Abstract
Cat masticatory muscle during regeneration expresses masticatory-specific myofibrillar proteins upon innervation by a fast muscle nerve but acquires the jaw-slow phenotype when innervated by a slow muscle nerve. Here, we test the hypothesis that chronic low-frequency stimulation simulating impulses from the slow nerve can result in masticatory-to-slow fiber–type transformation. In six cats, the temporalis muscle was continuously stimulated directly at 10 Hz for up to 12 weeks using a stimulator affixed to the skull. Stimulated muscles were analyzed by immunohistochemistry using, among others, monoclonal antibodies against masticatory-specific myosin heavy chain (MyHC), myosin binding protein-C, and tropomyosins. Under the electrodes, stimulation induced muscle regeneration, which generated slow fibers. Deep to the electrodes, at two to three weeks, two distinct populations of masticatory fibers began to express slow MyHC: 1) evenly distributed fibers that completely suppressed masticatory-specific proteins but transiently co-expressed fetal MyHCs, and 2) incompletely transformed fibers that express slow and masticatory but not fetal MyHCs. SDS-PAGE confirmed de novo expression of slow MyHC and β-tropomyosin in the stimulated muscles. We conclude that chronic low-frequency stimulation induces masticatory-to-slow fiber–type conversion. The two populations of transforming masticatory fibers may differ in their mode of activation or lineage of their myogenic cells.
Keywords
The jaw-closing muscles of the cat differ in allotype from limb muscles (Hoh and Hughes 1988) and in the fiber types and myofibrillar proteins they express (Hoh 2002). While cat limb muscles have slow and fast 2a and 2x fibers (Lucas et al. 2000), jaw-closing muscles of the cat have two unique fiber types: masticatory and jaw-slow. Masticatory fibers are known to express a set of jaw-specific myofibrillar proteins: masticatory myosin heavy chain (m-MyHC), masticatory light chain (Rowlerson et al. 1981; Qin et al. 1994; Qin et al. 2002), masticatory tropomyosin (m-Tm) (Rowlerson et al. 1983; Kang et al. 2010), and masticatory myosin binding protein-C (m-MBP-C) (Kang et al. 2010). Masticatory light chain-1 has recently been shown to be identical to atrial or embryonic light chain-1 (Reiser et al. 2010). Although masticatory myosin has a high ATPase (Rowlerson et al. 1981), masticatory fibers are associated with moderate speed of contraction (Hoh et al. 2007; Toniolo et al. 2008) but high tension cost (Saeki et al. 1987). The mobility of masticatory myosin heads and their protrusion towards thin filaments (Yamaguchi et al. 2010) may help explain the high Ca2+ sensitivity (Kato et al. 1985) and high maximal stress (Saeki et al. 1987; Toniolo et al. 2008) of these fibers. Jaw-slow fibers express slow MyHC associated with masticatory light chains (Sciote et al. 1995; Hoh et al. 2007); their cross-bridge cycling kinetics are considerably higher than those of limb-slow fibers (Hoh et al. 2007). They express β-tropomyosin and a slow isoform of MBP-C (Kang et al. 2010).
Mammalian limb muscle fibers are physiologically plastic; their phenotypic characteristics are under neural and hormonal control. Cross-innervation of fast and slow limb muscles tends to reverse their phenotypic properties (Buller et al. 1960; Hoh 1975; Hoh et al. 1980; Pette and Vrbova 1985). These effects are mediated by the pattern of nerve impulses, which differ for different fiber or motor unit types (Buller et al. 1960; Hennig and Lomo 1985). Slow-to-fast fiber transformation occurs in limb muscles under conditions of reduced impulse activity (Hoh et al. 1980; Baldwin et al. 1994; Stevens et al. 2000). In contrast, increasing functional load, or stimulating a fast muscle with sustained low-frequency impulses characteristic of slow motoneurons, leads to fast-to-slow fiber transformation (Salmons and Sreter 1976; Pette and Vrbova 1985). The surge in thyroid hormone levels that occurs during vertebrate ontogenesis (Hulbert 2000) is essential for normal muscle development, the hormone facilitating the transition from fetal to fast MyHC expression (Gambke et al. 1983; d’Albis et al. 1987; Adams et al. 1999). In mature animals, hyperthyroidism shifts fibers towards fast types, while hypothyroidism has the reversed effect (Zhong et al. 2010; Caiozzo and Haddad 1996). These thyroidal influences modulate the neural influence on muscle fiber types, including the effects of chronic low-frequency stimulation (Kirschbaum et al. 1990; Ianuzzo et al. 1991; Swoap et al. 1994).
Muscle fibers of the jaw allotype are also physiologically plastic. Satellite cell cultures (Kang et al. 2010) and uninnervated regenerates (Hoh and Hughes 1991) of cat masticatory muscle reveal their allotypic nature by expressing masticatory-specific myofibrillar proteins. Upon innervation by a limb fast muscle nerve, regenerating masticatory muscle continues to express masticatory myofibrillar proteins (Hoh and Hughes 1988; Kang and Hoh 2010). These proteins are also initially expressed upon innervation by a slow limb muscle nerve, but in the long term, most regenerated muscle fibers express only slow MyHC (Hoh and Hughes 1988) and acquire other phenotypic properties of jaw-slow fibers (Kang and Hoh 2010). It is postulated that myogenic cells of the jaw allotype express masticatory-specific myofibrillar proteins during myogenesis by default and that stimulation by the tonic, low-frequency impulses from slow motoneurons is responsible for diverting myofibrillar protein gene expression towards the jaw-slow phenotype (Kang and Hoh 2010).
In this work, we test the hypothesis that chronic low-frequency stimulation can convert masticatory fibers into jaw-slow fibers. We use a stimulator attached to the cat’s skull to stimulate the posterior temporalis muscle and examine stimulated muscles immunohistochemically using a battery of monoclonal antibodies (MAbs) against slow and masticatory-specific myofibrillar proteins. We show that such stimulation induces the transformation of masticatory fibers into jaw-slow fibers.
Methods
Stimulation of Cat Jaw Muscles
Six adult cats were used in this study, which was approved by the Animal Ethics Review Committee of The University of Sydney. The work was done in conformity with the Australian Code of Practice for the Care and Use of Animals for Scientific Purposes. Under general anesthesia (30 mg/kg sodium pentobarbitone administered intraperitoneally), a specially constructed stimulator measuring 2.5 cm3 was attached onto the skull. This was achieved by drilling two holes into the sphenoidal sinus and bonding the stimulator to the skull with dental acrylic. Platinum-wire electrodes, insulated by medical-grade silicone rubber tubing up to 4 mm from the tip, were inserted into nicks made in the fascia overlying the posterior temporalis muscle. They were placed 10 mm apart, each secured at two points by suturing to fascia. The electrodes were well away from the main bundle of the motor nerve, but some intramuscular branches might be sufficiently near to be stimulated. Identical electrodes not connected to the stimulator were placed in the contralateral temporalis muscle to serve as a control. The stimulator was powered by a stack of 10 calculator batteries (13.5 V). The posterior temporalis contains only masticatory fibers (Rowlerson et al. 1981; Hoh et al. 1988a). It was stimulated continuously with 1-millisecond pulses at 10 Hz for up to 12 weeks. The animals were inspected daily to ensure continuity of stimulation. Stimulus strength (8 V maximum) was adjusted to produce palpable local contractions without causing apparent distress to the animal. Both stimulated and control muscles were removed for study under general anesthesia after 10, 15, 21, 27, 42, and 84 days.
