Abstract
To investigate the possible role of vascular endothelial growth factor (VEGF) in the injured spinal cord, we analyzed the distribution and time course of the two tyrosine kinase receptors for VEGF, Flt-1 and Flk-1, in the rat spinal cord following contusion injury using a weight-drop impactor. The semi-quantitative RT-PCR analysis of Flt-1 and Flk-1 in the spinal cord showed slight upregulation of these receptors following spinal cord injury. Although mRNAs for Flt-1 and Flk-1 were constitutively expressed in neurons, vascular endothelial cells, and some astrocytes in laminectomy control rats, their upregulation was induced in association with microglia/macrophages and reactive astrocytes in the vicinity of the lesion within 1 day in rats with a contusion injury and persisted for at least 14 days. The spatiotemporal expression of Flt-1 in the contused spinal cord mirrored that of Flk-1 expression. In the early phase of spinal cord injury, upregulation of Flt-1 and Flk-1 mRNA occurred in microglia/macrophages that infiltrated the lesion. In addition, the expression of both receptors increased progressively in reactive astrocytes within the vicinity of the lesion, predominately in the white matter, and almost all reactive astrocytes coexpressed Flt-1 or Flk-1 and nestin. These results suggest that VEGF may be involved in the inflammatory response and the astroglial reaction to contusion injuries of the spinal cord via specific VEGF receptors.
Keywords
T
Vascular endothelial growth factor (VEGF) was originally regarded as a specific endothelial mitogen associated with angiogenesis, as well as a potent mediator of vascular permeability (Senger et al. 1983; Lennmyr et al. 1998). Recent studies have shown that VEGF is also a possible direct neurotrophic and neuroprotective factor (Sondell et al. 1999; Oosthuyse et al. 2001; Jin et al. 2002; Rosenstein et al. 2003; Sun et al. 2003; Han et al. 2004). Most of these studies have been performed on brain tissue that has undergone ischemic damage. Studies on the role of VEGF following spinal cord injury have been performed only by some groups. In a study of spinal cord contusion injury, Widenfalk et al. (2003) showed that VEGF treatment decreased the size of the lesion and the level of apoptosis, increased blood vessel density, and improved functional outcome. In addition, some studies have found that VEGF is upregulated in the injured spinal cord (Bartholdi et al. 1997; Sköld et al. 2000).
VEGF exerts its function through two high-affinity tyrosine kinases, the fms-like kinase (Flt-1, VEGF receptor-1) and the fetal liver kinase receptor (Flk-1, VEGF receptor-2) (Neufeld et al. 1999). Sköld et al. (2000) reported that VEGF receptors were induced in vessel-like structures and in some astrocytes following traumatic spinal cord injury. However, the temporal regulation and identification of the precise phenotypes of cells that express VEGF receptors in an in vivo model of spinal cord injury remain to be established.
We have therefore examined the spatiotemporal regulation of Flt-1 and Flk-1 mRNA in the rat spinal cord following acute contusion-induced spinal cord injury. The expression profiles for the ligand specificity of VEGF during insults should lead to a better understanding of the role of VEGF in spinal cord injury.
Materials and Methods
Animal Preparation
All experimental animal procedures were conducted with the approval of the Catholic Ethics Committee of the Catholic University of Korea and in accordance with the US National Institutes of Health “Guide for the Care and Use of Laboratory Animals” (NIH Publication No. 80-23, revised 1996). Adult male Sprague Dawley rats (270 ± 5 g) were used in this study. Surgical techniques used were similar to those described previously (Basso et al. 1996). The animals were deeply anesthetized intraperitoneally with 80 mg/kg ketamine and 10 mg/kg xylazine. The skin and muscles overlying the thoracic cord were separated and retracted under a dissecting microscope. Dorsal laminectomies at the level of the ninth thoracic vertebra (T9) were carried out to expose the dura mater. The impact rod of the New York University (NYU) weight-drop device was centered above T9 and dropped from a height of 25 mm to induce an incomplete or partial spinal cord injury. Following the lesion, the dorsal back musculature was sutured and the skin closed with surgical clips. After surgery, the animals were kept on a thermostatically regulated heating pad until completely awake. The urinary bladder of all rats was emptied manually twice a day until recovery of urinary function. Controls for these experiments were age-, sex-, and weight-matched laminectomy rats that underwent the same surgical procedures without having the weight dropped to induce injury. Animals with spinal cord injury and laminectomy controls were killed 1, 3, 7, or 14 days (n = 10 for each group at each time point) after the operation. At each time point, five rats were taken for RT-PCR analysis, and five for in situ hybridization histochemistry and immunohistochemistry. The animals were deeply anesthetized with 16.9% urethane (10 ml/kg) and killed by transcardial perfusion with a fixative containing 4% paraformaldehyde in 0.1 M phosphate buffer (pH 7.4). A 2-cm segment of spinal cord centered on the lesion/laminectomy site was removed and postfixed for 2 hr in the same fixative. After treatment with 30% sucrose solution, spinal cords were embedded in OCT compound and stored at −70C. Representative slides were stained with cresyl violet to assess tissue morphology and to determine the injury epicenter. For RT-PCR, animals were deeply anesthetized and decapitated. A 2-cm segment of spinal cord centered on the lesion/laminectomy site was removed and was quickly frozen in liquid nitrogen.
