Abstract
Gustducin is a guanosine nucleotide-binding protein functionally coupled with taste receptors and thus originally identified in taste cells of the tongue. Recently, bitter taste receptors and gustducin have been detected in the airways, digestive tracts and brain. The existing studies showing taste receptors and gustducin in the brain were carried out exclusively on frozen sections. In order to avoid the technical shortcomings associated with frozen sectioning, we performed immunofluorescence staining using vibratome-cut sections from mouse brains. Using a rabbit gustducin antibody, we could not detect neurons or astrocytes as reported previously. Rather, we found dense fibers in the nucleus accumbens and periventricular areas. We assumed these staining patterns to be specific after confirmation with conventional negative control staining. For the verification of this finding, we stained gustducin knockout mouse brain and tongue sections with the same rabbit gustducin antibody. Whereas negative staining was confirmed in the tongue, intensive fibers were constantly stained in the brain. Moreover, immunostaining with a goat gustducin antibody could not demonstrate the fibers in the brain tissue. The present study implies a cross immunoreaction that occurs with the rabbit gustducin antibody in mouse brain samples, suggesting that the conventional negative controls may not be sufficient when an immunostaining pattern is to be verified.
Introduction
The guanosine nucleotide-binding protein, gustducin (McLaughlin et al. 1992), was originally identified in taste cells of the tongue (Wong et al. 1996; Wong et al. 1999), and has therefore been functionally coupled with taste receptors. Bitter taste receptors and/or gustducin-expressing cells have been recently identified in tissues from airway (Tizzano et al. 2010; Braun et al. 2011; Barham et al. 2013) and digestive tract (Rozengurt 2006; Mazzoni et al. 2003). It has been suggested that gustducin and its coupled bitter taste receptors may be related to defensive functions against microbes, as airway bitter taste receptors can be activated by quorum-sensing molecules (Sbarbati et al. 2009; Tizzano et al. 2010; Lee et al. 2012; Lee et al. 2014) secreted by gram-negative bacteria (Conway et al. 2002; Frommberger et al. 2003).
Recently, taste receptors and gustducin were also detected in astrocytes or the cell bodies of neurons in various brain areas from the mouse (Ren et al. 2009) and rat (Shin et al. 2010; Singh et al. 2011; Dehkordi et al. 2012). It has also been reported that gustducin is expressed in the axon terminals of the rod bipolar cells in the mouse, rat and rabbit retina (Son et al. 2011). Given that some bitter ligands such as quinine (Albuquerque et al. 1981), caffeine, nicotine, morphine, parthenolide (Uematsu, et al. 2002; Rummel et al. 2011) and chloramphenicol (Thea and Barza 1989) can cross brain-blood barrier, the intracranial presence of bitter taste receptors or gustducin may have potential biological and pharmacological significance.
The quality of immunohistochemical staining depends largely on a wide range of factors, such as fixative and fixation duration; tissue processing, including heating and freezing; methods of antigen retrieval; the quality and concentration of the primary antibody; and the visualization method employed (Werner et al. 2000; Emerson et al. 2006; Bussolati and Leonardo 2008; Shi et al. 2008; Fung and Tam 2010). Haga and Yoshie (2010) demonstrated that changes in tissue conditions affected antigen preservation and thus resulted in an altered appearance of gustducin-stained taste cells in rat tongues. The existing studies that report on the presence of taste receptors and/or gustducin in the brain (Ren et al. 2009; Shin et al. 2010; Singh et al. 2011; Dehkordi et al. 2012) and retina (Son et al. 2011) were carried out exclusively on frozen sections. Frozen sectioning protocols however, may bring practical shortcomings to the samples, including degradation, autolysis and diffusion of the proteins, as well as compromised morphological features (Shi et al. 2008). These morphological and biochemical alterations to the samples may result in inconsistent results (Haga and Yoshie 2010). In order to avoid these technical shortcomings associated with frozen sectioning, we performed immunofluorescence staining on vibratome-cut sections from mouse brains. Surprisingly, we could not detect gustducin-positive neurons or astrocytes as shown in previous studies (Ren et al. 2009; Shin et al. 2010; Singh et al. 2011; Dehkordi et al. 2012). Instead, we found dense fibers in the nucleus accumbens and periventricular areas with the widely used and validated rabbit polyclonal antibody against gustducin α subunit gustducin, or GNAT3 protein.
Materials & Methods
Animals
Nine wild-type male C57BL/6 mice (8 weeks old) from The Jackson Laboratory (Bar Harbor, ME) were used to show gustducin immunoreactivity in the brain and tongue. Six gustducin-knockout mice (Wong et al. 1996) were used as a universal negative control. The procedures and protocols for all animal studies were approved by the Children’s Hospital of Philadelphia and University of Pennsylvania Institutional Animal Care and Use Committees in accordance with international guidelines on the ethical use of animals (National Research Council 1996).