Immunohistochemistry
Each muscle was cut into about 10 blocks, each of which was examined by horseradish peroxidase (HRP) immunohistochemistry as previously described (Hoh et al. 1988a) using various antibodies against myofibrillar proteins. Primary antibodies used were a polyclonal antibody raised in sheep against fetal (embryonic and neonatal) MyHCs (Hoh et al. 1988b) and the following MAbs: anti–m-MyHC 2F4 (Kang et al. 1994) (now available from Developmental Studies Hybridoma Bank, University of Iowa; Iowa City, IA), anti–m-Tm 1H2 and anti–m-MBP-C 3F10 (Kang et al. 2010), anti–slow MyHC NOQ-7-5-4D (Hoh et al. 1988b), anti–tropomyosin CH1 (Lin et al. 1985), and MAb 1A10 (Hoh et al. 1991). MAb 1A10 binds fetal MyHCs and jaw-slow, but not limb-slow, myosin. This capacity of MAb 1A10 to distinguish jaw-slow from limb-slow myosin is likely due to the association of masticatory myosin light chains with slow MyHCs in jaw-slow fibers (Hoh et al. 2007). MAb CH1 reacts with all jaw and limb muscle tropomyosin isoforms (Hoh et al. 1989). The secondary antibodies used were HRP-labeled rabbit anti-mouse and anti-sheep immunoglobulin antibodies (Dako; Glostrup, Denmark).
Biochemical Analysis of MyHCs and Tropomyosins
MyHCs from stimulated muscles and normal cat muscles were analyzed by SDS-PAGE and Western blotting. To provide reference fetal, slow, and masticatory MyHCs, crude myosins were extracted (with 100 mM sodium pyrophosphate, pH 8.3) from adult masseter (which contains masticatory and jaw-slow fibers) and soleus muscles from a four-day-old kitten. Myosin extracts and tissue sections (10–15 slices, 10 µm thick) were boiled in 100 µL sample buffer in an Eppendorf tube and cleared by centrifugation (bench microfuge) for one minute before application to the gel. SDS-PAGE was performed on 6% minislab gels (8 cm × 10 cm, 0.75 mm thick) according to the procedure of Danieli Betto et al. (1986). Gels were run at 4C at 120 V for about 16 hours, stained with Coomassie Brilliant Blue R-250 or Western blotted (Towbin et al. 1979); the primary antibody used was anti–slow MyHC, and the secondary antibody was HRP-coupled anti-mouse immunoglobulin antibodies.
SDS-PAGE of tropomyosins of stimulated muscles was done in 15% gels, using extracts of cryostat-sectioned tissues in sample buffer. The presence of β-tropomyosin was demonstrated by Western immunoblotting, using anti–tropomyosin MAb CH1 as the primary antibody and HRP-coupled anti-mouse immunoglobulin as the secondary antibody. Staining of the blotting membrane for protein was performed as described by Kumar and Lakshmi (1985).
Results
Control Temporalis Muscles
Immunohistochemical analysis of control muscles shows that electrode implantation without stimulation is associated with muscle regeneration and granulation tissue formation in the immediate neighborhood of the electrodes. Regenerating fibers up to 27 days after surgery expressed fetal, slow, and masticatory MyHCs (data not shown), consistent with earlier work on regenerating masticatory fibers (Hoh and Hughes 1988; Kang and Hoh 2010). Deep to the electrodes, all fibers stain only with anti–m-MyHC MAb. Control temporalis muscle fibers 42 and 84 days after surgery are virtually pure masticatory, indicating that regenerating fibers end up as masticatory fibers.
Immunohistochemistry of Stimulated Temporalis Muscles
Immunohistochemical examination of stimulated temporalis at 10 days reveals little difference from control muscle. Change is first observed at 15 days. Figure 1 shows serial sections of 15-day stimulated temporalis stained with anti–m-MyHC (Fig. 1A), anti–slow MyHC (Fig. 1B), anti–m-Tm (Fig. 1C), anti–m-MBP-C (Fig. 1D), MAb 1A10 (Fig. 1E), and anti–fetal MyHC (Fig. 1F) antibodies. The location of the stimulating electrodes is at the bottom of each panel. Subjacent to the electrodes, groups of contiguous fibers stain strongly with anti–slow MyHC, MAb 1A10, and anti–fetal MyHC antibodies, many of which also stain for masticatory-specific proteins, consistent with regenerating fibers. In the deeper region of this muscle are fibers (examples arrowed in each panel) that stain with anti–slow MyHC (Fig. 1B), MAb 1A10 (Fig. 1E), and anti–fetal MyHC (Fig. 1F) antibodies but are not stained with masticatory-specific MAbs. These transformed fibers are scattered among unchanged masticatory fibers.
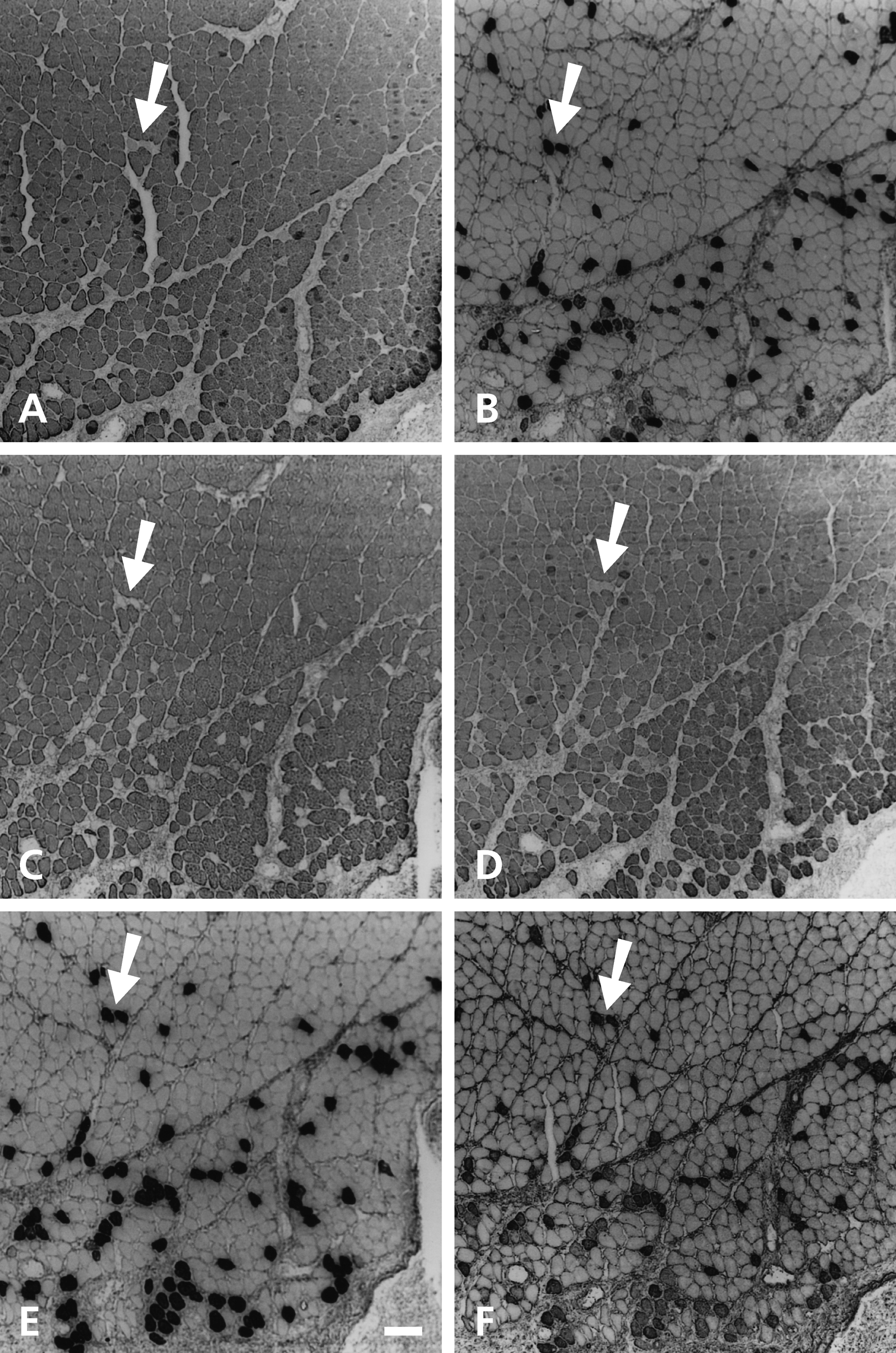
Cross-sections of temporalis muscle stimulated for 15 days stained with anti–m-MyHC (A), anti–slow MyHC (B), anti–m-Tm (C), anti–m-MBP-C (D), MAb 1A10 (E), and anti–fetal MyHC (F) antibodies. Stimulating electrodes are located at the bottom of each panel. Arrow points to two completely transformed fibers deep to the electrodes. Scale bar: 100 µm.