Oligonucleotide pairs for RT-PCR

GAPDH, glyceraldehyde-3-phosphate dehydrogenase.
Semi-quantitative RT-PCR
Total RNA was extracted from each cord sample using TRIzol reagent (Invitrogen; Carlsbad, CA). RNA samples were quantified by measuring optic absorbance at 260 nm with a spectrophotometer. First-strand cDNA was synthesized using Reverse Transcriptase M-MLV (Takara Korea Biomedical Inc.; Seoul, Korea). The reaction mixture consisted of 1 μg total RNA, 300 pmol Oligo (dT) primer, 0.5 mM dNTP mixture, 20 U RNase inhibitor, and 200 U RTase M-MLV (RNase H), all in 1 × RTase M-MLV buffer. The reaction mixture was incubated for 60 min at 42C and heated at 70C for 10 min. The mixture was then stored at −20C. Equal amounts (1 μl) of the reverse transcription products were then PCR amplified using Perfect Premix Version 2.1 (Ex Taq version; Takara Korea Biomedical, Inc.). Amplification commenced with denaturation at 94C for 4 min followed by 25–30 cycles at 94C for 30 sec, 58C for 30 sec, and 72C for 30 sec. The final extension was made at 72C for 10 min. One pmol of each primer was used in the amplification reaction. Ten μl of each PCR reaction product was electrophoresed into 1.5% (w/v) agarose gels containing ethidium bromide (1 μg/μl). For the semiquantitative measurements, we coamplified the Flk-1 and Flt-1 mRNAs with glyceraldehyde-3-phosphate dehydrogenase (GAPDH) mRNA and optimized the number of PCR cycles to maintain amplification in the linear range. RT-PCR products were quantified by photographic densitometry of the ethidium bromide–stained agarose gel, and Flt-1/GAPDH or Flk-1/GAPDH product ratios were calculated as indices of Flt-1 or Flk-1 mRNA expression. Five animals were used for PCR at each time point, and three measurements were made per animal. The primers used are listed in Table 1.
In Situ Hybridization Histochemistry
Antisense and sense riboprobes labeled with digoxigenin (DIG) and specific sequences for Flt-1 and Flk-1 were prepared using RT-PCR and in vitro transcription, as described previously (Choi et al. 2007). Briefly, total RNA was extracted from the hippocampi using TRIzol reagent (Invitrogen; Carlsbad, CA), and the RT-PCR product was cloned in a T-Easy vector (Promega; Madison, WI) and sequenced. The DIG-labeled sense and antisense Flt-1 or Flk-1 riboprobes were prepared by in vitro transcription using a DIG RNA labeling kit (Roche; Indianapolis, IN).
Horizontal cryostat sections (25-μm thick) of the spinal cord were hybridized with antisense or sense probes diluted in hybridization solution (150 ng/ml) at 52C for 18 hr. Hybridization was visualized using an alkaline phosphatase–conjugated sheep anti-DIG antibody (diluted at 1:2000) with 4-nitroblue tetrazolium chloride (0.35 mg/ml) and 5-bromo-4-chloro-3-indolyl phosphate (0.18 mg/ml) as substrates.
Double Labeling or Triple Labeling
Some sections were double labeled for glial fibrillary acidic protein (GFAP) or ED1; these are established markers for astrocytes and microglia/macrophages, respectively. After hybridization, as described above, the sections were incubated for 2 hr with biotin-conjugated mouse monoclonal anti-DIG antibody (Jackson ImmunoResearch, West Grove, PA; diluted 1:200) at room temperature. For double-immunofluorescence histochemistry, sections were incubated with a mouse monoclonal antibody to GFAP (Chemicon International, Inc., Temecula, CA; diluted at 1:500) or ED1 (Serotec Ltd, Oxford, UK; diluted at 1:50) overnight at 4C. The sections were then reacted with Cy3-conjugated streptavidin (Jackson ImmunoResearch; diluted 1:1000) and FITC-conjugated anti-mouse antibody (Jackson ImmunoResearch; diluted 1:50) for 2 hr at room temperature.
