Abstract
Overexpression of survivin has been associated with gemcitabine resistance in pancreatic adenocarcinoma, but previous studies have shown conflicting results. This study aims to determine its prognostic value in resected pancreatic adenocarcinoma with or without adjuvant therapy and its predictive value in adjuvant gemcitabine benefit in patients with resected pancreatic adenocarcinoma. This study included 118 patients who underwent pancreaticoduodectomy from 1999 to 2007, with no neoadjuvant chemoradiation. Forty-five patients received adjuvant gemcitabine. Survivin expression was assessed immunohistochemically and was graded as low (≤10% positive cells) and high (>10% positive cells) by recursive partitioning analysis. Prognostic factors, including tumor size, number of positive lymph nodes, perineural invasion, and stage, were identified for overall survival (OS) and progression-free survival (PFS) using Cox proportional hazards models. Multivariable analysis of the entire cohort revealed that both high survivin expression and perineural invasion predict significantly shorter OS (hazard ratio [HR] 2.0, p=0.01; HR 1.9, p=0.01, respectively) and shorter PFS (HR 1.9, p=0.04; HR 3.1, p=0.0006, respectively). Expression of survivin predicts neither OS nor PFS in patients treated with adjuvant gemcitabine. In summary, high expression of survivin is associated with shorter OS and PFS in patients with resected pancreatic adenocarcinoma after adjusting for other histopathological factors. However, survivin has no predictive value of adjuvant gemcitabine benefit.
Survivin, a member of the apoptosis inhibitor family, plays important roles in multiple cellular processes; it inhibits caspase-dependent or independent apoptosis, stabilizes microtubules during mitosis, and regulates cell cycle progression (Altieri 2003, 2008). Survivin is undetectable in most adult cells, although its presence is ubiquitous during fetal development. However, survivin has been shown to be overexpressed in many human cancers, which is independent of the mitotic index (Ambrosini et al. 1997; Altieri 2003). Clinically, survivin overexpression has been associated with more aggressive tumor subtypes and worse prognosis in neuroblastoma (Adida et al. 1998), non–small cell lung cancer (Monzo et al. 1999), glioblastoma (Chakravarti et al. 2002), and lymphoma (Schlette et al. 2004). Continuing investigation has been focusing on the development of a small-molecule inhibitor of survivin (Giaccone et al. 2009) and vaccine-based immunotherapy (Wobser et al. 2006) targeting survivin selectively in tumor cells.
Survivin has been studied in pancreatic ductal adenocarcinoma, a malignancy associated with a median overall survival of only approximately 18 months in patients with resectable tumor (Hidalgo 2010; Vincent et al. 2011). Preclinical studies had shown that pancreatic cancer cell lines with high survivin expression have an increased proliferation index and decreased radiosensitivity (Asanuma et al. 2000). Transfection with siRNA against survivin enhances pancreatic cancer cell sensitivity to gemcitabine (Liu et al. 2009). These findings have important clinical implications because gemcitabine-based chemotherapy and chemoradiation therapy are the mainstay of adjuvant treatment for resectable pancreatic adenocarcinoma (Neoptolemos et al. 2004; Oettle et al. 2007; Regine et al. 2008).
Previous small studies have shown conflicting results on the prognostic role of survivin in pancreatic adenocarcinoma (Ansari et al. 2011). Kami et al. (2004) studied 47 patients with pancreatic adenocarcinoma at all stages. Patients with positive survivin expression had a significantly decreased overall survival (OS) independent of radiation therapy. Tonini et al. (2005) confirmed this finding by demonstrating an association between cytoplasmic survivin expression and worse OS in 67 patients with resected pancreatic adenocarcinoma. However, these conclusions were challenged by four other studies (Sarela et al. 2002; Lee et al. 2005; Sagol et al. 2005; Sun et al. 2007) with similar sample size and proportion of tumors with positive survivin expression that did not show a significant association between survivin and OS.
To elucidate the prognostic and predictive roles of survivin, we evaluated 118 patients with resectable pancreatic adenocarcinoma. We aimed to determine whether survivin expression level measured by immunohistochemistry is prognostic in patients with resectable pancreatic adenocarcinoma and predictive of adjuvant gemcitabine benefit.
Materials and Methods
We retrospectively reviewed the medical records of 118 patients who underwent surgical resection for pancreatic adenocarcinoma at the Cleveland Clinic from October 1999 to December 2007. All surgical pathology reports were reviewed. In addition, all tumors were histologically reviewed and staged according to the seventh edition of the American Joint Committee on Cancer tumor, node, metastasis (TNM) classification system. Cases with gross evidence of intraductal mucinous papillary neoplasm (IMPN) in the main and branches of the pancreatic duct were excluded. In addition, cases with invasive adenocarcinoma arising from IPMN upon microscopic examination were also excluded. Patients with pancreatic ductal adenocarcinoma and a pathological stage of T1-3,N0-1,M0 were included. Patients with unresectable or metastatic disease, R2 resection, ampullary carcinoma, common bile duct carcinoma, and indolent pancreatic tumors such as mucinous cystadenocarcinoma, mucinous cystadenoma, and islet cell tumor were excluded. The study was approved by the institutional review board at the Cleveland Clinic.
