Abstract
We analyze the effect of chronic undernourishment on extensor digitorum longus (EDL) muscle maturation in the rat. Cytochrome c oxidase (COX) and alkaline ATPase histoenzymatic techniques were used to determine the relative proportion of different fiber types (oxidative/glycolytic and type I, IIa/IId, or IIb, respectively) and their cross-sectional area in control and undernourished EDL muscles at several postnatal (PN) ages. From PN days 15 to 45, undernourished EDL muscles showed predominance of oxidative and type IIa/IId fibers, but from PN days 60 to 90, there were a larger proportion of oxidative fibers and an equal proportion of type IIa/IId and IIb fibers. Meanwhile, in adult stages (from PN days 130–365), the relative proportion of fiber types in control and undernourished EDL muscles showed no significant differences. In addition, from PN days 15 to 90, there was a significant reduction in the cross-sectional area of all fibers (slow: 13–53%; intermediate: 24–74%; fast: 9–80%) but no differences from PN days 130 to 365. It is suggested that chronic undernourishment affects the maturation of fast-type muscle fibers only at juvenile stages (from PN days 15–45) and the probable occurrence of adaptive mechanisms in muscle fibers, allowing adult rats to counterbalance the alterations provoked by chronic food deprivation.
Keywords
It is generally accepted that muscles are composed of four different fiber types: slow, non-fatigable oxidative fibers (type I); two types of fast, non-fatigable oxidative- glycolytic fibers (type IIa and IId or intermediate); and fast, fatigable glycolytic fibers (type IIb; Armstrong and Phelps 1984). At birth, practically all the fibers in fast muscles show fetal or slow-type characteristics, and subsequently, most of them acquire metabolic and physiological characteristics corresponding to intermediate and/or fast-type fibers (Close 1964). The transformation from one type of muscle fiber to another during postnatal development (e.g., from slow to intermediate or from intermediate to fast fibers) implies the participation of a large variety of functional, molecular, and structural processes that may occur at a continuous, progressive, and dynamic manner during the postnatal development of the rat (Lehnert et al. 2007). In contrast, it has been assumed that an imposed dietary restriction may disturb the expression of intrinsic mechanisms that are involved in fiber transformation and energy production of muscles mostly constituted by fast fibers compared with those composed by slow fibers (Brozanski et al. 1991; Fiaccadori et al. 1995; White et al. 2000).
Dietary restrictions during critical stages of development (primarily pregnancy and lactation) generally produce drastic changes in the postnatal growth of skeletal muscles, including decreased muscle weight (Haltia et al. 1978; Goldspink and Ward 1979; Bedi et al. 1982; Lopes et al. 1982) and fiber size (Stickland et al. 1975). Some studies have indicated that prenatal or early postnatal undernutrition induces an increase in the relative proportion of type I fibers but decreases the number of type II fibers, causing a delay in muscle maturation (Brozanski et al. 1991; Prakash et al. 1993). In addition, a reduction in the number of nuclei in type II fibers with no change in the total number of fibers has been found in the muscles of young rats (Bayol et al. 2004). Early postnatal food deprivation induces an increase in the oxidative metabolism of muscle fibers by using more lipids than glucose (Lefaucheur et al. 2003). Meanwhile, the effects of both pre- and postnatal undernutrition (from gestation through weaning and/or juvenile stages of life) on young muscles increase the oxidative capability and the proportion of slow-oxidative fibers at the expense of fast-glycolytic fibers (Haltia et al. 1978; White et al. 2000). However, other studies have shown increased glycolytic activity and glycolytic enzymes accompanied by a decrease in oxidative capacity and oxidative enzymes in the gastrocnemius muscle of adult rats as a result of postnatal undernutrition (Raju 1975).
Because most studies have made use of different food paradigms to induce food deprivation at discrete periods of pre- and/or postnatal stages of life (e.g., during gestation and/or lactation periods) and only a few studies have used a chronic food deprivation paradigm (including pre- and postnatal stages), the information about the sequence of changes that undernutrition exerts on whole-muscle maturation is fragmented and inconclusive. To analyze the effect of chronic food deprivation on muscle maturation during most of the life span of animals, we characterize in this study the sequence of changes provoked by pre- and postnatal food deprivation on the metabolism, type, and cross-sectional area of fibers in rat extensor digitorum longus (EDL) muscle from early postnatal to adult stages of life. In summary, we found that in juvenile stages, there is an increment in the proportion of oxidative and type IIa/IId fibers accompanied by a reduction in the cross-sectional area of all fiber types in the undernourished EDL muscles, whereas no significant differences were found in adult stages as compared with the control group. Some data from this study have been previously reported (Ruiz et al. 2010).
Materials and Methods
Animals
Nineteen adult female Wistar rats (225 ± 25 g body weight) were obtained from CINVESTAV’s breeding house (CINVESTAV, Ciudad de México, DF, Mexico) and divided in two groups, which were subjected to different feeding conditions. The control group (C), consisting of female rats (n=9) and their offspring, had free access to commercial food (Formulab 5008 [Lab Diet, Brentwood, MO]; composition: crude protein not less than 23%, crude fat not less than 6.5%, crude fiber not more than 4%, ash not more than 8%, added minerals not more than 2.5%) throughout the entire experimental period, and the amount of food consumed was measured every day until the day of the experiment (Table 1). The chronically undernourished group (U) consisted of the remaining female rats (n=10), which received 50% of the food intake of control animals determined before from 3 to 4 weeks before mating through pregnancy and lactation (Chow and Lee 1964; Bedi 1994; Segura et al. 2001). The offspring were also undernourished after weaning (postnatal day 21) until their age at the experimental session (Table 1). After delivery, the sex and number of pups were determined, and the size of each litter was adjusted to 9 (usually 5 males and 4 females per litter). The experiments were performed only in male animals at 15, 21, 45, 60, 90, 130, and 365 postnatal days of age (PN; n=5–7 muscles per age and feeding condition). On the day of the experiment, 5 males were randomly selected from all the litters and weighed. All animals had unlimited access to water and were housed under the same environmental conditions of light/dark cycle and temperature (22–24C). Animal care and experiments were performed in accordance with the guidelines of the Mexican Official Norm (NOM-062-ZOO-1999) and the National Institutes of Health Guide for the Care and Use of Laboratory Animals (NIH Publication No. 8023, revised in 1996) and were approved by the Institutional Bioethical Committee for Care and Handling of Laboratory Animals (UPEAL-Protocol 0013-02; CINVESTAV). The animals were euthanized by an overdose of anesthetic (urethane).