To identify those astrocytes coexpressing VEGF receptors and nestin, we performed triple labeling using in situ hybridization for Flt-1 or Flk-1 and immunohistochemistry for nestin and GFAP. For triple labeling, some hybridized sections were incubated in a mix of rabbit polyclonal antibody to GFAP (Chemicon International, Inc.; diluted at 1:500) and mouse monoclonal antibodies to nestin (Biogenesis, Poole, UK; diluted at 1:100). The sections were then incubated in combination with Cy3-conjugated streptavidin (Jackson ImmunoResearch; diluted at 1:1000), Cy5-conjugated goat anti-mouse antibody (Jackson ImmunoResearch; diluted at 1:1000), and FITC-conjugated donkey anti-rabbit IgG (Jackson ImmunoResearch; diluted at 1:1000) for 2 hr at room temperature. Slides were viewed with a confocal microscope (MRC-1024; Bio-Rad Laboratories, Hercules, CA) using 488-nm line of an argon laser (for FITC) and 543-nm line (for Cy3) and 633-nm line (for Cy5) of a helium/neon laser and LaserSharp software (Bio-Rad Laboratories). Images were converted to TIFF format, and contrast levels were adjusted using Adobe Photoshop v. 7.0 (Adobe Systems; San Jose, CA).
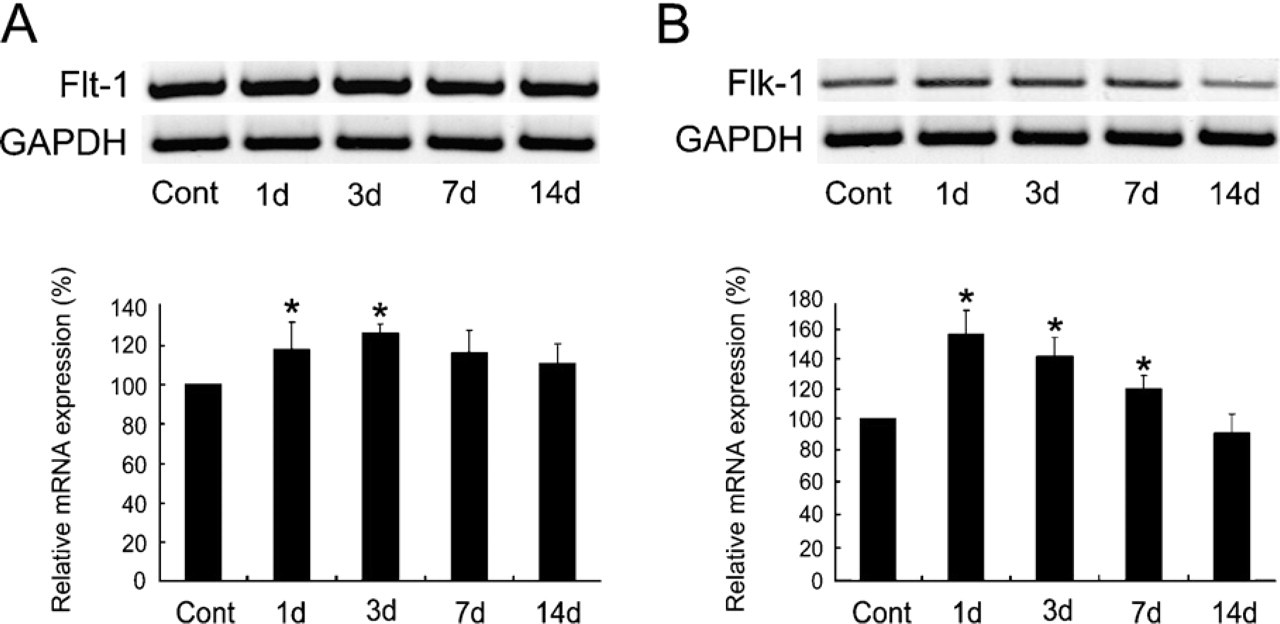
RT-PCR analysis of genes for Flt-1 (
Bromodeoxyuridine Administration and Triple Labeling
Sprague-Dawley rats (n = 8) were injected intraperitoneally each day for 14 days with the S-phase marker 5-bromo-2-deoxyuridine (BrdU; Sigma, St. Louis, MO) at a final concentration of 50 mg/kg body weight in phosphate-buffered saline (pH 7.5; buffering was with NaOH). Sections were obtained as described above and were double labeled for Flt-1 or Flk-1 in situ hybridization and BrdU/nestin immunohistochemistry. After in situ hybridization, BrdU labeling was detected by 30-min treatment of the sections with 2 N HCl. Neutralization was performed with 0.1 M borate buffer, and sections were then incubated with monoclonal rat anti-BrdU antibody (Accurate Chemical and Scientific Corporation; Westbury, NY) diluted at 1:50. Sections were incubated in mouse monoclonal antibodies to nestin (Biogenesis; diluted at 1:100), and then with a mixture of FITC-conjugated donkey anti-mouse IgG (Jackson ImmunoResearch; diluted at 1:50) and Cy3-conjugated goat anti-rat IgG (Jackson ImmunoResearch; diluted at 1:1000) for 2 hr at room temperature. Slides were viewed with a confocal microscope.
