Abstract
Aquaporin (AQP) is a water-selective channel protein. In the brain, AQPs play critical roles in the production of cerebrospinal fluid and in edema formation. In contrast, the expression and role of AQPs in spinal cord are unclear. We aimed to investigate the localization of AQP1 and AQP4 in normal rat spinal cord compared with the expression of marker proteins for astrocytes, neurons, and endothelial cells. Immunohistochemistry demonstrated that AQP1 and AQP4 are expressed along all levels of the spinal cord from the cervical to lumbar levels. AQP1 immunolabeling was observed in the dorsal horns in the gray matter, whereas the labeling was weak and mainly seen close to glia limitans in the white matter. AQP1 was co-labeled with marker proteins for unmyelinated neuronal fibers (peripherin) and endothelial cells (RECA-1) of blood vessels that had penetrated through the glia limitans. In contrast, AQP1 did not colocalize with GFAP, an astrocyte marker, at any level of the spinal cord. AQP4 was exclusively localized at the astrocytes, but AQP4 expression in spinal cord exhibited a less polarized and more spatial distribution than that of brain astrocytes. The observed characteristic localization and expression patterns of AQP1 and AQP4 could provide insights toward gaining an understanding of the role of AQPs in the spinal cord.
Introduction
Aquaporins (AQPs) are water channel proteins expressed widely in human body tissues, with high expression in kidney, salivary glands, lung, liver, muscle, and nervous system (Agre et al. 2002; Nielsen et al. 2002; Kwon et al. 2013). Thirteen mammalian aquaporins are known and they possess a common tetrameric structure with 20–50% amino acid homology. Cell membranes enriched with AQPs have high osmotic water permeability (Jung et al. 1994; Agre et al. 2002). Importantly, the central nervous system (CNS) is the site where many attempts have been made to establish the expression and role of AQPs. Previous studies have indicated the important role of AQP1 and AQP4 in the physiology and pathophysiology of cerebrospinal fluid (CSF) production and circulation and in the formation of brain edema in human patients (Amiry-Moghaddam et al. 2004; Verkman 2009). AQP9 expression has also been identified in mouse and rat brain and spinal cord (Elkjaer et al. 2000; Badaut et al. 2001, 2004; Oshio et al. 2004), but a comprehensive study with different AQP9 antibodies on wild-type and AQP9-knockouts indicates that there is a cross-reactivity with other proteins (Rojek et al. 2007). In contrast to the studies in brain tissues, however, little attention has been paid to the expression and role of water channel proteins in the spinal cord.
In the brain, AQP1 is exclusively expressed at the apical microvillar membranes of choroid plexus epithelial cells in rodents (Nielsen et al. 1993; Oshio et al. 2005) and humans (Mobasheri and Marples 2004), whereas no labeling is seen in neuronal cells, astroglial cells, and endothelial cells (Nielsen et al. 1993). In contrast, in spinal cord, it was previously demonstrated that AQP1 is expressed in primary afferent neurons, ependymal cells and astrocytic cell bodies, but not in endothelial cells (Shields et al. 2007; Nesic et al. 2008). Moreover, spinal cord injury has been shown to induce an increase in the expression of AQP1 in ependymal cells, dorsal horn fibers, neuronal cell bodies, and reactive scar-forming astrocytes (Nesic et al. 2008). Therefore, based on these previous studies, there is a significant difference in the AQP1 expression between brain and spinal cord tissues, particularly in terms of its expression in neuronal and astroglial cells. Further studies are thus needed to confirm the AQP1 localization in CNS.
AQP4 is predominantly present in the plasma membrane of astrocytes, with weaker expression previously noted in the basolateral membranes of ependymocytes (Rash et al. 1998). AQP4-labeled glial cells have been identified in the olfactory system, cortex, hippocampus, amygdaloid complex, thalamus, hypothalamus, subfornical organ, mesencephalon, cerebellum, and brain ventricular system (Venero et al. 2001). The strongest expression was observed at the borders between brain parenchyma and major fluid compartments, such as the blood brain barrier (BBB) and glia limitans, predominantly located in astrocytic foot processes (Nielsen et al. 1997; Rash et al. 1998). AQP4 expressed in astrocytic end-feet arranges into regular structures referred to as square arrays (Rash et al. 1998; Furman et al. 2003). Moreover, in the perivascular and subpial end-feet of astrocytes, AQP4 colocalizes with α-syntrophin, which belongs to the brain dystrophin complexes (Neely et al. 2001; Amiry-Moghaddam et al. 2003; Vajda et al. 2002). The role of AQP4 in the phases of formation and resolution of CNS edema has been demonstrated (Bloch et al. 2006; Manley et al. 2000; Papadopoulos et al. 2004a, 2004b; Papadopoulos and Verkman 2005), and hence brain and the respective compartments that are associated with the production and circulation of CSF and edema formation are important regions of interests in the studies focusing on AQP4. However, despite the importance of AQP4, little attention has been devoted to studying its expression in the spinal cord. To address this issue, we aimed to examine the detailed localization of AQP1 and AQP4 in normal rat spinal cord tissues using AQP1- or AQP4-specific antibodies and compared their expression with that of marker proteins for astrocytes, neurons, and endothelial cells.
Materials & Methods
Experimental Animals
Pathogen-free male Sprague-Dawley rats (270–310 g) were obtained from Charles River Laboratories (Orient Bio, Seongnam, Korea). The animal protocols were approved by the Animal Care and Use Committee of Kyungpook National University, and all animal experiments were conducted according to the guidelines of Kyungpook National University. Rats were housed in controlled temperature rooms (21 ± 1C) with a 12 hr light/dark schedule. Food and water intake were supplied ad libitum.
Perfusion Fixation, Spinal Cord Isolation and Sample Collection
The rats were anesthetized with isoflurane inhalation and perfusion fixation was performed according to the previous study (Gage et al. 2012). The diaphragm was incised along the entire length of the rib cage and a cut on the both sides through the rib cage up to the collarbone was made to allow for lifting the sternum away and providing a clear view of the heart and major vessels. A small incision was made on the posterior end of the left ventricle to pass a perfusion needle and place the tip of it into the ascending aorta. Then, an outlet incision was made in the right atrium and blood was flushed with warm normal saline (37C) for 30 sec, before switching to 4% paraformaldehyde in 0.01 M PBS (pH 7.4) for 3 min. Immediately after perfusion, the spinal cords were collected by hydraulic extraction method proposed by Meikle and Martin (1981). Briefly, a tip of the blunt needle attached to a 10-ml syringe filled with normal saline was placed in the hub of the lumbar canal. Then, after giving a firm and rapid push, the spinal cord popped out onto the working surface with very few histological artifacts. The spinal cord was divided on the basis of anatomic differences into lumbar enlargement representing lumbar part (L1–L5), cervical enlargement representing cervical part (C5–C1) and the thoracic part, which was in turn divided into thoracic lower (T12/13–T6) and thoracic upper (T6–T1) parts.
