Abstract
Amyloids are pathological intra- and extracellular fibrillar aggregates of polypeptides with a cross-β-sheet structure and characteristic tinctorial properties. The amyloid deposits commonly enclose several non-fibrillar components of the extracellular matrix. Their potential to regulate the formation and aggregation process of amyloid fibrils is still poorly understood. For a better understanding of the role of the extracellular matrix in amyloidosis, it is essential to gain deeper insights into the composition of amyloid deposits. Here, we utilized matrix-assisted laser desorption and ionization mass spectrometry imaging to identify extracellular matrix compounds in amyloid deposits. Using this technique, we identified and determined the spatial distribution of vitronectin within AApoAI-, ALλ-, ATTR- and AIns amyloid deposits and, using immunohistochemistry, validated the spatial overlap of vitronectin with amyloids in 175 cases with diverse types of amyloid in several different tissues.
Keywords
Introduction
Amyloids are pathological intra- and extracellular fibrillar aggregates of polypeptides with a cross-β-sheet structure (Sachse et al. 2008; Sunde et al. 1997) and characteristic tinctorial properties (Howie and Brewer 2009; Howie et al. 2008; Puchtler et al. 1962), which disrupt tissue and organ function and result in a broad spectrum of different amyloid diseases. Currently, 31 different autologous proteins and peptides have been identified to form amyloid fibrils (Sipe et al. 2014). Aside from the amyloid fibril proteins, the deposits always enclose other components, such as amyloid-P-component, the highly sulfated glycosaminoglycans, as well as proteoglycans and various serum-derived proteins (Röcken and Eriksson 2009), which are specifically enriched in the deposits and may contribute to the pathology. In addition, many of these components are constituents of the extracellular matrix (ECM) (Merlini and Bellotti 2003). However, the role of the ECM in amyloidogenesis is still ill-defined, and the discovery of novel non-fibrillar constituents of amyloid deposits may shed further light on the complex etiology and pathology of the disease.
In this study, we used matrix-assisted laser desorption and ionization mass spectrometry imaging (MALDI-MSI). This is a novel technology that combines mass spectrometry with two-dimensional imaging and is able to provide both the m/z values of peptides/proteins in tissue sections and their spatial distribution. Unlike conventional mass spectrometric technologies (Klein et al. 2011; Nasr et al. 2013; Roden et al. 2010; Rodriguez et al. 2008; Sethi et al. 2010, 2012; Vrana et al. 2009), in MALDI-MSI, a tissue section is mounted onto a glass slide or metal plate, and not homogenized, thereby maintaining the spatial resolution and facilitating a correlation with histoanatomical structures and pathologies; e.g., amyloid. The tissue section is covered with a matrix solution using a robotic nebulizer or spotter device. During this process, the analytes are extracted and will co-crystallize with the matrix, thereby maintaining their local position. The matrix consists of an organic compound that is able to absorb at the wavelength of the applied laser, which is an essential step of the desorption/ionization mechanism. The acquisition is conducted at distinct positions according to a two-dimensional raster of x,y-coordinates equal to the number of pixels in the image. For each position, a spectrum containing the detected masses and their intensities is obtained. The data are imported into an imaging software to reconstruct mass-resolved images of the tissue section (for further details see (Chughtai and Heeren 2010; Jones et al. 2012)).
By applying MALDI-MSI to four different amyloid cases and a validation cohort of 175 formalin-fixed and paraffin-embedded tissue samples from a large variety of different amyloid diseases analyzed by immunohistochemistry (IHC), we identified vitronectin (VTN) as a common constituent of amyloid.
Materials & Methods
Patients
Amyloid-laden, formalin-fixed, paraffin-embedded (FFPE) tissue samples from 175 patients with different types of amyloid were obtained from the Amyloid Registry of the University of Kiel. The samples included tissues from brain (8 patients), nose (1), lip (1), salivary gland (1), lung (8), heart (27), tongue (3), stomach (12), small intestine (10), large intestine (20), liver (6), spleen (1), kidney (19), bladder (5), carpal tunnel ligament and synovialis (19), intervertebral disc (1), lymph node (2), skin (4), subcutaneous fat (12), soft tissue (5) and bone marrow (10). The study was conducted after the surgical pathological investigations had been completed.