Tissue Processing
The animals were deeply anesthetized with 0.4 ml of 5% chloral hydrate. Intra-cardiac perfusion was performed with saline and 4% paraformaldehyde in 0.1 M phosphate buffer (pH 7.4). Brains were removed and post-fixed in the same fixative for 90 min at room temperature. Frontal sections were cut at 50-µm thickness with a Leica VT 1000s vibratome (Leica Microsystems Inc.; Buffalo Grove, IL). To minimize the number of animals sacrificed, the brain sections were collected in series for different immunostaining settings with an interval of 300 µm between two adjacent sections within an identical series.
Tongues from perfused mice were recovered and post-fixed for 3 hr at room temperature. The posterior half, comprising the foliate papillae along both lateral edges and a single circumvallate papilla at the central portion (Mistretta and Baum 1984; Mistretta et al. 1999), was embedded with agarose. Transverse sections at 100 µm in thickness were cut perpendicular to the surface and long axis of the tongue with the Leica vibratome. These tongue slices were collected in three series.
Immunofluorescence Staining
Single and double immunofluorescence staining was performed as previously reported (Xiong et al. 2012). Vibratome sections of the brain and tongue from both wild-type and gustducin-null mice were incubated with an affinity-purified rabbit polyclonal antibody against gustducin α subunit (sc-395; 1:1000 in PBS at a concentration of 0.2 µg/ml; Santa Cruz Biotechnology; Dallas, Texas). This Santa Cruz rabbit antibody recognizes the sequence of 70–120 amino acids of rat gustducin protein from its N-terminus. Visualization was performed with Alexa Fluor ® 594-conjugated goat anti-rabbit IgG (1:200, Molecular Probes; Grand Island, NY). Primary antibody incubation was applied for 90 min at room temperature and continued overnight at 4°C, and secondary antibodies were subsequently applied for 90 min at room temperature. Immunostained sections were counterstained with Hoechst (Molecular Probes), a nuclear dye to indicate the nuclei of all cells in the tissue for structural verification.
We then tested if a similar staining pattern could be obtained with another affinity purified polyclonal gustducin antibody raised in goat (LS-B4942; 1:500 in PBS at a concentration of 1 µg/ml; Lifespan Biosciences; Seattle WA). This Lifespan Biosciences goat antibody recognizes amino acids 304–318 of human gustducin at its C-terminus. Double immunofluorescence staining was also performed with both rabbit and goat polyclonal antibodies. Consecutively, Alexa Fluor ® 594-conjugated donkey anti-rabbit IgG (red) and Alexa Fluor ® 488-conjugated donkey anti-goat IgG (green) were used for the two gustducin antibodies in tongue and brain slices. We also performed double staining in brain sections using the rabbit polyclonal combined with a mouse monoclonal against microtubule-associated protein subtype 2 (MAP2, 1:500; Sigma-Aldrich; St. Louis, MO) or a rat polyclonal against glial fibrillary acidic protein (GFAP; 1:2; Developmental Studies Hybridoma Bank; Iowa City, Iowa). MAP2 is a dendritic marker for neurons from postnatal animals (Bernhardt and Matus 1984) and GFAP, a marker for astrocytes (von Koskull 1984). Combinations of Alexa Fluor ® 594-conjugated donkey anti-rabbit IgG (red) and Alexa Fluor ® 488-conjugated donkey anti-mouse IgG (green) or Alexa Fluor ® 488-conjugated donkey anti-rat IgG (green) were used for visualization.
Control Staining
Peptide blocking and omission of primary antibody were employed as the conventional “negative controls” (Haga and Yoshie 2010). We blocked the rabbit polyclonal with its immunizing peptide (Santa Cruz Biotechnology) before immunostaining. Different ratios of peptide to primary antibody were tested at 5-, 10- or 20-fold by weight (not by moles), reaching a concentration of 1, 2 or 4 µg/ml. The rabbit polyclonal and its blocking peptide were mixed and incubated on shaker for 90 min at RT and then overnight at 4°C. To the first series of brain or tongue sections, the pre-incubated peptide–antibody mixture was applied as a substitute for the original rabbit polyclonal during immunofluorescence staining, as described above. For the second series of sections, the rabbit polyclonal was omitted when performing immunofluorescence staining. A third series of sections were used for regular staining using the original rabbit polyclonal.
Confocal Imaging
Confocal images were acquired with Olympus Fluoview 1000 System (Olympus America, Center Valley, PA), as reported previously (Xiong et al. 2012). Consistent confocal settings were optimized and remained unchanged when imaging slices from different animals. The staining pattern was confirmed in at least three mice. For double immunofluorescence, a sequential imaging module was used to prevent signal leakage from one channel to another. A series of images was stacked to show the general distribution pattern, whereas single confocal images were used to show double staining at higher magnification. For brain structures, the nomenclature of Paxinos and Franklin (2001) was followed.
