Abstract
Autofluorescent compounds present in intestinal tissue often hinder the ability to utilize multiple, spectrally different, fluorophores. In addition, fixatives and blocking solutions may contribute to background autofluorescence or nonspecific immunofluorescent labeling. During immunofluorescence protocol development, autofluorescent pigments were observed in frozen bovine mid-ileal intestinal tissue sections. Coagulant fixatives, normal serum blocking, histochemical stains Sudan Black B (SBB) and 3,3′-diaminobenzidine (DAB), and spectral separation using imaging software were compared for their ability to reduce autofluorescence, as well as their effect on immunofluorescent labeling. Fluorescent pigments of frozen bovine mid-ileal intestinal tissue sections, most likely caused by eosinophils and lipofuscin, were masked successfully with a combination of DAB and SBB. Little to no statistical differences were observed for all other methods investigated; however, tissue fixed with 1:1 acetone methanol and 10% horse serum diluted in 0.05 M Tris buffer demonstrated lower mean fluorescence intensities. Spectral separation of specific immunofluorescent labeling from background autofluorescence is a simple method for removing unwanted fluorescence; however, successful separation is dependent on tissue and labeling quality.
Introduction
Inherently fluorescent components of intestinal tissue often hinder the ability to utilize multiple, spectrally different, fluorophores. Typical autofluorescence, caused by collagen and elastin, are most visible throughout green wavelengths.1,2 However, eosinophils and lipofuscin (fatty pigments contained within macrophages3,4), which can individually demonstrate a broad emission spectrum,1,2,5–8 are particularly problematic for immunofluorescence experiments and make it difficult to discern specific labeling from autofluorescence.
The successful chemical quenching of eosinophilic autofluorescence has been demonstrated previously,9–12 yet such treatment may also reduce immunofluorescent labeling of a target antigen. Alternatively, previous studies have also demonstrated reduction of these pigments using black histochemical stains to mask fluorescence. Sudan Black B (SBB) works via a mechanism of boundary surface adsorption, 13 staining lipids and fats in tissue black, whereas 3,3′-diaminobenzidine (DAB) is traditionally a chromogenic detection method of peroxidases, and when combined with a metal (e.g., nickel), produces a black precipitate. 14 Due to their abilities to stain unwanted fluorescent pigments black, SBB and DAB have been used in previous studies to mask lipofuscin8,15,16 and eosinophilic autofluorescence,17,18 respectively, in immunofluorescence experiments. However, the effect of these histochemical stains on autofluorescence and immunofluorescent labeling in frozen bovine intestinal tissue sections has not yet been demonstrated.
Other components of immunofluorescence experiments may also hinder the clear visualization of specific immunofluorescent labeling by causing auto-fluorescence and/or nonspecific immunofluorescent labeling. Fixation of the tissue section is an important step before labeling to ensure that the cellular components and processes are halted. 19 Although aldehydes tend to be the fixative of choice for frozen tissue sections, the use of these fixatives requires antigen retrieval to unmask epitopes. 20 Coagulating fixatives, such as ethanol, methanol, and acetone, do not require antigen retrieval, but may result in epitope degradation and/or increase in autofluorescence.19,20 One must also consider the blocking solution to be utilized. The addition of a blocking solution before incubation with the primary antibody is common in immunohistochemistry (IHC) as a method to reduce the binding of the primary and/or secondary antibody to endogenous Fc receptors in the tissue section, thereby reducing nonspecific labeling.14,17 However, it is possible that a blocking solution may increase autofluorescence and/or nonspecific immunofluorescent labeling if there is antigenic similarity between the blocking serum, the tissue, and/or the antibodies. In such cases, normal serum from a different species to the host of the secondary antibody could be used to block binding sites, as long as it does not cross-react with the primary or secondary antibody. 14
This article aimed to document observations of autofluorescence and nonspecific immunofluorescent labeling (as well as possible solutions to this problem) specifically in bovine tissue. The source of autofluorescent pigments of frozen bovine intestinal tissue was determined, and the ability of two histochemical stains to mask these pigments was assessed. In addition, we attempted to spectrally separate specific immunofluorescent labeling from nonspecific immunofluorescent labeling, as well as assessing the contribution of common coagulant fixatives and normal serum blocking solutions to background autofluorescence and nonspecific immunofluorescent labeling.
Methods
Snap-Freezing Protocol
A dry ice bath was prepared by combining 95% ethanol with dry ice and mixed until a slurry consistency was achieved. Isopentane (Sigma-Aldrich, St. Louis, MO) was added to a tin cup and the cup was placed into the dry ice bath. Mid-ileal intestinal samples were collected from one healthy cow and one cow naturally infected with Mycobacterium avium subspecies paratuberculosis (MAP), an enteric pathogen causing severe inflammation with a concomitant influx of macrophages. 21 The cows used in the current study were selected based on the presence of a high level of tissue autofluorescence when compared with other cow tissues within a cohort used for experimental study. The mid-ileal intestinal samples were washed with phosphate-buffered saline, pH 7.4, and a cross-section was positioned luminal side down on a section of liver covered with Tissue-Tek optimal cutting temperature (O.C.T.; Sakura Finetek, Torrance, CA). An O.C.T. liver sandwich was used to protect the villi during the freezing process and to ascertain tissue orientation following freezing. The intestine-liver sample was wrapped in foil and placed in the isopentane for at least 5 min. The snap-frozen sample was transferred to dry ice for transport to storage at −80C, where it remained until tissue sectioning could be performed.
Tissue Sections
The mid-ileal intestinal samples were removed from −80C and placed in a cryostat at −20C for at least 30 min before sectioning. Tissue samples were embedded in O.C.T., cut in 6-µm sections and adhered to ColorFrost Plus microscope slides (Thermo Fisher Scientific, Carlsbad, CA). Tissue sections were allowed to air-dry overnight at room temperature before fixing for 5 min at −20C. Tissue sections were stored at −80C until immunofluorescence staining could be performed.
Fixatives
Four fixatives were compared for their ability to fix tissue to slides without requiring antigen retrieval. The fixatives investigated were isopropanol, acetone, methanol, and 1:1 acetone methanol, and were compared with unfixed (air-dried) slides. The standard fixative for frozen tissue sections in our laboratory is 1:1 acetone methanol. The tissue sections were placed in the fixing medium at −20C for 5 min. Fixed slides were air-dried at −20C. For unfixed tissue, slides were allowed to air-dry at room temperature overnight before mounting. All slides were rehydrated with 0.05 M Tris buffer for 10 min before mounting in ProLong Gold Antifade Mountant (Thermo Fisher Scientific) and Richard-Allen Scientific “Slip-Rite” Cover Glass 1.5 (Thermo Fisher Scientific). The mounting medium was allowed to cure for at least 30 min at room temperature before imaging. The slides were assessed for autofluorescence using a confocal microscope with a 488-nm solid-state diode laser and emission collected between 500 and 550 nm. The fixative that demonstrated the lowest mean fluorescence intensity (MFI) was used for all remaining protocols.
