Abstract
Cystic Fibrosis Transmembrane conductance Regulator (CFTR) protein has recently been shown to be expressed in the human adult central nervous system (CNS). As CFTR expression has also been documented during embryonic development in several organs, such as the respiratory tract, the intestine and the male reproductive system, suggesting a possible role during development we decided to investigate the expression of CFTR in the human developing CNS. In addition, as some, although rare, neurological symptoms have been reported in patients with CF, we compared the expression of normal and mutated CFTR at several fetal stages. Immunohistochemistry was performed on brain and spinal cord samples of foetuses between 13 and 40 weeks of gestation and compared with five patients with cystic fibrosis (CF) of similar ages. We showed in this study that CFTR is only expressed in neurons and has an early and widespread distribution during development. Although we did not observe any cerebral abnormality in patients with CF, we observed a slight delay in the maturation of several brain structures. We also observed different expression and localization of CFTR depending on the brain structure or the cell maturation stage. Our findings, along with a literature review on the neurological phenotypes of patients with CF, suggest that this gene may play previously unsuspected roles in neuronal maturation or function.
Introduction
Cystic Fibrosis (CF) is a common autosomal recessive disorder that results from mutations in the gene encoding the cystic fibrosis transmembrane conductance regulator (CFTR) (Riordan et al. 1989). This protein mainly functions as a small conductance ATP- and cAMP-dependent chloride channel in secretory epithelial cells and has been shown to be expressed in the lung, liver, intestine, pancreas, genital excretory tract and sweat glands (Harris et al. 1991; Tizzano et al. 1993). In addition to its function as a chloride channel, CFTR is also involved in the regulation of other ion channel gating, as well as intracellular membrane trafficking and endo- and exocytosis (Guggino and Stanton 2006; Edwards and Kahl 2010). More than 1900 mutations in the CFTR gene have been described so far but the most frequent mutation is p.F508del, which accounts for approximately 65% of tested cases (Zielenski 2000).
The CF phenotype is only partly correlated with the genotype and encompasses a highly variable spectrum ranging from a focal form of male excretory infertility to a severe multi-organ disease (Zielenski 2000; Noone and Knowles 2001). Lung involvement is the major cause of mortality in these patients and is dominated by recurrent infections and obstruction, finally leading to respiratory failure. Over the past 20 years, CFTR protein and its mRNA expression have progressively been reported to be present in epithelial cells of many other organs, such as the kidney (Todd-Turla et al. 1996; Devuyst et al. 1996), smooth muscle cells, immune cells, cardiac myocytes (Robert et al. 2005) and neurons of the adult central nervous system (CNS) (Mulberg et al. 1995; 1998; Johannesson et al. 1997; Niu et al. 2009; Guo et al. 2009a; 2009b). In addition, several studies have reported the expression of CFTR very early during development in immature cells of the same organs as in adulthood, suggesting a previously unsuspected role for CFTR during development (Harris et al. 1991; Tizzano et al. 1993). These characteristics have predominantly been documented in the main systems affected in CF, particularly the respiratory tract, the intestine and the male reproductive system (Harris et al. 1991; Tizzano et al. 1993; Marcorelles et al. 2007; 2012). However, in the absence of clear genotype-phenotype correlations in CF, the developmental pattern of expression of CFTR has not been documented in organs that are not the major targets of the disease. As both peripheral and central neurological symptoms have sometimes been reported in patients with CF (Cavalier and Gambetti 1981; Cochran et al. 1991; El-Salem et al. 2010), we decided to investigate the expression of CFTR in the developing human CNS using immunohistochemistry and searched for, as previously described in the lung and male reproductive system (Marcorelles et al. 2007; 2012), possible differences in the expression and/or localization of the protein in deceased fetal and neonatal subjects with CF at different gestational ages.
Materials & Methods
Materials
Sixteen fetal and neonatal brains ranging from 13 to 40 weeks of gestation (WG) were selected for the present study. In each case, a complete autopsy had been performed with the written informed consent of the parents. Medical termination of the pregnancies has been accepted by the local ethical committee of the Prenatal Diagnosis Multidisciplinary Center according to the French Law. In ten cases, brains were macroscopically and microscopically free from detectable abnormalities and were used as controls (Table 1). CFTR expression in these control brains was compared with six fetal brains from patients with CF aged 13, 15, 19, 26 and 34 WG. The cause of death and genotype for each case is presented in Table 1.
Description of Fetal and Neonatal Cases Used in this Study.
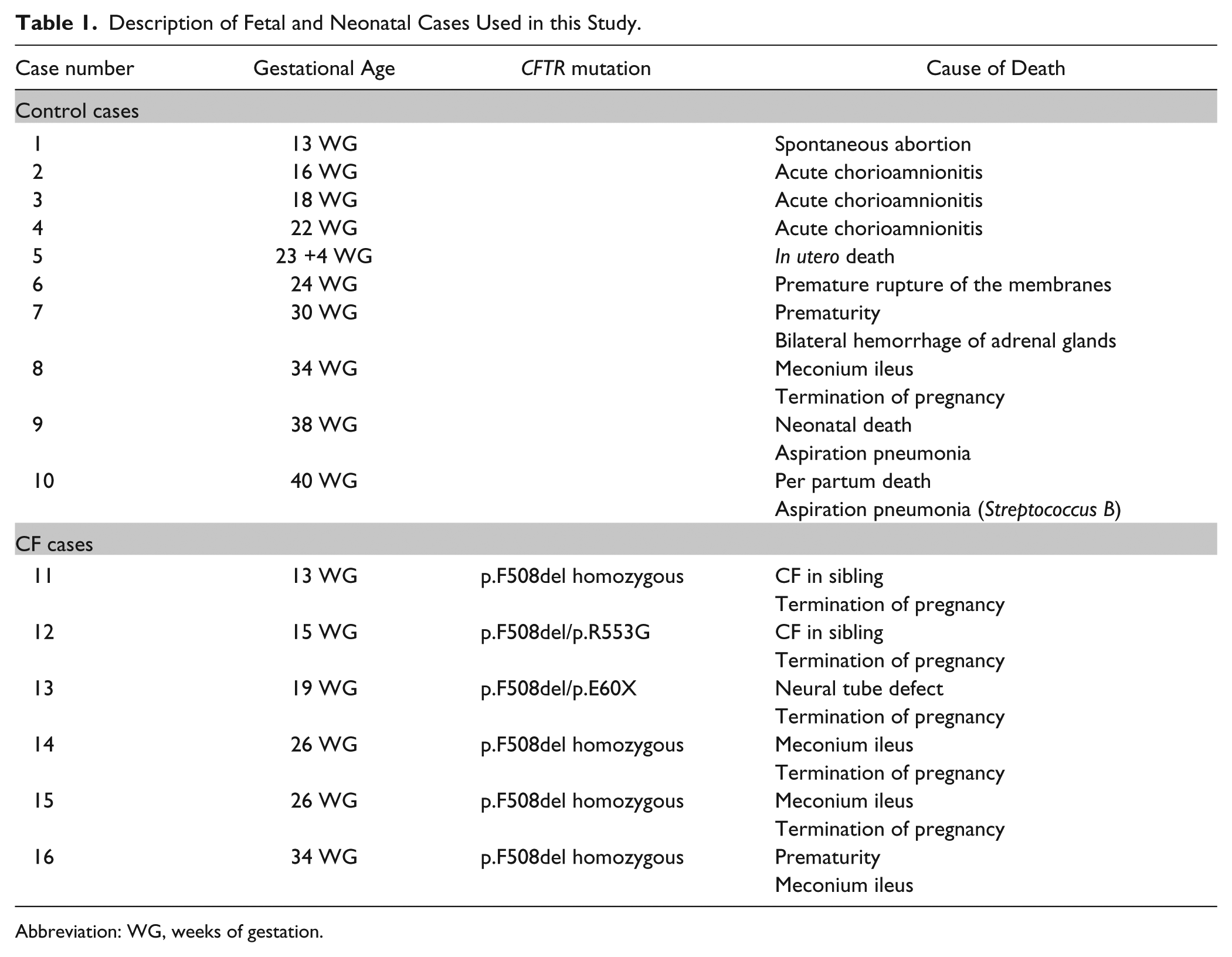
Abbreviation: WG, weeks of gestation.