Tissue samples from all 118 cases were either formalin fixed (105 patients, 89.0%) or Hollande’s fixed (13 patients, 11.0%) and paraffin embedded. The tumor-containing hematoxylin and eosin–stained slides were reviewed. Four tissue microarrays of tumors (duplicate, 1.5-mm cores from the area with the highest density of tumor cells) were constructed and used for the determination of survivin by immunohistochemistry (IHC). IHC staining was performed on a Discovery XT automated stainer (Ventana Medical Systems, Tucson, AZ) with diaminobenzadine (DAB) as the chromogen. Deparaffinized sections at 4 µm thickness were labeled with survivin antibody (Stefano et al. 2011) (mouse monoclonal antibody, clone 1C4A12, at 1:100 dilution at 37C for 1 hr; Zymed, San Francisco, CA) following antigen retrieval in Tris/borate/EDTA buffer, pH 8.0 to 8.5, for 8 min at 95C, 12 min at 100C, and then 8 min at room temperature. A secondary antibody (OmniMap anti-Mouse HRP; Ventana Medical Systems) was applied for 12 min at room temperature. Slides were then counterstained with Hematoxylin II (Ventana Medical Systems), post-counterstained with Bluing Reagent (Ventana Medical Systems), and visualized by light microscopy. Survivin immunoreactivity was assessed by recording the percentage of positive tumor cells with nuclear staining semiquantitatively, as nuclear immunoreactivity of survivin was previously reported in hepatocellular carcinoma cells when this antibody was used (Stefano et al. 2011). Appropriate negative controls (no primary antibody) were stained in parallel with each set of tumors studied. At least 400 tumor cells (range, 400–1200) were counted for the evaluation of survivin expression for each tumor.
Primarily descriptive statistical analysis was used. Categorical data were summarized as frequency counts and percentages. Continuous measures were summarized as means, standard deviations (SD), medians, and ranges. Sample size calculation and power estimation were not performed. OS was measured from the time of tumor resection to death. Progression-free survival (PFS) was measured from the time of tumor resection to disease progression or death. Time-to-event data were plotted using the Kaplan-Meier method with surviving patients censored at the date of last follow-up. Log-rank tests were used to evaluate different groups of patients. Cox proportional hazards model was used for multivariable analyses of potential prognostic indicators in a stepwise fashion. The entry criterion was p<0.1. The retention criterion was p<0.05. Hazard ratio (HR), 95% confidence interval (CI) for the HR, and corresponding p value were used to present Cox proportional hazards regression results. Recursive partitioning analyses (RPAs) were used to dichotomize survivin expression into a high-expression group and a low-expression group, as previously described (Kim et al. 2011; Xie et al. 2012). They were carried forward into the univariate and multivariable analyses. All the statistical analyses were performed using SAS 9.2 (SAS Institute; Cary, NC).
Results
Prognostic Value of Survivin in the Entire Cohort of Patients with Resectable Pancreatic Adenocarcinoma
We found 118 cases of pancreatic ductal adenocarcinoma with tumor resection, from October 1999 to December 2007. As shown in Table 1, 68 patients (57.6%) were male. The median age at the time of resection was 64.5 years (range, 35–93 years). The surgical procedures included the Whipple procedure in 95 patients (80.5%), distal pancreatectomy in 21 (17.8%), subtotal pancreatectomy in 1 (0.8%), and uncinate excision in 1 (0.8%). Six patients (5.1%) had stage IA disease, 17 (14.4%) had stage IB disease, 24 (20.3%) had stage IIA disease, and 71 (60.2%) had stage IIB disease. Thirty-eight patients (32.2%) had a positive surgical margin. The mean (SD) number of positive lymph nodes was 2.2 (3.3). Lymphovascular invasion was identified 57 patients (48.3%). Perineural invasion was identified in 77 patients (65.2%). No adjuvant treatment was given in 25 patients (21.2%), but 59 patients (50.0%) underwent adjuvant chemoradiation therapy, and 15 (12.7%) had adjuvant chemotherapy only. No patients received neoadjuvant chemoradiation. Treatment history for 15 patients (12.7%) was unknown. Forty-five patients (38.1%) received gemcitabine or gemcitabine-based regimens. The median follow-up was 13.8 months (range, 0.2–105.6 months). At the time of the last follow-up, 98 (83.0%) patients were dead.
