Abstract
The appropriate projection of axons within the nervous system is a crucial component of the establishment of neural circuitry. Draxin is a repulsive axon guidance protein. Draxin has important functions in the guidance of three commissures in the central nervous system and in the migration of neural crest cells and dI3 interneurons in the chick spinal cord. Here, we report that the distribution of the draxin protein and the location of 23C10-positive areas have a strong temporal and spatial correlation. The overexpression of draxin, especially transmembrane draxin, caused 23C10-positive axon bundles to misproject in the dorsal hindbrain. In addition, the overexpression of transmembrane draxin caused abnormal formation of the ganglion crest of the IX and X cranial nerves, misprojection of some anti-human natural killer-1 (HNK-1)-stained structures in the dorsal roof of the hindbrain, and a simultaneous reduction in the efferent nerves of some motoneuron axons inside the hindbrain. Our data reveal that draxin might be involved in the fascicular projection of cranial nerves in the hindbrain.
Introduction
In the early embryo, the anterior end of the neural tube enlarges and differentiates into the major brain subdivisions, including three expanding vesicles (forebrain, midbrain, and hindbrain) that are separated by two constrictions. The hindbrain is the most posterior vesicle of the embryonic brain. During the early stages of neural development, the hindbrain is transiently organized into segments along the anterior–posterior axis that lead to the formation of seven to eight metameres called rhombomeres.1,2 This transient segmental organization is necessary for the location of the cranial nerve exit points and neural crest cell migration from the dorsal hindbrain toward the branchial arch. Hindbrain motoneurons are born in the ventricular zone close to the floor plate, and they differentiate into specific subsets according to their rhombomere of origin. 3 The projecting efferent nerves from motoneurons combining with the projecting afferent nerves from the cranial sensory ganglia form the cranial nerves which connected with the brain at different position. The ventral basement membrane in the hindbrain neuroepithelium is both a substrate for migration and a boundary that constrains facial branchiomotor neurons (FBMNs) to the appropriate migratory path. 4
In both vertebrates and invertebrates, some of the cues governing axonal growth are also involved in neuronal migration.5 –8 Draxin is a repulsive axon guidance protein named for the “dorsal repulsive axon guidance protein.” 9 Draxin is required for the development of the spinal cord and the forebrain commissures, 9 and for the regulation of chick neural crest cell and dI3 interneurons migration.10,11 Draxin could inhibit several types of neuronal axon ourgrowth in vitro, such as the spinal cord commissural, the tectal and neocortical, and the lateral olfactory bulb neuronal axon outgrowth.9,12,13
To understand the molecular basis of draxin function during neural axon projections, we studied the effects of draxin during chick embryonic hindbrain formation. The 23C10 monoclonal antibody produced by Obata and Fujita 14 was found to recognize a protein widely expressed by many neural cells and axons of the nervous system. We found that this antibody was an excellent marker for neural axons in the chick hindbrain. Therefore, in this study, we used the 23C10 monoclonal antibody as a marker to examine the function of draxin during axonal projections in the chick hindbrain. The draxin protein was expressed in 23C10-positive areas in the peripheral area of the hindbrain basal zone. The overexpression of draxin, especially transmembrane draxin, caused misprojected 23C10-positive axon bundles in the dorsal hindbrain. In addition, the overexpression of transmembrane draxin caused abnormal formation of the ganglion crest of the IX and X cranial nerves, misprojection of some anti-human natural killer-1 (HNK-1)-stained structures in the dorsal roof of the hindbrain, and a reduction in the number of efferent nerves of some motoneuron axons inside the hindbrain. Our data revealed that draxin may have important functions during axonal fascicular projections, especially the axons which are from cranial nerves X inside the chick hindbrain.
Materials and Methods
Chick Embryos
All of the experiments were conducted in accordance with the guidelines for the care and use of animals approved by the Animal Care and Use Committee (Hebei Medical University, Hebei, China). Fertilized White Leghorn chick eggs were obtained from local commercial sources. All eggs were incubated at 38C until the embryos reached the desired Hamburger–Hamilton (HH) 15 stage. The embryos were removed from the eggs, stripped of their membranes, and fixed in 4% paraformaldehyde at 4C for 2 hr.
Immunohistochemical Staining and Statistical Analyses
For immunohistochemical staining, the sections were treated with blocking solution for 2 hr before being incubated in the primary antibody overnight at 4C. The following primary antibodies were used: anti-23C10 monoclonal antibody; anti-Neurofilament-L monoclonal antibody (cell signaling technology [CST]); anti-Neuron-specific β-III Tubulin antibody (Tuj-1); anti-HNK-1, anti-SC1, and anti-chick-Tag-1 monoclonal antibodies (from Prof. Tanaka); and rabbit anti-chick draxin polyclonal antibody. The rabbit anti-chick draxin polyclonal antibody and anti-23C10 monoclonal antibody were produced as previously described.9,14 We got anti-chick draxin polyclonal antibody and anti-23C10 monoclonal antibody from Prof. Tanaka. The selection of positive and negative controls for antibody validation was performed following the published guidelines for antibody validation. 16 Previously published article suggested that anti-23C10 monoclonal antibody could detect different neural structures, especially Purkinje cells, in embryonic day 11 chick cerebellum, and the staining of Purkinje cells was disappeared after embryonic day 18. 14 So we used Purkinje cells of embryonic day 11 chick cerebellum as positive control. Purkinje cells of embryonic day 19 chick cerebellum were used as negative control. After being washed extensively, the sections were incubated with Cy3-conjugated anti-mouse IgG or IgM (1:500 diluted in blocking solution).
For whole-mount immunostaining, the embryos were treated with methanol and 2% hydrogen peroxide (H2O2) before blocking. Anti-23C10 monoclonal antibody was incubated on the whole-mounts for 48 hr. After being washed extensively, the embryos were incubated with horseradish peroxidase (HRP)-conjugated anti-mouse IgG (1:300 diluted in blocking solution) for 48 hr. After being washed extensively, 3,3′-diaminobenzidine tetrahydrochloride (DAB) color development was performed for 20 min at room temperature.
All the measurements were performed by Image-Pro Plus software, and standard Student’s t-test was used for the statistical analyses.
In Ovo Electroporation
The electroporation of the expression vectors into the chick hindbrain was performed as previously described. 17 Myc- and His-tagged draxin cDNA was cloned into the pMES-IRES-EGFP vector. The following cDNAs were used in electroporation experiments: pMES-IRES-EGFP, pMES-draxin-IRES-EGFP, and pMES-draxin-Tm-IRES-EGFP. The membrane-bound form of draxin was constructed by fusing the transmembrane domain of SC1 18 at the C-terminal end of draxin. Plasmid DNA was concentrated to 4 mg/ml and supplemented with 0.1% Fast Green.
