Abstract
The expression of secretogranin III (SgIII) in chicken endocrine cells has not been investigated. There is limited data available for the immunohistochemical localization of SgIII in the brain, pituitary, and pancreatic islets of humans and rodents. In the present study, we used immunoblotting to reveal the similarities between the expression patterns of SgIII in the common endocrine glands of chickens and rats. The protein–protein interactions between SgIII and chromogranin A (CgA) mediate the sorting of CgA/prohormone core aggregates to the secretory granule membrane. We examined these interactions using co-immunoprecipitation in chicken endocrine tissues. Using immunohistochemistry, we also examined the expression of SgIII in a wide range of chicken endocrine glands and gastrointestinal endocrine cells (GECs). SgIII was expressed in the pituitary, pineal, adrenal (medullary parts), parathyroid, and ultimobranchial glands, but not in the thyroid gland. It was also expressed in GECs of the stomach (proventriculus and gizzard), small and large intestines, and pancreatic islet cells. These SgIII-expressing cells co-expressed serotonin, somatostatin, gastric inhibitory polypeptide, glucagon-like peptide-1, glucagon, or insulin. These results suggest that SgIII is expressed in the endocrine cells that secrete peptide hormones, which mature via the intragranular enzymatic processing of prohormones and physiologically active amines in chickens.
Keywords
Introduction
Secretory granules (SGs) in endocrine and neuroendocrine cells contain peptide hormones and bioactive amines. SGs also contain granin-family proteins and membrane-associated prohormone processing enzymes. The granin family consists of seven types of acidic secretory proteins: chromogranin A (CgA), chromogranin B (also known as secretogranin I; SgI), SgII (also known as chromogranin C), and SgIII–VI proteins (Huttner et al. 1991; Taupenot et al. 2003; Portela-Gomes et al. 2010; Bartolomucci et al. 2011). Although the functions of granin-family proteins are not completely understood, recent findings have suggested their involvement in several processes. The granins may be involved in sorting, sequestration, and storage of hormones inside SGs and in the regulation of the biogenesis of SGs and dense-core vesicles from the trans-Golgi network (TGN). They may also function as prohormones, giving rise to an array of peptide fragments as a result of proteolytic processing during carbohydrate and lipid metabolism and behavioral modulation in vivo (Zhang et al. 2006; Takeuchi and Hosaka 2008; Hotta et al. 2009; Hosaka and Watanabe 2010; Bartolomucci et al. 2011).
SgIII is a major granin-family protein originally identified as a product of the neuron-specific gene 1B1075 in mice (Ottiger et al. 1990; Kingsley et al. 1990; Dopazo et al. 1993). The distribution of SgIII has been investigated using immunocytochemistry and in situ hybridization in the nervous and endocrine tissues of various organisms. It has been examined in mice (Ottiger et al. 1990; Hosaka et al. 2002, 2005; Paco et al. 2010); in rat endocrine tissues, such as the anterior pituitary gland (Sakai et al. 2003) and pancreatic islets (Sakai et al. 2004); and in human pancreatic islets (Stridsberg et al. 2008). Immunoreactivity against SgIII in rats has been detected in neurons and astrocytes in the nervous system. It has been also found in thyrotrophs, mammotrophs, gonadotrophs, and corticotrophs in the anterior pituitary and in pancreatic α-, β-, and δ-cells. Immunoelectron microscopy has revealed that SgIII in rat endocrine cells in SGs is localized predominantly in the peripheral region. This localization suggests that SgIII has a role in anchoring the soluble contents of SGs by functioning as a membrane-associated protein (Sakai et al. 2003, 2004).
In our molecular, biochemical, and functional analyses of the role of SgIII in endocrine cells, we demonstrated that SgIII binds to CgA and carboxypeptidase E (CPE), a prohormone-processing enzyme. These associations are crucial for prohormone targeting and sorting within SGs in the regulated secretory pathway (Hosaka et al. 2002, 2005). The SgIII/CgA complex may play a key role in the biogenesis of SGs in mast cells; SgIII has multiple binding partners, such as the mast cell ion channel TRPA1 (Prasad et al. 2008). Furthermore, SgIII binds to cholesterol microdomains in the SG membrane from TGN and functions as an adapter for CgA (Hosaka et al. 2004). The knockdown of SgIII using siRNA in corticotroph-derived AtT20 cells severely impairs the intracellular retention of CgA; it causes an anomalous localization of barely detectable levels of CgA within the vacuoles in the TGN (Sun et al. 2013). These results suggest that SgIII functions as a sorting receptor for peptide hormones during SG formation and maturation.
The expression of SgIII has been confirmed in vertebrates (fishes, amphibians, reptiles, birds, and mammals), and appears to be correlated with the phylogeny of the regulated secretory pathway in their endocrine systems. Although there are some data on the types of SgIII-expressing endocrine cells in experimental rodents, the expression and systemic distribution of SgIII in these cells in species other than mammals is unclear. In the present study, we identified SgIII-expressing cells in chickens (Gallus gallus domesticus) using immunohistochemistry to examine the various cell types in endocrine glands and the gastrointestinal tract. We compared the expression profiles of SgIII in chickens and rats. We also examined the interaction between SgIII and CgA, and the expression of SG-associated proteins in chicken endocrine tissues.
Materials & Methods
Animals
Six male and three female White Leghorn chickens (L–M strain; G. gallus domesticus), aged 14 months, were purchased from Nissei Bio, Co., Ltd. (Hokkaido, Japan). Four Wistar male rats, aged 8 weeks, were purchased from CLEA Japan, Inc. (Tokyo, Japan), and six C3H/He male mice aged 12 weeks were purchased from Japan SLC, Inc. (Hamamatsu, Japan). After euthanasia by carbon dioxide inhalation, the chickens were processed for sample preparation. The rats and mice were deeply anesthetized by an intraperitoneal injection of sodium pentobarbital (0.1 mg/g body weight). All animal experiments were performed in accordance with the Guidelines for the Care and Use of Laboratory Animals at the Institute of Experimental Animal Science, College of Bioresource Sciences, Nihon University, and the Guidelines for Animal Care, the Regulation of Infectious Agents and Experimental Committee, Gunma University.
Primary Antibodies
The rabbit polyclonal anti-SgIII antibody (raised against rat SgIII peptides 373–471) used in this study was prepared as described previously (Hosaka et al. 2004). The antibody was diluted (1:1000) and then used for immunohistochemistry and immunoblotting. Other antibodies against SG-associated proteins and hormones used in the present study are listed in Table 1. We confirmed (by analysis of amino acid sequence homology of immunogens or checking the information provided by the manufacturers) that all of the listed antibodies react with chicken proteins and hormones. The normal species-specific IgGs used for immunohistochemical control staining were as follows: rabbit IgG (catalog no. sc-2027), mouse IgG (sc-2025), rat IgG (sc-2026), or guinea pig IgG (sc-2711) (Santa Cruz Biotechnology, Inc.; Dallas, TX).
Antibodies used against secretory granule-associated proteins and hormones.
Purchased from Santa Cruz Biotechnology, Inc. (Dallas, TX); LifeSpan BioSciences, Inc. (Seattle, WA, USA); Proteintech Group, Inc. (Chicago, IL); Sigma-Aldrich, Inc. (St. Louis, MO); Abcam Plc (Cambridge, UK); Millipore/Chemicon (Temecula, CA); Enzo Life Sciences International, Inc. (Plymouth Meeting, PA); BioPorto Diagnostics (Gentofte, Denmark); and Linco Research, Inc. (St. Charles, MO). bChromogranin A; cCarboxypeptidase E; dGastric inhibitory polypeptide (also known as glucose-dependent insulinotropic peptide); eGlucagon-like peptide-1.
Immunoblotting
For immunoblotting, three male chickens, two male rats, and two male mice were used. Endocrine glands from the chickens (pituitary, pineal, thyroid, parathyroid, ultimobranchial, and adrenal glands) and the brains from each animal were frozen on dry ice and stored at -80°C until use. Tissue sample preparation for SDS-PAGE and immunoblotting was performed as described previously (Gomi et al. 2007). The endocrine glands from rodents were used as positive controls. Pancreatic islets were isolated from mice as described previously (Gomi et al. 2005). Each protein extract was normalized for total protein content (20 µg for chicken tissues, 10 µg for rat tissues, and 5 µg for mouse tissues) and subjected to SDS-PAGE and immunoblotting using anti-SgIII antibodies. For the analysis of SG-associated proteins, primary antibodies against CgA, CPE, V-ATPase, and Rab27a were used (Table 1). We used the following peroxidase-conjugated secondary antibodies: anti-rabbit IgG (catalog no. 111-035-144), anti-mouse IgG (115-035-146), or anti-goat IgG (705-035-147) (1:5000 dilution; Jackson ImmunoResearch Laboratory, West Grove, PA). The Signal Enhancer Hikari reagent (Nacalai Tesque, Inc.; Kyoto, Japan) was used to enhance the signals in immunoblots for CPE and V-ATPase according to the manufacturer’s instructions.
