Abstract
Transient receptor potential (TRP) cation channels are emerging in vascular biology. In particular, the expression of the capsaicin receptor (TRPV1) was reported in vascular smooth muscle cells. This study characterized the arteriolar TRPV1 function and expression in the rat. TRPV1 mRNA was expressed in various vascular beds. Six commercially available antibodies were tested for TRPV1 specificity. Two of them were specific (immunostaining was abolished by blocking peptides) for neuronal TRPV1 and one recognized vascular TRPV1. TRPV1 was expressed in blood vessels in the skeletal muscle, mesenteric and skin tissues, as well as in the aorta and carotid arteries. TRPV1 expression was found to be regulated at the level of individual blood vessels, where some vessels expressed, while others did not express TRPV1 in the same tissue sections. Capsaicin (a TRPV1 agonist) evoked constrictions in skeletal muscle arteries and in the carotid artery, but had no effect on the femoral and mesenteric arteries or the aorta. In blood vessels, TRPV1 expression was detected in most of the large arteries, but there were striking differences at level of the small arteries. TRPV1 activity was suppressed in some isolated arteries. This tightly regulated expression and function suggests a physiological role for vascular TRPV1.
Keywords
Introduction
Capsaicin is the active component of hot chili peppers and acts as an irritant in humans (Szallasi and Blumberg 1999). The receptor that mediates the hot painful feeling upon capsaicin exposure is transient receptor potential channel vanilloid 1, TRPV1, which was first cloned and identified in sensory neurons (Tominaga et al. 1998). These physiological effects of capsaicin identified TRPV1 as a promising therapeutic target to modulate pain perception, and an extensive pharmaceutical effort was made to develop TRPV1 antagonists to relieve pain (Szallasi and Blumberg 1999). Hundreds of patents were filed and thousands of molecules developed to modulate TRPV1. However, this effort did not result in a breakthrough in pain treatment because of the physiological off-target effects of the developed TRPV1 antagonists, with subsequent work revealing that TRPV1 is involved in body temperature maintenance in addition to its other functions (Gavva 2008; Holzer 2008; Szallasi and Sheta 2012).
TRPV1 expression has been identified in various tissues in addition to sensory neurons. In particular, TRPV1 was found in the central nervous system (Toth et al. 2005) and in the peripheral blood vessels (Lizanecz et al. 2006). Later research on the effects of TRPV1 stimulation in blood vessels suggested both dilation and constriction effects upon TRPV1 stimulation (Kark et al. 2008). TRPV1-mediated dilation was found to be related to the perivascular sensory neuronal terminals, which were thought to release neurotransmitters (CGRP, substance P) upon stimulation and mediate vasodilation (Zygmunt et al. 1999). The vasoconstrictive properties of TRPV1, however, were much less-well characterized. Nonetheless, we (Czikora et al. 2012; Kark et al. 2008) and others (Cavanaugh et al. 2011) have recently shown that functional TRPV1 is expressed in arteriolar smooth muscle cells, where its activation results in an increase in intracellular Ca2+ concentration and vasoconstriction.
TRPV1-6 channels are gaining increasing attention in vascular biology. These cation channels demonstrate some selectivity to Ca2+ (Baylie and Brayden 2011). However, there is little consistency in the reports regarding the role of TRPV1 in vascular biology, with reports suggesting that even the same arteries can respond to capsaicin by dilation or constriction depending on the conditions (Baylie and Brayden 2011). These opposing effects on vascular diameter were explained by its localization in sensory neuronal terminals (mediating dilation) and in vascular smooth muscle cells (Kark et al. 2008).
We performed a detailed study here to reveal functional TRPV1 expression in various vascular tissues of the rat. First, antibodies that were specific for sensory neuronal TRPV1 and vascular TRPV1 expression were identified and the expression characterized. The data revealed that TRPV1 expression is not uniform in vascular beds, with some vessels expressing TRPV1 while others not in the same tissue section. Moreover, TRPV1 responses to capsaicin were different in isolated arteries where TRPV1 appears to be highly expressed, suggesting a tight regulation of TRPV1 sensitivity in arteriolar smooth muscle.
Materials & Methods
Materials and Solutions
Chemicals were from Sigma-Aldrich, unless stated otherwise. Capsaicin (8-methyl-N-vanillyl-trans-6-nonenamide) was dissolved in ethanol. Norepinephrine and acetylcholine were dissolved in distilled water.
Animals, Anesthesia and General Preparation for In Vivo Experiments
Male Wistar Kyoto (WKY/NCrl) rats (Charles River, Isaszeg, Hungary) were fed ad libitum (chow from Szinbad Kft, Godollo, Hungary). Rats were 250-450 g when experiments were started. Rats were anesthetized by 50 mg/kg i.p. thiopental. Animal experiments were carried out at and approved by the University of Debrecen, Medical and Health Science Center, and were in accordance with the standards established by the National Institutes of Health.
Total RNA and RT-qPCR
Tissue samples were prepared as described later for cannulated arteries. Reverse transcription-coupled quantitative PCR was performed as described previously (Bai et al. 2007). Briefly, total RNA was prepared using Trizol reagent (Life Technologies; Budapest, Hungary) according to the manufacturer’s instructions. RNA was treated with DNase and 0.5 μg RNA was reverse transcribed using a High Capacity cDNA Reverse Transcription Kit (Applied Biosystems; Foster City, CA). cDNA was purified on QIAquick PCR cleanup columns (Qiagen, Valencia, CA). cDNA at 10-fold dilution was used for qPCR reactions. The quantitative PCR reactions were performed using a LightCycler 480 system (Roche Applied Science; Basel, Switzerland) and a qPCR supermix (Biocenter; Szeged, Hungary) with the following primers: TRPV1 (fwd: 5’-gaatgacaccatcgctctgc; rev: 5’- aagagggtcaccagcgtcat) and 36B4 as control (fwd: 5’- ccccgtgtgaggtcacagta; rev: 5’- atgatcagcccgaaggagaa). TRPV1 expression was normalized for 36B4 expression. Finally, the products of the PCR amplification were run on a 2% agarose gel to verify their size. To check for the amplification of primer dimers, the template was omitted from the controls and melting curve analysis was performed.
Immunohistochemistry
Tissue samples (dorsal root ganglia, gracilis muscle, mesenterium, femoral muscle, aorta, carotid artery and the ears) were dissected from the rat and embedded in Tissue-Tek O.C.T compound (Electron Microscopy Sciences; Hatfield, PA). Cryostat sections (10-µm-thick) were prepared, fixed in acetone for 10 min and blocked with normal goat sera for 20 min (1.5% in PBS, Sigma-Aldrich; St. Louis, MO). TRPV1 was stained with anti-capsaicin receptor antibodies. Antibodies were obtained from Alomone Labs (Jerusalem, Israel) (anti-TRPV1-C, 3rd loop) Calbiochem (San Diego, CA) (anti-TRPV1-N), Osenses (Keswick, Australia) (3rd loop, 4th loop) and Neuromics (Edina, MN) (N-terminal). Details and dilutions of the antibodies are shown in Table 1. Blocking peptides (synthesized based on the immunogenic TRPV1 fragment used to develop the antibodies) were also used in some cases. Binding of the TRPV1-specific antibodies was visualized using fluorescent secondary antibodies (Table 1) by a Zeiss Meta confocal microscope (Zeiss; Oberkochen, Germany). Tissue sections were also co-stained with anti-smooth muscle actin (Table 1) or with a neurofilament-specific antibody (Table 1) in the blocking buffer. Pictures were processed by ImageJ software (NIH; Bethesda, MD) to calculate cross-sectional areas. Cross-sectional areas were calculated by applying the AUTO mode for the dorsal root ganglia pictures in both the Threshold and the Particle analysis menus (the single manual adjustment set the maximum area to 1000).
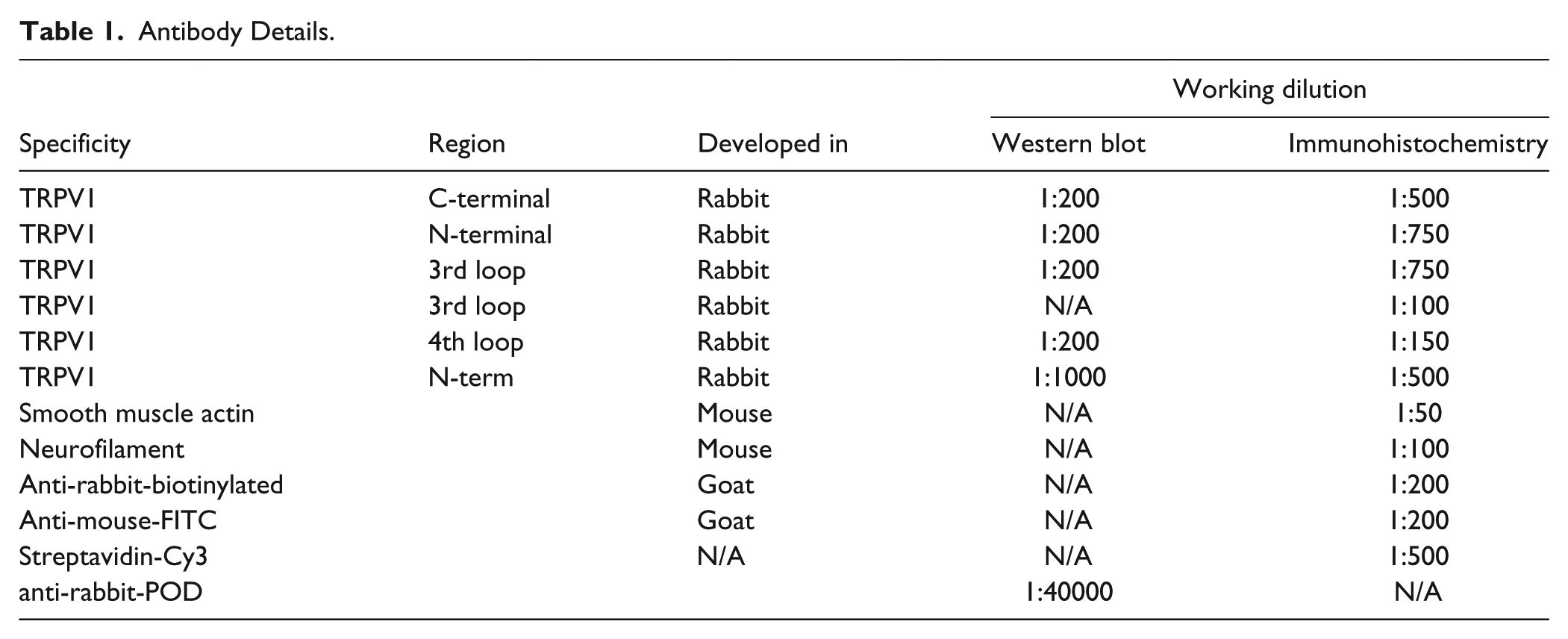
Antibody Details.
