Abstract
Laryngeal cartilages undergo a slow ossification process during aging, making them an excellent model for studying cartilage mineralization and ossification processes. Pig laryngeal cartilages are similar to their human counterparts in shape and size, also undergo mineralization, facilitating the study of cartilage mineralization. We investigated the processes of cartilage mineralization and ossification and compared these with the known processes in growth plates. Thyroid cartilages from glutaraldehyde-perfused male minipigs and from domestic pigs were used for X-ray, light microscopic, and transmission electron microscopic analyses. We applied different fixation and postfixation solutions to preserve cell shape, proteoglycans, and membranes. In contrast to the ossifying human thyroid cartilage, predominantly cartilage mineralization was observed in minipig and domestic pig thyroid cartilages. The same subset of chondrocytes responsible for growth plate mineralization is also present in thyroid cartilage mineralization. Besides mineralization mediated by matrix vesicles, a second pattern of cartilage mineralization was observed in thyroid cartilage only. Here, the formation and growth of crystals were closely related to collagen fibrils, which served as guide rails for the expansion of mineralization. It is hypothesized that the second pattern of cartilage mineralization may be similar to a maturation of mineralized cartilage after initial matrix vesicles–mediated cartilage mineralization.
Keywords
Introduction
Cell biological events leading to cartilage mineralization and ossification in human laryngeal cartilages are far from being understood. One good way to analyze these processes is provided by animal models, because in them fixation by glutaraldehyde perfusion can be used to guarantee good tissue preservation for transmission electron microscopy (TEM). With regard to the animal model, thyroid cartilages from miniature pigs and domestic pigs are well suited because they share strong similarities with the human tissues in shape and size. Mineralization and ossification of thyroid cartilage are observed in both human and pig cartilages.
Vesalius was first to describe ossification in the laryngeal cartilages of animals. 1 The few reports about mineralization processes in thyroid cartilages of animals are based on X-ray investigations. Friedrich mentioned gender differences during cartilage mineralization and ossification of horse laryngeal cartilages. 2 Furthermore, a significant increase of thyroid cartilage mineralization with advancing age was noted in this species. 3 In deer, Bejsovec observed a progression of mineralization and osteogenesis with advancing age. 4 Mineralization of laryngeal cartilages was also observed in dogs, 5 while ossification was detected in tiger thyroid cartilage. 6 In oxen, deer, common hares, and rabbits, mineralization and ossification were observed as preferably occurring at the insertion zones of laryngeal muscles. 7 Harrison and Denny also obtained similar results for the primate larynx. 8 However, TEM studies of cartilage mineralization of the laryngeal skeleton in animals are lacking, and the underlying mechanisms are not yet completely understood.
Therefore, in this study, we analyzed thyroid cartilage mineralization in minipigs, pigs, and cattle mainly by TEM analysis, focusing on chondrocytes involved in mineralization and on extracellular matrix components. For comparison, we also referred to the mineralization and ossification processes in human thyroid cartilage. 9 A prerequisite to investigating the course of events leading to mineralization is good preservation of cells and extracellular matrix. Therefore, we used different solutions for optimal fixation of cells and matrix.10–13 Thyroid cartilage mineralization was compared with the well-known mineralization process in growth plate.
Materials and Methods
Animals
The left halves of thyroid cartilages and proximal humerus growth plates from 17 male, castrated Munich minipigs (TROLL; Medical Service GmbH, Institute of Clinical Biochemistry, München, Germany; three aged 9–11 months, 13 aged 16–19 months, and one aged 32 months) and from five male castrated and one female domestic pigs (one aged 8 weeks, five aged 8 months, and one aged 3 years) were investigated by X-ray, light microscopic, and transmission electron microscopic methods.
To further analyze cartilage mineralization and ossification in the animal laryngeal skeleton, thyroid cartilages from six cows (one aged 25 months, two aged 2 years, two aged 3 years, and one aged 5 years) and from three bulls (two aged 2 years and one aged 3 years) were investigated by X-rays only.
Thyroid cartilage from a 48-year-old man was used for a comparison of the mineralization processes analyzed by X-rays.
The larynges of Munich minipigs (TROLL) were obtained from an authorized animal experiment performed by the Department of Oral and Maxillofacial Surgery of the medical faculty of Christian-Albrechts University of Kiel.
Fixation and Embedding for TEM
Minipigs were heparinized using Liquemin N 25000 (Roche Holding; Mannheim, Germany) to permit later fixation by perfusion. Animals were sacrificed by a supraphysiological dose of Tilest 500 (Parke-Davis GmbH; Berlin, Germany). After opening the thorax, the arterial system was flushed through the left ventricle with 5 to 8 liter Ringer solution at a pressure of 180 mbar. Afterward, the minipigs were perfused and fixed with a mixture of 2.5% glutaraldehyde and 1.5% formaldehyde dissolved in phosphate buffer (pH 7.4) at a pressure of 180 mbar. In the domestic pigs, which were killed in a slaughterhouse, no perfusion was applied.
To determine the degree of cartilage mineralization and ossification, X-rays of thyroid cartilage plates were performed with a mammography unit (Ergophos 4/Siemens; Siemens AG, Erlangen, Germany) with 50 mAs and 37.5 keV. Documentation was on Kodak X-Omat MA X-ray films (Carestream Health, Inc., Rochester, NY). Left thyroid cartilage plates were divided into nine segments (Fig. 1). A specimen of 2 × 2 × 1 mm3 was dissected out of each of the nine thyroid cartilage segments and out of the growth plate. These specimens were fixed by immersion. Our study was mainly focused on areas with cartilage mineralization and beginning ossification (segments 4B and 6, see Fig. 1) or regions with an interface between unmineralized and mineralized cartilage (segment 5B, see Fig. 1). As the features of cartilage mineralization and ossification in the other segments did not differ much from the selected ones, they were investigated only in some animals. For a good preservation of chondrocytes and extracellular matrix, we used the below immersion solutions. All immersion solutions were adjusted to an osmolarity of 330 mOsmol/l and a pH of 7.4. This procedure also contributed to maintaining the structures.
