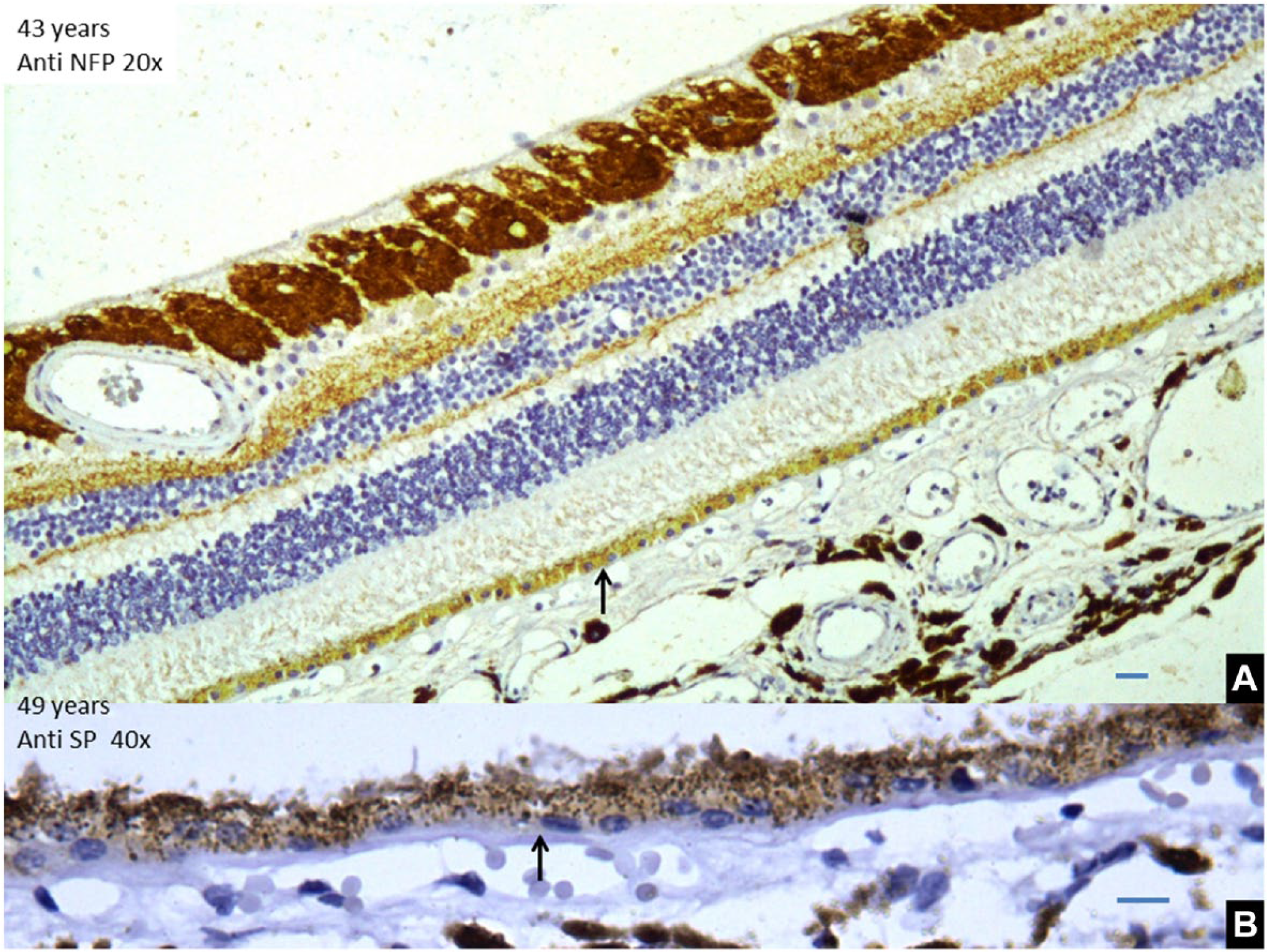
(A) Specimen of a 43-year-old retina showing RPE cells are low cuboidal cells with exposed nucleus and a decreased pigment content. Scale bar, 10 µm. (B) Thickening of Bruch’s membrane in a 49-year-old retina. Scale bar, 5 µm. Abbreviations: RPE, retinal pigment epithelium; NFP, neurofilamentary protein.
In the 58-year-old specimen and in those from early 60s, the RPE cells were attenuated. There was a small decrease in the height of the cell with a smaller, less intensely stained nucleus. The intracellular granules were further reduced in number. There were four to five interspersed areas where the RPE was stratified and was between two to three and five to six cells thick. In such areas of RPE proliferation, the cells were polyhedral with a central round nucleus and were filled with granules. One such area of stratified RPE cells was seen at the macula but the rest were present in the peripheral retina. In the latter half of the sixth decade, segments with the multilayer RPE were predominant. In most of the areas, the RPE was two cells thick but in the macular area, it was five to six cells thick. In one case in the early 70s (Fig. 3), apart from a few areas of simple epithelium, the whole expanse of the RPE was found to be two cells thick. In the areas with single cell thickness, many cells appeared binucleate. The macular RPE was two cells thick. In another case, the whole RPE was found to be attenuated throughout its expanse with a reduced cell size and fewer granules with no area of RPE proliferation. In the late 70s, the RPE cell size seemed to decrease further with almost squamous like cells and sparse granules (Table 2). During the 9th and 10th decades, progressive RPE attenuation was found (Fig. 4). In the 90s, the granules were severely depleted.