Western Blotting
Tissue samples (20 dorsal root ganglia and two carotid arteries) were dissected from the rat, pooled, and homogenized in 200 µl of SDS sample buffer (S3401, Sigma-Aldrich) using a glass tissue homogenizer. Cultured human embryonic kidney cells (HEK293, LGC Standards; Wesel, Germany) were transfected with a human TRPV1-expression plasmid (pdEYFP-C1 construct, RZPD; Berlin, Germany). HEK293 cells were maintained in Dulbecco’s modified Eagle’s medium (DMEM) supplemented with 10% heat-inactivated FBS, 2 mM glutamine, 100 U/ml penicillin, and 100 μg/ml streptomycin (all from Life Technologies) at 37C, 5% CO2. HEK293 cells (control and transfected) were collected from 100 mm diameter petri dishes and homogenized in 500 µl of SDS sample buffer. All homogenized samples were incubated at 100C for 10 min. Protein concentration was determined using a BSA standard. Protein (30 µg) was loaded onto 10% SDS-polyacrylamide gels and transferred onto nitrocellulose membranes. The membranes were then stained by a reversible protein staining dye (Ponceau S) and were cut into strips of the aforementioned samples and a prestained molecular weight standard (ProSieve QuadColor, Lonza; Rockland, MA) and probed with TRPV1-specific antibodies (Table 1). Blocking peptides (Table 1) were obtained from Alomone (ACC-030 antibody) and from EZBiolabs (Caramel, IN; PC547 antibody). Blocking peptides were used in a ratio of 1 µg peptide:1 µg antibody and were preincubated with their respective antibodies for 60 min at room temperature. All primary antibodies (Table 1) were indicated to work in western blotting, and we used dilutions suggested by the manufacturers (Table 1). Binding of the primary antibodies was detected by an anti-rabbit-POD secondary antibody (Sigma-Aldrich). Peroxidase reaction was detected by ECL (Western Lightning Plus ECL, PerkinElmer; Waltham, MA) and the signal was recorded by an imaging system (MF-Chemibis 3.2, Central European Biosystems; Budapest, Hungary). All membranes in the figures were developed together with an exposure time was 6 min. The intensity range was 0-15,984 (with recording in the range of 0-65,535).
Preparation of Cannulated Arterioles
Isolation of the skeletal (gracilis) muscle arterioles of the rat and diameter measurement of the arterioles were performed as described earlier (Lizanecz et al. 2006). Preparation of small mesenteric resistance arteries were performed likewise. The internal diameter of the arterioles was measured by video microscopy. Experiments were carried out in PSS (composition in mM: 110 NaCl, 5.0 KCl, 2.5 CaCl2, 1.0 MgSO4, 1.0 KH2PO4, 5.0 glucose and 24.0 NaHCO3 equilibrated with a gas mixture of 10% O2 and 5% CO2, 85% N2, at pH 7.4). After the development of spontaneous myogenic tone at 80 mmHg, arteriolar responses to acetylcholine (edothel dependent dilation, 1 nM–10 µM), to norepinephrine (vasoconstrictor, 1 nM–10 µM) and to capsaicin (TRPV1 agonist, 1 nM–10 µM) were measured. Agonists were applied in a cumulative fashion.
Measurement of Arteriolar Contractions under Isometric Conditions
Large arteries were prepared from the rat in Ca2+-free PSS. Vessel Segments (4-mm long) were fixed onto a contractile force measurement setup (DMT510A, Danish Myotechnology; Aarhus, Denmark). After fixing of the vessels on the setup in Ca2+-free PSS, the buffer was changed to PSS and vessels were stretched by 10 mN. Mounted arteries were incubated in PSS for 40–60 min (until force values were stabilized) at 37C. Experiments were initiated by the addition of the smooth muscle-dependent vasoconstrictive agent norepinephrine or U-46619 in a cumulative fashion (1 nM–10 μM). After reaching the maximal response, these agents were washed away and the vessels were incubated in PSS alone, until the contractile force decreased to the baseline level before the application of the vasoconstrictive agents. Finally, capsaicin was applied (1 nM–30 μM) to investigate TRPV1-–mediated vascular effects.
Data Analysis and Statistical Procedures
Arteriolar diameter was determined by measuring the distance between the intraluminal sides of the arteriolar wall (inner diameter). Data are shown as mean diameter ± S.E.M. Student’s t-tests were used to determine differences. Statistical analysis was performed using Microsoft Excel. P-values <0.05 were considered to be significant.
Results
Non-neuronal Expression of TRPV1
The expression of TRPV1 in vascular tissues was tested by RT-PCR (Fig. 1A) and qPCR (Fig. 1B) in isolated vascular preparations. TRPV1 expression was found to be the highest in dorsal root ganglia. Nonetheless, TRPV1 was also expressed in other tissues, albeit its expression was two orders of magnitude lower than that in dorsal root ganglia. TRPV1 mRNA expression was not detected in the isolated mesenteric artery (values were similar to those performed without template).
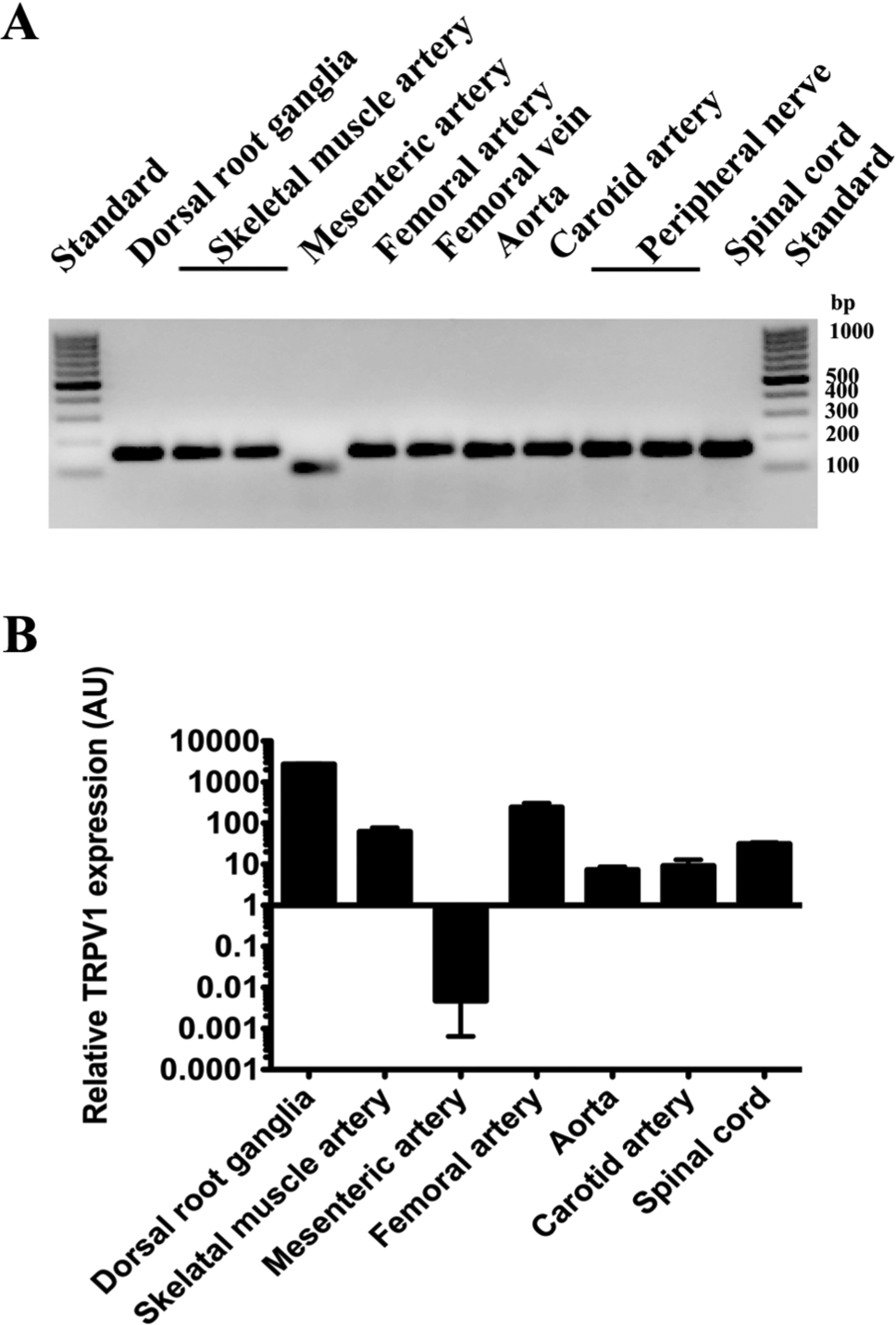
TRPV1 mRNA in peripheral tissues of the rat. TRPV1 expression was examined with RT-PCR (A) and qPCR (B) in peripheral tissues of the rat. Isolated mRNA (0.5 µg) from various tissue sources (isolated arteries, veins, nerves, dorsal root ganglia and spinal cord) was subjected to RT-PCR and qPCR with a primer set specific to rat TRPV1. (A) Reaction mixtures were loaded onto 2% agarose gels to separate PCR products. Bands at the apparent molecular size of 170 bp were in accordance with the expected size of the product, while the band in the mesenteric artery sample was nonspecific. (B) qPCR experiments revealed negligible expression of TRPV1 in mesenteric arteries (values were similar to those performed without template), but a reasonably high level of expression was found in other peripheral tissues (n=2–8; bars represent mean ± SEM).
Characterization of Antibodies Developed against TRPV1
A set of six antibodies developed against TRPV1 (Table 1) were tested on dorsal root ganglia of the rat. Among the six tested, two antibodies (anti-TRPV1-N and anti-TRPV1-C) stained specifically a subset of the neurons within the dorsal root, whereas three antibodies (Alomone 3rd loop, Osenses 3rd loop and Osenses 4th loop) did not give any cell-specific staining pattern and the last (Neuromics N-terminal antibody) had a rather nonspecific neuronal staining pattern under these conditions (Fig. 2). The anti-TRPV1-N and anti-TRPV1-C antibodies were tested in detail. Both the anti-TRPV1-N (red; Fig. 3A) and anti-TRPV1-C (red; Fig. 3B) antibodies stained a subset of cell bodies within the dorsal root ganglia of the rat. TRPV1-positive cells were also stained with a neurofilament-specific antibody (green; Fig. 3), although the intensity of the signal was weaker in TRPV1-expressing cells than TRPV1-negative cells. Images taken at a higher magnification in separate experiments confirmed this observation (Fig. 4A and Fig. 4C). TRPV1-specific immunostaining was negative when the anti-TRPV1 antibodies were pre-absorbed with their respective blocking peptides (anti-TRPV1-N, Fig. 4B; anti-TRPV1-C, Fig. 4D). The company datasheets for the TRPV1 antibodies (Fig. 2, Table 1) indicate that the antibodies are suitable for western blotting. Antibodies were therefore tested in western blots of homogenates of the dorsal root ganglia, carotid artery, and in HEK293 with or without TRPV1 overexpression (Fig. 5). Some of the antibodies (Alomone 3rd loop, Osenses 4th loop and Neuromics N-terminal, Fig. 5A-5C) gave only nonspecific signals (Fig. 5). Compared with the background staining (Fig. 5D), the anti-TRPV1-N antibody did not give any signal under these conditions (Fig. 5E). The anti-TRPV1-C antibody (Fig. 5F) was found to be applicable in western blotting, detecting TRPV1 in the dorsal root ganglia and in TRPV1-overexpressing HEK293 cells but not in untransfected HEK293 cells. Moreover, this TRPV1-specific signal disappeared in the presence of the blocking peptide.