Results
Semi-quantitative RT-PCR for Flt-1 and Flk-1 Following Spinal Cord Injury
The semi-quantitative RT-PCR analysis for Flt-1 and Flk-1 showed that mRNA of both receptors was upregulated in the injured spinal cord. The expression level of Flt-1 transcripts slightly increased in the spinal cord of animals subjected to contusion injury as early as 1 day later (Figure 1A). The levels increased to maximum values at 3 days and then declined. The expression of Flk-1 mRNA in the injured spinal cord showed a temporal pattern similar to that of Flt-1, but increased expression following injury was more prominent (Figure 1B).
Temporal Profiles of Flt-1 mRNA Following Spinal Cord Injury
The distribution and cellular localization of Flt-1 mRNA in the contused spinal cord was examined using in situ hybridization histochemistry. In agreement with a previous study (Islamov et al. 2004), constitutive expression of Flt-1 mRNA was found in neurons, endothelial cells, and some glial cells in the spinal cord of laminectomy control rats (Figures 2A–2C). Adjacent sections in these animals were routinely processed for in situ hybridization with the Flt-1 sense probe, and no specific cellular labeling was observed (data not shown).
Light microscopy of cresyl violet–stained tissue revealed that contusion injuries led to a characteristic egg-shaped zone of necrosis that extended rostrocaudally for several spinal cord segments (Basso et al. 1996; data not shown). At day 1 after injury, Flt-1 mRNA signals increased, and by day 3 (Figure 2D), Flt-1 mRNA signals had preferentially increased in the gray and white matter in close proximity to the weight-drop injury. In particular, Flt-1 expression was observed in cells with a rounded morphology in close proximity to the lesion (Figure 2E). In addition, hybridization signals were found in gray and white matter, presumably in astrocytes, although these signals were much more prominent in white matter (Figure 2F). Flt-1-labeled cells were also closely associated with blood vessels and were more prominent around the lesion (Figure 2F).
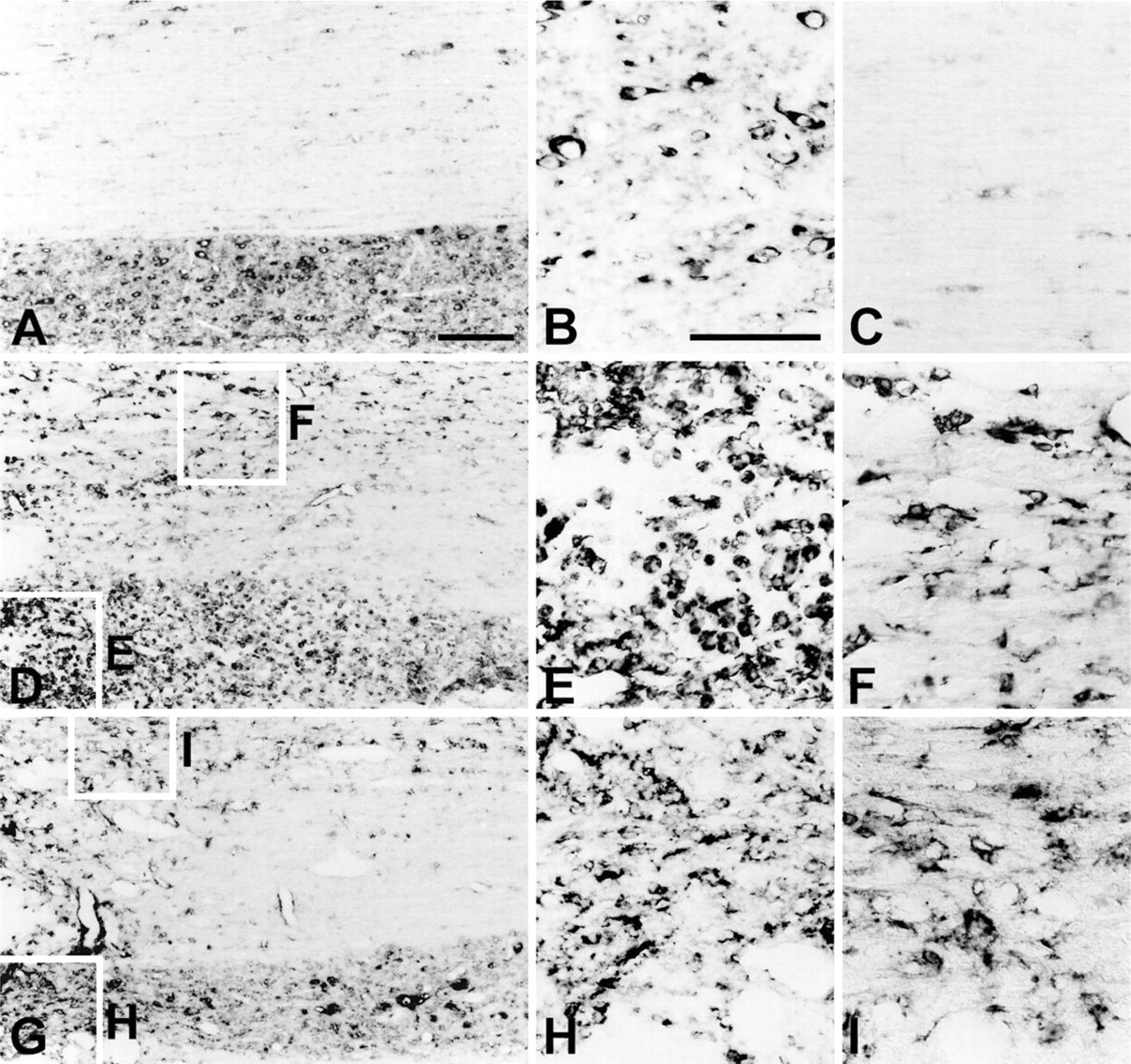
Changes in Flt-1 mRNA expression in the spinal cord of laminectomy controls (
At day 14 postinjury, the labeling intensity of Flt-1 mRNA remained at the level observed at day 3 (Figure 2G). Most signals were present in cells with astrocytic morphology located in the gray and white matter close to the lesion (Figures 2H and 2I). In addition, Flt-1 expression was observed in some cells with round or ameboid-shaped cell bodies.