Results
Draxin-Positive and 23C10-Positive Areas Are Detected in the Peripheral Area of the Hindbrain Basal Zone
Double immunohistochemistry staining was performed using an anti-chick draxin polyclonal antibody and the 23C10 monoclonal antibody on transverse sections of the chick embryonic hindbrain region at different stages. Hoechst counterstaining was used to show the hindbrain structure (Fig. 1). Using this method, the relationship between the distribution of draxin protein and the location of 23C10-positive axons was detected. At HH stages 18–19, the draxin protein was detected at the regions which are near the rhombic lip, and weakly dispersed punctate distribution of the draxin protein was detected in the peripheral area and the dorsal region of the hindbrain basal zone (Fig. 1A). At the same time, 23C10-positive areas, including projecting axons, were detected in the ventral peripheral area of the hindbrain basal zone (Fig. 1B). A proportion of the axons projected directly to the dorsal surface of the hindbrain basal zone. Both draxin-positive and 23C10-positive areas were detected inside the hindbrain, especially in the peripheral area of the hindbrain basal zone (Fig. 1C). At HH stages 21–22, the expression of draxin increased in the peripheral area of the hindbrain basal zone. Draxin protein was also detected in the regions which are near the rhombic lip and dorsal region of the hindbrain basal zone (Fig. 1D). At the same time, 23C10-positive axons and axonal bundles were detected in the ventral peripheral area of the hindbrain basal zone (Fig. 1E). The cranial nerves were formed and exited from the hindbrain. Strong labeling of both draxin-positive and 23C10-positive areas was detected in the peripheral area of the hindbrain basal zone (Fig. 1F). Ventral to the exiting points of cranial nerves in hindbrain, the expression of draxin was continued flaky distribution. In the exiting points of the cranial nerves and dorsal to the exiting points of cranial nerves in hindbrain, the expression of draxin was decreased and became discontinuous (Fig. 1F). The change in the expression pattern was measured by calculating the ratio of the draxin protein distribution area with the total area in the same section. The measurement method was showed by the dotted lines in Fig. 1D. White dotted lines mark the whole area in right half hindbrain. Pink dotted lines mark the draxin distribution area in the same half hindbrain. The average ratio of draxin protein distribution area with the total area in the left and right side in one section was used as the ratio for this section. Three sections from one embryo were counted, and the mean of these three sections was used for statistical analysis. At least 15 embryos for each HH stage were analyzed. The results are presented as the mean ± SD. At HH stages 18–19, the ratio of draxin protein distribution area to total area was 23.25% ± 2.85%. At HH stages 21–22, the ratio of draxin protein distribution area to total area was 41.83% ± 5.47%. The expression was increased at HH stages 21–22 compared with stages 18–19 (p < 0.05).
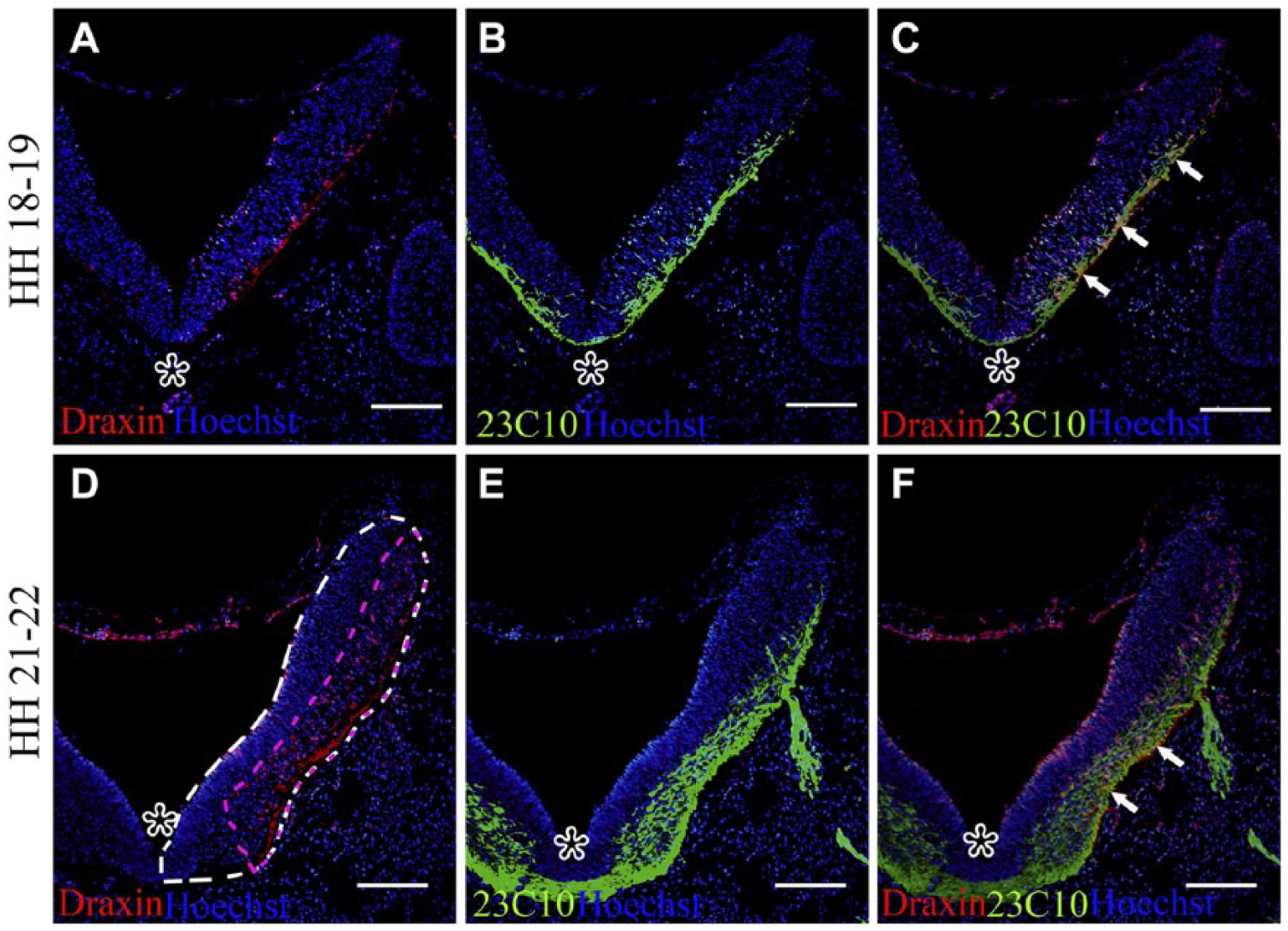
Distribution pattern of draxin and projection pattern of 23C10-positive axons in the embryonic chick hindbrain at different stages. The dorsal part is at the top. At level r7 of the chick hindbrain. Asterisks point to the ventral midline. Hoechst staining shows the structure of the sections. (A) At Hamburger–Hamilton (HH) stages 18–19, draxin protein was detected at the regions which are near the rhombic lip, and a weakly dispersed distribution of the draxin protein was detected in the peripheral area of the hindbrain basal zone. (B) Using the same section as A, 23C10-positive areas were detected in the ventral peripheral area of the hindbrain basal zone. (C) Overlay of A and B. Arrows point to the double-positive areas (yellow). (D) At HH stages 21–22, draxin protein expression was increased in the peripheral area of the hindbrain basal zone. White dotted lines mark the whole area in right half hindbrain. Pink dotted lines mark the draxin distribution area in the same half hindbrain. (E) Using the same section as D, 23C10-positive areas were detected in the ventral peripheral area of the hindbrain basal zone. (F) Overlay of D and E. Arrows point to the double-positive areas (yellow). Scale bar = 200 µm.