Results
Fibers Stained using Rabbit Polyclonal Antibody in Wild-type Mouse Brain
We used the Santa Cruz Biotechnology rabbit gustducin antibody to perform fluorescence immunostaining of sections from the whole brain ranging from the olfactory bulb through to the rostral spinal cord. Unlike previous studies showing gustducin-positive neuronal somata or astrocytes in a variety of brain regions (Ren et al. 2009; Dehkordi et al. 2012; Singh et al. 2012), we could not find positive cell bodies in the brain or spinal cord. Instead, we identified numerous gustducin-stained fibers (neuropile) in the nucleus accumbens (Fig. 1A–1C) and periventricular areas (Fig. 1D–1F). Over a period of two years, we tested three lots of the rabbit gustducin antibody and acquired the same staining pattern. In the nucleus accumbens, gustducin staining was seen solely in its core region (Fig. 1A). In the periventricular areas, gustducin staining was predominantly seen surrounding the ventral third ventricle (Fig. 1D). A small number of gustducin-stained fibers were distributed along the ventromedial bank of the lateral ventricle and the dorsal third ventricle. Fiber staining was occasionally distributed along the aqueduct but not seen around the fourth ventricle and the central canal of the spinal cord. Under high magnification, these stained fibers displayed a bead-like morphology (Fig. 1C and 1F; Insets). Double fluorescence staining demonstrated that these gustducin-stained fibers were not co-stained by either MAP2 (Fig. 2A–2C) or GFAP (Fig. 2D–2F), confirming that they do not belong to neuronal dendritic arbors or astrocytic fibers.
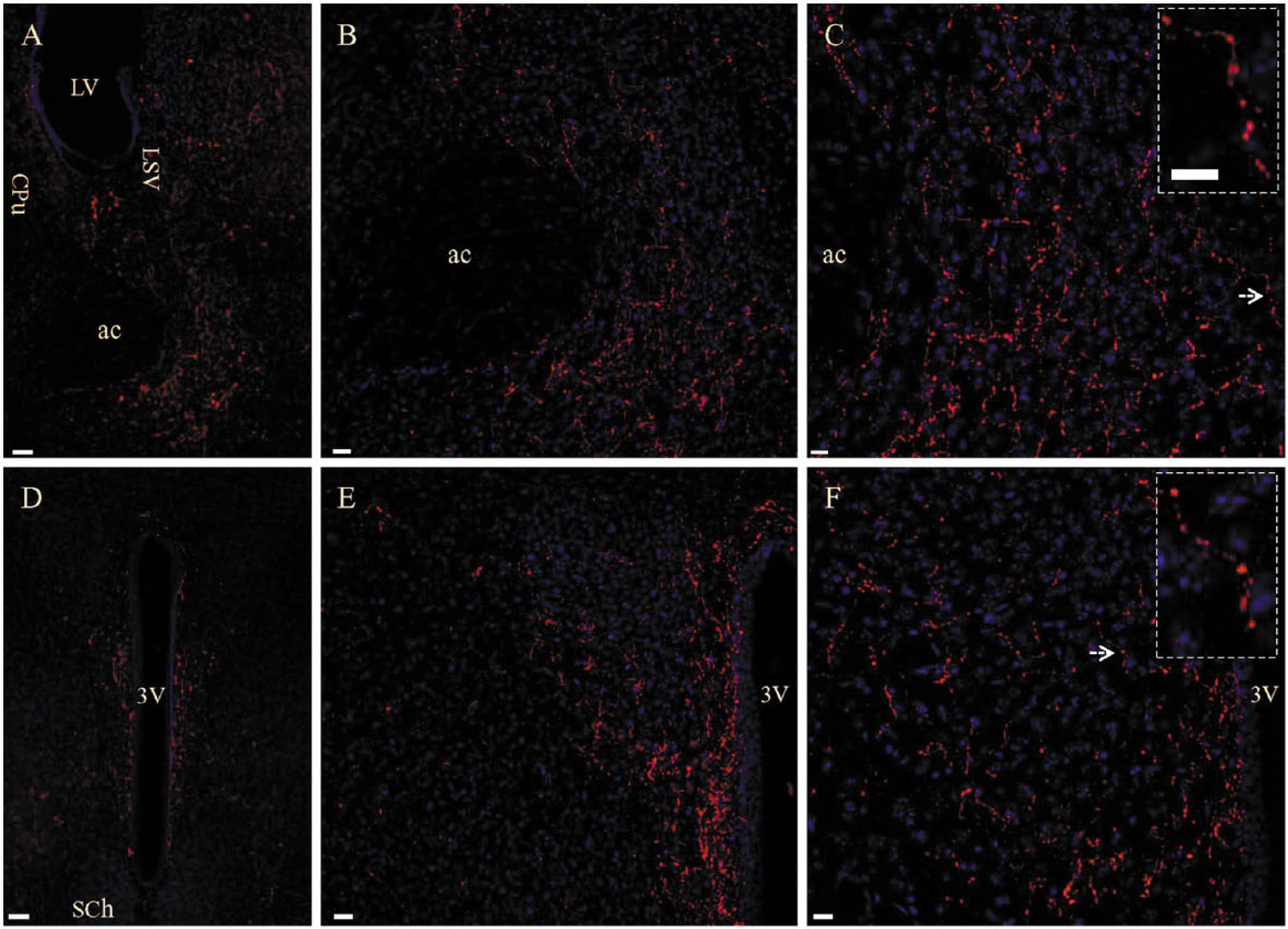