Figure 2 shows a montage of low-power photomicrographs of the 21-day stimulated muscle, stained with anti–slow MyHC MAb. This muscle has extensive granulation tissue at the electrode implantation site (top of Fig. 2), encroached by bands of slow-staining regenerating fibers, which also stain to varying degrees with anti–m-MyHC, MAb 1A10, and anti–fetal MyHC antibodies (data not shown). In sharp contrast to the unstimulated muscle, numerous slow-staining fibers are seen scattered throughout the thickness of the muscle.

A montage of low-power photomicrographs of a cross-section of 21-day stimulated temporalis stained with anti–slow MyHC MAb. Note the extent of slow staining fibers deep to the stimulating electrodes, which are located at the top. Scale bar: 200 µm.
Figure 3 shows serial sections of the deep fibers of the 21-day stimulated muscle at a higher magnification, stained with anti–m-MyHC (Fig. 3A), anti–slow MyHC (Fig. 3B), anti–m-Tm (Fig. 3C), anti–m-MBP-C (Fig. 3D), MAb 1A10 (Fig. 3E), and anti–fetal MyHC (Fig. 3F) antibodies. The smaller, transformed fibers seen at 15 days (staining with anti–slow MyHC, MAb 1A10, and anti–fetal MyHC antibodies but not with masticatory-specific MAbs) are also seen here. Comparing Figure 3B and 3E reveals a new subpopulation of fibers staining with anti–slow MyHC. These fibers are not stained by MAb 1A10 and anti–fetal MyHC antibody, but they retain staining with masticatory-specific MAbs. The unexpected mismatch between anti–slow MyHC and MAb 1A10 staining is probably due to the presence of a low level of slow MyHC and absence of fetal MyHCs in these fibers.
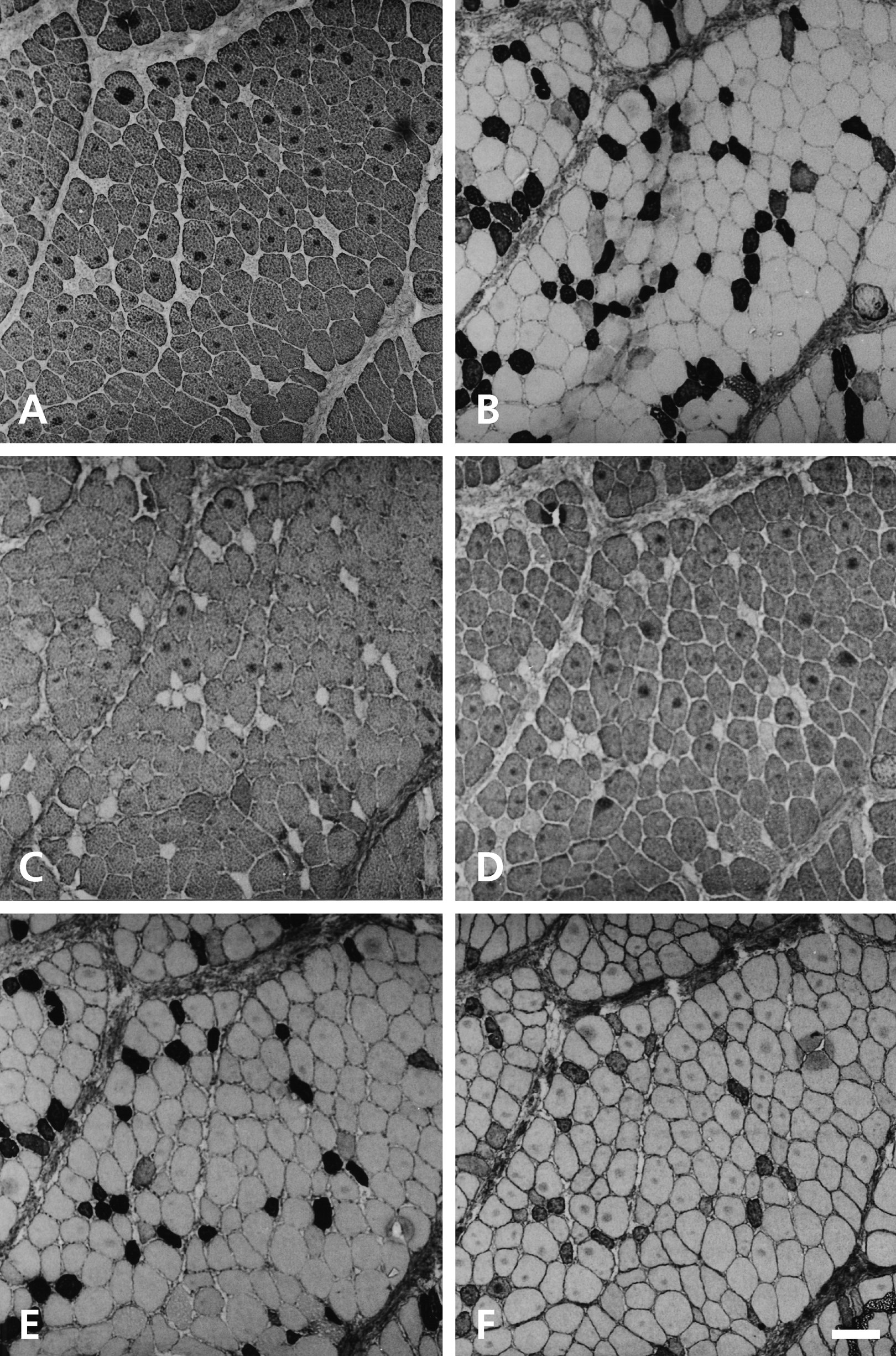
Semiserial sections of the deep region of the 21-day stimulated temporalis muscle shown in Figure 2 stained with anti–m-MyHC (A), anti–slow MyHC (B), anti–m-Tm (C), anti–m-MBP-C (D), MAb 1A10 (E), and anti–fetal MyHC (F) antibodies. Scale bar: 100 µm.
Figure 4 shows serial sections of a 27-day stimulated temporalis muscle subjacent to the stimulating electrodes stained with anti–m-MyHC (Fig. 4A), anti–slow MyHC (Fig. 4B), MAb 1A10 (Fig. 4C), and anti–fetal MyHC (Fig. 4D) antibodies. Most fibers here are stained with anti–m-MyHC (Fig. 4A) and anti–slow MyHC (Fig. 4B). There tends to be a reciprocal relationship in the intensity of staining for these two antibodies: vertical arrows point to a fiber strongly stained by anti–slow MyHC but weakly stained by anti–m-MyHC, and vice versa for horizontal arrows. Fibers staining for fetal MyHCs (Fig. 4D) constitute only a fraction of those staining for slow MyHC. The pattern of staining with MAb 1A10 (Fig. 4C) reflects that of slow MyHC (Fig. 4B) rather than that of fetal MyHCs (Fig. 4D), indicating a weaker presence of fetal MyHCs relative to the 15-day stimulated muscle (Fig. 1).