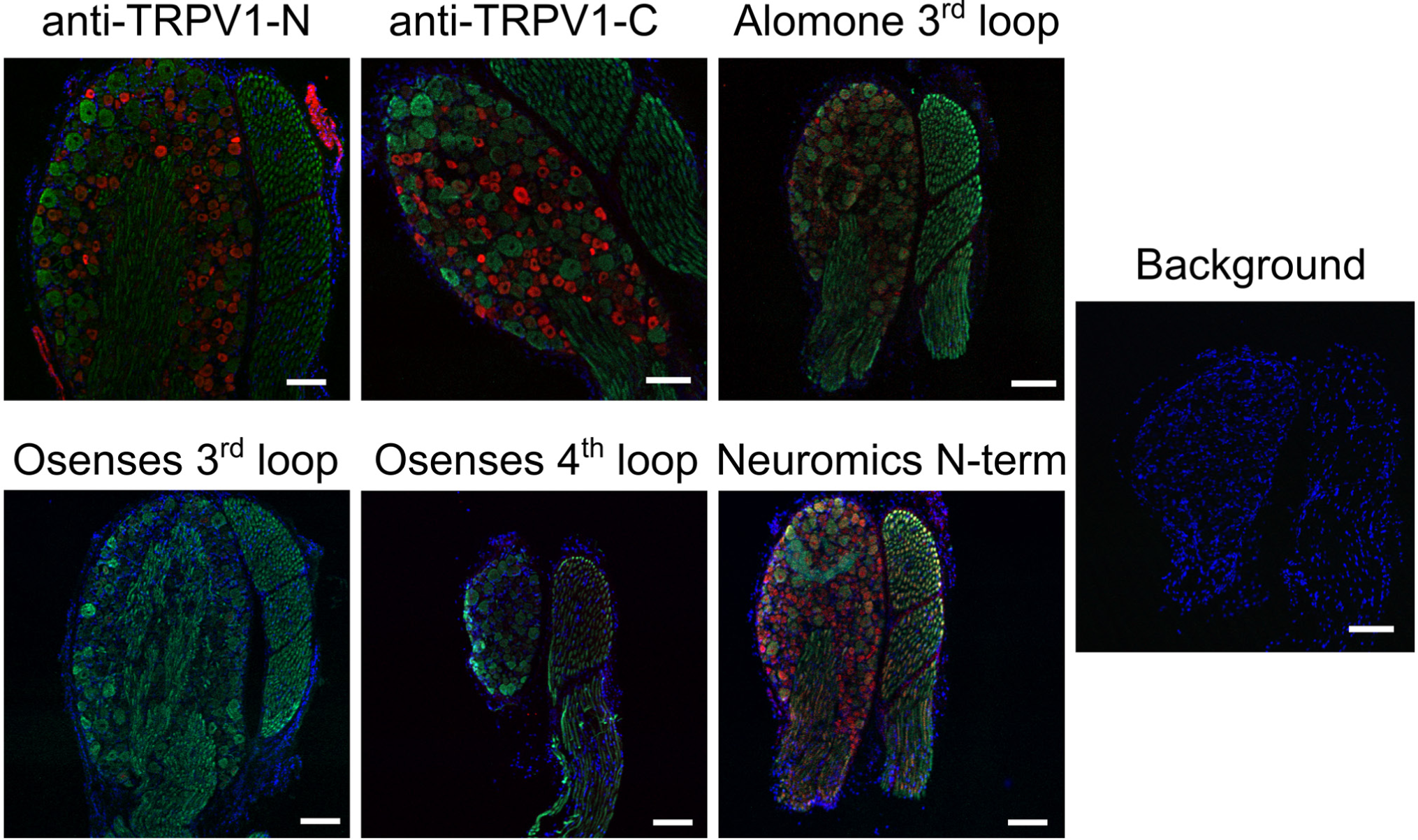
Specificity of TRPV1 antibodies. Six commercially available anti-TRPV1 antibodies were tested on dorsal root ganglia (cryostat sections) of the rat (red). Tissue sections were co-stained with a neurofilament-specific antibody (green, neurons). Nuclei were stained with a DAPI counterstain (blue). Background staining levels were checked by omitting the primary antibodies (and counterstaining with DAPI). Primary antibodies are indicated on the figure. Dilutions and details of the antibodies are summarized in Table 1. Bars represent 100 µm.
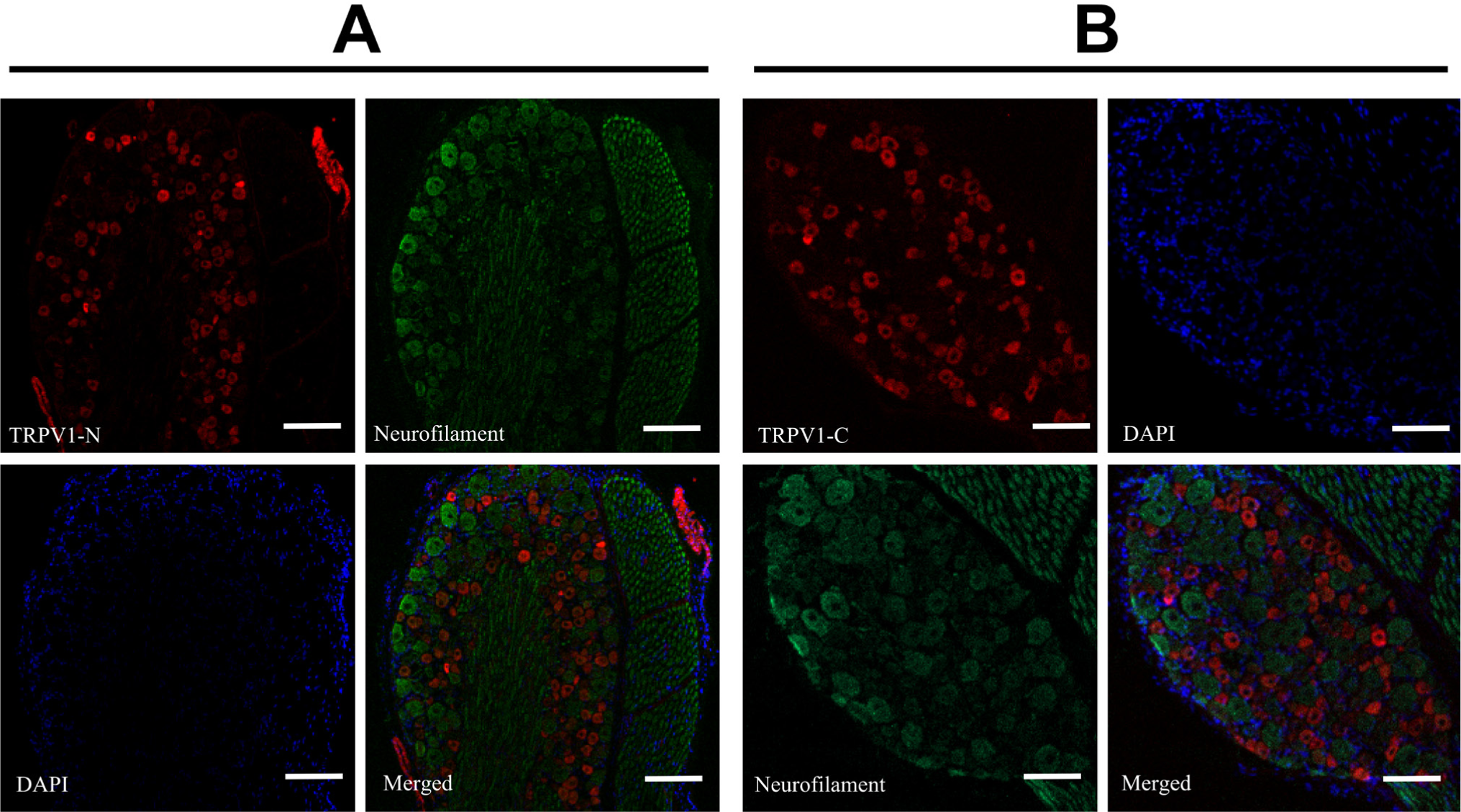
Colocalization of TRPV1 and neurofilament immunoreactivities. Rat dorsal root ganglia were stained with anti-TRPV1-N (A) and anti-TRPV1-C (B) antibodies (red), together with a neurofilament-specific antibody (green; neurons) and DAPI counterstain (blue; nuclei). The merged images for these three channels are shown. Bars represent 100 µm.
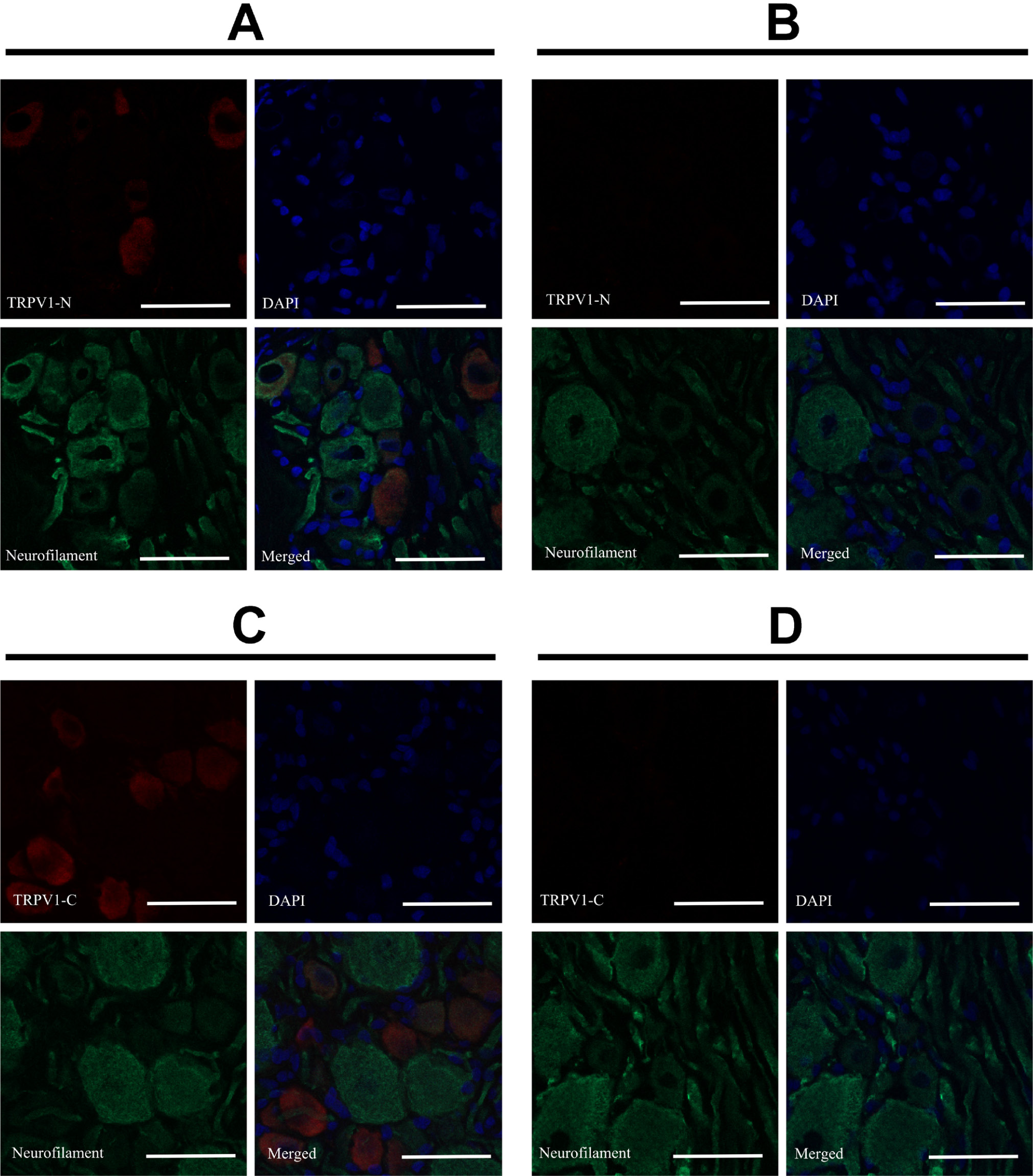
Specificity of neuronal TRPV1 staining. Dorsal root ganglia of the rat were stained with anti-TRPV1-N (A and B) or anti-TRPV1-C (C and D) antibodies (red); together with a neurofilament-specific antibody (green; neurons) and DAPI counterstain (blue; nuclei). Blocking peptides were synthesized according to the sequence of the immunogenic TRPV1 fragment to investigate the specificity of the TRPV1 staining. Positive staining disappeared when the antibodies were pre-incubated with the blocking peptides (B and D). Bars represent 50 µm.