Histology and Immunohistochemistry
Amyloid was detected by Congo red staining viewed under polarized light to show green birefringence and Congo red fluorescence. Immunohistochemical classification was carried out as described in detail elsewhere (Kebbel and Röcken 2006; Yumlu et al. 2009; Schönland et al. 2012; Gioeva et al. 2013) with commercially available monoclonal antibodies directed against AA amyloid, vitronectin (Merck Millipore; Darmstadt, Germany), insulin (BioGenese) and β2-microglobulin (β2M), and polyclonal antibodies directed against amyloid P-component, λ-light chain, κ-light chain, fibrinogen, and Aβ amyloid (all from DAKO; Hamburg, Germany). In addition, we used non-commercially available polyclonal antibodies directed against apolipoprotein AI (anti-apoAI), transthyretin (TTR3), λ-light chain (AL1, AL3 and AL7), and κ-light chain (AK3) (Kebbel and Röcken 2006; Schönland et al. 2012; Gioeva et al. 2013).
Immunostaining for vitronectin was evaluated first by documenting the presence of any vitronectin immunostaining within amyloid deposits, and subsequently by categorizing the percentage of the vitronectin-positive amyloid areas for each case as 0%, ≤10%, 11-90%, >90%.
Mass Spectrometry
Chemicals
Modified sequence-grade trypsin was purchased from Promega (Mannheim, Germany) and α-cyano-4-hydroxycinnamic acid (α-CHCA) from LaserBio Labs (Sophia-Antipolis Cedex, France). Double-distilled water (DDW) was obtained from Carl Roth (Karlsruhe, Germany) and xylene from BüFa (Lübeck, Germany). Acetonitril, ethanol were purchased from Merck (Darmstadt, Germany) and trifluoroacetic acid (TFA), ammonium bicarbonate, octyl-α/β-glucoside (OcGlc), red phosphorous and acetone from Sigma-Aldrich (Steinheim, Germany).
Biological Tissues
Five micrometer sections of FFPE human liver, heart (two different cases) and subcutaneous abdominal tissue containing AApoAI-, ALλ-, ATTR- and AIns amyloid, respectively, were prepared using a microtome (Leica Biosystems; Nussloch, Germany). The sections were mounted onto histological glass slides (SuperFrost® Plus, Menzel-Gläser) and dried at 54°C overnight. Tissue sections were stored at 4°C until further use.
Dewaxing and Rehydration
For dewaxing (removal of paraffin), FFPE tissue sections were immersed in xylene for 2 × 10 min, then rehydrated for 2 × 2 min in 99% ethanol, 70% ethanol, 50% ethanol, followed by 2 × 1 min in DDW. Tissue sections were dried at room temperature in a desiccator under a light vacuum of -400 mBar for at least 15 min before trypsin and matrix deposition.
On-Tissue Digestion
Trypsin solution (20 µg/ml) was prepared in 50 mM ammonium bicarbonate buffer (pH 8.1) containing 0.5% OcGlc. Deposition onto the tissue section was done using a SunCollect Micro Fraction Collector/MALDI Spotter (SunChrom, Friedrichsdorf, Germany) in spraying mode at a flow rate of 20 µl/min over 10 layers. After trypsin deposition, the section was incubated for 2 hr at 37°C in a humid environment using a homemade incubation chamber.
Matrix Deposition
Five milligram of α-CHCA was dissolved in 1 ml ACN:H2O (50:50) containing 0.1% TFA and sonicated for 5 min. The matrix solution was sprayed onto the tissue section using the SunCollect Micro Fraction Collector/MALDI Spotter. The deposition was performed with an increasing flow rate over the first three layers. The first layer was performed at 10 µl/min, the second at 20 µl/min, and the final eight layers at 30 µl/min.
MALDI-MS-Imaging
The instrument was calibrated before the MALDI-MSI experiment using signals of red phosphorous (oversaturated in acetone) between 100 and 3,000 m/z. Data were acquired in positive-ion mode (V-mode, sensitivity) using a MALDI SYNAPT G2-S system (Waters Corporation, Manchester, UK) operating with a 1 kHz Nd:YAG laser. To define the regions for acquisition, digital scans (Epson® Perfection 1640SU) of tissue sections were imported into the High Definition Imaging (HDI) software (Waters Corporation). All image data were acquired in the mass range from 700 to 3,000 m/z with a spatial resolution at 200 µm and 1,000 laser shots per position. Peptide images were generated for the 1,000 most intense signals with HDI software using a bin size of 0.1 Da for the mass range 900-3,000 m/z and an external lock mass ([Glu1]-fibrinopeptide) for recalibrating the data.
