Abstract
Matrix metalloproteinase-20 (MMP-20) expression is widely regarded as tooth-specific, with expression limited to dental hard tissues. Necessary for sound enamel formation, MMP-20 and MMP-2 proteolytically process dentin sialophosphoprotein (DSPP) into dentin sialoprotein, dentin phosphoprotein, and dentin glycoprotein during tooth formation. In the mid-2000s, three members of the
Keywords
Introduction
The expression of matrix metalloproteinase-20 (MMP-20) is widely regarded as tooth-specific, and required for the development of structurally sound dental enamel (Llano et al. 1997; Grant et al. 1999; Turk et al. 2006; Bartlett et al. 2011). Only sporadic reports of its expression in non-odontogenic tissues have been reported (Väänänen et al. 2001). Earlier reports indicate that MMP-20 and MMP-2 proteolytically process dentin sialophosphoprotein (DSPP)—a member of the
A decade ago, we reported the expression of all five members of the SIBLING family in metabolically active duct systems of the salivary and eccrine sweat glands, and the nephron (Ogbureke and Fisher 2004, 2005, 2007). We also confirmed that the specific in vitro binding and activation of MMP-2, MMP-3, and MMP-9 by three SIBLINGs—bone sialoprotein, (BSP), osteopontin (OPN), and dentin matrix protein1 (DMP1), respectively (Fedarko et al. 2004)—also occurs in biological systems along with the possible biological relevance of such interactions (Ogbureke and Fisher 2004, 2005, 2007). Furthermore, our most recent report documented the expression of MMP-20 in human oral squamous cell carcinoma (OSCC), along with its co-expression and specific interaction with DSPP, thereby establishing MMP-20 as the cognate MMP partner for DSPP (Saxena et al. 2015).
Based on these previous reports, we hypothesized that MMP-20 is expressed, and co-expresses and interacts with DSPP in salivary glands. We investigated this hypothesis in normal human major salivary glands and cell lines using immunohistochemistry (IHC), immunofluorescence (IF), western blot (WB), quantitative RT-PCR, and in situ proximity ligation assay (iPLA) techniques.
Materials & Methods
Tissue Sections and Cells, Culture Conditions, and Antibodies
Paraffin-embedded, normal, non-pathological, human major salivary glands sections were obtained from de-identified surgical wastes resulting from surgical procedures for pathology not involving the salivary glands but for which the glands were wholly or partially sacrificed. The human normal salivary gland cell line, HSG, has been published (Nagy et al. 2007), and was a kind gift from Dr. Anita Joy (Southern Illinois University, Edwardsville, IL). The human oral keratinocytes (HOK) whole cell lysate (cat# 2616) was purchased from ScienCell (Carlsbad, CA), whereas the immortalized human oral keratinocyte (HOK16B) cell line (Park et al. 1995) was a kind gift from Dr. Nadarajah Vigneswaran (University of Texas School of Dentistry, Houston, TX). The OSC2 cell line (Osaki et al. 1994) was a kind gift from Dr. Stephen Hsu (Georgia Regents University, GA). Archived paraffin sections of human OSCC used as positive controls for MMP-20 expression have been published (Saxena et al. 2015). HSG and HOK16B cells were cultured in keratinocyte growth medium supplemented with growth factors, cytokines, and other supplements (KGM-2, CC3103, CC- 4152; Lonza, Allendale, NJ). The OSC2 cell line was maintained in Dulbecco’s modified Eagle’s medium/Nutrient Mixture F-12 (DMEM/F-12, 11330-032; Life Technologies, Grand Island, NY) containing 10% fetal bovine serum supplemented with 1% penicillin/streptomycin. The mouse monoclonal anti-DSPP antibody (LFMb-21) used in this study has previously been characterized in detail and published (Ogbureke and Fisher 2004; 2005; Chaplet et al., 2006), and was purchased from Santa Cruz Biotechnology (sc-73632; Dallas, TX). The recently published rabbit polyclonal anti-MMP-20 antibody (Saxena et al., 2015) was made by conjugating the human MMP-20 peptide sequence YRLFPGEPKWKK (108-119/483) to keyhole limpet hemocyanin (KLH), and was purchased from Bioss Inc. (bs-5788R; Woburn, MA). These and other antibodies used in this study are summarized in Table 1.
Antibodies used in this study.