Immunohistochemistry
To have the desired orientation of the tissue sections, spinal cord segments were put in plastic straws with a direction marker indicating caudal to cephalic direction before placing them in the histological cassettes. The samples were subsequently immersion fixed in 4% paraformaldehyde in 0.01 M PBS (pH 7.4) for 1 hr followed by dehydration in ethanol (70, 96 and 99% each for 2 hr), clearing in xylene overnight, and, finally, embedding in paraffin (Lee et al. 2011). The paraffin-embedded tissues were cut (2-μm thickness) on a rotary microtome (Leica Microsystems, Wetzlar, Germany). The sections were dewaxed and rehydrated. For immunoperoxidase labeling, endogenous peroxidases were blocked by immersion in a solution prepared by dissolving 2.4 ml of 30% H2O2 in 240 ml of absolute methanol for 10 min at room temperature. To reveal antigens, sections were put in 1 mmol/l Tris solution (pH 9.0) supplemented with 0.5 mM EGTA and heated using a microwave oven for 10 min. Thereafter, the sections were cooled for 30 min in the same buffer. Nonspecific binding of IgG was precluded by incubating the sections in 50 mM NH4Cl for 30 min, followed by blocking in PBS supplemented with 1% BSA, 0.05% saponin, and 0.2% gelatin. The sections were incubated overnight at 4C with primary antibodies diluted in PBS supplemented with 0.1% BSA and 0.3% Triton X-100. The following primary antibodies were used: rabbit polyclonal AQP1 (1:1000–1:4000; Terris al. 1996; Kwon et al. 2005), rabbit polyclonal AQP4 (1:1000–1:3000; Terris et al. 1995; Christensen et al. 2006), rabbit polyclonal AQP4 (AB2218, 1:35,000–1:40,0000, EMD Millipore; Billerica, MA;), mouse monoclonal AQP4 (ab9512, 1:100, Abcam; Cambridge, MA), mouse monoclonal rat endothelial cell antigen-1 (RECA-1, ab9774, 1:50, Abcam), rabbit polyclonal transient receptor potential cation channel subfamily V member 4 (TRPV4, AB39260, 1:1000, Abcam), mouse monoclonal glutamine synthase (GS, MAB302, 1:1,000, EMD Millipore), mouse monoclonal glial fibrillary acidic protein (GFAP, MAB3402, 1:1,000, EMD Millipore), mouse monoclonal neurofilament protein 200 kDa (NF200, MAB5262, 1:2000, EMD Millipore), and mouse monoclonal peripherin (MA3-16724, 1:50, Thermo Scientific; Wilmington, DE). After rinsing with PBS supplemented with 0.1% BSA, 0.05% saponin, and 0.2% gelatin for 3×10 min, the sections were incubated in horseradish peroxidase-conjugated secondary antibodies (Dako; Glostrup, Denmark) diluted 1:200 in PBS supplemented with 0.1% BSA and 0.3% Triton X-100, followed by incubation with diaminobenzidine. The microscopy was carried out using a Leica DMRE light microscope. Immunofluorescence labeling was prepared using Alexa fluor secondary antibodies diluted 1:200 (anti-rabbit Alexa fluor 488, anti-mouse Alexa fluor 594) and DAPI (in 1:10,000 solution prepared from 5 mg/ml stock). After a final wash with PBS, the coverslips were mounted with anti-fade agent (#P36930, Life Technologies; Carlsbad, CA). Laser scanning confocal microscopy was carried out (Zeiss LSM 5 EXCITER, Jena, Germany). The specificity of AQP1 and AQP4 antibodies was validated by peptide pre-absorption control experiments in the rat kidney, brain, and spinal cord sections using the peptide against which the antisera was raised (Terris et al. 1996; Terris et al. 1995).
Results
AQP1 Expression
Immunolabeling of AQP1 was of a similar intensity from the cervical region to the lumbar levels of the rat spinal cord. The highest intensity of AQP1 immunolabeling was seen in the dorsal horns as a scattered, spotted pattern, especially in laminae I and II (Fig. 1A and 1B). In laminae III and IV, AQP1 was mostly expressed at the lateral and medial boundaries of the dorsal horns (Fig. 1A). In lamina V, the immunolabeling showed a tangled, filamentous pattern (Fig. 1A and 1C). In other parts of the gray matter, AQP1 expression was seen as dispersed dots along the medial boundary of the dorsal horn up to lamina X and throughout the immediate surroundings of the central canal (Fig. 2D). In white matter, AQP1 immunolabeling was sporadic but present along all levels of the spinal cord and was mostly found close to the glia limitans (Fig. 1D and Fig. 2CI–2CIII).
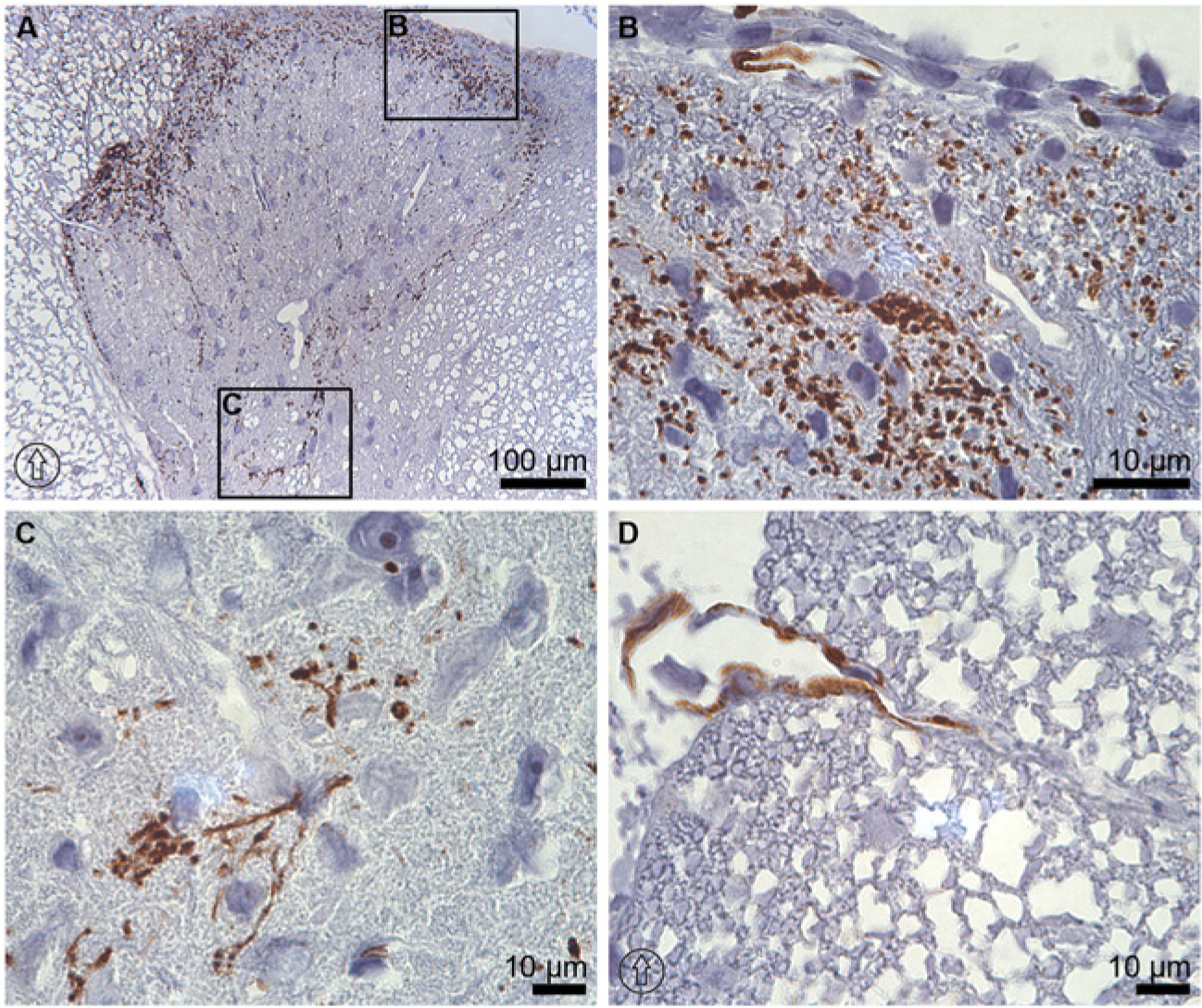
Immunoperoxidase microscopy of AQP1 at the thoracic level of rat spinal cord tissue. Expression of AQP1 in the dorsal horn at thoracic level of the spinal cord (A) was strong in laminae I and II (B). (C) Immunolabeling of AQP1 was also observed at the lateral and medial boundaries of the dorsal horns up to lamina V, where the immunolabeling pattern appeared as a tangled filamentous arrangement. (D) In white matter, AQP1 labeling was sparse and found mostly close to the glia limitans. The dorsal orientation of the spinal cord is indicated by encircled arrows.