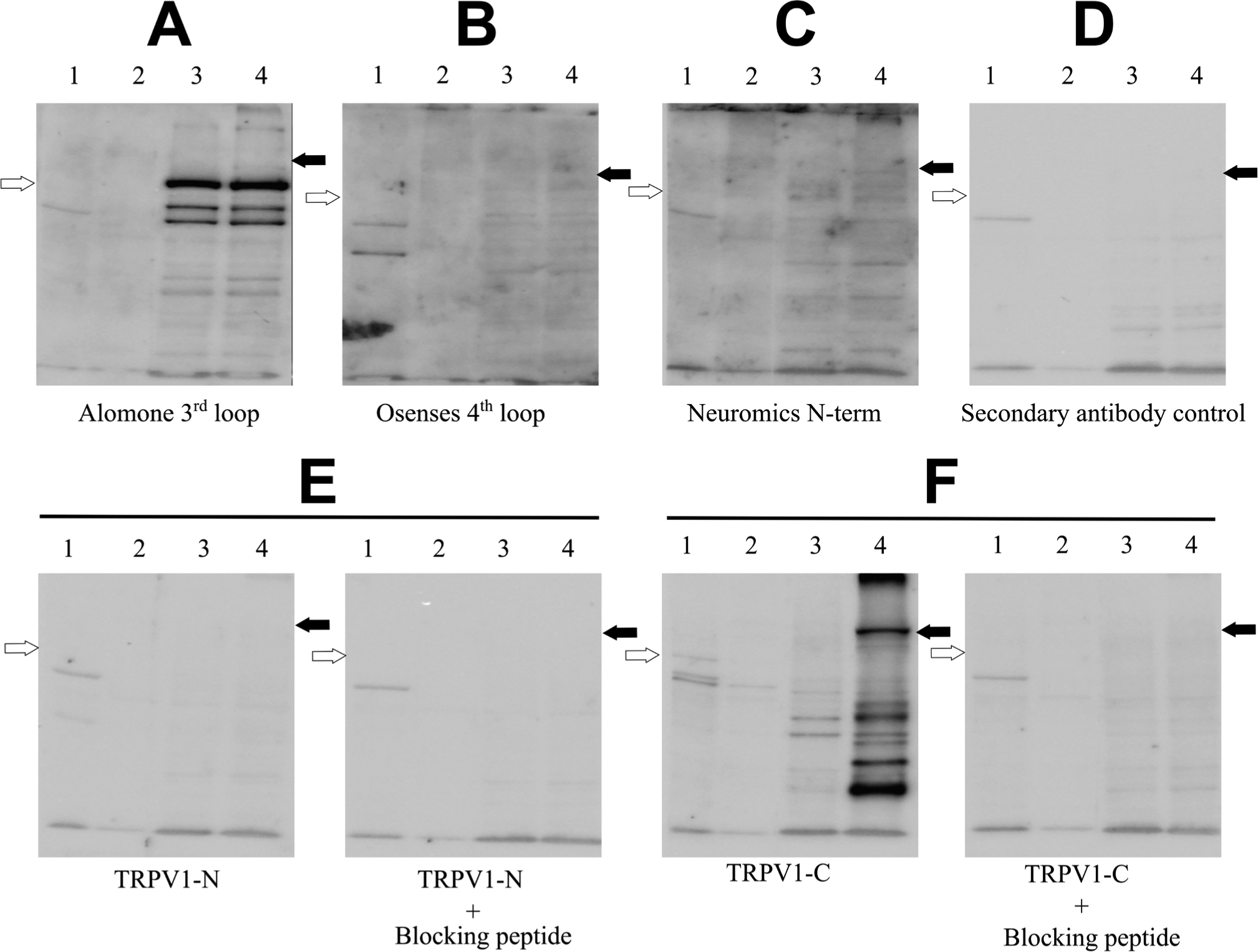
Specificity of TRPV1 antibodies in western blotting. Dorsal root ganglia (1) and the carotid artery (2) were harvested from the rat. HEK293 (3) and HEK293 cells transfected with GFP-TRPV1 (4) were cultured in cell culture dishes. Protein lysates from each sample were prepared in SDS sample buffer, separated on 10% polyacrylamide gels (30 µg protein/well) and transferred onto nitrocellulose membranes for TRPV1 antibody staining. Dilutions were as per the manufacturer’s recommendation or 1:50 (anti-TRPV1-N antibody). (A) Alomone antibody; (B) Oseneses 4th loop, and (C) Neuromics N-terminal were assessed and compared with (D) secondary antibody alone (no primary antibody). (E, F) Blocking peptides, synthesized according to the sequence of the immunogenic TRPV1 fragment, were preincubated with the primary antibody to ascertain specificity of the staining. Membranes were incubated simultaneously with the secondary antibody (goat anti-rabbit–POD) and ECL was used for the detection. All of the membranes were developed simultaneously in a Chemibis 3.2 imager with an exposure time of 6 min. Empty arrows point to the expected position of endogenous TRPV1 (lanes 1, 2 and 3), and filled arrows to the position of GFP-TRPV1 (lane 4 only).
Characterization of TRPV1-positive Structures in the Dorsal Root Ganglia
The anti-TRPV1-N and anti-TRPV1-C antibodies were used to investigate TRPV1 expression in the rat. The cross-sectional area of TRPV1-positive neurons was measured in the dorsal root ganglia (anti-TRPV1-N, Fig. 6A and anti-TRPV1-C, Fig. 6B). Both antibodies stained the small-diameter neurons (Fig. 6E and 6F, respectively, cross-sectional diameter was 200-600 µm2), which was in accordance with the size of the sensory neurons. Surprisingly, anti-TRPV1-N antibody also gave a TRPV1-like immunoreactivity in vascular beds within (Fig. 6A) and around (Fig. 3A) the dorsal root ganglia. We next tested the colocalization of TRPV1 with smooth muscle actin. The anti-TRPV1-N antibody stained some but not all of the smooth muscle cells (Fig. 6B), with some vessels in the section also positive for TRPV1. Anti-TRPV1-C antibody did not stain smooth muscle cells (Fig. 6D). The functional expression of TRPV1 in sensory neurons is well established, but its expression in the vasculature is a relatively novel concept. Thus, we next sought to investigate this vascular expression of TRPV1 using a combination of immunohistochemistry and functional measurements.
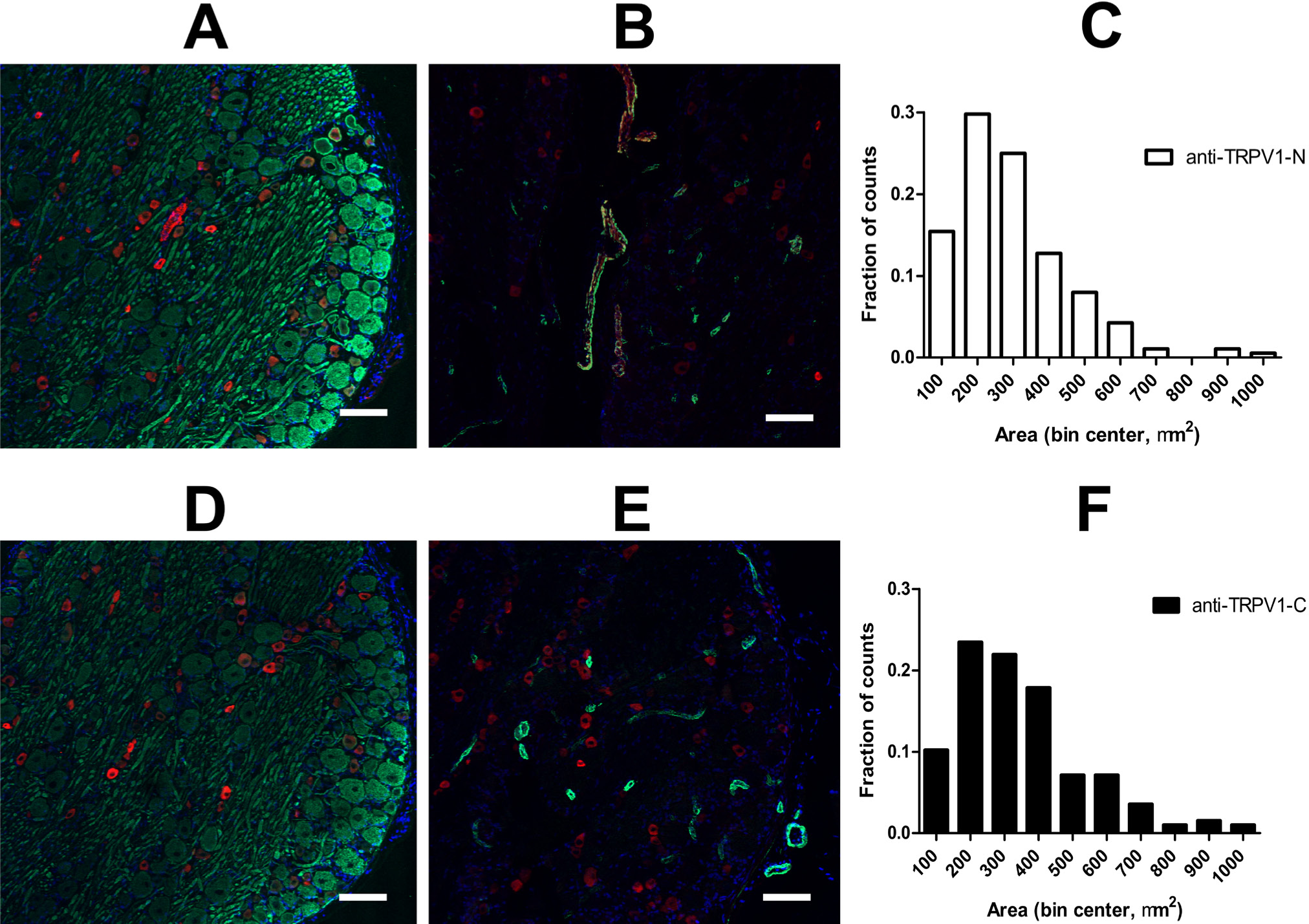
TRPV1 expression in sensory neurons. The selectivity of the anti-TRPV1-N (A and B) and anti-TRPV1-C (C and D) antibodies were tested in the dorsal root ganglia of the rat. Cryostat sections (10 µm) were stained using TRPV1 antibodies (red), a neurofilament-specific antibody (green; A and C) or an antibody against smooth muscle actin (green; B and D). Nuclei were stained with DAPI (blue). Cross-sectional area of the stained cells was calculated and plotted on histograms (anti-TRPV1-N, panel E, anti-TRPV1-C, panel F). Evaluations were made with ImageJ software.