Overexpression of Draxin Caused 23C10-Positive Axons to Misproject to the Dorsal Roof Area of the Hindbrain
To determine the in vivo function of draxin during the development of 23C10-positive axonal projections, we overexpressed either the secreted or transmembrane forms of the draxin cDNA in the chick hindbrain by in ovo electroporation. The empty vector was electroporated as the control. Secreted draxin, transmembrane draxin, or the control vector was first overexpressed in the chick hindbrain at HH stages 13–14, and 23C10 immunostaining was performed at HH stages 25–26 by whole-mount immunohistochemistry. The dorsal view of the hindbrain region is shown with the head at the upper right side of each panel. The electroporated side was the right side hindbrain of each embryo (Fig. 2). There were no apparent 23C10-positive axons in the dorsal roof of the hindbrain region (Fig. 2A, B). After the overexpression of secreted draxin, there were a few misprojected 23C10-positive axons in the dorsal roof of the electroporated side of the hindbrain region (arrowheads in Fig. 2C, D). Very few axons were also found in the contralateral side of the hindbrain in some embryos (arrow in Fig. 2C, D). After the overexpression of the transmembrane form of draxin, 23C10-positive axons and thick axonal bundles were misprojected in the dorsal roof of the electroporated side of the hindbrain region (Fig. 2E, F). Arrowheads in Fig. 2E, F indicated some of these misprojected axons. Some axons were also found in the contralateral side of the hindbrain (arrows in Fig. 2E, F). The number of obvious abnormal axons in dorsal roof area of the hindbrain was counted. The results are presented as the mean ± SD. At least 15 embryos were used for analyses. Quantification indicated that the mean abnormal axon number of the control group was 0.37 ± 0.05, the mean abnormal axon number of the secreted draxin overexpression group was 8.2 ± 0.17, and the mean abnormal axon number of the transmembrane draxin overexpression group was 27.3 ± 4.9. The differences among the three groups were statistically significant (p < 0.05; Fig. 2G). This result indicates that the transmembrane form of draxin has a stronger function than secreted draxin. Other markers, such as Tuj-1, could also detect the misprojected axons in the dorsal roof of hindbrain after transmembrane draxin overexpression by whole-mount immunostaining (Fig. 2H). The distribution pattern of misprojected Tuj-1 positive axons was similar with 23C10-positive axons in the dorsal roof of hindbrain.
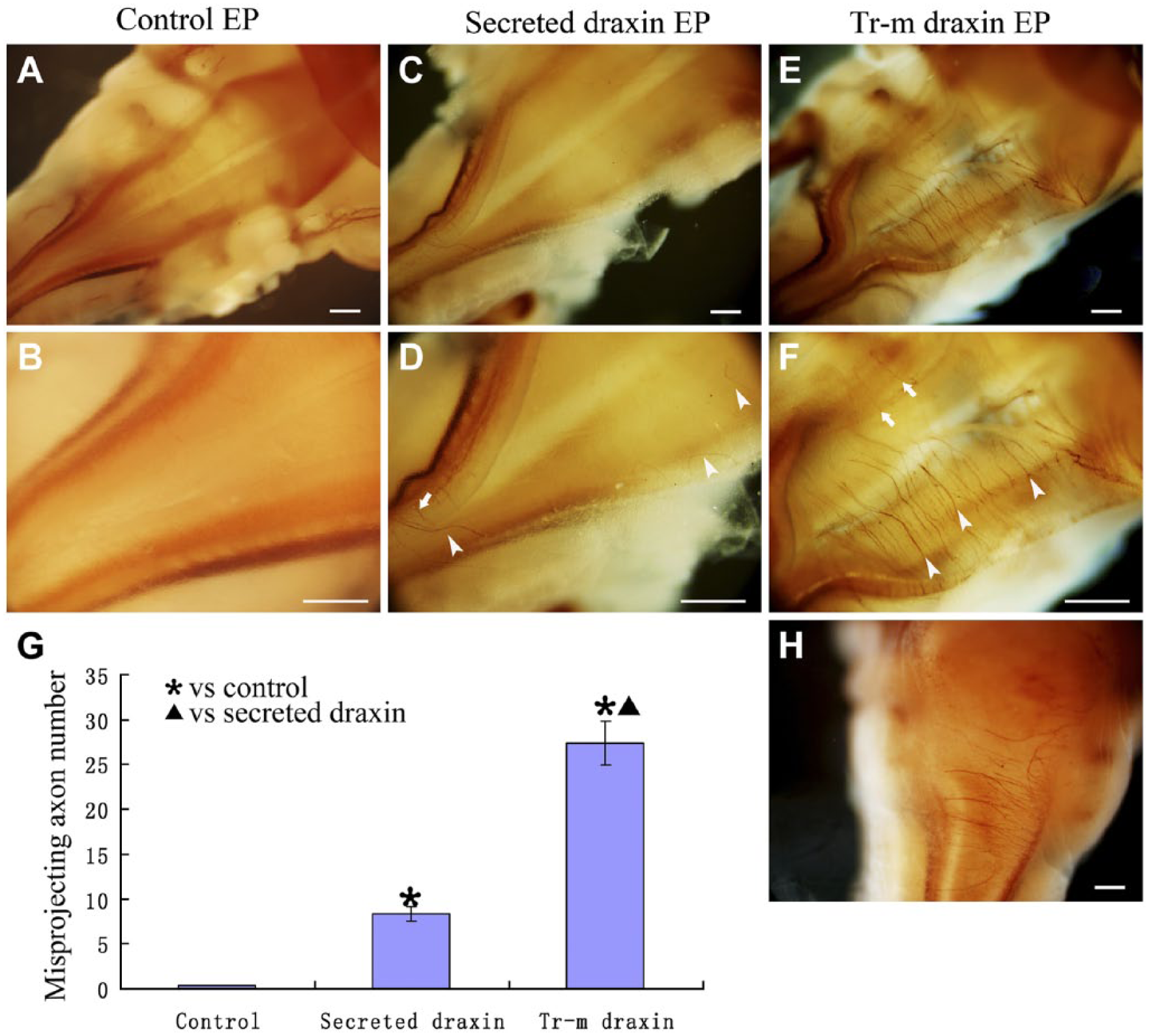
Draxin overexpression caused misprojection of 23C10-positive axons in the whole-mount hindbrain. Dorsal view of the whole-mount hindbrain (the head is on the upper right) and right side was electroporated. (A) Whole-mount 23C10 immunostaining after control vector overexpression. (B) Higher magnification of the same embryo in A. (C) Whole-mount 23C10 immunostaining after secreted draxin overexpression. (D) Higher magnification of the same embryo in C. (E) Whole-mount 23C10 immunostaining after transmembrane draxin overexpression. (F) Higher magnification of the same embryo in E. (G) Column diagram shows the statistical difference of the misprojecting axons in different groups. Arrowheads point to the misprojected axons in the electroporated side. Arrows point to the misprojected axons in the contralateral side. (H) Whole-mount Tuj-1 immunostaining after transmembrane draxin overexpression. Abbreviations: EP, electroporation; Tr-m, transmembrane. * indicate transmembrane or secreted draxin-overexpressed group compares with control group and ▲ indicate transmembrane draxin-overexpressed group compares with secreted draxin-overexpressed group (p < 0.05). Scale bar = 300 µm.
To further examine these misprojected axons, 23C10 immunostaining using transverse sections was performed at the same stages with whole-mount staining. The electroporation effectively induced the ectopic expression of draxin as shown by co-induced enhanced green fluorescent protein (EGFP) labeling, 9 such that the EGFP-positive side was the overexpressed side. After the overexpression of the control vector, there were no apparent 23C10-positive axons in the dorsal roof area of the hindbrain region (Fig. 3A–C). After the overexpression of secreted draxin, there were limited misprojected 23C10-positive axons in the dorsal part of the electroporated side of the hindbrain region (Fig. 3D–F). After the overexpression of the transmembrane form of draxin, 23C10-positive axons and thick axonal bundles were misprojected in the dorsal roof of the electroporated side of the hindbrain region (Fig. 3G–I). Immunostaining of the sections reconfirmed that the transmembrane form of draxin had a stronger function than secreted draxin.