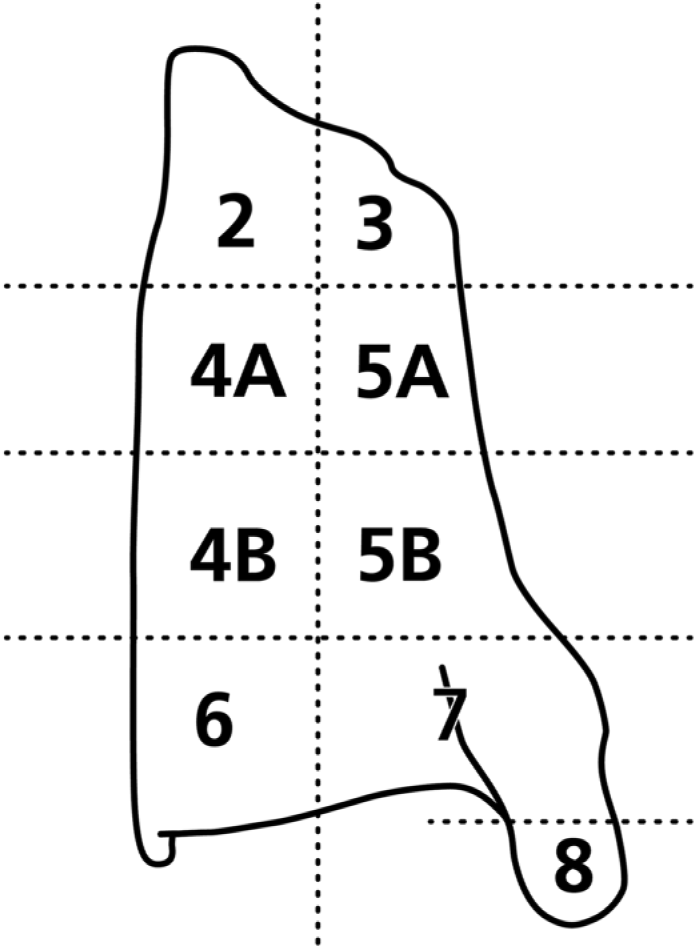
Diagram of a left thyroid cartilage plate in view from outside explaining the dissection. Thyroid cartilage plate was divided into nine segments. Specimens of 2 × 2 × 1 mm size were dissected out of each segment.
Solutions for Immersion Fixation and Postfixation
Immersion solutions used for good preservation of thyroid cartilage chondrocytes and components of extracellular matrix are summarized in Table 1.
Solutions for Immersion Fixation and Postfixation Suited for Different Transmission Electron Microscopic Analyses.
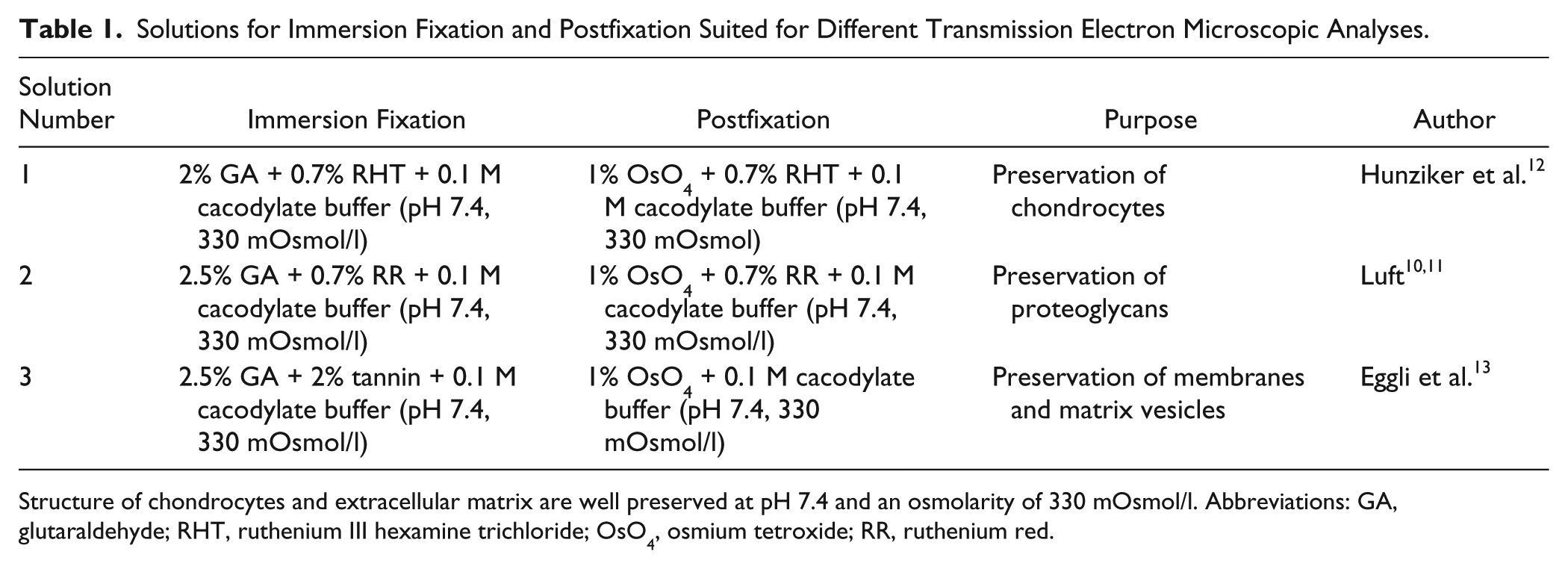
Structure of chondrocytes and extracellular matrix are well preserved at pH 7.4 and an osmolarity of 330 mOsmol/l. Abbreviations: GA, glutaraldehyde; RHT, ruthenium III hexamine trichloride; OsO4, osmium tetroxide; RR, ruthenium red.
For chondrocytes, immersion fixation 1 was used.14,15 The fixation solution contained 2% glutaraldehyde + 0.7% ruthenium III hexamine trichloride + 0.1 M cacodylate buffer. For postfixation, the solution contained 1% osmium tetroxide (OsO4) + 0.7% ruthenium III hexamine trichloride + 0.1 M cacodylate buffer.
For proteoglycans, immersion fixation 2 was used.10,11 The fixation solution contained 2.5% glutaraldehyde + 0.7% ruthenium red + 0.1 M cacodylate buffer. For postfixation, the solution contained 1% OsO4 + 0.7% ruthenium red + 0.1 M cacodylate buffer.
For membranes and matrix vesicles, immersion fixation 3 was used. 13 The fixation solution contained 2.5% glutaraldehyde + 2% tannin + 0.1 M cacodylate buffer. The postfixation solution contained 1% OsO4 + 0.1 M cacodylate buffer.
The postfixed specimens were washed repeatedly in 0.1 M cacodylate buffer (330 mOsmol/l; pH 7.4) and dehydrated in several series of ethanol. Specimens were partly embedded in araldite (five minipigs) and partly in Spurr’s embedding resin (12 minipigs and two domestic pigs). This was done to test which kind of embedding medium was better suited for analyzing mineralized tissues.
Cutting of Sections
Specimens were sawed out of the Spurr and araldite blocks, mounted on supporting blocks, and trimmed with razor blades under a magnifying glass. For light microscopy, 1-µm semithin sections were cut with glass knives on a microtome (Ultracut E; Reichert Jung, Leica Micro-Systems GmbH, Wetzlar, Germany). For TEM, 50 to 70 nm thick ultrathin sections were cut using a diamond knife (DuPont Knife 24124; DuPont, Wilmington, DE). Better cutting was possible if Spurr-embedded specimens were used whereas araldite-embedded specimens revealed more difficult cutting.
The sections were caught on formvar-coated copper grids (Stegnetze Typ 00841180, Drawing: R150A ∅3, Stork Veco B. V., Eerbeek, The Netherlands). After drying, the sections were contrasted in uranyl acetate (Merck 8473; Merck KGaA, Darmstadt, Germany) for 15 min and in lead citrate (Merck 7398; Merck KGaA) for another 15 min.