Methods
Eight-µm, paraffin-embedded sections from multiple brain regions were mounted onto coated slides (Silanized Slides S 3003 Dako, Trappes, France) and dried overnight in a convection oven (37°C). Sections were deparaffinized in three baths of xylene and then re-hydrated in a series of ethanol solutions. Induced epitope retrieval was performed by immersion in a citrate buffer solution, pH6, at 95–99C for 1 hr. Slides were then rinsed in distilled water and allowed to cool. Different pre-treatments and dilutions were tested to determine the optimal dilution (1/150) for the monoclonal anti-human CFTR antibody (Mab24-1; R&D Systems, Lille, France), which was raised against amino acids 1377-1480 of the C-terminal domain. Endogenous peroxidase activity was removed by Peroxidase Blocking Reagent (S2001, Dako, Trappes, France) for 10 min. Non-specific binding was blocked with 10% bovine serum albumin (BSA) (A-3174, Sigma-Aldrich, Saint Quentin, France). Incubation with the primary mouse monoclonal antibody was performed in BSA diluted 1:10 for 90 min at room temperature. Sections were washed three times in PBS and incubated with the Peroxidase-Conjugated Envision + Dual Link System-HRP (K4061, Dako) for 30 min and finally washed in PBS. Labeling was visualized using 3’-diaminobenzidine in a chromogen solution (Liquid DAB + Substrate Chromogen System, K3468, Dako). Slides were rinsed in tap water, counterstained with Mayer’s Hematoxylin and mounted in mounting medium. Negative controls were obtained by omission of the primary antibody or the use of other antibodies of known reactivity such as VEGF (Sentilhes et al. 2010) or ARX, a transcription factor widely expressed in the developing brain and giving only nuclear labeling. In addition, the specificity of our CFTR labeling was carefully checked using different anti-CFTR antibodies (MAB3480 from Chemicon, Temecula, CA; MAB25031 and MAB1660 from R&D Systems, Abington, UK) and by testing them in parallel on lung, kidney and vas deferens sections (positive controls) and on heart and thymus sections (negative controls) (Todd-Turla et al. 1996; Devuyst et al. 1996; Marcorelles et al. 2007; 2012).
Results
At 13 WG, in the control case, CFTR was widely expressed in many brain structures (Fig. 1A). In the developing cortex, the neuroepitheli lining the lateral ventricle exhibited a strong diffuse expression of CFTR whereas cells in the ventricular/subventricular zone (VZ/SVZ) of the developing cortex had only a perinuclear rim labeling (Fig. 1B–1C). Radial glial cells appeared negative. In layer I of the developing cerebral cortex, Cajal-Retzius cells, which are transient neurons playing important roles in regulating neuronal migration and corticogenesis, were strongly positive, whereas neurons in the developing cortical plate had only a perinuclear rim with more diffuse cytoplasmic labeling extending within neuronal processes (Fig. 1D), similar to radially migrating neurons (Fig. 1E). In the developing ganglionic eminences, only the neuroepithelium was faintly labeled. The developing basal ganglia (caudate nucleus, putamen and globus pallidus) were also positive, especially the globus pallidus, where large neurons showed perinuclear and diffuse cytoplasmic labeling with enrichment in neuronal processes (Fig. 1F–1G). The thalamus and the hypothalamus were also strongly positive, as well as the neuroepithelial lining the third and fourth ventricles (Fig. 1H–1J). CFTR expression in the choroid plexuses appeared diffuse and cytoplasmic but slightly polarized (Fig. 1K). At the infratentorial level, CFTR was strongly expressed in neurons of the pons, cranial nerves, brainstem nuclei and olivary inferior complex. In most of the cerebral structures observed, CFTR labeling appeared mostly perinuclear and diffuse with cytoplasmic enrichments sometimes located in neuronal processes. However, in the dentate nucleus, olivary inferior complex and cranial nerves, neurons displayed additional cytoplasmic large vesicular-like staining (Fig. 1L).