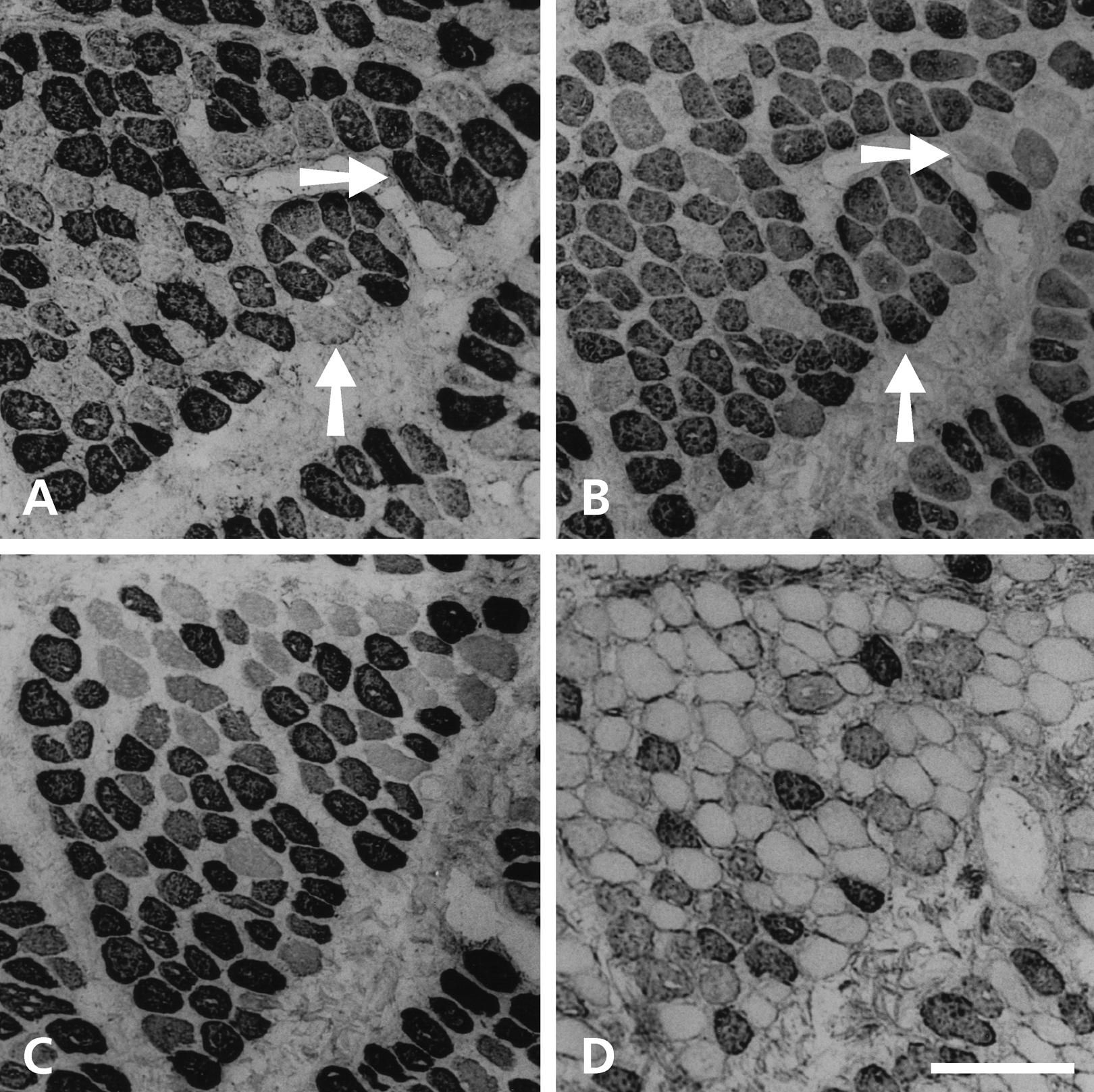
Serial sections of the 27-day stimulated temporalis muscle beneath the stimulating electrodes stained with anti–m-MyHC (A), anti–slow MyHC (B), MAb 1A10 (C), and anti–fetal MyHC (D) antibodies. Horizontal arrows point to a fiber strongly stained by anti–m-MyHC but weakly stained by anti–slow MyHC, while vertical arrows point to a fiber staining strongly with anti–slow MyHC but weakly with anti–m-MyHC. Scale bar: 100 µm.
Figure 5 shows serial sections of a 42-day stimulated temporalis muscle deep to the stimulating electrodes stained with anti–m-MyHC (Fig. 5A), anti–slow MyHC (Fig. 5B), MAb 1A10 (Fig. 5C), and anti–fetal MyHC (Fig. 5D) antibodies. Compared with the deep region of 21-day stimulated muscle (Fig. 3), a larger proportion of fibers stains with anti–slow MyHC. The two distinct populations of slow-staining fibers are even more obvious now: 1) a small, evenly dispersed population of completely transformed fibers (strongly stained with anti–slow MyHC and MAb 1A10 but not stained with anti–m-MyHC) that still weakly stain with anti–fetal MyHC (example arrowed), and 2) a clustered population of large, incompletely transformed fibers (staining more weakly with anti–slow MyHC and MAb 1A10, but still staining strongly with anti–m-MyHC but not with anti–fetal MyHC). The acquisition of some staining with MAb 1A10 by the latter group of fibers suggests an increase in their content of slow MyHC relative to the 21-day stimulated muscle (Fig. 3).
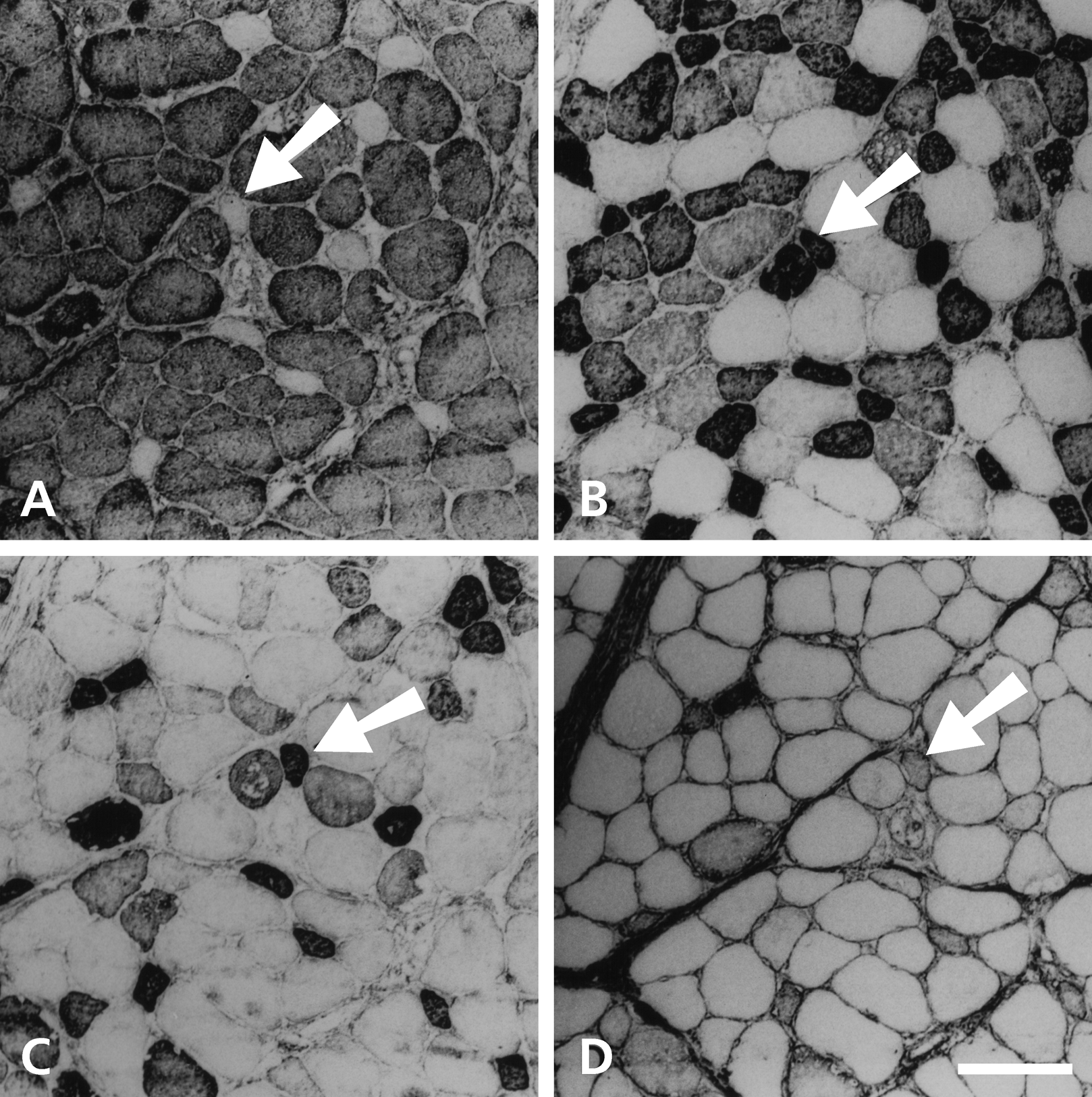
Serial sections of the 42-day stimulated temporalis muscle stained with anti–m-MyHC (A), anti–slow MyHC (B), MAb 1A10 (C), and anti–fetal MyHC (D) antibodies. The arrow in each panel points to an example of the small, completely transformed fiber. Scale bar: 100 µm.