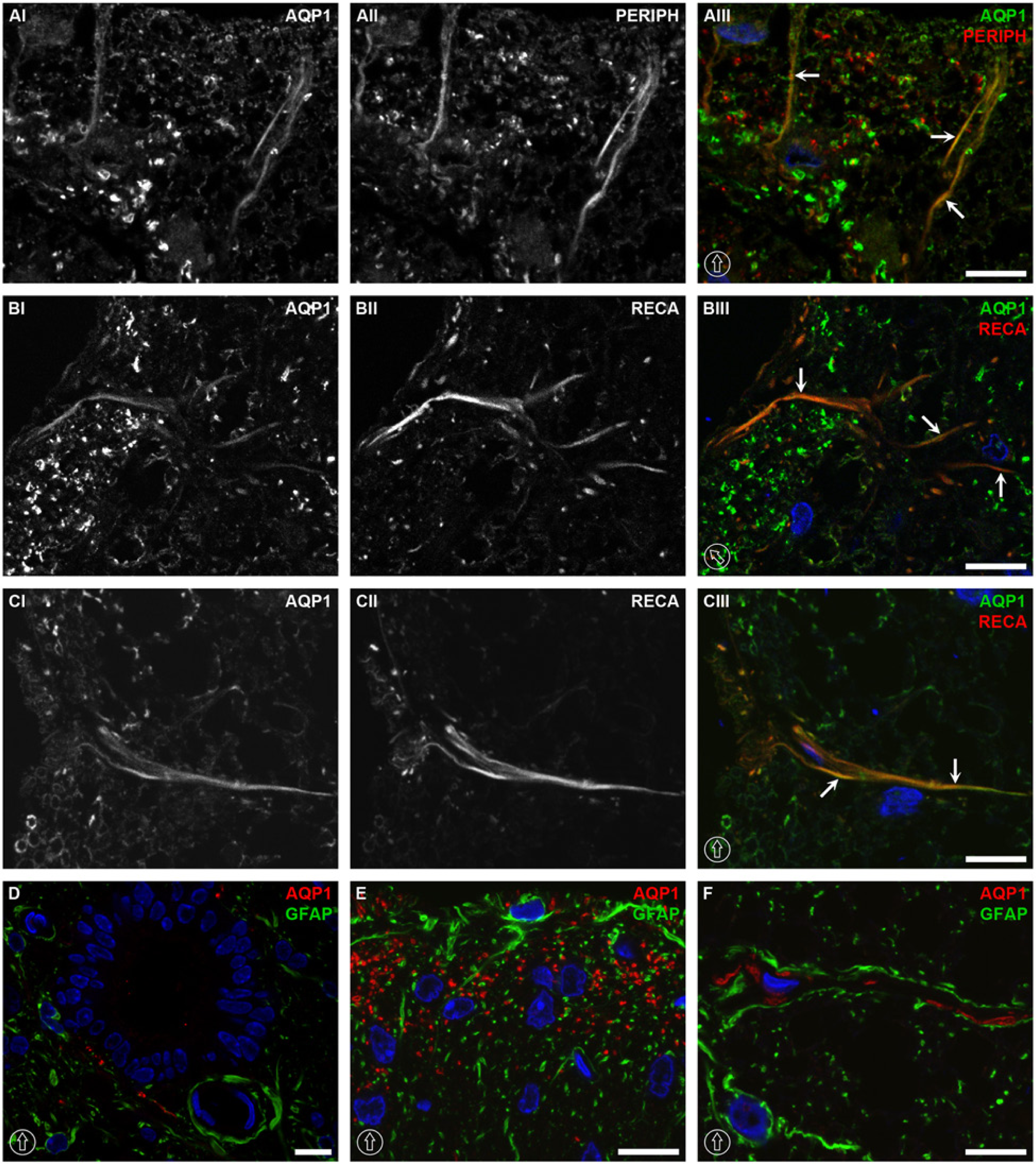
Colocalization of AQP1 with neuronal, endothelial and astrocytic markers at the thoracic level of the rat spinal cord. AQP1 was colocalized with peripherin in laminae I and II of the dorsal horn (AI–AIII), with weaker signal intensity up to lamina III. Colocalization between AQP1 and RECA-1 was found likely in the small arterioles and capillaries branching from the arterial vasocorona, which penetrated into the spinal cord core in the dorsal horn of gray matter (BI–BIII) and white matter (CI–CIII). No co-labeling was noticed between AQP1 and GFAP in AQP1-expressed structures around the central canal (D), dorsal horn (E), or white matter (F). The dorsal orientation of the spinal cord is indicated by encircled arrows; colocalization by arrows. The blue signal in the images represents DAPI staining of the nuclei. AQP1, aquaporin-1; GFAP, glial fibrillary acidic protein; PERIPH, peripherin; and RECA, rat endothelial cell antigen-1. Scale bars, 10 µm.
Colocalization analysis demonstrated that AQP1 was associated with peripherin, a marker for unmyelinated neuronal fibers (Fornaro et al. 2008; Goldstein et al. 1991). The intensity of the colocalization was strong in laminae I and II (arrows in Fig. 2AIII). The immunolabeling intensity decreased and was restricted to linear structures in lamina III (data not shown). Although colocalization between AQP1 and peripherin was obvious in laminae I and II, a large fraction of AQP1 labeling did not colocalize with peripherin (Fig. 1AI–1AIII). In white matter, AQP1 was colocalized with RECA-1, an endothelial cell marker (Duijvestijn et al. 1992), mainly in the immediate vicinity of the glia limitans in the white matter (Fig. 2CI–2CIII). Some co-labeled signals were also observed in lamina I and II of the dorsal horn in the gray matter (Fig. 1BI–1BIII). In contrast, fiber-like or filamentous structures labeled by AQP1 in lamina V (Fig. 1C) were not co-labeled with any of the examined antibodies (peripherin, RECA-1, GFAP, and NF200).
AQP4 Expression
Like AQP1 expression, the immunolabeling intensity of AQP4 was similar and unchanged from the cervical to lumbar levels. AQP4 was highly expressed in astrocytes in the white and gray matter. In white mater, AQP4 was expressed in a radial pattern protruding from the gray matter to the glia limitans along astrocytes processes (Fig. 3C, 3E and 3F). In gray matter, AQP4 was expressed in a laminar pattern, where the strongest immunolabeling was seen at the laminae I and II in the dorsal horns and glia limitans (Fig. 3A and 3B). AQP4 labeling was also abundant in lamina IX in the ventral horns, where it was localized to the astrocytes adjacent to capillaries and around motor neurons (Fig. 3C and 3D); it was not, however, observed in the neuronal somata (Fig. 3D). Other areas demonstrating AQP4 expression were in the surroundings of the central canal in lamina X (Fig. 4DI). Blood vessels were encircled by AQP4 labeling (Fig. 3D).