Direct MALDI-MS/MS
Instrument calibration and definition of regions to be acquired was performed as described above for MS measurements; the spatial resolution and acquisition mode were also equivalent with those applied in MS-mode. Fragmentation was carried out in the transfer cell of the TriWave region (Pringle et al. 2007). Peptides were fragmented utilizing an optimized 20 eV collision energy ramp according to the precursor mass (PM). All data were acquired in the mass range from 100 to PM+50 m/z and processed with the MassLynx software (Waters Corporation). The masses were corrected with an external lock mass ([Glu1]-fibrinopeptide) and deisotoped using the MaxEnt 3 algorithm. The resultant peak list was submitted to a MASCOT (Matrix Science, London, UK) query search against the Swiss-Prot database (2012_03). The parent and fragment ion tolerances were set at 25 ppm and 0.2 Da, respectively. One missed cleavage and methionine oxidation as variable modifications were also included.
Results
MALDI-MS-Imaging on FFPE Amyloid-laden Human Tissue Sections
In the first set of experiments, we used FFPE liver tissue from a 68-year-old woman with hereditary AApoAI amyloidosis, heart tissue from a 51-year-old man with ALλ amyloidosis, heart tissue from a 62-year-old woman with hereditary ATTR amyloidosis, and subcutaneous abdominal tissue from a 38-year-old woman with AIns amyloidosis. For all four cases, unknown peptides at m/z 1646.815, 1422.661 and 1314.682 were detected when conducting the MALDI-MSI experiment. Figure 1B–1D displays the spatial distribution of the AApoAI amyloid-laden liver tissue exhibiting local maxima of intensity, thus showing protein deposits typical for amyloid plaques. Comparing the peptide mass images with the ApoAI-immunostaining (Fig. 1A) revealed a similar distribution pattern for the positive-stained amyloid areas and the local intensity maxima of all three peptides. This result indicates the co-localization of these peptides within amyloid deposits. These observations were also made for the ALλ-, ATTR- and AIns amyloid cases (Supplemental Fig. 2). MS spectra of amyloid deposits are given in the supplementary section for all four samples (Supplemental Fig. 2).
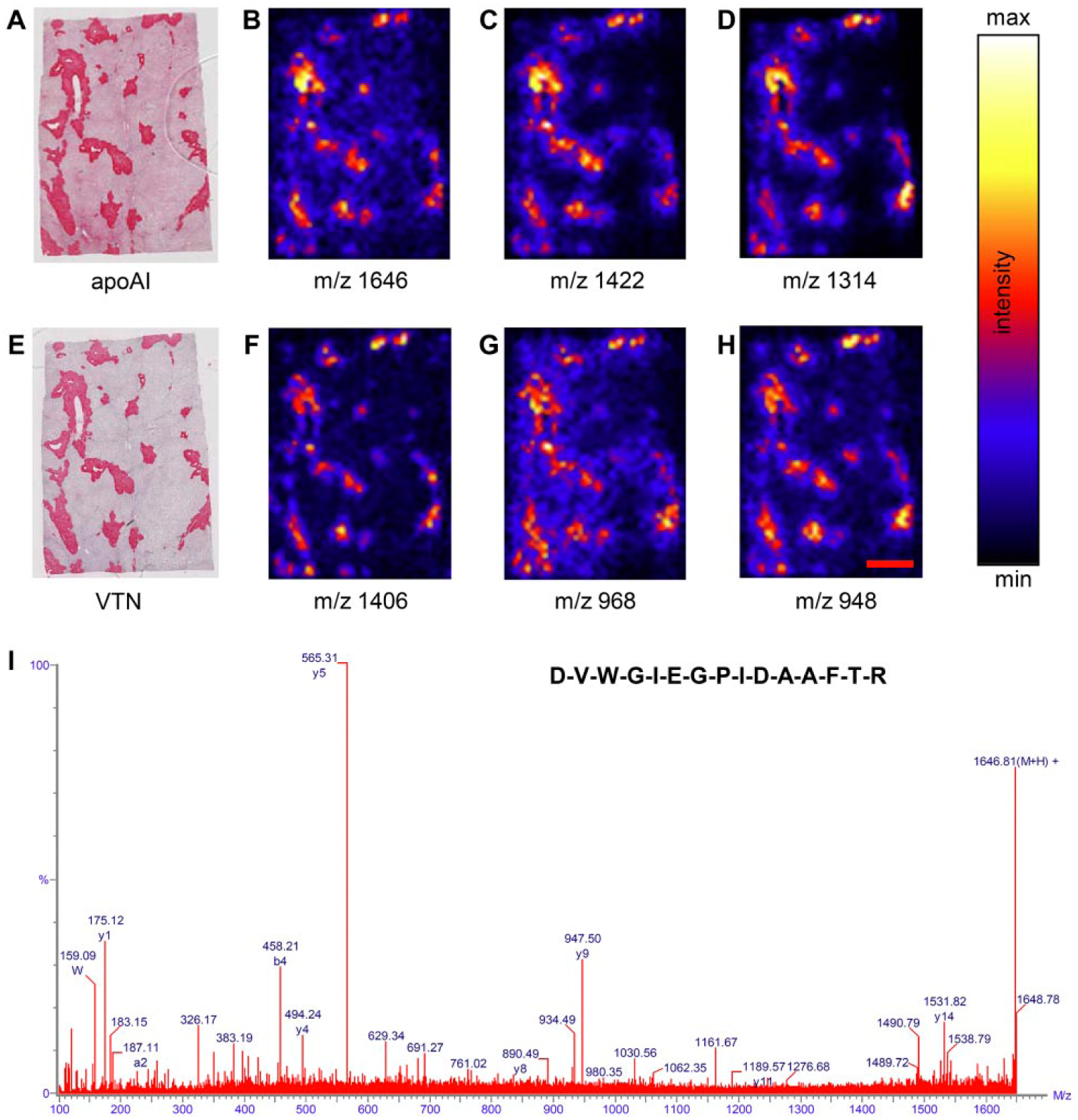