Blocking With Normal Serum
The standard blocking solution for IHC protocols in our laboratory includes a 10% normal serum blocking solution. Goat and horse normal serum (Equitech-Bio, Inc., Kerrville, TX) were compared as 5% and 10% dilutions in 0.05 M Tris buffer and 0.05 M Tris buffer with 0.2% Tween-20 and 0.9% sodium chloride (NaCl). Following tissue rehydration as described previously, tissue sections were blocked with 100 µL of blocking solution for 30 min at room temperature in a humidified chamber. Slides were then mounted as described previously and assessed for autofluorescence using a confocal microscope with a 488-nm solid-state diode laser and emission collected between 500 and 550 nm. Following assessment of tissue sections for autofluorescence, blocking solutions that demonstrated the lowest MFI were assessed for effects on immunofluorescent labeling and compared with unblocked tissue. Immediately following the completion of blocking incubation, the blocking solution was poured from the slide. The slides were not rinsed in-between blocking and primary antibody incubation. Unblocked tissue was incubated with 100 µL of 0.05 M Tris for 30 min at room temperature in a humidified chamber as a replacement for the blocking solution. Slides were mounted as described previously. Immunofluorescent labeling (see the “Immunofluorescence Protocol” section) was assessed using a confocal microscope with a 488-nm solid-state diode laser and emission collected between 500 and 550 nm. The blocking solution that demonstrated the lowest MFI was used for all remaining protocols that required incubation with a primary and secondary antibody.
Masking of Autofluorescence
SBB (Eastman Kodak Company, Rochester, NY) and DAB (Vector Laboratories, Burlingame, CA) were compared for their ability to quench or mask autofluorescence. The SBB was prepared as 0.3% in 70% ethanol stirred in the dark for 2 hr, 16 and DAB was prepared as per manufacturer’s instructions. Following tissue rehydration as described previously, tissue sections were incubated with 100 µL of DAB, SBB, or 100 µL each of DAB and SBB for 10 min at room temperature in a humidified chamber. Slides with SBB (SBB and DAB + SBB) were rinsed with 70% ethanol and all slides were washed for 5 min in 0.05 M Tris buffer with 0.2% Tween-20 and 0.9% NaCl buffer. Slides were then mounted as described previously and assessed for autofluorescence using a confocal microscope with a 488-nm solid-state diode laser and emission collected between 500 and 550 nm. Following the assessment of tissue sections for autofluorescence, all masking combinations were also assessed for effects on immunofluorescent labeling. Following tissue rehydration, slides were incubated with a masking solution, which was followed by incubation of primary and secondary antibodies with or without a blocking solution. Slides were mounted as described previously and MFI was assessed using a confocal microscope with a 488-nm solid-state diode laser and emission collected between 500 and 550 nm.
Immunofluorescence Protocol
The primary antibody used in this study was a monoclonal mouse antimacrophage surface antigen (clone AM-3K; Abnova, Taipei, Taiwan), diluted 1:200 in 0.05 M Tris buffer. The primary antibody was fluorescently labeled with a cross-adsorbed Alexa Fluor 488 goat anti-mouse IgG (heavy and light chain) secondary antibody (Thermo Fisher Scientific), diluted 1:1000 in 0.05 M Tris buffer.
Tissue sections were removed from −80C and allowed to equilibrate to room temperature for 10 to 20 min. A liquid blocker “Pap” pen was used to draw a hydrophobic barrier around the tissue and allowed to dry. Following tissue rehydration, blocking and/or autofluorescence quenching, the primary and secondary antibodies were incubated for 60 min each at room temperature in a humidified chamber. The slides were washed following the primary and secondary antibody incubations three times for 5 min, alternating 0.05 M Tris and 0.05 M Tris with 0.2% Tween-20 and 0.9% NaCl buffers. Slides were mounted as described previously.
Confocal Imaging
The tissue sections were examined with an A1 Resonance Plus inverted microscope (Nikon, Melville, NY) equipped with a four-laser gallium–arsenide–phosphide/normal photomultiplier tube (PMT) detector unit (DU4; GaAsP, 488 and 561; PMT, 405 and 640), Galvano resonant scanner, and Nikon Imaging Software (NIS) Elements Advanced Research software (version 4.50.00). Images were acquired by sequential scanning to avoid fluorescence cross-over using a 405/488/561/640 dichroic mirror. Following manual visualization of slides before imaging, intestinal autofluorescence was observed to be most abundant and brightest throughout green wavelengths. All slides were imaged using a 488-nm solid-state diode laser and 525/50 bandpass filter, with emission collected between 500 and 550 nm. All images were captured using a 20× Plan Apo lambda objective (1024 × 1024 pixels), numerical aperture 0.75, pinhole 1.2 AU, and exposure 6.2 sec per pixel dwell. Detector sensitivity (gain) and laser power settings were kept the same for all collected images to allow comparisons between images, treatments, and cows. On collection of each image, the NIS Elements Advanced Research software automatically calculates the MFI for the whole image. A total of five images per slide were collected to perform statistical analysis.
Spectral Separation
Acquisition of spectral images (515 × 512 pixels), encompassing up to 32 channels (420–740 nm), was performed using an A1-DUS spectral detector (Nikon). The Nyquist theorem was used to create the best possible image for the chosen optical resolution. On selecting this option, thresholding was performed using a spectral frequency look-up table. Spectral frequency was thresholded to between 2000 and 3000 to avoid oversaturation. Once thresholded, two regions of interest (ROIs) were selected, one representing labeling with the Alexa Fluor 488 secondary antibody, and one representing background autofluorescence/nonspecific immunofluorescent labeling. The spectral profiles for each ROI were then compared with a library reference for Alexa Fluor 488, provided by the NIS Elements Advanced Research software (Fig. 1). It was particularly important that there be a high degree of similarity between the spectral profiles for the Alexa Fluor 488 ROI and the Alexa Fluor 488 library reference. Once the spectral profiles of the ROI were defined and added to the reference library, unmixing of previously collected spectral images for the labeled unblocked and blocked slides was performed. Using the Spectral Unmixing Settings option in the NIS Elements Advanced Research Software, the defined ROIs for Alexa Fluor 488 labeling and background were added to the unmixing elements table. Selecting “Unmix” identifies only those spectral profiles that match the ROIs added to the unmixing elements table, resulting in two images: one image that identifies the Alexa Fluor 488 ROI and one image that identifies the background ROI.