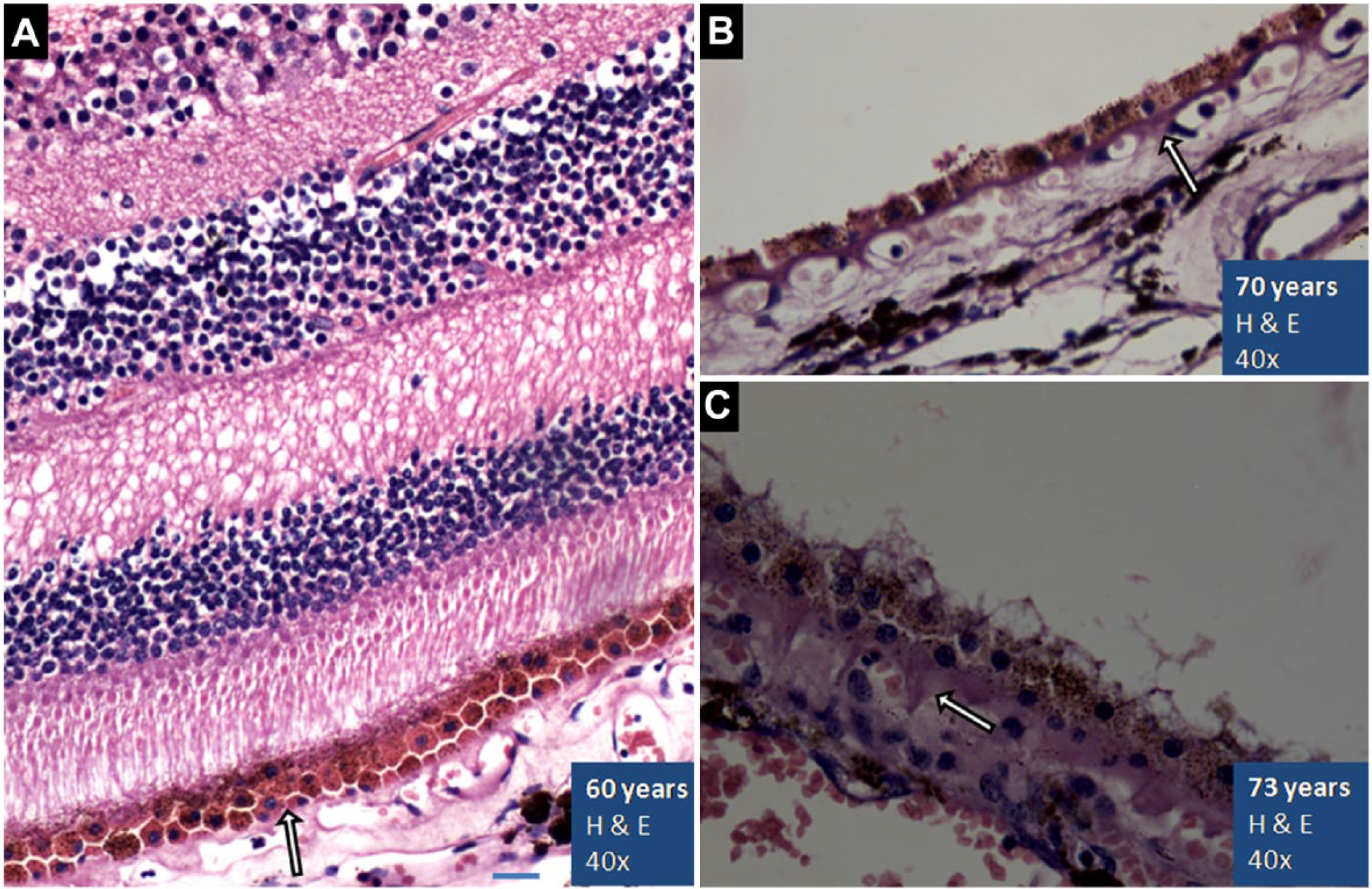
(A) The H&E stained (40×) specimen of a 60-year-old retina. The macular region is shown as evident by multilayered ganglion cells. The RPE is stratified, 2- to 3-cell-thick layer, with polyhedral cells (arrow). A longitudinal section of capillary can be seen in the inner plexiform layer (IPL). (B) A 70-year-old peripheral retina at 40×. RPE cells are attenuated with reduced pigmentation. (C) Stratified, pigment-filled polyhedral cells of a 73-year-old peripheral retina at 40×. A gradual increase in Bruch’s membrane thickening with hyalinization spreading to intercapillary pillars is shown in all the sections (arrow). Scale bar, 5 µm. Abbreviation: RPE, retinal pigment epithelium.