MMP, matrix metalloproteinase; DSPP, dentin sialophosphoprotein; BSP, bone sialoprotein; OPN, osteopontin; DMP1, dentin matrix protein1; MEPE, matrix extracellular phosphoglycoprotein.
Immunohistochemistry
Unless otherwise stated, reagents were from Biocare Medical (Concord, CA). Standard immunoperoxidase techniques were performed on archival formalin-fixed, paraffin-embedded human parotid, submandibular and sublingual salivary gland sections (4–5 μm) with MACH4 universal HRP (M4U534H)/AP (M4U536H) polymer kit. Briefly, following deparaffinization in xylene and rehydration through a series of ethanol and water, antigen retrieval was performed as previously described (Saxena et al. 2015) followed by endogenous peroxidase quenching by treating with peroxidase blocking reagent for 10 min. Sections were then treated with background punisher (BP974H) for 20 min to reduce non-specific binding. Thereafter, sections were incubated overnight at 4°C with DSPP and MMP-20 antibodies diluted in Da Vinci Green diluent before incubating with MACH4 polymer for 30 min at room temperature. Double-stain immunolabeling for the co-detection of MMP-20 and DSPP was carried out on serial sections of salivary glands with DSPP and MMP-20 antibody cocktail and MACH2 double-stain 2, mouse-HRP + rabbit-AP kit (MRCT525) following the manufacturer’s instructions, with modifications.
Chromogen detection was performed either with 3,3’-Diaminobenzidine (DAB; BDB2004), Warp Red (WR806H) for single-stain sections, or with any appropriate combination of two chromogens for double-labeling. Sections were counterstained with hematoxylin. Negative control salivary gland sections were treated with Universal Negative Control Serum (NC498). Positive controls consisted of sections of human OSCC known to express MMP-20 and DSPP, whereas tissue negative controls for MMP-20-DSPP expression consisted of normal oral mucosa known to lack MMP-20 and DSPP expression. Representative photographic images were captured using an Eclipse Ni-E microscope with Nikon DS-U3 digital camera and NIS Elements AR software (Nikon; Melville, NY).
Western Blotting
WB was performed on whole cell extracts of HSG, HOK (negative control), and OSC2 (positive control) cells to determine the expression of MMP-20 and DSPP as well as the expression of other SIBLINGs and their cognate MMPs. The total amount of protein was quantitated, and equal amounts loaded and resolved with 4–20% Criterion TGX (567-1094; Bio-Rad, Hercules, CA) SDS-polyacrylamide gel electrophoresis. This was followed by electrophoretic transfer using a Trans-Blot Turbo Transfer System (170-4155EDU, Bio-Rad) to a low fluorescence PVDF membrane (20130403, Bio-Rad). Proteins were detected using respective primary and secondary antibodies, and photographed using a LiCor Odyssey scanner (Lincoln, NE). Band intensity was normalized using actin, and quantified using ImageJ (NIH; Bethesda, MD).
Real Time PCR
Total RNA was extracted from HSG cells using Trizol reagent (15596-026; Life Technologies, Grand Island, NY) following a standardized protocol. The concentration of each sample was measured using a NanoDrop spectrophotometer (Wilmington, DE). All primers were designed using NCBI Primer-Blast (http://www.ncbi.nlm.nih.gov/tools/primer-blast/), and are listed in Table 2. One microgram of total RNA was reverse transcribed using iScript RT Supermix (1708841; Bio-Rad). qRT-PCR was performed from synthesized cDNA on a real-time qPCR machine using iTaq Universal SYBR Green PCR Master Mix (1725124, Bio-Rad). A standard curve was generated from three serial dilutions of cDNA. Samples, including negative controls, were analyzed in triplicate, and PCR products were verified using dissociation curve analysis. mRNA levels were normalized to GAPDH and analyzed using Bio-Rad CFX manager software.
Primers Used for RT-PCR.
DSPP, dentin sialophosphoprotein; OPN, osteopontin; BSP, bone sialoprotein; DMP1, dentin matrix protein1; MEPE, matrix extracellular phosphoglycoprotein; MMP, matrix metalloproteinase.