Gustducin-stained fibers in nucleus accumbens and periventricular areas in wild-type mouse. Rabbit polyclonal antibody against gustducin α subunit (GNAT3 protein) was used. (A) Gustducin staining in the nucleus accumbens and ventral part of the lateral septal nucleus (LSV). ac, anterior commissure; CPu, caudatus-putamen; LV, lateral ventricle. (B) Higher magnification of gustducin staining in the nucleus accumbens in (A). (C) Higher magnification of stained components in (B) exhibiting a bead-like morphology (inset). (D) Gustducin staining in the periventricular area surrounding the ventral third ventricle (3V). SCh, suprachiasmatic nucleus. (E) Higher magnification of gustducin stained fibers in (D). (F) Bead-like appearance (inset) of gustducin-stained fibers in (E) under higher magnification. Arrows in (C) and (F) indicate a single fiber highlighted in the Inset. Scale (A and D), 50 µm; (B and E), 20 µm; (C and F), 10 µm; insets, 10 µm.
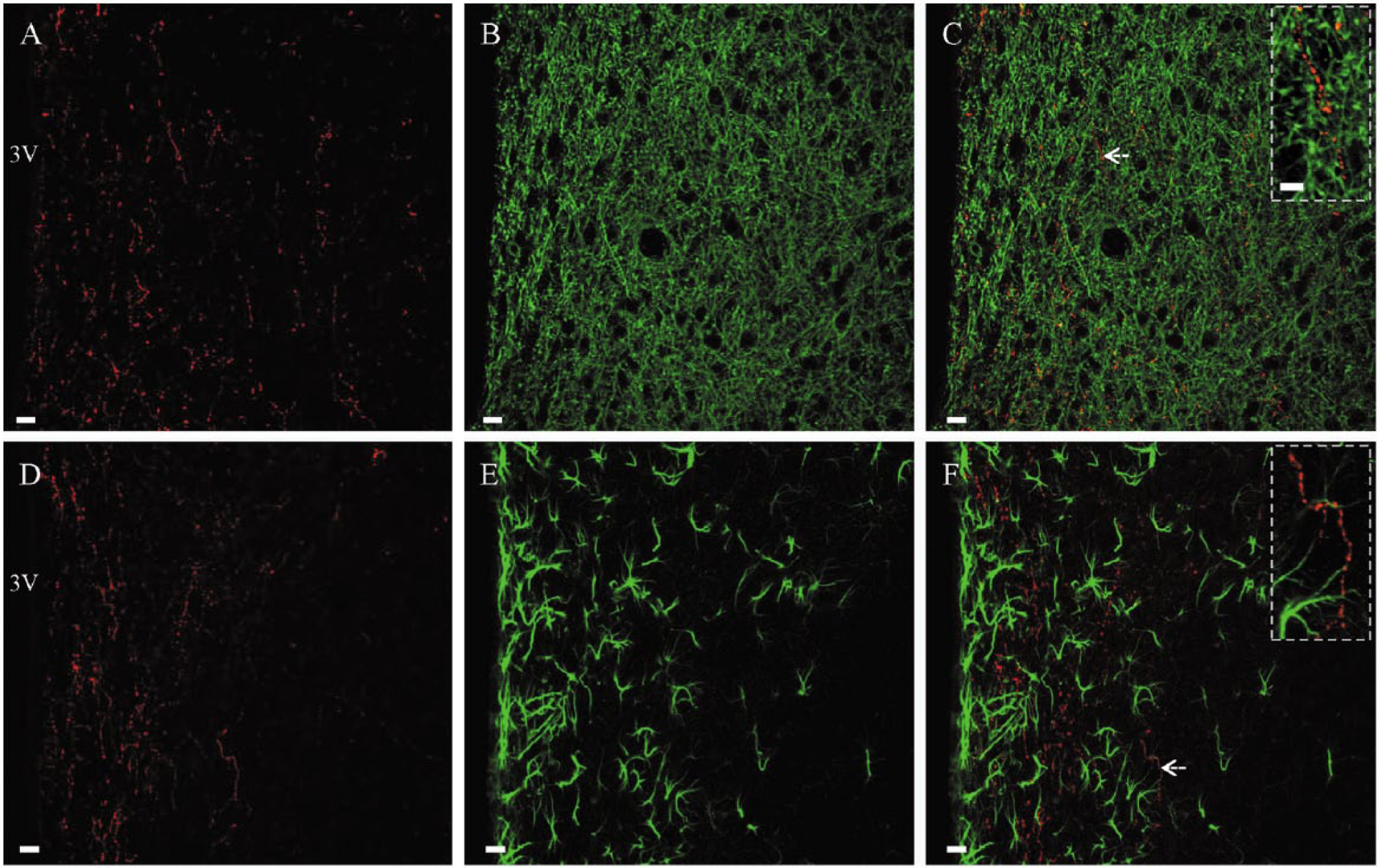
Gustducin-stained fibers may be axonal structures. (A–C) Co-staining of rabbit anti-gustducin (A, red) and mouse anti-microtubule associated protein subtype 2 (MAP2; B, green) showed that these gustducin-stained fibers were not MAP2-positive (C). (Inset) Higher magnification of a single fiber in C (arrow) stained with gustducin only. Some gustducin-stained neuropiles were present in a very close contact with MAP2-positive dendrites (yellow). (D–F) Co-staining of rabbit anti-gustducin (D, red) and rat anti-glial fibrillary acidic protein (GFAP; E, green) showed that these fibers were not GFAP-positive (F). (Inset) Higher magnification of the single fiber in F (arrow) stained by gustducin only. Scale (A–F), 10 µm; insets, 5 µm.
Control Staining in Tongue Slices