Temporal Profiles of Flk-1 mRNA Following Spinal Cord Injury
The spatiotemporal distribution pattern of Flk-1 labeling in the contused spinal cord closely matched that of Flt-1. Hybridization signals for Flk-1 mRNA in laminectomy control animals were localized in neurons, endothelial cells, and some glial cells in the spinal cord (Figures 3A–3C), in agreement with previous findings (Islamov et al. 2004). At day 1 after spinal cord injury, Flk-1 mRNA hybridization was upregulated in gray and white matter around the lesion (data not shown), and became more evident at day 3 (Figures 3D–3F). This prominent enrichment was seen in rounded cells with the morphology of microglia/macrophages, especially in close proximity to the lesion (Figure 3E). In addition, Flk-1 mRNA hybridization signals were found in cells with astrocytic morphology in the vicinity of the lesion, predominately in the white matter (Figure 3F). At day 14 after spinal cord injury, Flk-1 mRNA was still abundant in the white and gray matter close to the lesion, and most signals were present in cells with astrocytic morphology (Figures 3G–3I).
The Phenotype of Cells Expressing Flt-1 and Flk-1
To identify the cell types expressing Flt-1 mRNA following spinal cord injury, a combination of in situ hybridization and immunohistochemistry was performed. As shown in Figures 4A–4C, in the contused tissue at day 3 after injury, most of the Flt-1-expressing cells in close proximity to the epicenter of the lesion were round, ED1-positive cells. In contrast, in the contused spinal cord at day 14 postinjury, the vast majority of Flt-1-expressing cells in close proximity to the epicenter of the lesion were labeled with GFAP (Figures 4D and 4E) and nestin (Figure 4F). A subpopulation of cells double labeled for Flt-1 and nestin showed BrdU labeling (Figures 4G–4I).
In addition, double labeling with Flk-1 mRNA and GFAP or ED1 revealed that most of the cells expressing Flk-1 at days 3 and 14 postinjury were ED1-positive microglia/macrophages and reactive astrocytes, respectively (Figures 5A–5E). Almost all reactive astrocytes expressing Flk-1 also expressed nestin (Figures 5D–5F), and some cells that were positively labeled for Flk-1 and nestin were also positively labeled for BrdU (data not shown).
Discussion
This study is the first to provide a detailed characterization of the time course and cellular localization of two tyrosine kinase receptors for VEGF, Flt-1 and Flk-1, in a rat model of spinal cord injury. Expression of Flt-1 and Flk-1 in in situ hybridization and the semiquantitative RT-PCR showed similar overall patterns in the contused spinal cord. Although mRNAs for Flt-1 and Flk-1 were constitutively expressed in neurons, endothelial cells, and some astrocytes in control animals, their upregulation was induced in association with reactive astrocytes and microglia/macrophages in the vicinity of the lesion within 1 day of contusion injury, and persisted for at least 14 days following injury. In the early phase of spinal cord injury, most of the increase in Flt-1 and Flk-1 expression could be attributed to ED1-positive cells, which had the morphology of activated macrophages and infiltrated the epicenter of the injury. However, the expression of both receptors progressively increased in reactive astrocytes in the vicinity of the lesion, predominately in the white matter.
VEGF receptors have been shown to be upregulated in the injured spinal cord (Sköld et al. 2000; Widenfalk et al. 2003; Islamov et al. 2004). However, these studies do not agree about the phenotypes of the cells expressing the VEGF receptors. Islamov et al. (2004) found increases in Flt-1 immunoreactivity in neurons following sciatic nerve injury. In contrast, Sköld et al. (2000) and Widenfalk et al. (2003) found enhanced expression of VEGF receptors in vascular endothelial cells and some astrocytes, and in endothelial cells, respectively. These discrepancies could be because of differences in experimental methods or in the animals. Our data support that VEGF receptors are upregulated in the injured spinal cord, and provide details of the expression patterns of these receptors with double-labeling techniques.