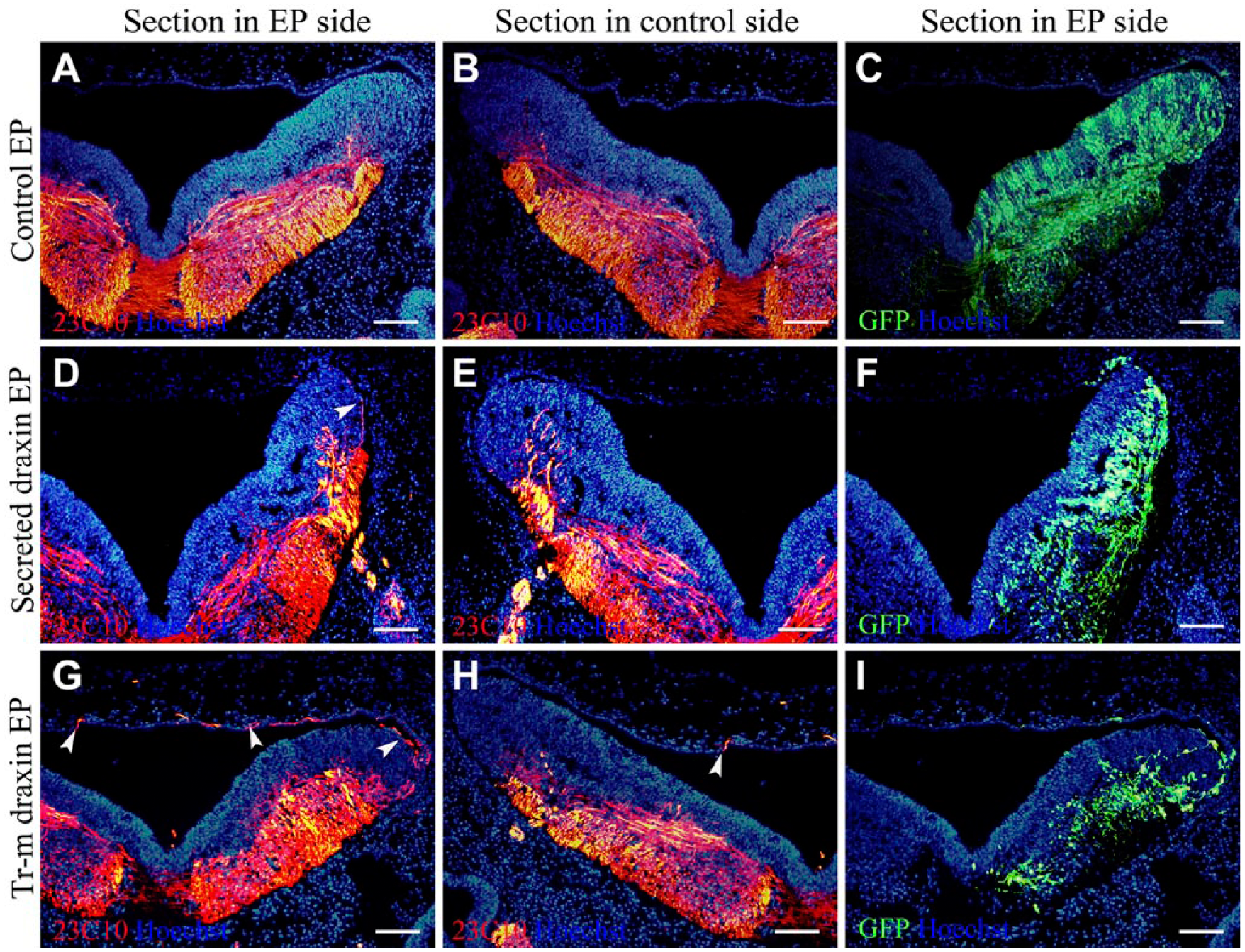
Draxin overexpression caused misprojection of 23C10-positive axons in the hindbrain sections. The dorsal part is at the top in transverse section, and the enhanced green fluorescent protein (EGFP)-positive side was overexpressed. The sections were sampled at level r6-r7 of the chick hindbrain. Hoechst staining shows the structure of the sections. (A–C) The same section after control vector overexpression was used. (A) 23C10 immunostaining of the electroporated side. (B) 23C10 immunostaining of the contralateral side. (C) EGFP expression. (D–F) The same section after secreted draxin overexpression was used. (D) 23C10 immunostaining of the electroporated side. (E) 23C10 immunostaining of the contralateral side. (F) EGFP expression. (G–I) The same section after transmembrane draxin overexpression was used. (G) 23C10 immunostaining of the electroporated side. (H) 23C10 immunostaining of the contralateral side. (I) EGFP expression. Arrowheads point to the misprojected axons in the electroporated side. Abbreviations: EP, electroporation; GFP, green fluorescent protein; Tr-m, transmembrane. Scale bar = 100 µm.
Different Neural Markers Detected the Misprojected Axons After Overexpression of Transmembrane Draxin in the Chick Hindbrain
To further confirm which type of axonal misprojection caused the deficiency of ganglion crest rootlets of cranial nerves IX and X, immunostaining for several neural markers and the chick neural crest marker (HNK-1) was performed after overexpression of transmembrane draxin. In embryos overexpressing transmembrane draxin, neural markers, such as neurofilament (Fig. 4A–C) and Tuj-1 (Fig. 4E–G), detected the misprojected axons in the dorsal roof of the hindbrain. A proportion of the neurofilament-positive axons (Fig. 4A–C) and Tuj-1-positive axons (Fig. 4E–G) were also detected on the contralateral side of the same embryo. Anti-HNK-1-positive axons were also detected in the dorsal roof of the hindbrain region (Fig. 4I–K). There were no neurofilament, Tuj-1, and HNK-1 positive axons in the dorsal roof of the hindbrain region after control vector overexpression (Fig. 4D, H, L). To reveal the relationship between the markers mentioned above with the 23C10-positive axons, we stained adjacent serial sections with each marker and the 23C10 monoclonal antibody. All the 15 embryos showed simultaneous labeling for neurofilament and 23C10 in axons of the dorsal roof. In addition, all the 15 embryos showed simultaneous labeling for Tuj-1 and 23C10 in axons of the dorsal roof. Fourteen of 15 embryos showed simultaneous labeling for HNK-1 and 23C10 in axons of the dorsal roof. The 23C10 signal was collocated with neurofilament, and Tuj-1 antibody declared again that 23C10 is one good marker for axonal projection in the chick hindbrain. The collocation of 23C10 and the HNK-1 demonstrated that the misprojected axons might be related with neural crest-derived sensory nerve fibers.