Immunoprecipitation
For co-immunoprecipitation analysis, chicken pituitary and adrenal glands were homogenized in 50 mM MES (pH 5.5) containing 150 mM NaCl, 10 mM CaCl2, 1% Triton X-100, and 1× protease inhibitor cocktail (cOmplete Mini, Roche Diagnostics, Mannheim, Germany). The homogenates were kept on ice for 20 min and then centrifuged at 20,000 ×g for 10 min. The supernatants were collected, and the protein concentrations determined using a CBB protein assay reagent (Nacalai Tesque). The soluble extracts (1 mg of total protein) were used for immunoprecipitation with 5 µl of anti-SgIII antibody or normal rabbit IgG (1.2 µg, Santa Cruz Biotechnology, sc-2027), as described previously (Han et al. 2008). One-third of the precipitated immunocomplexes were separated using SDS-PAGE, and the interaction between SgIII with CgA was analyzed by immunoblotting using anti-CgA antibodies.
Immunohistochemistry
Chicken endocrine tissues were collected and fixed in Bouin’s fixative for 3 days. Tissue blocks were then dehydrated and embedded in paraffin. Five-micrometer-thick tissue sections were mounted on silane-coated glass slides and processed for immunohistochemistry. The sections were deparaffinized in xylene and then rehydrated in a graded series of ethanol and water solutions. Hematoxylin and eosin (H&E) staining was performed to observe the gross histological features of the tissues. For SgIII immunohistochemistry, deparaffinized sections were immersed in 0.01 M sodium citrate buffer (pH 6.0) and processed using microwave antigen retrieval, as described previously (Gomi et al. 2013). Then, the sections were treated with Dako REAL Peroxidase-Blocking Solution (Dako; Glostrup, Denmark) and blocked with 5% normal goat serum (Vector Laboratories; Burlingame, CA) in PBS. After incubation with primary antibodies overnight at 4°C, the sections were rinsed in PBS and then incubated with secondary antibodies using the HRP-labeled polymer technique with an EnVision Dual Link System–HRP kit (Dako). The sections were then visualized using a peroxidase substrate kit (SK-4100, Vector Laboratories) and counterstained with Mayer’s hematoxylin. To verify the cross-reactivity of the anti-SgIII antibody against chicken tissues, the diluted antibody was incubated with the corresponding antigen (GST-fused rat SgIII peptides 373–471, concentration 5 µg/ml) at 4°C for 12 hr, and the chicken tissue sections were immunostained as described previously (Sakai et al. 2003). To obtain negative controls, the sections were incubated with the mixture of the diluted anti-SgIII antibodies that were pre-treated with absorbing SgIII peptides 373–471 (concentration 5 µg/ml) and normal rabbit IgG (concentration 1 µg/ml) instead of the primary antibodies. Images were acquired using a BX51 microscope equipped with a DP72 CCD camera (Olympus; Tokyo, Japan) and also digitized with the DP2-BSW software.
Double Fluorescence Immunohistochemistry
To analyze the co-expression of SgIII and gut hormones in chicken digestive organs, double immunofluorescence staining was performed as described previously (Gomi et al. 2013). The sections were first incubated with a mixture of primary antibodies and then with a secondary antibody combination of Alexa 555-conjugated anti-rabbit IgG and Alexa 488-conjugated anti-mouse, anti-rat, or anti-guinea pig IgG (1:2000 dilution; Invitrogen, Carlsbad, CA). After washing in PBS, the sections were counterstained with Hoechst 33342 (Nacalai Tesque). To analyze the co-expression of SgIII and CgA in the adrenal gland, a secondary antibody combination of Alexa 488-conjugated anti-rabbit IgG and Alexa 555-conjugated anti-goat IgG was used. For negative controls, the sections were incubated with the mixture of absorbed anti-SgIII antibodies and normal animal IgGs corresponding to the animal species of anti-hormone antibodies. The fluorescence images were acquired using a Zeiss Axio Imager M1 equipped with an AxioCam MRm CCD camera (Carl Zeiss Meditec, Inc.; Dublin, CA). Images were digitized using the Zeiss Axiovision software.
Results
Comparison of SgIII Expression in Chicken, Rat and Mouse
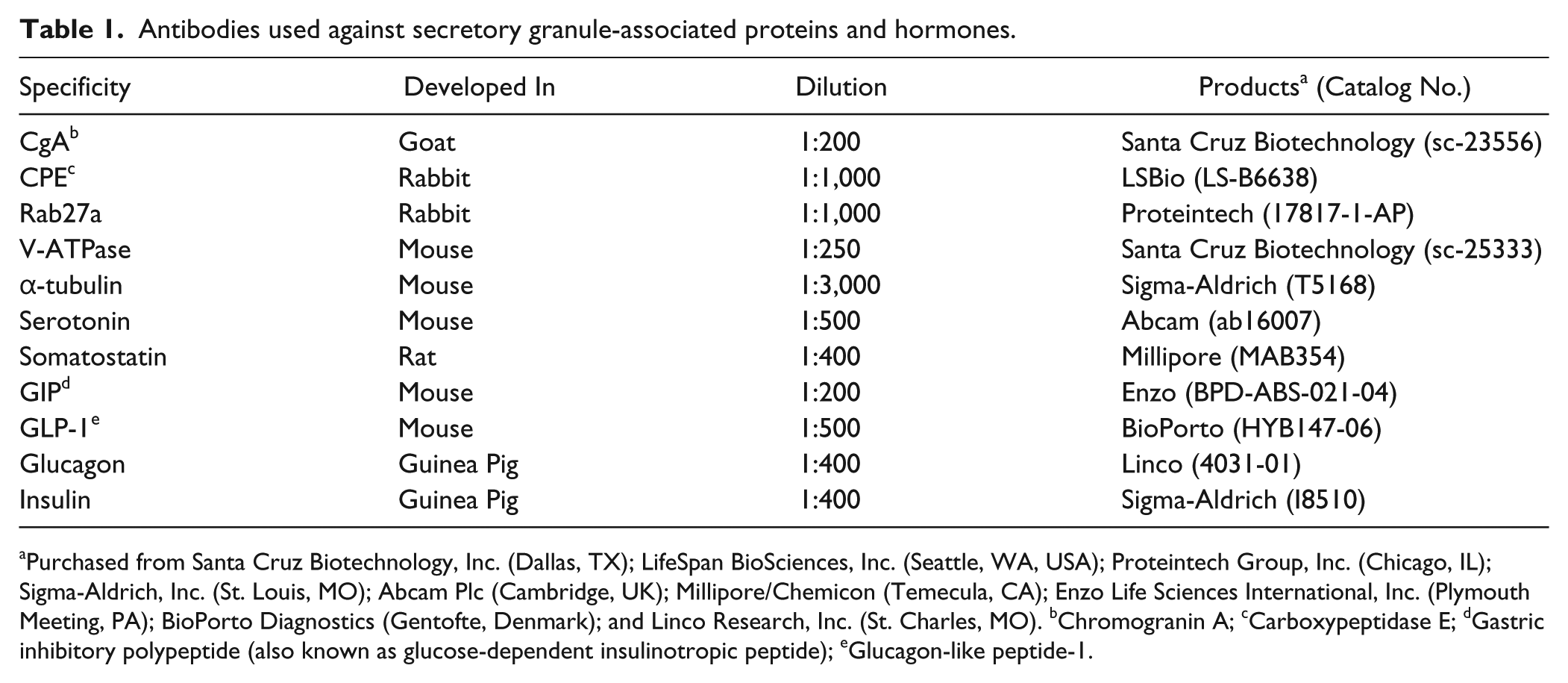
We first compared the amino acid sequences of the SgIII protein in chicken, rat and mouse (Supplementary Fig. 1). The chicken protein consisted of 470 amino acids and had 67.9% homology to rat SgIII, whereas mouse protein had 95.8% homology. The following three major functional domains were identified in rat SgIII (Hosaka and Watanabe 2010): cholesterol-binding domain (amino acids 40–186), CgA-binding domain (214–373), and CPE-binding domain (374–471) had 75.5%, 62.1%, and 75.5% homology to the corresponding domains of chicken SgIII, respectively. We then examined the expression of SgIII in chicken, rat and mouse nervous and endocrine tissues by immunoblotting using anti-SgIII (rat SgIII 373–471) antibodies. A comparison of the C-terminal amino acid sequences of SgIII in these animals is shown in Figure 1A. Although 98 of the 99 amino acid residues were identical in rat and mouse (98.9% identity), only 75 of the 99 residues were identical in chicken and rat (75.7% identity). Nevertheless, immunoblot and immunohistochemical analyses revealed that the anti-SgIII antibody cross-reacted with chicken SgIII (Fig. 1B, 1C). Immunoblotting revealed strong expression of SgIII in the mouse pituitary gland and pancreatic islets and rat pituitary gland. In chickens, relatively strong expression was observed in the pituitary and adrenal glands. The immunoreactive signals were detected as several bands that were considered as the precursor and proteolytically processed forms of SgIII (Sakai et al. 2004; Paco et al. 2010). The overall expression pattern of SgIII in chicken endocrine tissues was similar to that in the rat, except that no expression was observed in the thyroid gland. Equivalent SgIII signals were observed in the rat thyroid gland and chicken ultimobranchial glands, the lower vertebrate-specific extrathyroidal endocrine tissue that gives rise to the calcitonin-producing C cells (Fig. 1B). The cross-reactivity of anti-SgIII in chicken endocrine tissues was immunohistochemically confirmed by antibody adsorption by the immunogen; the immunoreactive signal in the pituitary and adrenal sections disappeared (Fig. 1C).