Food Consumption per Rat of the Control (C) and Chronically Undernourished (U) Groups
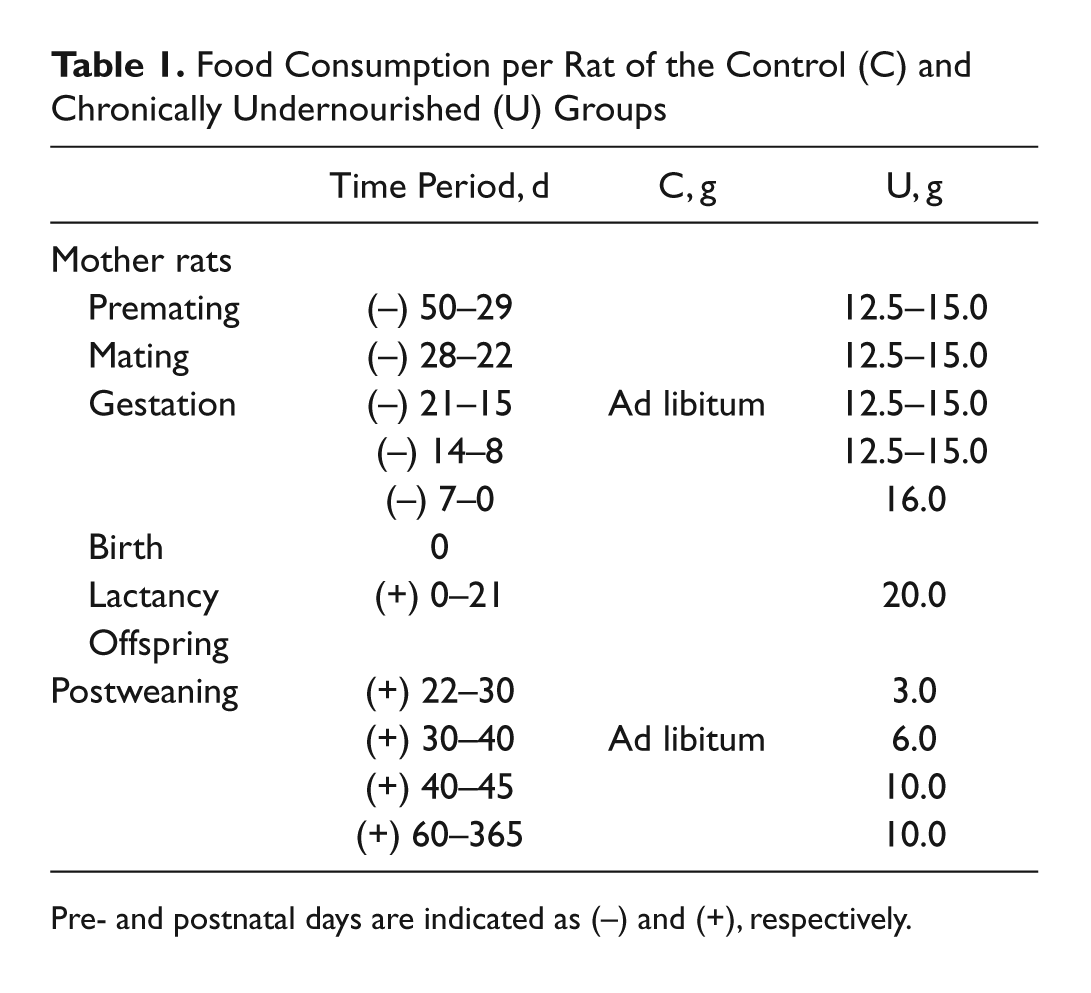
Pre- and postnatal days are indicated as (−) and (+), respectively.
Histoenzymatic Analysis
The EDL muscles from the left or right hind limb were quickly removed, weighed, and immersed in 2-methyl butane that was cooled to near freezing with liquid nitrogen. Subsequently, the middle section of the muscle was mounted on specimen holders in a cryoprotectant solution (Tissue-Tek® OCTMT; Finetek, Torrance, CA). Serial transverse sections of 8 to 10 µm thickness were cut on a cryostat microtome at −20C (CM-1100; Leica Microsystems, Nussloch, Germany). The sections were mounted on glass coverslips for staining. In consecutive sections of the EDL muscle, the oxidative metabolism of the muscle fibers was inferred by staining for cytochrome c oxidase (COX; Seligman et al. 1968; Novikoff and Goldfischer 1969). In brief, the samples were incubated at 37C for 60 min in 1% w/v of 3,3′-diaminobenzidine (Sigma-Aldrich; St. Louis, MO) and cytochrome c from equine heart (Sigma-Aldrich; Shanghai, China) in 0.2 M PBS (pH 7.6). The samples then were washed three times for 5 min in deionized water and dehydrated. Coverslips then were mounted with glycerogel (2% gelatin, 50% glycerol, 0.5% phenol). In the subsequent serial tissue sections, the myofibrillar ATPase activity at pH 9.4 was visualized using an alkaline ATPase (pH 9.4) technique (modified from Guth and Samaha 1970). In brief, the muscle sections were submerged in a preincubation solution of 0.01 M Tris base (Bio-Rad; Hercules, CA) and 0.018 M CaCl2 (pH 9.4) for 20 min. The samples then were washed three times for 5 min with deionized water and subsequently incubated at 37C for 1 hr in 1.5% w/v of adenosine-5′-triphosphate (Sigma-Aldrich; St. Louis, MO) in a preincubation solution at pH 9.4. Later, the samples were incubated in 2% CaCl2 for 5 min, transferred to 2% w/v CoCl2 solution for another 5 min, and transferred to 10% v/v (NH4)2HSO4 for 3 min. Then, the sections were washed, dehydrated, and mounted with glycerogel on coverslips, as described above. Photomicrographs of each muscle section were taken