Immunofluorescence
Cells plated on coverslips in a 6-well dish overnight were washed 3 times with 1×PBS and fixed in 3% paraformaldehyde for 20 min before permeabilization with 0.1% Triton X-100 for 10 min. Cells were then treated with blocking buffer (1×PBS, 3% goat serum) for 1 hr at room temperature, followed by overnight incubation at 4°C with primary antibodies to DSPP and MMP-20 diluted in blocking buffer. Coverslips were washed 3 times with 1×PBS and incubated with secondary antibodies for 1 hr before mounting with Prolong Gold Anti-Fade Reagent with DAPI (P36931; Life Technologies).
In Situ Proximity Ligation Assay
In situ Proximity Ligation Assay (iPLA) was used to verify specific cellular interactions of MMP-20 with DSPP in fixed human major salivary gland tissue sections and in HSG cells. Prior to iPLA, cells were fixed in 4% paraformaldehyde at 4°C for 15 min; fixed, paraffin-embedded salivary gland tissue sections were deparaffinized and subjected to antigen retrieval. iPLA was performed using the HRP/NovaRed detection kit (DUO92012; Olink Biosciences, Sigma-Aldrich, St. Louis, MO) according to the manufacturer’s protocol. Briefly, tissue sections were incubated with primary anti-MMP-20 polyclonal (rabbit) antibody and anti-DSPP monoclonal (mouse) antibody or with the appropriate normal rabbit/mouse IgG. Sections were then incubated with corresponding secondary antibodies conjugated to oligonucleotide PLA probes (MINUS and PLUS) for 1 hr at 37°C. Rolling circle amplification (RCA) was performed using T4-ligase, as described by the manufacturer (Olink Bioscience). HRP/NovaRed oligonucleotides were used to detect RCA products in the tissue sections. Protein interactions were observed as brown, punctate signals and were captured using a Nikon Eclipse Ni-microscope and NIS Elements AR software.
Statistical Analyses
Results were analyzed using either a Student’s t-test or one-way analysis of variance (ANOVA) with subsequent post-hoc Tukey’s pairwise analysis using GraphPad Prism v6 (San Diego, CA). Data are presented as the mean ± SEM, with statistically significant differences indicated by P values less than or equal to 0.05.
Results
MMP-20 Expression in Normal Human Salivary Glands
To investigate the expression of MMP-20 in salivary glands, we performed IHC on fixed tissue sections of parotid, submandibular, and sublingual salivary glands, and surveyed the normal HSG cell line for MMP-20 expression by WB and qRT-PCR. As shown in Figure 1, there was strong positive immunoreactivity (red) for MMP-20 in ducts of the parotid (Fig. 1A) and submandibular (Fig. 1B) glands as well as punctate expression (Fig. 1A, 1B; arrows) in their serous acini and demilunes, respectively. Immunoreactivity for MMP-20 was present (red) but less intense in the ducts of the sublingual glands (Fig. 1C). However, there was complete negative immunoreactivity for MMP-20 in the mucous acini of all three major glands (Figs. 1A–1C). A human OSCC section known to express MMP-20 was used as a positive control (Fig. 1D, red staining). Negative controls consisted of normal oral mucosa known to be negative for MMP-20 (Fig. 1E) and salivary gland sections treated with pre-immune IgG (Fig. 1F). WB (Fig. 1G) and qRT-PCR (Fig. 1H) analyses confirmed the significant upregulation of MMP-20 in human HSG cells at the protein (p<0.001) and mRNA levels (p<0.005), respectively. This contrasts with barely detectable expression levels of MMP-20 in human oral keratinocyte (HOK) lysates that were used as a negative control. OSC2 cells, which are known to upregulate MMP-20 (Saxena et al. 2015), were used as a positive control. Furthermore, MMP-2, MMP-3, and MMP-9, along with their respective cognate SIBLING partners, BSP, OPN, and DMP1, respectively, as well as MEPE, were expressed in the HSG cells at the protein (Fig. 1G) and mRNA (Fig. 1H) levels.