Figure 6 shows serial sections of a 84-day stimulated temporalis muscle subjacent to the electrodes, stained with anti–m-MyHC (Fig. 6A), anti–slow MyHC (Fig. 6B), MAb 1A10 (Fig. 6C), and anti–fetal MyHC (Fig. 6D) antibodies. Anti–slow MyHC and MAb 1A10 now stained fibers in an identical manner, there being a high density of stained fibers near the electrodes, at the top right corner of the photomicrograph, but stained fibers are more sparse away from the electrodes, at the bottom left. The pattern of staining for anti–slow MyHC is roughly reciprocal to that for m-MyHC antibody, although it is clear that many slow fibers co-express m-MyHC. None of the fibers now stained with anti–fetal MyHC antibody.
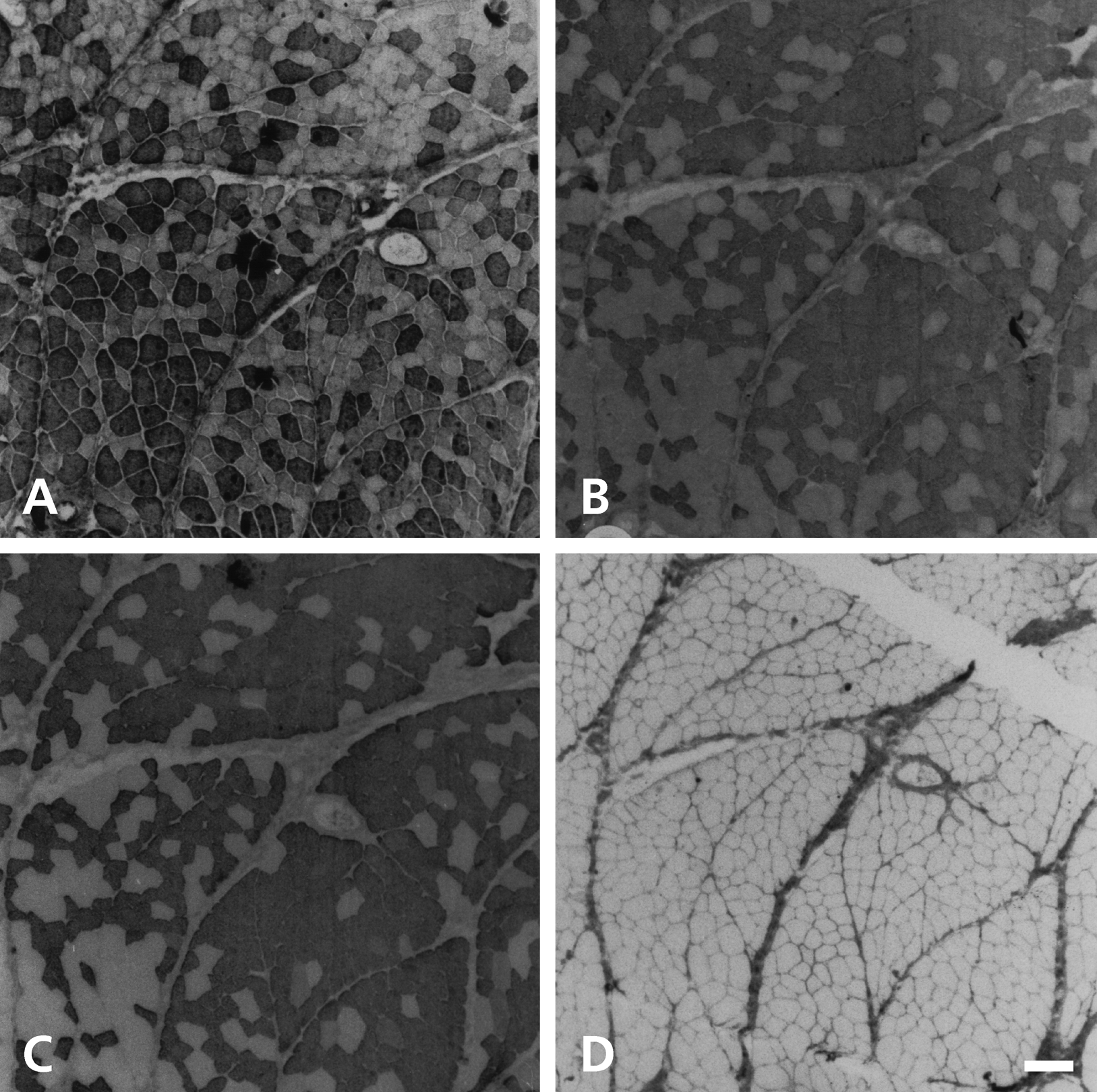
Serial sections of the superficial region of the 84-day stimulated temporalis muscle stained with anti–m-MyHC (A), anti–slow MyHC (B), MAb 1A10 (C), and anti–fetal MyHC (D) antibodies. Scale bar: 100 µm.
Figure 7 shows serial sections of the 84-day stimulated temporalis deep to the electrodes stained with anti–m-MyHC (Fig. 7A), anti–slow MyHC (Fig. 7B), anti–m-Tm (Fig. 7C), and anti–m-MBP-C (Fig. 7D) antibodies. The scattered first population of completely transformed fibers can still be seen (examples arrowed). Compared with the 42-day stimulated muscle, the second population of transformed fibers has intensified in staining with anti–slow MyHC MAb, but the staining patterns of these slow fibers for the masticatory-specific proteins vary. Staining for anti–m-MBP-C (Fig. 7D) is largely complementary to that for anti–slow MyHC (Fig. 7B), indicating successful suppression of m-MBP-C in these slow fibers. Anti–m-MyHC (Fig. 7A) stains nearly all fibers except those that are completely transformed, while some of these anti–m-MyHC staining fibers have lost staining for m-Tm (Fig. 7C). These results indicate that by 84 days, the second population of slow fibers is incompletely transformed: they manage to suppress m-MBP-C completely and m-Tm partially but failed to suppress m-MyHC.