Semiquantitative Data of the Chorioretinal Junction in Each Decade.
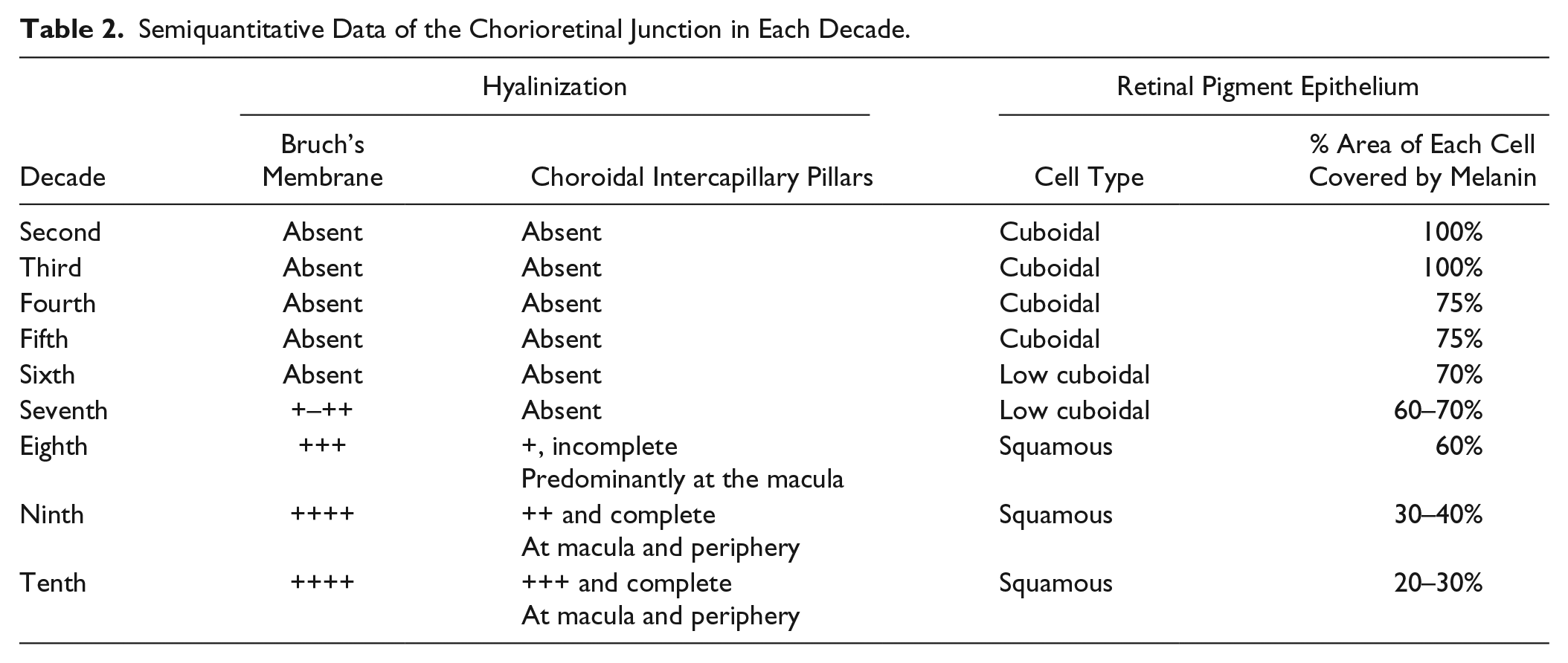
(A) Retina, 83 years old, immunolabeled by anti-collagen IV antibodies (20×): Thickened BM of the RPE and choroid capillaries. (B) PAS-stained retina (40×), 87 years old. Increased hyalinization of BM and intercapillary pillars. (C) 78 years old, thickened BM and depletion of melanin granules (H&E, 40×). (D) 91 years old, RPE with squamous cells and thick BM. (E) 95 years old, reduction in the thickness of retina with merging of outer and inner nuclear membranes. RPE is filled with melanin granules and shows age-related melanin reduction is not universal (H&E, 40×). (F) Two-cell-thick RPE of a 99-year-old, reduced cell size and hyalinized BM (H&E, 20×). Scale bar, 5 µm. Abbreviation: BM, Bruch’s membrane; RPE, retinal pigment epithelium.
From the apical aspect of the RPE cells, thin eosinophilic extensions were seen going into the layer of rods and cones. These extensions were devoid of granules and seen in the form of a sheet. There was found to be a gradual senescent reduction in these apical microvilli.