Characterization of Functional TRPV1 Expression in Different Vascular Tissues of the Rat
Vascular smooth muscle cells of blood vessels within the gracilis muscle of the rat were positively stained with an anti-TRPV1-N antibody (Fig. 7B), whereas anti-TRPV1-C antibody did not produce a specific staining pattern (Fig. 7D). Neither antibody stained the neurites in this tissue type (Fig. 7A and 7C, respectively). TRPV1-positive (anti-TRPV1-N antibody) arteries were isolated and the effect of the TRPV1 agonist, capsaicin, was tested. Capsaicin evoked a robust constriction in these arterioles, which was comparable to that evoked by norepinephrine (Fig. 7E).
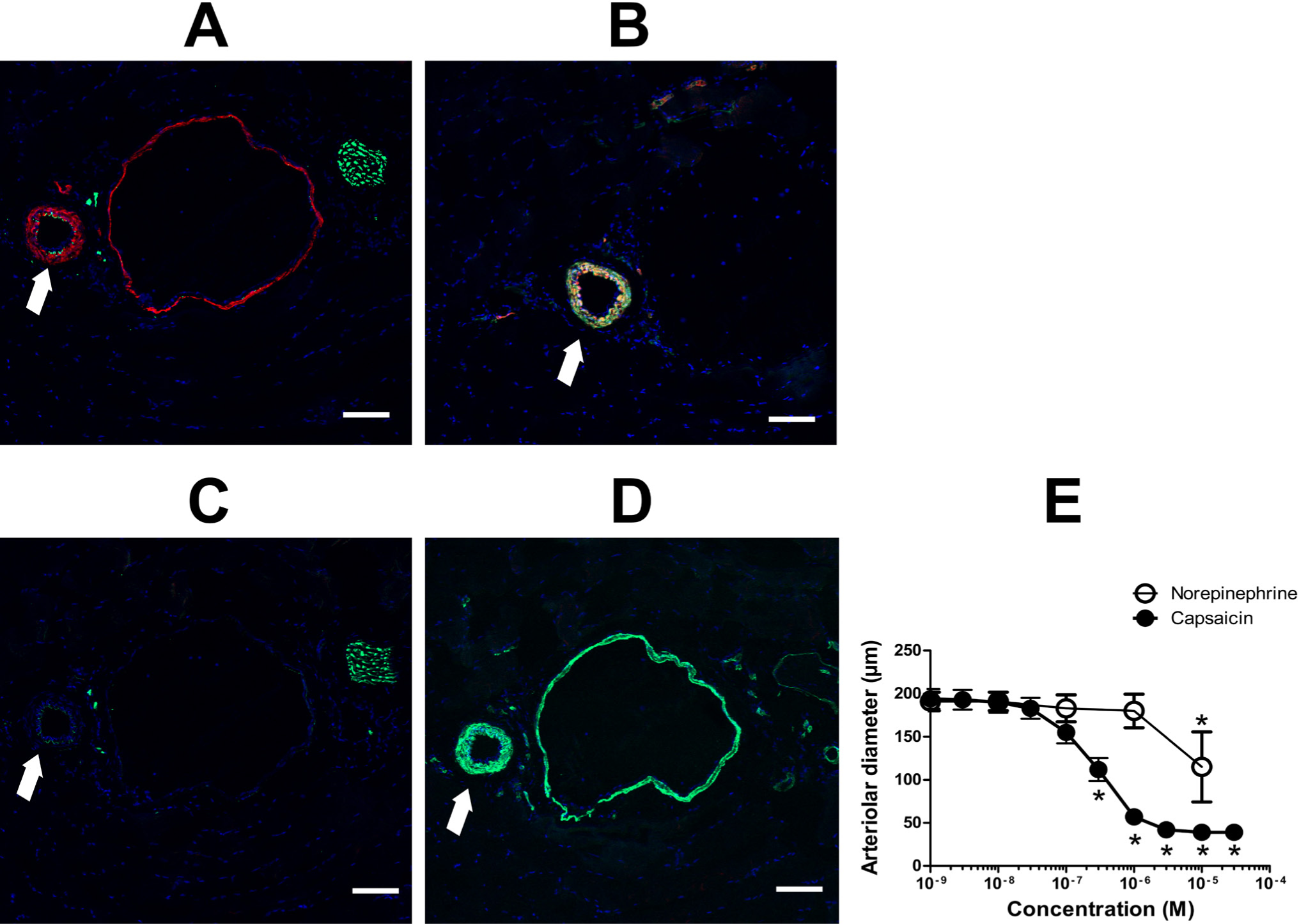
Functional expression of TRPV1 in skeletal muscle blood vessels. Cryostat sections were prepared from the gracilis muscle of the rat (10 µm) and were stained using anti-TRPV1-N (A and B) or anti-TRPV1-C (C and D) antibodies (red). Sections were co-stained with antibodies against neurofilament (green; A and C) or smooth muscle actin (green; B and D). The same arteries (arrows) were isolated and mounted on an isobaric (cannulated) setup. (E) Concentration-response to capsaicin (a TRPV1-specific agonist) and to norepinephrine. Data are the mean ± SEM of five independent experiments. Asterisks indicate significant differences as compared with the initial (before treatment) values.
These conflicting staining patterns of the vascular tissue by the two TRPV1 antibodies were further investigated using blocking peptides. Smooth muscle staining with anti-TRPV1-N antibody (Fig. 8A) was blocked by the immunogenic TRPV1 fragment (Fig. 8B), confirming the specificity of the TRPV1 staining. On the other hand, there was no signal above the background in the case of the anti-TRPV1-C antibody (Fig. 8C and 8D).
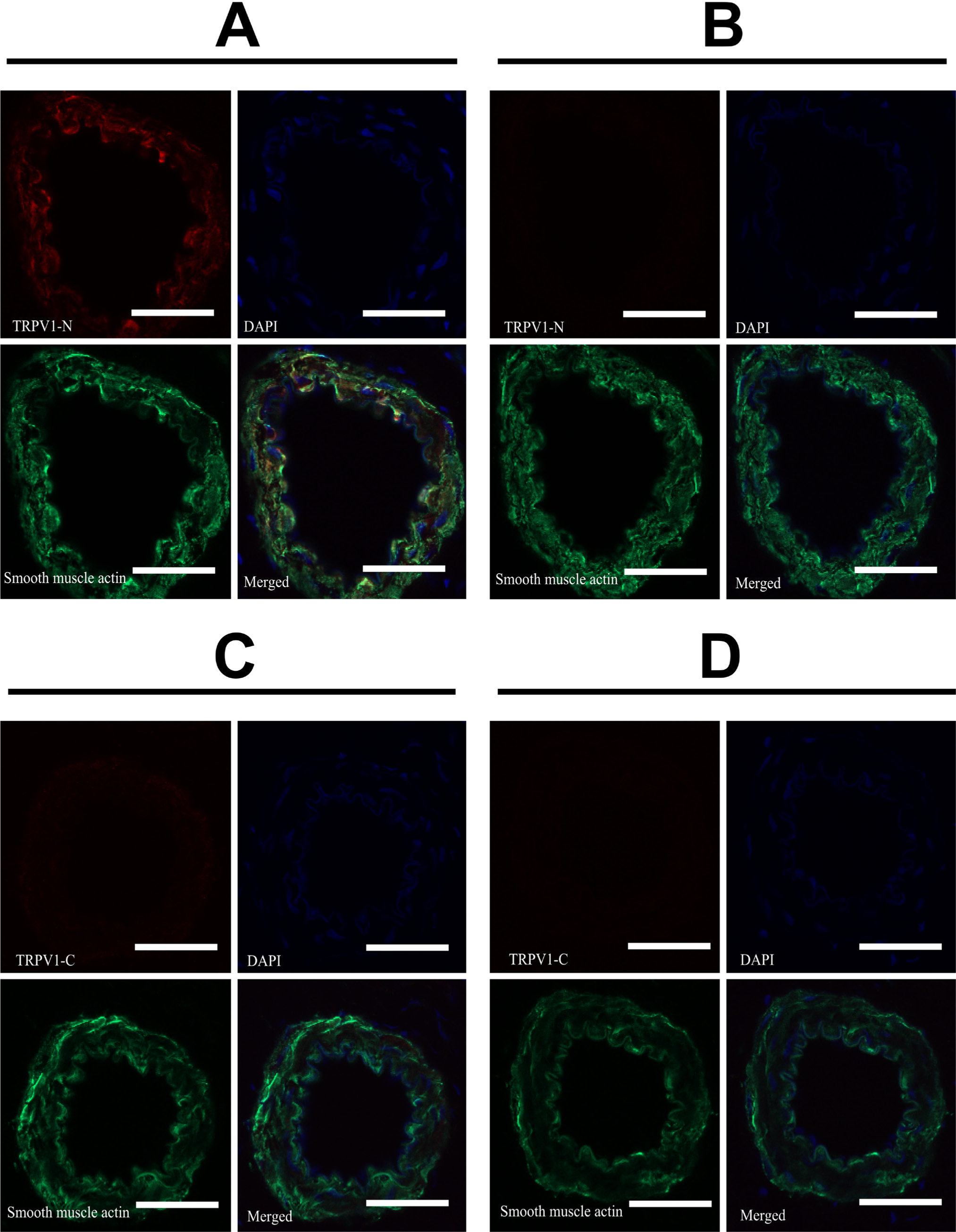
TRPV1 expression in smooth muscle cells. Smooth muscle expression of TRPV1 was investigated in detail. Gracilis muscle tissue sections of the rat were probed with anti-TRPV1-N (A and B; red) or anti-TRPV1-C (C and D; red) as well as anti-neurofilament (green) and DAPI (blue). Merged images are shown. (B, D) TRPV1-specific antibodies were pre-absorbed with blocking peptides to show specificity of staining (compare A with B; C with D). Bars represent 100 µm.
An inhomogeneous staining pattern was found in the mesenteric tissue with the anti-TRPV1-N antibody (Fig. 9A and 9B), while the anti-TRPV1-C antibody (Fig. 9C and 9D) again failed to show specific staining. Some of the blood vessels were positive for TRPV1, while others were not within the same tissue section (Fig. 9A and 9B). Capsaicin had no functional effect, although norepinephrine evoked substantial vasoconstriction (Fig. 9E).