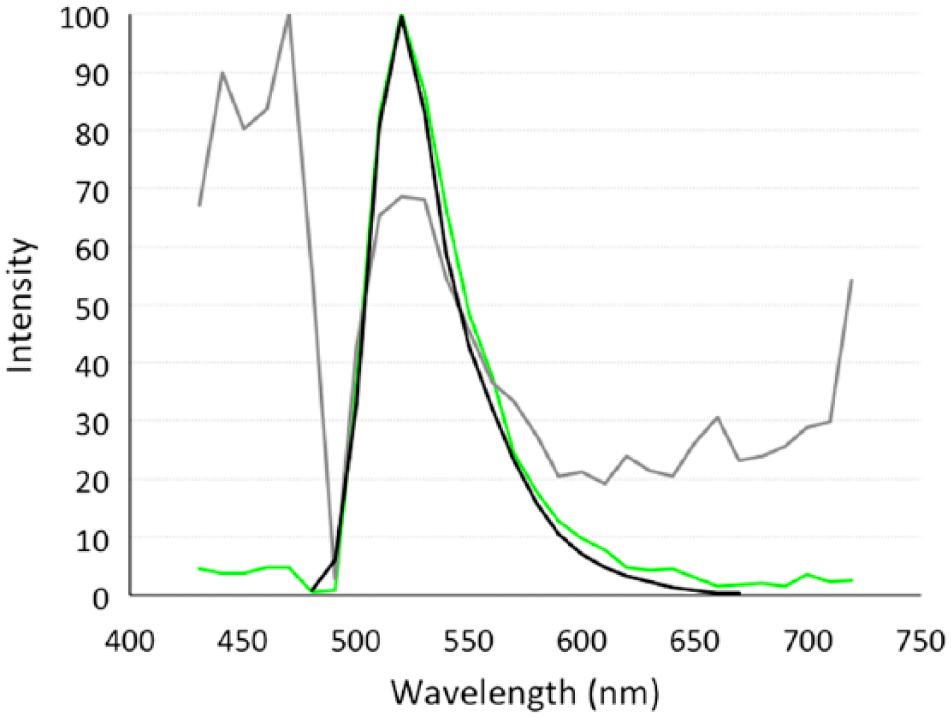
An example of the spectral profile used to separate macrophage surface antigen, clone AM-3K, immunofluorescently labeled with Alexa Fluor 488 (green) from background (gray) in frozen bovine mid-ileal intestinal tissue. Tissue was blocked with 10% horse serum and compared with the Nikon Imaging Software (NIS) Elements library reference for Alexa Fluor 488 (black), excitation 488 solid-state diode laser and emission bandwidth 525/50 nm. Scale bar = 50 µm.
Statistical Analysis
When no apparent differences were observed between the two cows (n=5), the results were combined to compare each treatment within each protocol (n=10).
Descriptive statistics, including mean, standard deviation (SD), and 95% confidence intervals (CIs) were calculated for each treatment, within each protocol, and for each cow, using SAS Enterprise Guide, version 4.3.
Results
Fixative
Differences in the fluorescence intensity of intestinal tissue autofluorescence from a control cow and a cow naturally infected with MAP were observed with the investigated fixatives (Fig. 2); however, differences between fixative treatment groups were not significant (p>0.05). Methanol and 1:1 acetone methanol demonstrated the lowest fluorescence intensity for unlabeled tissue when compared with unfixed/air-dried tissue, with MFIs of 160.0 and 160.5 pixels, respectively (Table 1). Fixation with either acetone or isopropanol proved disadvantageous with observed increases in tissue autofluorescence when compared with unfixed/air-dried tissue (Table 1). When comparing between cows, fixation with methanol resulted in the lowest MFI for both the healthy control cow and the MAP cow, with a 22.2% and 7.3% decrease, respectively (Table 1); however, only the MFI for the healthy control cow was significantly lower (p<0.05) when compared with unfixed/air-dried tissue (Table 1).
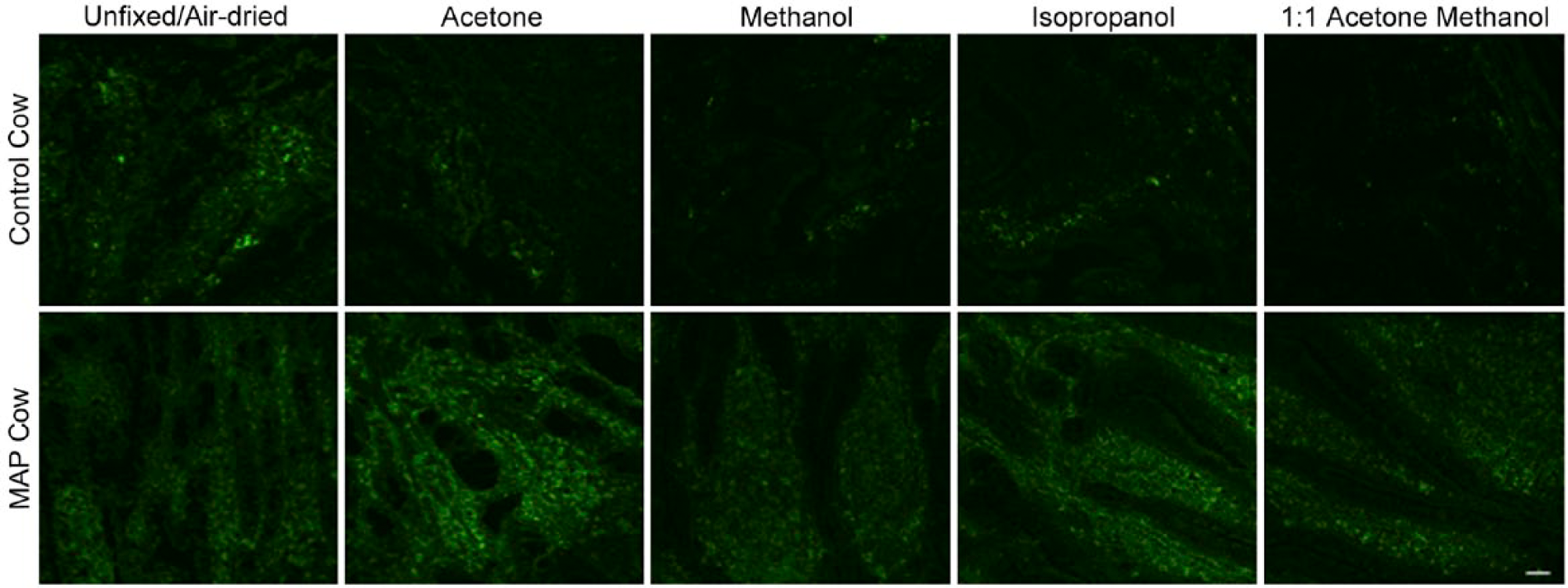
Autofluorescence observed in frozen bovine mid-ileal intestinal tissue collected from a healthy control cow and MAP cow. Tissue was unfixed/air-dried, or fixed with acetone, methanol, isopropanol, or 1:1 acetone methanol, excitation 488 solid-state diode laser and emission bandwidth 525/50 nm. Scale bar = 50 µm. Abbreviation: MAP = Mycobacterium avium subspecies paratuberculosis.
Mean Fluorescence Intensity of Inherent Tissue Autofluorescence of Mid-Ileal Intestinal Tissue Fixed With Acetone, Methanol, Isopropanol, or 1:1 Acetone Methanol, Compared With Air-Dried Unfixed Tissue.