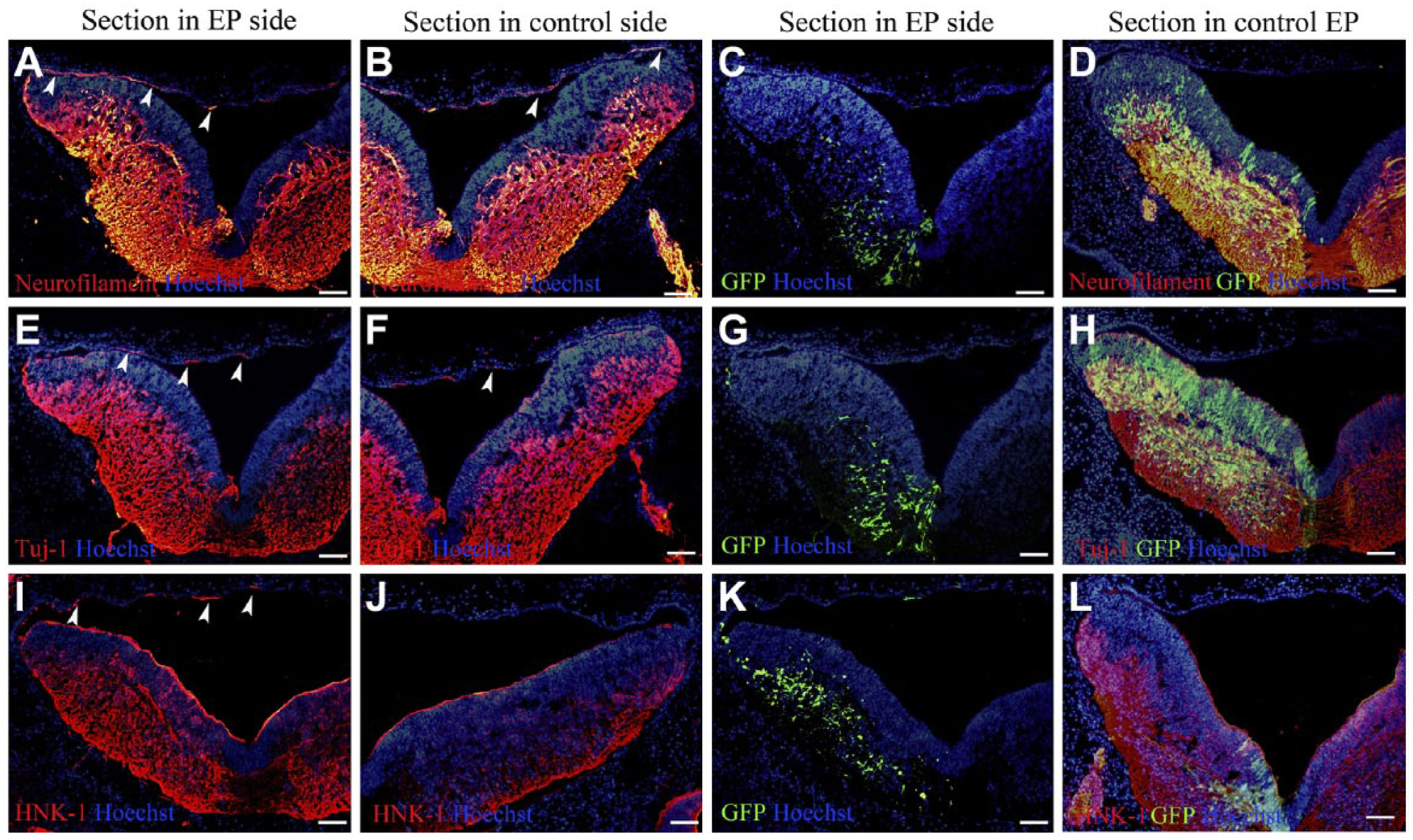
Different neural markers could detect the misprojected axons in the dorsal roof of the hindbrain. Control vector-overexpressed embryos (D, H, and L) and transmembrane draxin-overexpressed embryos (except D, H, and L) were used at level r6-r7 of the chick hindbrain. Hoechst staining shows the structure of the sections. (A) Neurofilament staining of the electroporated side. (B) Neurofilament staining of the contralateral side using the same section as A. (C) Enhanced green fluorescent protein (EGFP) expression using the same section as A showed that the overexpressed side was the left side. (D) Neurofilament staining of the electroporated side. EGFP expression showed that the overexpressed side was the left side. (E) Tuj-1 staining of the electroporated side. (F) Tuj-1 staining of the contralateral side using the same section as E. (G) EGFP expression using the same section as E showed that the overexpressed side was the left side. (H) Tuj-1 staining of the electroporated side. EGFP expression showed that the overexpressed side was the left side. (I) Human natural killer-1 (HNK-1) staining of the electroporated side. (J) HNK-1 staining of the contralateral side using the same section as I. (K) EGFP expression using the same section as I showed that the overexpressed side was the left side. (L) HNK-1 staining of the electroporated side. EGFP expression showed that the overexpressed side was the left side. Arrowheads indicate some of the misprojected axons in both electroporation (EP) side and the contralateral side. Abbreviation: GFP, green fluorescent protein. Scale bar = 100 µm.
Overexpression of the Transmembrane Form of Draxin Caused an Apparent Deficiency of the Ganglion Crest of Cranial Nerves IX and X in the Chick Hindbrain
To further examine all of the axonal structures and to identify the source of the misprojected axons in the chick hindbrain, we performed whole-mount nerve immunostaining using the 23C10 antibody. Draxin was overexpressed at HH stages 13–14 in the chick hindbrain, and the embryos were analyzed at HH stages 25–26. The lateral view of the electroporated side and the contralateral side from each embryo is shown. Several ganglion crest rootlets of cranial nerves IX and X were shown to leave the hindbrain and converged to form thick cranial nerves at this stage. 19 After overexpression of the control vector, the ganglion crest rootlets of cranial nerves IX and X formed normally in both the electroporated side and the control side (Fig. 5A–C). After overexpression of secreted draxin, there was no significant difference between the electroporated side and the control side. The ganglion crest rootlets of cranial nerves IX and X formed normally on the control side. The shape of the ganglion crest rootlets of cranial nerves IX and X appeared to be normal on the electroporated side of the same embryo (Fig. 5D–F). After overexpression of the transmembrane form of draxin, a significant difference was found between the electroporated side and the control side. The ganglion crest rootlets of cranial nerves IX and X formed normally on the control side. However, the normal structure of ganglion crest rootlets of cranial nerves IX and X was missing on the electroporated side of the same embryo. Only a few ganglion crest rootlets of cranial nerves IX and X projected from the hindbrain (Fig. 5G–I). The number of obviously thick ganglion crest rootlets of cranial nerves was counted in each side of the embryo. The ratio of the rootlets number in the electroporated side to the number of rootlets in the contralateral side in each embryo was counted as relative nerve root number. At least 15 embryos were used for analyses. The relative nerve root number in control electroporated embryos was 0.97 ± 0.17, the relative nerve root number in secreted draxin electroporated embryos was 0.93 ± 0.15, and the relative nerve root number in transmembrane draxin electroporated embryos was 0.42 ± 0.07. The relative nerve root number was significantly decreased in the transmembrane draxin-overexpressed embryos (p < 0.05; Fig. 5J).