Measurements of Mineralization and Ossification
Mineralized cartilage areas, together with ossified areas from representative radiographs of the left and right thyroid cartilage plates from minipigs, domestic pigs, and cattle, were measured using the software SigmaScan Pro 5 (Systat Software GmbH; Erkrath, Germany). The radiographs were scanned and the areas were encircling by the use of the software.
The percentage of mineralized and ossified cartilage areas was determined in dependence from the total area of the respective thyroid cartilage plate.
Evaluation by Light Microscopy and TEM
Semithin sections were evaluated by light microscopy using a Zeiss Axiophot microscope (Zeiss; Jena, Germany). Specimens suited for TEM were selected at light microscope magnifications. TEM analysis was performed at EM 900 (Zeiss).
Statistical Analysis
Ossification values of the respective samples were averaged and analyzed with the statistical software Stata12.0 (StataCorp; College Station, TX) in mean percentage ± standard error of the mean (SEM). For illustration purposes, a box plot was created, which illustrates the distribution of smear values and difference between species.
Results
Analysis of Radiographs
Out of radiographs from 17 male minipigs, five male and one female domestic pigs, six cows, and three bulls, we choose only the characteristic ones showing the progress of cartilage mineralization and ossification. In radiographs of thyroid cartilage, spotted and opaque cartilage mineralization characterized by a honeycomb structure can be distinguished clearly from ossification characterized by filigree trabecular architecture reminding of a spider’s web. Radiographs of thyroid cartilages from perfusion-fixed minipigs and domestic pigs or cattle obtained from slaughterhouse did not differ in quality. The high quality of perfusion-fixed material additionally treated by immersion fixation and postfixation (see Table 1) was only visible when light microscopical and TEM techniques were applied.
In thyroid cartilages from about 11-month-old male and female miniature pigs, the male tissues were somewhat larger in height and breadth than the female ones (Fig. 2A and B). Cartilage mineralization had extended to approximately the whole thyroid cartilage plate. However, the dorsal half was not as densely mineralized, particularly in the female specimen (Fig. 2A and B). Bone formation was restricted to the ventral-caudal edge of the thyroid cartilage near the origin of the vocalis muscle (Fig. 2A and B). Compared with the 11-month-old animal, cartilage mineralization was much denser in the thyroid cartilage of a 32-month-old minipig (Fig. 2C). Only the dorsal one third had remained unmineralized. Ossification had extended and now included the ventral-caudal edge and the inferior horn (Fig. 2C). In thyroid cartilage of an 8-week-old male domestic pig, cartilage mineralization was missing (Fig. 2D). In an 8-month-old male domestic pig, cartilage mineralization has included the lower ventral one fourth of thyroid cartilage and parts of the inferior horn (Fig. 1E). Ossification was observed at the ventral-caudal edge. In a 3-year-old male domestic pig, by contrast, cartilage mineralization had spread over nearly the entire thyroid cartilage plate, leaving only a small cartilage window above to the inferior horn unmineralized (Fig. 2F). The lower half of the near ventral border, including the ventral-caudal edge and the inferior horn up to its base, had been ossified (Fig. 2F).
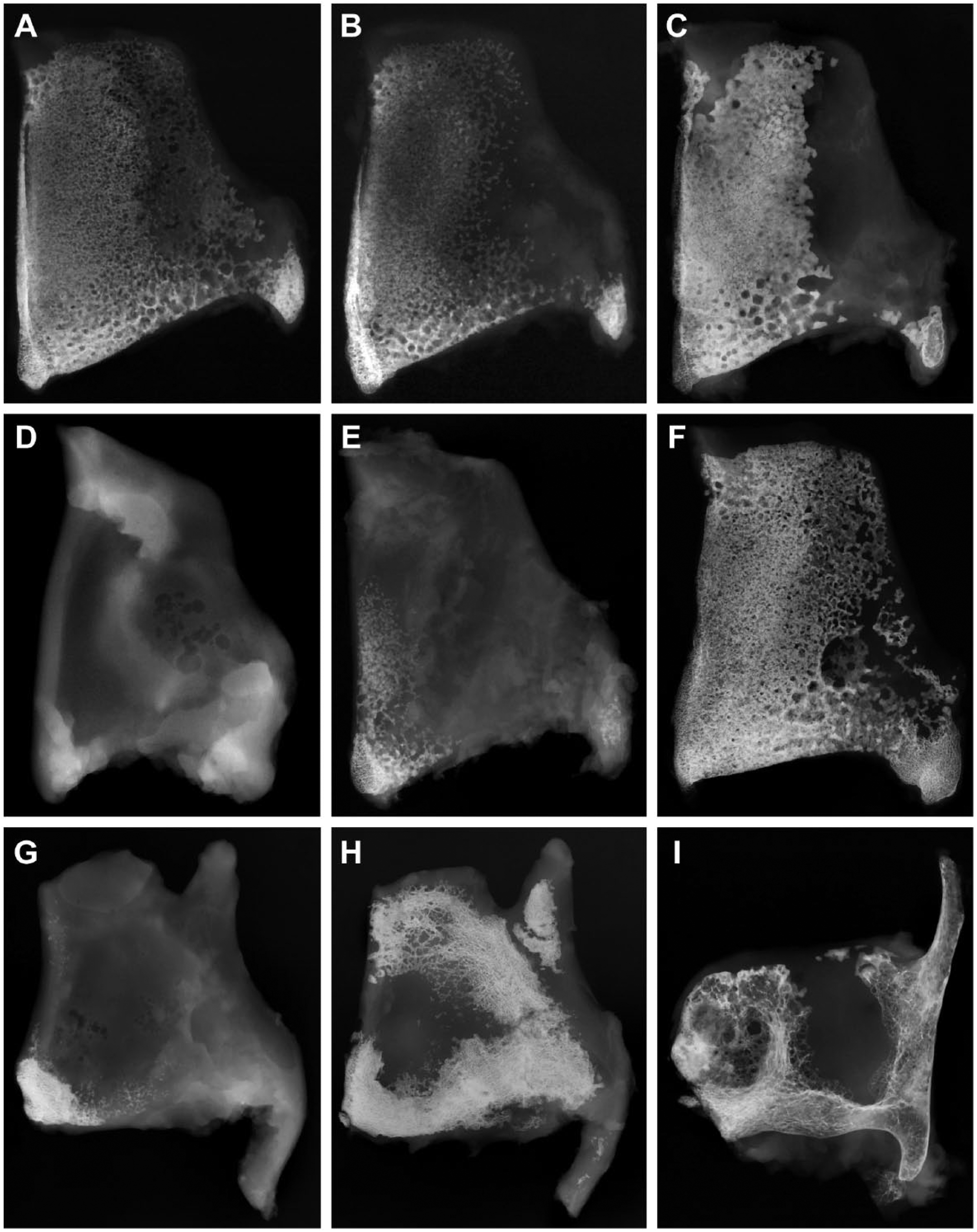
X-ray films from left thyroid cartilage plates from different species showing the extension of cartilage mineralization and ossification. (A) Nearly the whole thyroid cartilage plate is mineralized in an 11-month-old male minipig. Ossification is limited to the inferior ventral edge and the ventral rim. (B) Only three quarters of the thyroid cartilage show cartilage mineralization in a female minipig of the same age. Ossification is limited to the inferior ventral edge and the ventral rim. (C) In a 32-month-old male minipig, somewhat more than half of thyroid cartilage is mineralized. The inferior ventral edge, as well as the inferior horn, shows centers of ossification. (D) No cartilage mineralization was seen in thyroid cartilage of a male domestic pig aged 8 weeks. (E) Mineralized cartilage is limited to a ventral lower strip and the inferior horn in an 8-month-old male domestic pig. The ventral lower edge contains a bony nucleus. (F) Nearly the whole thyroid cartilage plate is mineralized, and ossification is seen in the ventral inferior edge and inferior horn in a 3-year-old male domestic pig. (G) In thyroid cartilage from a 2-year-old bull, only the lower ventral edge shows signs of cartilage mineralization and discrete ossification. (H) By contrast, thyroid cartilage plate of a 5-year-old cow presents broad strips of bone located at the ventral, dorsal, and lower border, including the upper horn. Cartilage mineralization is seen in the upper part of the thyroid cartilage plate while a central cartilage window has remained unmineralized. (I) Thyroid cartilage plate from a 48-year-old man is extensively ossified, leaving only two windows of cartilage unmineralized. In contrast to animals, cartilage mineralization is only discrete. It was limited to small rims at the bone–cartilage interface.