SgIII expression in chickens, rats, and mice. (A) Comparison of the C-terminal amino acid sequences of SgIII corresponding to the immunogen used to generate the anti-SgIII (rat amino acids 373–471) antibody in rats, mice and chickens. Vertical bars indicate identical amino acids. (B) Immunoblot analysis using anti-SgIII antibodies in the brain and endocrine glands. Rat (Rt; 10 µg of protein), mouse (Mo; 5 µg of protein), and chicken (Ch; 20 µg of protein) tissue extracts were analyzed using SDS-PAGE (10% polyacrylamide gel) and immunoblotting. Small arrows in the upper panels indicate the immunoreactive bands (labeled by anti-SgIII antibodies) corresponding to the precursor and proteolytically processed forms of SgIII. The lower panels show the control α-tubulin immunoblots. (C) Cross-reactivity of the anti-SgIII antibodies in chicken endocrine tissues. Chicken pituitary and adrenal sections were incubated either with anti-SgIII antibody adsorbed on the purified SgIII (rat amino acids 373–471)/GST fusion protein (5 µg/ml) as an immunogen (right panel) or without immunogen adsorption (left panel). Abbreviations: aa, amino acids; gl, gland. Scale, 40 µm.
Expression of SG-associated Proteins and Protein–Protein Interactions between SgIII and CgA in Chicken Endocrine Tissues
Next, we examined the expression of SG-associated proteins in chicken endocrine and cerebral tissues. In addition to CgA, we analyzed the expression of CPE, V-ATPase (a proton-pump, acidifying intracellular organelles such as secretory vesicles and SGs; Schoonderwoert et al. 2000) and Rab27a (involved in the transport of secretory vesicles; Tolmachova et al. 2004) in the secretory cells. Equal amounts of protein extract (20 µg) from different tissues were analyzed by immunoblotting using antibodies against CgA, CPE, V-ATPase, and Rab27a (Fig. 2). The expression levels of CPE, V-ATPase (localized in the intermembrane space of SGs), and Rab27a (localized on the outer membrane of SGs) were similar in each endocrine tissue. However, CgA was highly expressed in the pituitary and adrenal glands, and its expression profile closely resembled that of SgIII. In addition, the expression of these SG-associated proteins could not be detected in the thyroid gland. Because SgIII binds specifically to CgA (Hosaka et al. 2002), we next examined the interaction between these proteins in chicken tissues. Co-immunoprecipitation analysis using anti-SgIII antibodies and tissue extracts prepared from the chicken pituitary and adrenal glands confirmed the binding of CgA to SgIII (Fig. 3A, 3B). Double fluorescence immunohistochemistry revealed that the levels of co-expression of SgIII and CgA varied in different SgIII-expressing tissues (Fig. 3C). In the pituitary gland, some of the SgIII-positive cells co-expressed CgA. In the adrenal medullary parts and pancreatic islets, all the cells expressing SgIII also expressed CgA. These results suggest that CgA interacts with SgIII in chicken endocrine glands in vivo. Our results are consistent with the reports of biochemical studies in cultured cell lines (Hosaka et al. 2002, 2005; Han et al. 2008) and immunocytochemical analysis of rat pituitary and islet cells (Sakai et al. 2003, 2004).
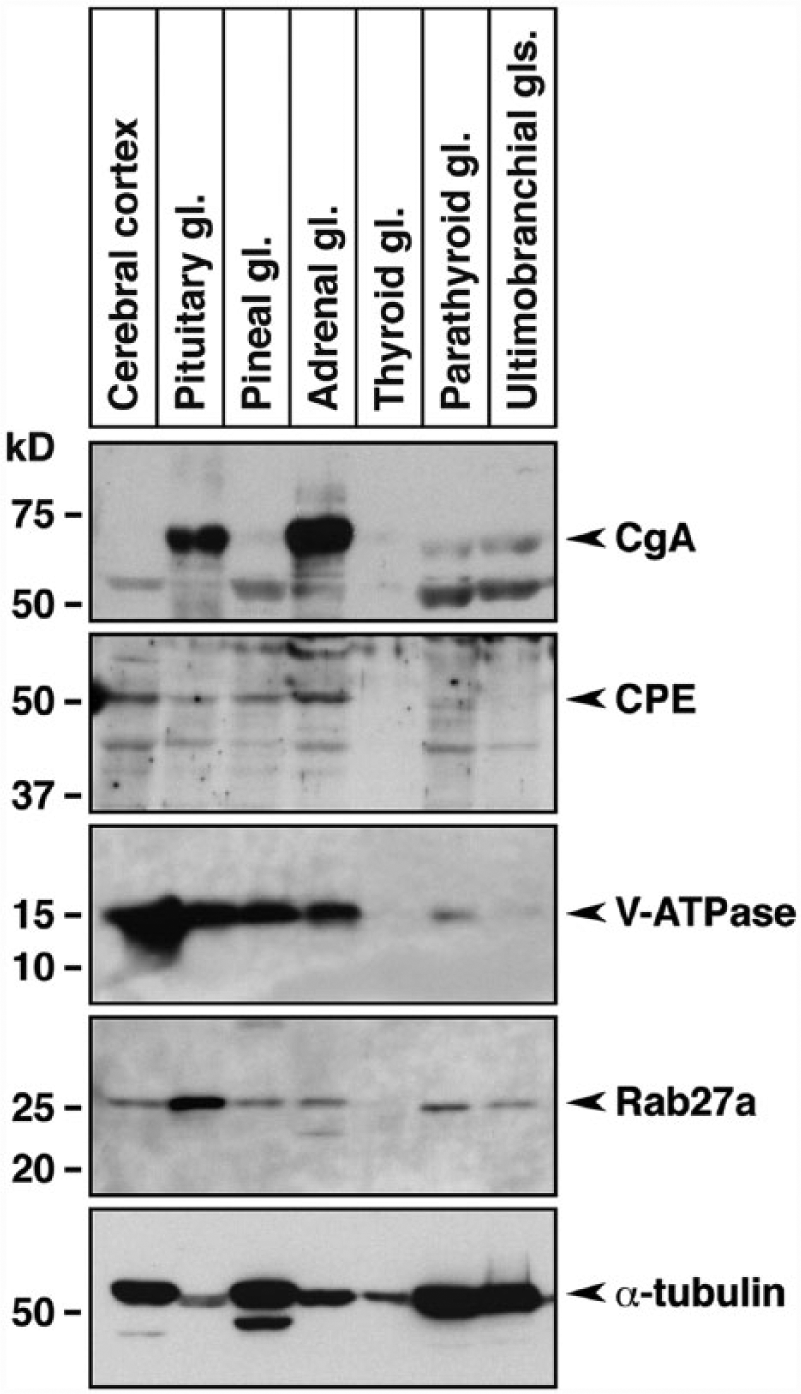
Expression of secretory granule-associated proteins in chicken endocrine tissues. Equal amounts of protein extracts (20 µg) from the indicated tissues were subjected to SDS-PAGE, followed by immunoblotting using antibodies against CgA, CPE, V-ATPase and Rab27a. The bottom panel shows the control α-tubulin immunoblots. Abbreviation: gl, gland.
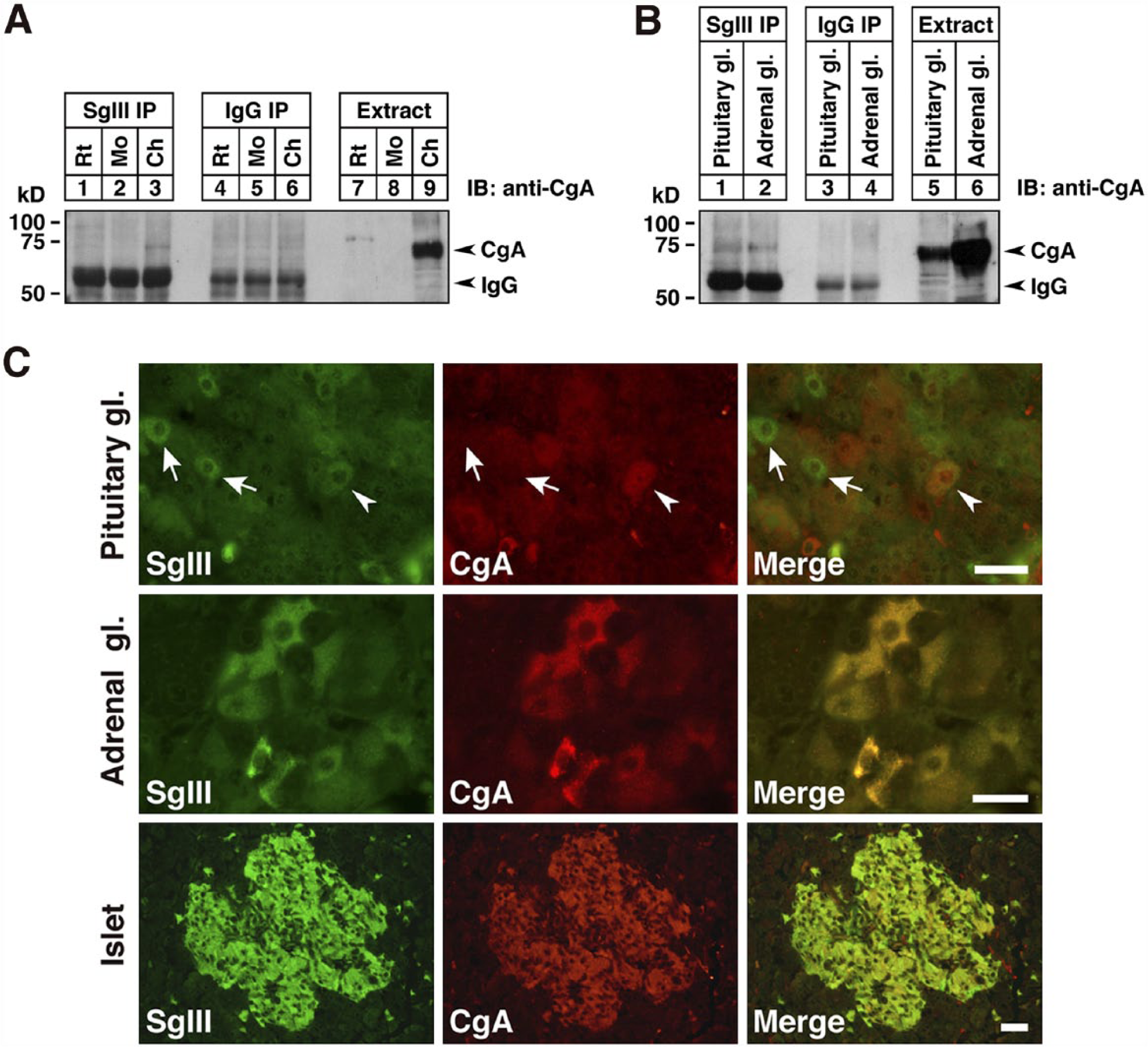
Co-expression and interaction of SgIII and CgA proteins in chicken endocrine tissues. (A) Co-immunoprecipitation of SgIII and CgA in extracts of the pituitary gland (1 mg of protein) from rat, mouse and chicken. Immunoprecipitates with anti-SgIII antibodies (lanes 1, 2, and 3), control rabbit IgG (lanes 4, 5, and 6), and the starting tissue extracts (15 µg; lanes 7, 8, and 9) were analyzed by immunoblotting using goat polyclonal antibodies specific for chicken CgA. (B) Co-immunoprecipitation of SgIII and CgA in the chicken pituitary and adrenal glands. Tissue extracts were immunoprecipitated using anti-SgIII antibodies (lanes 1 and 2), control rabbit IgG (lanes 3 and 4), and the starting tissue extracts (15 µg; lanes 5 and 6). (C) Co-expression of SgIII (green) and CgA (red) in the chicken anterior pituitary lobe, medullary parts of the adrenal gland, and pancreatic islets visualized using double fluorescence immunohistochemistry. Arrowhead indicates double-immunolabeled anterior pituitary cell, and arrows indicate anterior pituitary cells that do not express CgA. Abbreviations: gl, gland; IP, immunoprecipitates. Scale, 20 µm.