Immunohistochemical staining of apoAI (A) and vitronectin (VTN; E) shows high intensity staining for amyloid deposits in formalin-fixed, paraffin-embedded amyloidogenic liver tissue sections. MALDI-MS images display the spatial distribution of tryptic peptides belonging to VTN (B−D), SAP (F) and apoE (G, H) within the tissue section. For all peptides, high signal intensities were detected in the amyloid deposits. The MALDI-MS/MS spectrum (I) displays the fragmentation pattern of the tryptic peptide at m/z 1646 identified as VTN. Scale, 2 mm.
MALDI-MS/MS of Vitronectin on FFPE Amyloidogenic Human Tissue Sections
Direct MALDI-MS/MS was carried out to identify the three peptides at m/z 1646.815, 1422.661 and 1314.682 observed in the MSI experiment. When submitting the processed data to a MASCOT query search, all three peptides were identified as tryptic peptides of the glycoprotein vitronectin (Table 1). An example of the MS/MS spectra is given in Fig. 1I. The three identified peptides are proteotypic; this describes peptides with amino acid sequences unique for vitronectin, allowing unambiguous identification unlike so-called shared peptides that harbor sequences that could be part of other proteins. This result was validated by immunostaining with an antibody against vitronectin (Fig. 1E). The positive red staining in the IHC image and the local maxima of intensity in the peptide images demonstrate a similar distribution within the tissue section. These findings indicate that vitronectin is present within AApoAI amyloid deposits, particularly when comparing IHC images with a congruent staining pattern (Fig. 1A, 1E). Similar validations were carried out for ALλ-, ATTR- and AIns amyloid cases (Supplemental Fig. 2 and Supplemental Table 1).
Identification of Peptides in Amyloidogenic Human Liver Tissue.
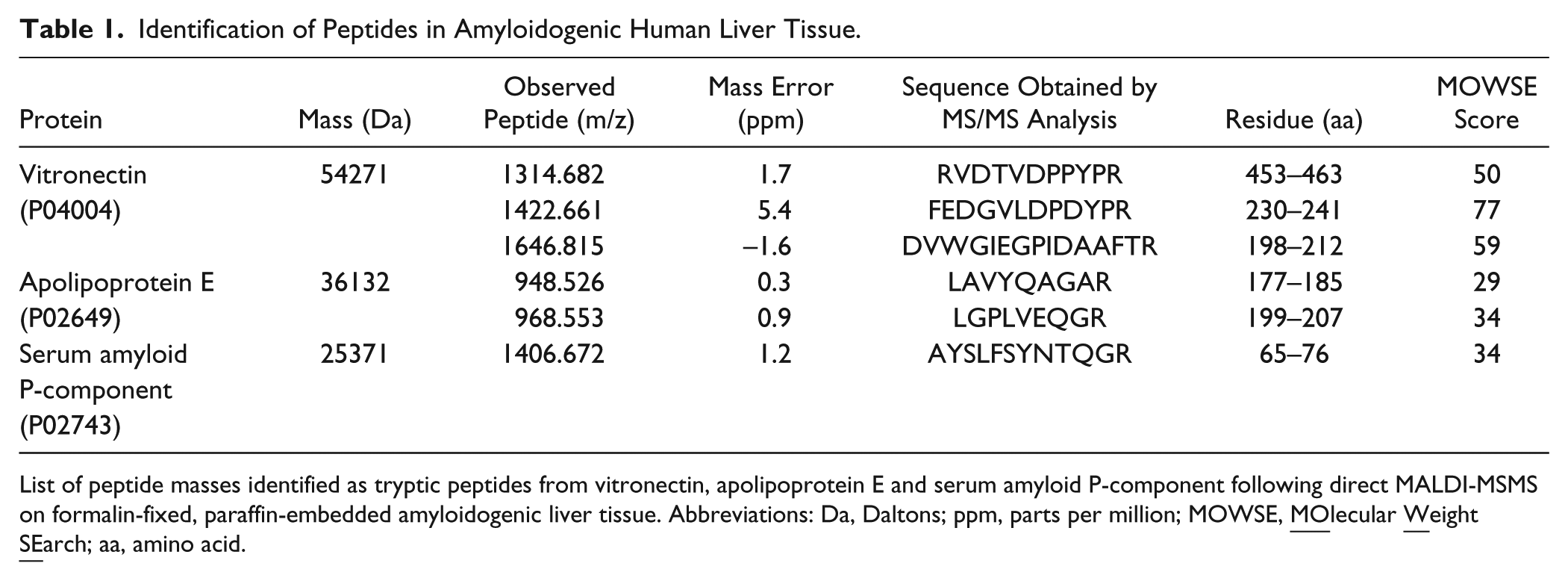
List of peptide masses identified as tryptic peptides from vitronectin, apolipoprotein E and serum amyloid P-component following direct MALDI-MSMS on formalin-fixed, paraffin-embedded amyloidogenic liver tissue. Abbreviations: Da, Daltons; ppm, parts per million; MOWSE,