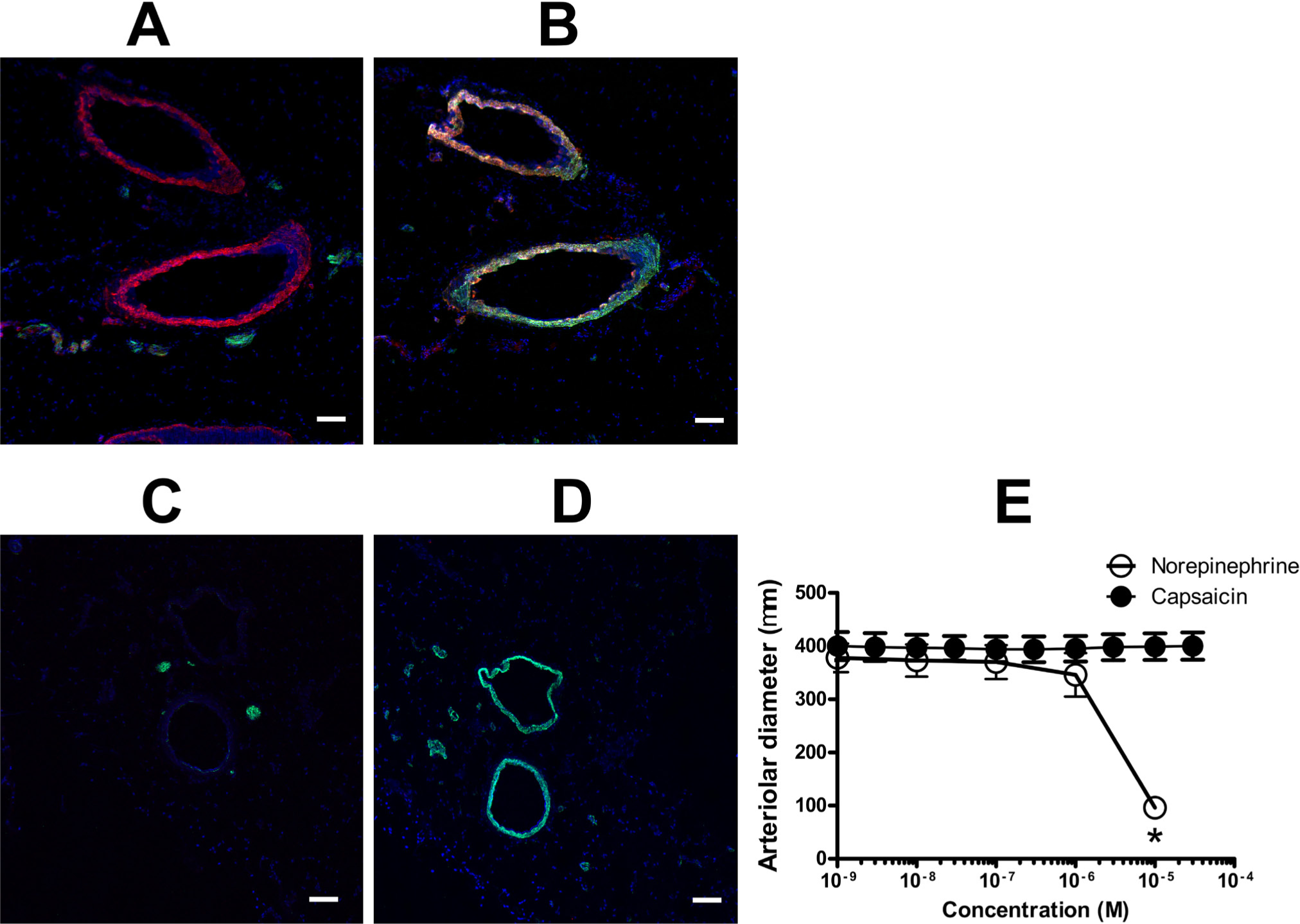
Expression of TRPV1 in mesenteric blood vessels. Cryostat sections were prepared from the mesenteric tissue (near the small intestine; 10 µm) and TRPV1 expression (red) was detected using anti-TRPV1-N (A and B) or anti-TRPV1-C (C and D) antibodies. Sections were co-stained with antibodies against neurofilament (green; A and C) or smooth muscle actin (green; B and D). (E) Isolated, third-order arteries from the same tissue were mounted on an isobaric vascular setup and the responses to capsaicin and norepinephrine were recorded. Data are the mean ± SEM of five independent experiments. Asterisk indicates a significant difference as compared with the initial (before treatment) value. Bars represent 100 µm.
The anti-TRPV1-N antibody gave a strong positive staining for sections of the femoral artery (Fig. 10B), whereas the anti-TRPV1-C antibody showed a weak background staining in skeletal muscle cells (Fig. 10D). Capsaicin had no effect in the functional measurements on these (isolated) arteries, compared with the constrictions evoked by norepinephrine (Fig. 10E). None of the peripheral neurites were stained by these antibodies (Fig. 10A and 10C).
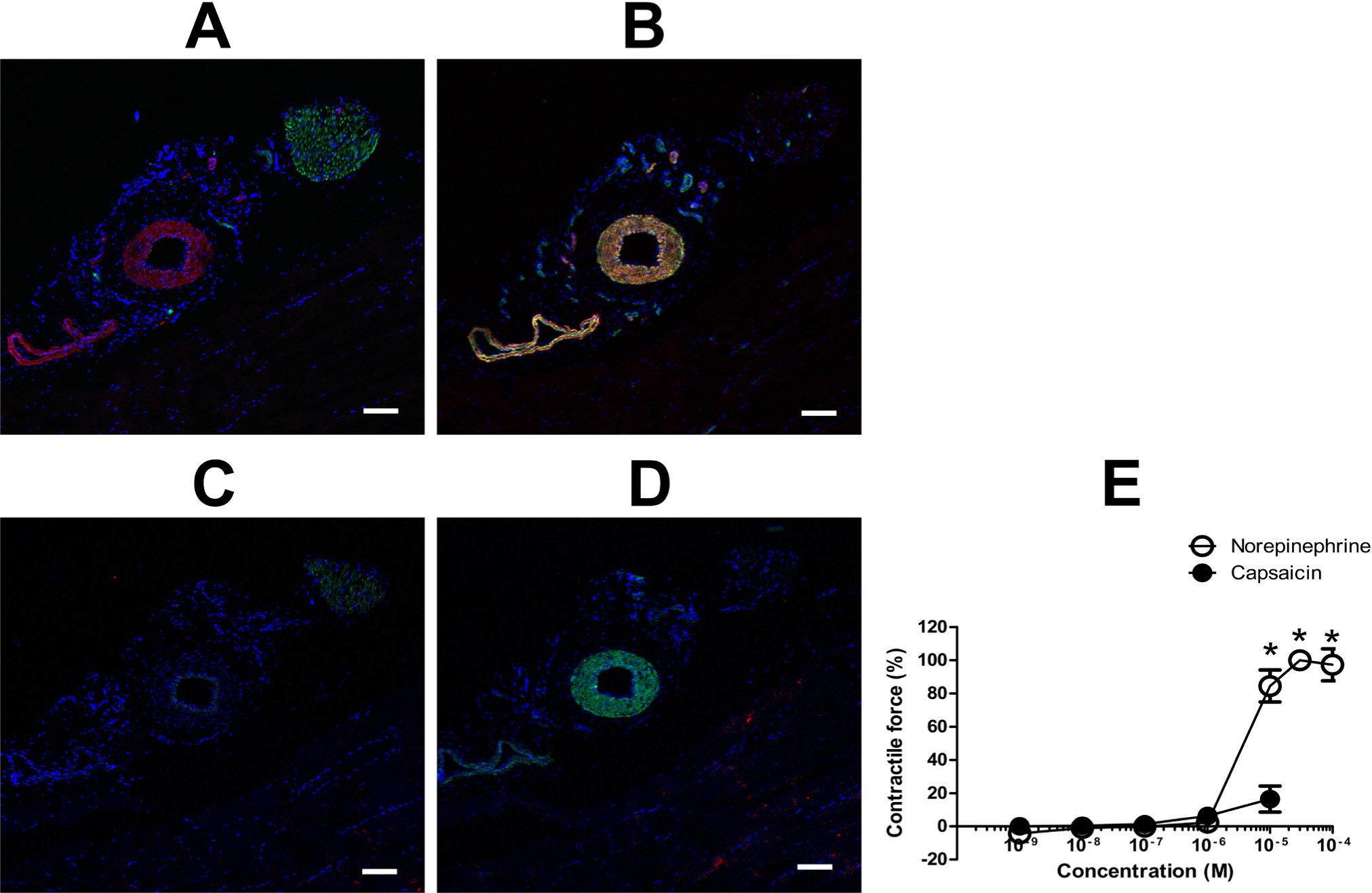
Expression of TRPV1 in the femoral artery. Femoral artery tissue sections were probed with anti-TRPV1-N (red; A and B) or anti-TRPV1-C (red; C and D), and anti-neurofilament (green; A and C) or anti-smooth muscle actin (green; B and D), and counterstained with DAPI (blue). (E) The same arteries were mounted on an isometric contractile force measurement system and responses to capsaicin (TRPV1-specific agonist) and norepinephrine were measured. Data are the mean ± SEM of four independent experiments. Asterisks indicate significant differences as compared with the initial (before treatment) constrictions. Bars represent 100 µm.
We next examined TRPV1 staining of the aorta. The aorta was positively stained for TRPV1 using the anti-TRPV1-N antibody (Fig. 11B), but not with the anti-TRPV1-C antibody (Fig. 11D). Capsaicin had no effect on the isolated rings, whereas norepinephrine evoked substantial constrictions (Fig. 11E). There was no neuronal staining in these tissue sections (Fig. 11A and 11C).