Expression of SgIII in Chicken Endocrine Tissues
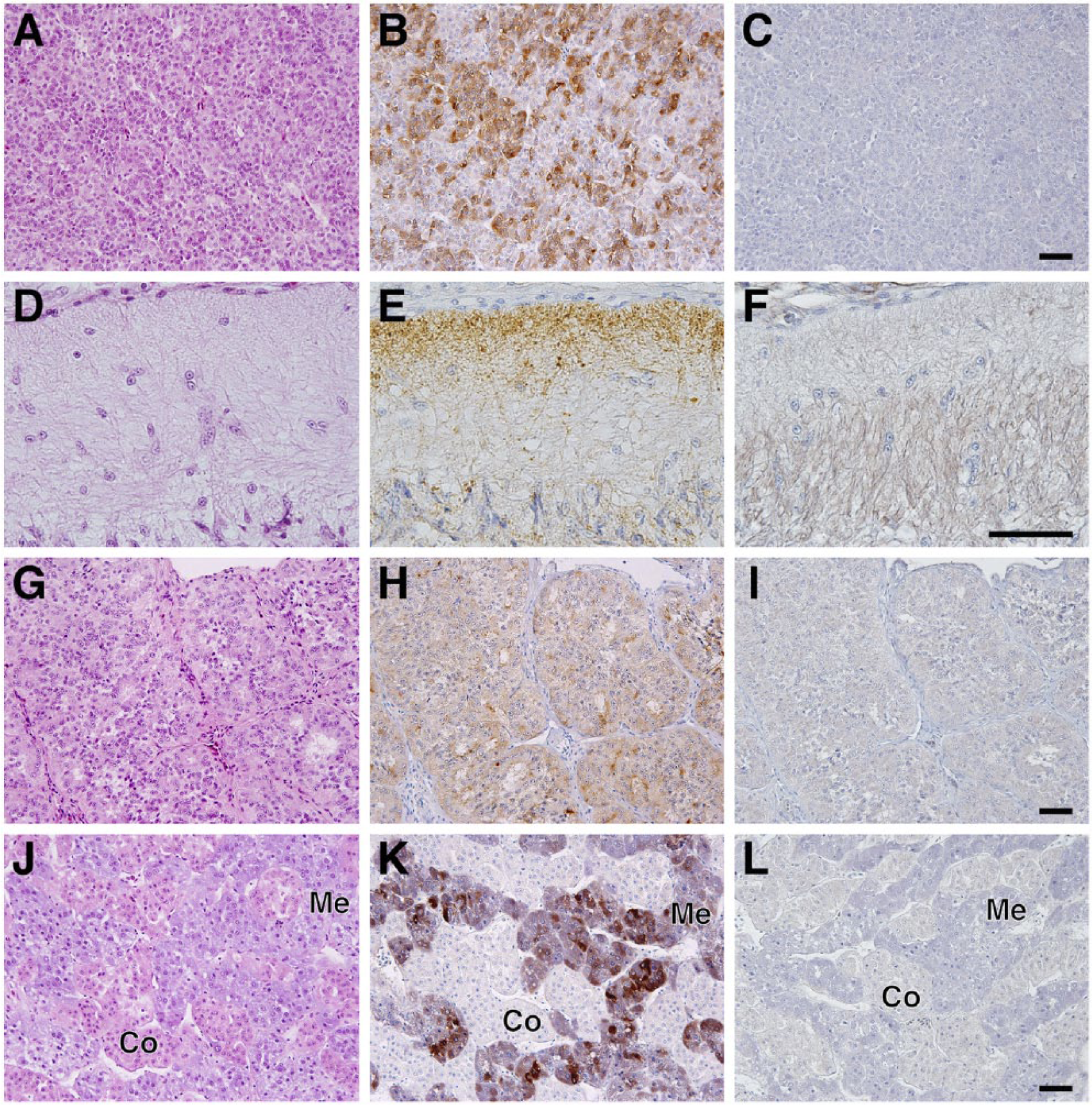
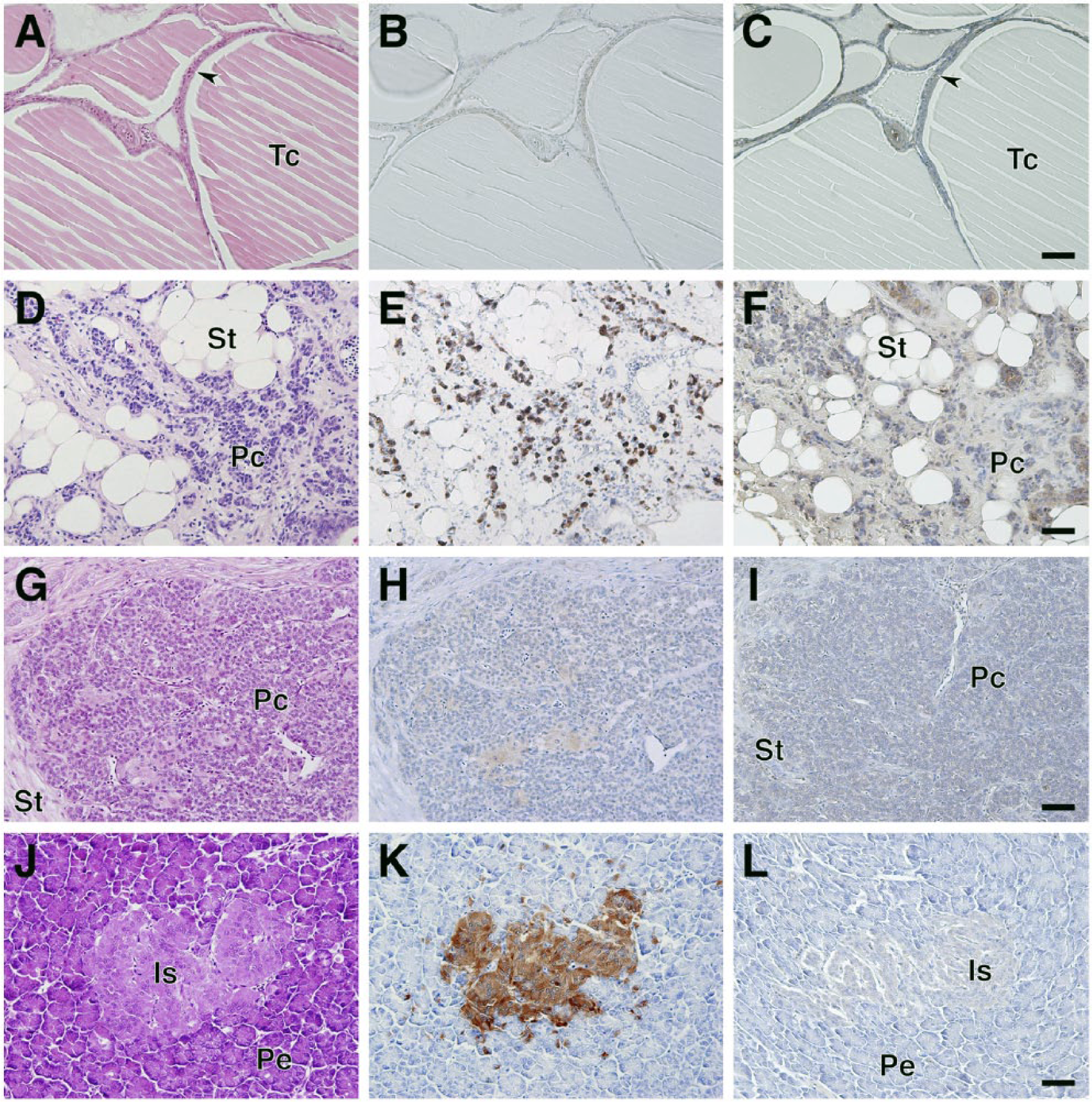
Paraffin-embedded tissue sections were utilized in general histological observations using H&E staining, and antigen retrieval techniques were used for the immunohistochemical analysis of SgIII, as reported previously (Gomi et al. 2013). SgIII expression was first examined in the typical endocrine tissues (pituitary, pineal, adrenal, thyroid, parathyroid, and ultimobranchial glands and pancreatic islets). In the anterior pituitary lobe, the basophilic endocrine cells identified by H&E staining expressed SgIII with varying immunoreactive intensities (Fig. 4A–4C). In the posterior pituitary lobe, the terminal region of endocrine neuron processes was immunoreactive for SgIII (Fig. 4D–4F). Relatively uniform immunoreactivity against SgIII was observed in the pineal gland (Fig. 4G–4I). Unlike in mammals, whose adrenal glands consist of two distinct structures (the outer cortex and inner medulla), the intermingled arrangement of cortical and medullary regions is characteristic of the chicken adrenal gland. The immunoreactivity against SgIII was restricted to the medullary regions, which are composed of basophilic endocrine cells (Fig. 4J–4L). In the chicken thyroid gland, no immunoreactivity was observed in follicular cells, which was consistent with the data obtained from immunoblotting (Figs. 1B, 5A–5C). In the chicken, parafollicular cells in the mammalian thyroid gland existed independently as ultimobranchial glands and contained SgIII-immunoreactive cells (Fig. 5D–5F). In the parathyroid gland, very weak immunoreactivity was observed in parathyroid cells (Fig. 5G–5I). Finally, SgIII was expressed in the pancreatic islet cells in chickens (Fig. 5J–5L).