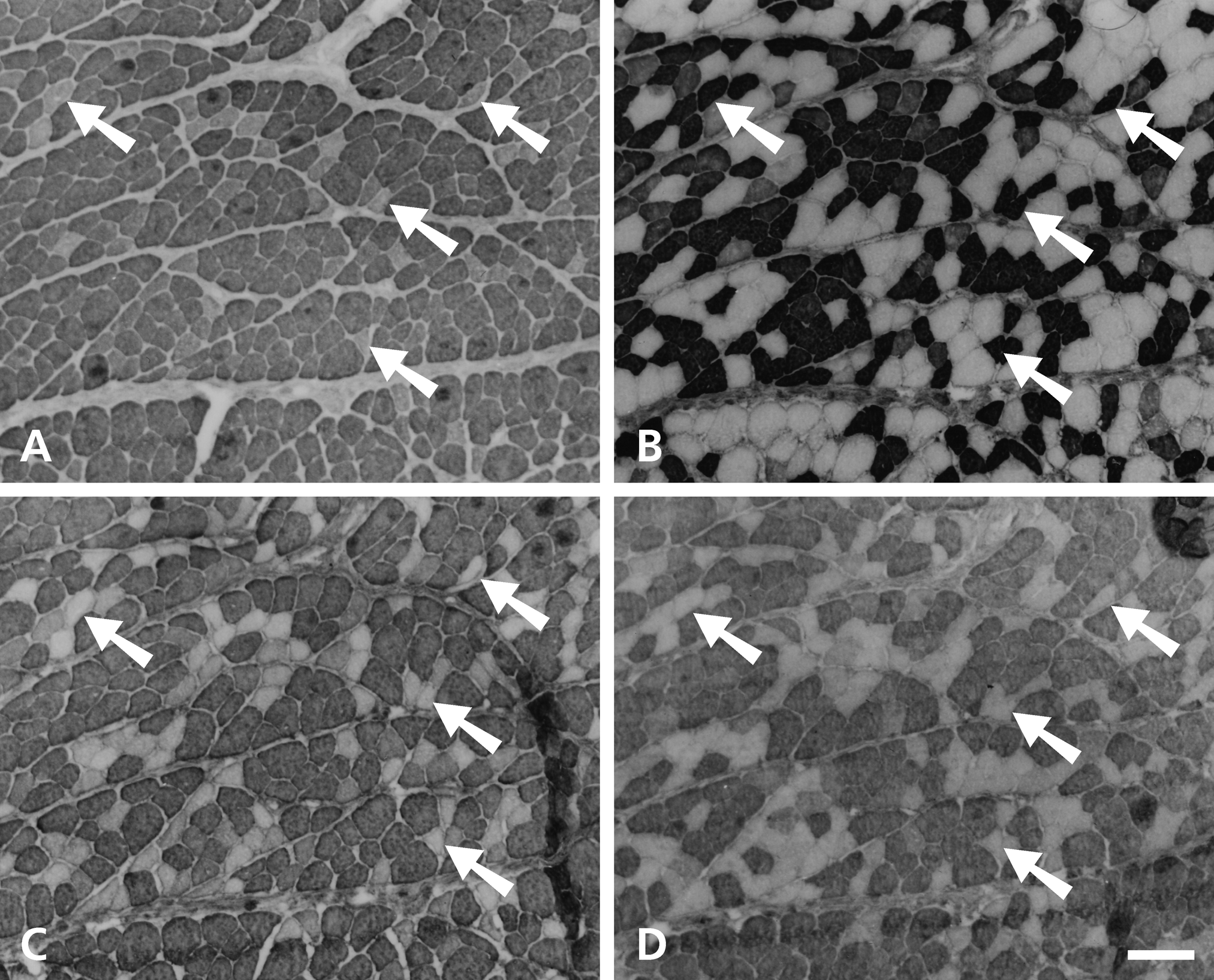
Serial sections of the deep region of the 84-day stimulated temporalis muscle stained with anti–m-MyHC (A), anti–slow MyHC (B), anti–m-Tm (C), and anti–m-MBP-C (D) antibodies. Arrows point to examples of completely transformed fibers. Scale bar: 100 µm.
Table 1 gives a summary of the time course of changes in immunoreactivities of masticatory fibers induced in different regions of the temporalis muscle by chronic low-frequency stimulation.
Summary of the Time Course of Changes in Immunoreactivities of Masticatory Fibers Induced in Different Regions of the Temporalis Muscle by Chronic Low-Frequency Stimulation

Note: Reactivities decrease in the order ++ > + > ± > −. Heterogeneous reactivities within a population are indicated by symbols separated by a slash.
Biochemical Analysis of Slow MyHC and β-tropomyosin in Stimulated Muscles
The presence of slow and fetal MyHCs in stimulated muscles is shown by SDS-PAGE (Fig. 8). To provide controls for slow, fetal, and masticatory MyHCs, kitten soleus and adult masseter myosins are analyzed. Soleus myosin from a four-day-old kitten (Fig. 8A) reveals two bands, fetal and slow MyHCs. Adult masseter (Fig. 8B) shows a major, m-MyHC component that has mobility intermediate between slow and fetal MyHCs and a weaker, slow MyHC component having the same mobility as that in kitten soleus. Slow MyHC is known to be absent in unstimulated temporalis. Temporalis muscle stimulated for 27 days (Fig. 8C) shows three components: slow MyHC and m-MyHC of approximately equal intensity and a faint fetal MyHC component, consistent with immunohistochemistry illustrated in Figure 4. The presence of slow MyHC in cryostat sections of other stimulated muscles has also been confirmed by SDS-PAGE (data not shown).
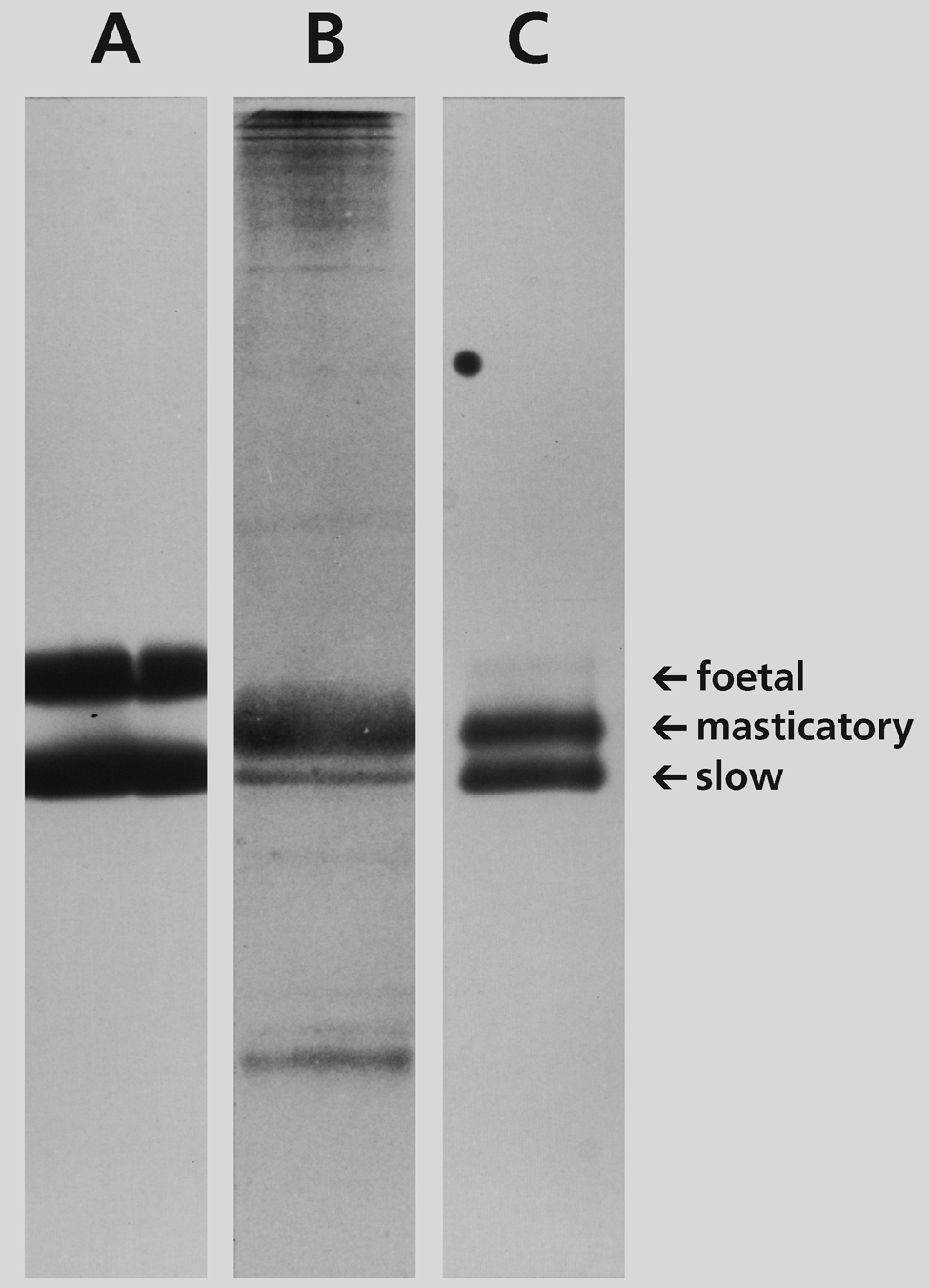
SDS-PAGE of MyHCs extracted from cryostat sections from soleus muscle of a four-day-old kitten (A), normal adult cat masseter muscle (B), and cat posterior temporalis muscle stimulated for 27 days (C). Arrows point to the locations of fetal, masticatory, and slow MyHCs. Note the presence of a slow MyHC component in the masseter (B) and a strong, slow MyHC band and a faint, fetal MyHC band in the stimulated temporalis (C).