In a 2-year-old bull, only small parts of cartilage near the ventral-caudal edge of the thyroid cartilage plate were mineralized, while a circumscribed area of this region was ossified (Fig. 2G). In a 5-year-old cow, large areas of the thyroid cartilage plate including the upper horn and parts of the inferior horn were ossified, leaving only a ventral–central window of the plate unmineralized (Fig. 2H).
In a 48-year-old man, major areas of thyroid cartilage were ossified (Fig. 2I). Only a small ventral and a somewhat larger dorsal window have remained unmineralized.
Scanning of cartilage mineralization together with ossification in some representative radiographs of left and right thyroid cartilage plate showed that these processes were highly developed in minipigs when compared with domestic pigs and cattle (Fig. 3A). Domestic pigs surpassed cattle significantly in cartilage mineralization and ossification (Fig. 3A). When scanning was focused on ossification only, it became clear that percentual ossification in the sense of cartilage mineralization was low in all three species (Fig. 3B).
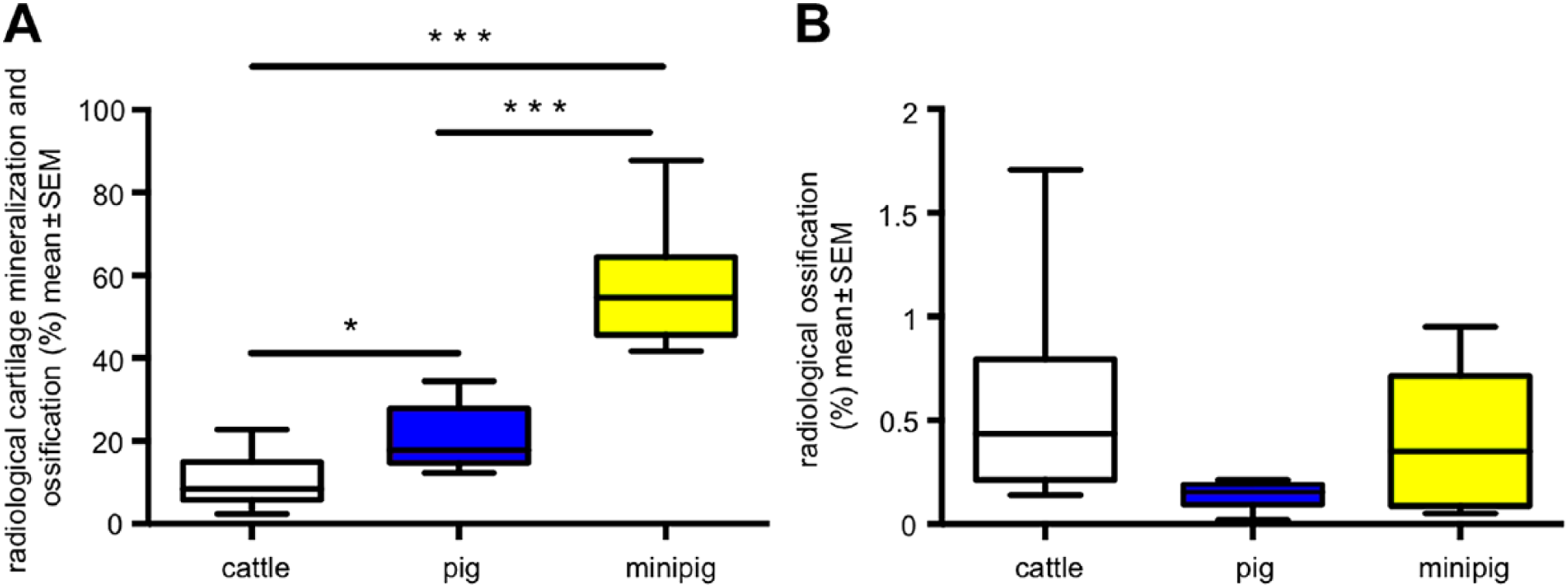
X-ray analysis of thyroid cartilage plates from minipigs (
Analysis by Light Microscopy and TEM
Chondrocytes of growth plate and thyroid cartilage
In growth plates of miniature pigs, chondrocytes were arranged by columns and showed a different morphology (Fig. 4A). Small resting chondrocytes turned into oval proliferating chondrocytes (Fig. 4B). In the upper hypertrophic zone, chondrocytes tended to mature and develop a rounded shape (Fig. 4C). In the lower hypertrophic zone, chondrocytes mineralized the longitudinal septa (Fig. 4D). On this framework, osteoblasts deposited bone to build primary spongiosa. Therefore, primary spongiosa contained a core of mineralized cartilage, which was covered by bone.