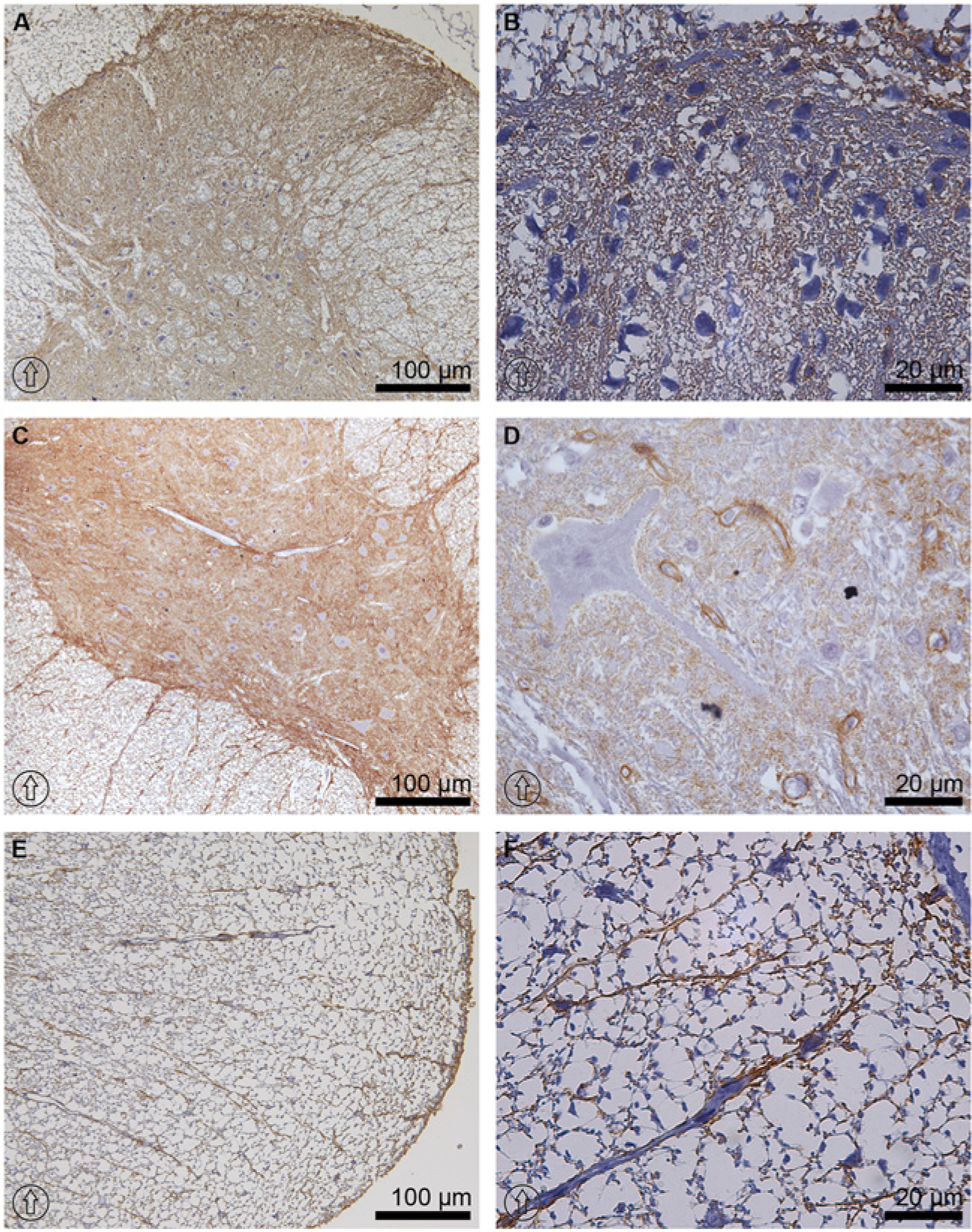
Immunoperoxidase microscopy of AQP4 at the thoracic level of rat spinal cord tissue. AQP4 was highly expressed along the whole spinal cord. It was most abundant in laminae I and II, and in the glia limitans of the dorsal horn (A and B). In the ventral horn, AQP4 labeling intensity was higher in lamina IX (C and D). Lamina IX consisted of large motor neurons, wherein the AQP4 signal was attributed to circular structures of capillaries and astrocytes processes, but was not found in the neuronal somata (D). In white matter, AQP4 was labeled in astrocytic fiber bundles protruding from the gray matter up to the glia limitans (E and F). The dorsal orientation of the spinal cord is indicated by encircled arrows.
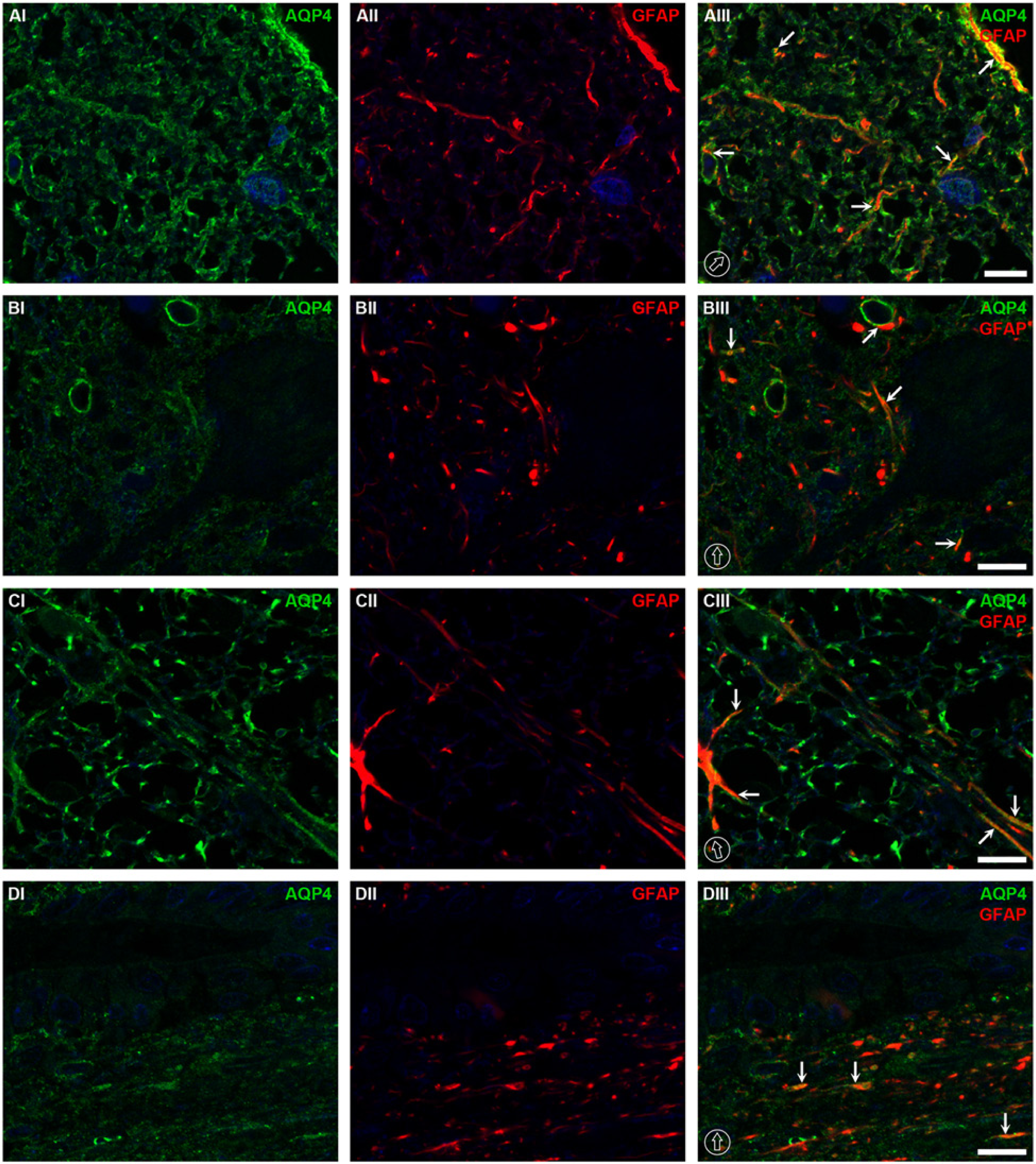
Colocalization of AQP4 and GFAP at the thoracic level of rat spinal cord tissue. In the dorsal horns, the highest co-expression between AQP4 and GFAP was observed in the glia limitans and laminae I and II (AI–AIII). Weaker colocalization of AQP4 and GFAP was present in the ventral horns, as demonstrated in lamina IX, which consists of large motor neurons (BI–BIII). The colocalization was also found at the circular structures of capillaries highlighted by AQP4 with red spots of GFAP, possibly astrocytic processes (BIII). In white matter, AQP4 was co-labeled with GFAP, especially along the astocytic processes (CI– CIII). In lamina X around the central canal, co-labeling of AQP4 and GFAP was also observed (DI–DIII). The dorsal orientation of the spinal cord is indicated by encircled arrows; colocalization by arrows. The blue signal in the images represents DAPI staining of nuclei. AQP4, aquaporin-4; GFAP, glial fibrillary acidic protein. Scale bars, 10 µm.