using a digital camera (AxioCam MRc; Zeiss, Jena, Germany) mounted on an inverted microscope (Axio Observer.D1; Zeiss). The fibers in the COX-stained muscle slices were classified according to the following criteria: dark fibers were identified as oxidative, whereas light fibers were considered glycolytic (see Fig. 1). Using the alkaline ATPase technique, light, gray, and dark fibers were identified as slow type I (S), fast type IIb (F), and intermediate type IIa/IId (I), respectively (see Fig. 3; Soukup and Jirmanová 1979; Miyabara et al. 2005). Because fiber types in EDL muscles are not evenly distributed (Pullen 1977; Kernell 1998; Wang and Kernell 2001), the total number of each fiber type was determined in both control and undernourished EDL muscles with the help of a custom program in the MATLAB (MathWorks; Natick, MA) language written in our laboratory. The proportion of each fiber type was expressed as a percentage of the total number of fibers constituting the EDL muscles. Subsequently, the ratio of oxidative (O) to glycolytic (G) fibers (RO/G) and the ratio of intermediate (I) to fast (F) fibers (RI/F) were determined. Subsequently, the transversal area of randomly selected fibers (100 fibers of each type) was measured in sections from five different muscles per age and feeding condition with the Sigma Scan Pro program (version 4.01; Systat Software, Chicago, IL) and expressed in square micrometers. The average growth rate of muscles was determined from the linear regression of the initial ascending phase in the cross-sectional area × postnatal age relationship (between 15 and 45 days of life; see Fig. 5A, C, E) obtained for each fiber type in control and undernourished muscles.
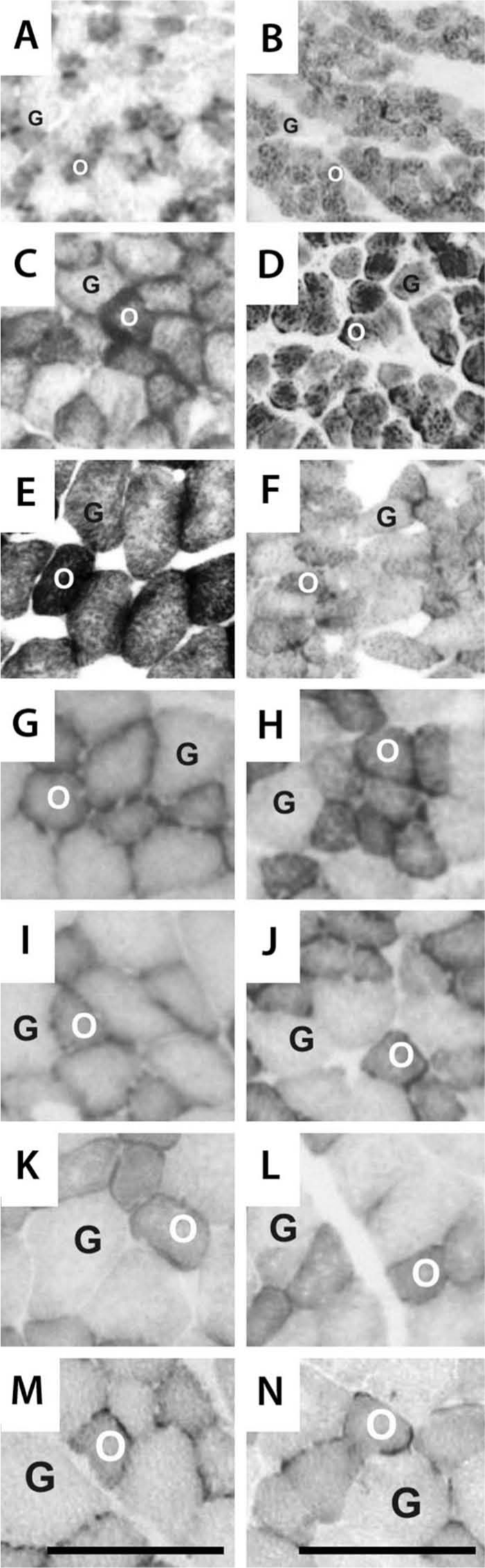
Photomicrographs of extensor digitorum longus muscles stained with the Cox technique at different postnatal ages from control (left column) and undernourished (right column) rats, respectively. (A, B) Postnatal day (PN) 15; (C, D) PN21; (E, F) PN45; (G, H) PN60; (I, J) PN90; (K, L) PN130; (M, N) PN365. Oxidative (O) and glycolytic (G) fibers were identified by their staining intensity as indicated in text. Calibration bars = 100 µm for all panels.