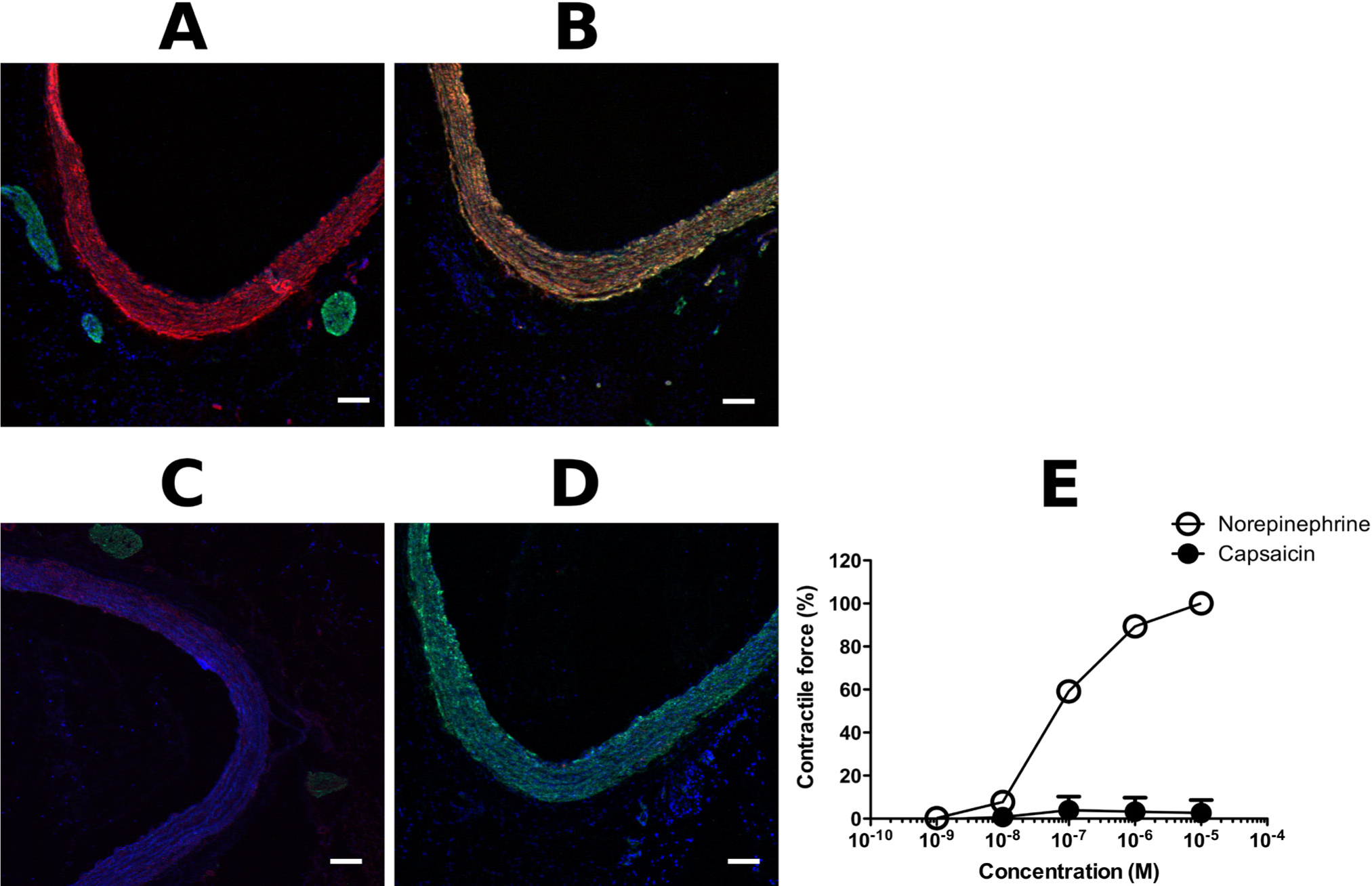
Expression of TRPV1 in the aorta. Rat aorta tissue sections were probed with anti-TRPV1-N (red; A and B) or anti-TRPV1-C (red; C and D), and anti-neurofilament (green; A and C) or anti-smooth muscle actin (green, B and D), and counterstained with DAPI (blue). (E) Contractions to capsaicin and norepinephrine were tested in an isometric contractile force measurement system. Data are the mean ± SEM of six independent experiments. Asterisks indicate significant differences as compared with the initial (before treatment) contractile forces. Bars represent 100 µm.
We also tested TRPV1 staining of the carotid artery. Again, the anti-TRPV1-N antibody stained the smooth muscle layer of the tissue (Fig. 12B), whereas the anti-TRPV1-C antibody demonstrated no specific staining (Fig. 12D). Nonetheless, capsaicin evoked a partial constriction in carotid arteries, which was about 20% of the maximal constriction achieved by the thromboxane A2 agonist U-46619 (Fig. 12E). Besides vessel staining, no neuron-specific staining was found in these sections (Fig. 12A and 12C).
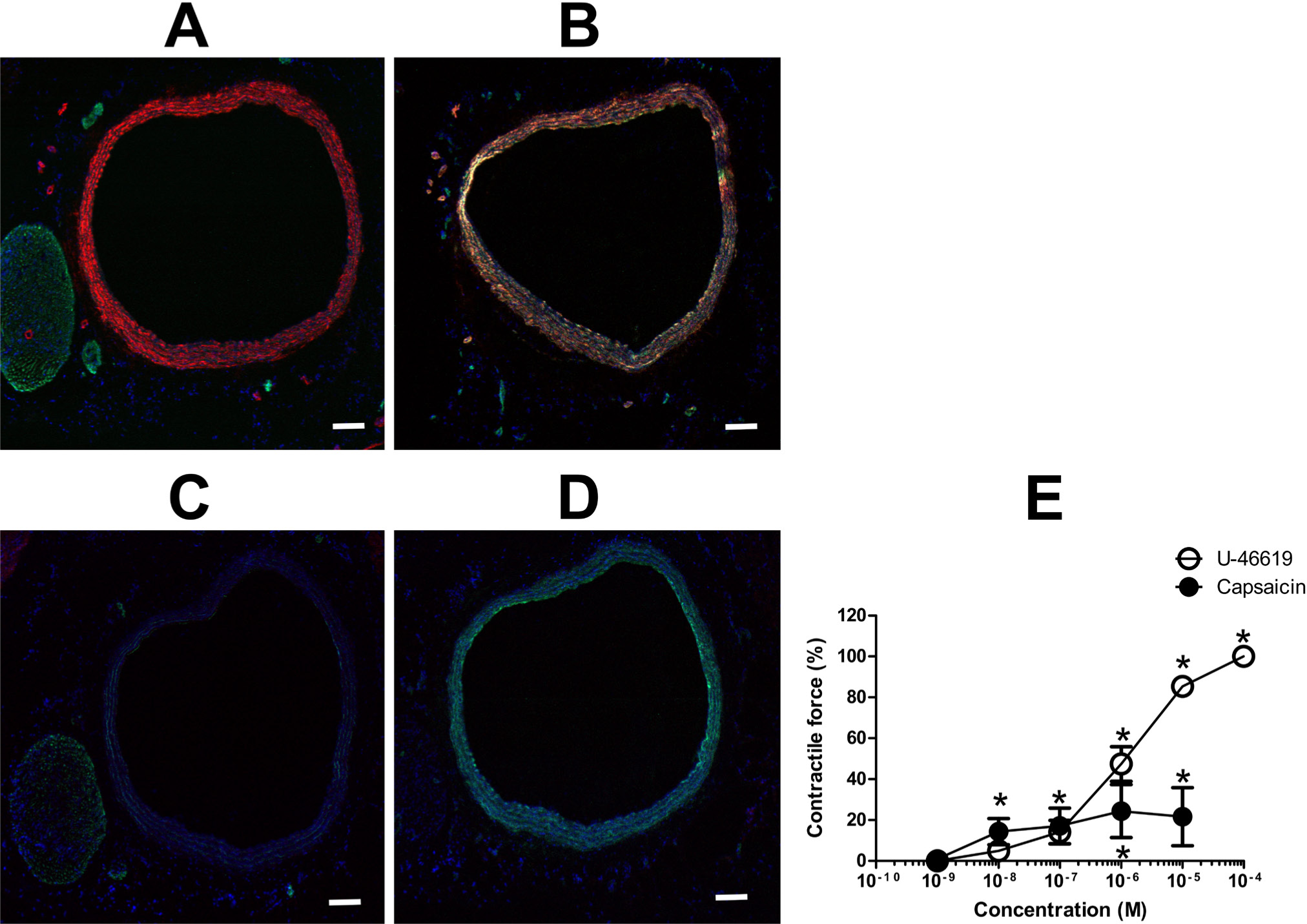
Expression of TRPV1 in the carotid artery. Cryostat sections were prepared from the carotid artery of the rat (10 μm) and were stained using anti-TRPV1-N (A and B) or anti-TRPV1-C (C and D) antibodies (red). Sections were also co-stained with antibodies against neurofilament (green; A and C) or smooth muscle actin (green; B and D). The same arteries were isolated and mounted on an isometric (ring) setup. (E) Concentration-responses to capsaicin and to U-46619 (a thromboxane A2 receptor agonist). Data are the mean ± SEM of eight independent experiments. Asterisks indicate significant differences as compared with the initial (before treatment) contractile forces. Bars represent 100 μm.
Finally, a tissue important in the thermoregulation was tested. Staining of the ears of the rat revealed that both the anti-TRPV1-N and anti-TRPV1-C antibodies can stain peripheral axons (Fig. 13A and 13C, respectively). The anti-TRPV1-C antibody did not stain specifically other structures (Fig. 13D). In contrast, the anti-TRPV1-N antibody again stained some of the blood vessels (Fig. 13B). It is important to note that a very limited number of blood vessels were stained, and they appeared to have a larger diameter (Fig. 13B).
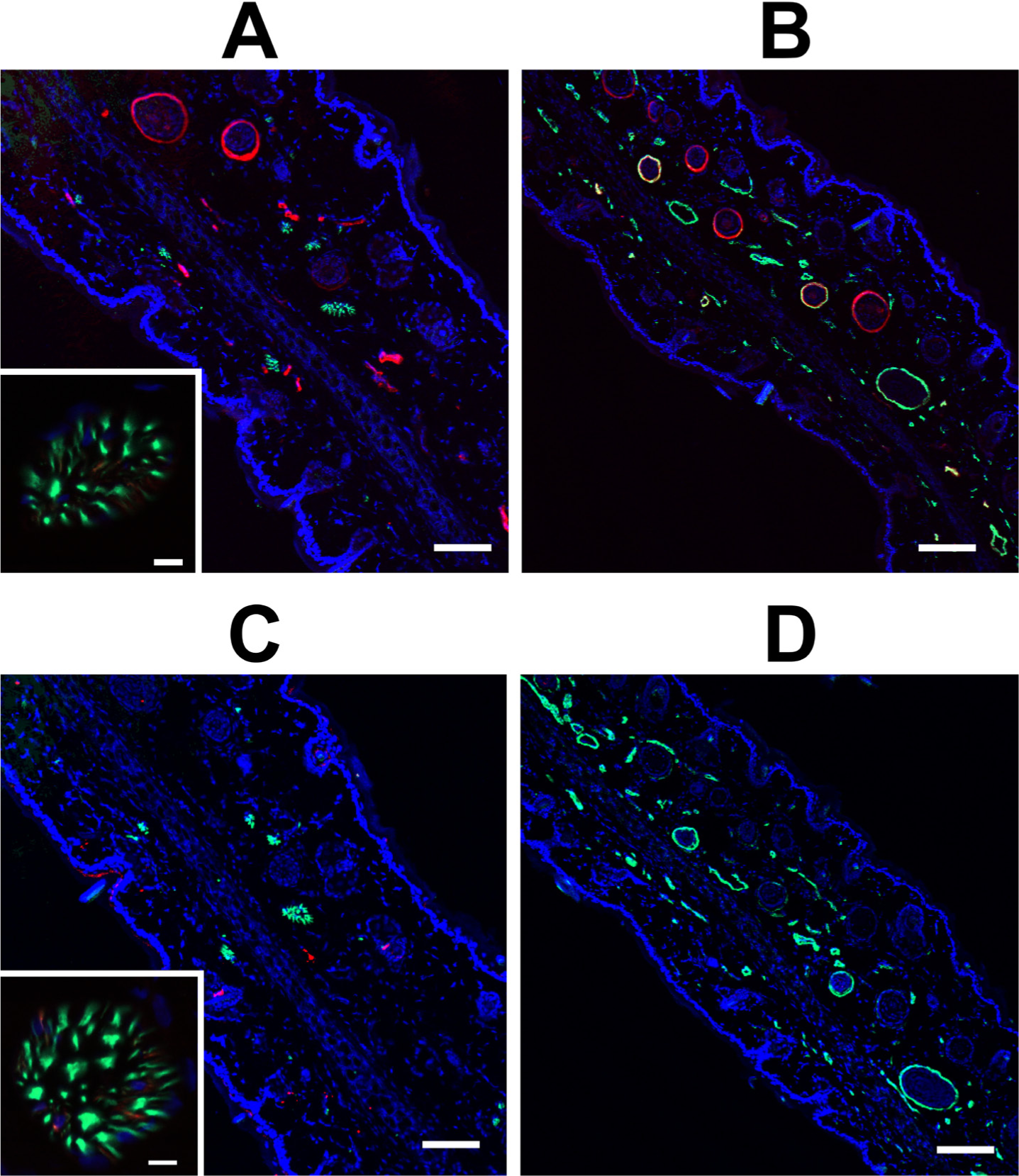
Expression of TRPV1 in the skin. Immunohistochemistry was performed in the skin from the hindpaw of the rat. Sections were stained with anti-TRPV1-N (red, A and B) or anti-TRPV1-C (red, C and D), and anti-neurofilament (green, A and C) or anti-smooth muscle actin (green, B and D), and counterstained with DAPI (blue). Colocalization of TRPV1-positive neurites with anti-neurofilament is shown in the insets at a higher magnification. Bars, 100 µm; inset bars,10 µm.