Fluorescent Microscopy
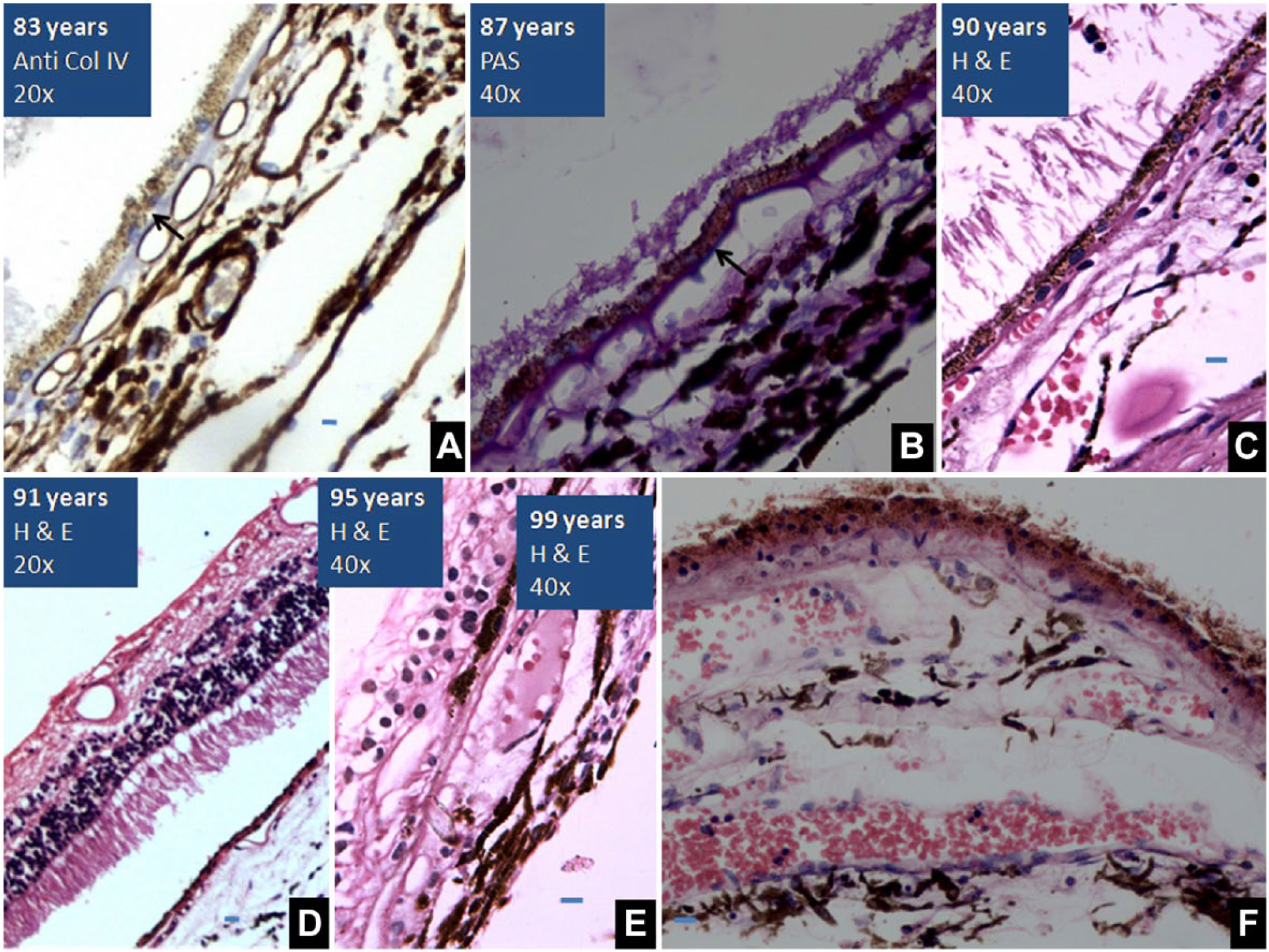
We examined the H&E slides under the fluorescent microscope, to differentiate the pigment present in the RPE cells (Fig. 5). Lipofuscin is an auto-fluorescent pigment whereas melanin is not. In specimens of the third decade, only melanin was found whereas specimens of later years showed progressive accumulation of lipofuscin. In specimen of the seventh decade, lipofuscin was present at the base whereas melanin occupied the apical areas of the cell. We observed a gradual decline in melanin with age but it was not universal. The specimens of the 10th decade showed brilliant fluorescence fully covering the cells. In the retina from the 99-year-old, both the pigments were present (Fig. 4).
Retina as seen under the fluorescent microscope. Solid arrows indicate RPE whereas the dotted arrows are marking Bruch’s membrane. A gradual decrease in the melanin content (seen as black nonfluorescent granules) and an increase in the lipofuscin-induced yellow fluorescence can be appreciated with advancing age. Scale bar, 5 µm. Abbreviation: RPE, retinal pigment epithelium.