Several lines of evidence suggest that VEGF may be involved in the astroglial response in a variety of pathophysiological processes. First, VEGF and Flk-1 expression increases in reactive astrocytes in the hippocampus following entorhinal deafferentation (Wang et al. 2005). Second, VEGF induces astroglial proliferation and maintenance during repair following brain injury (Krum et al. 2002; Krum and Khaibullina 2003; Brockington et al. 2004; Yasuhara et al. 2004; Mani et al. 2005). Third, following spinal cord injury, VEGF and its receptors are upregulated in some reactive astrocytes surrounding the lesion (Sköld et al. 2000). Treatment with VEGF protein (Widenfalk et al. 2003) or treatment with VEGF protein combined with VEGF165 coding (Facchiano et al. 2002) improves functional outcome and decreases secondary degeneration in animal models of spinal cord injury, and reactive astrocytes play a pivotal role in the healing process after spinal cord injury (Okada et al. 2006). Our data support these views and provide evidence that a portion of the neuroprotective effects of VEGF are mediated via the astroglial reaction after spinal cord injury.
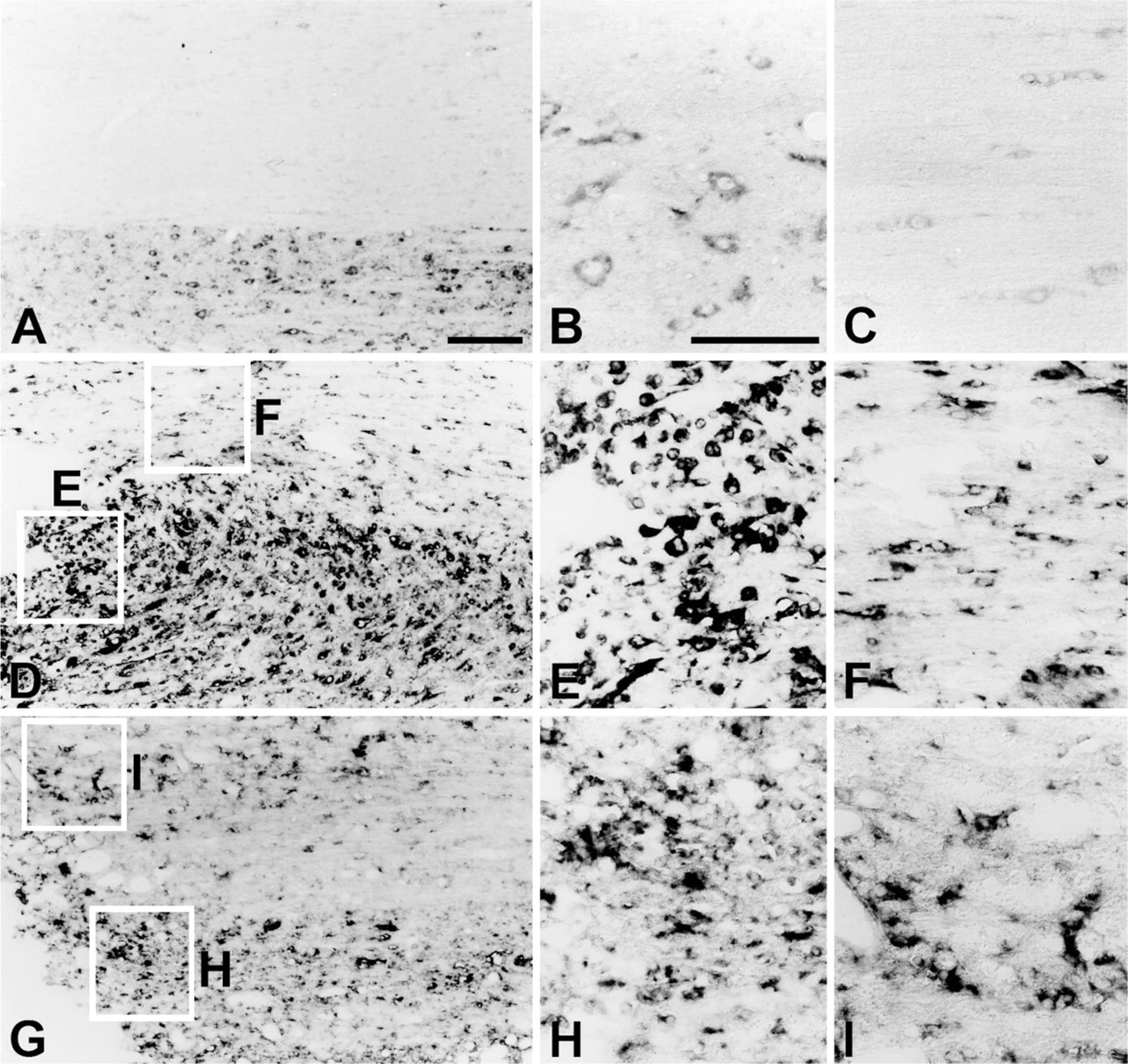
Changes in Flk-1 mRNA expression in horizontal sections of the spinal cord of laminectomy control (
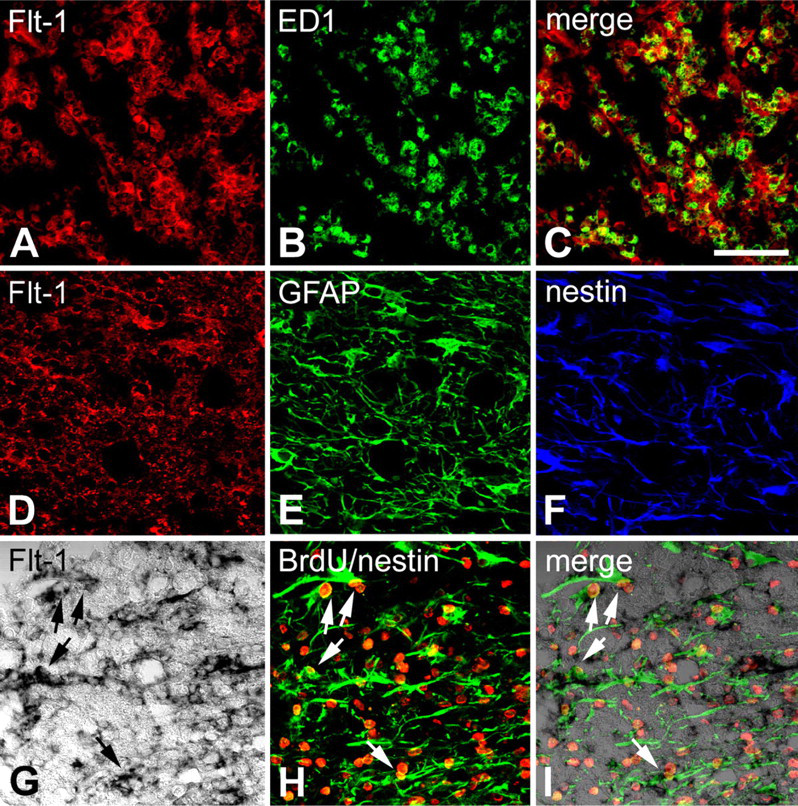
Identification of phenotypes of cells expressing Flt-1 in the spinal cord at day 3 (