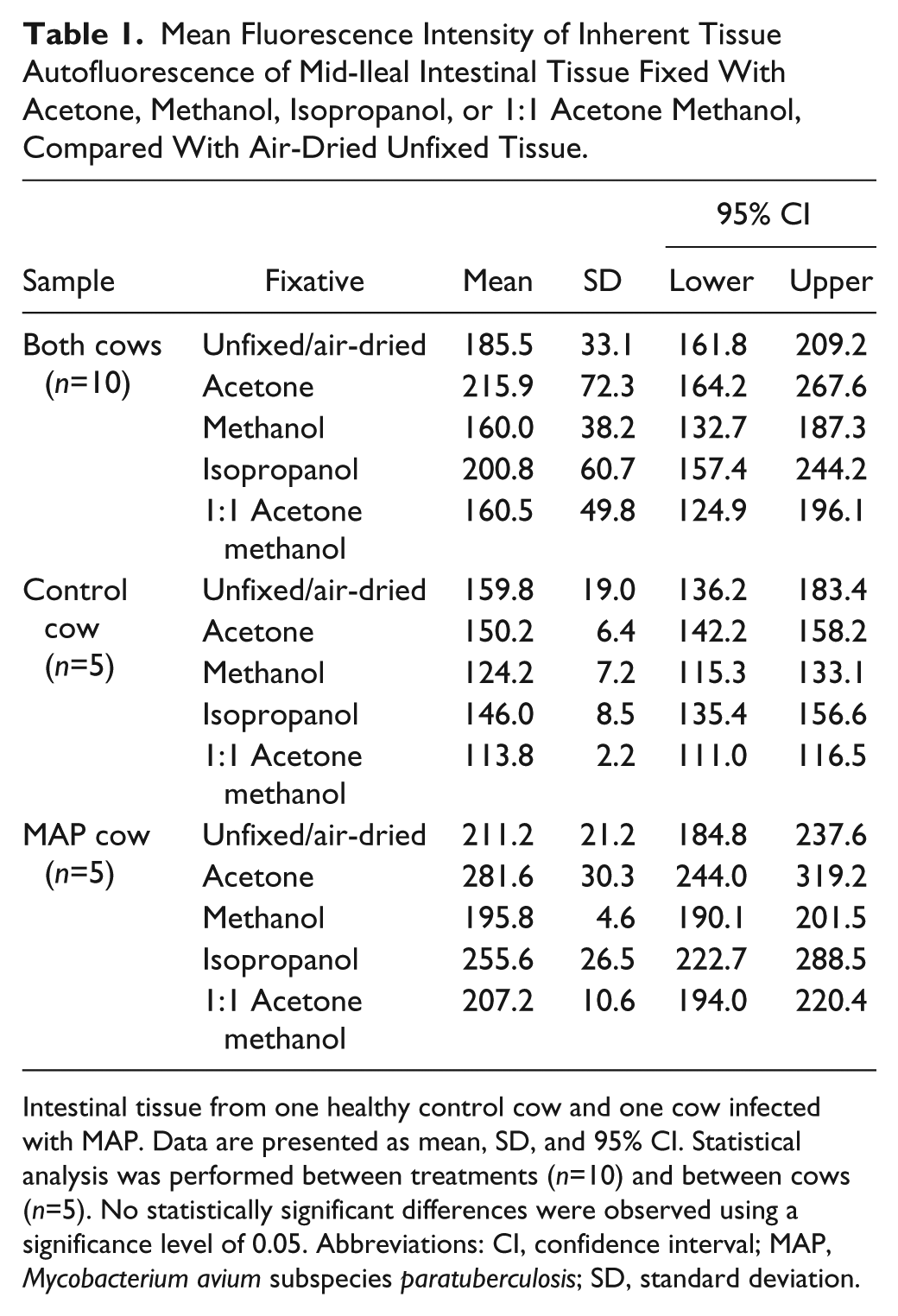
Intestinal tissue from one healthy control cow and one cow infected with MAP. Data are presented as mean, SD, and 95% CI. Statistical analysis was performed between treatments (n=10) and between cows (n=5). No statistically significant differences were observed using a significance level of 0.05. Abbreviations: CI, confidence interval; MAP, Mycobacterium avium subspecies paratuberculosis; SD, standard deviation.
Blocking With Normal Serum
Intestinal tissue autofluorescence was visible for all blocking solutions investigated, for both cows, although differences were not significant (p>0.05). When comparing between treatments, and on average, normal serum diluted in 0.05 M Tris buffer demonstrated a lower MFI for unlabeled tissue when compared with normal serum diluted in 0.05 M Tris buffer with 0.2% Tween-20 and 0.9% NaCl (Table 2). In addition, horse serum demonstrated a lower MFI when compared with goat serum, regardless of dilution buffer (Table 2), although concentration of the serum did not further affect this reduction. On average, the MAP-infected cow demonstrated a higher MFI when compared with the healthy control cow; however, differences between treatments within each cow were not significantly different (p>0.05). In addition, no significant differences were observed in MFIs of macrophage surface antigen immunofluorescent labeling when comparing tissue blocked with 5% and 10% horse serum diluted in 0.05M Tris buffer and unblocked tissue (p>0.05).
Mean Fluorescence Intensity of Inherent Tissue Autofluorescence of Mid-Ileal Intestinal Tissue Blocked With 5% or 10% Horse or Goat Serum Diluted in 0.05 M Tris Buffer or 0.05 M Tris Buffer With 0.2% Tween-20 and 0.9% NaCl.