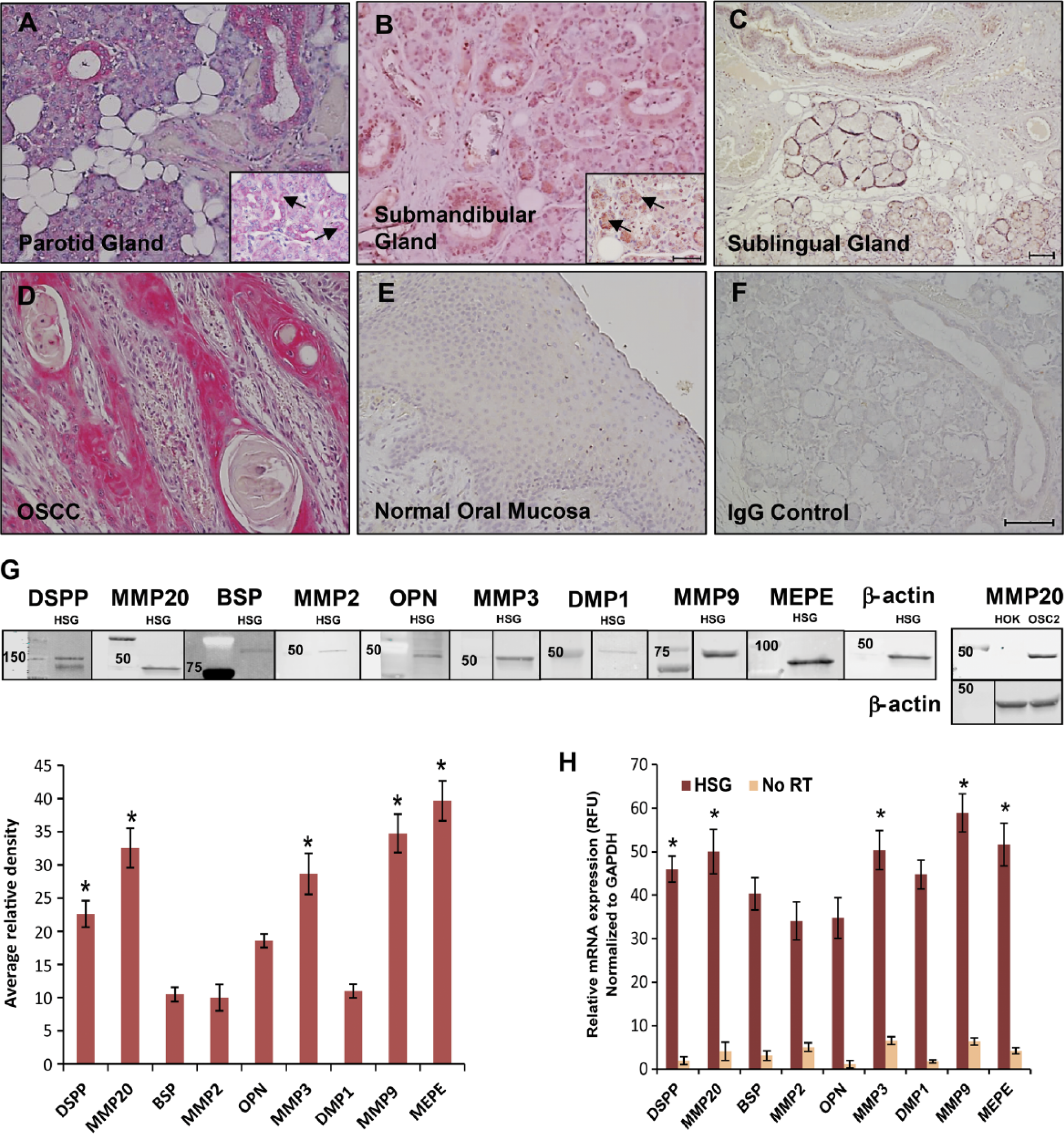
Expression of matrix metalloproteinase-20 (MMP-20) MMP-20 in normal human salivary glands. Positive immunoreactivity (red) for MMP-20 in ducts of human parotid (A), submandibular (B), and sublingual (C) glands with punctate (arrows in inset), focal, serous acinar immunoreactivity. There is negative immunoreactivity for MMP-20 in the mucous acini of all three major glands. Sections of human oral squamous cell carcinoma (OSCC), known to show positive immunoreactivity to MMP-20, was used as a tissue positive control (D), whereas sections of normal oral mucosa, known to be negative for MMP-20, was used as a tissue negative control (E). (F) Pre-immune IgG negative control on salivary gland section. (G) Western blots (WB) and quantitative analysis of protein expression of MMPs and SIBLINGs in human normal salivary gland (HSG) cells. Protein levels of MMP-20, dentin sialophosphoprotein (DSPP), MMP-3, MMP-9, and matrix extracellular phosphoglycoprotein (MEPE) were significantly higher (p<0.001) than that of bone sialoprotein (BSP), MMP-2, osteopontin (OPN), and dentin matrix protein1 (DMP1) in HSG cells. WB analysis of MMP-20 in human oral keratinocyte (HOK) lysate and the OSC2 human oral squamous cell carcinoma (OSCC) cell line were used as negative and positive controls, respectively. β-actin was used for normalization. Average relative density is shown below the blots as a bar graph. (H) Quantitative real-time RT-PCR analysis shows significant up-regulation (p<0.005) of mRNA levels of MMPs and SIBLINGs in HSG cells. GAPDH was used as the endogenous control. Values are mean ± SE. n=3. Scale, 100 μm. Abbreviation: SIBLINGs,