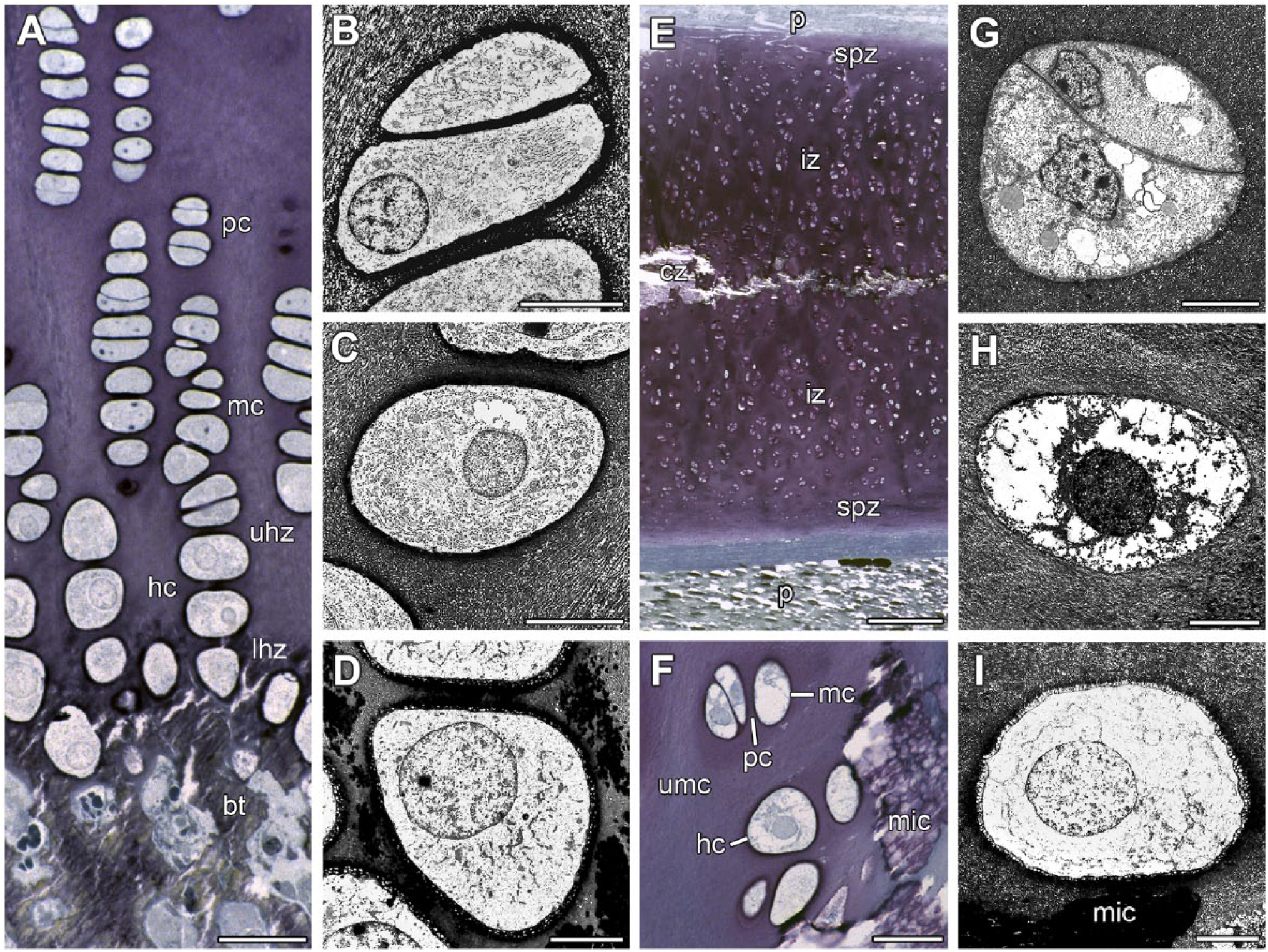
Types of chondrocytes in proximal humerus growth plate of a 32-month-old male minipig (A–D) and in thyroid cartilages of a 9-month-old minipig (E) and a 16-month-old (F–I) minipig. (A) Proximal humerus growth plate shows a columnar arrangement of chondrocytes. The following types of chondrocytes are distinguishable: pc, mc, and hc. In the metaphysis, bt can be seen. (B) pc possess an oval shape. Bar = 7 µm. (C) mc have developed a rounded shape. (D) Cartilage mineralization appears in the longitudinal septa beneath hc. (E) Cartilage segment 4B—Outer limitations of thyroid cartilage is represented by both P. The following zones of different chondrocytes can be distinguished: spz, iz, and cz. Extracellular matrix of the cz is partly mineralized. (F) Cartilage segment 6—An interface between umc and mic shows hc in the vicinity of mic. At some distance from mic, pc and mc are observed. (G) Segment 5B—Two pc lay close together as a pair. Bar = 7 µµ. (H) Segment 6—An mc has a rounded shape. (I) Segment 6—An hc is located near mic. Fixation under addition of RHT (A–I). Toluidine blue (A, E, F). Bar = 7 µm (B–D, G–I), 30 µm (A, F), and 300 µm (E). Abbreviations: pc, proliferating chondrocyte; mc, maturing chondrocyte; hc, hypertrophic chondrocyte; bt, bony trabecles; uhz, upper hypertrophic zone; lhz, lower hypertrophic zone; P, perichondria; spz, subperichondrial cartilage zone; iz, intermediate cartilage zone; cz, central cartilage zone; umc, unmineralized cartilage; mic, mineralized cartilage; RHT, ruthenium III hexamine trichloride.
Chondrocytes of thyroid cartilage from minipigs can also be attributed to zones. However, a column-like appearance of cells was generally missing (Fig. 4E). The subperichondrial zones located at the outer and inner side of thyroid cartilage contained unmineralized cartilage with small proliferating cells, which were positioned in pairs (Fig. 4F and G). Toward the center of the cartilage plate, two intermediate zones with more oval-shaped maturing cells followed (Fig. 4F). Some of these chondrocytes were located in the vicinity of mineralized cartilage (Fig. 4F). The central cartilage zone was characterized by mineralized cartilage (Fig. 4E). Here, hypertrophic chondrocytes were located within or in the vicinity of mineralized cartilage (Fig. 4H and I). These rounded cells were the biggest ones in the thyroid cartilage. Hypertrophic chondrocytes have a large nucleus and contain more cytoplasm than proliferating and maturing chondrocytes (Fig. 4G–I). Compared with the chondrocytes in the other zones, hypertrophic chondrocytes possessed the highest number of cell organelles (Fig. 4H and I). By contrast, vacuoles were found more often in proliferating and maturing chondrocytes (Fig. 4G) than in the hypertrophic ones.
Cartilage mineralization in growth plate and in thyroid cartilage
As expected, extracellular matrix was better preserved in perfusion-fixed minipigs (Fig. 5I) when compared with domestic pigs derived from slaughterhouse (Fig. 5C, D, and I).
