Abstract
Obesity induces chronic, low-grade inflammation, which increases the risk of colon cancer. We investigated the preventive effects of Bardoxolone methyl (BARD) on high-fat diet (HFD)-induced inflammation in a mouse colon. Male C57BL/6J mice (n=7) were fed a HFD (HFD group), HFD plus BARD (10 mg/kg) in drinking water (HFD/BARD group), or normal laboratory chow diet (LFD group) for 21 weeks. In HFD mice, BARD reduced colon thickness and decreased colon weight per length. This was associated with an increase in colon crypt depth and the number of goblet cells per crypt. BARD reduced the expression of F4/80 and CD11c but increased CD206 and IL-10, indicating an anti-inflammatory effect. BARD prevented an increase of the intracellular pro-inflammatory biomarkers (NF-қB, p NF-қB, IL-6, TNF-α) and cell proliferation markers (Cox2 and Ki67). BARD prevented fat deposition in the colon wall and prevented microbial population changes. Overall, we report the preventive effects of BARD on colon inflammation in HFD-fed mice through its regulation of macrophages, NF-қB, cytokines, Cox2 and Ki67, fat deposition and microflora.
Introduction
Obesity increases the risk of developing colon cancer (Liu et al. 2012; Bardou et al. 2013). Studies have shown that the incidence of colon cancer is correlated with body size and body weight (Kono et al. 1999; Bassett et al. 2010). The risk of developing colon cancer is doubled in overweight or obese people as compared with healthy people with normal weight (Giovannucci et al. 1995). Diet-induced obese mice have high levels of inflammation and an increased risk of carcinogenesis (Erdelyi et al. 2009). Colon cancer is the third-most diagnosed cancer in the world (Ferlay et al. 2015); because of this, preventing obesity-associated colon cancer is important.
Consumption of HFD is a key factor in the development of obesity, which increases fat deposition and causes changes to the intestinal structure and morphology. Increased fat deposition has been observed in the intestine of diet-induced obese mice and is a risk factor for inflammatory bowel disease (Douglass et al. 2012; Paik et al. 2013). The chronic consumption of a HFD can affect colon crypts, which is accompanied by a disruption to the colon epithelium, as well as reduced goblet cell number, and increased epithelial cell proliferation (Bird and Stamp 1986; Paturi et al. 2010).
Inflammation is another risk factor for colon cancer (Giugliano et al. 2006; Day et al. 2013; Fang et al. 2013). It has been suggested that gut inflammation and oxidative stress induced by a HFD have carcinogenic effects in mice (Erdelyi et al. 2009). For instance, a HFD increases nuclear factor-κ light-chain enhancer of activated B cells (NF-қB) activation in the epithelial cells of the small intestine and colon in mice (Ding et al. 2010; Kim et al. 2012a). Moreover, a HFD elevates tumor necrosis factor alpha (TNF-α) protein by 72% in mouse colonic mucosa (Liu et al. 2012). A HFD also increases colon cancer cell proliferation, as observed by an increase in the level of nuclear antigen Ki67 (Ki67) expression (Park et al. 2012). Ki67 is a cell proliferation marker used to assist in the diagnosis of colon cancer (Shepherd et al. 1988; Takano et al. 1996; Nabi et al. 2008). Moreover, overweight people have a higher level of Cox2 mRNA in the colon as compared with people with weight in the normal range (Delage et al. 2007). A HFD promotes colitis in mice, which is associated with a shortened colon and an elevated level of Cox2 (Kim et al. 2012a). Compared with mice fed a normal diet, those fed a HFD are more susceptible to carcinogen-induced colon tumorigenesis. In particular, a HFD increases the development of polyps and aberrant crypt foci in colon cancer mouse models (Baltgalvis et al. 2009; Padidar et al. 2012). Furthermore, the gut microbiota composition correlates to body-weight gain, as well as to the local and systemic inflammation associated with human obesity (Verdam et al. 2013). Strategies to modulate gut microflora may help to prevent the risk of colon-associated complications in obesity.
Bardoxolone methyl (BARD) is a synthetic pentacyclic triterpenoid compound and a derivative of oleanolic acid. In other reports, this compound and its analog RTA 405 are effective in the suppression of body weight in obese humans and body fat in obese mice, respectively (Manenti et al. 2012; Chin et al. 2013). Anti-inflammatory, anti-obesity and anti-cancer effects of BARD have been demonstrated in preclinical and clinical studies (Liby et al. 2009; Saha et al. 2010; Tran et al. 2013; Wu et al. 2014). BARD and its analog 2-cyano-3,12-dioxooleana-1,9-dien-28-oic acid-9,11-dihydro-trifluoroethyl amide are immunomodulators and inhibitors of NF-қB signalling and various inflammatory mediators (ie TNF-α, Cox2) (Deeb et al. 2007; Aminzadeh et al. 2014; Choi et al. 2014). BARD suppresses the colitis-associated colon inflammation in mice and protects human colon epithelial cells from radiation-induced neoplastic transformation (Eskiocak et al. 2010; Choi et al. 2014). In this study, we investigated the cellular mechanism of BARD in the colons of mice fed a HFD by assessing the expression of various inflammatory and anti-inflammatory mediators, the degree of fat deposition and the changes in colon microbiota.
Materials & Methods
Experimental Procedures on Animals
Male, 12-week-old, C57BL/6J mice were obtained from the Animal Resource Centre (Perth, WA). After 1 week acclimatization, the mice were divided into three groups (n=7): (1) mice fed a normal lab chow diet (LFD group, Fat/Protein/Carbohydrate = 5:18:77 as % energy fat) (Vella Stock Feeds, Doonside, NSW); (2) mice fed a high-fat diet (HFD group, Fat/Protein/Carbohydrate = 40:15:45 as % energy fat) (SF11-095, Speciality Feeds, WA); and (3) mice fed the same HFD and supplemented with BARD (CAS: 218600-53-4, Shanghai Haoyuan Chemexpress Co., Ltd) in drinking water at dose of 10 mg/kg body weight (HFD/BARD group). It has been previously shown that a HFD (over 20 weeks) induces significant intestinal inflammation in C57BL/6J mice (Axling et al. 2012; Shen et al. 2014). We therefore chose 21 week of BARD intervention to observe the preventive effects of this compound on HFD-induced colon inflammation. We used BARD at dose 10 mg/kg body weight based on a previous study in which BARD mixed in the diet has potent anti-inflammatory and anti-tumor effects in mice (Liby et al. 2009; Wu et al. 2014). Since our aim was to study obesity, we chose to supplement BARD into the drinking water so as to avoid BARD from potentially reducing the palatability of the HF diet, which may influence food intake. We have previously reported that oral BARD administration at a dose of 10 mg/kg can suppress body weight in HFD mice (Camer et al. 2015b; Dinh et al. 2015b). At week 21 of treatment, all mice were sacrificed for tissue collection. Whole colons were washed in phosphate-buffered saline and cut longitudinally. Part of the colon was kept frozen at -80°C for oil-red O staining of neutral fat while the remaining part of the colon was rolled and fixed in 4% paraformaldehyde. The fixed tissues were embedded in paraffin for histology and immunohistochemistry. The cecal content was collected and stored at -80°C for an analysis of microbial population. All procedures were approved by the Animal Ethics Committee of the University of Wollongong, NSW, Australia, and complied with the Australian Code of Practice for the Care and Use of Animals for Scientific Purposes. The approval ID for this study is AE12/15.
Histology and Immunohistochemistry
Paraffin-embedded colons were cut to 5-µm sections and stained with hematoxylin and eosin (POCD Scientific, Artarmon, NSW). Histological photographs of the stained sections were taken using a Leica microscope (Leica Microsystems, Wetzlar, Germany). ImageJ 1.46r software (http://imagej.nih.gov/ij/download.html) was used for measurements of the average crypt length and the number of goblet cells per crypt. Crypt lengths were measured using a ×10 objective and dimensions were calculated using a graticule (μm). A total of 42 individual crypts from 7 mice (6 crypts per mouse) in each group was used for measurement of colon crypt lengths, as described in previous studies (Steinbrecher et al. 2002; Li et al. 2007). The number of goblet cells per crypt was counted from 42 individual crypts in each group (n=6 crypts per mouse; n=7 mice per group) in accordance with a previous study (Taraban et al. 2011). Quantification was performed in a blind, randomized order.
To determine the effect of BARD on fat deposition in mouse colons, colons were stained with oil-red O. Briefly, frozen colon tissue was mounted in optimum cutting temperature medium, cut at 10 μm, and fixed in 10% formalin for 10 min. Colons were washed with distilled water and incubated in 100% propylene glycol for 2 min. The sections were then incubated with 0.5% oil-red O (Sigma-Aldrich Pty. Ltd, Sydney, NSW, Australia) at 60°C for 7 min. Following this, all sections were directly washed with 85% propylene glycol for 1 min, and washed twice with distilled water. The stained sections were then counterstained with hematoxylin (POCD Scientific, Artarmon, NSW), washed thoroughly with tap water, and cover-slipped with mounting medium for imaging. Six fields per sample (n=7) (×10) was used to measured oil-red O staining by ImageJ 1.46r software.
We used immunohistochemistry to determine the expression of different inflammatory and proliferative biomarkers in the colon crypts, which is in accordance with a top-down morphogenesis of colorectal tumorigenesis described in a previous report (Shih et al. 2001). The antibodies used in this study have been previously employed and are reliable biomarkers for detecting gut inflammation and carcinogenesis in humans and mice (Charalambous et al. 2009; Vitor et al. 2009; Steinhagen et al. 2012; Göranzon et al. 2013). The immunohistochemical procedure was based on our previous work (Dinh et al. 2015a). The primary antibodies used for immunohistochemical staining included: anti-F4/80 (ab6640) at 1:1000, anti-CD11c (ab33483) at 1:500 or anti-CD206 (ab64693) at 1:3000, anti-IL-10 (ab34843) at 1:400, anti-Ki67 (ab15580) at 1:500 from Abcam Inc (Cambridge, MA); NF-қBp65 (sc-109) at 1:100, phosphorylated NF-қBp65 (sc-33020) at 1:100, IL-6 (sc-7920) at 1:100, IL-1β (sc-7884) at 1:100, TNF-α (sc-8301) at 1:100 from Santa Cruz Biotechnology (Dallas, TX). Secondary antibodies were used at 1:250 dilution: rabbit anti-rat IgG biotinylated secondary antibody (ab6733), goat anti-Armenian hamster IgG H&L biotin (ab5744), and goat anti-rabbit IgG H&L biotin (ab6720), from Abcam Inc. Negative controls for all immunostaining were carried out by omitting the primary antibody. Within each group, six fields per sample (n=7) were used to quantify the morphological staining of all biomarkers. CD11c (×20), CD206 (×20), phosphorylated NF-kBp65 (×40), and Ki67 were quantified as the percent of positive cells, whereas F4/80, NF-kBp65, Cox2, IL-1β, IL-6, TNF-α, IL-10 (×20) were quantified as the percentage positive area per image.