To verify the specificity of the rabbit gustducin antibody, we performed double immunofluorescence staining in tongue slices (Fig. 3) using both rabbit and goat gustducin antibodies. In wild-type mice, taste cells were simultaneously stained with both rabbit (red) and goat (green) antibodies, showing a perfect co-staining (Fig. 3A, yellow). In gustducin-knockout mice, tongue slices were not stained by either of the two antibodies (Fig. 3D).
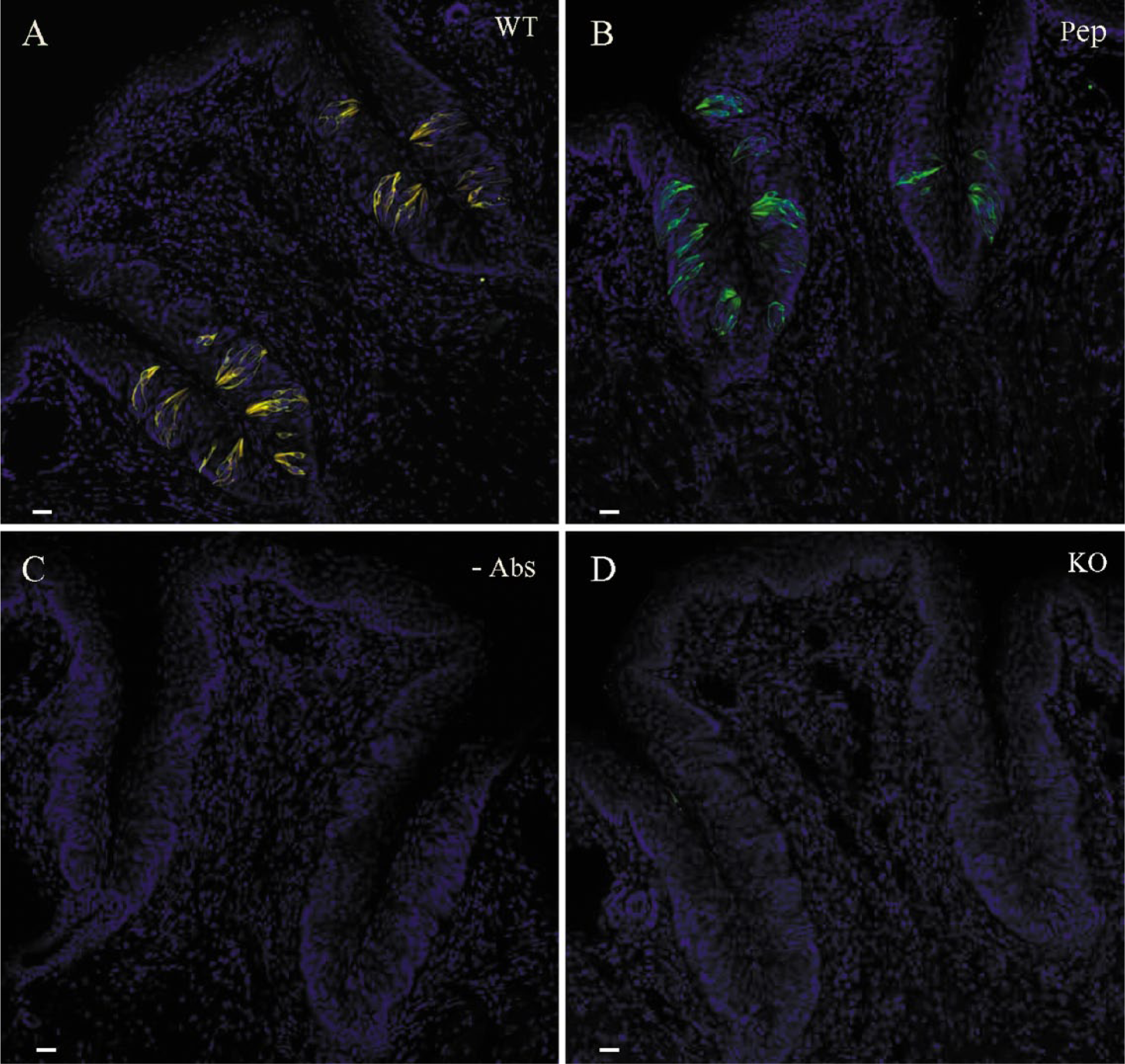
Gustducin and control staining in the circumvallate papilla of the tongue from wild-type (A–C) and gustducin knock-out mice (D). (A) Double staining with both gustducin antibodies raised from rabbit (red) and goat (green) showed perfect co-staining (yellow). (B) Pre-incubation with immune peptide for the rabbit antibody (Pep) completely blocked immunostaining from this antibody, leaving goat antibody staining (green) unaltered. (C) Omission of both antibodies (-Abs) resulted in no staining. (D) Double staining with both gustducin antibodies showed no positive staining in the tongue from gustducin knockout mice. Scale, 20 µm.
We then performed peptide blocking in tongue slices from wild-type mice using different ratios of the immune peptide to the rabbit gustducin antibody (5:1, 10:1 and 20:1). Complete blocking of the rabbit gustducin antibody staining could be reached only at the ratio of 20:1 (peptide concentration at 4 µg/ml) in tongue slices, leaving taste cells stained solely by the goat antibody (Fig. 3B, green). On the other hand, omission of both rabbit and goat polyclonal antibodies resulted in no staining in tongue slices (Fig. 3C), suggesting that the secondary antibodies gave no detectable non-specific staining in the present protocol.