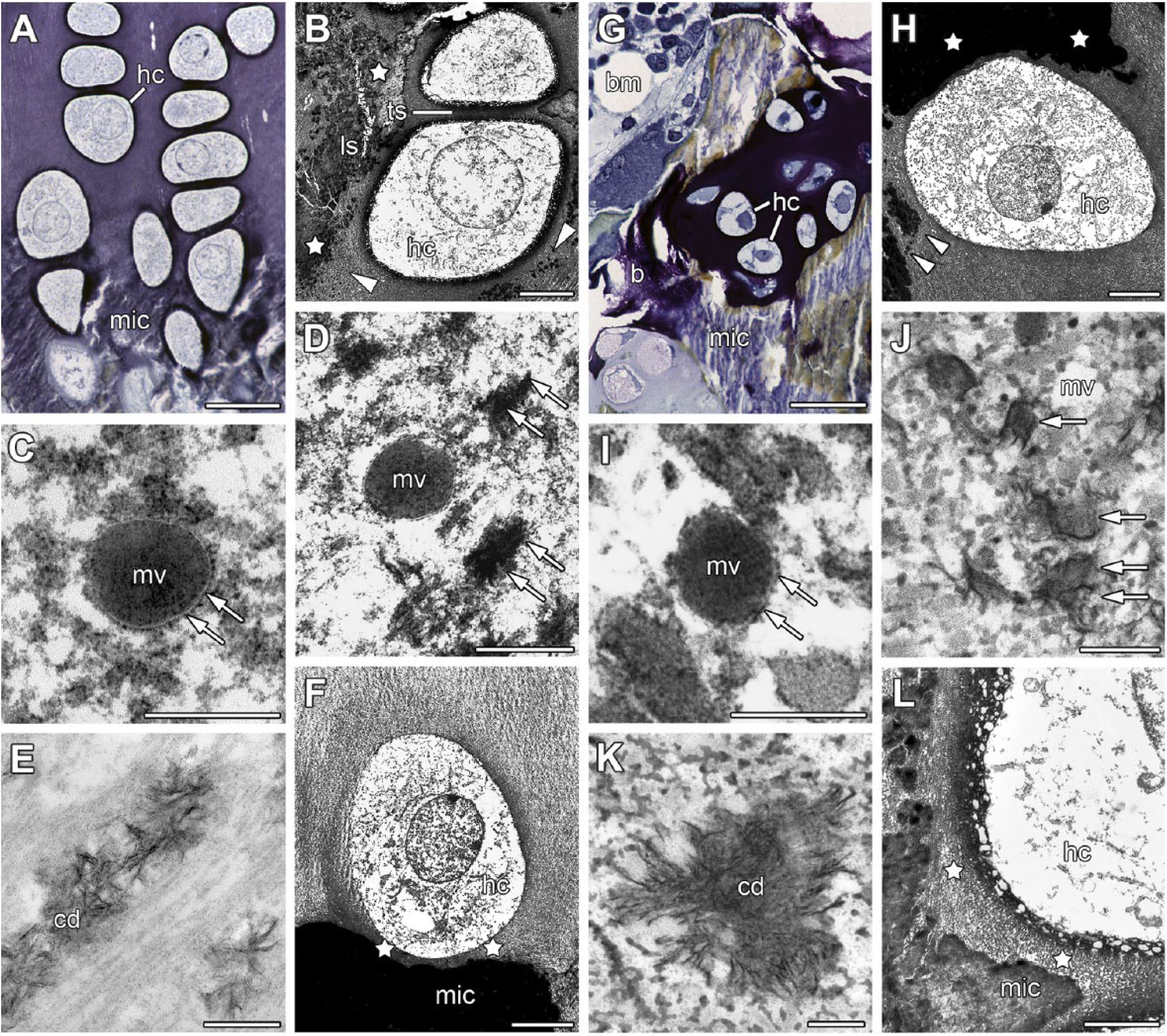
Mineralization of the proximal humerus growth plates of a 32-month-old male minipig (A, B, E, F) or an 8-week-old male domestic pig (C, D) and of thyroid cartilages from a 10-month-old minipig (G, J), a 16-month-old minipig (H, L), a 9-month-old minipig (K), or from an 8-month-old domestic pig (I). (A) In the lower growth plate, mic is observed in ls between hc. (B) Only ls are mineralized by hc while ts remain unmineralized (arrowheads show beginning, asterisks show advanced cartilage mineralization). (C) The longitudinal septum of the growth plate contains an mv. Arrows point to the vesicular membrane. (D) In thyroid cartilage segment 6, small druses (arrows) constructed from apatite crystals near an mv characterize the beginning of cartilage mineralization. (E) In ls of growth plate, calcium cd can be seen. (F) In the lower growth plate, pericellular matrix (stars) around an hc remains unmineralized. (G) In thyroid cartilage segment 6, mic and incipient b are observed in the neighborhood of hc. In addition, bm has developed. (H) In thyroid cartilage segment 6, beginning (arrowheads) and advanced (stars) cartilage mineralization can be seen. (I) In thyroid cartilage segment 6, an mv embedded in collagen fibrils is shown. Arrows point to vesicular membrane. (J) In the longitudinal septum of the growth plate, small druses (arrows) are located near to an mv. (K) Also in thyroid cartilage segment 9, calcium cd characterize the beginning of cartilage mineralization. (L) Also in thyroid cartilage segment 6, pericellular matrix (stars) around an hc is unmineralized. Fixation under addition of RHT (A, B, F, H, L), RR (G, K), or tannin (C, D, E, I, J). Toluidine blue (A, G). Bar = 0.2 µm (C, I, J, K), 0.4 µm (D, E), 2 µm (L), 7 µm (B, F, H), and 40 µm (A, G). Abbreviations: mic, mineralized cartilage; hc, hypertrophic chondrocytes; ls, longitudinal septa; ts, transverse septa; mv, matrix vesicle; cd, containing druses; b, bone deposition; bm, bone marrow; RHT, ruthenium III hexamine trichloride; RR, ruthenium red.
Hypertrophic chondrocytes appeared in the vicinity of mineralizing cartilage areas in both growth plate (Fig. 5A and B) and thyroid cartilage (Fig. 5G and H). In both organs, matrix vesicles appeared in mineralizing cartilage areas close to hypertrophic chondrocytes. Matrix vesicles were extracellular 100-nm-diameter membrane-invested particles, selectively located within still unmineralized cartilage. The intravesicular matrix was homogeneous and osmiophilic (Fig. 5C and I). In growth plate, matrix vesicles could be observed in mineralizing longitudinal septa. They were absent in transverse septa, which did not mineralize. In thyroid cartilage, matrix vesicles were seen near to the mineralizing central cartilage zone. However, some matrix vesicles were also observed in the transition zone between the intermediate and central cartilage zones.
Both in growth plate (Fig. 5D) and thyroid cartilage (Fig. 5J), the first crystalline apatite mineral was seen as a needle-like dense precipitate with the matrix vesicle. With accumulation, growth, and following rupture of the vesicular membrane, crystals were exposed to the extravesicular environment. Afterward, little calcified nodules which have grown by conglomeration of some crystals were observed. The first calcified nodules were not much larger than matrix vesicles (Fig. 5D and J). The calcified nodules grew into druses by the apposition of additional apatite crystals (Fig. 5E and K). Islands of mineralized cartilage developed by fusion (Fig. 5B and H). The areas provided for cartilage mineralization were the longitudinal septa in growth plate and the central cartilage zone in thyroid cartilage. In both organs, a small pericellular rim surrounding the hypertrophic chondrocytes remained unmineralized (Fig. 5F and L). That pericellular unmineralized matrix was about 2 µm in breadth.
Different patterns of thyroid cartilage mineralization
In light microscopy, preservation of nucleus and cell organelles was better in perfusion-fixed minipigs (Fig. 6A) when compared with domestic pigs obtained from slaughterhouse (Fig. 6D). The difference was also observed on the ultrastructural level by comparing Fig. 6E (specimen of a domestic pig) with Fig. 6F (specimen of a minipig).