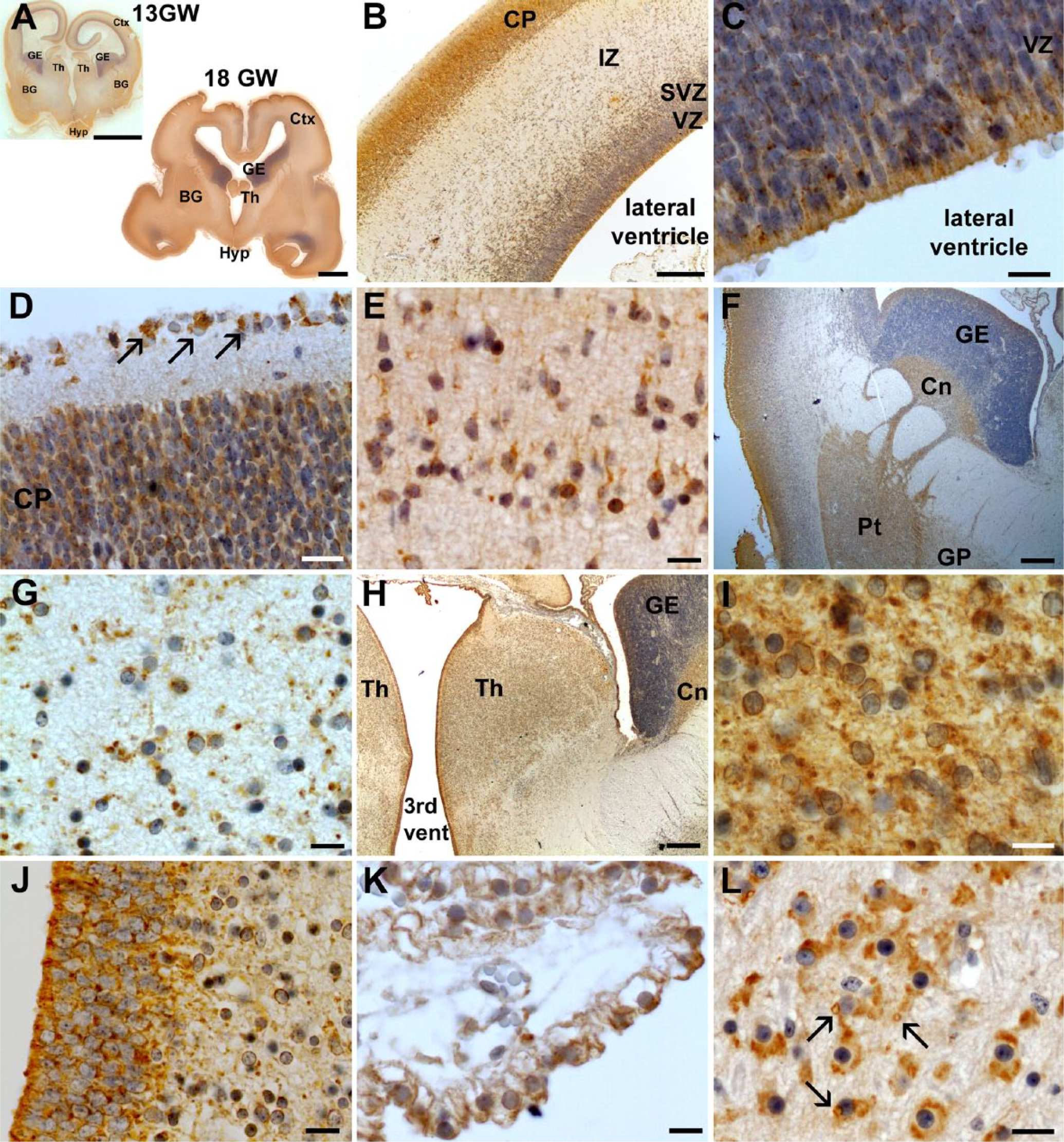
CFTR expression at 13 WG. (A) At this stage, CFTR is expressed in neurons of several brain structures. CFTR expression is found in the developing cortex (ctx) (B), where it is particularly expressed in the neuroepithelium of the ventricular zone (VZ) (C), in the cortical plate (CP) and in Cajal-Retzius cells (see arrows) (D), and in neurons of the intermediate zone (IZ) (E). (F) In the ganglionic eminences (GE), CFTR is only expressed in the neuroepithelium lining the lateral ventricle. In the basal ganglia (BG), CFTR is only faintly and diffusely expressed in the caudate nucleus (Cn) and putamen (Pt) but is clearly present in large neurons of the globus pallidus (GP), showing a perinuclear localization and diffuse cytoplasmic expression with local enrichments (G). CFTR is also strongly expressed in the developing thalamus (Th) (H–I) and in the neuropithelium lining the third ventricle (J). In the choroid plexuses, CFTR localization appears slightly polarized with more expression at the apical side (K). At the infratentorial level (developing cerebellum, pons and medulla), CFTR is also strongly expressed but shows a different type of labeling with the apparition of vacuole-like structures (arrows) (L). Scale bars: A, 4 mm; B, F, H, 0.2 mm, D, E, G, I–L, 10 µm, C, J, 5 µm.
In the corresponding CF case (13 WG, homozygous for the p.F508del mutation), CFTR expression was generally very weak, probably due to the reduced amount of mutated protein produced. In the developing cortex, the neuroepithelial lining was negative (Fig. 2A) whereas neurons of the cortical plate and Cajal-Retzius cells were only faintly positive (Fig. 2B). The ganglionic eminences, the developing hippocampus and hypothalamus were completely negative. Basal ganglia and thalamus displayed a very faint and diffuse cytoplasmic labeling (Fig. 2C–2D). Similarly, the neuroepithelium of the third and fourth ventricles, as well as the choroid plexuses, were very faintly labeled (Fig. 2E–2F). Interestingly, at the infratentorial level, CFTR was expressed in brainstem nuclei and cranial nerves at a level almost similar to the control (Fig. 2G–2I), suggesting that the absence or low expression of CFTR in some parts of the brain, such as the cortex, hippocampus or hypothalamus, may be due to a delay in the appearance of the protein, as previously observed in other tissues (Marcorelles et al. 2007; 2012). In all structures, CFTR labeling in the CF case was only diffuse and cytoplasmic. Positivity along the perinuclear rim was very rarely observed (Table 2).