Colocalization analysis demonstrated that AQP4 was co-expressed with GFAP at the astrocytes in the laminae I and II in the dorsal horns and glia limitans of the gray matter (Fig. 4AI–4AIII), and in astrocytes in the white matter (Fig. 4CI–4CIII). Less-intense colocalization of AQP4 with GFAP was noticed in laminae IV–VIII in the gray matter, where AQP4 labeling was mostly expressed along the outline of capillaries, co-labeled with GFAP, presumably astrocytic end-feet (data not shown). Moreover, co-labeling of AQP4 and GFAP was also noticed as a linear structure of astrocytic processes, especially in the ventral horn of lamina IX close to large motor neurons (Fig. 4BI–4DIII). Around the central canal in lamina X, the degree of colocalization between AQP4 and GFAP was slightly higher than that in other parts of the gray matter, excluding laminae I and II, and the glia limitans (Fig. 4DI–4DIII).
In contrast, colocalization between AQP4 and glutamine synthase (GS) was rarely seen in the spinal cord (Fig. 5A, 5C, 5E and 5G), indicating that AQP4 was mostly present in astrocytes around the capillaries (i.e., end-feet) and astrocytic processes; GS is predominantly found in the astrocytic cell body (Fig. 5A, 5C and 5E).
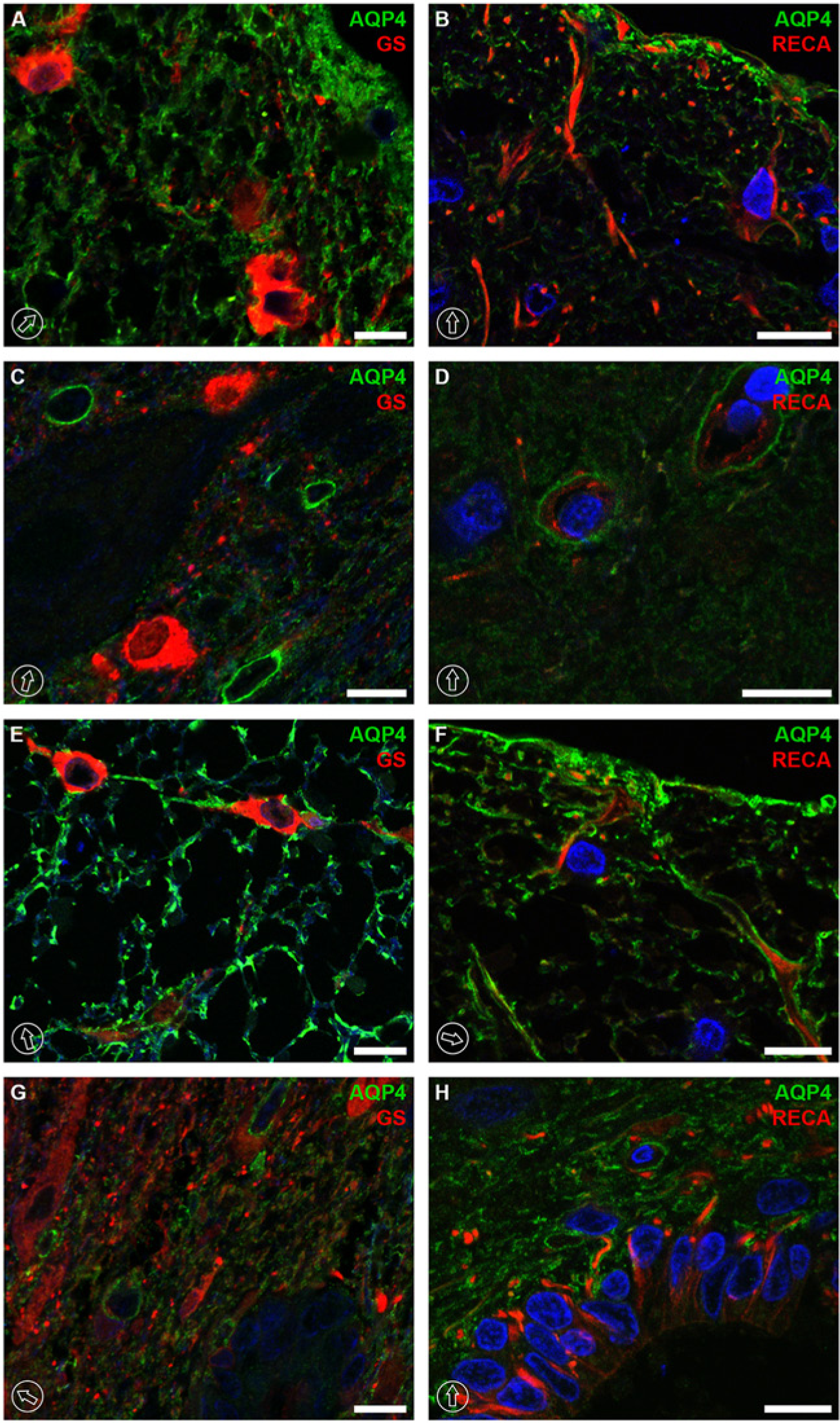
Colocalization of AQP4 and GS or AQP4 and RECA-1 at the thoracic level of rat spinal cord tissue. Colocalization was rarely seen between AQP4 and GS along the whole spinal cord in the dorsal horns (A), ventral horns (C), white matter (E) and around the central canal (G). GS was expressed in the cell body of astrocytes, whereas AQP4 was restricted to the astrocytic plasma membrane around the capillaries or astrocytic processes. No co-labeling was encountered between AQP4 and RECA-1 (B, D, F, and H). RECA-1 immunolabeling was encircled by AQP4 in vascular units in the ventral horns (D), or their labeling patterns were adjacent in laminae I and II (B), as well as in lamina X with the central canal (H) or in the white matter (F). The dorsal orientation of the spinal cord is indicated by encircled arrows. The blue signal in the images represents DAPI staining of nuclei. AQP4, aquaporin-4; GS, glutamine synthase; and RECA, rat endothelial cell antigen-1. Scale bars, 10 µm.
No co-labeling was encountered between AQP4 and RECA-1. RECA-1 immunolabeling was closely encircled by AQP4 in the neurovascular units (Fig. 5D). In white matter, RECA-1 immunolabeling was rarely found and mostly presented close to the glia limitans near the expression of AQP4 (Fig. 5F). In other laminae, such as I and II (Fig. 5B) as well as lamina X with the central canal(Fig. 5H), the immunolabeling signal was more or less adjacent to that of AQP4.
The expression of transient receptor potential cation channel subfamily V member 4 (TRPV4) has been reported previously in the kidney, liver and rat brain (Liedtke et al. 2000; Strotmann et al. 2000; Delany et al. 2001), with remarkable analogous expression to that of AQP4 in the superficial layers of the neocortex and the astrocytic end-feet (Benfenati et al. 2007). Thus, we examined whether immunolocalization of TRPV4 is associated with AQP4 in the spinal cord. The results demonstrated that TRPV4 immunolabeling was similar to the expression pattern of AQP4, but colocalization between the two proteins was not clearly seen. In the gray matter of spinal cord, TRPV4 expression represented a similar distribution to that of AQP4 (TRPV4 in Fig. 6A and 6B vs AQP4 in Fig. 3A and 3B), but it was not present in the glia limitans (Fig. 6A and 6B). In the ventral horns, TRPV4 labeling was found in the immediate vicinity of the plasma membrane of neuronal cells and in the fiber-like structures projecting toward the white matter (Fig. 6C and 6D). Moreover, TRPV4 immunolabeling was seen in the neuronal cell bodies (arrowheads in Fig. 7B). Importantly, no colocalization between TRPV4 and GFAP was found in AQP4-rich structures of laminae I and II in the dorsal horns (Fig. 7A). Furthermore, no clear overlapping between TRPV4 and AQP4 was observed in the laminae of the dorsal horn (Fig. 7C) or ventral horn (Fig. 7E) in the gray matter, white matter (Fig. 7F), at the border between the gray and white matter close to the sulcal artery (Fig. 7GI-GIII) or in other gray matter regions, as presented in lamina VIII (Fig. 7HI–7HIII).