In addition, direct MALDI-MS/MS identified tryptic peptides of apolipoprotein E (apoE) and serum amyloid P-component (SAP) on the AApoAI amyloid-laden liver tissue, which are both known to be common components of amyloid deposits (Table 1). Figure 1F−1H show the spatial distribution of the corresponding peptides at m/z 1406.672 (SAP), and 968.553 and 948.526 (apoE) with an intensity pattern similar to the peptides of vitronectin and the apoAI-immunostaining.
Immunohistochemical Validation of the Presence of Vitronectin within Diverse Types of Amyloids
The results of the MALDI-MSI experiments led to the suggestion that vitronectin may be a common component of amyloids. Subsequently, the presence and histoanatomical distribution of vitronectin was validated in 175 amyloid-containing tissue samples from 175 patients with diverse types of amyloid, including 107 (61%) men and 68 (39%) women. The age of the patients ranged from 23 to 94 years. The tissue samples of all patients showed variable amounts of amyloid deposits, demonstrating the typical apple-green birefringence in polarized light after Congo red staining. Immunostaining with an antibody against vitronectin was positive in the amyloid deposits of 170 (98%) cases (Table 2). To visualize the spatial distribution of vitronectin in different amyloid types, Congo red-stained tissue sections were observed with fluorescence microscopy (Fig. 2). This method offers the advantage that even small amyloid deposits, which are sometimes difficult to recognize in polarized light, can be visualized as an orange fluorescence. Table 2 shows the number of cases that stained positive for vitronectin for each amyloid type. Subsequently, we scored the extent of vitronectin immunostaining of the amyloid deposits into one of four categories: 0%, ≤10%, 11-90%, >90% of the amyloid area. For all AFib- and AApoAI amyloid cases, vitronectin immunostaining was seen to cover more than 90% of the amyloid area. This degree of coverage was less frequently observed for AA (85% of cases), ALκ (58%), ATTR (54%), Aβ (33%), ALλ (29%), Aβ2M (0%), AIns (0%) amyloid cases, indicating that some amyloid types tend to enclose vitronectin more extensively than others. Furthermore, grouping the cases according to the type of amyloid revealed that the amount of vitronectin in amyloid deposits varies not only with the amyloid type, but also within each group.
Validation Cohort for 175 Amyloid Cases Investigated using Immunohistochemistry with Anti-Vitronectin.