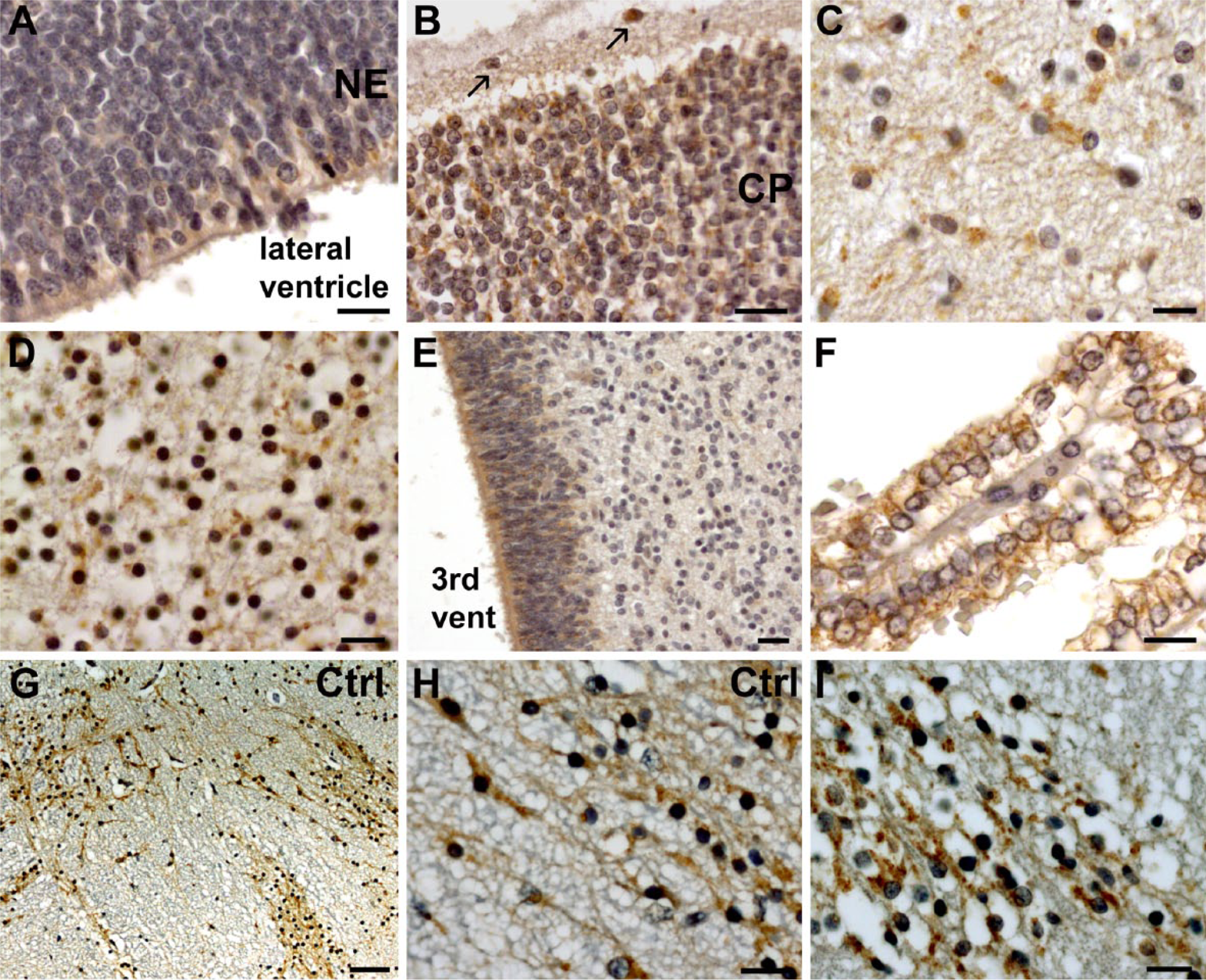
CFTR expression in the 13 WG CF case. In the developing cortex, the neuroepithelium is almost negative (A) whereas Cajal-Retzius cells (indicated by arrows) and neurons of the cortical plate are weakly positive (B). Neurons of the globus pallidus (C) and the thalamus (D) were positive but more diffusely and weakly labeled than in the control (Fig. 1G and 1I). Similarly, staining in the neuroepithelium, such as the one lining the third ventricle (E), was only weakly positive. Although the choroid plexuses expressed CFTR in the CF case, its localization was more diffuse and less polarized (F) than in the control. At the infratentorial level, CFTR was clearly expressed in several structures, such as neurons of the cranial nerves (G) shown at low magnification where it exhibited similar level of expression (I) as compared with the 13 WG control (H). Scale bars: A, G, 5 µm, B–D, F, H, I, 10 µm, E, 100 µm.
CFTR Protein Expression in the Developing Central Nervous System.
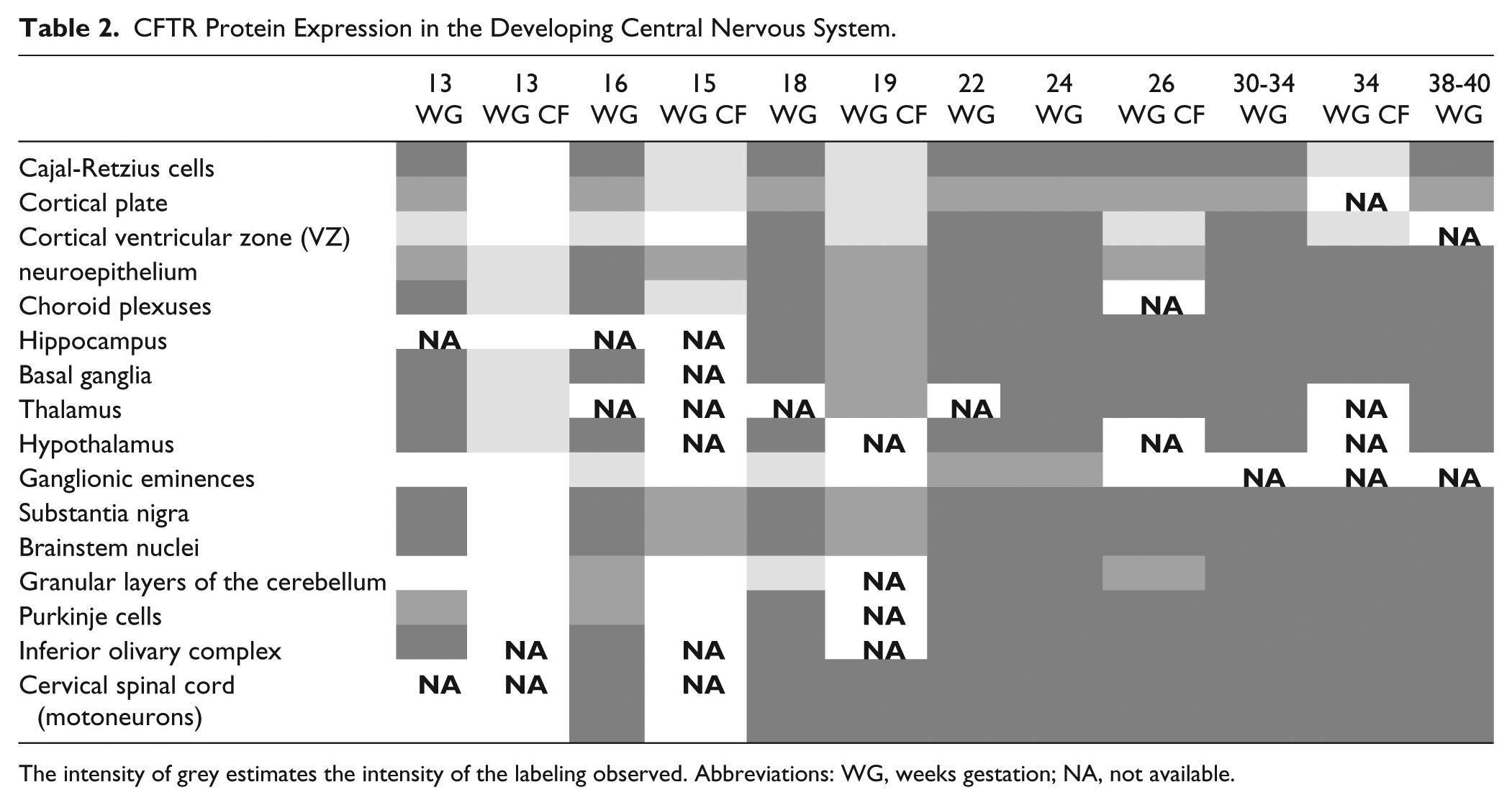
The intensity of grey estimates the intensity of the labeling observed. Abbreviations: WG, weeks gestation; NA, not available.