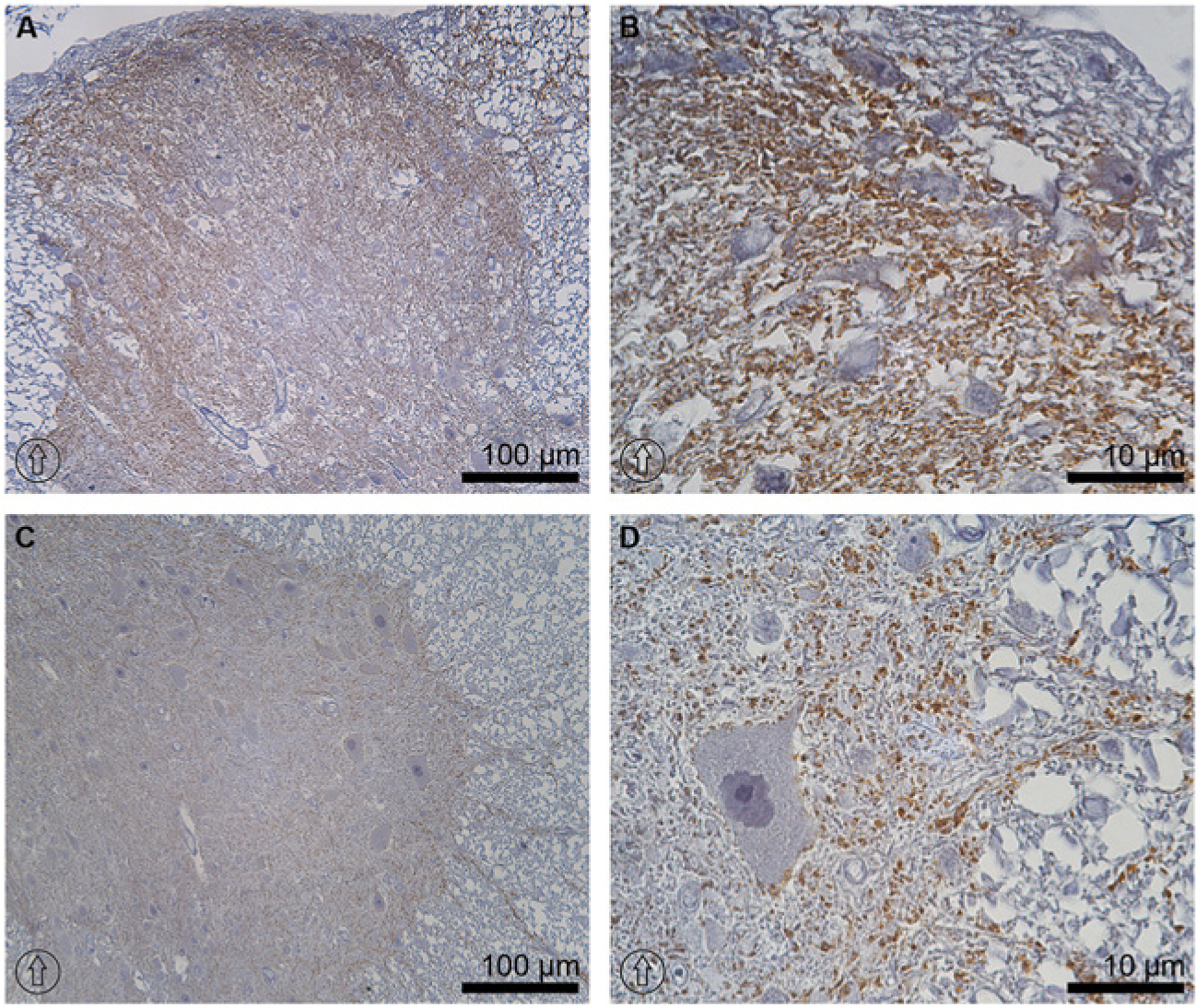
Immunoperoxidase microscopy of TRPV4 at the thoracic level of rat spinal cord tissue. The labeling of TRPV4 was seen in the dorsal horn of the gray matter, with slightly higher intensity in laminae I and II (A). No labeling of TRPV4 was seen in the glia limitans or in the dorsolateral fasciculus contiguous to lamina I (B). TRPV4 signal was found in the immediate vicinity of the neuronal plasma membrane in the ventral horn and in the fiber-like structures projecting toward the white matter (C and D). The dorsal orientation of the spinal cord is indicated by encircled arrows.
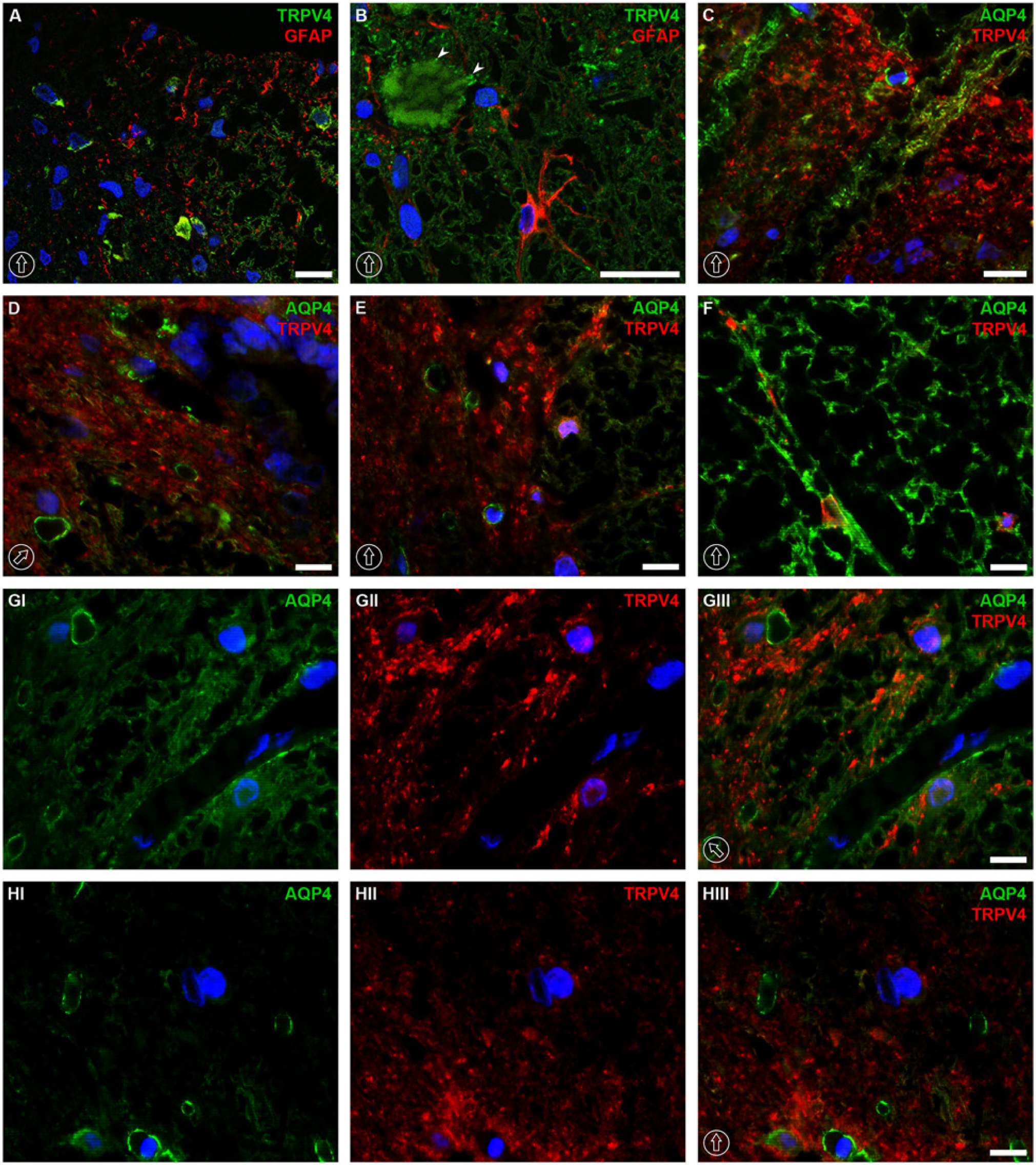
Colocalization of AQP4 and TRPV4 or TRPV4 and GFAP at the thoracic level of rat spinal cord tissue. No colocalization was noticed between TRPV4 and GFAP in the dorsal horn (A) or in the ventral horn (B). AQP4 was not colocalized with TRPV4 in AQP4-enriched structures in the dorsal horn (C), surroundings of the central canal (D), ventral horn (E) or in the white matter (F). The lack of colocalization between TRPV4 and AQP4 was also demonstrated at the border between the gray matter and white matter close to sulcal artery (GI–GIII) and in lamina VII of the gray matter (HI–HIII). The dorsal orientation of the spinal cord is indicated by encircled arrows; arrowheads in panel B indicate TRPV4 labeling in neuronal cell body. The blue signal in the images represents DAPI staining of nuclei. AQP4, aquaporin-4; GFAP, glial fibrillary acidic protein; and TRPV4, transient receptor potential cation channel subfamily V member 4. Scale bars, 10 µm.