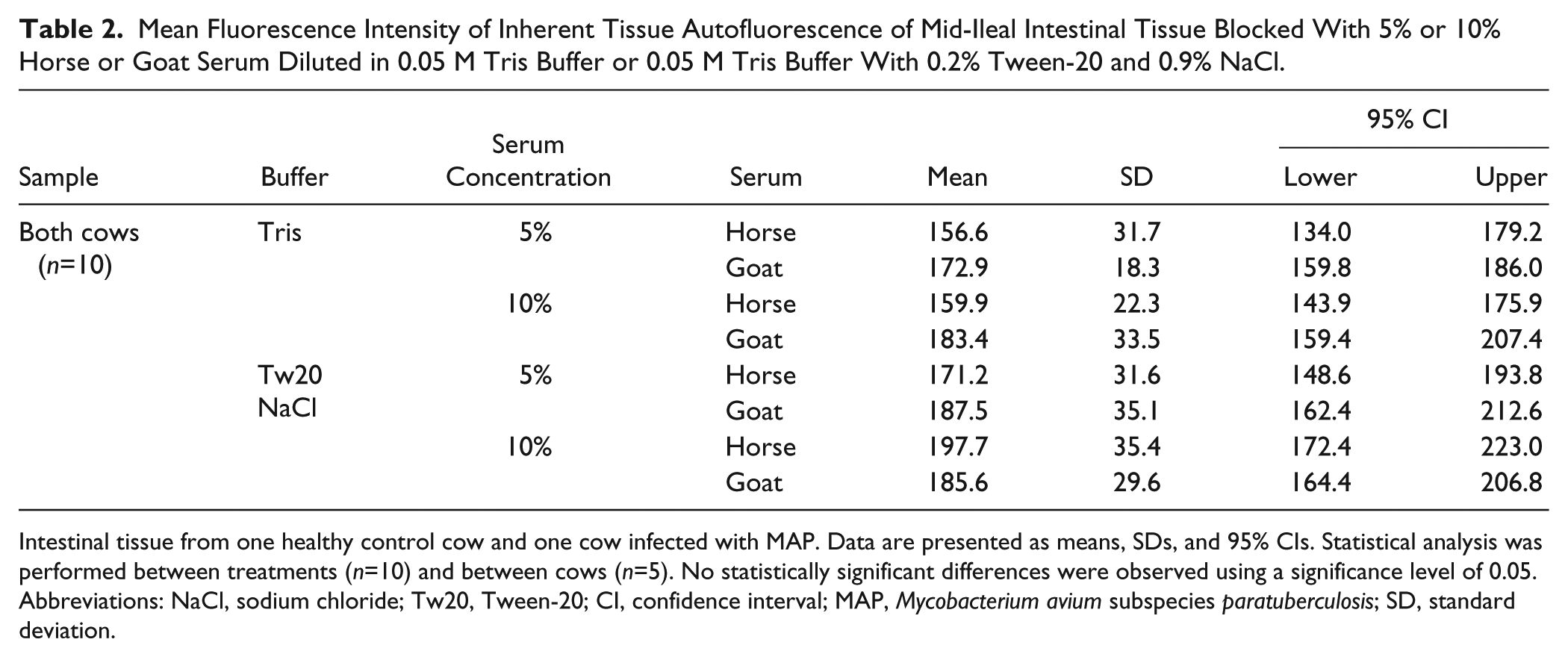
Intestinal tissue from one healthy control cow and one cow infected with MAP. Data are presented as means, SDs, and 95% CIs. Statistical analysis was performed between treatments (n=10) and between cows (n=5). No statistically significant differences were observed using a significance level of 0.05. Abbreviations: NaCl, sodium chloride; Tw20, Tween-20; CI, confidence interval; MAP, Mycobacterium avium subspecies paratuberculosis; SD, standard deviation.
Masking of Autofluorescence
A comparison of DAB, SBB, and DAB + SBB stains to quench autofluoresence was performed and is highlighted in Fig. 3. Although an additional variable of unblocked tissue was compared with previously optimized conditions of 10% horse serum in 0.05 M Tris buffer, no significant differences were observed; thus, the results presented constitute effects of the quenching reagents on tissues blocked with 10% horse serum only.
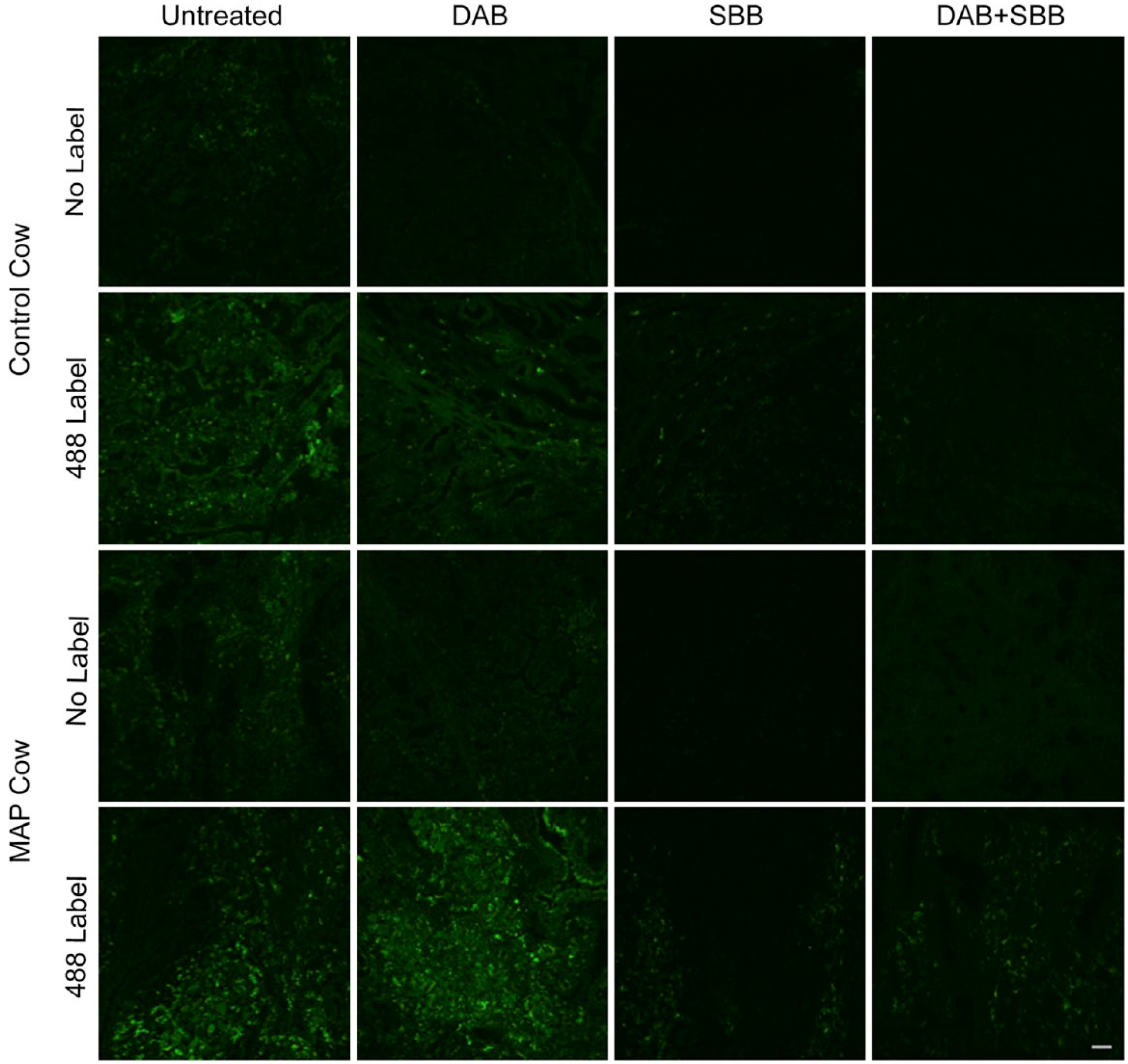
Autofluorescence and immunofluorescent labeling of macrophage surface antigen (clone AM-3K) with Alexa Fluor 488, observed in frozen bovine mid-ileal intestinal tissue collected from a healthy control cow and MAP cow. Tissue was fixed with 1:1 acetone methanol, blocked with 10% horse serum diluted in 0.05 M Tris buffer and autofluorescence masked with DAB, SBB, DAB + SBB, or untreated, excitation 488 solid-state diode laser and emission bandwidth 525/50 nm. Scale bar = 50 µm. Abbreviations: MAP, Mycobacterium avium subspecies paratuberculosis; DAB, 3,3′-diaminobenzidine; SBB, Sudan Black B.