Bruch’s Membrane
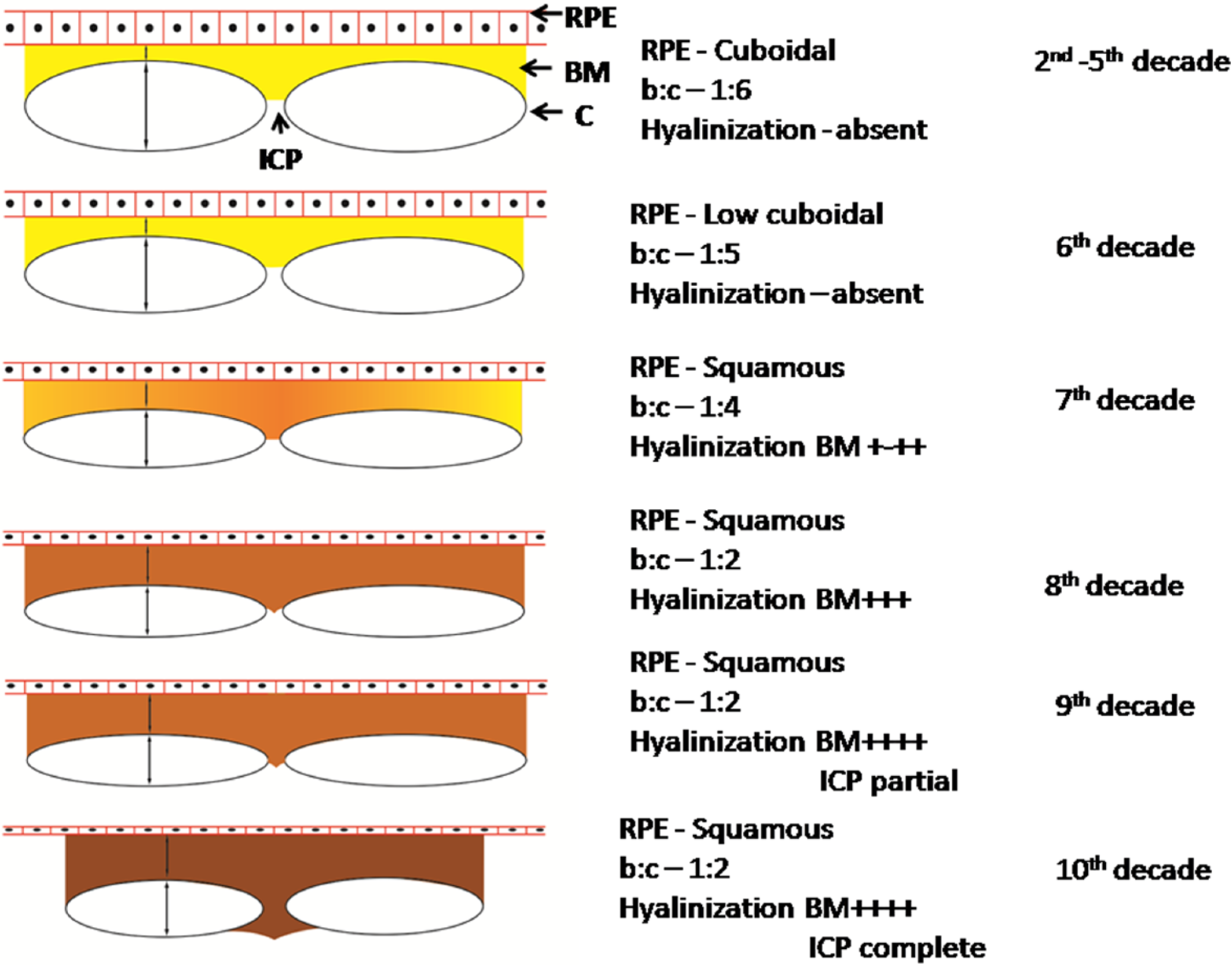
The RPE cells rest on the basement membrane that fuses with the basement membrane of the choroidal capillaries to form BM (Fig. 6). In the second decade, it was seen as a thin but distinct membrane. The ratio of BMs membrane to the diameter of the abutting choroidal capillary (b:c) was 1:6 or less. These changes are better appreciated in the slides labeled with anti-SP or anti-NFP antibodies (Figs. 1 and 2).
Diagram depicting age-related changes at the chorioretinal junction from 2nd to 10th decades. Arrows show the diameter of the BM and diameter of the choroidal capillary (C). Darkening in the BM denotes increasing hyalinization. Abbreviations: BM, Bruch’s membrane; RPE, retinal pigment epithelium.
In the early 60s, BM was found to be thickened and hyalinized. The ratio of the BM thickness to the diameter of the underlying choroidal capillary decreased to 1:4. At the macula, BM was much more thickened and its ratio with the capillary diameter decreased to 1:3 (Fig. 3A). In the late 60s, the hyalinization of the BM was increased but it was not extending into the intercapillary pillars in the choroid. In the early 70s, the hyalinization of BM was more advanced and extending in between the underlying choroidal capillaries in the peripheral retina (Fig. 3B). At the macula, BM was thicker than that seen in the peripheral retina with a BM width to capillary diameter ratio of 1:3 to 1:2, and intercapillary pillars were completely hyalinized (Fig. 3C). In the latter half of the 70s, BM was thicker. In the 9th and 10th decades, BM was seen as a thick undulating membrane and at places the ratio of BM to the capillary diameter was decreased to 1:2. The hyalinization of intercapillary pillars was also noted in the peripheral retina.
The thickening of BM was best demonstrated by immunolabeling of the antibodies against the collagen IV that localized to the basement membrane of the RPE and basement membrane of choroidal capillaries, and also by PAS staining (Fig. 4A and B).