Real-time PCR (qRT-PCR) was used to quantify microbial strains from cecal content. One hundred mg of cecal content from each mouse was used for genomic DNA extraction with Isolate DNA kit (Bioline, Sydney, Australia). DNA concentration was determined using Agilent Bioanalyzer (Agilent Technologies, Santa Clara, CA).
For Quantitative real-time PCR (qPCR), 20 ng DNA was used per PCR reaction (20 µl). PCR was performed using SensiFast™ SYBR No-ROX reagents (Bioline). Different primers were used to quantify dominant bacterial strains, including the Gram-positive Bifidobacteria, Lactobacilli, and the Gram-negative Bacteroides-Prevotella, which were purchased from Sigma-Aldrich (Sydney, Australia) (Table 1). Quantification was carried out using Lightcycler 480 real-time PCR system (F. Hoffmann-La Roche Ltd, Switzerland), with a two-step PCR protocol. The PCR conditions were 95°C for 3 min, followed by 40 cycles of 95°C for 5 sec, 60°C for 10 sec, and 72°C for 10 sec. A positive value was determined when a crossing point (CP) was smaller than 35 (Durant et al. 2009). Relative bacterial population was quantified as per the Livak method (2-ΔΔT) using universal primer values as a reference (Condezo-Hoyos et al. 2014).
Primers for qPCR.
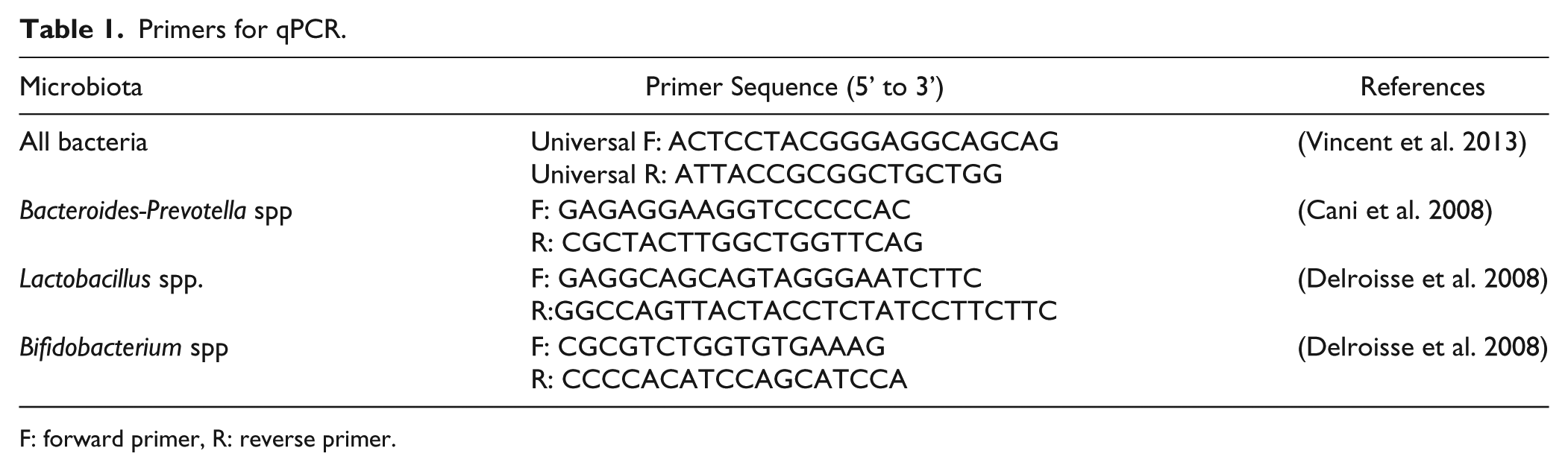
F: forward primer, R: reverse primer.
Statistical Analysis
Sample size in the present study was adopted from a previous study (Gu et al. 2013) and confirmed by a power calculation using JMP software (SAS Institute Inc., Cary, NC). We used SPSS 19 package (SPSS, Chicago, IL) to perform a one-way analysis of variance test, and a least significant difference post-hoc analysis for data analysis. We used the Pearson’s test for correlation among the histological and immunohistochemical staining parameters. All values are expressed as mean ± SEM. Statistical significance was considered at p<0.05.
Results
BARD Improves Morphology in the Colons of HFD-fed Mice
HFD mice had shorter colons, as compared with LFD and HFD mice administered BARD (Fig. 1A). HFD/BARD mice had significantly reduced colon weight per colon length (0.017 g/cm; Fig. 1B). This is 26% (p<0.01) lower than the LFD mice (0.024 g/cm) and 34% (p<0.001) lower than the HFD control mice (0.026 g/cm).
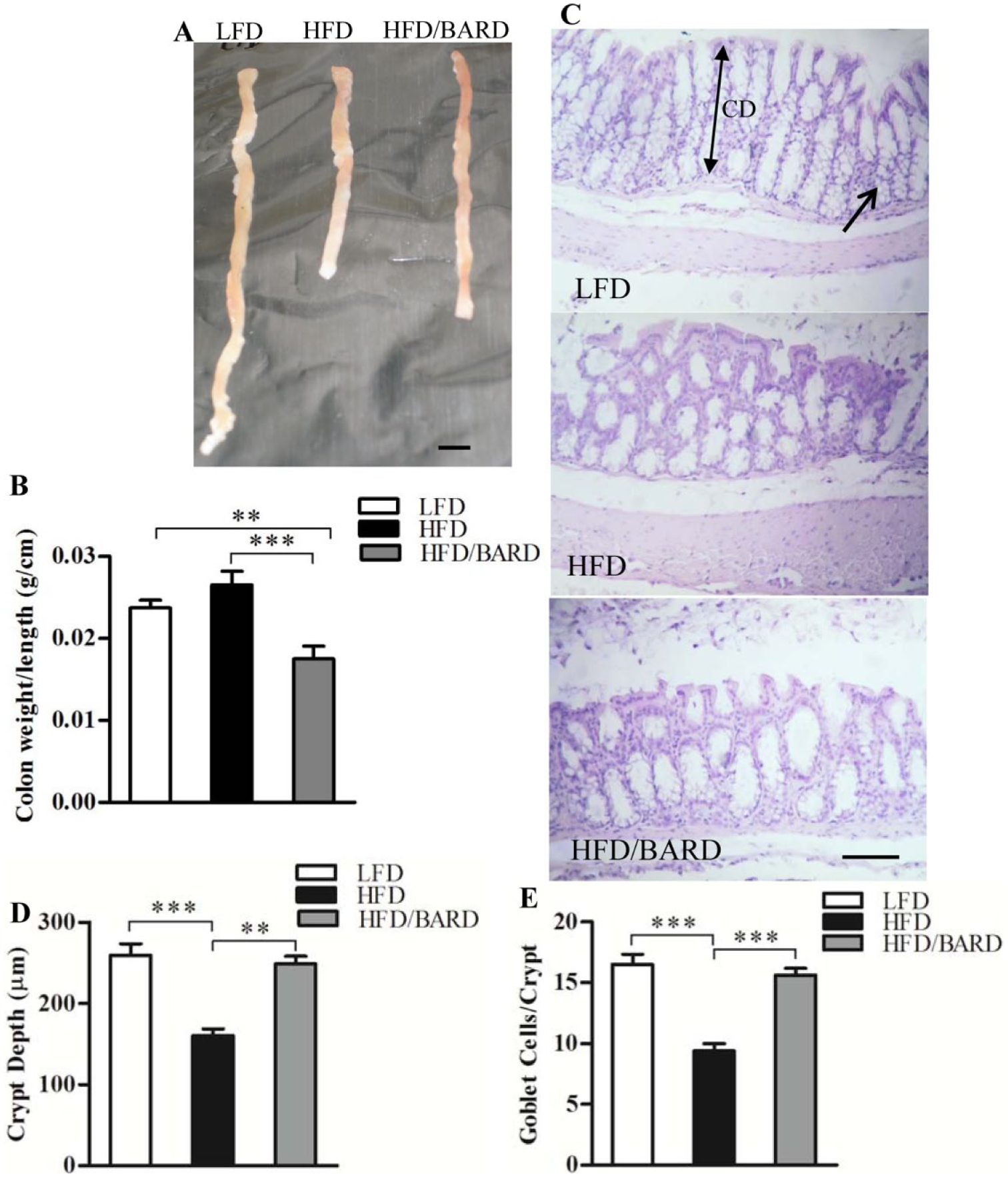
Effect of bardoxolone methyl (BARD) on colon morphology in mice fed a low-fat diet (LFD), a high-fat diet (HFD), or a high-fat diet supplemented with BARD (HFD/BARD). (A) Mouse colons. (B) Colon weight per length (g/cm) (n=7). (C) Hematoxylin and eosin stained sections (×10). The arrow indicates goblet cells, whereas the double-headed arrow indicates the colon crypt depth (CD). (D) Crypt depth (µm). (E) Number of goblet cells per crypt. All data are presented as mean ± SEM. A total of 42 crypts from 7 mice in each group were used for the analysis of crypt depth and number of goblet cells per crypt. **p<0.01; ***p<0.001. Scale (A) 1 cm; (C) 100 µm.
We investigated the effect of BARD on colon crypt depth and on the number of goblet cells per crypt in HFD mice by histological staining (Fig. 1C). Colon crypt depth in HFD mice was 38% (p<0.001) shorter than that in LFD mice (Fig. 1D). Compared with the HFD control, HFD/BARD mice had longer colon crypts (+55%, p<0.001), similar to the LFD mice. Similarly, the HFD mice had half as many goblet cells per crypt in the colon as LFD mice and HFD/BARD mice (p<0.001) (Fig. 1E). These data demonstrate that BARD improves colon morphology in HFD mice.
BARD Suppresses Infiltration of Inflammatory Macrophages in the Colons of HFD-fed Mice
The effect of BARD on macrophage infiltration was determined through the expression of F4/80 (total) and CD11c (M1 or inflammatory macrophage phenotype) immunostaining in the colon crypts (Fig. 2A–2C). HFD mice had 3.4% F4/80-positive area compared with 1.1% in LFD mice (Fig. 2D). Compared with the HFD control, HFD/BARD mice had significant reduction in F4/80-positive area (p<0.001). Similarly, the HFD mice had an increased level of CD11c-positive cells (2.8%), whereas the number of CD11c cells was reduced in the colons of both HFD/BARD and FD mice (Fig. 2E).