MMP-20 Co-expression with DSPP in Normal Human Salivary Glands
Based on reports of the binding of three SIBLINGs to, and their activation of, specific MMPs (BSP/MMP-2, OPN/MMP-3, DMP1/MMP-9) in biochemical and biological systems (Fedarko et al. 2004; Ogbureke and Fisher 2004; Ogbureke and Fisher 2005; Ogbureke and Fisher 2007), we hypothesized that MMP-20 co-expresses and potentially interacts with DSPP in salivary glands. Representative double-stain IHC results (serial tissue sections) show co-immunoexpression of MMP-20 with DSPP on human salivary gland tissue sections, with cytoplasmic, perinuclear, and punctate nuclear localizations (Figs. 2A–2C; arrows). A representative hematoxylin and eosin (H&E)-stained section of normal salivary gland is shown in Figure 1D, whereas Figure 1E shows a representative pre-immune IgG negative control. Double immunostaining for DSPP (red) and MMP20 (brown) in OSCC, which was used as tissue positive control, is shown in Figure 1F. Immunofluorescence (IF) results show MMP-20–DSPP co-expression in HSG cells (Figs. 2G–2I). Figure 2J shows IgG negative IF in HSG cells, whereas HOK16B (Fig. 2K) and OSC2 (Fig. 2L) cells represent negative and positive cell controls, respectively, for MMP-20–DSPP co-expression (Saxena et al. 2015).
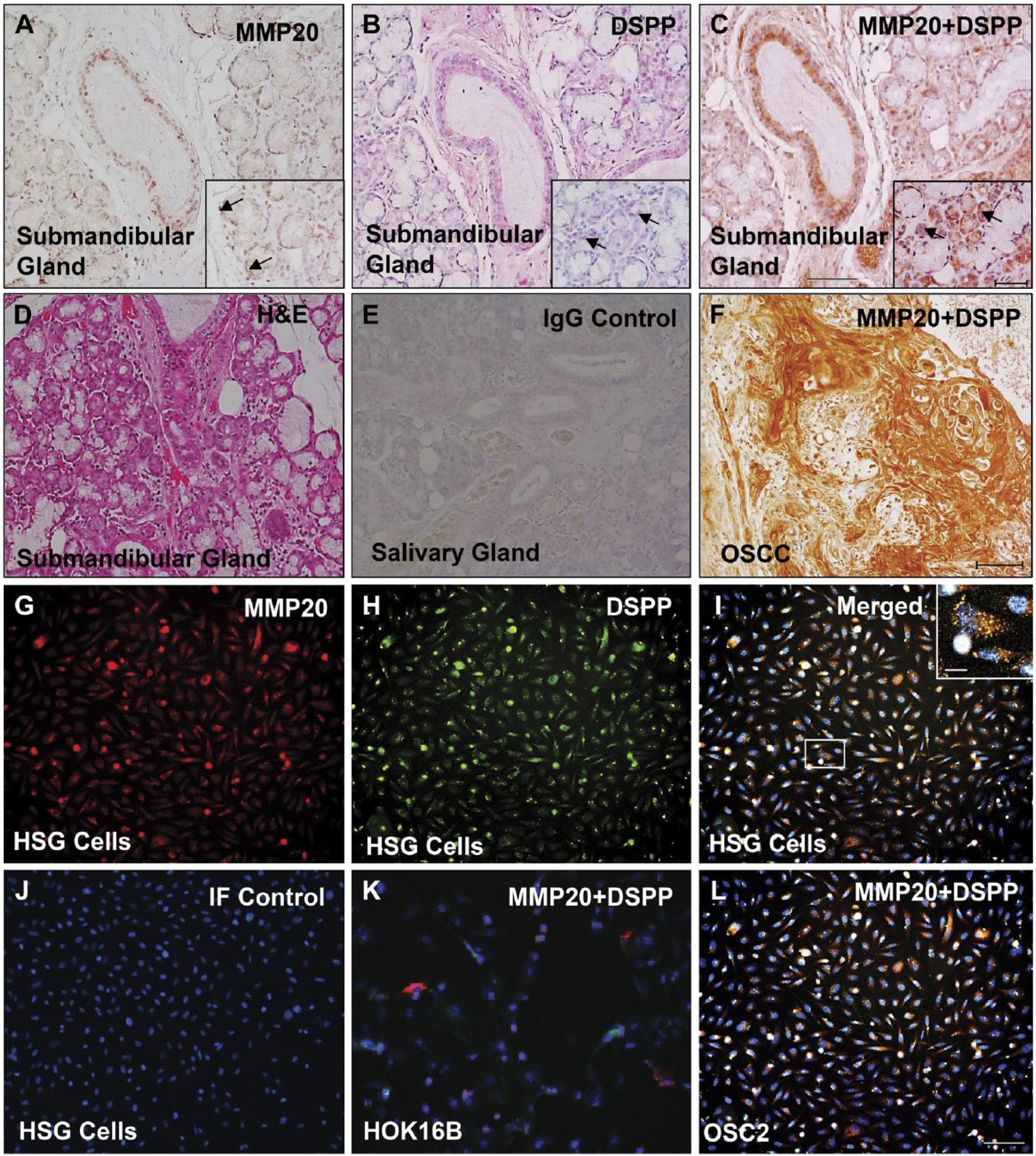
Co-expression of matrix metalloproteinase-20 (MMP-20) and dentin sialophosphoprotein (DSPP) in human salivary glands. MMP-20 positive immunoreactivity (brown; A), DSPP positive immunoreactivity (red; B), and MMP-20–DSPP co-immunoreactivity (brown, HRP; red, alkaline phosphatase) in ducts and acinar cells (A–C; arrows in inset) of the submandibular gland (C). (D) Hematoxylin and eosin (H&E)-stained submandibular