The b:c ratio was analyzed to calculate the absolute percentage variation and decade-wise percentage variation (Table 3). The absolute percentage variation of b:c is the b:c ratio at the second decade, and is taken as optimum or 100% with variation being 0%. The absolute percentage variation in each decade gives the percentage difference from the optimum. The decade-wise percentage variation of b:c denotes the difference in the b:c ratio as compared with the preceding decade.
Decade-Wise Variation in the Ratio of Bruch’s Membrane Thickness to the Choroidal Capillary Diameter (b:c).
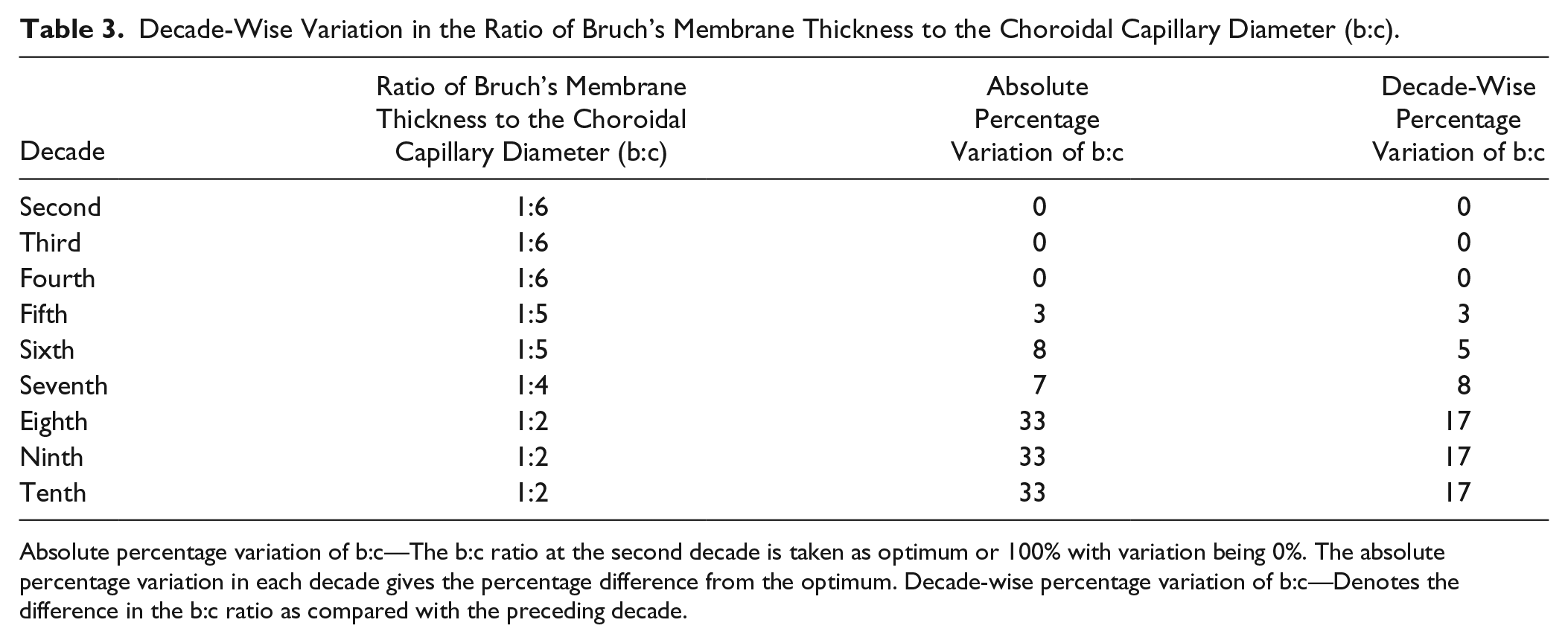
Absolute percentage variation of b:c—The b:c ratio at the second decade is taken as optimum or 100% with variation being 0%. The absolute percentage variation in each decade gives the percentage difference from the optimum. Decade-wise percentage variation of b:c—Denotes the difference in the b:c ratio as compared with the preceding decade.