AQP1 and AQP4 Expression
In the spinal cord, the immunolabeling intensity of AQP1 was overwhelmed by that of AQP4. The characteristic patterns of AQP1 and AQP4 labeling were compared. The main structure where AQP1 was abundantly expressed was the dorsal horn near the glia limitans (Fig. 1A and 1B and Fig. 8B), whereas AQP4 was abundantly expressed along the whole glia limitans in the rat spinal cord (Fig. 3A and 3E). AQP1-labeled structures co-labeled with RECA-1 in white matter were presumably penetrating arterioles from pial arteries (Fig. 2CI–2CIII). These arterioles were surrounded by GFAP-expressing astrocytic end-feet (Fig. 2F). In contrast, no colocalization was found between the structures labeled by RECA-1 and AQP4 (Fig. 5F), where AQP4 labeled astrocytes and RECA-1 labeled endothelial cells in arterioles, respectively. In contrary, AQP1 and RECA-1 co-expression was not found in the neurovascular units of the spinal cord (Fig. 8C), whereas the neurovascular units were strongly encircled by AQP4 (Fig. 5D).
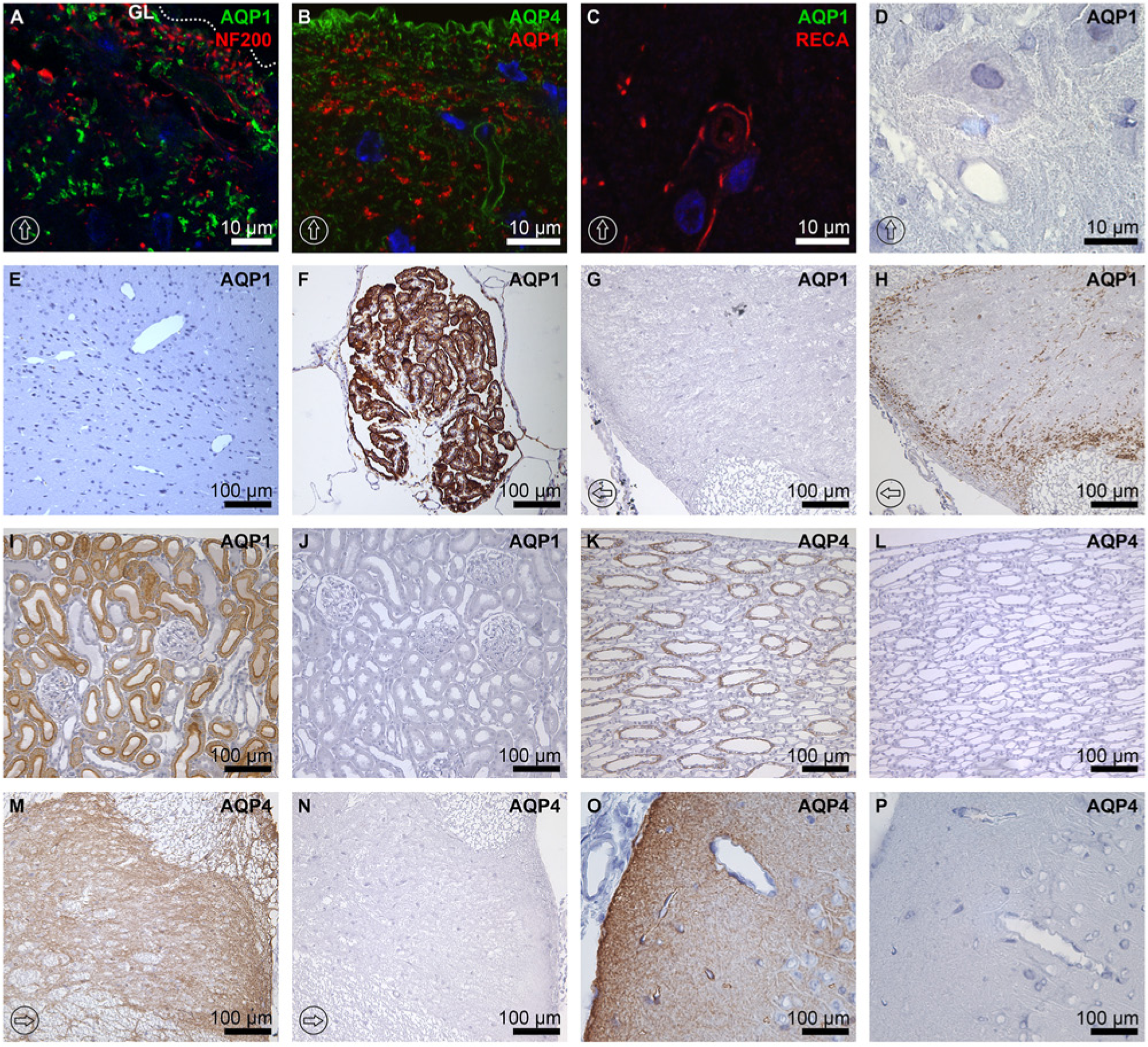
Comparison of AQP1 and AQP4 labeling and peptide pre-absorption control study. AQP1 was immunolabeled in dorsal horn in a scattered spot pattern, especially in laminae I and II, but it was not expressed in the glia limitans of the dorsal horn (GL in panel A, indicated by a dotted line) and was not co-labeled with NF200 (A) or AQP4 (B). No AQP1 labeling was detected along the vascular units in the gray matter labeled with RECA-1 (C), which were tightly encircled by AQP4 (Figure 5D). AQP1 immunolabeling was not detected in vascular structures in the gray matter of the spinal cord (D) or in the neuronal cells of the brain (E), whereas the epithelium of the choroid plexus was abundantly labeled by AQP1 (F). (G–P) Peptide pre-absorption control study. AQP1 and AQP4 antibodies labeled the proximal tubule of the kidney cortex (I) and collecting ducts (K), respectively, whereas no labeling was seen with peptide-pre-absorbed antibodies (J and L). Moreover, AQP1 and AQP4 antibodies labeled the rat spinal cord (H and M) and AQP4 antibodies labeled the rat brain (O), respectively, whereas no labeling was seen with peptide-pre-absorbed antibodies (G, N, and P). AQP1 control staining in the brain was made at approximately the position of Bregma -4.52 mm in (F), whereas (E) represents the parietal association cortex. The dorsal orientation of the spinal cord is indicated by encircled arrows; GL and dotted lines in panel A indicate the glial limitans. The blue signal in the images represents DAPI staining of nuclei. AQP1, aquaporin-1; AQP4, aquaporin-4; NF200, neurofilament protein 200 kDa; and RECA-1, rat endothelial cell antigen-1.
Importantly, no colocalization was seen between AQP1 and NF200, a marker for myelinated neuronal fibers (Groeneweg et al. 1993), in any parts of spinal cord (Fig. 8A), and no AQP1 immunolabeling was seen in the neuronal somata (Fig. 8D). Moreover, AQP1 was not colocalized with GFAP, an astrocyte marker (Groeneweg et al. 1993), at any level of the rat spinal cord (Figure 2D-F).