Discussion
There are numerous reports about TRPV1 expression in non-neuronal tissues. Here we made an effort to characterize these TRPV1 populations in the peripheral tissues of the rat. The presence of TRPV1 mRNA was established first. Our qPCR analysis (Fig. 1B) suggested that TRPV1 is indeed expressed in vascular preparations, although the expression level was about two orders of magnitude lower than that in the dorsal root ganglia. After establishing the presence of TRPV1 in vascular tissues, an effort was made to identify antibodies suitable for detecting TRPV1 expression. Six commercially available antibodies were tested. We found that only two of the six tested antibodies were selective for TRPV1 in dorsal root ganglia sections. These antibodies (referred as anti-TRPV1-N and anti-TRPV1-C) were then characterized in detail. We showed that one antibody (anti-TRPV1-C) was able to detect endogenous (dorsal root ganglia) and exogenous (transfected HEK293 cells) TRPV1 in western blotting. Moreover, TRPV1 staining of sensory neurons (anti-TRPV1-N and anti-TRPV1-C antibodies) and vascular smooth muscle cells (anti-TRPV1-N antibody) was blocked by the antigenic peptides, suggesting that both of these antibodies are specific for TRPV1.
We next concentrated our efforts on examining TRPV1 expression in blood vessels, with staining performed in parallel with functional measurements on the same arteries where possible. We first tested arteries present within the gracilis muscle tissue, because we have previously provided data to suggest functional TRPV1 expression in skeletal muscle arteries (Czikora et al. 2012; Kark et al. 2008; Lizanecz et al. 2006). Indeed, using the anti-TRPV1-N antibody, TRPV1 was found to be abundantly expressed in all blood vessels within the gracilis muscle. Interestingly, the anti-TRPV1-C antibody staining was not positive in this tissue, suggesting that the anti-TRPV1-C antibody does not recognize vascular smooth muscle-located TRPV1; however, the antibody can detect TRPV1 in sensory neurons in western blotting and immunohistochemistry. This discrepancy in staining may lead one to argue that the vascular smooth muscle staining observed with the anti-TRPV1-N antibody is artifactual; however, there are many reasons why this is unlikely: (1) Vascular TRPV1 staining was blocked by the TRPV1-specific antigenic peptide (Fig. 8); (2) Vascular TRPV1 expression is in accordance with the constrictive effect of the TRPV1 agonist capsaicin. (Capsaicin-mediated vasoconstriction is absent in TRPV1-/- mice (Czikora et al. 2012), which strongly suggests that a capsaicin response is specific for TRPV1); (3) TRPV1 mRNA is present in the isolated arteriolar preparations (Fig. 1); and (4) Earlier reports by an independent group also showed functional arteriolar TRPV1 expression (Cavanaugh et al. 2011).
Assuming this staining to be specific, the goal of the present work was to study TRPV1 expression and function in isolated arteries from a set of rat tissue samples, using the anti-TRPV1-C antibody as a TRPV1 expression marker in vascular tissue. There were several important observations. First, it appears that the TRPV1 is not uniformly expressed in the vascular tissue, with TRPV1 only expressed in a subset of blood vessels in some tissues (in particular, mesenteric arteries and skin). The observed differences in TRPV1 staining within the same tissue sections suggest a complex regulation of TRPV1 expression at the level of the individual vessels. Another surprising observation was the wide range of functional responses of the TRPV1-positive (anti-TRPV1-N antibody) arteries. Whereas arteries from the gracilis muscle responded to capsaicin with a robust constriction—which was comparable to that of those evoked by norepinephrine (representing the maximal physiological vasoconstriction in this particular case)—other arteries (e.g., the carotid artery) had a limited functional TRPV1 response, even if an apparently high level of TRPV1 expression was found. Moreover, some of the arteries expressing TRPV1 did not contract upon capsaicin supplementation (femoral and mesenteric arteries and aorta).
These apparent contrasts between TRPV1 function and expression may be explained by many factors. It is possible that TRPV1 expression is regulated at the cellular level. One of the observations supporting this hypothesis is that only a portion of isolated coronary smooth muscle cells responded to capsaicin by increasing intracellular Ca2+ concentrations (Czikora et al. 2012). Another possibility is that TRPV1 expression is regulated at the level of the individual blood vessel (Cavanaugh et al. 2011). This is supported by the staining patterns in this study that showed uniform staining in the smooth muscle layer in a given blood vessel; although, some of the blood vessels were stained whereas others were not, even within the same tissue slice. It needs to be mentioned that this vessel-specific staining pattern has also been reported earlier with genetically engineered mice, where TRPV1 was genetically labeled with a fluorescent protein; however, the same group was not able to stain unmodified TRPV1 in the same location (Cavanaugh et al. 2011). Finally, it is also possible that the activity of vascular TRPV1 is controlled by posttranslational modifications. In this case, the physiological activity of vascular TRPV1 may be regulated by sensitization-desensitization of the receptor. Indeed, previous work demonstrates that vascular TRPV1 can be desensitized via a process involved the activation of the Ca2+-dependent phosphatase, calcineurin (Lizanecz et al. 2006). An alternative explanation for the differences in the arteriolar response to capsaicin (in the case of the arteries of the gracilis muscle, as well as the femoral and carotid arteries and aorta) may be the existence of different TRPV1 isoforms. All of the preparations showed strong staining with the anti-TRPV1-N antibody. Nonetheless, at least three different TRPV1 splice variants have been suggested, which have a low sensitivity for agonists (such as the capsaicin used here) (Eilers et al. 2007; Tian et al. 2006; Vos et al. 2006; Wang et al. 2004). Moreover, these splice variants may associate with the dominant (functional) form of the TRPV1, and inhibit its activity (without affecting its apparent expression when tested by immunohistochemistry). Here, we performed a morphological study, in which we found striking differences in the TRPV1 staining pattern within arteries in close proximity (on the same tissue section) and showed that large arteries with apparently similar TRPV1 expression have strikingly different capsaicin sensitivity. Further studies are required to identify the reasons for these differences.
Regardless of the complicated regulation, the main question for this study was the functional role of TRPV1 in vascular biology. TRPV1-mediated vasoconstriction was reported three decades ago. Donnerer and Lembeck (1982) described that capsaicin evokes a three-phase response in the rat, with the second phase described as a vasoconstrictive response that was independent of sensory innervation (Donnerer and Lembeck 1982). Later, Duckles also described the direct effect of capsaicin on vascular smooth muscle cells (Duckles 1986). The presence of TRPV1 in vascular tissue was further confirmed after the identification of its gene. TRPV1 staining in the smooth muscle layer of epineurial arteries, together with a contractile response to capsaicin and resiniferatoxin, was also noted previously (Davidson et al. 2006), and recently, functional TRPV1 expression in the smooth muscle cells of intrapulmonary arteries was related to pulmonary hypertension (Martin et al. 2012). In addition to the confirming these earlier results, our data show that arteriolar TRPV1 expression is tightly regulated (arteries within a couple of micrometers show high or undetectable level of TRPV1 expression) and that TRPV1 responses are suppressed in some cases, in spite of uniformly high TRPV1 expression.
Our data provide evidence that TRPV1 represents another significant vascular TRP channel, and suggest that TRPV1 is in its sensitized state in some skeletal muscle arteries (such as those isolated from the gracilis muscle) and in the coronary arteries (Czikora et al. 2012), but it is inhibited in other blood vessels. It is also known that TRPV1 stimulation has divergent effects in vivo: it may evoke neurogenic vasodilation (e.g., in the skin) or vascular constrictions (e.g., in the skeletal muscle) (Kark et al. 2008). These divergent effects suggest that the systemic regulation of TRPV1 may be an effective tool to regulate blood distribution between tissues. Moreover, these divergent effects are in accordance with the immunohistochemical data presented here. It is expected that arteries with sensory neuronal innervation, but without vascular TRPV1 expression (such as blood vessels in the skin; Fig. 13), respond to TRPV1 stimulation by dilation. In contrast, arteries with high smooth muscle TRPV1 expression and without apparent sensory neuronal innervation (such as the gracilis artery; Fig. 8) are expected to respond to the same TRPV1 activation by constriction. This work adds an important addition to this hypothesis in that TRPV1 expression in the arteries appears to not be necessarily functionally active. Indeed, we show that although TRPV1 is expressed in the large arteries, the activity of this receptor appears to be suppressed.
Taken together, this study made an effort to investigate the functional expression of TRPV1 in the rat. We found that TRPV1 expression in the vascular smooth muscle is regulated at the level of the individual blood vessels: some blood vessels showed intense TRPV1 immunostaining, whereas nearby vessels were negative. Moreover, the activity of the smooth muscle-expressed TRPV1 appeared to be suppressed in some cases. Nonetheless, TRPV1 was found to be widely expressed in the vasculature rat, suggesting a physiological role for these cation channels in vascular biology.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The work is supported by the TAMOP-4.2.2/B-10/1-2010-0024 and TÁMOP 4.2.2.A-11/1/KONV-2012-0045 (to IÉ, ZP and AT), TÁMOP-4.2.2.A-11/1/KONV-2012-0025 (to PB) projects. These projects are implemented through the New Hungary Development Plan, co-financed by the European Social Fund. In addition, the study was supported by the Hungarian Academy of Sciences OTKA (K84300 to AT and PD83473 to PB) and Bolyai János Research Fellowship (to AT and PB), by the National Office for Research and Technology (Baross Gábor ÉletMent (to AT) and Seahorse (to PB) grants) and by the University of Debrecen (Mec-8/2011 to PB).