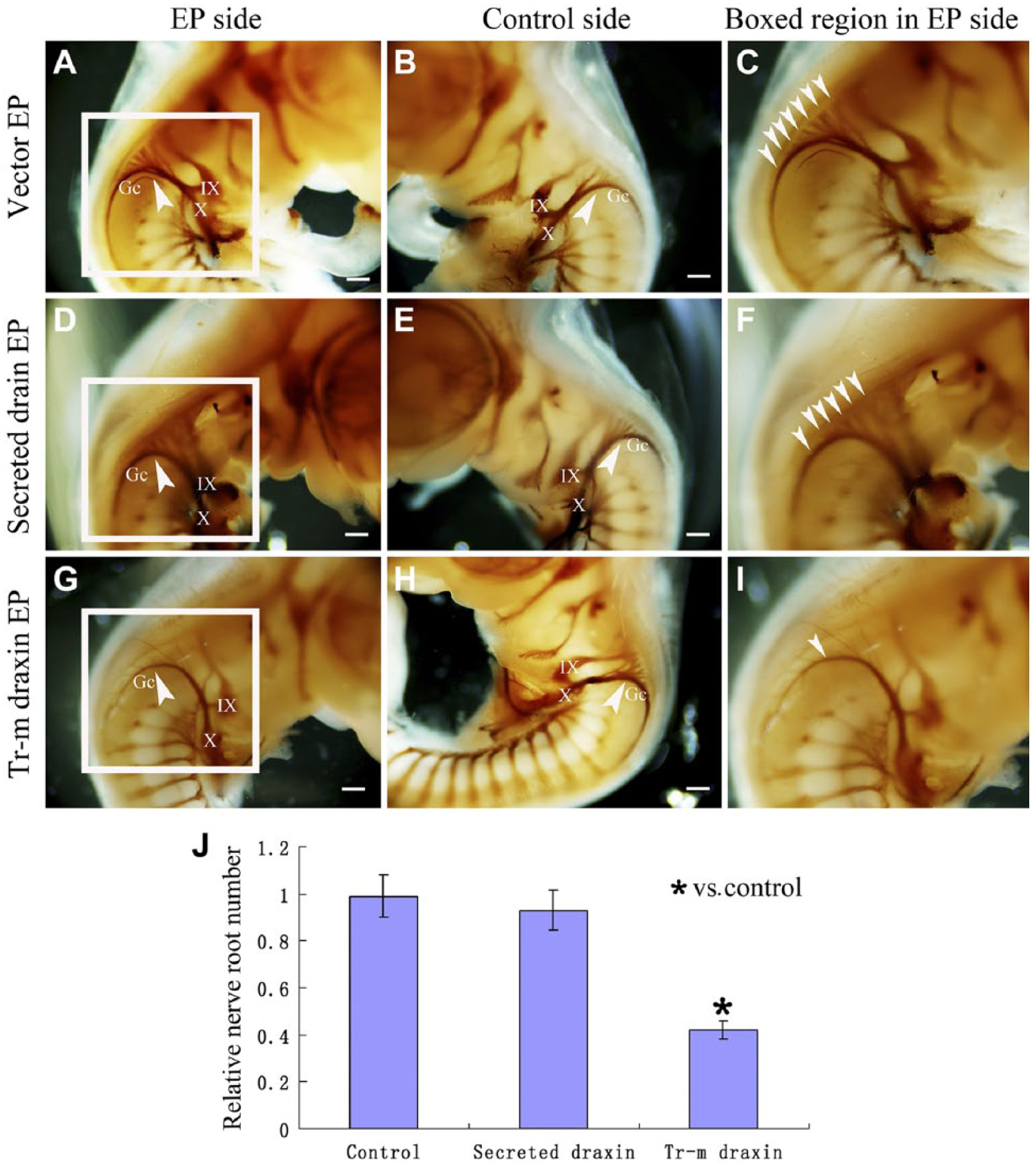
Overexpression of transmembrane draxin causes abnormal formation of the rootlets of ganglion crest of cranial nerves (Gc) in the chick hindbrain. Lateral view of 23C10 whole-mount immunostaining of Hamburger–Hamilton (HH) stages 25–26 chick hindbrain. (A) Electroporated side after control vector electroporation. The arrowhead indicates the normally formed Gc. (B) Control side using the same embryo as A. The arrowhead indicates the normally formed Gc. (C) Higher magnification of the boxed region in A. Arrowheads indicate all the thick rootlets of the Gc. (D) Electroporated side after secreted draxin electroporation. The arrowhead indicates that no obvious abnormality was found. (E) Control side using the same embryo as D. The arrowhead indicates that the Gc was formed normally. (F) Higher magnification of the boxed region in D. Arrowheads indicate all the thick rootlets of the Gc. (G) Electroporated side after transmembrane draxin electroporation. The arrowhead indicates that the normal structure of Gc rootlets, which just left the hindbrain, was missing. (H) Control side using the same embryo as G. The arrowhead indicates that the Gc was formed normally. (I) Higher magnification of the boxed region in G. Arrowheads indicate all the thick rootlets of the Gc. (J) Column diagram shows the statistical difference of the relative nerve root number in different groups. Abbreviations: EP, electroporation; Tr-m, transmembrane. Gc, ganglion crest of cranial nerves X; X, cranial nerves X; IX, cranial nerves IX. *p < 0.05. Scale bar = 300 µm.
Misprojected Axons Were Not Commissural or Motoneuron Axons, but the Abnormal Formation of the Ganglion Crest Rootlets of Cranial Nerves IX and X Might Be Related With the Motor Axonal Projection
The chick commissural axon-specific marker, Tag-1, was also used, and no obvious Tag-1-positive axonal misprojections could be detected after overexpression of transmembrane draxin (Fig. 6A–C). At lower magnification, no obvious Tag-1-positive commissural axons could be detected in the dorsal roof of the hindbrain. In the same section, the ventral projections of the commissural axons in all of the embryos were significantly affected (Fig. 6A). Co-induced green fluorescent protein (GFP) signaling indicated the ectopic expression of Draxin (Fig. 6B). Very limited Tag-1-positive axons were detected in the dorsal roof of the hindbrain at higher magnification (Fig. 6C). Two of 15 embryos showed this type of limited labeling of Tag-1-positive axons in the dorsal roof, but all the 15 embryos showed the inhibition of the ventral projections of the commissural axons. To quantify the inhibition of the ventral projections of the commissural axons, we measured the relative anti-Tag-1 integral optical density (IOD) value. Ligature between the midpoint of the roof plate and floor plate was used to separate the section into two parts. The IOD value in each part was measured. The ratio of IOD value between the electroporated side and the contralateral side was counted as the relative anti-Tag-1 IOD value. The relative anti-Tag-1 IOD value in normal embryos was used as the control. A lower relative anti-Tag-1 IOD value indicated that the Tag-1-positive signaling was reduced. This quantification assay indicated that Tag-1-positive signaling was significantly decreased on the electroporated side with draxin overexpression (Fig. 6D). One motoneuron marker, SC1, also showed a reduction in the number of efferent projections from the hindbrain (Fig. 6E–H). Thick axonal bundles inside the hindbrain could be detected on the control side, and these axons passed through the ventral peripheral area of the hindbrain basal zone to form the root of the cranial nerve (Fig. 6G). Co-induced GFP signaling indicated the ectopic expression of Draxin (Fig. 6F). Thick axonal bundles inside the hindbrain could also be detected on the electroporated side. However, the number of axons that passed through the ventral peripheral area of the hindbrain basal zone was reduced (Fig. 6E). Moreover, the root of the cranial nerve became narrow or was absent on the electroporated side (Fig. 6E). We measured the width of axon bundles which passed through the ventral peripheral area of the hindbrain basal zone. We also measured the largest width of the projecting axon bundle inside the hindbrain basal zone. The ratio between the width of axon bundle passing through and the largest width of the projecting axon bundle inside the hindbrain basal zone in the same side was counted as the relative width of axon bundle. The lower relative width of the axon bundle indicated that the axons passing through the ventral peripheral area were reduced. This quantification indicated that the relative width of the axon bundle was significantly decreased on the electroporated side compared with the control side (Fig. 6H).