Localization of SgIII in the chicken pituitary, pineal, and adrenal glands. Sections of the anterior (A–C) and posterior (D–F) pituitary lobes, pineal gland (G–I), and adrenal gland (J–L) were stained with H&E (A, D, G, and J) and immunostained with anti-SgIII antibodies (B, E, H, and K). Sections incubated with a mixture of the absorbed anti-SgIII antibodies and normal rabbit IgG were used as negative controls (C, F, I, and L). Abbreviations: Co, cortical parts of the adrenal gland; Me, medullary parts of the adrenal gland. Scale, 50 µm.
Localization of SgIII in the chicken thyroid, ultimobranchial, and parathyroid glands and pancreatic islets. Sections of thyroid gland (A–C), ultimobranchial glands (D–F), parathyroid gland (G–I), and pancreatic islets (J–L) stained with H&E (A, D, G, and J) and immunostained with anti-SgIII antibodies (B, E, H, and K). Negative control sections (C, F, I, and L). Abbreviations: Tc, colloid of the thyroid; St, stroma; Pc, parenchyma; Is, pancreatic islets; Pe, exocrine parts of the pancreas. Arrowheads indicate thyroid follicular cells. Scale, 50 µm.
Expression of SgIII in the Chicken Gastrointestinal Tract
The gastrointestinal tract contains endocrine cells that secrete more than a dozen different gut hormones with various physiological effects in the digestive system. The chicken gastrointestinal tract is composed of several unique structures. We analyzed SgIII expression in the chicken esophagus (crop), stomach (proventriculus and gizzard), and small and large intestines. No immunoreactive cells were observed in the crop (data not shown). The proventriculus (glandular stomach) and gizzard (muscular stomach) are equivalent to the mammalian proper gastric gland and pyloric part, respectively. SgIII-immunoreactive cells in these tissues were sparsely distributed in the superficial and deep proventricular glands (Fig. 6A–6C), as well as in the ventricular gizzard glands (Fig. 6D–6F). In the small intestine, SgIII-positive cells were localized in the intestinal villi and crypts of the duodenum (Fig. 6G–6I), jejunum (data not shown), and ileum (Fig. 6J–6L). Furthermore, SgIII-positive cells were found in the intestinal villi and crypts of the cecum (Fig. 6M–6O) and rectum (data not shown).
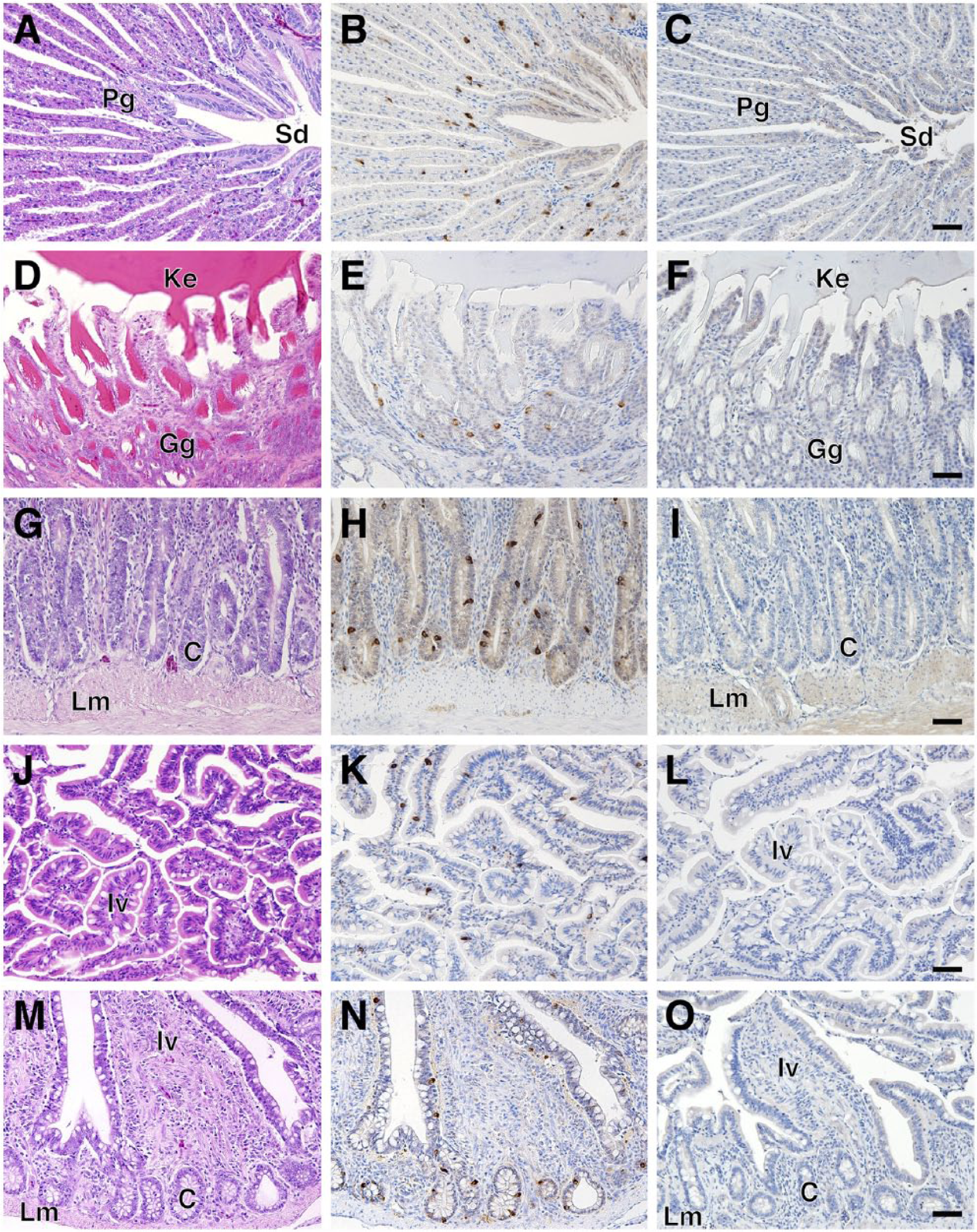
Localization of SgIII in the chicken alimentary canal. Sections of the proventriculus (A–C), gizzard (D–F), duodenum (G–I), ileum (J–L), and cecum (M–O) stained with H&E (A, D, G, J, and M) and immunostained with anti-SgIII antibodies (B, E, H, K, and N). Negative control sections (C, F, I, L, and O). Abbreviations: Pg, deep proventricular glands; Sd, secretory duct; Ke, keratinous substances; Gg, gizzard’s glands; C, crypt (intestinal gland); Lm, lamina muscularis; Iv, intestinal villus. Arrowheads indicate thyroid follicular cells. Scale, 50 µm.