Tissues treated with SBB demonstrated a significantly lower (p<0.05) MFI compared with DAB and DAB + SBB, averaging 91.2, 147.0, and 123.2 pixels, respectively. Similar trends were noted for both cows, regardless of infection status (Fig. 3).
Further evaluation of the effects of masking reagents on specific immunofluorescent labeling of macrophage surface antigen (AM-3K) showed that treatment with SBB (alone or in combination with DAB) resulted in a significant reduction (44.8% and 43.9%, respectively) in labeling compared with tissues treated with DAB alone (9.2%; Table 3).
Mean Fluorescence Intensity of Inherent Tissue Autofluorescence (No Label) and Macrophage Surface Antigen (488 Label) of Mid-Ileal Intestinal Tissue Masked With DAB, SBB, or a Combination of DAB and SBB for Tissue Blocked With 10% Horse Serum.
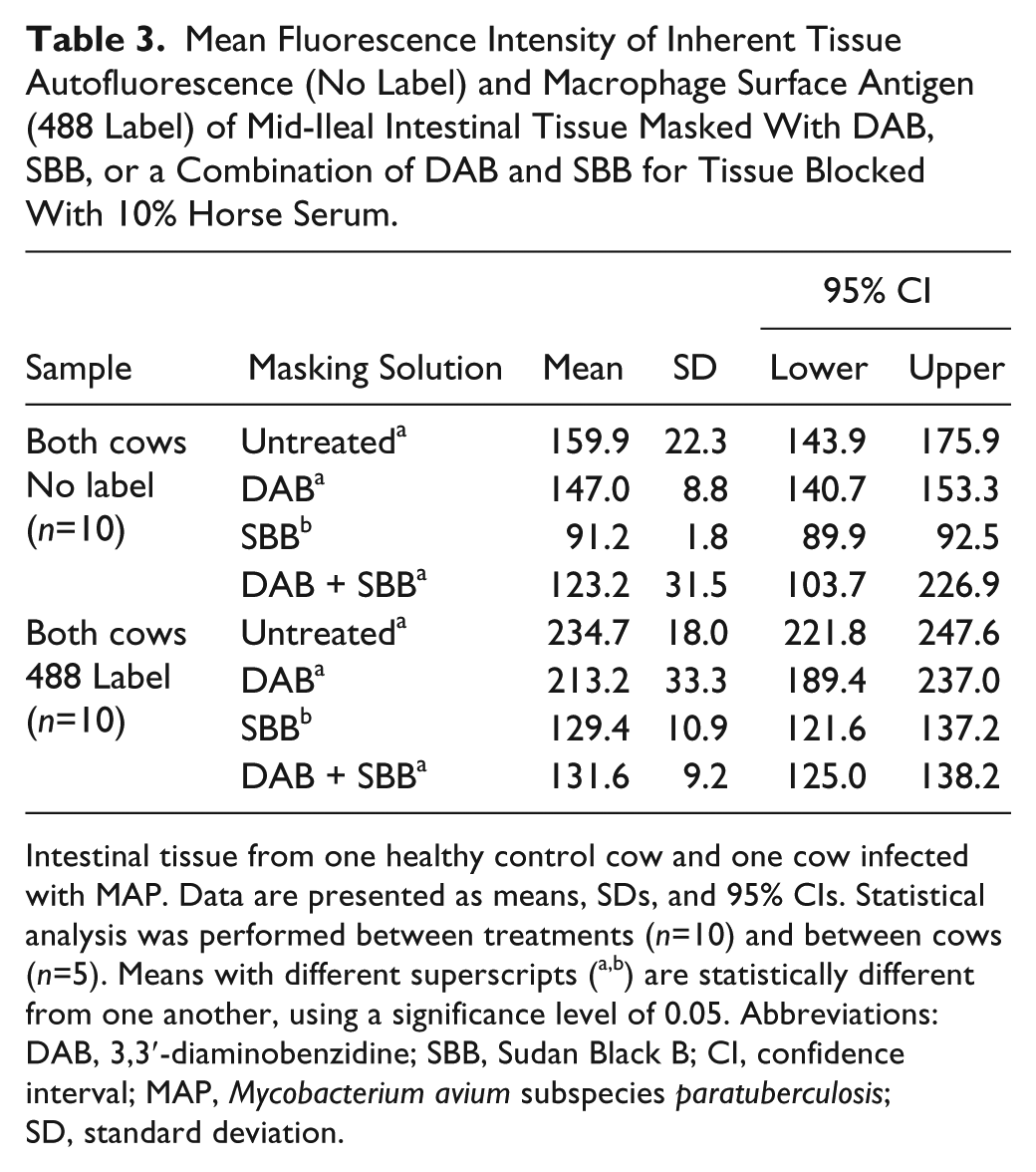
Intestinal tissue from one healthy control cow and one cow infected with MAP. Data are presented as means, SDs, and 95% CIs. Statistical analysis was performed between treatments (n=10) and between cows (n=5). Means with different superscripts (a,b) are statistically different from one another, using a significance level of 0.05. Abbreviations: DAB, 3,3′-diaminobenzidine; SBB, Sudan Black B; CI, confidence interval; MAP, Mycobacterium avium subspecies paratuberculosis; SD, standard deviation.
Spectral Separation
Again, an additional variable of unblocked tissue was compared with tissue blocked with 10% horse serum diluted in 0.05 M Tris buffer; however, no significant differences were observed, and therefore, the results presented are for spectral separation of the immunofluorescent label for tissue blocked with 10% horse serum only.
The MAP cow demonstrated a significantly lower MFI (37.0 pixels) for ROI classified as AM-3K-488, when compared with the healthy control cow (79.2 pixels; p<0.05; Table 4). The MAP cow also demonstrated a significantly higher MFI (206.8 pixels compared with 173.6 pixels) for the ROI classified as background (p<0.05). The observed difference in spectral separation between cows was also confirmed by immunofluorescent labeling (Fig. 4).
Mean Fluorescence Intensity of Spectrally Separated Macrophage Surface Antigen (AM-3K-488) and Background in Mid-Ileal Intestinal Tissue Blocked With 10% Horse Serum.