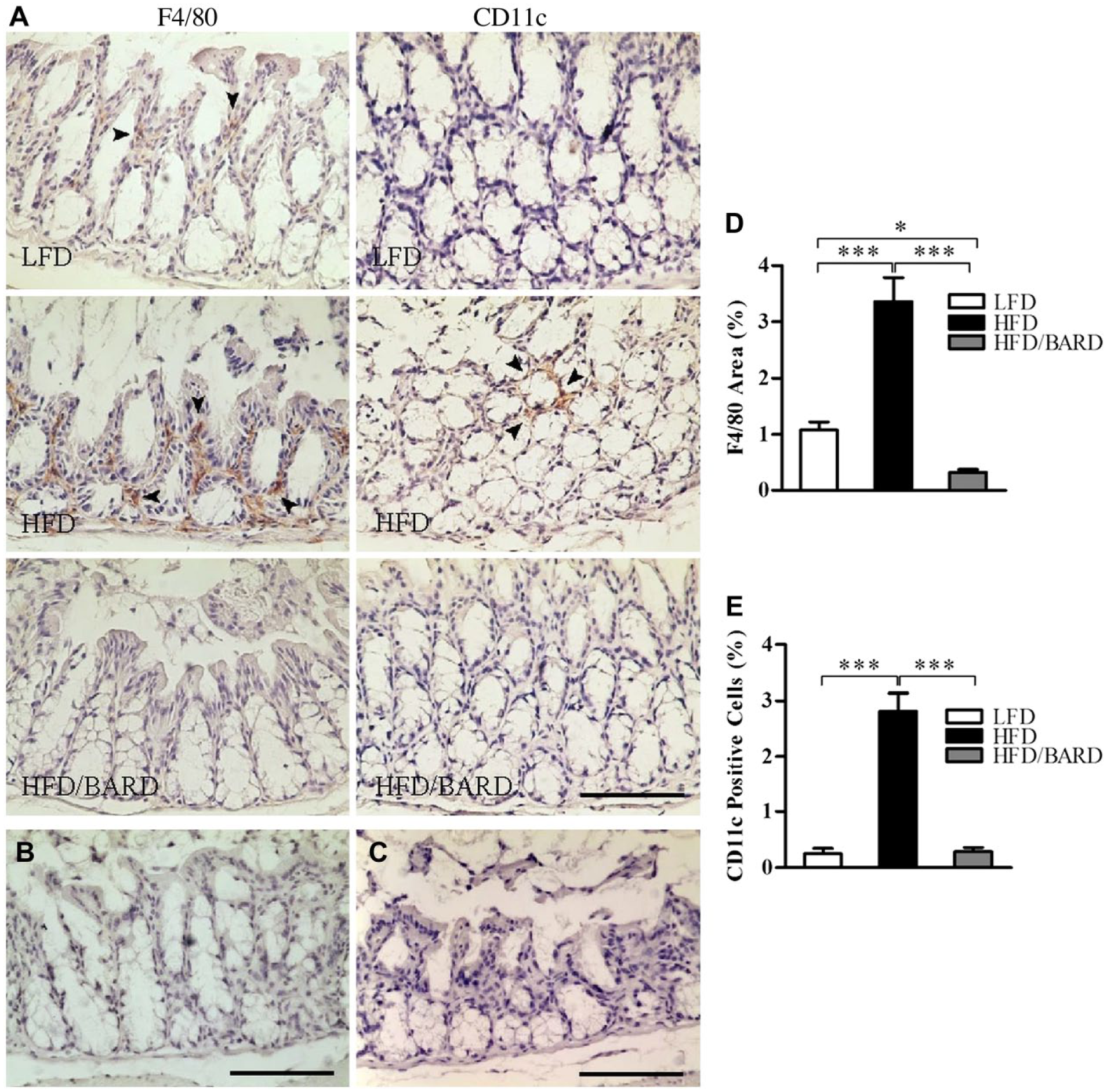
Effect of bardoxolone methyl (BARD) on the expression of F4/80 and CD11c macrophages in the colon crypts of mice fed a low-fat diet (LFD), a high-fat diet (HFD), or a high-fat diet supplemented with BARD (HFD/BARD). (A) F4/80- and CD11c-stained sections (×20). (B) Negative F4/80-stained section (×20). (C) Negative CD11c-stained section (×20). The arrowheads indicate F4/80 and CD11c staining (brown). (D) F4/80-positive area (%). (E) CD11c-positive cells (%). Six fields per sample (n=7) were used for statistical analysis. All data are presented as mean ± SEM. *p<0.05, ***p<0.001. Scale, 100 µm.
BARD Suppresses NF-қB Activation and Pro-inflammatory Cytokines in the Colons of HFD-fed Mice
We investigated the effect of BARD on NF-қB activation by assessing protein expression of total NF-қBp65 and its phosphorylation (pNF-қBp65) in the colon crypts (Fig. 3A–3D). The NF-қBp65-positive area in the colon crypts of HFD mice was 14.8%, which was significantly higher than that in the LFD (6%) and HFD/BARD (4.6%) mice (Fig. 3E; p<0.001). HFD mice had more pNF-қBp65 positive cells (36%) in the colon crypts as compared with LFD and HFD/BARD mice, which had reduced proportion of pNF-қBp65-stained cells (Fig. 3F; p<0.001). This reduction of NF-қBp65 by BARD was correlated with a reduction in phosphorylated (p)NF-қBp65 levels (Fig. 3G). These results therefore demonstrate the anti-inflammatory effect of BARD through a suppression of NF-қB activity.
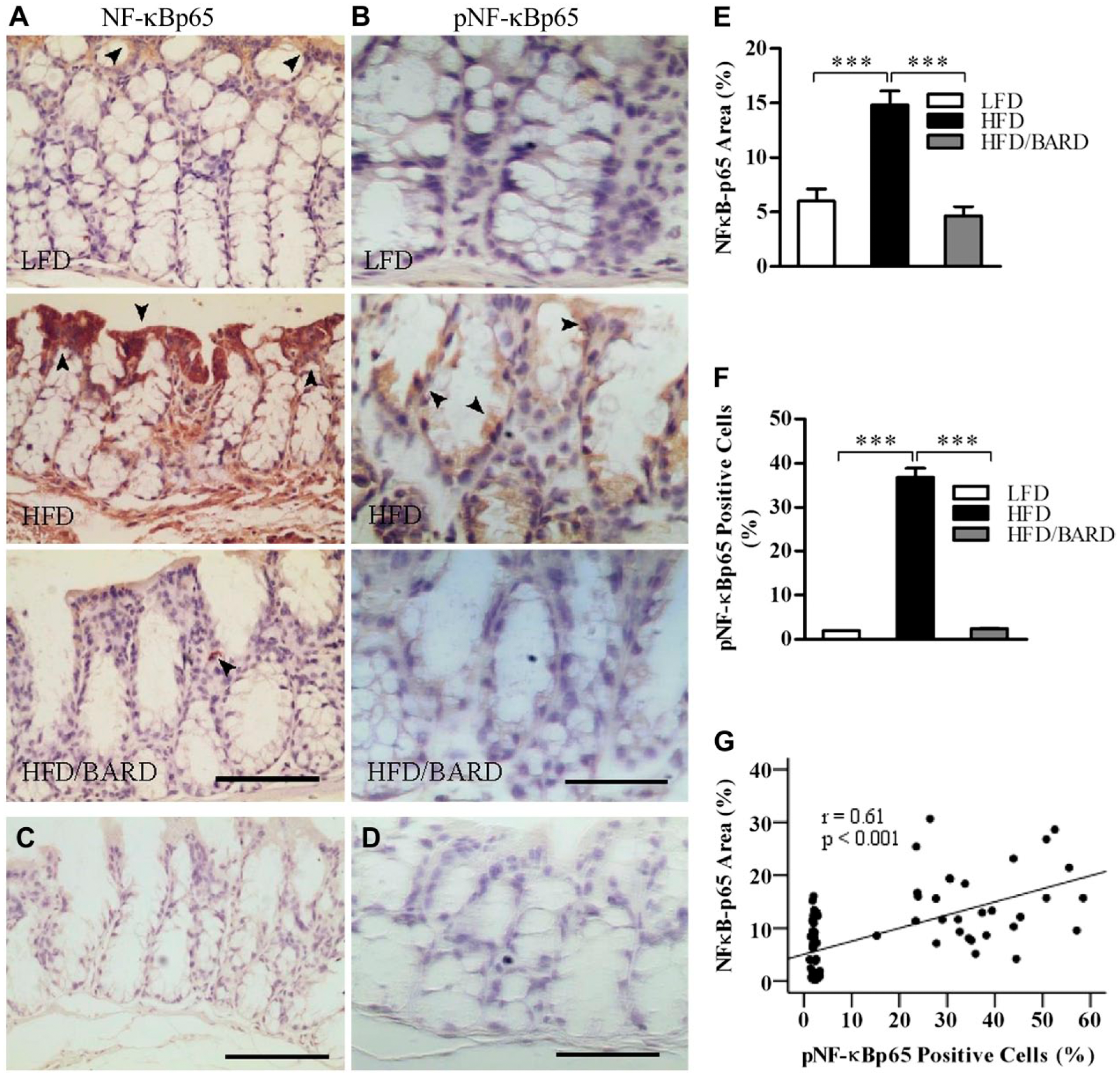
Effect of bardoxolone methyl (BARD) on the expression of NF-қB in the colon crypts of mice fed a low-fat diet (LFD), a high-fat diet (HFD), or high-fat diet supplemented with BARD (HFD/BARD). (A) NF-қBp65-stained sections (×20). (B) Phosphorylated NF-қBp65-stained sections (×40). (C) Negative NF-қBp65-stained section (×20). (D) Negative phosphorylated NF-қBp65-stained section (×40). The arrowheads indicate NF-қBp65-positive areas and phosphorylated NF-қBp65 positive cells (brown). (E) NF-қBp65-positive area (%). (F) Phosphorylated NF-қBp65-positive cells (%). (G) Correlation between NF-қBp65 and phosphorylated NF-Bp65. Six fields per sample (n=7) were used for statistical analysis. All data are presented as mean ± SEM ***p<0.001. NF-қBp65: p65 nuclear factor-κ light-chain enhancer of activated B cells; pNF-қBp65: phosphorylated NF-қBp65. Scale (A, C) 100 µm; (B, D) 50 µm.
We further investigated the effect of BARD on the expression of pro-inflammatory cytokines within the colon crypts (Fig. 4A–4D). In LFD mice, percent of positive areas of pro-inflammatory cytokines were 1.2%, 0.9% and 2.9% for IL-6, TNF-α, IL-1β, respectively (Fig. 4E). In contrast, HFD had higher percentage of positive areas of IL-6 (4.5 %), TNF-α (3%), and IL-1β (8.8%). HFD mice administered BARD had a reduced percent of pro-inflammatory cytokine-positive areas, by 2.3% for IL-6, 1.4% for TNF-α, and 1.8% for IL-1β. The reduction in pro-inflammatory cytokine-positive staining in response to BARD was also correlated with decreased levels of total F4/80 and CD11c macrophages (Table 2).