gland. (E) Representative pre-immune IgG negative control. (F) Double immunostaining for DSPP (red) and MMP20 (brown) in OSC2 human oral squamous cell carcinoma (OSCC), as a representative tissue positive control. Immunofluorescence for MMP-20 (red; G), DSPP (green; H), and their co-expression (yellow hue; I) in human normal salivary gland (HSG) cells. (J) IgG negative control. Co-expression of DSPP (green) and MMP20 (red) in immortalized human oral keratinocytes, HOK16B, was used as a cell negative control (K), whereas OSC2 cells were used as a cell positive control (L). Scale, 100 μm; (C, inset) 50 μm.
MMP-20 May Interact with DSPP in Normal Human Salivary Glands
To form the foundation for future investigation of the function/mechanism(s) of any potential MMP-20–DSPP interaction in normal salivary gland physiology, we sought preliminary evidence of such interaction. As shown in Figure 3A, iPLA on salivary gland tissue sections showed cytoplasmic, perinuclear, and punctate nuclear signals, indicating MMP-20 and DSPP co-expression and interaction. Significantly, the MMP-20–DSPP interaction was very intense and specific, and precluded MMP-20 from interacting with the other three SIBLINGs, OPN (Fig. 3B), BSP (Fig. 3C), and DMP1 (Fig. 3D), whose MMP partners, MMP-2, MMP-3, and MMP-9, respectively, are also known. Sections of human OSCC known to co-express MMP-20 and DSPP, used as a tissue positive control, were also positive for the MMP-20–DSPP interaction (Fig. 3E). On the other hand, normal oral mucosa, which is known to express neither MMP-20 nor DSPP and was used here as a tissue negative control, was negative for the MMP-20-DSPP interaction (Fig. 3F). The IgG negative control is shown in Figure 3G.