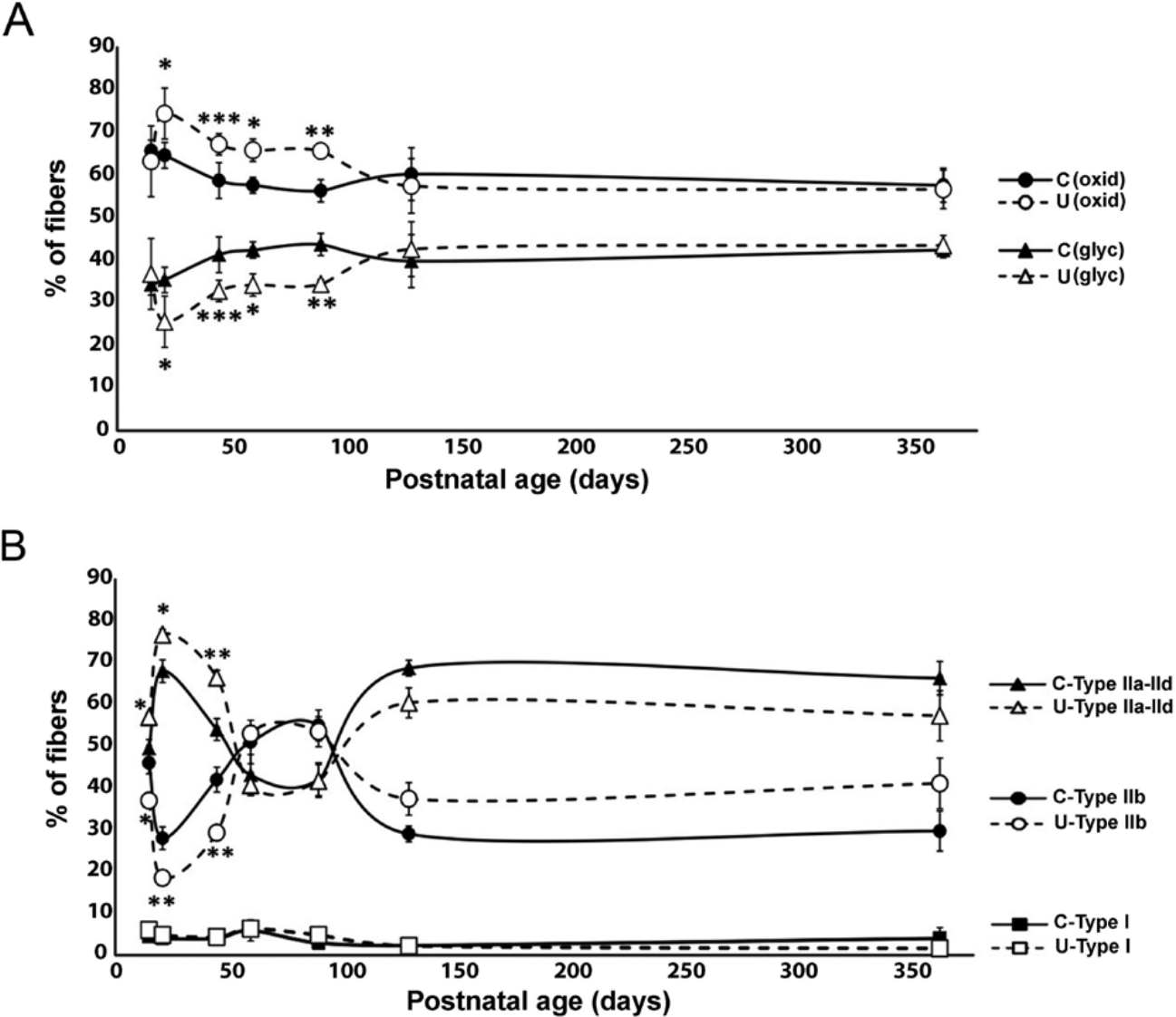
The effects of chronic undernutrition on the proportions of fiber types in the extensor digitorum longus (EDL) muscle during the postnatal development of the rat. (A) Graph illustrating the mean ± SD percentages of fibers showing oxidative and glycolytic activity in the EDL muscles from control (●, ▲, respectively) and undernourished (○, Δ, respectively) rats at different postnatal ages (from postnatal day [PN] 15 to PN365). (B) Same format as above for EDL muscle sections stained with the alkaline ATPase staining technique (pH 9.4), but in this case, the fibers were identified according to their staining intensity as type IIa/IId (I), type IIb (F), and type I (S) in control (●, ■, ▲, respectively) and undernourished (○, □, Δ, respectively) muscles, as illustrated in (A and B). Unpaired t-test, *p<0.05; **p<0.01; ***p<0.001.
Statistical Analysis
All values are expressed as the mean ± SD. The differences between the different age groups and feeding conditions were assessed by the unpaired Student’s t-test and Welch’s correction for groups. The statistical analyses were performed by using the GraphPad Prism program (version 5.0; GraphPad Software, San Diego, CA).
Results
Body and EDL Muscle Weight and Total Number of Fibers
At practically all ages (from PN15–PN130), the body weight of undernourished animals was significantly lower than that of control animals (28%–68%; Table 2; p<0.01 to p<0.001), but at PN365, body weight was not significantly different between the groups (Table 2). In addition, the gain of body weight in undernourished animals was less (1.6 ± 0.02 g body weight/d) than that of control animals (3.2 ± 0.04 g body weight/d). In concordance with the latter, a significant reduction in EDL muscle weight (from 41%–61%; Table 2; p<0.01 to p<0.001) was observed in undernourished rats at practically all of the postnatal ages analyzed, with the exception of PN365, when no significant weight differences were observed between control and undernourished muscles. Meanwhile, the total number of fibers in undernourished EDL muscles was not different from that of controls at practically all of the postnatal ages analyzed, with the exception of PN15, when undernourished muscles contained a lesser number of fibers than control muscles (Table 2; p<0.05). These results indicate that chronic undernutrition reduces the weight of the EDL muscle without affecting the total number of fibers in it, from weaning to the adult stages of the rat.