Recent studies have shown that VEGF stimulates neurogenesis in the developing and adult nervous systems, in addition to playing a role in hematopoiesis, vasculogenesis, and angiogenesis (reviewed in Brockington et al. 2004; Yasuhara et al. 2004). VEGF receptors are expressed in neural progenitor cells in the developing retina (Yang and Cepko 1996) and in rat neural stem cells and cultured mouse cortical neurons (Jin et al. 2002; Maurer et al. 2003; Schänzer et al. 2004). In addition, VEGF stimulates neurogenesis in vivo and in vitro (Jin et al. 2002; Schänzer et al. 2004). Interestingly, in the injured rat spinal cord, reactive astrocytes show nestin immunoreactivity, which suggests that they may acquire the potential of neural stem cells (Lang et al. 2004). In our experiments, almost all reactive astrocytes expressing Flt-1 or Flk-1 in the contused spinal cord also expressed nestin. Moreover, our triple-labeling studies showed that a subpopulation of BrdU labeling overlapped with nestin/Flt-1 or nestin/Flk-1 expression. Overall, our data suggest that the Flt-1-or Flk-1/nestin/BrdU-labeled astrocytes may be a type of neural stem cell, and that VEGF may stimulate endogenous progenitors via VEGF receptors in the contused spinal cord. However, considering that VEGF does not cause any apparent difference in BrdU incorporation into astrocytes in animals with spinal cord contusion injury (Widenfalk et al. 2003), the possibility that nestin expression reflects the reversal of astrocytes to a more immature phenotype during the reactive state cannot be excluded.