Control Staining in Brain Slices
Because gustducin immunostaining was completely abolished in the tongue slices (Fig. 3D) from gustducin knockout mice (Wong et al. 1996), we expected negative staining in the brain slices from these mice as well. To our surprise, we consistently detected a high density of stained fibers along the ventral third ventricle and nucleus accumbens core. A small number of stained fibers were also seen in periventricular areas along the lateral ventricle, dorsal third ventricle and aqueduct. Similarly as in wild-type mice, no gustducin-stained neuronal somata or astrocytes were identified in brain slices from gustducin knockout mice. We then performed double immunostaining with both rabbit and goat antibodies to mouse brain slices. The goat gustducin antibody (green) did not stain the periventricular and accumbens fibers in wild-type (Fig. 4A and 4B) or knockout mice (Fig. 4D and 4E), leaving a single staining of the rabbit gustducin antibody (Fig. 4, red). A close comparison showed that there was no obvious difference in the general distribution pattern of these fibers in the gustducin-knockout mice (Fig. 4D and 4E) as compared with the wild-type mice (Fig. 4A and 4B). Under high magnification (Fig. 4; insets), stained fibers in knockout mice (Fig. 4D and 4E) exhibited a similar morphology to that of wild-type mice (Fig. 4A and 4B). Finally, conventional control experiments were performed in brain slices. We found that fiber staining by the rabbit gustducin antibody could not be completely blocked (Fig. 4c, red) even at the concentration 4 µg/ml of blocking peptide. As anticipated, omission of both rabbit and goat gustducin antibodies resulted in no staining in brain slices (Fig. 4F).