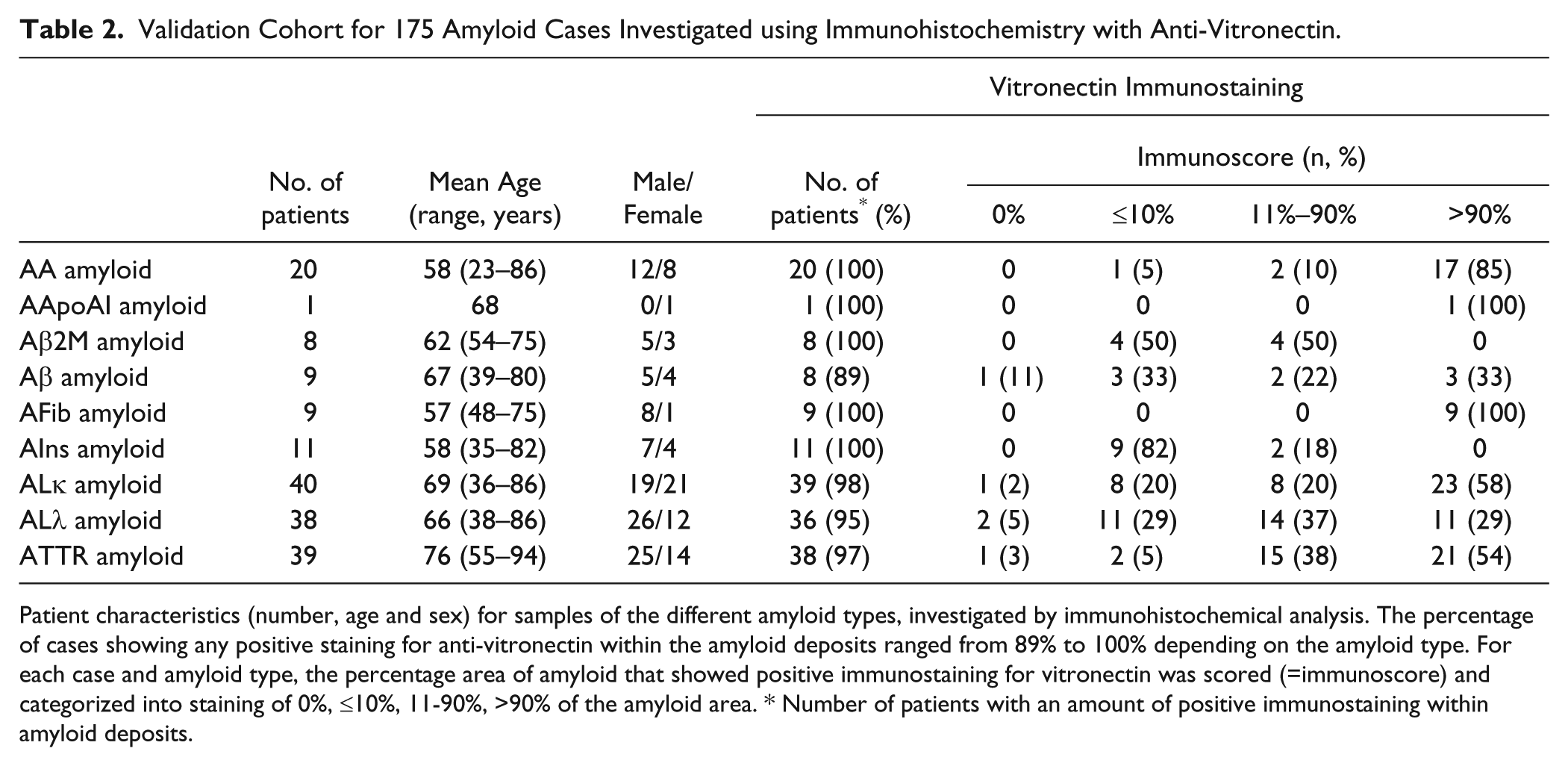
Patient characteristics (number, age and sex) for samples of the different amyloid types, investigated by immunohistochemical analysis. The percentage of cases showing any positive staining for anti-vitronectin within the amyloid deposits ranged from 89% to 100% depending on the amyloid type. For each case and amyloid type, the percentage area of amyloid that showed positive immunostaining for vitronectin was scored (=immunoscore) and categorized into staining of 0%, ≤10%, 11-90%, >90% of the amyloid area. * Number of patients with an amount of positive immunostaining within amyloid deposits.