Specificity of AQP1 and AQP4 Antibodies
Specificity of AQP1 and AQP4 antibodies was confirmed by immunoperoxidase microscopic examination of the rat brain cortex, choroid plexus, kidney, and spinal cord (Fig. 8E–8P). AQP1 labeling was not seen in the rat brain cortex (Fig. 8E), whereas AQP1 was abundantly expressed in the choroid plexus epithelium (Fig. 8F). Moreover, AQP1 was highly expressed in the rat kidney proximal tubules (Fig. 8I) and spinal cord (Fig. 8H), whereas peptide pre-absorbed AQP1 antibodies did not label the proximal tubules (Fig. 8J) or spinal cord (Fig. 8G). AQP4 expression was seen in the kidney collecting duct principal cells (Fig. 8K), whereas the labeling was not seen in the sections labeled by peptide pre-absorbed AQP4 antibodies (Fig. 8L). Moreover, AQP4 labeling was seen in the rat spinal cord (Fig. 8M) and in the rat brain (Fig. 8O), whereas the labeling was abolished by peptide pre-absorbed AQP4 antibodies (Fig. 8N and 8P), indicating the specificity of the AQP1 and AQP4 antibodies.
Discussion
We demonstrated that AQP1 was abundantly expressed in laminae I and II of the dorsal horn, consistent with previous studies (Oshio et al. 2006; Nesic et al. 2008; Shields et al. 2007). Other parts of the gray matter showed weak AQP1 labeling in a form of dispersed dots, mostly encountered in the medial boundary of the dorsal horn and lamina X. AQP1 expressed in laminae I and II was co-labeled with peripherin, a marker for unmyelinated neuronal fibers (Fornaro et al. 2008; Goldstein et al. 1991). Interestingly, approximately two thirds of AQP1 expressed in laminae I and II was not co-labeled by peripherin, whereas peripherin expression almost fully overlapped with that of AQP1 labeling. The co-expression was also detectable in some fiber-like structures in lamina III, albeit with a lower labeling intensity. The results suggest that AQP1 is likely to be expressed in nocioceptive C fibers of the dorsal horn, consistent with previous studies (Shields et al. 2007; Zhang and Verkman 2010) reporting the co-expression of AQP1 and nocioceptive fiber markers (i.e., substance P-positive neurons) in the dorsal root ganglions with nearly 90% coverage.
In this study, a noticeable finding for AQP1 expression was its colocalization with an endothelial cell marker RECA-1 (Duijvestijn et al. 1992) in both the white matter (Figure 1I) and gray matter (Figure 1H), close to the glia limitans. The structures co-labeled by AQP1 and RECA-1 were likely to be the small arterioles and capillaries branching from the arterial vasocorona, which penetrates into the spinal cord core, similar to the AQP1 expression in the vasculature of other organs (Nielsen et al. 1993). But AQP1 expression in endothelial cells was weaker or undetectable at sites where contact was made with astrocytes. In contrast, AQP1 expression was not seen in endothelial cells of the neurovascular units in the spinal cord, consistent with the findings observed in the brain (Nielsen et al. 1993; Kobayashi et al. 2001). In the brain, reports indicate an absence of AQP1 expression in endothelial cells, the cells that constitute the neurovascular units and blood-brain barrier (Nielsen et al. 1993; Kobayashi et al. 2001). This finding was also confirmed by our observations in the present study, where we found that AQP1 labeling was only seen in the choroid plexus of the rat brain, but not in endothelial, neuronal, or astroglial cells of the rat brain cortex. However, a few previous reports show that AQP1-labeled blood vessels are occasionally found in the cerebral parenchyma of rat and human brains (Saadoun et al. 2002; Wilson et al. 2010), and this needs to be further examined.
The role of AQP1 expression in the spinal cord also needs to be elucidated. In the brain, the localization of AQP1 suggests an important role of AQP1 in CSF production and in nociceptive stimulation. AQP1-null mice demonstrated significantly decreased swelling and shrinkage of the choroid plexus epithelium in response to perfusion with hypotonic or isotonic artificial CSF. In addition, the CSF production rate and intracranial pressure in response to focal insult was lower in AQP1-null mice (Oshio et al. 2005). On the contrary, only a few attempts have been made to determine the functional role of AQP1 in the spinal cord and dorsal root ganglion. The studies on AQP1-null mice suggest that AQP1 might be involved in the process of acute thermal and chemical pain (Oshio et al. 2006). These suggestions were based on the findings of impaired firing of action potentials in small dorsal root ganglion neurons (Zhang and Verkman 2010). In contrast, Shields et al. (2007) did not find significant differences in pain processing between AQP1-null and wild type mice. Interestingly, AQP1 could be involved in the swelling of AQP1-expressing cells in the spinal cord; i.e., unmyelinated neuronal cells after spinal cord injury. Indeed, Nesic et al. (2008) demonstrated that AQP1 was up-regulated in surviving neurons after spinal cord injury, and hence the role of AQP1 needs to be further investigated, particularly in spinal cord injury patients.
We demonstrated that AQP4 expression was found in laminae I and II, at the boundaries of the ventral horns, and in the regions surrounding the central canal and glia limitans. In particular, AQP4 was expressed in the cell membrane and processes of astrocytes around neurons and blood vessels, consistent with previous studies (Vitellaro-Zuccarello et al. 2005). AQP4 was colocalized with GFAP at all levels of the spinal cord and, importantly, AQP4 labeling was observed close to neuronal somata and blood vessels, but was not colocalized with neuronal markers (NF-200 and peripherin) or endothelial cell markers (RECA-1). In brain tissues, previous studies have shown that AQP4 is highly polarized to perivascular end-feet of astrocytes (Nielsen et al. 1997; Rash et al. 1998), whereas, in the spinal cord, AQP4 is more evenly expressed in the cell membranes. This expression pattern was particularly seen in the white mater, whereas the co-labeling with GFAP became noticeably decreased in laminae IV–VIII of the gray matter.
The functional role of AQP4 in the CNS, particularly with regards to the formation and resolution phase of brain edema, has been extensively studied. AQP4-deficient mice exposed to cytotoxic brain edema show improved neurological outcomes and longer survival rate than wild-type mice (Manley et al. 2000; Papadopoulos and Verkman 2005). However, several other studies have demonstrated opposing results. For example, AQP4-knockout mice manifested reduced water elimination, which resulted in increased intracranial pressure, inferior neurological scoring and marked hydrocephalus as compared with their wild-type counterparts in the disease models of focal cortical-freeze injury, intraparenchymal saline infusion, brain tumor and obstructive hydrocephalus (Papadopoulos et al. 2004a; Bloch et al. 2006). The outcome induced by AQP4 deletion signifies the bidirectional water flux through AQP4, which is driven by osmotic gradients.
The observed characteristic expression of AQP4 with a less polarized and more spatial distribution in the astrocytes in the spinal cord as compared to that seen in the brain might suggest a different role for AQP4 in the spinal cord. The main function of AQP4 in physiological conditions could be water transport from the blood or the CSF to the spinal cord parenchyma or vice versa. In pathological conditions of spinal cord injury, a biphasic change in AQP4 expression levels was demonstrated in astrocytes, revealing early down-regulation and subsequent up-regulation of the protein (Nesic et al. 2006). AQP4 up-regulation associated with activated astroglial cells suggests strong implications for AQP4 in the development of neuropathies like cell swelling or persistent pain progression (Nesic et al. 2005; 2006; 2010; Nagelhus et al. 2013).
In summary, we demonstrated the immunolocalization of AQP1 and AQP4 in the rat spinal cord. The expression pattern and labeling intensity of AQP1 and AQP4 were unchanged along all levels of the spinal cord. AQP1 was co-labeled with markers for unmyelinated neuronal fibers and endothelial cells in small blood vessels penetrating through the glia limitans, but not with GFAP, an astrocytic marker. AQP4 was exclusively localized to astrocytes, consistent with labeling in the brain; however, AQP4 expression in the spinal cord exhibited a less polarized and more spatial distribution than that of brain astrocytes. The role of AQPs in the spinal cord needs further investigation, particularly in terms of their role in spinal cord injury.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the