Body Weight, Muscle Weight, and Total Number of Fibers of Control (C) and Undernourished (U) Extensor Digitorum Longus (EDL) Muscles
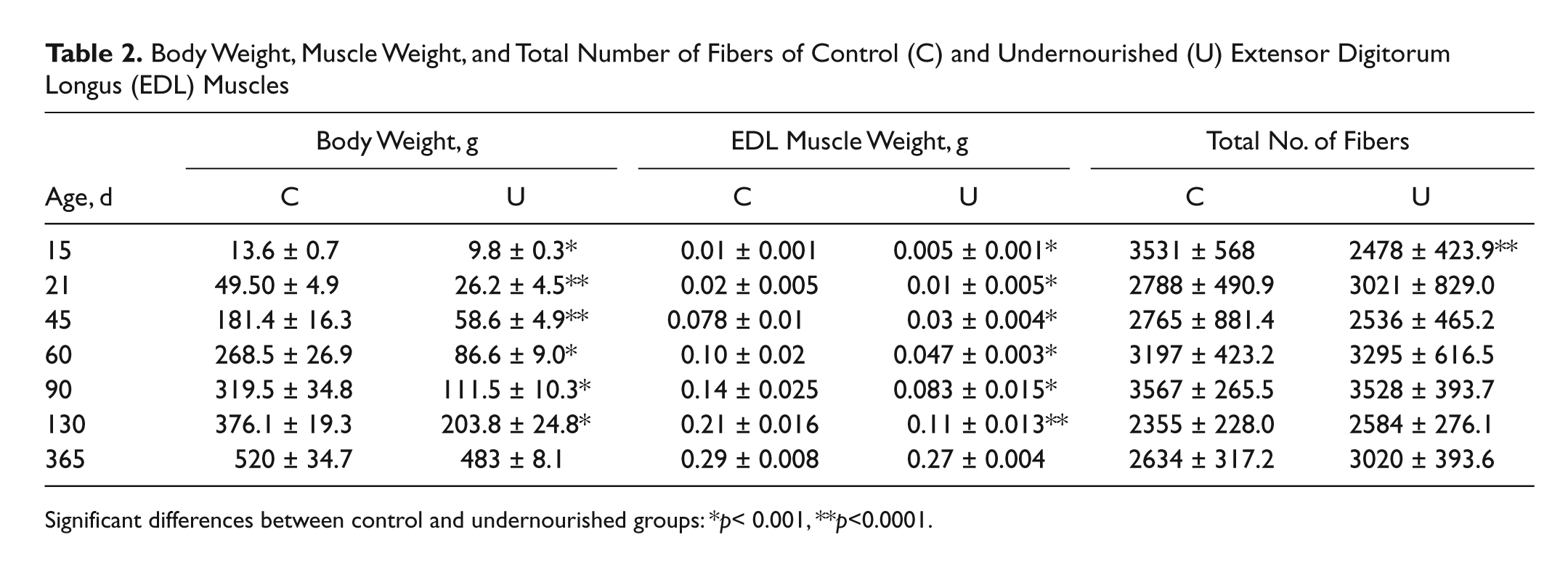
Significant differences between control and undernourished groups: *p< 0.001, **p<0.0001.
Relative Composition of Fiber Types in EDL Muscle during the Postnatal Development of the Rat
Photomicrographs of EDL muscle sections stained with the COX and alkaline ATPase techniques from control and undernourished rats with different postnatal ages are illustrated in Figures 1 and 3. Oxidative (O) muscle fibers appeared dark or gray; glycolytic fibers (G) showed a lighter or paler coloration in both the control and undernourished EDL muscles stained with the COX technique, and slow type I (S), fast type IIb (F), and intermediate type IIa/IId (I) showed light, gray, and dark staining with the alkaline ATPase technique fibers, respectively (see Materials and Methods). Note that from PN15 to PN90, all types of fibers in undernourished muscles showed a smaller size than did controls.
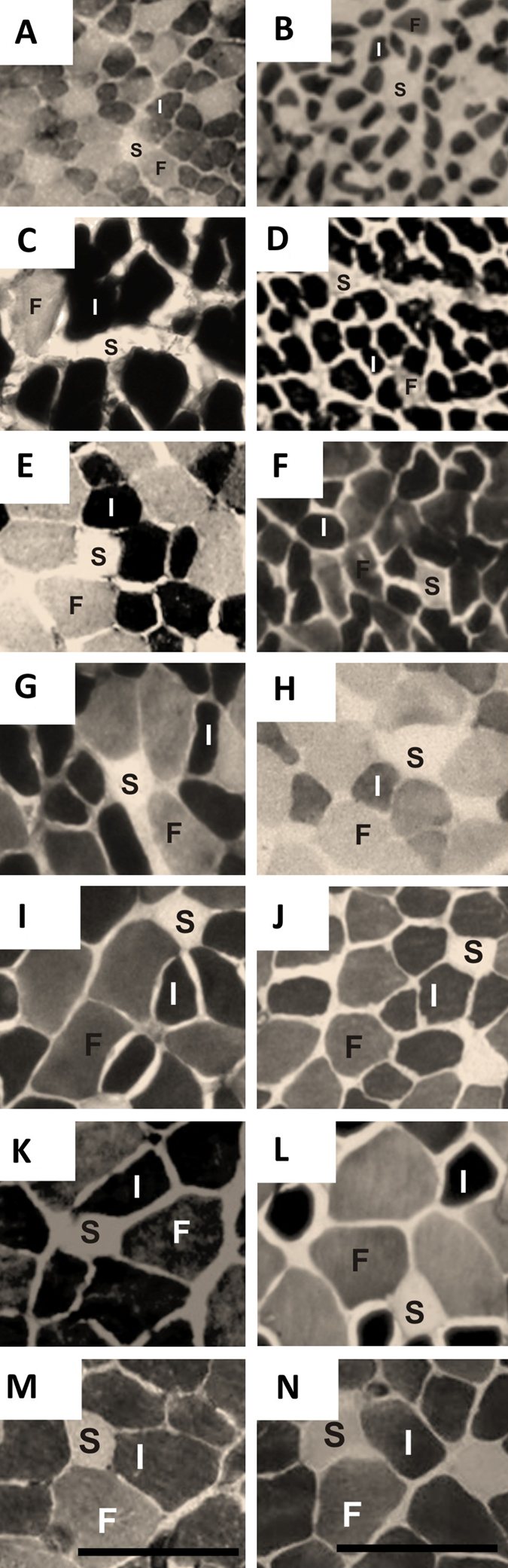
Photomicrographs of extensor digitorum longus muscles from control (left column) and undernourished (right column) rats stained with the alkaline mATPase technique (pH 9.4) at different postnatal ages. Same format as Fig. 1. (A, B) Postnatal day (PN) 15; (C, D) PN21; (E, F) PN45; (G, H) PN60; (I, J) PN90; (K, L) PN130; (M, N) PN365. The fibers were identified according to their staining intensity as type IIa/IId (I), type IIb (F), and type I (S). Calibration bars = 100 µm for all panels.