In the 16 and 18 WG control cases, CFTR was expressed in the same cerebral structures as at 13 WG but with stronger expression (Fig. 1A, Table 2). In the developing cortex, Cajal-Retzius cells were still strongly positive, and several tangentially and radially orientated neurons in the intermediate zone (IZ) exhibited mild perinuclear immunoreactivity with diffuse cytoplasmic staining extending to leading processes. As observed at 13 WG, the caudate nucleus and putamen were only very weakly positive but large neurons of the globus pallidus were more strongly labeled, with the appearance of white, large and small vesicular-like structures (Fig. 3A). The same pattern of cellular labeling was also observed in the thalamus, hypothalamus (from 18 WG) and in large neurons of the substantia innominata and nucleus basalis of Meynert, which appear at all stages very strongly labeled, even at low magnification (Fig. 3B–3C). In small neurons of the same structures, only a perinuclear rim was detected. The ganglionic eminences were still negative for staining, with the exception of the neuroepithelium lining the lateral ventricle, which was faintly positive. In the developing cerebellum, CFTR expression was observed in Purkinje cells, as well as in external and internal granular cell layers, predominating in the proliferative compartment of the external granular cell layer (Fig. 3D). CFTR was still present in cranial nerves and in most of most of the peduncular and pontine nuclei, usually showing a perinuclear and diffuse cytoplasmic labeling except in noradrenergic neurons of the locus coeruleus and in motor neurons, where the punctuate and vesicular pattern appeared enriched under the plasma membrane (Fig 3E–3F, Table 2).
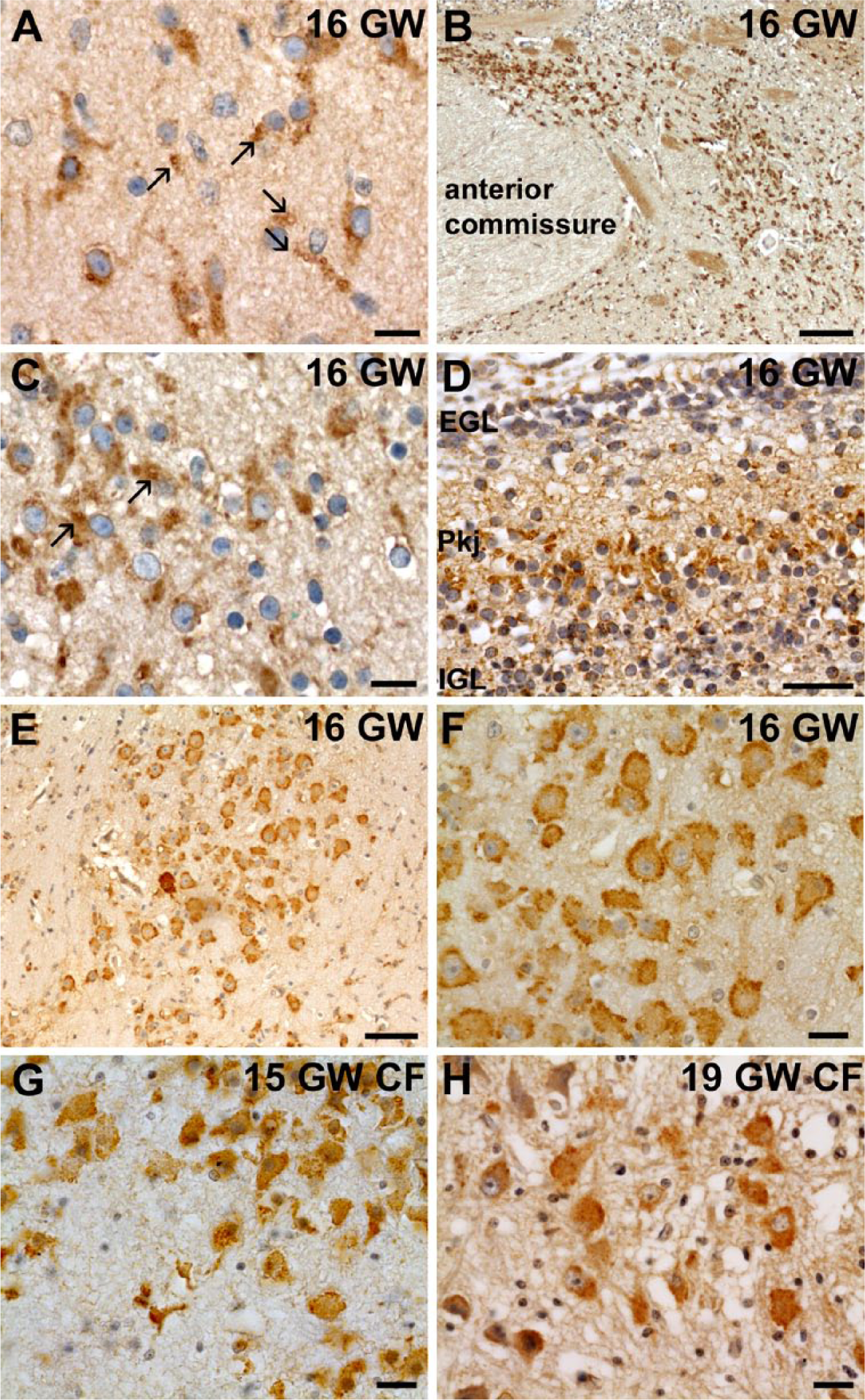
Expression of CFTR between 15 and 19 WG. At 16 WG, CFTR antibody labeled small and large cytoplasmic vesicular-like structures (arrows) in large neurons of the globus pallidus whereas small neurons only had a perinuclear staining (A). This type of labeling was also observed in neurons of the substantia innominata and nucleus basalis of Meynert (C), which expressed strongly CFTR, as shown at low magnification (B). In the developing cerebellum, differentiating Purkinje cells (Pkj) were already strongly labeled as the proliferating compartment of external (EGL) and internal granular layers (IGL) cells (D). At the infratentorial level, large neurons of the locus coeruleus expressed strongly CFTR (E) at 16 WG with vesicular-like enrichment under the plasma membrane (F). Interestingly, this pattern of expression was also found, albeit weaker, in the locus coeruleus of CF cases at 15 WG (G) and 19 WG (H). Scale bars: A, C, F–H, 10 µm, B, 100 µm, D, E, 50 µm.