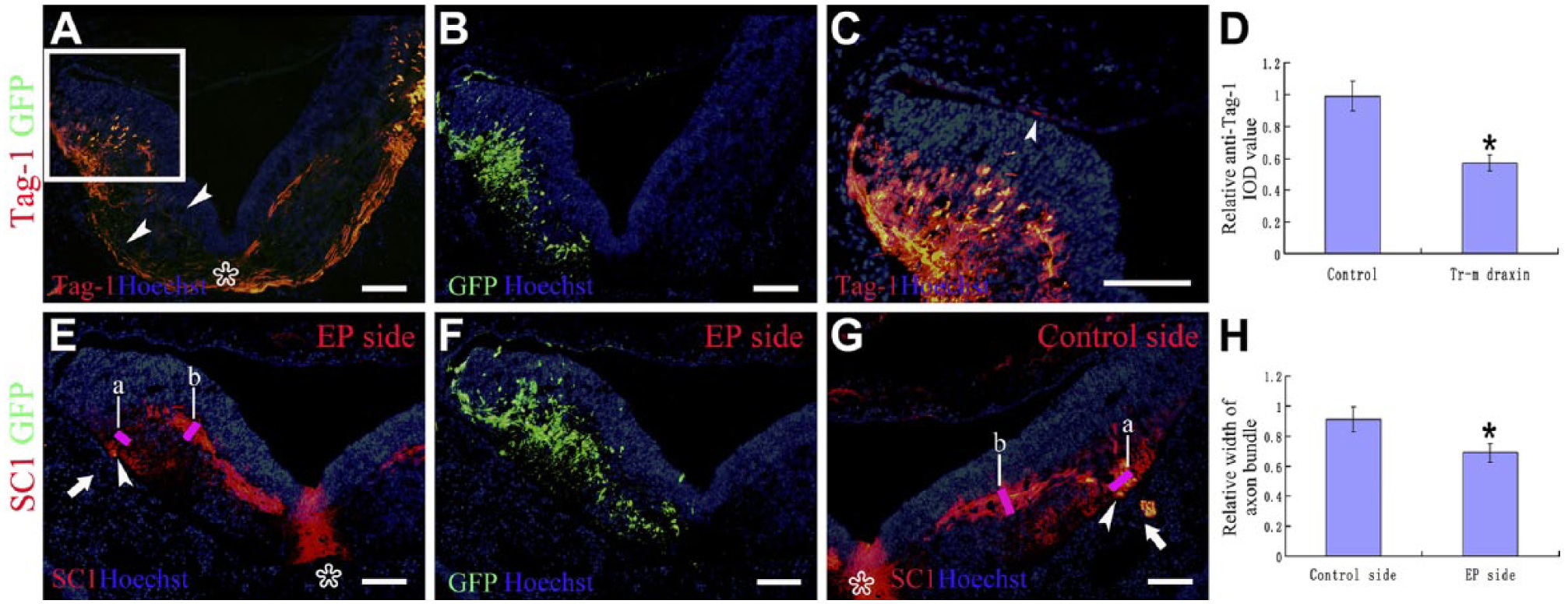
Abnormal formation of the Gc rootlets might be related to the motor axon projections. Transmembrane draxin-overexpressed embryos were used. At level r7 of the chick hindbrain. Asterisks point to the ventral midline. Hoechst staining shows the structure of the sections. (A) Chick Tag-1 staining at low magnification. Arrowheads indicate that the ventral projection of the commissural axons was significantly affected. (B) Enhanced green fluorescent protein (EGFP) expression using the same section as A showed that the overexpressed side was left. (C) Higher magnification of the boxed region in A. Arrowheads showed a few misprojected axons could be detected. (D) Column diagram shows the statistical difference of the relative anti-Tag-1 integral optical density (IOD) value in different groups. (E) SC1 staining on the electroporated side. Arrowheads showed that the number of motoneuron axons passing through the ventral peripheral area of the hindbrain basal zone was reduced. Arrows showed that the root of the cranial nerve was absent on the electroporated side. (F) EGFP expression using the same section as E showed that the overexpressed side was left. (G) SC1 staining of the contralateral side using the same section as E. Arrowheads showed many motoneuron axons passed through the ventral peripheral area of the hindbrain basal zone. Arrows showed the root of the cranial nerve on the control side. No SC1-positive axons could be detected in the dorsal roof of the hindbrain on either side. (H) Column diagram shows the statistical difference of the relative width of axon bundle in different groups: a points the width of axon bundle which passes through the ventral peripheral area of the hindbrain basal zone, and b points the largest width of the projecting axon bundle inside the hindbrain basal zone. Abbreviations: GFP, green fluorescent protein; EP, electroporation. *p < 0.05. Scale bar = 100 µm.
Discussion
A signal sequence trap screen was performed to identify the repulsive axon guidance protein draxin, which is a secreted protein that is widely expressed in the central nervous system. 9 Draxin is required for the development of the spinal cord and the forebrain commissures. 9 Draxin is also involved in the migration of the neural crest 10 and of the dI3 interneurons in the chick spinal cord, 10 and is expressed in the rhombic lip and migratory stream of some pre-cerebellar neurons in the developing hindbrain of mice. 20 However, the potential role of draxin in the axonal projection in the chick hindbrain is largely unknown. We first examined the temporal and spatial correlation between the distribution pattern of the draxin protein and the development of 23C10-positive axon projections in the chick hindbrain (Fig. 1). At different developmental stages, the structures that were labeled with both draxin and 23C10 were always located inside the hindbrain, especially in the peripheral area of the basal zone. At the same time, there was a scattered punctate distribution of draxin protein located around the 23C10-positive axon bundles inside the peripheral area of the hindbrain basal zone. This result lets us to assume that draxin protein distribution and the projection of 23C10-positive axons had a strong spatial and temporal correlation, and draxin might be involved in the regulation of 23C10-positive axon projections, such as fascicular projection, to their final destination in the peripheral area of the hindbrain basal zone. The expression of draxin in the exiting points of the cranial nerves was decreased and discontinuous compared with the expression of draxin in ventral region of exiting points of cranial nerves in hindbrain (Fig. 1F). This result lets us to speculate that the changes of draxin expressing around the exiting point of cranial nerves might be involved in restricting the projection of cranial nerves from the hindbrain.
To examine the probable function of draxin during the development of 23C10-positive axons, we performed in vivo experiments and confirmed that draxin plays an important role in the fascicular projection of 23C10-positive axons in the chick hindbrain. Overexpression of draxin, especially the transmembrane form of draxin, caused axons and axon bundles to misproject in the dorsal roof area of the hindbrain, which is the thin roof plate of the fourth ventricle. However, the overexpression of the control vector did not cause any changes in the dorsal roof area of the hindbrain (Figs. 2 and 4). The reason for the very weak function of secreted draxin overexpression may be that draxin is a secreted protein with a high affinity for lateral basement membranes. 9 This suggests that after electroporation, the overexpressed secreted draxin protein was widely diffused inside the hindbrain and the lateral basement membranes. However, after the overexpression of the transmembrane form of draxin, the draxin protein was tethered to the membrane of cell bodies which were electroporated inside the hindbrain. This gave more time for transmembrane draxin to impact cells and axons than the secreted form of draxin. Moreover, during neural cell movement, the possibility is high that transmembrane form of draxin may directly influence and change the projection of surrounding axons.