The graph in Figure 2A illustrates the average percentage of oxidative and glycolytic fibers in the control and undernourished muscles at various postnatal ages in the rat. Unlike the control muscles, the undernourished muscles had a higher percentage (15% to 21% more) of oxidative fibers, accompanied by a corresponding significant decrement in the percentage of glycolytic fibers from PN21 to PN90 (17% to 14% less; Fig. 2A; p<0.01 to p<0.05). However, the percentage of oxidative and glycolytic fibers in both the control and undernourished muscles was not significantly different from PN130 to PN365 (Fig. 2A; p>0.05). From PN21 to PN90, the average ratio of oxidative to glycolytic fibers (RO/G) in the undernourished muscles (range, 1.4–3.1) was higher than in control ones (range, 1.3–1.9), and no differences were found at PN15, PN130, or PN365 (Fig. 4A). The mean percentage of type I fibers present in undernourished muscles was not significantly different from control muscles at any postnatal age (Fig. 2B). However, the percentage of type IIa/IId fibers in the undernourished EDL muscles from PN15 to PN45 was significantly higher than that in control muscles, which was correlated with a proportional decrease in the percentage of type IIb fibers at the same postnatal ages (Fig. 2B; p<0.05 to p<0.01). These proportions of fibers were reversed at PN130 and PN365, when the percentage of type IIb fibers was higher than type IIa/IId fibers (Fig. 2B), but no significant differences were found between fiber types in both groups of muscles at those ages. From PN15 to PN45, the ratio of type IIa/IId with respect to type IIb fibers (RI/F) in undernourished muscles was higher than that of control muscles (Fig. 4B; p<0.01 to p<0.05). Meanwhile, at adult stages (from PN130–PN365), the RI/F ratio was less than that of the control group (Fig. 4B). These results may indicate that chronic undernutrition exerts a differential effect on the proportion of muscle fiber types at different postnatal stages of development, with a greater effect in juveniles than in adults.
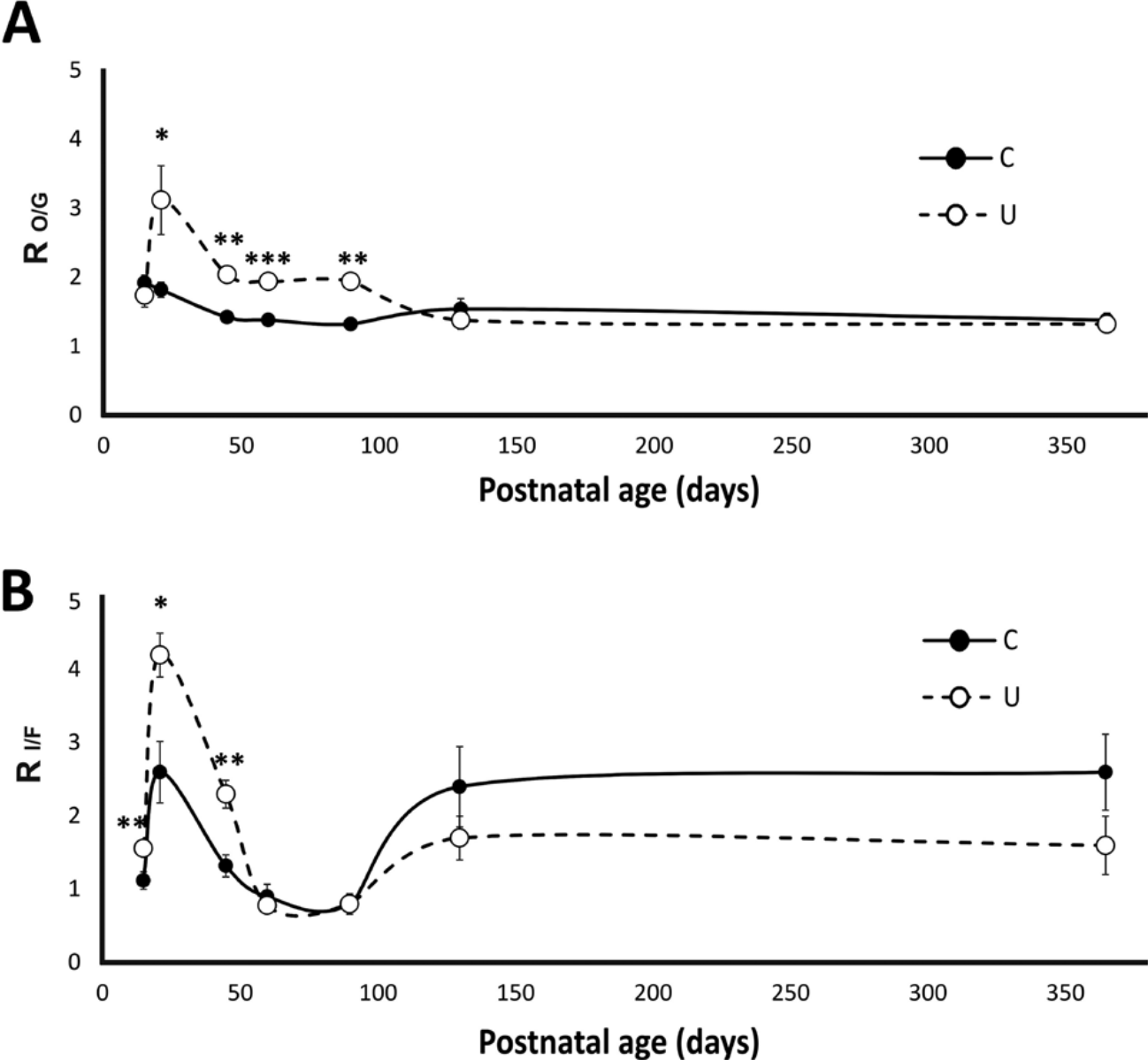
The effect of chronic undernutrition on the fiber type ratio of extensor digitorum longus (EDL) muscles during different postnatal stages of the rat. (A) Changes in the mean ± SD ratios of the oxidative (O) and glycolytic (G) fibers in the control (C) group’s and the undernourished (U) group’s EDL muscles stained with the COX technique (RO/G) at different postnatal ages (from 15–130 days of postnatal life). (B) Same as (A) but showing the changes in the mean ± SD ratios of type IIa/IId (I) and type IIb fibers (F) in EDL muscles with the alkaline ATPase (RI/F) technique (n=5 per age and feeding condition). See the Materials and Methods section for further details. Significant differences between control and undernourished muscles were tested with the unpaired t-test. *p<0.05; **p<0.001; ***p<0.0001.