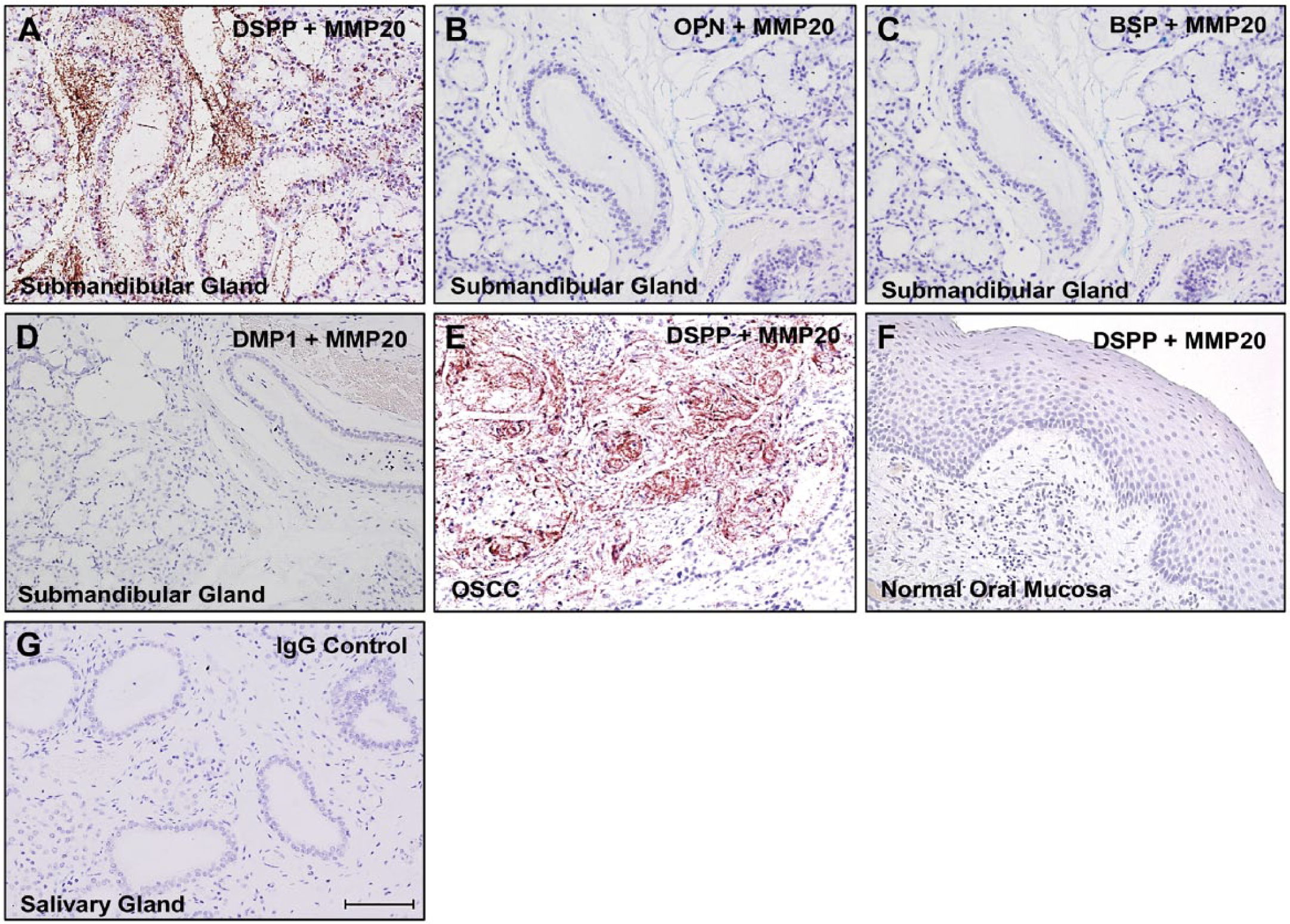
Matrix metalloproteinase-20 (MMP-20) and dentin sialophosphoprotein (DSPP) interaction in normal human salivary glands. In situ proximity ligation assay (iPLA) depicting MMP-20 interactions (brown dots) with DSPP (A). There was no interaction of MMP-20 with osteopontin (OPN; B), bone sialoprotein (BSP; C), or dentin matrix protein1 (DMP1; D) in normal human salivary gland sections, confirming the specificity of the MMP-20–DSPP interaction. A section of human OSCC was used as a tissue positive control (E), whereas normal oral mucosa was used as a tissue negative control (F). The IgG negative control is shown in (G). Scale,100 μm.