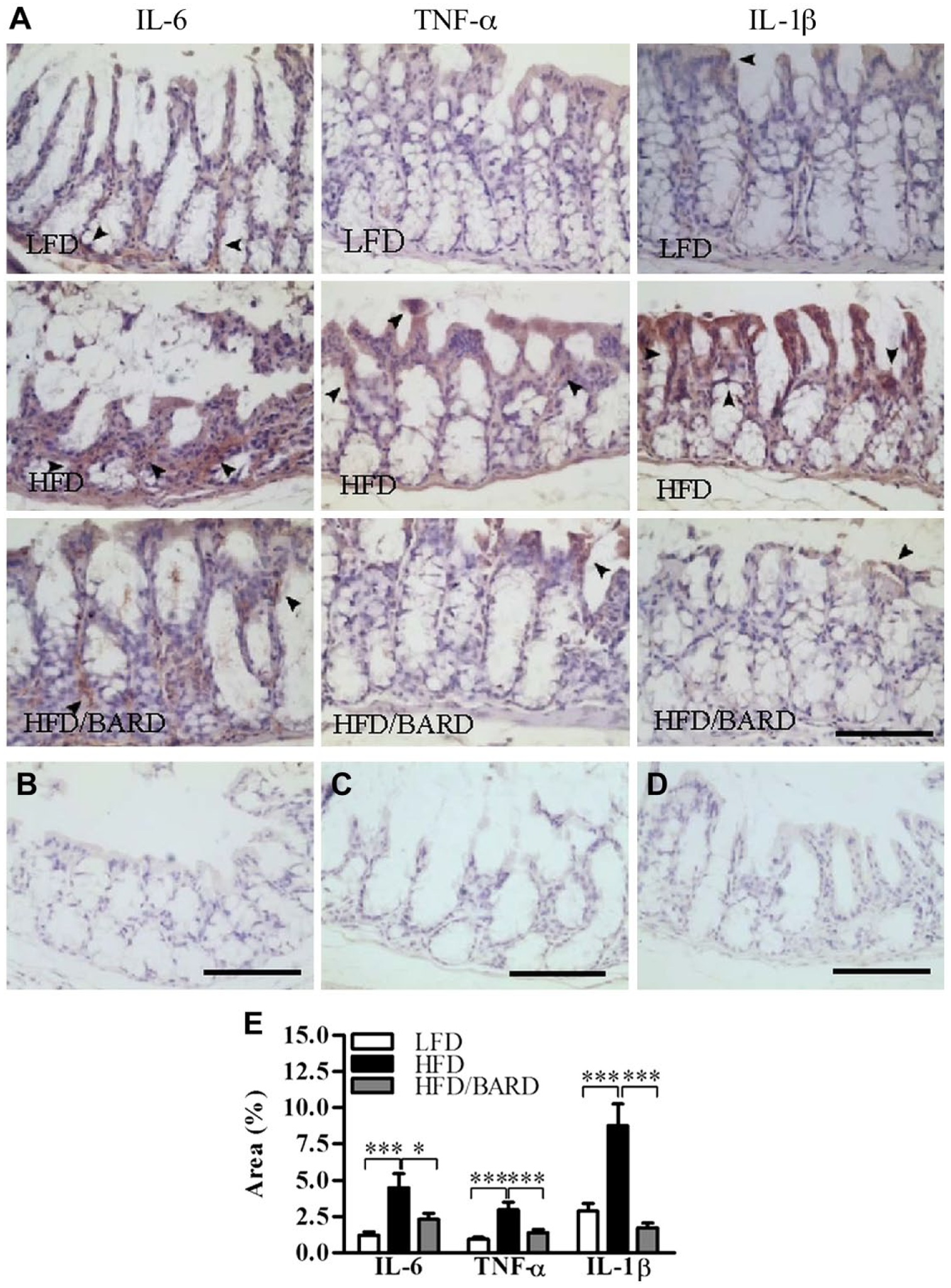
Effect of bardoxolone methyl (BARD) on the expression of pro-inflammatory cytokines in colon crypts of mice fed a low-fat diet (LFD), a high-fat diet (HFD), or a high-fat diet supplemented with BARD (HFD/BARD). (A) IL-6-, TNF-α-, and IL-1β-stained sections (×20). (B) Negative IL-6-stained section (×20). (C) Negative TNF-α-stained section (×20). (D) Negative IL-1β-stained section (×20). The arrowheads indicate IL-6-, TNF-α-, and IL-1β-positive areas (brown). (E) IL-6-, TNF-α-, and IL-1β-positive area (%). Six fields per sample (n=7) were used for statistical analysis. All data are presented as mean ± SEM. *p<0.05, ***p<0.001. IL-6: interleukin 6; IL-1β: interleukin 1 beta; TNF-α: tumor necrosis factor-alpha. Scale, 100 µm.
Correlation between Macrophages and Cytokines.
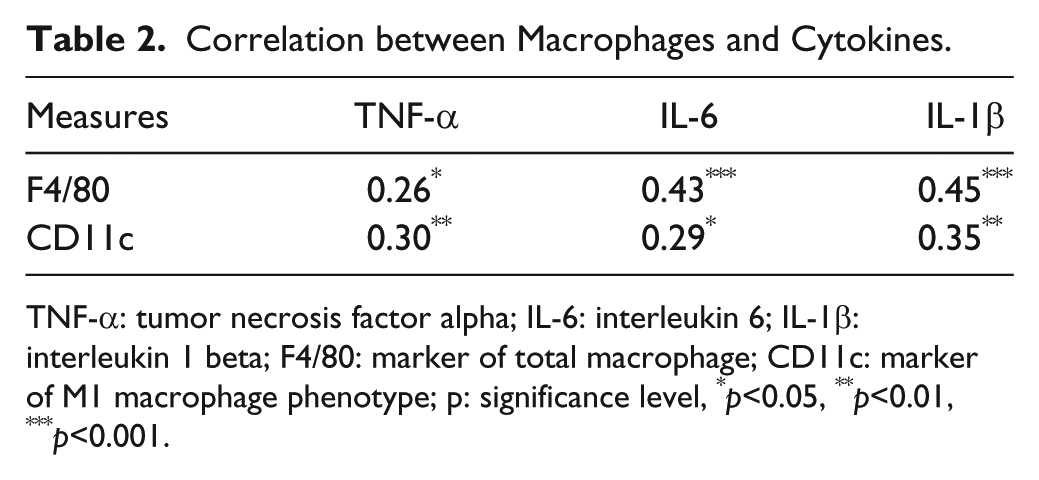
TNF-α: tumor necrosis factor alpha; IL-6: interleukin 6; IL-1β: interleukin 1 beta; F4/80: marker of total macrophage; CD11c: marker of M1 macrophage phenotype; p: significance level, *p<0.05, **p<0.01, ***p<0.001.
BARD Suppresses Cox2 Protein Expression in the Colons of HFD-fed Mice
We investigated the effect of BARD on the expression of Cox2 protein in the colon crypts, which is an inflammatory and proposed cancer marker in gut epithelial cells (Fig. 5A and 5B). Compared with mice fed a LFD, those fed a HFD were positive for Cox2 immunostaining (0.3% positive area; Fig. 5C). Similar to LFD mice, HFD/BARD mice were almost negative for Cox2 staining. The reduced level of Cox2 protein in HFD/BARD mice also correlated with the reduced levels of total and phosphorylated NF-қBp65 (Fig. 5D and 5E, respectively).
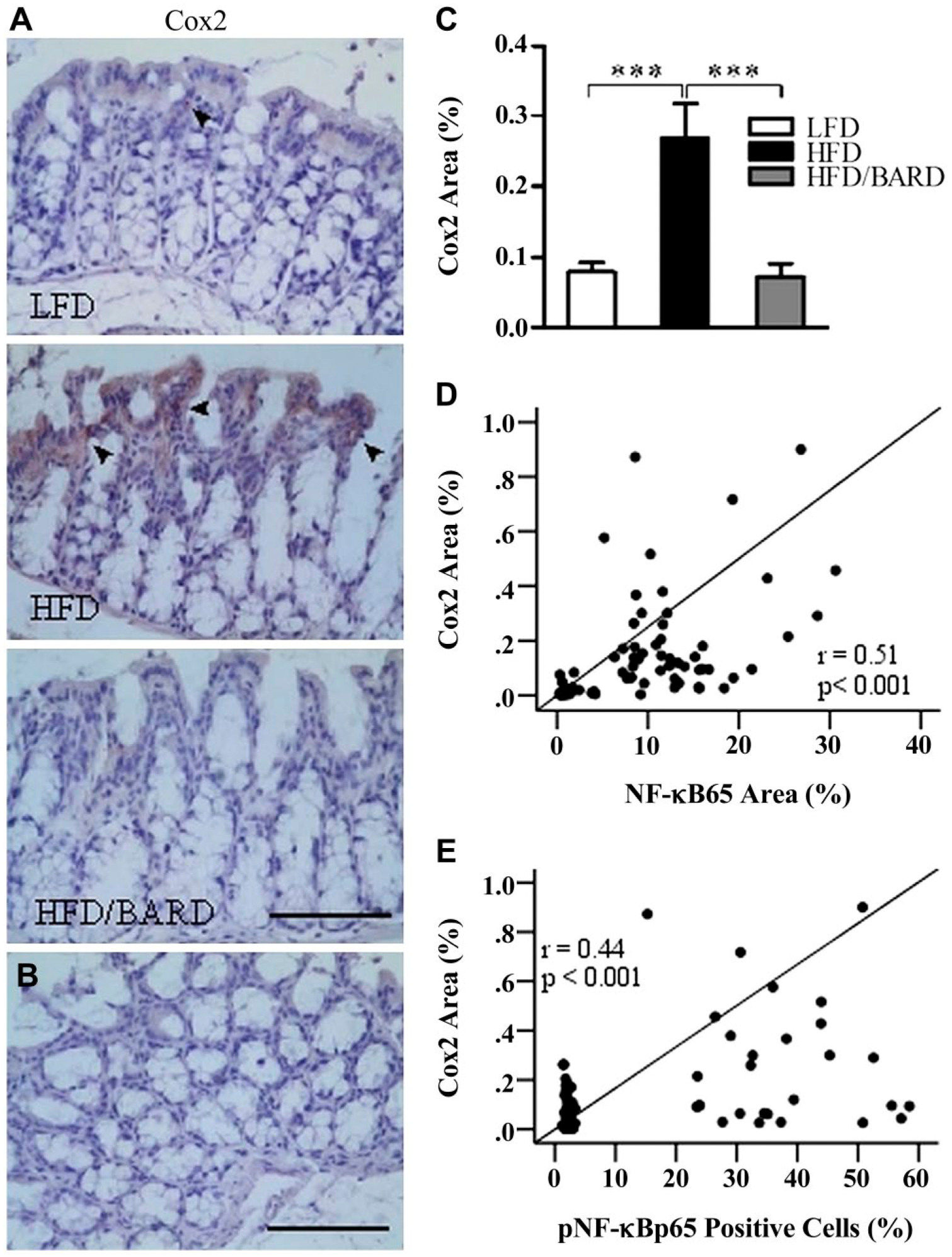
Effect of bardoxolone methyl (BARD) on the expression of Cox2 in the colon crypts of mice fed a low-fat diet (LFD), a high-fat diet (HFD), and a high-fat diet supplemented with BARD (HFD/BARD). (A) Cox2-stained sections (×20). (B) Negative Cox2-stained section (×20). The arrowheads indicate Cox2-positively stained areas (brown). (C) Cox2-positive area (%). (D) Correlation between Cox2 and NF-қBp65. (E) Correlation between Cox2 and pNF-қBp65. Six fields per sample (n=7) were used for statistical analysis. All data are presented as mean ± SEM. ***p<0.001. Cox2: cyclooxygenase 2; NF-қBp65: p65 nuclear factor-κ light-chain enhancer of activated B cells; pNF-қBp65: phosphorylated NF-қBp65. Scale, 100 µm.