Cross-Sectional Area of Fibers in the EDL Muscle during the Postnatal Development of the Rat
The graphs in Figure 5A, C, E illustrate the mean cross-sectional area of type I, IIa/IId, and IIb fibers in the EDL muscle of control and undernourished rats at different postnatal ages (from PN15–PN365). As illustrated in graphs, the different fiber types in undernourished muscles showed significant reductions in their cross-sectional area from PN15 to PN90, as compared with control ones (Fig. 5A, C, E, respectively; p<0.001 to p<0.01). At PN365, the fibers in both control and undernourished EDL muscles showed no significant differences in their cross-sectional area but exhibited a slight decrease in thickness (hypotrophic effect). It is noteworthy that the three different types of fibers in control muscles showed an exponential growth phase (hypertrophy) at earlier postnatal ages of life (from PN15–PN45), reaching their maximal cross-sectional area at young adult stages (around PN60–PN130). By comparison, the fibers of undernourished muscles grew almost linearly (Fig. 5B, D, F), reaching their highest cross-sectional area at later postnatal ages than that in controls (around PN90–PN130).
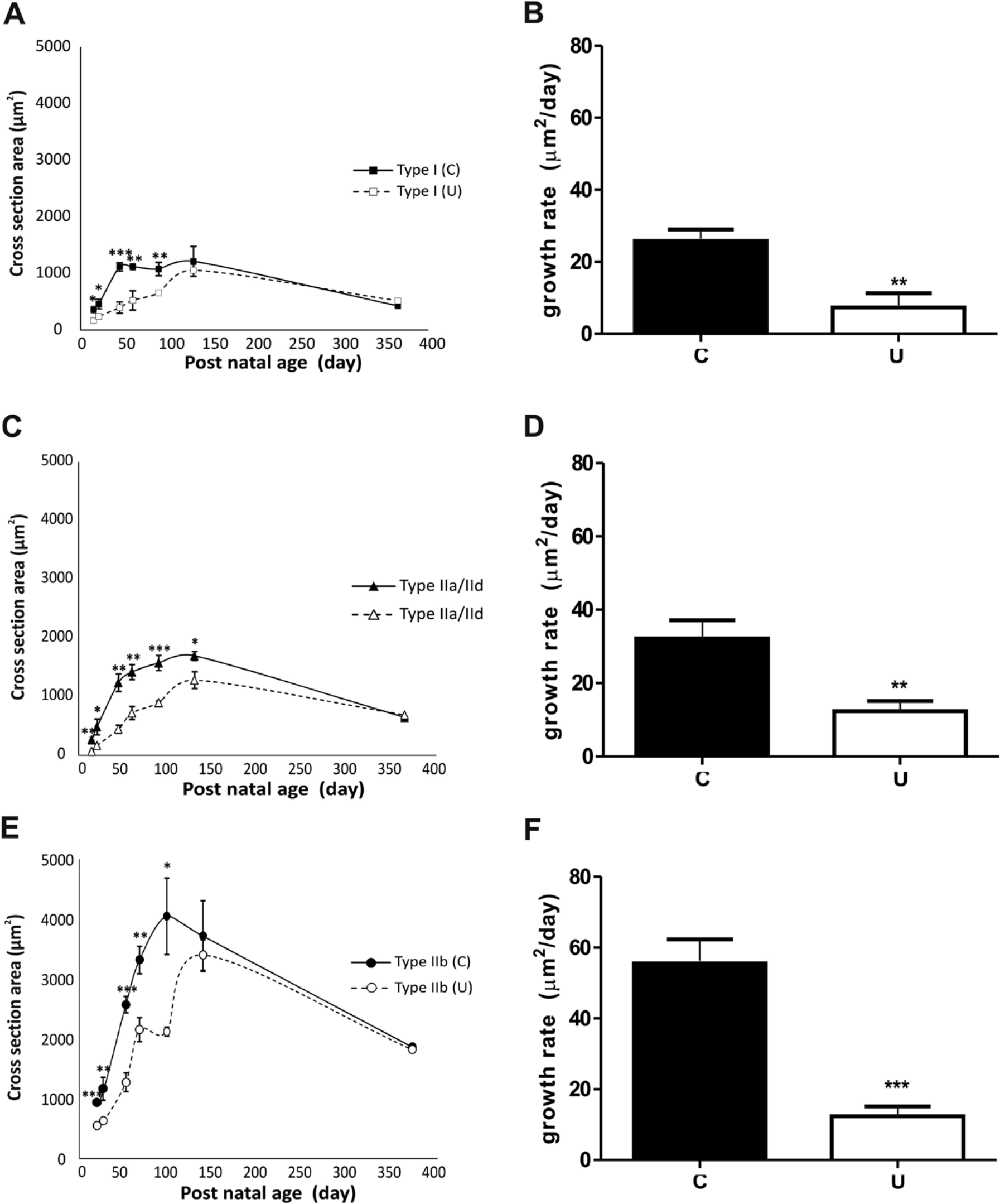
The effects of chronic undernutrition on the cross-sectional areas and growth rates of fiber types in the extensor digitorum longus muscle at different postnatal ages of the rat. (A, C, E) Graphs illustrating the mean ± SD cross-sectional areas of type I, IIa/IId, and IIb fibers from control (C; ■, ▲, ●, respectively) and undernourished muscles (U; ○, □, Δ, respectively). (B, D, F) Graphs indicating the mean ± SD growth rate of the different fiber types (S, I, F) in control (■) and undernourished (□) muscles, respectively. Significant differences were obtained with the two-way analysis of variance test for data illustrated in (A), (C), and (E). *p<0.05; **p<0.01; ***p<0.001.