Discussion
Our recent documentation of the expression of MMP-20 and its co-expression with DSPP in oral cancer indicated that MMP-20 may be more widely expressed in different tissues, including normal tissues, than previously thought (Saxena et al. 2015). The purpose of this study therefore was to investigate MMP-20 expression and to verify its co-expression and provide evidence of an interaction with DSPP in human salivary glands. Our data provide clear evidence of the expression of MMP-20, its co-expression, and interaction with DSPP in cells of the salivary glands. Significantly, iPLA results, which indicate specificity of the MMP-20–DSPP interaction (to the exclusion of the other SIBLINGs with known MMP partners; Figs. 3A–3D), confirm MMP-20 as the cognate MMP partner for DSPP. MMP-20 and DSPP can therefore be expected to form spontaneously in vitro with the same high-affinity (nM) complexes observed in vitro for BSP and MMP-2, OPN and MMP-3, and DMP1 and MMP-9 (Fedarko et al. 2004). Furthermore, our current results reinforce prior evidence that the SIBLING–MMP pairing described in vitro may also be important in vivo in non-mineralizing soft tissues (Ogbureke and Fisher 2004, 2005, 2007; Saxena et al. 2015). Although our previous reports documented the expression of the SIBLINGs and their known MMP partners in the normal salivary glands tissues (Ogbureke and Fisher 2004), here we provide, for the first time, additional evidence of the corresponding expression of all five SIBLINGs and known cognate MMPs in a normal HSG cell line.
Our laboratory and those of others have reported the expression of MMP-2, MMP-3, and MMP-9 in the ducts of normal human salivary glands, nephrons, and sweat ducts (Nagel et al. 2004; Ogbureke and Fisher 2004; 2005; 2007). The same expression pattern is recapitulated for their corresponding tissue inhibitors of metalloproteinases (TIMPs; Fedarko et al. 2004). Significantly, the appropriate SIBLING partner was previously shown to be able to reactivate a TIMP-inactivated MMP upon binding in vitro, suggesting that the MMPs may remain active in the ducts/saliva, even in the presence of these inhibitors (Fedarko et al. 2004; Ogbureke and Fisher 2007).
Previous and relatively recent reports show the up-regulation of some SIBLINGs in different tumors, including oral cancers (Bellahcéne et al. 1996; Waltregny et al. 1998; Chaplet et al. 2003; 2006; Ogbureke et al. 2007). At the same time, reports have indicated that the up-regulation of MMP-2, MMP-3, and/or MMP-9 is associated with increased aggressiveness of tumors (Birkedal-Hansen et al. 1993; Brabletz et al. 1999; Thomas et al. 1999; Ogbureke et al. 2007, 2012). Combined, these observations have led to the suggestion that SIBLING–MMP complexes may aid the metastatic processes of tumor cells (Bellahcéne et al. 2008). However, because adult salivary ductal cells are relatively stationary—unlike the metastasizing malignant cells—they may lack the ability to degrade connective tissue/basement membranes as a result of a SIBLING–MMP interaction (Ogbureke and Fisher, 2004). This supports our earlier assertion that SIBLING–MMP complexes in normal adult tissues, such as the salivary gland, may be involved in the normal turnover of cell-surface and/or the repair of pericellular matrix proteins damaged by oxidative by-products of these metabolically active cells (Ogbureke and Fisher, 2004, 2007).
Some MMPs have been detected in saliva (Wu et al. 1997), where they may participate in the processing of specific salivary proteins in transit within the ducts or within the intraoral environment, as previously suggested (Ogbureke and Fisher, 2004). Although we could not locate any report of the detection of MMP-20 in normal human saliva or serum, Wang et al. (2001) were able to detect quantifiable recombinant MMP-20 added to human saliva using the one-step sandwich enzyme immunoassay (EIA) system. It is therefore conceivable that MMP-20 may be secreted and, subsequently, detected in saliva (and serum) of patients with pathological conditions, such as OSCC in which MMP-20 is substantially upregulated.
In summary, we report the expression of MMP-20 in all major human salivary gland ducts, with expression more intense and prominent in the parotid and submandibular glands than in the sublingual glands. This novel finding suggests that MMP-20 expression in normal, non-pathological human tissues may be less tissue-restricted as hitherto thought. Significantly, its co-expression and potential interaction with DSPP supports a paradigm for the central role for SIBLING–MMP interactions in normal salivary gland physiology. An outstanding challenge remains the identification of specific protein substrates of the SIBLING–MMP complexes in normal, non-mineralizing tissues, and the precise biological significance of MMP-20–DSPP interaction in physiological and pathological processes in non-dental tissues.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