BARD Suppresses the Ki67 Index in the Colons of HFD-fed Mice
Figure 6A shows a significant change in the expression of Ki67 immunoactivity in the colon epithelial cells among the three groups of mice (Fig. 6A and 6B). In Fig. 6C, colons of HFD mice had 19.6% Ki67-positive cells as compared with 5.9% for LFD and 2.5% in HFD/BARD mice (p<0.001). The percent of Ki67-positive cells in HFD/BARD mice was comparable with that of LFD mice, suggesting that BARD has an anti-proliferative effect. There was a moderate correlation between Ki67 expression and the levels of total and phosphorylated NF-қB (Fig. 6D and 6E, respectively) and pro-inflammatory cytokines TNF-α, IL-6, and IL-1β (Fig. 6F, 6G and 6H, respectively).
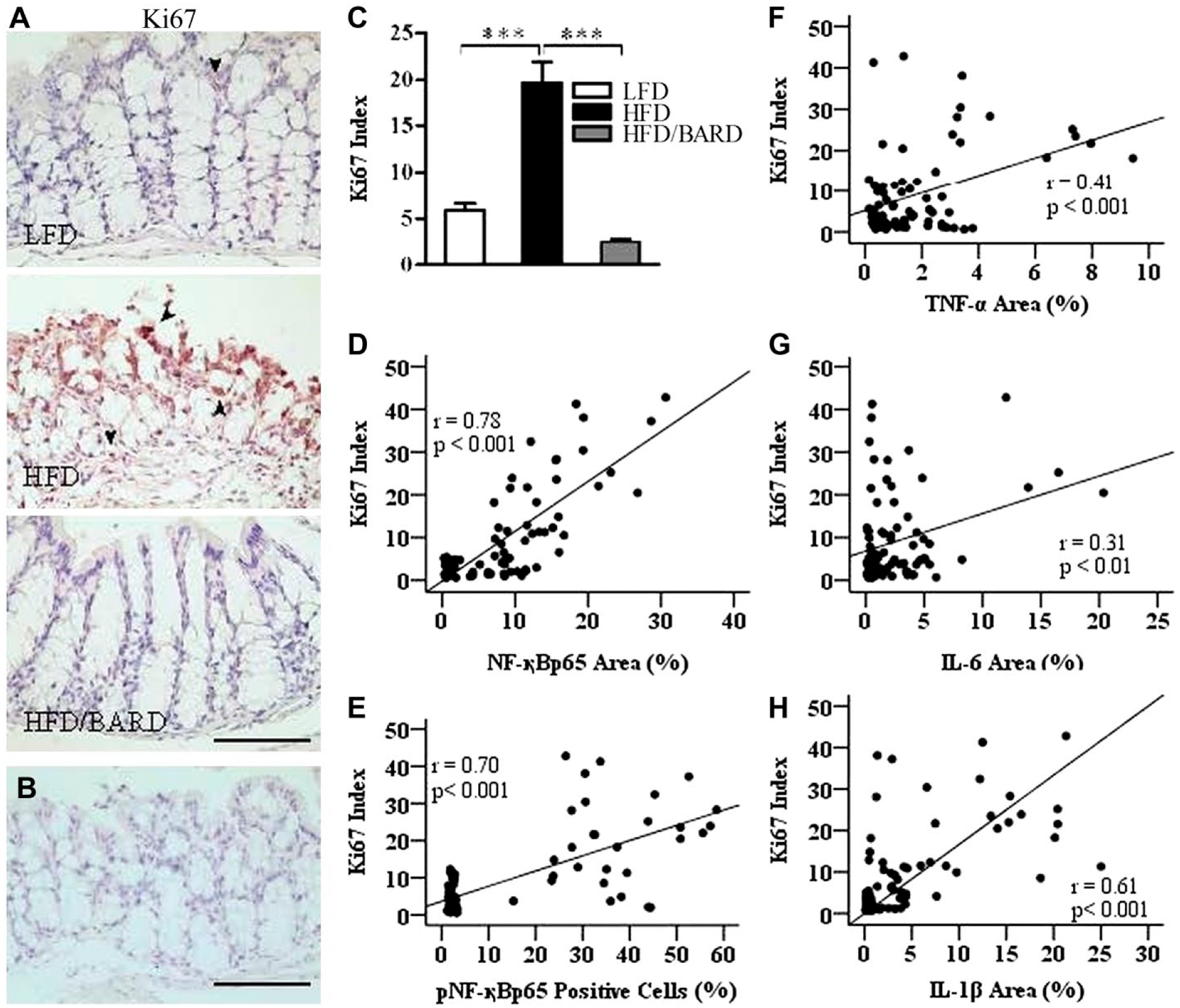
Effect of bardoxolone methyl (BARD) on the expression of Ki67 in colon epithelial cells in mice fed a low-fat diet (LFD), a high-fat diet (HFD), or a high-fat diet supplemented with BARD (HFD/BARD). (A) Ki67-stained sections (×20). (B) Negative Ki67-stained section (×20). The arrowheads indicate brown nuclear staining for Ki67. (C) Ki67-positive cells (%). (D) Correlation between Ki67 and total NF-қBp65. (E) Correlation between Ki67 and pNF-қBp65. (F) Correlation between Ki67 and TNF-α. (G) Correlation between Ki67 and IL-6. (H) Correlation between Ki67 and IL-1β. Six fields per sample (n=7) were used for statistical analysis. All data are presented as mean ± SEM. ***p<0.001. NF-қBp65: p65 nuclear factor-κ light-chain enhancer of activated B cells; pNF-қBp65: phosphorylated NF-қBp65; TNF-α: tumor necrosis factor alpha; IL-6: interleukin 6; IL-1β: interleukin 1 beta. Scale, 100 µm.
BARD Increases M2 Macrophage and Anti-inflammatory Cytokines in the Colons of HFD-fed Mice
We further investigated the effects of BARD on the expression of anti-inflammatory macrophages (M2 macrophage phenotype) and anti-inflammatory cytokines by assessing the level of epithelial CD206-positive cells and the IL-10-positive area in colon crypts, respectively (Fig. 7A–7C). The percentage of CD206-positive cells in HFD mice were 3.6%, which is half that of the LFD mice (7.5%) (p<0.001) (Fig. 7D). However, the percentage of CD206-positive cells was higher in HFD/BARD mice (6.7%) as compared with HFD mice (p<0.01). In Fig. 7E, the percentage of IL-10-positive area in HFD mice was 0.8%, which is lower than that in LFD mice (7.3%) and in HFD/BARD (7.5%) (p<0.001). The increase in M2 macrophages and IL-10 proteins in the colon crypts suggests a potent anti-inflammatory effect of BARD in HFD-fed mice.
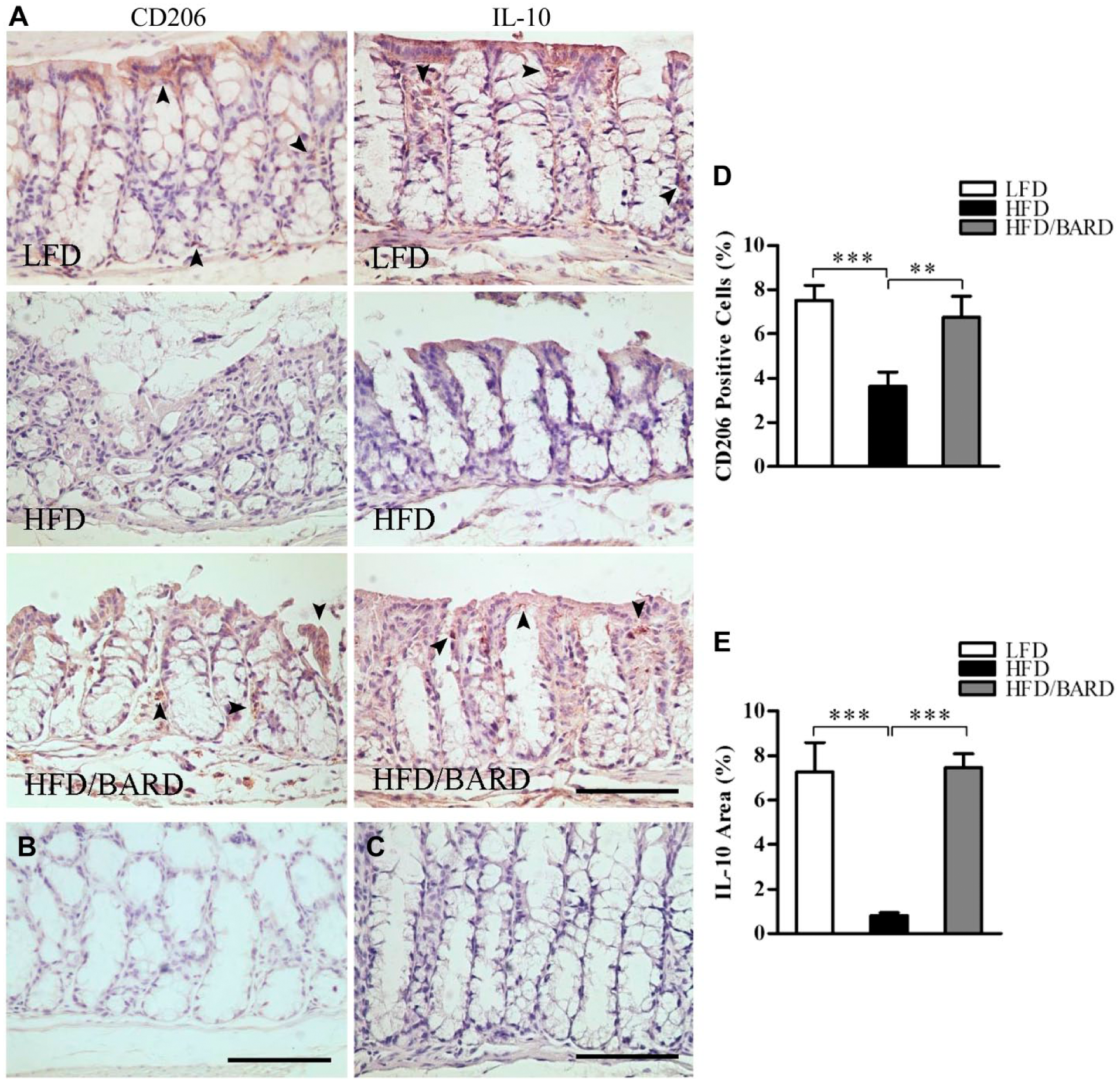
Effect of bardoxolone methyl (BARD) on the expression of M2 macrophages and anti-inflammatory cytokine IL-10 in colon crypts mice fed a low-fat diet (LFD), a high-fat diet (HFD), and a high-fat diet supplemented with BARD (HFD/BARD). (A) CD206- and IL-10-stained sections (×20). (B) Negative CD206-stained section (×20). (C) Negative IL-10-stained section (×20). The arrowheads indicate CD206-positive cells and IL-10-positive areas (brown). (D) CD206-positive cells (%). (E) IL-10-positive area (%). Six fields per sample (n=7) were used for statistical analysis. All data are presented as mean ± SEM. **p<0.01, ***p<0.001. IL-10: interleukin 10. Scale, 100 µm.