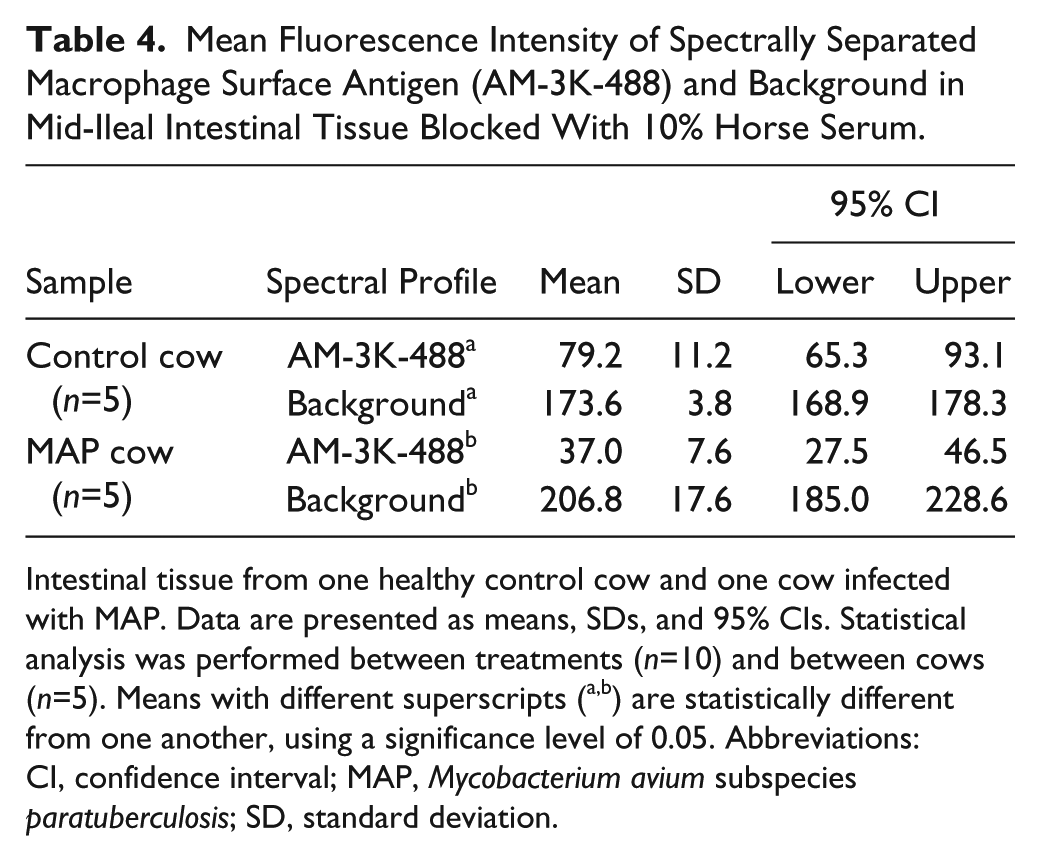
Intestinal tissue from one healthy control cow and one cow infected with MAP. Data are presented as means, SDs, and 95% CIs. Statistical analysis was performed between treatments (n=10) and between cows (n=5). Means with different superscripts (a,b) are statistically different from one another, using a significance level of 0.05. Abbreviations: CI, confidence interval; MAP, Mycobacterium avium subspecies paratuberculosis; SD, standard deviation.
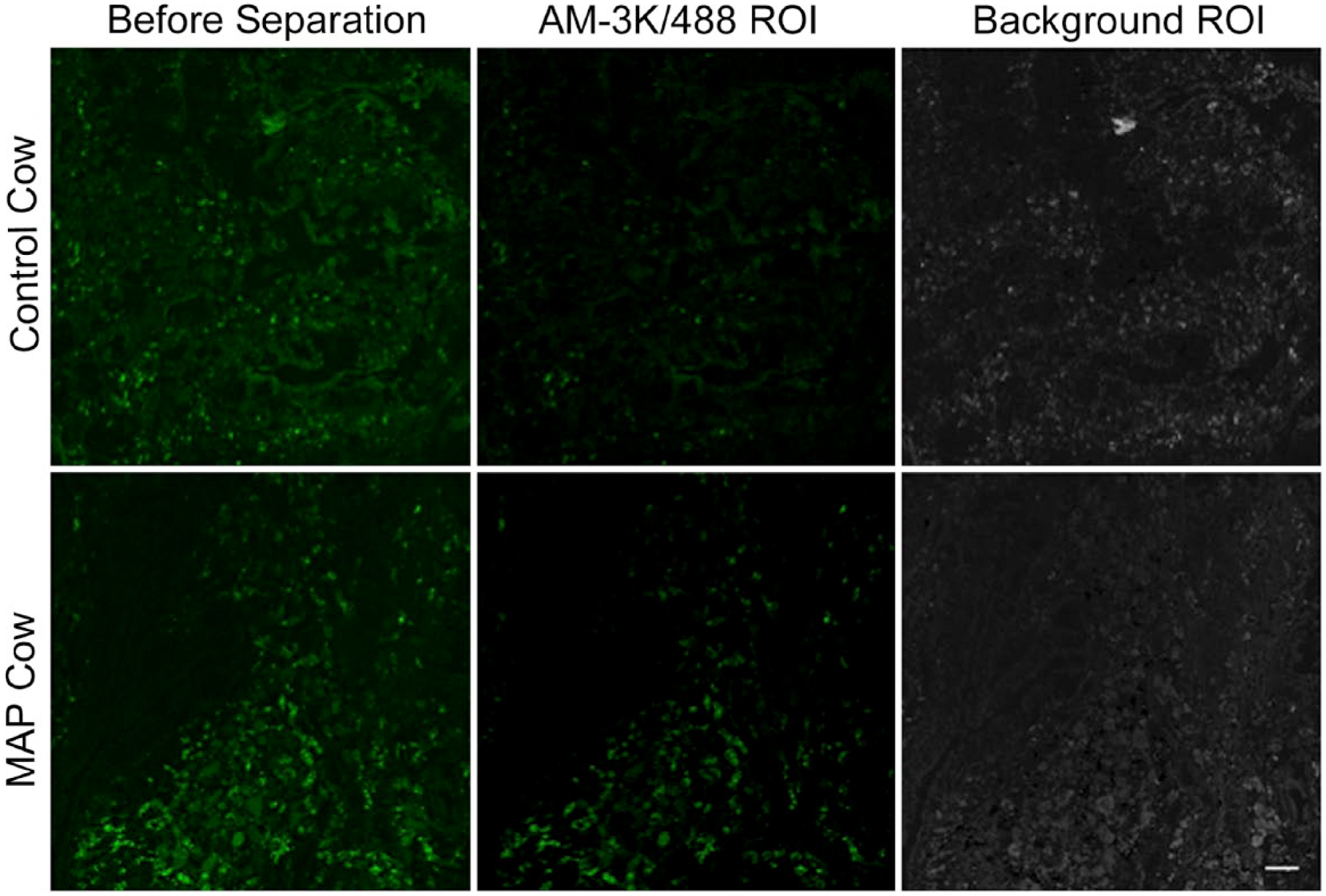
Immunofluorescent labeling of macrophage surface antigen (clone AM-3K) observed in frozen bovine mid-ileal intestinal tissue collected from a healthy control cow and MAP cow. Tissue was fixed with 1:1 acetone methanol and blocked with 10% horse serum diluted in 0.05 M Tris buffer. ROIs were used to spectrally separate AM-3K-488-specific immunofluorescent labeling from nonspecific background, excitation 488 solid-state diode laser and emission bandwidth 525/50 nm. Scale bar = 50 µm. Abbreviations: MAP, Mycobacterium avium subspecies paratuberculosis; ROI, region of interest.