The cranial sensory ganglia of the vertebrate head are derived from two diverse populations of migratory cells that originate in the neurogenic placodes and the neural crest. 21 The patterning of neural crest cell migration within the hindbrain region is essential for directing the epibranchial neuronal cells inward to establish their central connections. 22 The neural crest cells normally migrate in tightly packed neural crest cell streams, and overexpression of some types of repulsive axon guidance proteins in the chick hindbrain, such as Sema 3A and Sema 3F, results in disrupted stream migration. 23 In the zebrafish hindbrain, the FBMNs undergo a chain-like tangential migration from their birthplace to their final destination. During this migration, the pioneer neuron and its axon are crucial to later migrating cells. 24 Chick dorsal root ganglion axons enter the spinal cord through the entry zone (the restricted dorsolateral portion of the spinal cord) and then run longitudinally in a fasciculated manner, forming the prospective dorsal funiculus. After a waiting period following the entry into the marginal zone of the spinal cord, these axons invade the mantle layer. 25 Overexpression of draxin, especially transmembrane form of draxin, caused some HNK-1-positive axons to appear in the dorsal roof of the hindbrain (Fig. 4). It is possible that overexpressed draxin caused changes in some cells inside the hindbrain, which may be the projection targets of the cranial sensory ganglia cells. As a result, axons projecting from the cranial sensory ganglia to the mislocated cells altered their targeting to the dorsal roof of the hindbrain; however, more evidence is needed to support this speculation. Another possibility was that overexpression caused increased draxin distribution in the hindbrain, especially in the exiting point of cranial nerves. As a result, the regulating function of draxin was increased and caused the afferent sensory axons from the cranial sensory ganglia to misproject to the dorsal roof of the hindbrain.
To examine the location from which the misprojected 23C10-positive axons originated, we performed whole-mount nerve immunostaining following electroporation. After overexpression of transmembrane draxin, a significant difference was found between the electroporated side and the control side. The normal structure of the ganglion crest rootlets of cranial nerves IX and X was missing on the electroporated side of the embryo (Fig. 5). The reason for the overexpression of secreted draxin that did not cause obvious changes in the ganglion crest rootlets of cranial nerves IX and X might be the weak function of secreted draxin. In the whole-mount immunostaining preparation of embryos overexpressing secreted draxin, only a few axons were found to misproject to the dorsal roof of the hindbrain, and this minor loss of normally projecting axons could not cause an obvious difference by whole-mount immunostaining. We further analyzed the embryos with a more significant phenotype using the commissural axon-specific marker, Tag-1. The ventral projection of the commissural axon was significantly affected, but we could not detect obviously misprojected commissural axons in the dorsal area. Only in a few embryos, very limited commissural axons were detected at higher magnification (Fig. 6), which is not consistent with an abundant misprojection of axons in the dorsal roof of hindbrain. At the same time, we could not detect misprojected motor neuronal axons in the dorsal roof of the hindbrain (Fig. 6). Combining the above results with the fact that neural crest cells avoid draxin expression sources, 9 we can speculate that the misprojected axons might be sensory afferent fibers of cranial nerves. It is possible that secreted and transmembrane draxin-overexpressing cells could affect the projection of axons from the surrounding cells, especially the axons from the cerebral ganglion cells. Because of the repulsive function of draxin, some projecting axons, especially sensory ganglia nerves, might be displaced their normal location to the dorsal roof of the hindbrain. Direct effects of draxin on this effect must be examined in future studies. We also found that a reduced number of motoneuron axons pass through the ventral peripheral area of the hindbrain basal zone in the draxin-overexpressed side (Fig. 6). This result suggested that the abnormal formation of the ganglion crest rootlets of cranial nerves IX and X might be caused by the inhibition of the outgrowth of efferent projections of the motor axons of these cranial nerves. But we could not exclude the possibility that some formed motoneuron axons inside the hindbrain basal zone changed their direction to the dorsal roof abnormally by the inhibitory function of overexpressed draxin, because that not all of the misprojected 23C10-positive axons were also HNK-1 positive. It is possible that the overexpression will increase the distribution of draxin in the hindbrain, especially in the lateral basement membrane. As a result, the inhibitory function of draxin at the exit point of the cranial nerves might be increased. Finally, the formation of the motor axons of these cranial nerves was decreased, and the projection of formed motoneuron axons was changed. These two possibilities suggested that overexpressed draxin might affect the efferent and afferent projections of the cranial nerves simultaneously.
The 23C10 antibody can detect many types of neural axons (including the motoneuronal and sensory neural axons) during the normal development of the chick hindbrain. Compare with the distribution pattern of Tuj-1, HNK-1, and Tag-1 antibodies, together with colocalization detection (Fig. 4), we demonstrate that 23C10 antibody could detect all the Tuj-1 positive neurons. At the same time, 23C10 antibody might detect most HNK-1 positive sensory neural cells. The distribution pattern of draxin was different between the peripheral area of hindbrain basal zone and around the exiting point of cranial nerves. The scattered punctate distribution of draxin around the 23C10-positive axons in the peripheral area of the hindbrain basal zone might involve in the formation of fascicular projection of axon bundles. The continued flaky distribution of draxin that was located ventrally to the exiting point of cranial nerves might function to regulate the proper projecting location of cranial nerve fibers, because the root of cranial nerve fibers was just adjacent to the dorsal boundary of the continued flaky distributed draxin. Gain-of function data indicated that the draxin expression in the hindbrain tissue has a strong repellant function in regulating the normal axonal projections of the cranial nerves, particularly in regulating the regular bundling of cranial nerves IX and X axonal projections.
Netrin receptor deleted colon cancer (DCC) had been shown to be a receptor for draxin in regulating axon projection in mice. 26 Unfortunately, we could not detect the expression of DCC in the chick hindbrain (data not shown). However, a DCC homologue has not yet been identified in the chicken genome. 27 Neucrin, which is identical to draxin, is a unique secreted Wnt antagonist and plays roles in neural development in zebrafish. Neucrin knockdown affected specification of the dorsal region in the midbrain and the hindbrain, suppressed neuronal differentiation, and caused increased cell proliferation and apoptosis in developing neural tissues. 28 We plan to analyze the functional receptor of draxin during the axonal projection in chick hindbrain and the function of draxin in neuronal differentiation and proliferation in the chick hindbrain in future studies.
We observed some 23C10-positive axons on the contralateral side in which the electroporation was not performed. This observation might have been caused by the crossing of an axon from the draxin-overexpressing side of the embryo. It is possible that the overexpressed draxin was located within the axons and crossed to the contralateral side with these axons. After crossing the ventral midline, the draxin protein would contact the axons on the contralateral side and function similarly to its effect on the overexpressed side to alter the axon projections.
Footnotes
Author Contributions
SZ participated in drafting the manuscript. LW and LK performed section immunohistochemistry staining. GH and JD performed whole-mount immunohistochemistry staining. SL performed statistical analysis. HC and HT participated in the design of this article. YS performed in the design of this study and drafting the manuscript. All authors read and approved the final manuscript.
Competing Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by the National Natural Science Foundation (31100789), the China Postdoctoral Science Foundation (2012T50244), the Project sponsored by Scientific Research Foundation (SRF) for Returned Overseas Chinese Scholars (ROCS), State Education Ministry (SEM), and the PhD Programs Foundation of the Ministry of Education of China (20111323120006).