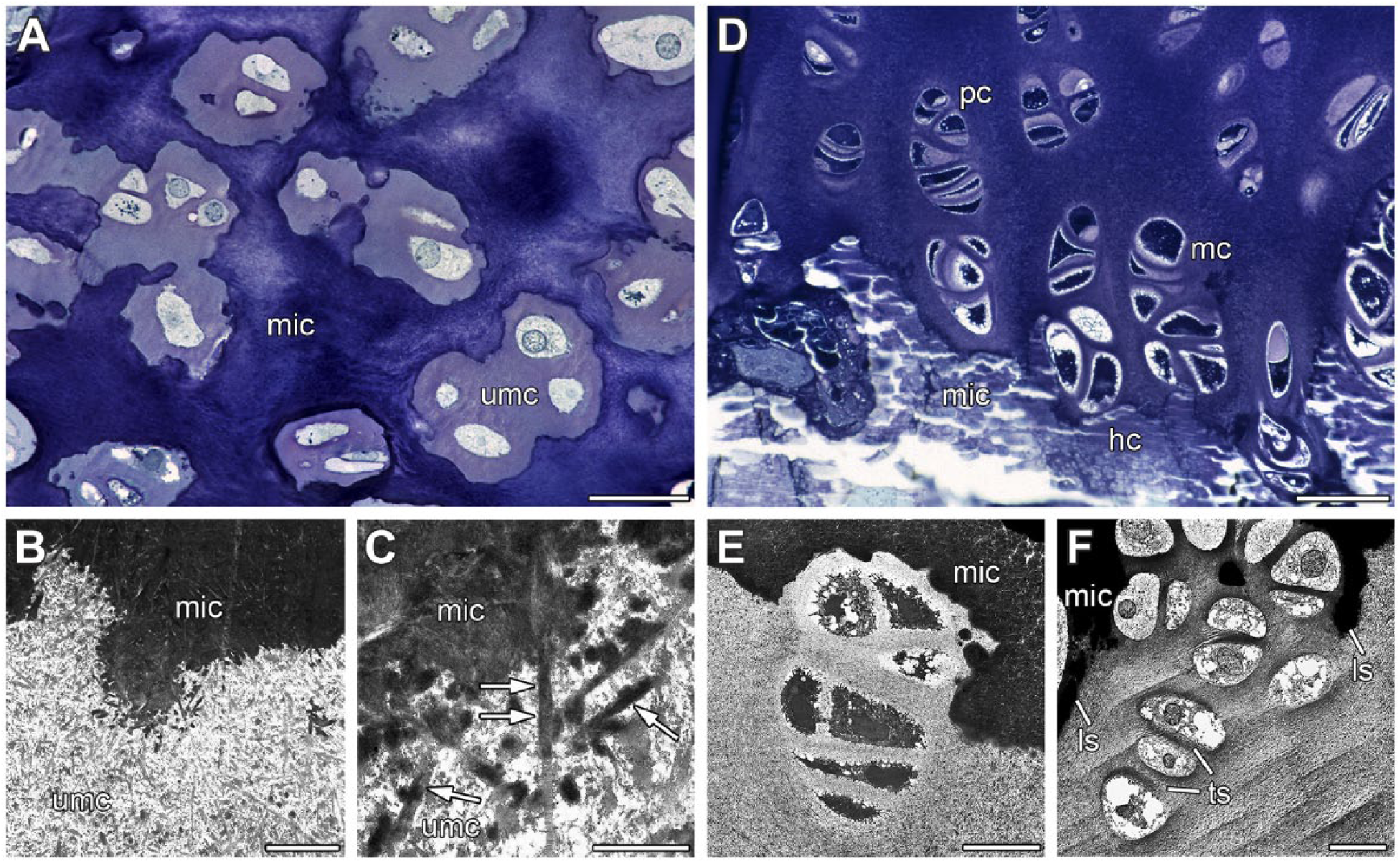
Different forms of thyroid cartilage mineralization at a 10-month-old male minipig (A) and a 32-month-old male minipig (B, C, F) or an 8-month-old domestic pig (D, E). (A) In thyroid cartilage segment 6, islands of umc are located within mic. (B) In thyroid cartilage segment 6, an interface between umc and mic cartilage is visible. Collagen fibrils are clearly visible in umc. (C) In thyroid cartilage segment 6, apatite crystals (arrows) developed along collagen fibrils of umc. (D): In thyroid cartilage segment 6, chondrocytes have formed columns, reminiscent of the respective structures in growth plate. pc can be distinguished from mc and hc. (E) Thyroid cartilage segment 6 of a domestic pig shows a bay of mic with a column-like formation of chondrocytes. (F) Thyroid cartilage segment 6 of a minipig also shows a bay of mic with a column-like formation of chondrocytes. As in growth plates, some ls showed cartilage mineralization while ts remain unmineralized. Fixation under addition of RR (A) or tannin (B–E) or RHT (F). Toluidine blue (A). Bar = 1 µm (C), 3 µm (B), 15 µm (E, F), 30 µm (A), and 50 µm (D). Abbreviations: umc, unmineralized cartilage; mic, mineralized cartilage; pc, proliferating chondrocyte; mc, maturing chondrocyte; hc, hypertrophic chondrocyte; ls, longitudinal septa; ts, transverse septa; RR, ruthenium red; RHT, ruthenium III hexamine trichloride.
Thyroid cartilage showed another pattern of cartilage mineralization besides mediation by matrix vesicles. This pattern occurred in already mineralized cartilage areas which were sometimes interrupted by islands of unmineralized cartilage (Fig. 6A). The islands contained proliferating, maturing, and hypertrophic chondrocytes (Fig. 6A). Using light microscopy, a clear border between mineralized and unmineralized cartilage was observed (Fig. 6A). However, using TEM, unfinished and progressing cartilage mineralization was seen (Fig. 6B).
In this second pattern of thyroid cartilage mineralization, matrix vesicles and typical calcification nodules were lacking (Fig. 6C). Crystal formation and crystal growth showed a close relation to collagen fibrils. Collagen fibrils were used as guide rails for the expansion of mineralization (Fig. 6C). Both mineralization patterns occurred simultaneously in thyroid cartilage. However, cartilage mineralization along collagen fibrils was seen less frequently. Furthermore, this second mineralization pattern appeared in areas where mineralization mediated by matrix vesicles was completed.
In some areas of thyroid cartilage, chondrocytes were not arranged according to the three zones, but rather formed columns with proliferating, maturing, and hypertrophic chondrocytes known from growth plates (Fig. 6D). As in growth plates, only longitudinal septa showed cartilage mineralization while transverse septa remained unmineralized (Fig. 6E and F). However, in contrast to growth plates, not every longitudinal septum revealed mineralization (Fig. 6F).
Discussion
A description of cartilage mineralization and ossification of animal laryngeal cartilages is lacking at the ultrastructural level. Here, we refer to mineralization of minipig and domestic pig thyroid cartilage, because both species were readily comparable with the human tissue as to form and size. We used X-ray, light microscopic, and especially transmission electron microscopic methods. The mineralization process was compared with the well-known processes in growth plates. However, despite we spent effort on best preservation of cells and matrix for TEM analyses, our study is limited by the number of specimens investigated.
As assumed, better preservation of cells, cell organelles, and extracellular matrix was reached in the perfusion-fixed minipigs treated additionally with special immersion and postfixation solutions (see Table 1) when compared with domestic pigs obtained from slaughterhouse. This can be recognized by comparing Figs. 5J and 6A (specimen from minipigs) with Figs. 5C, D, I, and 6D (specimen from domestic pigs).