Discussion
It has been assumed that a severe nutritional restriction significantly affects the growth and maturation of tissues and cells, and when undernourishment occurs earlier, its effects are more severe, generalizing the alterations to organs and/or systems in the organism (Ihemelandu 1985; Zhu et al. 2006). According to our data, the body and muscle weights of chronically undernourished animals were significantly lower than those of control animals. Similar results have been reported in other studies (Rowe 1968; Beverly et al. 1991; Segura et al. 2001), in which a close relationship between low food intake and reduced body and muscle weight of the offspring has been suggested. In this regard, Bayol and colleagues (2004) suggested that there is no relationship between maternal food intake during gestation and the postnatal body growth rate in the offspring, but we found in this study that the body growth of chronically undernourished animals was inferior to that of control animals (see Results). The differences between both studies possibly are related to the feeding paradigms employed; Bayol and colleagues used maternal food deprivation followed by ad libitum food restitution, whereas in this study, we maintained the food restriction throughout the whole life span of animals. In addition, previous studies have suggested that the reduction in muscle weight of undernourished animals is associated with the loss of tissue proteins and/or decreases in the fiber area and volume (hypotrophy) or a reduction in the total number of fibers (hypoplasia; Rowe 1968). Some studies have shown that prenatal undernutrition decreased by 19% to 34% the total number of fibers in the lumbrical and soleus muscles of newborn rats (Ameredes et al. 1999). Similar alterations have been found in pigs and ewes with different feeding protocols to provoke undernutrition than that used in this study (Wilson et al. 1988). Other studies affirm that the number of muscle fibers does not change during the postnatal development of the undernourished rat (Dwyer et al. 1994; Dermitas and Özcan 2012). In agreement with the latter, our results indicate that chronic undernourishment has essentially no effect on the number of fibers in the EDL muscle at any postnatal age, with the exception of PN15, when there is a noticeable reduction in the number of fibers, which probably is associated with a delay in the hypoplasia process due to an early effect of undernourishment; this assumption requires a more detailed analysis than that carried out in this study. According to the latter, it could be assumed that the observed decrement in muscle weight of juvenile and adult undernourished animals is mostly due to a loss of cellular components, instead of a reduction in the number of muscle fibers (Haltia et al. 1978).
On the other hand, our data also indicate that, at juvenile stages, all fiber types in undernourished EDL muscles showed a smaller cross-sectional area and a lower growth rate than control muscles. It is noteworthy that the growth rate of type IIb fibers was the most affected, followed by type IIa/IId, and the least altered was that corresponding to type I fibers from juvenile and young adult undernourished animals (PN15–PN90). However, at adult stages, the cross-sectional area of all fibers was similar in control and undernourished muscles. This suggests that chronic undernourishment induces a delay and/or alteration in the hypertrophy process of the EDL muscle fibers in the juvenile and young adult rat (see Goldspink 1964; Rowe 1968).
The histoenzymatic evidence obtained in this study indicates that chronic undernourishment induces a relative increase in the proportion of fibers showing oxidative metabolism at the expense of glycolytic fibers in the EDL muscle in late preweaning, juvenile, and adult stages (PN15–PN90). In addition, a significant increase in the relative proportion of intermediate type IIa/IId fibers was accompanied by a proportional decrease in the relative proportion of type IIb fibers from PN15 to PN45. Our results partially agree with those reported in previous studies, which found that, at juvenile stages of life, several food deprivation paradigms (applied at different postnatal stages of development) induce an increase in the proportion of oxidative compared with glycolytic fibers in distinct fast muscles (Bedi et al. 1982; Lewis et al. 1986; Yamaguchi et al. 1993; White et al. 2000). The relatively prolonged period of time in which EDL fibers show a predominance of oxidative metabolism in the undernourished muscles may serve as a protective mechanism against food deprivation, similar to that observed in skeletal muscles subjected to extreme conditions of enhanced neuromuscular activity (Close 1964; Howells et al. 1978; Raju 1975; Sieck et al. 1989; White et al. 2000). Based on the consideration that muscles containing type II fibers with lower oxidative capacity are more easily fatigued, whereas muscles constituting mostly type I fibers with higher oxidative capacity are more fatigue resistant (Burke and Edgerton 1975; Donoghue et al. 2005), as well as the results obtained in this study indicating that undernourished EDL muscles contain an increased proportion of fibers showing oxidative metabolism concomitantly with a higher proportion of type IIa/IId fibers at PN21 to PN45, it could be argued that chronic undernourishment induces an increase in muscle fatigue resistance at those ages. In accordance with this hypothesis, an increase in the fatigue index (higher resistance to fatigue) of diaphragm muscles in the undernourished rat has been demonstrated previously (Sieck et al. 1987). In summary, the evidence obtained in this study suggests that chronic undernourishment affects the fiber growth of the EDL muscle by decreasing their cell volume and/or protein content, and such alterations seem to occur concomitantly with changes in the transformation and/or differentiation from type IIa/IId to type IIb fibers and the enhancement of oxidative metabolism in the EDL muscle at the early and juvenile stages of postnatal life in the rat.
Conclusion
The evidence obtained in this study suggests that chronic undernourishment negatively influences muscle maturation, particularly at the early and juvenile stages of life, when the fibers increase in thickness and volume. These changes are accompanied by an enhancement in the fiber oxidative metabolism and a modification in the transformation and/or differentiation of type IIa/IId to type IIb fibers in the EDL muscle. These changes could indicate the probable occurrence of adaptive mechanisms that allow muscles to counterbalance, in a plastic and dynamic manner, the alterations provoked by chronic food deprivation.
Footnotes
Acknowledgements
We thank American Journal Experts for editing the English in this text, E. Velazquez and P. Reyes for computing assistance, and Dr. Maria Eugenia Mendoza Garrido and M. en C. Cesar Gabriel Toriz González for their valuable support in obtaining photomicrographs.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was partially supported by fellowships to Ismael Jiménez-Estrada from the Sistema Nacional de Investigadores and to Azucena Ruiz-Rosado from the Consejo Nacional de Ciencia y Tecnología, México.