Deposits at the Junctions
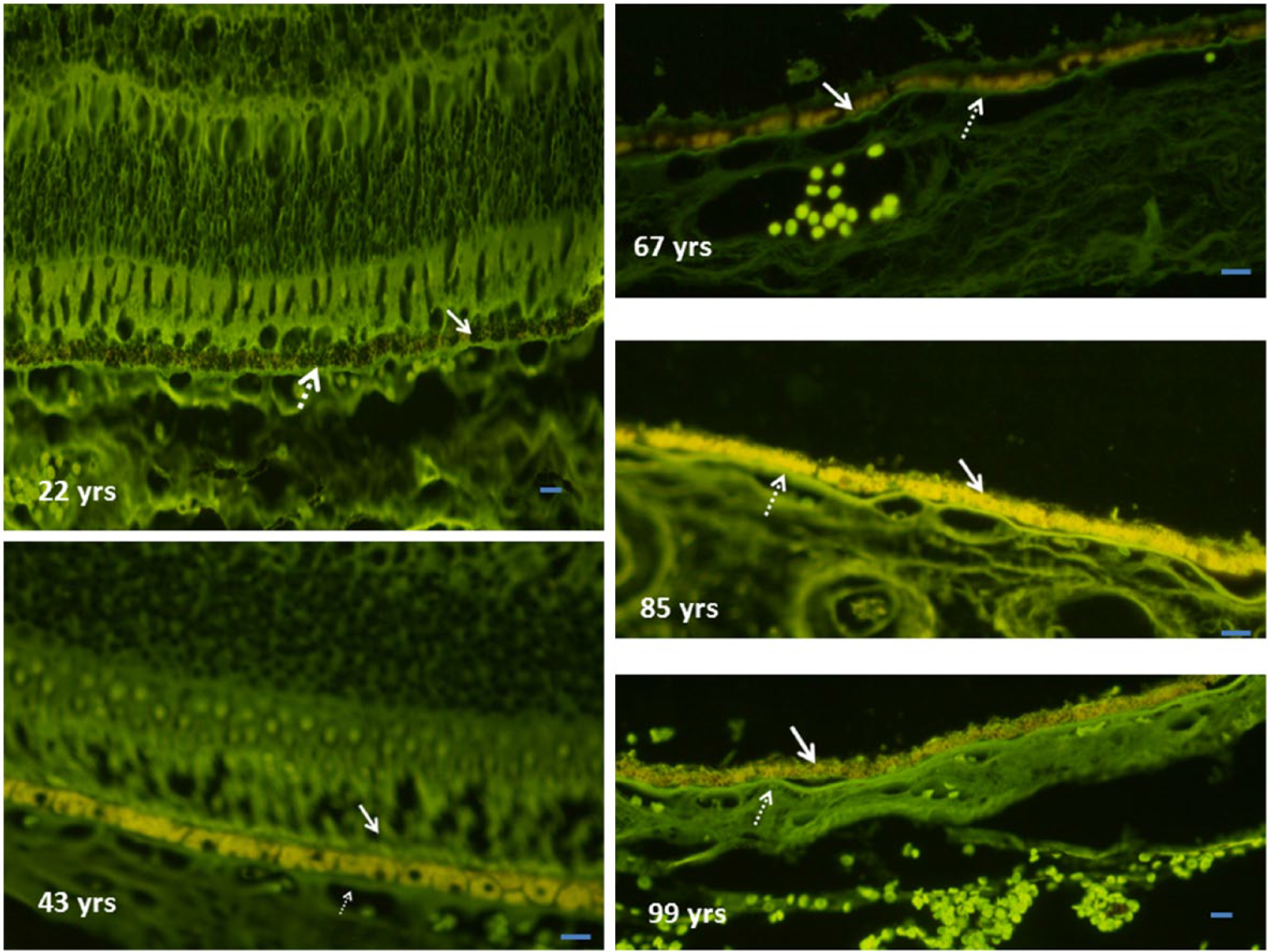
Up till the late 60s, no accumulation was seen beneath the RPE cells and their basement membrane. Dry drusens (Fig. 7) were first seen in the 67-year-old retina. They were two in number, seen in the peripheral retina as well-defined, rounded accumulation of a homogeneous material between the RPE cell and its basement membrane. In the eighth and ninth decades, two to five dry drusens were seen but many specimens did not have such deposits. In the early 90s, the number increased to six to eight. At 95 years, multiple drusens were present throughout the membrane. The size of deposits was increased with two deposits of huge proportions and striated appearance. At 99 years, multiple drusens were observed. Wet drusens and basilinear deposits were not found in any of the specimens.
Dry nodular drusens. (A) Retina of a 77-year-old showing dry nodular drusen, present between RPE and basement membrane. RPE cells are squamous in shape (H&E, 40×). (B) Multiple drusens in an 84-year-old specimen. (C) An 85-year-old retina with two drusens and attenuated RPE is seen in the field (H&E, 40×). (D) A 95-year-old retina studded with large drusens with striated appearance (H&E, 40×). Scale bar, 5 µm. Abbreviations: RPE, retinal pigment epithelium; ICP, intercapillary pillar; BM, Bruch’s membrane.
Discussion
The RPE performs highly specialized metabolic and transport functions essential for homeostasis of the neural retina. The photoreceptors shed about 5% of their outer segment mass daily. These spent tips of the rods and cones are received through apical microvilli of the RPE and phagocytosed. 12 One RPE cell supports 30 to 50 photoreceptors. The apical microvilli were seen as an eosinophilic sheet present at the base of the photoreceptor layer interdigitating with the outer most part of the photoreceptor layer. These projections were found to decrease with age with a marked reduction in the 80s and 90s as also reported by Bonhilla. 6
With aging the RPE becomes more pleomorphic, with the macular RPE becoming narrower with an increased height and the opposite occurring in the periphery. The peripheral RPE cells become broader, lower, vacuolated, and pleomorphic with age. 13 In our study, attenuation of the RPE was evident at 60 years of age with cells reduced to squamous shape in the late 70s. Narrowing of the RPE was found in peripheral as well as macular retina. In some specimens belonging to the seventh decade, large polyhedral RPE cells arranged in layers were present, but such areas were seen at both the macula and periphery.