Scanning of thyroid cartilages showed that cartilage mineralization is the main event in aging of laryngeal cartilages from minipigs, domestic pigs, and cattle. By contrast, ossification is the dominant event in human thyroid cartilage, while cartilage mineralization is restricted to small areas at the bone–cartilage interface. 9
Both thyroid cartilage and growth plates contained three types of cells: proliferating, maturing, and hypertrophic chondrocytes. In contrast to growth plate, proliferating chondrocytes were located in the subperichondrial cartilage zone of thyroid cartilage. As described for growth plates, proliferating chondrocytes have a flat form.15–17 Maturing chondrocytes known from the upper hypertrophic zone of growth plates are located in the intermediate cartilage zone of thyroid cartilage. As shown for growth plates, maturing chondrocytes become larger and cell organelles are increased in comparison with proliferating chondrocytes.15,18,19 Hypertrophic chondrocytes occur only in the central cartilage zone of thyroid cartilage. Hypertrophic chondrocytes were the largest cells, both in thyroid cartilage and growth plate.17,18 Compared with the proliferating chondrocytes, the cell surface of hypertrophic chondrocytes has increased 4-fold while cellular volume has increased as much as 10-fold.15,18 Taken together, thyroid cartilage and growth plate contained the same set of specialized chondrocytes required for mineralization of extracellular cartilage matrix.
Matrix vesicles are observed in thyroid cartilage and growth plate in the vicinity of hypertrophic chondrocytes and mineralizing cartilage. In thyroid cartilage, most matrix vesicles are found in the central cartilage zone. By contrast, in growth plate, matrix vesicles are located in the longitudinal septa between hypertrophic chondrocytes exclusively, whereas transverse septa do not contain any.20–24 Matrix vesicles are osmiophilic and their innermost matrix is more electron dense compared with the perivesicular matrix.25,26 The vesicles must not been mistaken for cross-sections of cytoplasmic extensions belonging to chondrocytes.22,25 The enzyme alkaline phosphatase responsible for the intake of calcium and phosphate into the vesicles is a main marker of matrix vesicles.21,26–28 Calcium was identified in matrix vesicles by autoradiography. 29 In both thyroid cartilage and growth plate, matrix vesicles are pinched off from specific regions of the outer plasma membrane of chondrocytes.22,26,27,29,30 Taken together, matrix vesicles budding off from chondrocytes are observed not only in growth plate but also in thyroid cartilage.
Matrix vesicles are found in mineralizing areas of thyroid cartilage. However, the significance of matrix vesicles in cartilage mineralization is discussed controversially. Several authors agree that the vesicles are responsible for cartilage mineralization.21,27,31–33 Mitchell and Shepard claimed there is no correlation between matrix vesicles and cartilage mineralization. 34 Calcium and phosphate are enriched within the vesicles. After passing the solubility quotient, initial crystals grow in matrix vesicles. Anderson and Bonucci have proved the nature of apatite crystals by electron diffraction.21,22 Arsenault and Hunziker obtained similar results with dark-field electron microscopy. 31 In vesicles of thyroid cartilage and growth plate, the first crystals are connected to the innermost layer of the vesicular membrane.20,25,30,31,35 Later on the vesicular membrane is disrupted and apatite crystals are exposed to the extracellular cartilage matrix, serving as sprouts for the formation of further crystals.18,22,32,36,37 In this way, globe-like druses are formed. The globe-like structure of druses in minipig thyroid cartilage has also been shown by scanning electron microscopy. 38 Coherent mineralized cartilage areas developed with further growth of these druses.20,22,39–41 To summarize, in thyroid cartilage and growth plate, first apatite crystals originated in matrix vesicles. Further growth of crystals into druses occurs after rupture and disappearance of these vesicles.
Initial cartilage mineralization is mediated by matrix vesicles in both thyroid cartilage and growth plate. However, a second cartilage mineralization process with late onset is present in thyroid cartilage. Here, collagen fibrils are used as leading tracks for the mineralization process. Apatite crystals take their origin at the collagen fibrils of the extracellular matrix of minipig thyroid cartilage. Bonucci and Anderson also reported similar results in their analyses of cartilage mineralization.25,39 We assume that cartilage mineralization in thyroid cartilage is first mediated by matrix vesicles, then second by collagen fibrils.
We conclude that, in contrast to humans where thyroid cartilage ossifies nearly completely by advanced age, cartilage mineralization is the main event during aging of minipig and domestic pig or cattle thyroid cartilage, while only discrete ossified areas are observed. The same set of chondrocytes as described for growth plates is also responsible for pig thyroid cartilage mineralization. Matrix vesicles and the formation of apatite crystals mediate cartilage mineralization in both organs. Only thyroid cartilage reveals a second pattern of cartilage mineralization in pigs—mediated by collagen fibrils.
Compared with the human larynx, thyroid cartilage of pigs shows a very early cartilage mineralization near to the insertion zone of the vocalis muscle (segments 4B and 6, see Fig. 1). In human, this area is mineralizing and ossifying relatively late in age and also only in male. Possibly, this exceptional feature contributes to the high variability of the human voice which especially appears by listening to the voice of speakers and chamber singers. However, progressing gender-related ossification of laryngeal cartilages, particularly in the area of the anterior commissure, could be one factor among others (i.e., loss and changes of elastic fibers, changes of collagen fibrils, loss of proteoglycans, reduced secretion potency of laryngeal glands, epithelial metaplasia with increase in squamous epithelium, changes in the laryngeal musculature) leading to stiffening of the complete laryngeal skeleton favoring voice changes with advanced age. 42 This might by a reason why chamber singers sometimes are affected by a reduction of voice quality about the age of 50 and why the human voice changes continuously throughout life.
Footnotes
Acknowledgements
We would like to thank Mrs. Antje Haupt, Mrs. Elisabeth Schöngarth, and Mr. Horst Mrohs (Anatomical Institute of Christian-Albrechts-University of Kiel (CAU), Kiel, Germany) for their excellent technical assistance. Furthermore, we thank Mrs. Heidi Waluk and Mrs. Heide Siebke (Anatomical Institute of CAU, Kiel, Germany) for their help in the photographic work. In addition, we thank Mr. Marco Gösswein (Institute of Anatomy 2, Friedrich-Alexander-University [FAU] Erlangen-Nürnberg, Germany) for his help with the layout and Mr. Jörg Pekarsky (Institute of Anatomy 2, FAU Erlangen-Nürnberg, Germany) for graphical assistance. We cordially thank Prof. Dr. Ernst Hunziker (M. E. Müller Institute for Biomechanics, University of Bern, Switzerland) for his support and help concerning fixation techniques in transmission electron microscopy (TEM).
Competing Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
HC, BT, and FP contributed to the conception and design of the study. HC, MS, BF, and RH performed the experiments. SH performed the statistical analysis. HC, MS, BF, RH, BT, and FP contributed to the analysis and interpretation of data. All approved the submitted manuscript.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was supported in part by the German Research Foundation (Deutsche Forschungsgemeinschaft [DFG], Program Grant PA 738/9-2).