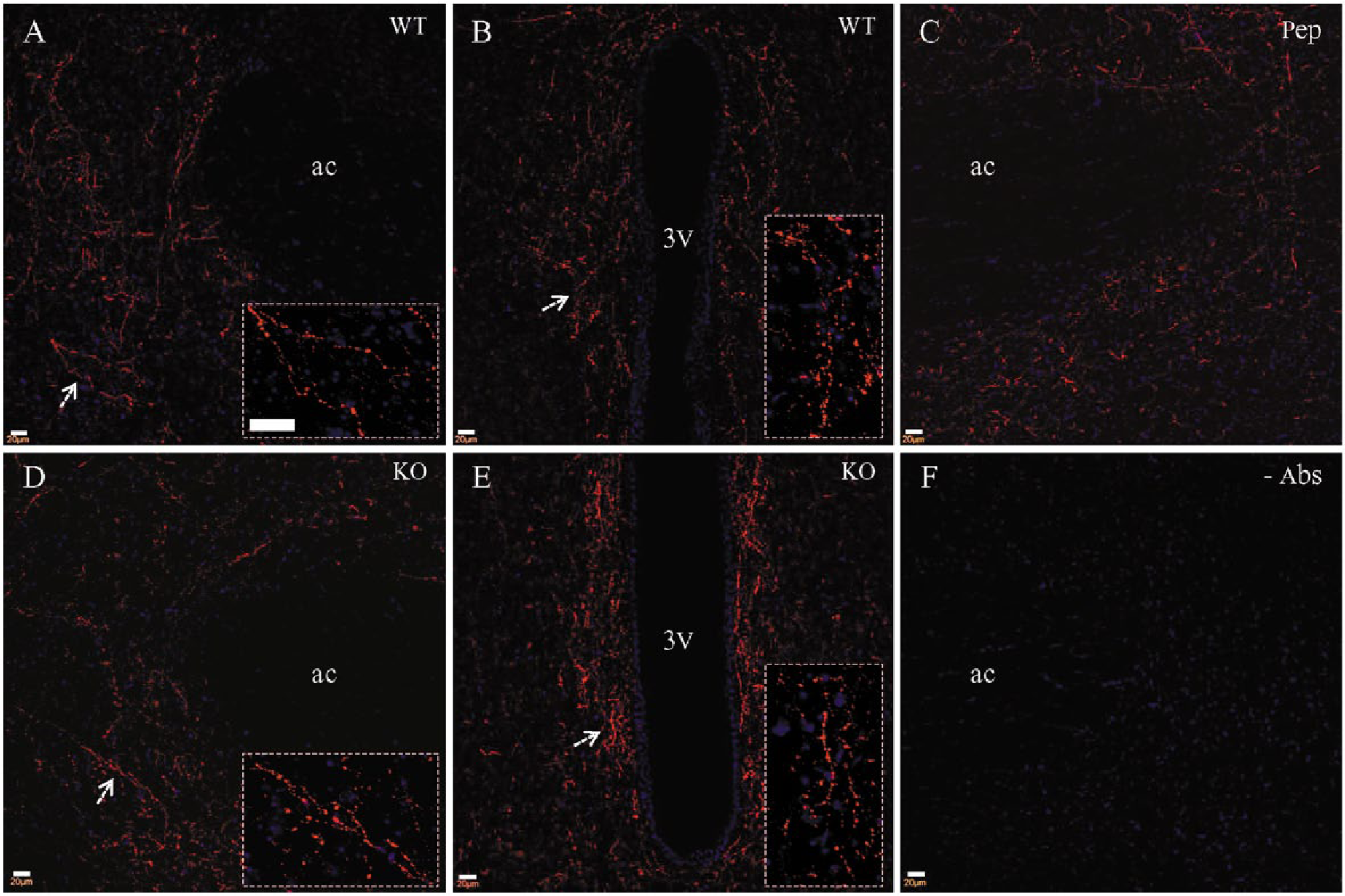
Fibers stained by the rabbit antibody (red) are not stained by the goat antibody. Rabbit antibody-stained fibers in the nucleus accumbens (A) and periventricular area (B) from wild-type mouse. (C) Pre-incubation with immune peptide for the rabbit antibody could not block immunostaining from this antibody in the brain. Rabbit antibody-stained fibers in nucleus accumbens (D) and periventricular area (E) from gustducin-knockout mouse. A similar distribution pattern and bead-like morphology (insets) are seen from wild-type (A and B) and knockout (D and E) mice. Arrows indicate the position of the higher magnification images (insets). (F) Omission of both primary antibodies resulted in no staining at all. Scale (A–F), 20 µm; insets, 20 µm.
Discussion
Using a rabbit polyclonal gustducin antibody from Santa Cruz Biotechnology (sc-395), we appeared to observe specific staining of fibers in the mouse nucleus accumbens and periventricular areas. The bead-like morphology and non-dendritic/non-astrocytic identity highly suggested that these fibers may belong to axonal structures. Because a conventional “negative control” with omission of the primary antibody resulted in no staining, we assumed that these stained fibers were reflective of gustducin expression with discrete localization patterns in the central nervous system. However, this conclusion was erroneous, because the rabbit polyclonal staining was similar in gustducin-knockout mice (Wong et al. 1996). Additionally, the rabbit polyclonal staining observed in both wild-type and knockout mice could not be confirmed with a goat polyclonal gustducin antibody from Lifespan Biosciences (LS-B4942). It is highly possible that a cross immunoreaction occurred between an unidentified molecule in these fibers and the rabbit polyclonal antibody.