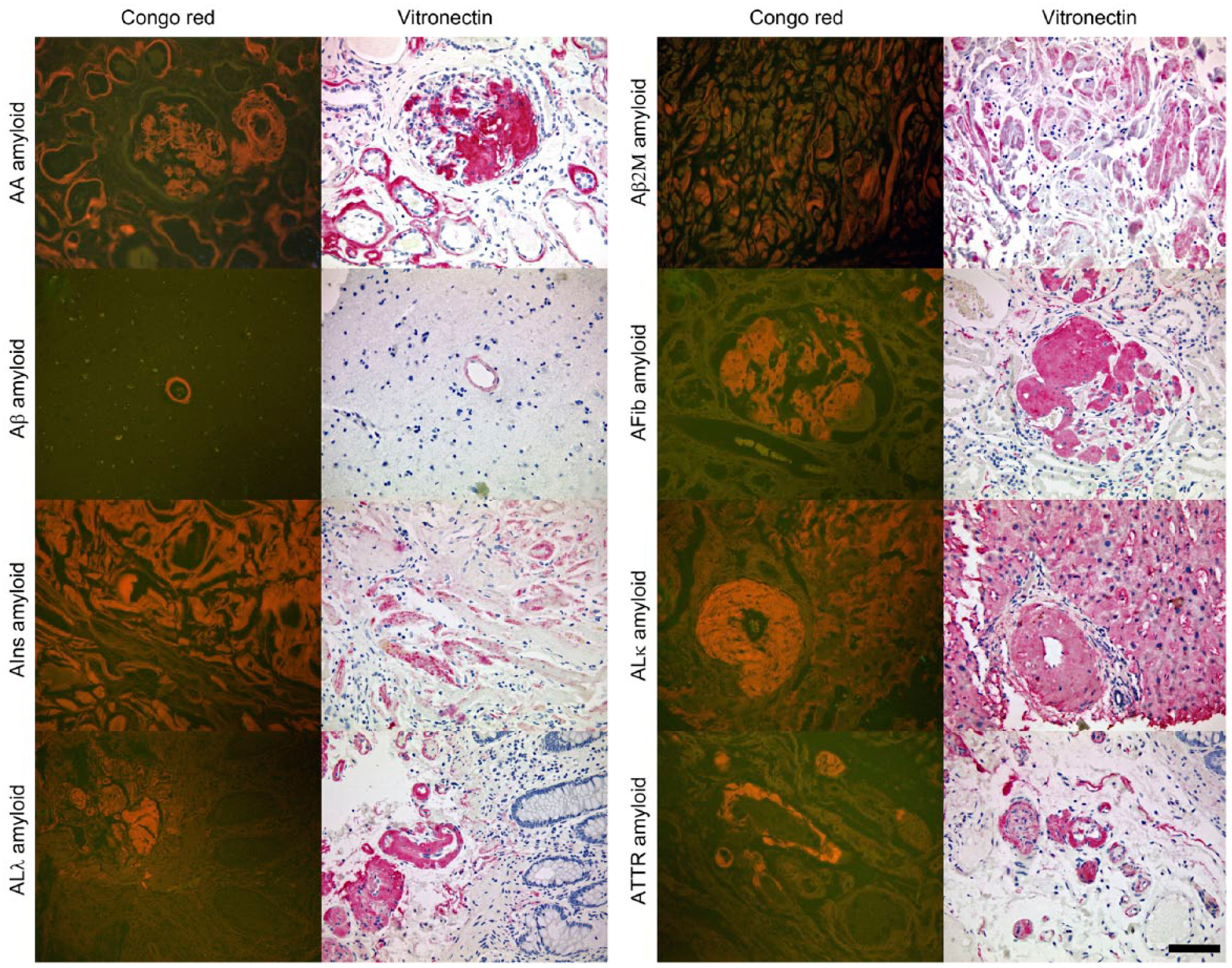
The presence of vitronectin within amyloid deposits is illustrated for several amyloid types: AA- and AFib amyloid both in kidney; ALλ- and ATTR amyloid in the large intestine; Aβ amyloid in brain; AIns amyloid in subcutaneous fat; Aβ2M amyloid in carpal tunnel ligament; and ALκ amyloid in liver. Congo red staining is observed with fluorescence microscopy, displaying amyloid deposits in orange. The case of AIns amyloid demonstrates that the distribution of vitronectin does not always cover the whole amyloid area. Scale, 80 µm.
Discussion
In our study, MALDI-MSI was used for the first time to identify the specific and spatial enrichment of vitronectin in amyloid deposits in four different cases (AApoAI, ALλ, ATTR, AIns). Our results were confirmed by immunohistochemistry in a validation cohort of 175 separate patients, including samples from 22 different histoanatomical locations and 9 different amyloid types. Thus, vitronectin is revealed as a common constituent of amyloid deposits. Using a significantly larger cohort, our finding confirms and extends upon previous observations made by others: the presence of vitronectin has been reported for Aβ amyloid in the brain of 15 patients (Akiyama et al. 1991), for AL amyloid in neuropathy of 3 patients (Zanusso et al. 1992) and for renal AA- and AL-amyloidosis of 20 and 6 patients (Dahlbäck et al. 1987).