In the corresponding 15 WG CF case (compound heterozygous p.F508del/p.R553G), a diffuse cytoplasmic neuronal expression was observed in the same brain structures as the control but with a much weaker and more diffuse expression (Table 2). In general, the perinuclear staining was less visible and the vesicular-like pattern was very rarely observed, except in the thalamus or the locus coeruleus where the expression of mutated protein looked similar to the control (Fig. 3G). In the 19 WG CF case (compound heterozygous p.F508del/p.E60X), CFTR expression was very weak, more so than in the other CF cases (Table 2), probably due to the severity of the mutations. The neuroepithelium and the choroid plexuses were negative, as well as the cerebral cortex, with the exception of the Cajal-Retzius cells, which were faintly positive. The other brain structures were very weakly labeled and the pattern of expression of CFTR appeared more perinuclear and generally more diffuse than in controls of similar ages, except in motor neurons or in the locus coeruleus, where the labeling under the plasma membrane could be observed in some cells similarly to that seen in the control (Fig. 3H).
At 22 and 24 WG, the cortical VZ/SVZ were strongly positive, with cytoplasmic and perinuclear CFTR expression (Fig. 4A–B). Many radially and tangentially orientated neurons were also immunopositive in the IZ (Fig. 4D). In the choroid plexuses, a stronger expression of CFTR began to emerge at the level of cell-cell junctions. In the neuroepithelium of the third and fourth ventricles, differentiating tanycytes and ependymal cells strongly expressed CFTR (Fig. 4E). Similarly, the VZ of the ganglionic eminences was also positive although the rest of the cells corresponding to differentiating interneurons did not express CFTR (or very weakly). The hippocampus, thalamus, and hypothalamus were strongly immunoreactive (Fig. 4G), although not all cells expressed CFTR. At the infratentorial level, as in earlier stages, CFTR expression was detected in the neurons of cranial nerves, substantia nigra, red nucleus, and with a similar type of staining as earlier stages in cerebral peduncles, pons, cerebellum and medulla (Table 2).
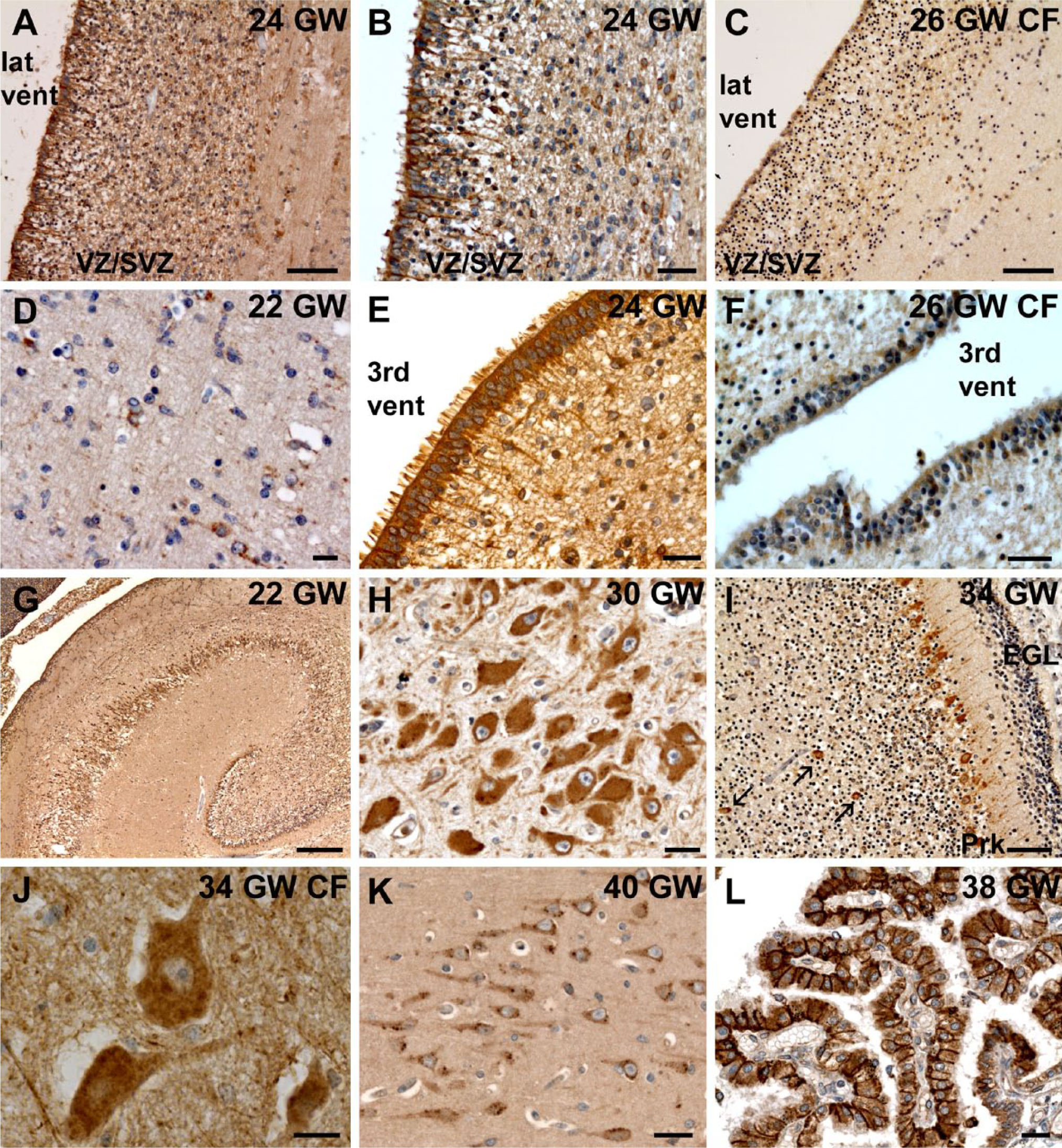
CFTR expression between 22 and 40 WG. At 22 and 24 WG, CFTR antibody labeled intensely the developing ependymal cells lining the lateral ventricle (A–B). However, in the 26 WG CF case, the neuroepithelium was scarcely positive (C) and ependymal cells looked not as well differentiated as in the control (Fig. 4A). Many neurons were labeled in the ventricular/subventricular zone (VZ/SVZ) (A–B) and in the intermediate zone (IZ) (D) of the cortex in the control. Similar to the cortex, the neuroepithelial lining the third ventricle was strongly and well differentiated in the control (E), in contrast to the 26 WG CF case (F). At 22 WG, CFTR was strongly expressed in the hippocampus (G). In most of the cerebral structures, as in the dentate nucleus, a perinuclear and intense cytoplasmic staining with reinforcement under the plasma membrane was observed (H). In the developing cerebellum, CFTR was expressed in Golgi II cells (see arrows) in addition to Purkinje (Prk) and granular cells in the external granular layer (EGL) (I). In most of the cells, such as motor neurons (J), CFTR labeling in the 34 WG CF case looked cytoplasmic diffuse without much perinuclear or cytoplasmic enrichment contrarily to what was observed in the control, as for example in the hippocampus (K). From 24 WG, CFTR labeling in the choroid plexus was enriched at the apical and lateral side of cells (L). Scale bars: G, 300 µm, A, C, I, 50 µm, B, E, F, K, L, 20 µm, D, H, J, 10 µm.
In the corresponding CF patient (26 WG, homozygous for the p.F508del mutation), as for other CF cases, CFTR immunostaining was globally weaker and more generally cytoplasmic, with less perinuclear and vesicular-like patterns observed than in the control (Table 2). Interestingly, there was no tanycyte differentiation and the pattern of immunoreactivity of the neuroepithelium was very weak (Fig. 4C, 4F).