Identification of the Cell Types Expressing SgIII in the Chicken Gastrointestinal Tract and Pancreatic Islets
The types of gastrointestinal endocrine cells (GECs) and pancreatic islet cells displaying SgIII immunoreactivity were identified by double immunofluorescence staining with antibodies against gut hormones [glucagon, insulin, somatostatin, glucagon-like peptide-1 (GLP-1), gastric inhibitory polypeptide (GIP), and serotonin] (Table 1). A summary of the co-expression of gut hormones and SgIII is shown in Table 2. In the proventriculus, GECs expressing glucagon, somatostatin, GLP-1, GIP, and serotonin co-expressed SgIII in the superficial proventricular glands (Fig. 7). The same result was obtained in the deep proventricular glands (data not shown). In contrast, only serotonin was co-expressed with SgIII in GECs in the gizzard (Fig. 8A–8C). In the duodenum, a similar distribution pattern of SgIII-immunolabeled GECs in the proventriculus was observed (Fig. 9). However, some serotonin-expressing cells did not co-express SgIII (Fig. 9M–9O). In the cecum, only serotonin-expressing cells co-expressed SgIII (Fig. 8D–8F), whereas only GIP-expressing cells, but not serotonin-expressing cells, co-expressed SgIII in the rectum (Fig. 8G–8L). Thus, the pattern of co-expression of serotonin and SgIII in GECs varied in the different regions of the chicken gastrointestinal tract.
Summary of Co-Expression of Gut Hormones and SgIII.
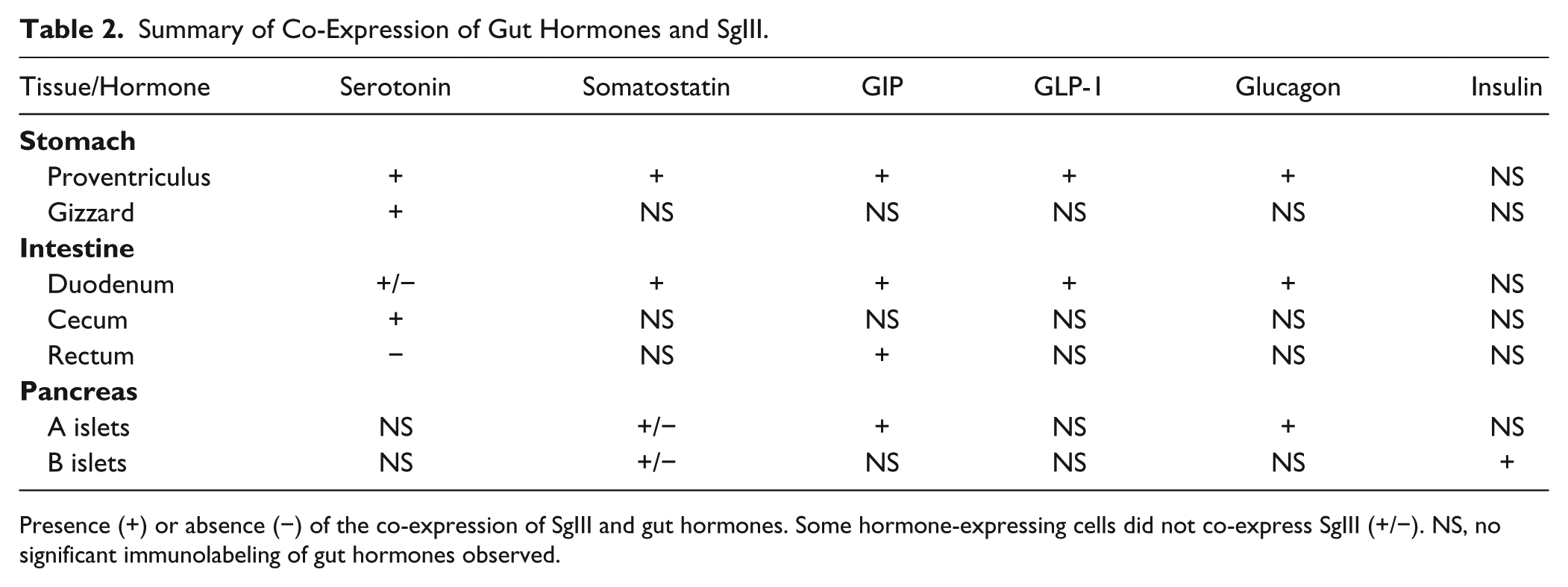
Presence (+) or absence (−) of the co-expression of SgIII and gut hormones. Some hormone-expressing cells did not co-express SgIII (+/−). NS, no significant immunolabeling of gut hormones observed.
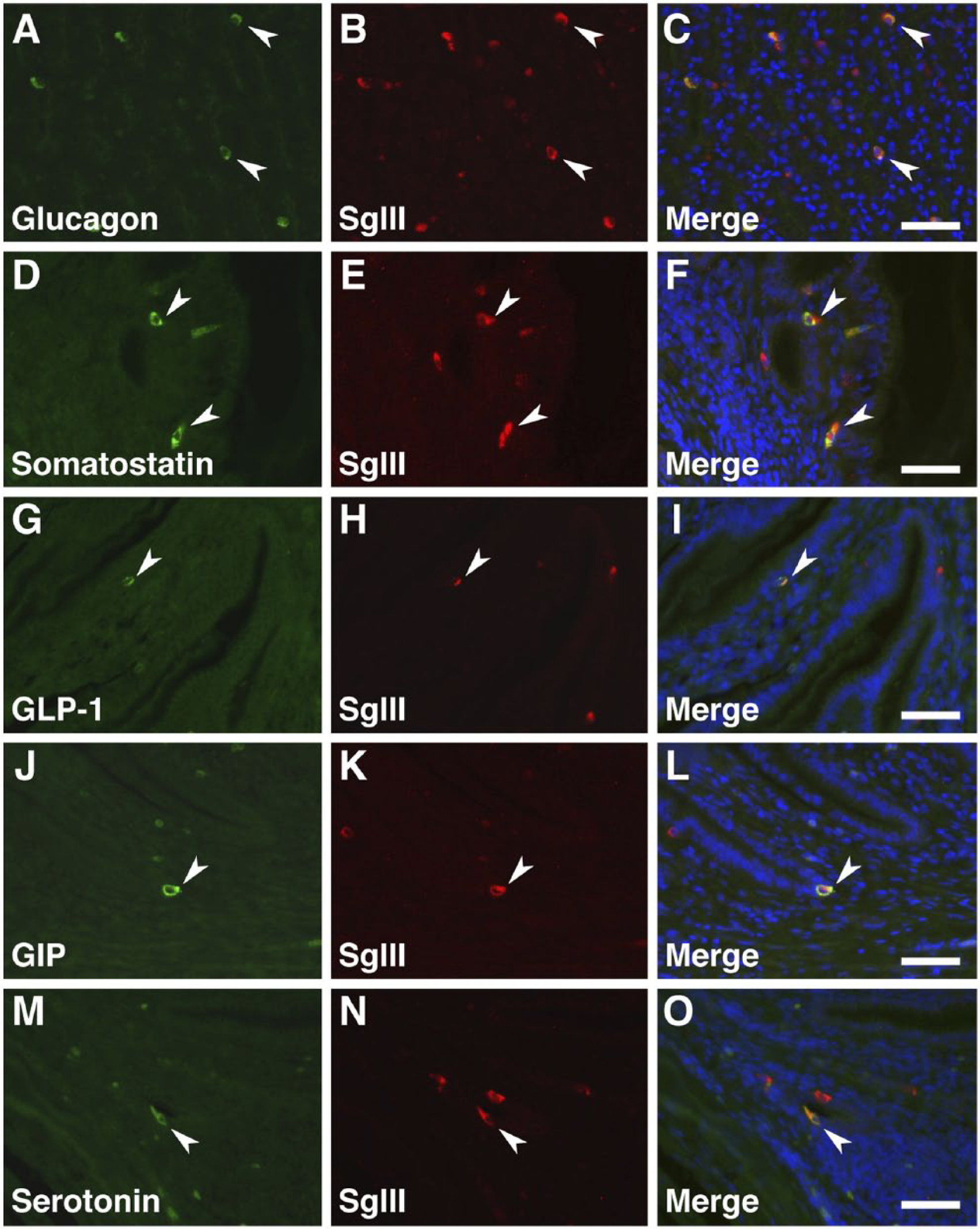
Double immunofluorescence staining for SgIII and gut hormones in the chicken proventriculus (superficial proventricular glands). Sections were incubated with anti-SgIII antibodies and a combination of anti-glucagon (A–C), anti-somatostatin (D–F), anti-GLP-1 (G–I), anti-GIP (J–L), and anti-serotonin (M–O) antibodies. Arrowheads indicate double-immunolabeled GECs. Blue fluorescence reflects nuclear counterstaining. Scale, 50 µm.