The RPE melanin is of neural ectoderm origin and is accumulated as a by-product of tyrosine and dihydroxyphenylalanine oxidation. Lipofuscin is another intracellular auto-fluorescent pigment that originates mainly from the phagocytosed outer segments of the photoreceptors. The RPE has a well-developed and very active lysosomal system. Hence, the accumulation of lipofuscin is seen as a sign of lysosomal aging and a universal index of senescence.14,15 An age-related increase in the retinal lipofuscin contents has been documented,4,7 but it is believed that there is gradual loss of melanin and accumulation of lipofuscin. It is suggested that the accumulation of lipofuscin in the aged RPE is connected to the RPE functional degeneration either by “clogging” of the cytoplasm by the accumulated granules, or by increased oxidative stress in the cell. 6 Through light microscopy, we found a gradual but steady decline in the intracellular granule content of the RPE (Table 2). This depletion became very striking in the ninth decade. We expected to find an increase in the intracellular granules in the later decades because of the age-related increase in the lipofuscin pigment. Therefore, the tissues were analyzed under the fluorescent light to differentiate between the nonfluorescent melanin and the brilliantly fluorescent lipofuscin. The fluorescence was found to be uniform, suggesting that lipofuscin was so abundant that the resultant fluorescence seems to fill the cell completely and is not seen as granules (Fig. 4). This is in concurrence with the findings of Wing et al., 4 who reported that the in older eyes, the lipofuscin granules form the clumps and fill the entire RPE cell cytoplasm.
The sub-RPE nodular drusen seems to be a physiological process of aging. These drusens are excrescences formed on the inner aspect of BM and are composed of a granular substance, lipid, protein, crystalline deposits of calcium, and residual bodies. There are several histopathological types of drusens, including hard, soft, confluent, and large drusens. Although multiple classifications exist, most clinicians use size to classify drusens: small or “hard” (<63 μm) and soft, intermediate (>63 to <125 μm), and large (>125 μm) drusen. When hard or soft drusens coalesce to the point of losing their boundaries, they are then classified as “diffuse.” Hard drusens are nonspecific and age related, whereas soft drusens are considered pathognomonic of age-related macular degeneration (AMD).6,16 The hard drusen is said to be present in 83% of autopsy eyes with no documented ocular disease. 17 We observed two to three dry drusens first at the age of 67 years. At the age of 95 and 99, the retinal RPE was studded with dry drusens. But the appearance of drusen was not a constant feature as some specimens from the eighth decade did not show any. The age-related and pathological changes in the human BM are documented and include thickening and changes in its composition. 18 The BM is an acellular extracellular meshwork found between the RPE and the choroid and is known to undergo increased thickening during aging. 19 May 20 reported that thickening of the BM starts in the periphery and macular thickening was first observed from 45 years of age. Other authors proposed its linear correlation with age.21,22 The thickening of BM leads to a decrease in its solubility and elasticity. 23 The age-related hyalinization of BM and intercapillary pillars of choroid could modify the diffusion characteristics, which in turn compromises the retinal metabolism. 21 We have recorded gradual thickening and hyalinization spreading into intercapillary pillars with age. The ratio of thickness of the BM to capillary diameter reduced from 1:6 or less in the 2nd decade to 1:2 in the 10th decade. In the specimens from the 10th decade, the membrane was very thick with complete hyalinization of intercapillary pillars (Fig. 6, Table 3). It has been postulated that a progressively thickened BM could mechanically compress the choriocapillaris and cause a decrease in choriocapillaris’ diameter. 24
AMD is an exaggeration of normal aging changes. In pathological conditions like AMD, the structural changes within the BM are seen one or two decades before the cellular changes in the RPE. These changes within the BM can induce modifications in the attachment, survival, proliferation, and gene expression profiles of the overlying RPE.25–27 Sun et al. 5 have demonstrated with direct evidence that senescent changes in the BM reduce the phagocytic ability of the RPE which in turn increases the BM changes causing further reduction in the phagocytosis.
Our results support that physiological changes at the chorioretinal junction have linear correlation with age. Given the many supporting functions that the RPE serves for the retina, it is important to decipher the age-related changes in this epithelium to understand age-related changes in vision. Moreover, these physiological senescent changes are found in a more pronounced form in the pathological state of AMD, leading to severe loss of vision. Hence, a chronological description of these changes in adults might aid clinicians as well as pathologists by providing baseline data.
Limitations of the Study
We have limited this study to the retinal section taken parallel to the equator, near the posterior pole only, as other portions of the globe were utilized for other studies and not available. Thus, it is likely that our study does not capture the full spectrum of changes occurring with age. Nonetheless, we can conclude that for this section, there are notable age-related changes.
Footnotes
Competing Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
TG designed the study, analyzed the data, and drafted the manuscript; NS performed the immunohistochemistry and helped with the manuscript; JA carried out the morphometric and immunofluorescent study; and DS was involved in conception of the study, execution, and acquisition of material. All authors have read and approved the final manuscript.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