From 30 WG, CFTR pattern of expression was globally similar to what had been observed at earlier stages (Table 2). In general, large neurons had strong, cytoplasmic granular immunostaining, most likely corresponding to Nissl bodies, in addition to plasma membrane and perinuclear expression (Fig. 4H, Table 2). In small neurons, only perinuclear expression was detected. Most of the cerebral structures had labeled neurons at this stage. The ganglionic eminences, putamen and caudate nucleus, which were negative or very weakly positive at earlier stages, showed intense perinuclear labeling. In the developing cerebellum, Golgi II cells began to express CFTR in addition to Purkinje and granular cells (Fig. 4I).
The pattern of CFTR expression in the corresponding CF patient (34 WG, homozygous for the p.F508del mutation) was similar to the control case. However, most neurons displayed a diffuse vesicular cytoplasmic labeling and no clear accumulation under the plasma membrane could be observed (Fig. 4J, Table 2). Ependymal cells, in particular tanycytes, looked less differentiated than those in the control at the same age and in the choroid plexuses, CFTR expression was cytoplasmic and diffuse, without any clear polarization.
At 38 to 40WG, the expression of CFTR was very strong in every brain structures studied (Table 2). The perinuclear and vesicular-like pattern, which was more often observed at earlier stages at the infratentorial level or in the substantia innominata, was now observed in most of the cerebral structures, even in neurons of the cortical plate or hippocampus (Fig. 4K). In ependymal cells, CFTR immunoreactivity was concentrated at the apex. In the choroid plexuses, CFTR expression was enriched at the apical and lateral sides of the cells (Fig. 4L).
Discussion
Only few studies have previously investigated CFTR expression in the rodent CNS. They have reported its expression in the cerebral cortex of adult rat, especially in layer V, and in several other brain regions, such as the midbrain, pons, medulla oblongata, hypothalamus, thalamus, hippocampus, choroid plexuses and ependyma (Mulberg et al. 1995; Johannesson et al. 1997; Hincke et al. 1995). The first report of CFTR expression in human brain focused on the anterior part of the hypothalamus (Mulberg et al. 1998), as it helped to explain the increased energy expenditure, growth failure and delay in pubertal development or infertility reported in CF patients (Mulberg et al. 1998; Shepherd et al. 1998). In agreement with this study, we observed that the hypothalamus and, from 22 WG, the thalamus in control cases displayed an intense signal.
More recently, Guo and colleagues reported a much broader expression of CFTR in the adult human brain, spinal cord and sympathetic ganglia (Niu et al. 2009; Guo et al. 2009a; 2009b). In agreement with these studies, we never found CFTR expression in radial glial cells or astrocytes but exclusively in neurons. In control cases, CFTR was detected both in the soma and neuronal processes, but fiber tracts, particularly pyramidal tracts, were always negative, suggesting that CFTR may only be expressed in dendrites and in the most proximal part of axons. CFTR does not seem to be preferentially expressed in one neuronal type, as it was found to be expressed in all cortical layers, Cajal-Retzius cells, Purkinje cells and motor neurons. In addition, we observed CFTR expression in some migrating neurons in the developing cortex and cerebellum. Interestingly, we observed that CFTR expression followed a certain dynamic pattern of expression intensity and cellular localization appearing first in infratentorial structures (inferior olivary complex, motor neurons, cranial nerves, brainstem nuclei…), hypothalamus and substantia innominata whereas most of the other brain structures progressively caught up during development with the cerebral cortex (except for Cajal-Retzius cells) being the last (Table 2). We also noted that in the developing cerebral cortex, the VZ and CP were faintly labeled at early stages but both became progressively more intensevely labeled during development, suggesting that CFTR may be required for neuronal maturation and differentiation, as well as in ependymal cell maturation in the neuroepithelium.