Because normal cat jaw-slow muscle fibers express β-tropomyosin, whereas masticatory fibers do not (Kang et al. 2010), the presence of this isoform is examined in stimulated muscles. Figure 9 shows protein-stained SDS-PAGE transblots (Fig. 9A, 9C, 9E, 9G) and Western blots (Fig. 9B, 9D, 9F, 9H) of myofibrillar proteins from control temporalis (Fig. 9A, 9B), temporalis stimulated for 42 days (Fig. 9C, 9D), temporalis stimulated for 84 days (Fig. 9E, 9F), and control soleus muscle (Fig. 9G, 9H). The primary antibody used in the Western transblots, MAb CH1, reacts with all cat skeletal tropomyosin isoforms (Hoh et al. 1989). Here, CH1 stains the two components in soleus muscles: the faster migrating α-tropomyosin and the slower β-tropomyosin. Control temporalis has only a single m-Tm band with the same mobility as α-tropomyosin, while the stimulated muscles show a weak β-tropomyosin band and a strong m-Tm band. This result is consistent with the small proportion of fibers in this muscle not stained for m-Tm (Fig. 7B) and thus likely to express β-tropomyosin.
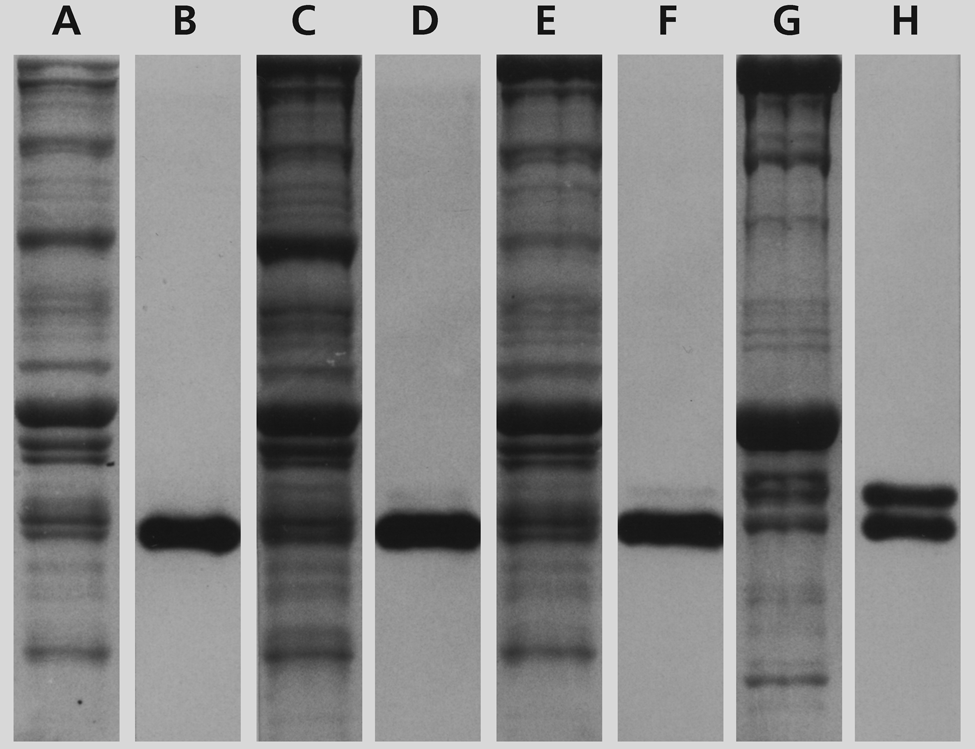
SDS-PAGE and Western blots of myofibrillar proteins extracted from cryostat sections of stimulated temporalis and control soleus muscles. The lanes show protein-stained transblots (A, C, E, G) and corresponding Western blots (B, D, F, H) using MAb CH1 of myofibrillar proteins from control temporalis (A, B), temporalis stimulated for 42 days (C, D), temporalis stimulated for 84 days (E, F), and control soleus muscle (G, H). MAb CH1 binds m-Tm in control and stimulated temporalis muscles (B, D, F), and α and β tropomyosins in the soleus muscle (H). Note the weak presence of β tropomyosin in the stimulated temporalis (D, F).
Discussion
Transformation of Masticatory Fibers into Jaw-Slow Fibers by Chronic Low-Frequency Stimulation
The overall result of this investigation is that chronic low-frequency stimulation of the cat posterior temporalis muscle does transform masticatory fibers into jaw-slow fibers. Electrode implantation without stimulation is associated only with the local muscle regeneration and granulation tissue formation, with no change in masticatory fibers deep to the electrode. The regenerated fibers end up as masticatory fibers. In stimulated muscles, regeneration subjacent to the electrodes is more extensive, presumably because of stimulation-induced metabolic stress. Regenerated fibers in stimulated muscles, by contrast, end up expressing slow MyHC, as previously reported for regenerated masticatory muscle innervated by the soleus nerve (Hoh and Hughes 1988; Kang and Hoh 2010). Apparent direct transformation of masticatory fibers into slow fibers occurs deep to the electrodes commencing after two to three weeks, progressively involving more fibers with prolonged stimulation, as masticatory-specific myofibrillar proteins are suppressed to varying degrees. The appearance in stimulated muscles of slow MyHC and β-tropomyosin, characteristic of jaw-slow fibers, has been confirmed by SDS-PAGE and immunoblotting. The present results support the view (Hoh and Hughes 1988; Kang and Hoh 2010) that jaw-slow fibers in regenerated masticatory muscle innervated by the soleus nerve are induced by chronic low-frequency stimulation mediated by the soleus nerve. They also provide evidence that such stimuli can directly transform masticatory fibers into jaw-slow fibers.
Our results using a muscle of the jaw allotype resemble those of chronic low-frequency electrical stimulation of limb fast muscle (Pette 1984) but show distinct allotype-specific features. The transformation involves suppression of masticatory-specific myofibrillar proteins, and the transformed fibers share immunohistochemical characteristics of normal jaw-slow fibers in reacting with MAb 1A10, which binds fetal MyHCs but does not bind limb-slow myosin. This difference in immunoreactivity between jaw and limb-slow myosins is thought to arise from their different myosin light chain compositions (Hoh et al. 2007), suggesting that transformed jaw-slow fibers express embryonic light chain-1 and masticatory light chain-2 rather than limb-slow light chain isoforms. β-tropomyosin, normally absent in masticatory fibers, is induced in transformed jaw-slow fibers.