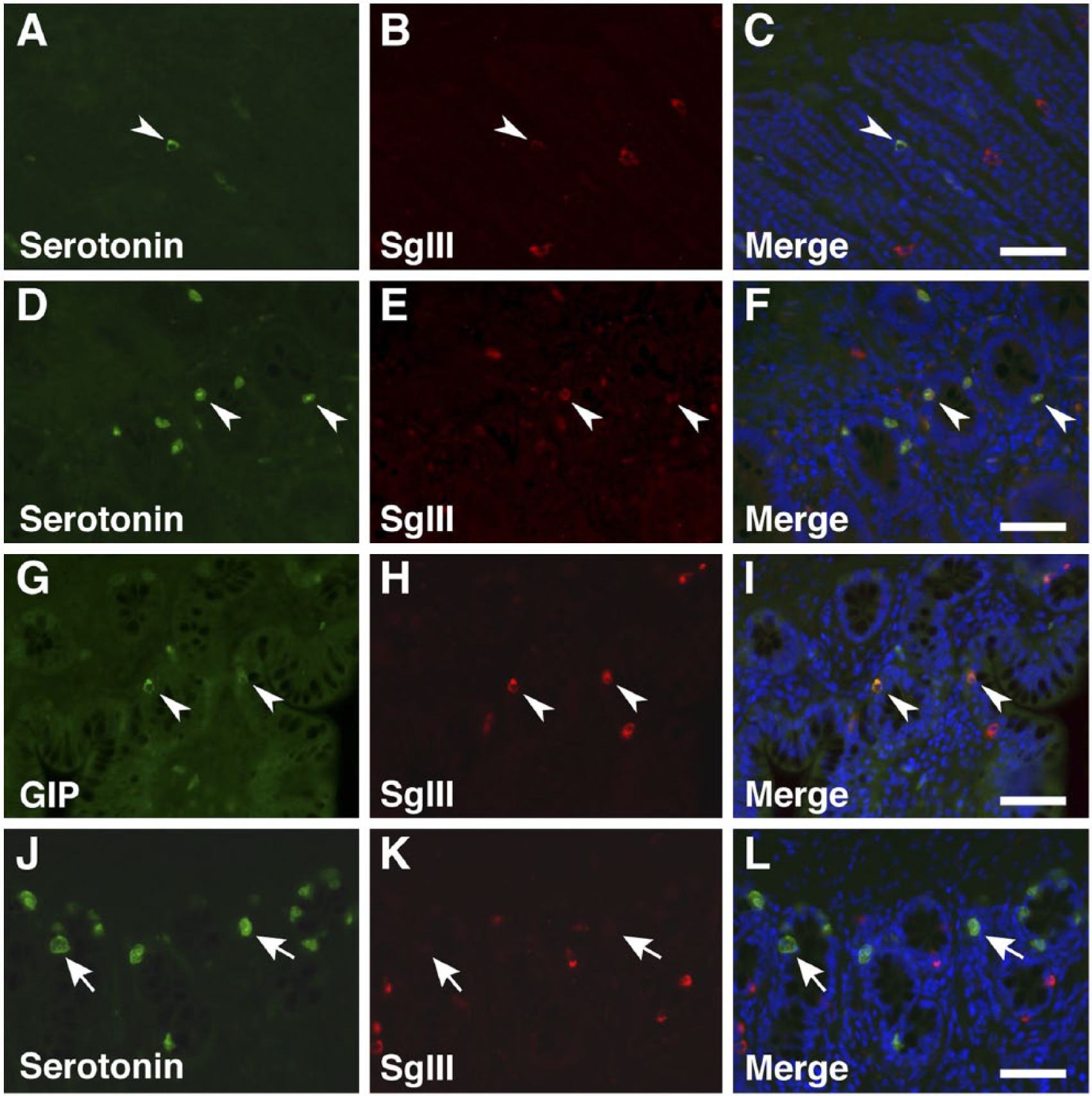
Double immunofluorescence staining for SgIII and gut hormones in the chicken gizzard, cecum, and rectum. Sections were incubated with anti-SgIII antibodies and a combination of anti-serotonin (gizzard, A–C; cecum, D–F; rectum, J–L) and anti-GIP (rectum, G–I) antibodies. Arrowheads indicate double-immunolabeled GECs, and arrows indicate GECs that do not express SgIII. Blue fluorescence reflects nuclear counterstaining. Scale, 50 µm.
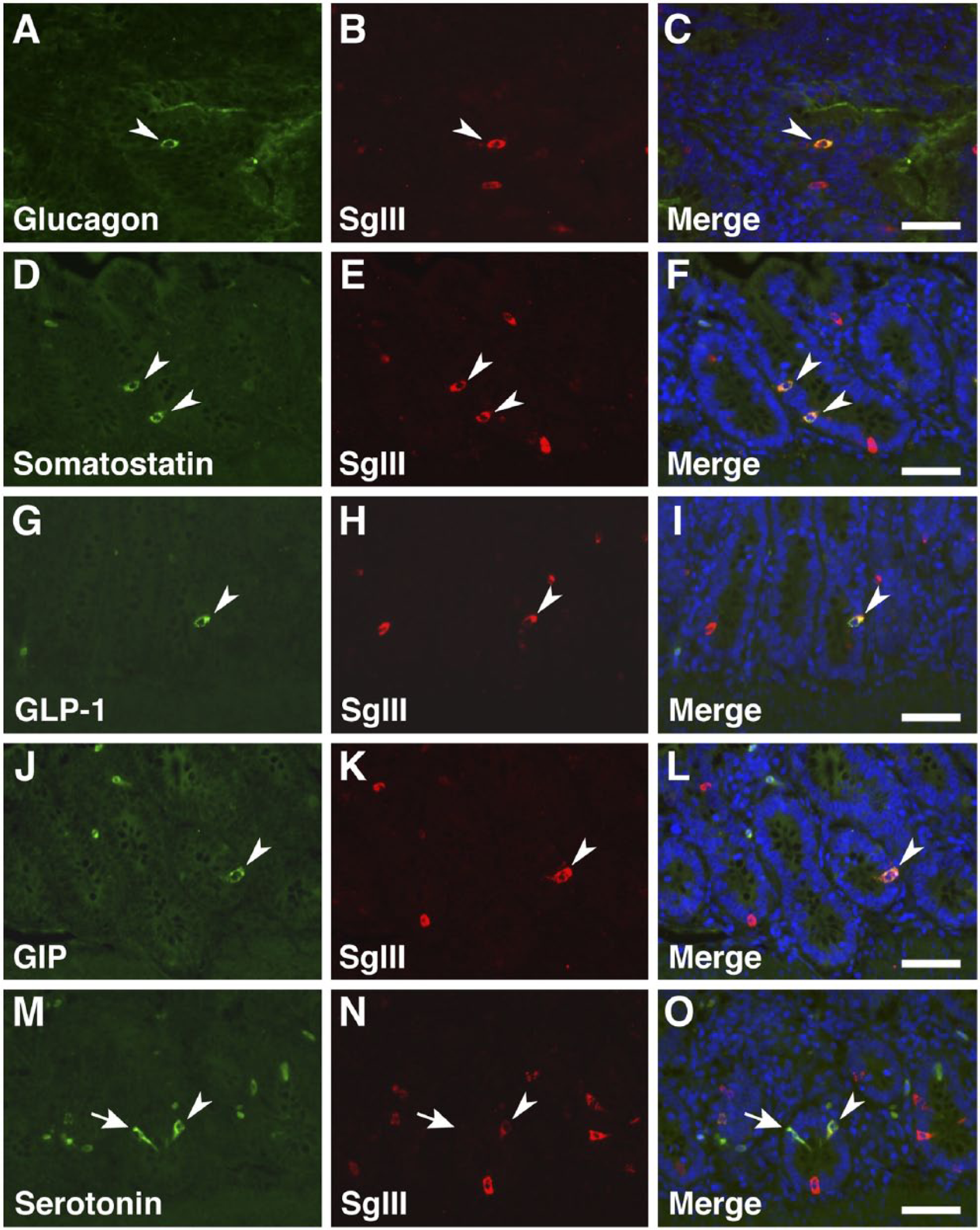
Double immunofluorescence staining for SgIII and gut hormones in the chicken duodenum. Sections were incubated with anti-SgIII antibodies and a combination of anti-glucagon (A–C), anti-somatostatin (D–F), anti-GLP-1 (G–I), anti-GIP (J–L), and anti-serotonin (M–O) antibodies. Arrowheads indicate double-immunolabeled GECs, and arrows indicate GECs that do not express SgIII. Blue fluorescence reflects nuclear counterstaining. Scale, 50 µm.
Previous studies have reported the expression of SgIII in α-, β-, and δ-cells in the pancreatic islets of rats (Sakai et al. 2004) and the human α-, β-, and PP cells, but not in δ-cells (Stridsberg et al. 2008). We examined the cell types expressing SgIII in chicken pancreatic islets. Chicken pancreatic islets contain unique structures, such as large and α-cell-dominant A islets (Fig. 10A), relatively small-sized and β-cell-dominant B islets (Fig. 10D), and a small number of sparsely distributed δ-cells (Fig. 10G). In addition to α-, β-, and δ-cells, there are GIP-positive cells distributed in A islets (Fig. 10J). The co-expression of glucagon and GIP, but not glucagon and GLP-1, was observed in chicken α-cells (data not shown). Pancreatic islet cells expressing glucagon, insulin, somatostatin, and GIP co-expressed SgIII (Fig. 10A–10C, 10D–10F, 10G–10I, 10J–10L). However, some somatostatin-expressing δ-cells did not co-express SgIII (Fig. 10G–10I).

Double immunofluorescence staining for SgIII and pancreatic islet hormones in chickens. Sections were incubated with anti-SgIII antibodies and a combination of anti-glucagon (A–C), anti-insulin (D–F), anti-somatostatin (G–I), and anti-GIP (J–L) antibodies. Arrowheads indicate double-immunolabeled GECs, and arrows indicate GECs that do not express SgIII. Blue fluorescence reflects nuclear counterstaining. Scale, 50 µm.