Vitronectin is a multifunctional glycoprotein expressed in liver (Jenne et al. 1989; Seiffert et al. 1991), brain (Walker and McGeer 1998), retina (Anderson et al. 1999; Hageman et al. 1999; Ozaki et al. 1999) and vascular smooth muscle cells (Dufourcq et al. 1998). It appears in blood and in the ECM as an intact 75 kDa polypeptide or as two polypeptide chains (65 kDa and 10 kDa) linked by an intramolecular disulfide bridge (Hayman et al. 1983; Preissner 1991). Its amino acid sequence contributes 54 kDa to this molecular mass. Given that the primary structure contains multiple binding sites for other macromolecules, vitronectin is involved in several biological processes (Preissner 1991; Schvartz et al. 1999). For example, interaction of the Arg-Gly-Asp (RGD) sequence with specific integrin receptors mediates the adhesion, spreading and migration of cells (Barnes et al. 1983; Basara et al. 1985; Cherny et al. 1993). Furthermore, association of vitronectin with components of the ECM is facilitated by a collagen-binding domain and a polycationic heparin-binding domain (Suzuki et al. 1984; Gebb et al. 1986; Ishikawa-Sakurai and Hayashi 1993). Because amyloid deposits are known to contain several components and proteins of the ECM (Röcken and Eriksson 2009), vitronectin may be another interesting ECM component involved in amyloid pathology. A study investigating the influence of ECM compounds on the expression of the β-amyloid precursor protein (APP) and its processing to amyloidogenic derivatives in fibroblasts showed that vitronectin increases the amount of both species (Bronfman et al. 1996). This finding lends support to the hypothesis that the extracellular accumulation of vitronectin may precede the formation of amyloid. Vitronectin is able to form amyloid fibrils in vitro (Shin et al. 2008) and thereby may act as a nidus for the induction and acceleration of amyloid formation. A similar mechanism has been described by others, where the oral and intravenous administration of amyloid fibrils into a susceptible animal functions as an “amyloid-enhancing-factor” (Lundmark et al. 2002, 2005). However, as we did not observe vitronectin in every case, an opposite mechanism may also be possible: vitronectin is incorporated after primary accumulation of amyloid fibrils, where the amyloid fibril serves as a nidus for the accumulation of vitronectin.
The observed varying amounts of vitronectin within amyloid deposits provides evidence that both ECM composition and type of amyloid may affect the specific and spatial accumulation of vitronectin. This is because amyloids of systemic amyloidosis are exposed to different extracellular environments and amyloids originating from different precursor proteins possess dissimilar physico-chemical properties. In order to predict if and how vitronectin participates in amyloidogenesis, several essential questions have to be answered in future investigations: (1) What is the order of events: Do amyloid fibrils accumulate first or does vitronectin? (2) How do ECM composition and amyloid type affect vitronectin accumulation within the amyloid deposits? (3) Does the primary structure of vitronectin contain binding sites that would facilitate linkage to amyloidogenic polypeptides?
These concerns aside, in this work, we demonstrated the potential of MALDI-MSI as a useful assisting tool for studying amyloid composition by revealing the co-localization of vitronectin, apolipoprotein E and serum amyloid P-component in AApoAI amyloid deposits. Furthermore, through immunohistochemistry, we show vitronectin to be a variable component of the matrix, with several different local and systemic amyloid types. The influence of vitronectin on the formation and aggregation of amyloid fibrils is still unclear and requires further investigations.
Footnotes
Acknowledgements
The authors thank Prasath Somasundaram and Dennis Linke for their technical assistance.
Declaration of Competing Interests
The authors declared no potential competing interests with respect to the research, authorship, and/or publication of this article.
Author Contributions
Study concept and design was done by MW, AT, and CR. Clinical data were acquired by HS, surgical pathological data by SK and CR and mass spectra data by MW and AT. The data were analyzed and interpreted by MW, AT, SK and CR. Drafting of the manuscript and critical revision of the manuscript for important intellectual content was done by all authors. CR obtained funding. Administrative, technical, or material support was provided by AT, SK and CR. The study was supervised by AT and CR.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by grants of the Germany Research Foundation (Grant-No. Ro 1173/11) and the Federal Ministry of Education and Research (GERAMY).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