BARD Prevents Fat Deposition in the Colon of HFD-fed Mice
Figure 8A–8C shows oil-red O staining of fat in the colons of HFD-fed mice. HFD mice had 0.8% oil-red O-positive areas in the colon wall and crypts, which is twice as high as that of LFD mice (p<0.05; Fig. 8D). Compared with HFD mice, HFD/BARD mice had reduced oil-red O-positive staining (p<0.01), similar to LFD mice. Table 3 shows that BARD reduced the amount of fat deposition in the colons of HFD-fed mice, which correlated with the reduction of F4/80 macrophages (p=0.0001), pNF-қBp65 (p=0.0165), TNF-α (p=0.0337). Reduced fat deposition by BARD also correlated with the increase in the anti-inflammatory marker IL-10 (p=0.0226). These data suggest that HFD-induced fat deposition may contribute to inflammatory progression in the mouse colon and that BARD can prevent such effects.
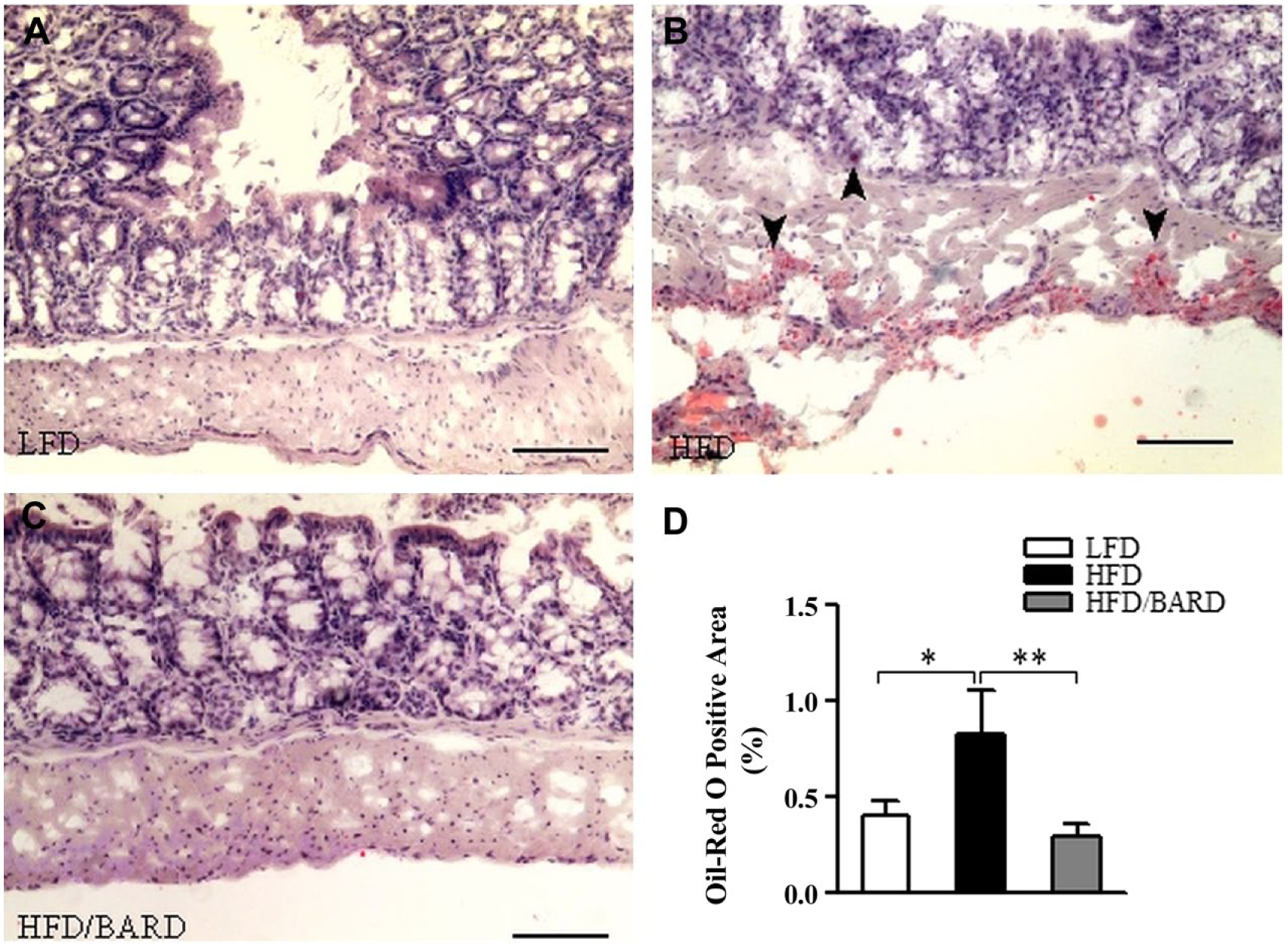
Effect of bardoxolone methyl (BARD) on fat deposition in the colon mucosa and wall of mice fed a low-fat diet (LFD), a high-fat diet (HFD), or a high-fat diet supplemented with BARD (HFD/BARD). (A–C) Oil-red O-stained sections (×10). (D) Oil-red O-positive areas (%). Six fields per sample (n=7) were used for statistical analysis. All data are presented as mean ± SEM. *p<0.05, **p<0.01. Scale, 100 µm.
Correlation between Adiposity and Inflammatory Modulators.
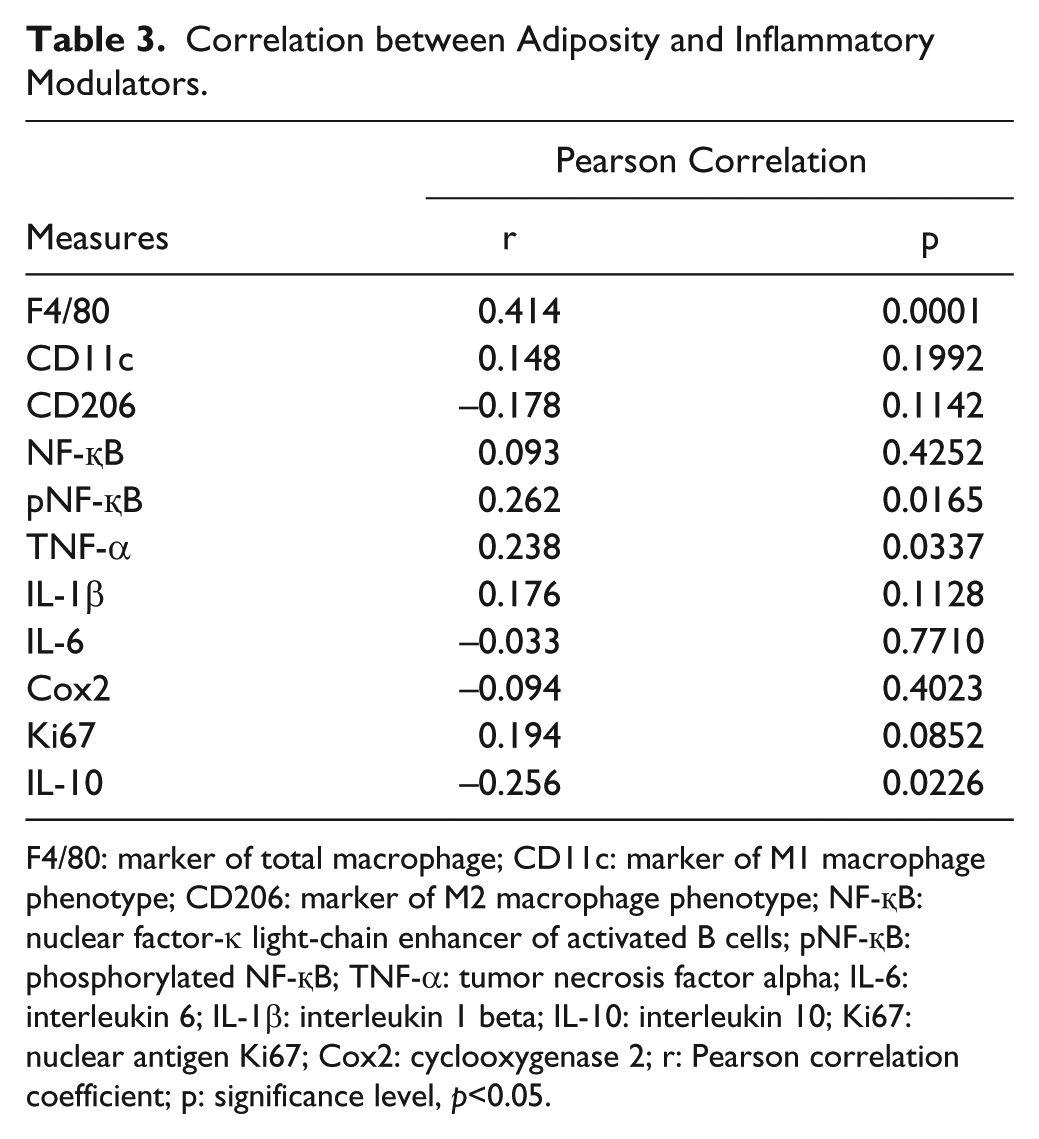
F4/80: marker of total macrophage; CD11c: marker of M1 macrophage phenotype; CD206: marker of M2 macrophage phenotype; NF-қB: nuclear factor-κ light-chain enhancer of activated B cells; pNF-қB: phosphorylated NF-қB; TNF-α: tumor necrosis factor alpha; IL-6: interleukin 6; IL-1β: interleukin 1 beta; IL-10: interleukin 10; Ki67: nuclear antigen Ki67; Cox2: cyclooxygenase 2; r: Pearson correlation coefficient; p: significance level, p<0.05.