Discussion
Our previous reports suggest that SgIII functions as a sorting receptor for peptide hormones during the formation and maturation of SGs (Hosaka et al. 2002, 2004, 2005; Sun et al. 2013). During the sorting process, the binding of SgIII to CgA and CPE is a critical step for targeting prohormones to the cholesterol microdomains, which can also bind to SgIII, within the SG membrane of the TGN pathway (Takeuchi and Hosaka 2008). These multifaceted binding properties of SgIII mediate the selective recruitment of prohormones, the processing and modification of prohormones, and the condensation (aggregation) of mature hormones (Hosaka and Watanabe 2010). This function of SgIII is preserved among vertebrate secretory cells; a recent bioinformatics study has confirmed the expression of SgIII in most vertebrate species (searched the NCBI database). However, little is known about cell types and animal species that express SgIII apart from those cells in the brain, pituitary gland, and pancreatic islets of mice, rats and humans (Ottiger et al. 1990; Hosaka et al. 2002; Sakai et al. 2003, 2004; Stridsberg et al. 2008; Paco et al. 2010). In the present study, we used the chicken as the model species and examined the expression and properties of SgIII in standard endocrine glands. We also used immunohistochemistry to examine SgIII expression in different tissues and/or cell types in the endocrine glands and gastrointestinal tract.
Analysis of SG-associated protein expression showed that CgA and SgIII were strongly expressed in the pituitary and adrenal glands. CPE, V-ATPase, and Rab27a were also expressed in the SgIII-positive chicken endocrine glands. These results suggest that the significant expression of SgIII and SG-associated proteins in chicken endocrine glands may be related. As one of the representative examples of the interaction of SgIII and its binding protein, the co-localization and interaction between SgIII and CgA was confirmed in vivo using double fluorescence immunohistochemistry and co-immunoprecipitation. However, in the anterior pituitary cells, some of the SgIII-positive cells expressed CgA, whereas, in the adrenal medullary parts and pancreatic islets, all the SgIII-positive cells expressed CgA. The different patterns of co-expression of SgIII and CgA in the chicken pituitary and islet cells were consistent with our previous observations in rat pituitary and islet cells (Sakai et al. 2003; 2004). In the rat pituitary cells, the SgIII and CgA co-localization is restricted to gonadotrophs; other types of SgIII-expressing cells strongly express CgB (Sakai et al. 2003). When the expression of CgA in gonadotrophs of castrated male rats is suppressed by the continuous administration of estradiol, the large CgA-containing SGs disappear, whereas the smaller SGs, containing SgII, are retained (Watanabe et al. 1998). These results also suggest that there are multiple mechanisms in the sorting of peptide hormones and in the biosynthesis of SGs, other than the mechanism based on the interaction of SgIII and CgA. Indeed, the knockdown of SgIII in pituitary-derived AtT-20 cells has disclosed a latent redundancy in the sorting and biosynthesis of SGs, where another granin protein, SgII, behaves like SgIII (Sun et al. 2013). Conversely, after the knockdown of SgII in PC12 cells, the number of SGs significantly decreases but they are not completely depleted (Courel et al. 2010). Furthermore, we have demonstrated that the interaction between SgIII and CPE facilitates prohormone sorting within SGs in CPE-deficient Cpefat/fat mice, whose expression levels of SgIII and CgA are substantially elevated (Hosaka et al. 2005). These results are consistent with the idea that CPE functions as a sorting receptor in proopiomelanocortin targeting to SGs in the pituitary cells (Cool et al. 1997).
Comparative immunoblotting revealed that both the precursor and processed forms of SgIII were present in chicken tissues, which was consistent with previous observations in Xenopus, rat, and mouse tissues (Holthuis et al. 1996; Sakai et al. 2004; Han et al. 2008; Paco et al. 2010). Furthermore, immunohistochemical staining showed the expression of SgIII in the chicken pineal gland, adrenal medullary tissue, parathyroid gland, ultimobranchial glands, and GECs in the gastrointestinal tract. However, no SgIII expression could be demonstrated in the thyroid gland of chickens using either immunoblotting or immunohistochemistry. This suggests that the immunoreactive signal in the rat thyroid gland observed during immunoblotting originates from parafollicular cells (calcitonin-producing C cells) and not from thyroid hormone-producing follicular cells (unpublished observations). In agreement with this observation, additionally, a small band of the same size was observed in immunoblots from the rat thyroid gland and chicken ultimobranchial glands, which supports for the previous result.
Analysis of the expression of SgIII and gut hormones in the chicken gastrointestinal tract revealed that glucagon, somatostatin, GLP-1, GIP, and serotonin were co-expressed with SgIII in the proventriculus and duodenum. In contrast, the co-expression of SgIII and serotonin in the gizzard and cecum and with GIP in the rectum was limited (Table 2). We noted that the co-expression pattern of SgIII and serotonin in chicken GECs varied among the different regions of the gastrointestinal tract. In mammals, serotonin-expressing enterochromaffin (EC) cells are widely distributed in the stomach and small and large intestines. These cells play an important role in the regulation of secretion, motility, and sensation. In the small intestine, serotonin is secreted in response to chemical and mechanical stimuli; it activates peristalsis, intestinal secretion and vagal afferents, and occasionally causes diarrhea or vomiting as a pathophysiological response (Osaka et al. 1971; Fujita and Kobayashi 1977; Iwanaga et al. 1993). Serotonin secreted in the large intestine regulates the activity of neighboring epithelial cells and goblet cells in the crypts (Kuramoto et al. 2007). The EC cells in the small and large intestines are also classified into subpopulations on the basis of the co-expression of serotonin and physiologically active peptides such as substance P, Met-enkephalin, or sorbin (Pearse and Polak 1975; Sokolski and Lechago 1984; El Fadil et al. 1997). Furthermore, the patterns of co-localization of chromogranins (CgA, CgB, and CgC) and serotonin vary in human gastrointestinal EC cells (Portela-Gomes et al. 1997). Therefore, functional or subtype-related differences in EC cells in the small and large intestines may be related to the different co-expression patterns of SgIII and serotonin in chicken GECs. Interestingly, in the chicken, we found different expression patterns of SgIII and serotonin in the cecum and rectum. In the cecum, EC cells expressed both proteins but the rectal EC cells did not. The exact reason for this difference is unclear. However, the physiology of intestinal motility in the cecum and rectum is different. In the chicken, the digesta stay longer in the cecum than in the rectum and cecal motility is controlled by circadian rhythm (Isshiki and Nakahiro 1975). Differential expression of chromogranins (CgA and CgB) and other secretory proteins (such as substance P, peptide tyrosine-tyrosine, neurotensin, and secretin) in EC cells of the large intestine (Salvi et al. 1996) may be related to the different co-expression patterns of SgIII and serotonin.
In the pancreas, SgIII was clearly co-expressed in glucagon- and GIP-producing α-cells in A islets and insulin-producing β-cells in B islets. Surprisingly, although both GLP-1 and GIP are expressed in α-cells in rat pancreatic islets (Gomi et al. 2013), only GIP-positive cells co-expressed SgIII in chicken pancreatic islets. The chicken islet cells stained positive for insulin, glucagon, somatostatin and GIP but were negative for GLP-1 and serotonin. As in both rat (Sakai et al. 2004) and human (Stridsberg et al. 2008) islets, SgIII expression in somatostatin-producing δ-cells in the chicken islets was either absent or at very low levels. In contrast, somatostatin-producing GECs in the proventriculus and duodenum co-expressed SgIII. It is interesting that two active somatostatin peptides, SS-28 and SS-14 (generated by the proteolytic processing of prosomatostatin), are present in mammals and chickens and that different peptide fragments are found in gastric or intestinal D-cells and pancreatic δ-cells (Ravazzola et al. 1983; Francis et al. 1990; Takayanagi et al. 1996). The differences in the proteolytic processing of prosomatostatin may be related to the expression intensity of SgIII in the gastrointestinal D-cells (strong) and pancreatic δ-cells (weak or absent) in chickens. However, no suitable antibody against chicken pancreatic polypeptide for double immunofluorescence staining with anti-SgIII antibody is available. Further studies should be undertaken to examine the expression of SgIII in PP cells in chicken islets.
We were unable to identify all the SgIII-expressing cell types in the gastrointestinal tract because more than a dozen different gut hormones are secreted from various GEC subsets. Further studies are needed to classify the cells that express SgIII. Nevertheless, our study revealed that SgIII is expressed in endocrine glands and GECs that secrete bioactive amines or peptide hormones via the proteolytic processing and terminal modification of prohormones. Therefore, the expression of SgIII may be related to the specific modification and proteolytic processing of prohormones (Bataille 2007). This assumption is also supported by the lack of SgIII expression in endocrine cells in the thyroid gland or in the cortical parts of the adrenal gland that lacks these properties.
We observed different SgIII expression levels in various types of endocrine cells. These quantitative differences were probably caused by varying expression levels of SgIII-interacting molecules, such as CgA, CgB, CPE and SgII (Sakai et al. 2003; Hosaka et al. 2005; Hotta et al. 2009). The expression levels of granin proteins in the pituitary endocrine cells are affected by hormonal treatment (Scammell et al. 1986; Anouar and Duval 1992; Thompson et al. 1992; Watanabe et al. 1998). The expression of molecules belonging to the family of prohormone convertases may also affect the expression level of SgIII. These possibilities should be resolved in future studies.
In summary, we performed an extensive protein expression analysis in the chicken endocrine tissues. We demonstrated that SgIII was expressed systemically in endocrine cells, in the SGs that contain and secrete peptide hormones (via the intragranular proteolytic processing of prohormones) and physiologically active amines (catecholamines, serotonin, and melatonin). This study is the first to describe an avian SgIII expression profile and provides useful information about granin-family proteins in these animals.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Grants-in-Aid from the
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