The intracellular pattern of CFTR expression we observed was similar to previous descriptions in adult human and rodent nervous system (Mulberg et al. 1998; Guo et al. 2009a; 2009b). At early stages in control cases, CFTR was predominantly observed around the nucleus. In migrating neurons, a diffuse cytoplasmic labeling was also observed in leading processes, consistent with the previously reported role of CFTR (Bradbury et al. 1994) in vesicular trafficking, which is known to be important for cellular migration. At later stages, when neurons began to differentiate, the labeling appeared more diffuse and cytoplasmic, often enriched under the plasma membrane (called “3” in Table 2, Fig. 3E–3F), probably corresponding to the transport of functional CFTR at the plasma membrane. From mid-gestation, we observed in an increasing number of cerebral structures, neurons with large cytoplasmic vacuole-like structures (called “2” in Table 2, Fig. 1L, 3A, 3C), which disappeared after 34 WG in control cases and were rarely observed in CF patients (Table 2). These vacuole-like structures may correspond to trans-Golgi CFTR trafficking. From the third trimester, the CFTR antibody clearly labeled structures probably corresponding to Nissl bodies, especially in the hippocampus, in Purkinje cells, sensory nuclei and motor neurons of the cervical spinal cord. We tried to characterize these intracellular patterns using double-labeling experiments with different markers of endosomes, trans-Golgi network or lysosomes but, unfortunately, the CFTR antibody we used in this study—which was, in our hands, the only one to give nice and reproducible results on paraffin brain sections—did not work in double-labeling experiments.
The CFTR gene mutation p.F508del found in the CF cases of our study is a class II mutation leading to a defective maturation of the protein (Zielenski 2000). Whereas the normal, newly-synthesized CFTR protein is glycosylated in the endoplasmic reticulum (ER) and further modified in the trans-Golgi network to become functional, the misfolded F508del protein is retained in the ER, where it is subsequently degraded by the ubiquitin-proteasome system (Guggino and Stanton 2006). A fraction of the mutated protein is still directed to the cellular plasma membrane and possesses a residual activity but has a decreased half-life (Tizzano et al. 1993). In our study, the pattern of expression of mutated CFTR was similar to that in the control cases. We only observed a global delay in the appearance of the immunostaining, possibly due to the smaller amount of protein produced, along with a slight delay in neuronal maturation, as suggested by the pattern in the neuroepithelium or the type of cellular labeling observed in CF patients compared with controls (Table 2). These data, combined with the absence of cerebral malformation reported in CF patients, suggest that the F508del protein may have a residual function that is sufficient for neuronal differentiation and/or normal function, or that mutated CFTR may be functionally compensated by other gene(s). Along the same lines, we observed a more severe effect on both CFTR expression and localization in the 19 WG patient, with general, very weak labeling throughout the brain and an absence of signal in the developing cortex, neuroepithelium and choroid plexuses. This is in agreement with the genotype of this patient who has, in addition to the p.F508del mutation, a class I mutation (p.E60X) leading to the absence of the protein.
Although CF is generally not associated with malformations to the CNS, a recent study has reviewed nine unrelated cases presenting with an association between CF and Chiari I malformation (Patel et al. 2011). The etiology of this association remains unexplained although it has been suggested that it may result from the recurrent coughing and wheezing or metabolic and electrolyte imbalances that occur characteristically in CF. In this study, we observed in mutated cases a slight delay in maturation, especially at the level of the neuroepithelium. It is thus possible that a delay in the maturation of the neuroepithelium and the choroid plexuses in CF patients may cause defects in the cerebrospinal fluid outflow and possibly contribute to hydrocephalus.
Both peripheral and central neurological signs have been reported in patients with CF. Peripheral nerve dysfunction had originally been attributed to malabsorption and vitamin deficiency but vitamin E supplementation sometimes failed to prevent these complications (Cavalier and Gambetti 1981). More recently, nerve conduction studies showed electrophysiological abnormalities of peripheral nerves and derangement of autonomic response in children with CF (El-Salem et al. 2010). In autopsy reports of adults with CF, axonal dystrophy has been described in 75% of the brains as well as in the few examined spinal cords (Cavalier and Gambetti 1981; Cochran et al. 1991). Even if these symptoms may result from a complex association of diabetes, autoimmunity and vitamin deficiency, a primary CFTR defect is also likely to be involved as shown by the recent report of peripheral nervous system defects observed in CFTR-/- pigs at birth (Reznikov et al. 2013).
Concerning CFTR protein function, recent works have studied CFTR activity in motor neurons of the spinal cord or brainstem nuclei in rats during neonatal development (Ostroumov et al. 2007; 2011; Morales et al. 2011) and have shown that CFTR contributes to the relatively depolarized equilibrium potential for synaptic inhibition, an important process to control hyperexcitability and seizure-predisposition in neonates (Ostroumov et al. 2007). It has also been shown that CFTR plays a role in postsynaptic inhibition of motor neurons from the trigeminal motor nucleus during rapid-eye-movement (REM) sleep (Morales et al. 2011). Recently, Ostroumov and colleagues observed that the gene activities of the chloride inward transporter, NKCC1, and CFTR were positively correlated postnatally and, using different blockers for these channels, demonstrated that inhibition of CFTR or NKCC1 activity produced a negative shift in GABA/glycine reversal potential of spontaneously occurring synaptic events measured after block of excitatory transmission, suggesting that CFTR operated together with NKCC1 to produce depolarizing GABA/glycine-mediated synaptic events (Ostroumov et al. 2011). The importance of chloride in the control of neuronal migration and differentiation is well known (Ben-Ari 2006) and, as with NKCC1, CFTR may also play a role in these processes. Besides, a recent study revealed that attention-hyperactivity disorders are not uncommon, maybe even under evaluated, in children with CF (Georgiopoulos and Hua 2011), suggesting a possible role of CFTR in the development and/or maturation of neuronal subpopulations.
In conclusion, we here describe for the first time a strong and widespread neuronal expression of CFTR in the human developing CNS. Although none of the CF patients studied here had any cerebral abnormality, there was a slight delay in CFTR expression in several brain structures. We also observed different expression and localization of CFTR depending on the brain structure and the cell maturation stage.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