When a target antigen or an unidentified molecule shares an epitope in common with the immune peptide of a tool antibody, a cross immunoreaction occurs (Berzofsky et al. 2003). This is also the case when the molecule has a structurally similar epitope to that of the immune antigen. Cross reactivity between specific antibodies and unrelated proteins is gaining increased attention, with one report showing that two widely used mouse monoclonal antibodies against the master cell cycle regulator Cdk1 cross-react with the centrosomal protein Cep152 (Lukinavičius et al. 2013). Here, we have provided another example of cross immunoreactivity; although, the nature of the cross-reacting molecule is unknown as yet. A search through Protein BLAST (National Library of Medicine) shows that a putative candidate protein called GTP-binding protein like 1 (AGBL1) has a similar sequence to that of the immune peptide from Santa Cruz Biotechnology. Thus, cross reaction might be expected between the rabbit polyclonal and protein AGBL1. However, our hypothesis remains untested because of the lack of an ideal AGBL1 antibody and a paucity of literature about the distribution pattern of this protein in the brain.
Using the same rabbit gustducin antibody from Santa Cruz Biotechnology (sc-395), we identified dense fiber staining exclusively in the nucleus accumbens and periventricular areas in vibratome sections. It is worth emphasizing that we could not find positive staining in neuronal somata or astrocytes. However, previous studies using frozen sections (Shin et al. 2010; Dehkodi et al. 2012) claimed positive staining in neuronal somata and astrocytes. This discrepancy may be attributed to the differences in tissue processing. In some circumstances, such as improper cryoprotection or fluctuations in the cutting temperature, frozen sectioning may introduce damage to the tissue, which includes protein loss or diffusion, and/or morphological changes (Shi et al. 2008). These morphological and biochemical alterations may result in inconsistent immunostaining patterns (Haga and Yoshie 2010). We noticed that Dehkodi et al. (2012) showed gustducin-positive astrocytes in rat medullary pyramids, but not in other brain areas. Using a similar protocol, Shin et al. (2010) reported intense, astrocytic staining in the hippocampus of the mouse, rat and rabbit.
The best control for specific staining in tissues should be either negative staining in gene knockout mice or co-staining with two antibodies that are made from different animal species and designed to recognize different sequences of the target protein (Lorincz and Nusser 2008; Saper 2009). However, many previous immunohistochemical studies solely adopt the conventional “negative control” by either omitting or blocking the primary antibody prior to staining (Ruiz-Avila et al. 1995; Haga and Yoshie 2010; Shin et al. 2010; Singh et al. 2011; Son et al. 2011; Dehkordi et al. 2012). Peptide blocking might just confirm that the primary antibody is specific for the target peptide sequence (not the entire protein), which is competing against the immune peptide for binding to the primary antibody. To estimate the necessary amount of the immune peptide to block the staining, certain factors particular to the target molecule (in this case, gustducin protein) must be taken in consideration: (1) the affinity of the gustducin antibody to the protein, and (2) the concentration of gustducin in the tissue. A large amount of gustducin protein in the tongue slices might therefore require a much higher concentration of blocking peptide in order to block the immunostaining (see Results). Simply omitting the primary antibody, on the other hand, is just a control to rule out non-specific binding of the secondary antibody, which may result in a detectable signal. Both strategies, however, cannot eliminate the possibility of non-specific binding of the primary antibody to tissues or cells of interest. Any non-specific binding of the secondary antibody may be diminished, at least in part, by a normal serum blocking step prior to immunostaining. The primary antibody’s non-specific binding appears more likely to result in detectable false staining due to the magnifying effect of subsequent immunoreaction steps. So far, there is no evidence to show whether immune peptide-bound primary antibody has the same ability to non-specifically bind to sample tissues as the unblocked antibody. Therefore, it is unclear if the immune peptide-primary antibody mixture can really represent the original antibody in this matter.
Without proper controls, the previous reports of taste receptors and/or gustducin in the brain (Ren et al. 2009; Shin et al. 2010; Singh et al. 2011; Dehkordi et al. 2012) and retina (Son et al. 2011) might be artificial. Rather than the conventional negative controls of pre-incubation with immune peptides or omission of primary antibodies, a better negative control may be necessary, especially when a new immunostaining pattern is to be verified. Tissues from knockout mice may be an ideal negative control for non-specific staining (Lorincz and Nusser 2008; Saper 2009). Sometimes, knocking out certain key proteins may be lethal to mice. Additionally, gene knockout may not have any relevance to staining human or non-mouse tissues. In these cases, temporal or local gene knockdown may be an optimal selection. When genetic manipulation is unavailable, testing different tissue processing protocols or various sources of primary antibodies in parallel may be an option.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by a grant from The Flight Attendants