Discussion
Naturally fluorescence components of intestinal tissue often limit the use of green fluorophores in immunofluorescence. Typical autofluorescence is most visible throughout green wavelengths, 2 making it difficult to discern specific immunofluorescent labeling and can eliminate the use of green fluorophores in multicolor experiments. In the current study, inherent autofluorescence in bovine intestinal tissue was most apparent when excited with 488 nm (emission 500–550 nm) and 561 nm (emission 570–620 nm) diode lasers. Statistical analysis was restricted to autofluorescence observed when tissue was excited using the 488-nm diode laser, as fluorescence intensity was brightest in this emission range, and more difficult to discern from specific immunofluorescent labeling (data not shown).
Autofluorescence caused by eosinophils is due to flavin dinucleotide (FAD), reduced nicotinamide-adenine dinucleotide (NADH) and reduced nicotinamide-adenine dinucleotide phosphate (NADPH). The emission of autofluorescence caused by FAD, NADPH, and NADH has been demonstrated between 500 and 560 nm, 2 445 and 520 nm, 2 and between 440 and 470 nm, 22 respectively. Lipofuscin pigments are residual bodies of undigested or oxidized lipids thought to result from the enzymatic separation of cellular organelles within lysosomes. 23 Lipofuscin can demonstrate autofluorescence across a broader emission range than eosinophils, and has been demonstrated using ultraviolet, fluorescein, rhodamine, and Cy5 filters, 8 and using 488- and 543-nm lasers. 15 Based on this previous research, and the significant level of autofluorescence observed when using the 488 and 561 lasers, we concluded that the autofluorescence observed in our frozen bovine intestinal tissue sections was most likely due to eosinophils (FAD and NADPH), lipofuscin, or a combination of both.
The masking of resident autofluorescent components of tissue has previously been performed using both DAB 18 and SBB.8,15,16 In the current study, the MFI of tissue treated with DAB was higher when compared with tissue treated with SBB. This finding indicates that the mid-ileal tissue contained more autofluorescence due to lipofuscin, rather than eosinophils, as the MFI recorded is a result of any inherent autofluorescence not quenched by the assigned treatment. This result was also replicated when comparing between cows; however, the MAP cow demonstrated significantly higher MFI for tissue treated with DAB and a combination of DAB and SBB, when compared with the healthy control cow. It is likely that the disease status of the MAP cow contributed to higher levels of eosinophilic and lipofuscin pigments. The gastrointestinal tract is the primary site for normal eosinophil and macrophage residence, and both are involved in the host response to pathogens.24–26 In a study by Monif, 27 cows determined to have moderate to high levels of acid-fast bacilli in the mid-ileum tended to have low numbers of eosinophils. However, this is in contrast to the MAP cow used in the current study, as acid-fast staining of mid-ileal tissue showed a high number of acid-fast bacilli (data not shown). Conversely, a high level of pathogen infiltration is often associated with a higher number of intestinal macrophages. 21 In addition, the addition of SBB (either alone or in combination with DAB) demonstrated the greatest reduction in MFI of the 488-nm label. This result is most likely the “truest” indication of the MFI of the fluorescent label alone; however, it is possible that this reduction in MFI also includes a reduction in immunofluorescent labeling. Therefore, the effect of SBB on immunofluorescent labeling should be individually assessed for each fluorescence label to be utilized.
Of the coagulant fixatives investigated, methanol and a combination of 1:1 acetone methanol demonstrated the lowest MFIs; however, as methanol alone can denature proteins, 19 its use in conjunction with acetone and a cold fixation procedure, helps maintain enzyme activity. 19
The current study also observed no significant differences between blocking solutions containing 5% and 10% dilutions of normal serum, although horse serum (on average) demonstrated a lower MFI when compared with goat serum. It is possible that the antigenic similarity between bovine and goat IgG 28 contributes to background autofluorescence; thus, goat serum may be unsuitable for inclusion in a protocol with bovine tissues. In addition, blocking solutions that contain Tween-20 and NaCl may also contribute negatively to MFI, despite being purported to reduce hydrophobic, ionic, and electrostatic interactions. 29
In addition, no significant differences were observed for immunofluorescent labeling for unblocked and blocked tissue. This result is in agreement with a study by Buchwalow, 30 who found no differences in immunostaining between blocked and unblocked tissue sections for paraffin-embedded, frozen, and paraformaldehyde and acetone fixed tissue sections. This study concluded that endogenous Fc receptors do not retain their ability to bind the Fc portion of IgG after routine fixation, and thus, the inclusion of a blocking solution in IHC protocols is unnecessary. 30
The current study also assessed the ability of imaging software to spectrally separate specific immunofluorescent labeling from background. Although there were no significant differences between unblocked and blocked tissue, there were significant differences between cows. Despite ROIs being defined and collected in the same way for both cows, the healthy control cow demonstrated a significantly higher MFI for the AM-3K label compared with the MAP cow. However, it was noted that specific immunofluorescent labeling in the MAP cow tissue was more intense and widespread throughout the tissue, when compared with the healthy control cow tissue, which showed more, lower intensity labeling. The author’s believe that this was due to the incorrect identification of background/nonspecific immunofluorescent labeling as the fluorescent label. Although spectral profiles are independent of fluorescence intensity, it is possible that the more specifically that a marker is fluorescently labeled (e.g., increased expression due to disease status), the more accurately the software can identify the spectral profiles, resulting in a more accurate separation.
In summary, frozen bovine intestinal tissue sections contain inherent fluorescent pigments caused by lipofuscin and eosinophils, with an emission range between 500 and 550 nm and 570 and 620 nm, and abundance of these fluorescent pigments seems associated with tissue-specific immune responses. Masking of the fluorescent pigments was most successful when using SBB (either alone or in combination with DAB); however, this may also result in a reduction in immunofluorescent labeling. A fixative consisting of 1:1 acetone methanol demonstrated less influence on background autofluorescence, whereas the use of blocking solutions may in fact be unnecessary. The spectral separation of specific immunofluorescent labeling from background may be the simplest solution for “removing” unwanted fluorescence; however, the accuracy of this method is dependent on the quality of the immunofluorescent labeling to be separated. Although many of the experiments performed in this study yielded results that were not statistically significant, it should be stressed that there is not a “one-protocol-fits-all” approach for immunofluorescence or, indeed, IHC experiments. Researchers should reassess their protocols whenever an element (tissue, reagent, antibody) is adjusted, which will result in the most biologically relevant data possible.
Footnotes
Acknowledgements
The authors would like to thank Judith Stasko and Adrienne Shircliff of the National Animal Disease Center Histology and Microscopy Services Unit, for their technical experience and expertise.
Competing Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
JRS developed original concept and obtained funding. CJJ performed protocol development, immunohistochemistry, image analysis, and drafted the manuscript. All authors have read and approved the final manuscript.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was funded through the National Animal Disease Center, Agricultural Research Service, US Department of Agriculture.