BARD Prevents the Alteration of Gut Microbiota in HFD Mice
To investigate the effect of BARD on gut microbiota, relative DNA abundance of Gram-positive bacteria Bifidobacterium sp., Lactobacillus spp., and Gram-negative bacteria Bacteroides-Prevotella spp. was determined by real-time PCR. The relative DNA abundance of Bacteroides-Prevotella spp. was significantly reduced in HFD-fed mice, which was half that of LFD mice (Fig. 9A; p<0.001). BARD significantly increased the relative DNA abundance of Bacteroides-Prevotella spp., which was 3-fold higher than that in HFD mice (p<0.001). The relative abundance of Bidifobacterium spp. in HFD/BARD mice was similar to the DNA abundance in LFD mice (Fig. 9B; p<0.001), and significantly upregulated compared to mice on HFD (p<0.001). The HFD also increased the relative DNA abundance of Lactobacillus spp., which is 8-fold higher than that in LFD mice (Fig. 9C; p<0.001). HFD/BARD mice had reduced DNA level of Lactobacillus spp. compared with HFD mice (p<0.001). In Table 4, the reduction in the populations of Bidifobacterium spp. and Lactobacillus spp. in response to BARD supplementation correlated with the decrease in inflammatory biomarkers (NF-қBp65, pNF-қBp65, TNF-α, IL-1β, IL-6), Cox2, and Ki67 and the increase in the anti-inflammatory cytokine IL-10. Conversely, BARD induced an increase in the Gram negative Bacteroides-Prevotella spp. population, and this correlated with the increase in IL-10 and the decrease in inflammatory biomarkers. These data therefore demonstrate that HFD disturbs the gut microbiota population, which can be prevented by BARD.
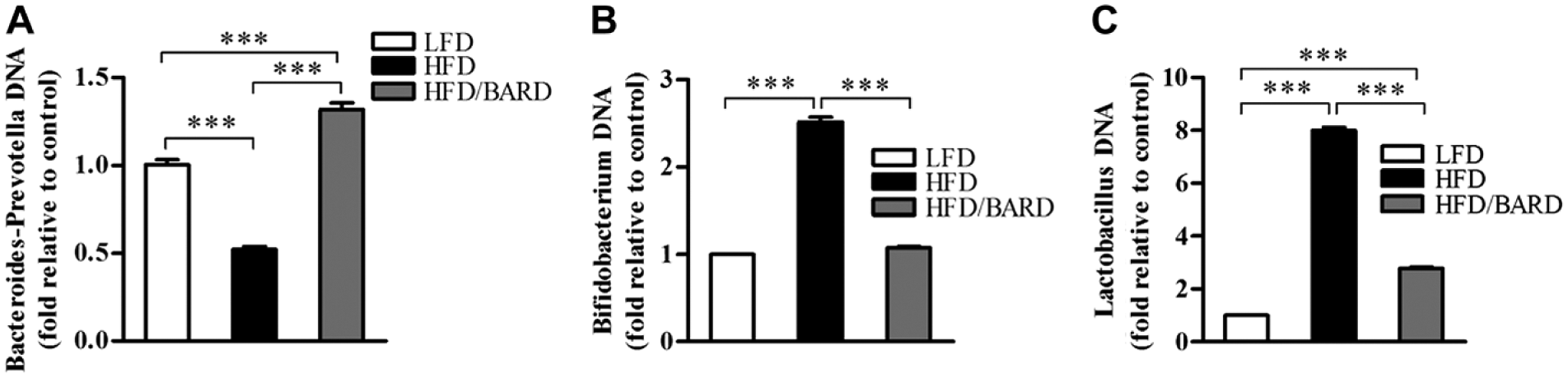
Effect of bardoxolone methyl (BARD) on relative DNA abundance of fecal bacteria in mice fed a low-fat diet (LFD), a high-fat diet (HFD), or a high-fat diet supplemented with BARD (HFD/BARD). (A) Relative DNA abundance of Bacteroides-Prevotella spp. (B) Relative DNA abundance of Bifidobacterium spp. (C) Relative DNA abundance of Lactobacillus spp. All data are presented as mean ± SEM (n=7). ***p<0.001.
Correlation between Fecal Microbiota and Inflammatory Biomarkers in Mouse Colons.
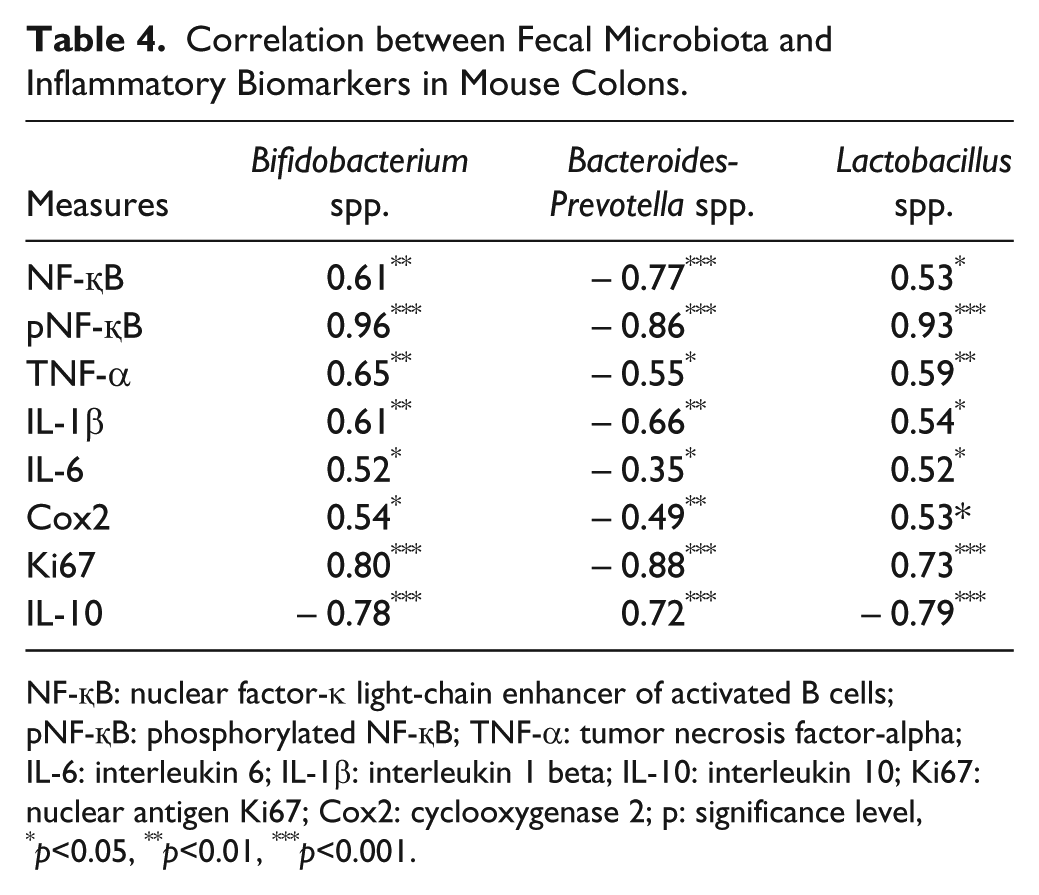
NF-қB: nuclear factor-κ light-chain enhancer of activated B cells; pNF-қB: phosphorylated NF-қB; TNF-α: tumor necrosis factor-alpha; IL-6: interleukin 6; IL-1β: interleukin 1 beta; IL-10: interleukin 10; Ki67: nuclear antigen Ki67; Cox2: cyclooxygenase 2; p: significance level, *p<0.05, **p<0.01, ***p<0.001.
Discussion
BARD inhibits colon cancer cell growth by inducing apoptosis, which is associated with NF-қB suppression (Gao et al. 2010). Furthermore, a mouse study has suggested that BARD can treat colitis-associated colon cancer as it reduces colon tumors and inflammation-driven colon carcinogenesis (TNF-α, IL-6, IL-1β, Cox2) (Choi et al. 2014). The present study investigated the anti-inflammation and anti-cell proliferation effects of BARD in the colons of mice fed a HFD. Fat deposition and microbial population changes were also determined along with these changes in the mouse colon.
In the present study, chronic HFD (for 21 weeks) feeding in mice led to a shortened colon, associated with a reduced length of colon crypts and a reduced number of goblet cells per crypt, which could be reversed by BARD. Consistent with our result, mice administered HFD for 35 weeks have shortened colon and crypt lengths, which is associated with the upregulation of inflammation and cancer-associated genes (Deol et al. 2015). However, mice fed a HFD for 10 and 15 weeks have been reported to have elongated colon crypts as compared with the LFD group (Bird and Stamp 1986). Also, mice on an acute HFD for 30 days have normal colon crypt length, similar to that of control mice (Basson et al. 2004). It appears that a HFD causes changes in mouse colon crypt length depending on the duration of exposure to a HFD. Moreover, the loss of goblet cells in the colon has been implicated in tissue injury in a colitic mouse model (Xiao et al. 2007). HFD induces goblet cell elimination, crypt destruction, and the infiltration of inflammatory cells in the colons of chemical-induced colitic rats (Morampudi et al. 2014). In the present study, the normalisation of colon crypts by BARD indicates its effectiveness in the prevention of HFD-induced changes in colon morphology and inflammation. The effects of BARD on colon crypts in our study are consistent with a previous study showing that BARD increases colon length and reduces colon thickness in colitis-associated mice (Choi et al. 2014).
Macrophage infiltration is a common characteristic of inflammation in the colons of diet-induced obese mice (Erdelyi et al. 2009; Lam et al. 2012; Liu et al. 2012). We found an increased level of F4/80 and CD11c immunohistochemical staining in the colons of HFD-fed mice, which could be suppressed by BARD. These findings in the colon coincide with observations in white and brown adipose tissue of mice fed a HFD or a HFD plus BARD (Dinh et al. 2015a; Dinh et al. 2015b). The modulating effects of BARD on infiltrating macrophages are consistent with the immunomodulation function of most triterpenes (Honda et al. 1997; Gao et al. 2008; Ikeda et al. 2008; Kulkarni et al. 2013). Colitic mice fed a HFD develop severe inflammation in the colon accompanied by an exaggerated level of infiltrating macrophages (Teixeira et al. 2011). F4/80 and CD11c macrophages are found in the colon, and an increased level of these cells is often seen in inflammatory colons and in carcinogenesis (Takada et al. 2010; Rivollier et al. 2012; Hume and Gordon In Press). These results suggest a potential role of BARD in preventing tumorigenesis and cancer.