Only a limited number of myofibrillar proteins have been examined in this study. Earlier studies on limb muscles showed that chronic low-frequency stimulation induced qualitative and quantitative changes in a wide spectrum of proteins, including enzymes of energy metabolism, Ca2+-handling proteins, and myofibrillar proteins (Pette 1984). Recent proteomic analyses of stimulated limb muscle have identified changes in 41 proteins, including those involved in ion homeostasis, capillarization, and stress response (Donoghue et al. 2005; Donoghue et al. 2007). It is likely that proteomic analysis of stimulated cat masticatory fibers will also reveal changes in expression of proteins in these functional categories.
Masticatory-to-Slow Fiber Transformation via Muscle Regeneration
Masticatory fibers are not normally subject to the metabolic demands of continuous activity. The stress induced by the sudden onslaught of continuous stimulation at 10 Hz is likely to induce some muscle cell death, followed by regeneration from satellite cells. We observed regeneration principally subjacent to the electrodes, where stimulus strength is most intense. These regenerating fibers initially express fetal, masticatory, and slow MyHCs, but fetal and masticatory MyHCs are progressively suppressed, leaving only slow MyHC by 84 days. These changes under the influence of chronic stimulation are very similar to those in regenerating masticatory muscle innervated by the soleus nerve (Kang and Hoh 2010). Muscle regeneration has also been reported to contribute to the appearance of slow fibers in rabbit fast muscle chronically stimulated via its nerve (Maier et al. 1986).
Masticatory-to-Slow Fiber–Type Transformation Deep to the Stimulating Electrodes
Deep to the stimulating electrodes, responding muscle fibers are likely to have only been intermittently activated and may thus escape cell death. These fibers may also be protected from damage by the opening of K-ATP channels due to the stimulation-induced depletion of intracellular ATP, leading to membrane hyperpolarization and reduced excitability (Flagg et al. 2010). We observe two immunochemically distinct populations of transforming fibers here. The first population to transform appears on day 15. These fibers tend to be of small diameter and dispersed in distribution, initially co-expressing slow and fetal MyHCs, while m-MyHC, m-MBP-C, and m-Tm are simultaneously suppressed. Fetal MyHC expression becomes weak by 42 days and absent by 84 days. In rabbit fast muscle subject to chronic low-frequency stimulation, transient expression of fetal MyHCs has also been reported (Maier et al. 1988). The second population of fibers to transform emerges around 21 days. Initially, these fibers express slow MyHC weakly and do not suppress masticatory-specific myofibrillar proteins. These fibers are larger and do not express fetal MyHCs at any stage. They remain incompletely transformed by 84 days, expressing slow MyHC strongly, suppressing m-MBP-C completely, suppressing m-Tm to a lesser extent, but persist in expressing m-MyHC.
Possible Cellular Basis for the Two Populations of Deep Fibers Undergoing Transformation
Two possible cellular mechanisms may be considered for generating the heterogeneous response of the deep masticatory fibers to chronic stimulation. First, diversity may arise from how the muscle fibers are activated. Some deep fibers are likely to have been stimulated directly, while others are stimulated via intramuscular nerve fibers. The dispersed first population of fibers may belong to motor units whose nerve fibers are stimulated subjacent to the electrodes. Perhaps a greater consistency of activation via the nerve may have led to rapid focal degeneration and regeneration of the dispersed fibers of the motor units, resulting in fibers of small size that transiently express fetal MyHCs. Directly stimulated deep muscle fibers may have been spared of degeneration and regeneration by being intermittently activated and undergo direct, incomplete masticatory-to-slow transformation without transiently expressing fetal MyHCs.
Another possible cellular basis for generating the heterogeneous response to chronic stimulation may be the diverse developmental origins of masticatory fibers from myogenic cells. Primary and secondary myotubes of somitic muscles are known to derive from distinct lineages of myoblasts (Dunglison et al. 1999; Pin et al. 2002) and may mature into phenotypically identical fast 2b or 2x fibers after going through different developmental progressions of MyHC expression (Narusawa et al. 1987; Hoh et al. 1988b; Zhang and McLennan 1998). It has been postulated that phenotypically identical fibers with different developmental histories may respond differently to fiber type–changing stimuli (Pin et al. 2002; Zhong et al. 2010). Hypothyroidism shifts fast 2b fibers of primary origin towards slow fibers, but fast 2b and 2x fibers of secondary origin only shift towards 2a fibers (Zhong et al. 2010). Chronic low-frequency stimulation acts synergistically with hypothyroidism in inducing fast-to-slow MyHC expression as well as shifting 2b and 2x fibers towards type 2a. These stimulation-induced actions are opposed by thyroid hormone (Kirschbaum et al. 1990), and stimulation probably also differentially targets fibers of different developmental origins.
These considerations on developmental histories of fibers of the limb allotype and their responses to various stimuli suggest that parallel mechanisms may operate to regulate fibers of the jaw allotype. The apparently homogeneous masticatory fibers in the posterior temporalis also arise from primary and secondary myotubes, which also differ in their developmental progressions of MyHC expression (Hoh et al. 1988a). While the effects of thyroid hormone manipulations on cat masticatory muscles are unknown, a parallel mechanism here would have hypothyroidism inducing slow MyHC expression in masticatory fibers of primary origin, but not masticatory fibers of secondary origin. Thus, the first population of transformed fibers may be derived from primary myotubes, while the second population may be from secondary myotubes. This view is supported by the dispersed topography of the first population of transformed fibers, which resembles that of primary myotubes in developing temporalis muscle (Hoh et al. 1988a).
Possible Molecular Pathways that Transform Masticatory Fibers into Jaw-Slow Fibers
Two signal transduction mechanisms initiated by the sustained rise of intracellular Ca2+ during chronic low-frequency muscle stimulation have been proposed for activity-dependent fast-to-slow fiber–type transformation in limb muscles. The first involves activation of the Ca-calmodulin–dependent phosphatase calcineurin. Calcineurin then dephosphorylates the nuclear factor of activated T-cells (NFAT), which induces expression of slow muscle genes (Chin et al. 1998; McCullagh et al. 2004) and suppression of fast muscle genes (Rana et al. 2008). The second mechanism involves activation of intranuclear Ca-calmodulin kinase, which phosphorylates class II histone deacetylases (HDAC) in the nucleus, allowing HDAC to exit from the nucleus, thereby removing the HDAC repression of nuclear factor MEF2 activation of slow fiber–type gene expression (Liu et al. 2005; Mu et al. 2007). A further mechanism that enhances MEF2 activation may be the removal of HDAC by ubiquitination and its subsequent degradation (Potthoff et al. 2007).
We propose that in muscles of the jaw allotype, the same signaling pathways are used to transform masticatory fibers into jaw-slow fibers. Whether these signaling pathways are actually involved in transforming masticatory fibers following chronic low-frequency stimulation, and how different genes are targeted in muscles of different allotype, and fibers with different developmental histories, are interesting questions for further investigation.
Footnotes
Acknowledgements
The authors thank Dr. A.W. Everett for MAb 1A10, Dr. R.B. Fitzsimons for MAb NOQ-7-5-4D, Dr. J.J.-C. Lin for MAb CH1, M. Oetojo and H. Shoemaker for the design and construction of the stimulator, and E. Hoh for assistance with the figures.
The author(s) declared no potential conflicts of interest with respect to the authorship and publication of this article.
This work was supported by grants from the Muscular Dystrophy Association of the United States of America and the National Health and Medical Research Council of Australia.