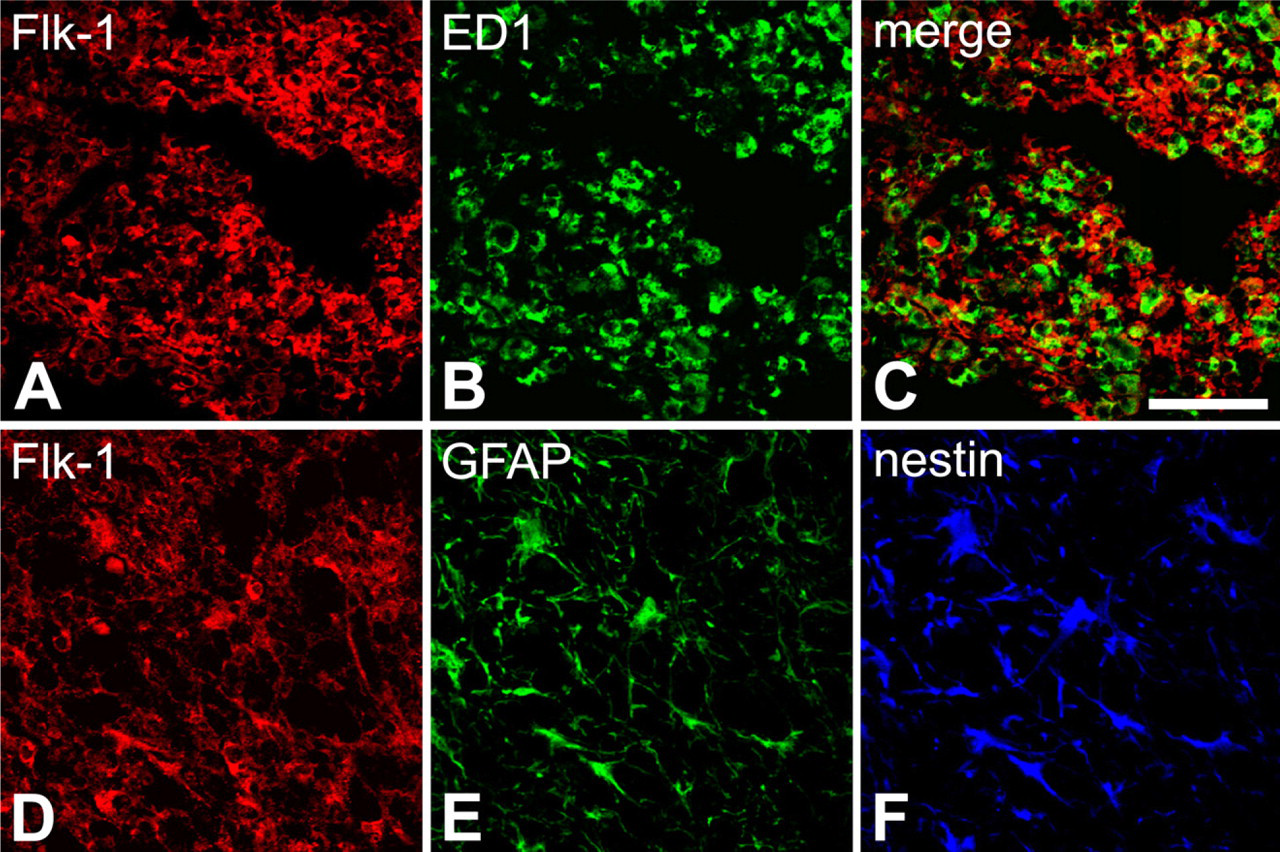
Identification of phenotypes of cells expressing Flk-1 in the spinal cord at day 3 (
Flt-1 and Flk-1 were also induced in ED1-positive cells, which had the same morphology as activated macrophages. The ED1 antigen is expressed in the membranes of phagolysosomes in microglia and macrophages and is upregulated during phagocytosis (Bauer et al. 1994; Damoiseaux et al. 1994). It has been proposed that administration of VEGF to the brain causes migration of monocytes/macrophages (Proescholdt et al. 1999; Croll et al. 2004). Flt-1 expressed on the surface of monocytes/macrophages is involved in ligand-dependent cell migration (Sawano et al. 2001). In addition, Forstreuter et al. (2002), in their in vitro study, found that microglial cells expressed Flt-1 mRNA and protein, and that VEGF induced chemotaxis and proliferation of microglial cells. Therefore, our data suggest that VEGF enables monocytes/macrophages to enter the spinal cord through breaches in the blood–spinal cord barrier or by a change in the permeability of this barrier via specific VEGF receptors, and that VEGF is involved in the activation and migration of intrinsic microglia following spinal cord injury.
In summary, our data show that both VEGF receptors are upregulated in microglia/macrophages and reactive astrocytes in the vicinity of the lesion in the contused spinal cord. Almost all reactive astrocytes expressing Flt-1 or Flk-1 also expressed nestin immunoreactivity, and some of these cells showed BrdU labeling. These data suggest that VEGF may be involved in inflammation and in the astroglial reaction via specific VEGF receptors following acute spinal cord contusion in rats.
Footnotes
Acknowledgements
This research was supported by a grant (M103KV010010-06K2201-01010) from the Brain Research Center of the 21st Century Frontier Research Program funded by the Ministry of Science and Technology, the Republic of Korea, and by a grant (SC3112) from the Stem Cell Research Center of the 21st Century Frontier Research Program funded by the Ministry of Science and Technology, Republic of Korea.
We gratefully acknowledge the technical assistance of Mr. Hong Lim Kim.