In inflamed intestinal mucosa, activated NF-қB is highly expressed in macrophages and epithelial cells (Rogler et al. 1998). As well as reducing macrophage invasion, BARD also reduces total and phosphorylated NF-қB proteins. NF-қB is an important target of anti-cancer drugs, and an increasing number of studies have demonstrated BARD’s effect in reducing NF-қB activation (Pergola et al. 2011). Similar effects have been observed with ursolic acid triterpene, which inhibits the NF-қB activation in human intestinal epithelial cells and peritoneal macrophages in IL-10-deficient mice (Chun et al. 2014). This is consistent with known inhibitory effects of most pentacyclic triterpenes (Takada and Aggarwal 2003; Liby and Sporn 2012; Hwang et al. 2014). From this data, we have shown that NF-қB plays an essential role in HFD-induced colon inflammation and we suggest that BARD is an effective chemo-preventive agent.
The increased activation of NF-қB in the colon of HFD-fed mice was associated with an elevated level of pro-inflammatory cytokines (Kim et al. 2012a). Studies have shown that BARD induces anti-inflammatory effects via modulating the pro-inflammatory response (Auletta et al. 2010), as found in the present study. A high level of TNF-α enhances the development of inflammatory bowel disease and colon cancer in mice and rats (Sands and Kaplan 2007; Jain and Bird 2010; Flores et al. 2012). The inhibition of this cytokine is important for preventing intestinal inflammation (Kojouharoff et al. 1997; Popivanova et al. 2008). TNF-α, IL-6, and IL-1β have been identified as a cluster of inflammatory markers that increase in colon cancer (Ong et al. 2010; Klampfer 2011). The suppressive effect of BARD on these pro-inflammatory cytokines is consistent with most pentacyclic triterpenoids; for instance, supplementation with ursanic, oleanic, and lupanic triterpenoids in T84 colon carcinoma cells strongly suppresses the inflammatory genes associated with inflammatory bowel diseases (Mueller et al. 2013). A reduction in the level of pro-inflammatory cytokines is correlated with a reduction in the number of infiltrating macrophages. This indicates that BARD’s suppression of pro-inflammatory cytokines may occur partly through modulation of immune cells. This is consistent with betulinic acid triterpene, which reduces TNF-α expression in mice (Oliveira Costa et al. 2014).
Inflammatory cells have an elevated level of Cox2 (Masferrer et al. 1994). We observed a reduction in Cox2 protein expression in HFD mice also given BARD. In one study on an LPS-exposed macrophage cell line, oleanolic acid and ursolic acid derivatives were shown to suppress Cox2 protein and mRNA expression (Suh et al. 1998). Similarly, a synthetic oleanolic acid analog, 18alpha-olean-12-ene-3beta-23,28-triol, was previously shown to reduce Cox2 expression in human colon HT-29 cancer cells (Janakiram et al. 2008). A BARD analog, RTA dh404, reduced renal Cox2 expression, and reduced phosphorylated NF-қBp65 in rats with chronic kidney disease (Aminzadeh et al. 2014). Similar to RTA dh404, we found that a reduction in Cox2 protein is correlated with a reduction in total and phosphorylated NF-қBp65. Previously, BARD has been found to suppress NF-қB leading to a reduction in Cox2 in human leukemia cell lines (Shishodia et al. 2006). Another study has shown that the combined treatment of triterpenoid α and β-amyrin reduces the immunohistochemical staining level of Cox2 and phosphorylated NF-қBp65 in the colons of colitic mice (Vitor et al. 2009). Similar effects have been described for other triterpenes including maslinic acid and betulinic acid (Takada and Aggarwal 2003; Hsum et al. 2011). Cox2 inhibitors (such as celecoxib) have been used for the primary prevention of colon tumors and suppression of adipose inflammation in HFD-induced obese rats (Sheng et al. 1997; Half and Arber 2009; Hsieh et al. 2010). These results thus demonstrate the anti-inflammatory effect of BARD on Cox2 suppression and NF-қB regulation.
A HFD increases Ki67 in various tissues, including the prostate gland and the small intestine in mice (Petit et al. 2007; De Wit et al. 2008; Benesh et al. 2013). We found that a HFD increases Ki67 (a cell proliferation marker) in the epithelial cells of colon crypts in mice, and that BARD can prevent this HFD-induced increase in Ki67. A similar effect has been observed in azoxymethane-induced colon cancer mice, as shown by a reduced number of tumors and Ki67 expression (Choi et al. 2014). Increased Ki67 expression has been consistently observed in colon tumors in humans and mice (Evans et al. 2006; Vinuesa et al. 2012). Epithelial Ki67 as a biomarker has been used to investigate some cancers and colorectal polyps (Deckert et al. 1989; Aubert et al. 2002; Aslan et al. 2005; Nabi et al. 2008). The upregulation of Ki67-positive proliferating cells reflects a failure of epithelial cells to repress DNA synthesis during their migration to the crypt surface, which leads to adenoma formation and tumorigenesis (Deschner and Lipkin 1975; Shih et al. 2001). A cell culture study on human colon crypts has shown that Ki67 in normal tissue is expressed in the basal crypt, which is undetectable after 3–5 days of the crypt cycle; however, Ki67-positive cells with budding structures are continuously and extensively expressed in adenomatous crypts (Dame et al. 2014). The increased Ki67 index in the shortened crypts in the present study likely reflects the HFD-associated promotion of an adenomatous environment in the mouse colon. These results are consistent with a previous study showing the protective role of BARD in colonic epithelial cells (Kim et al. 2012b). Overall, increased Ki67, NF-қB, and pro-inflammatory cytokines in the present study suggest a risk of tumorigenesis in HFD-induced obesity. Furthermore, the reduced Ki67 index and the lower level of inflammatory modulators by BARD, which is associated with an improvement in the colon crypts to resemble that of normal, LFD controls, suggests anti-proliferative effects and regulation of colon epithelial homeostasis, inflammation and tumorigenesis by BARD.
M2 macrophages in colon lamina propria are known to release the anti-inflammatory cytokine IL-10 in response to gut stimuli such as microbiota (Rivollier et al. 2012). CD206 is a biomarker of M2. This study shows that a HFD reduces CD206 and IL-10 expression, which can be reversed by BARD. It is reported that a high level of IL-10 and CD206 causes resistance to inflammatory bowel disease (Bain et al. 2013). M2 macrophages reduce colitis through IL-10 expression in mice (Zhu et al. 2014). Several studies have demonstrated the anti-inflammatory effects of pentacyclic triterpenes through the induction of IL-10 (Chen et al. 2012; Martín et al. 2014). Ursolic acid, α- and β-amyrin increase the expression of IL-10 protein in mouse colons (Vitor et al. 2009; Jang et al. 2014). Our data are also consistent with the known anti-cancer effects of BARD (Gao et al. 2010; Hong et al. 2012; Choi et al. 2014).
We investigated the effect of oral BARD on lipid deposition in the colons of HFD-fed mice. HFD-induced fat accumulation in the colon wall and mucosa were reduced by BARD. We have previously observed the reducing effects of oral BARD on liver, white and brown adipose tissue in HFD-fed mice (Camer et al. 2015a; Dinh et al. 2015a; Dinh et al. 2015b; Dinh et al. 2015c). An increased lipid accumulation has been observed in the intestinal mucosal of HFD-induced and genetically-induced obese mice, as assessed by increased oil-red O staining of neutral fat and excess triacylglycerol (Douglass et al. 2012). The present data therefore suggests the potential of BARD in preventing HFD-induced fat deposition in the intestine.
A previous study has implicated an overflow of dietary fat to the distal intestine as a causative factor for microbiota alterations in the gut, contributing to obesity and metabolic syndrome (de Wit et al. 2012). We found that BARD prevents the alteration of microbiota in HFD mice, with an increase in Bacteroides-Prevotella spp. and a reduction in Bidifobacterium spp. and Lactobacillus spp. The presence of Bacteroides-Prevotella spp. protects humans from inflammation, and induces obesity resistance (De Filippo et al. 2010). A calorie-restricted diet prevented the growth of both Bidifobacterium spp. and Lactobacillus spp., species whose presence tends to that accompany excessive weight loss in overweight adolescents (Santacruz et al. 2009). BARD has been identified as a suppressor of body weight, with a significant weight loss effect in obese people and in patients with chronic kidney disease (Manenti et al. 2012). Additionally, we have previously reported the preventive effect of BARD in HFD-induced body weight in mice (Dinh et al. 2015a; Dinh et al. 2015b). Therefore, the effect of BARD treatment in preventing an alteration to gut microbiota induced by a HFD in obese mice may contribute to BARD’s ability to prevent HFD diet-induced obesity.
In conclusion, this study shows that mice on a chronic HFD have colon inflammation, as assessed by the upregulation of inflammatory biomarkers in the colon epithelium. This was shown by an increase in M1 macrophage invasion and an elevated level of NF-қB, pro-inflammatory cytokines, Cox2 and Ki67. Suppressing these inflammatory markers is important for the prevention of colon inflammation. BARD decreased the expression of pro-inflammatory cytokines, Cox2 and Ki67 while increasing the expression of the anti-inflammatory marker IL-10. This may relate to the anti-cancer effects observed in an existing study (Choi et al. 2014). Moreover, BARD prevents fat deposition in the colon wall of HFD mice, and this is accompanied by a normalisation of colon microbiota. The outcome of the present study suggests a need for further investigations using BARD to prevent inflammatory disease and obesity-associated colon inflammation and colon cancer. This study suggests a future application of BARD in the prevention of obesity-associated colon disorder in humans. However, the use of BARD should be prohibited in patients with advanced chronic kidney disease and those susceptible to cardiovascular disease to avoid possible heart failure-related side effects, as observed in previous clinical trials (Chin et al. 2014). Further studies on the molecular mechanisms of chronic kidney diseases and cardiovascular disease in animal models will also help to understand the potential side effects of BARD, which may assist in the future applications of BARD in human patients.
Footnotes
Acknowledgements
We would like to thank Ms Linda Cohen for her editorial revision of this manuscript.
Author Contributions
CD carried out the experiments, data analysis and statistical analysis and wrote the manuscript. YY, AS and XH were responsible for the experiment design and helped interpret the results. QZ has contributed to the discussion section and helped interpret the results. PZ contributed to microbial quantification. All authors have contributed to and approved the final manuscript.
Competing Interests
The authors declared no potential competing interests with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