Patient Demographics, Clinical Characteristics, and Histopathological Features (n=118)
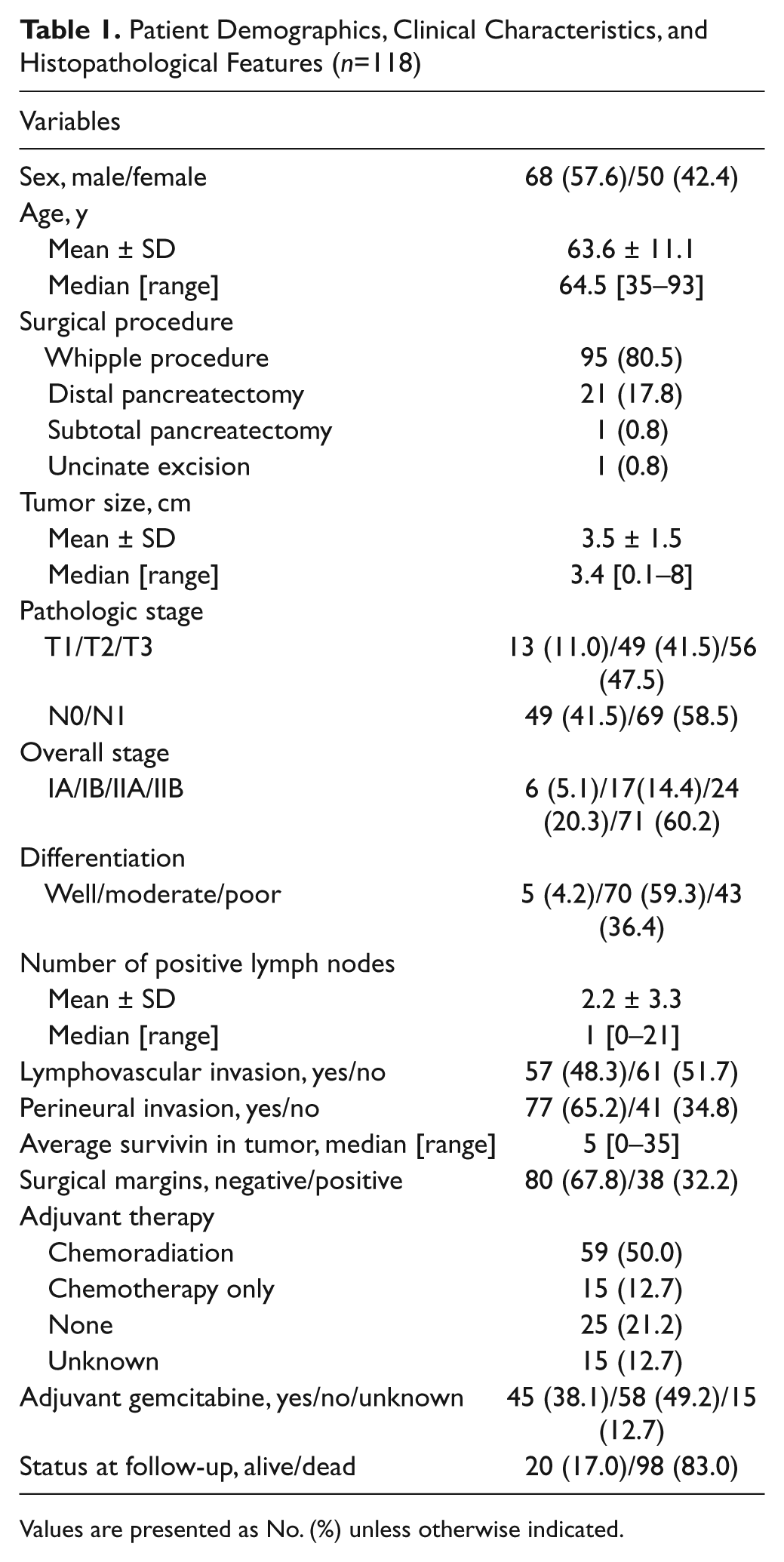
Values are presented as No. (%) unless otherwise indicated.
Examining the staining index (%) of survivin by RPA, a cutoff value of 10% was determined for OS and PFS for the entire cohort. Four tumors with a high and low staining index of survivin are illustrated in Figure 1. For OS, 95 patients were segregated into the survivin low-expression group and 23 patients into the survivin high-expression group. For PFS analysis, 65 patients were grouped into the survivin low-expression group and 16 patients into the survivin high-expression group. Univariate analysis for survival included survivin protein expression, TNM stage, tumor size, number of positive lymph nodes, overall stage, lymphovascular invasion, perineural invasion, adjuvant therapy, sex, age, tumor grade, status of surgical margins, and adjuvant gemcitabine. As shown in Table 2, tumor size, number of positive lymph nodes, overall stage IIA/I and IIB/I, perineural invasion, and N stage were associated with worse OS. Similarly, tumor size, number of positive lymph nodes, lymphovascular invasion, perineural invasion, and N stage were associated with decreased PFS. As depicted in Figure 2, for patients with high survivin expression in tumors, there was a trend to shorter OS (median OS 13.1 vs 16.1 months; p=0.11). The same conclusion was reached for PFS (median PFS 11.1 vs 15.9 months; p=0.06).
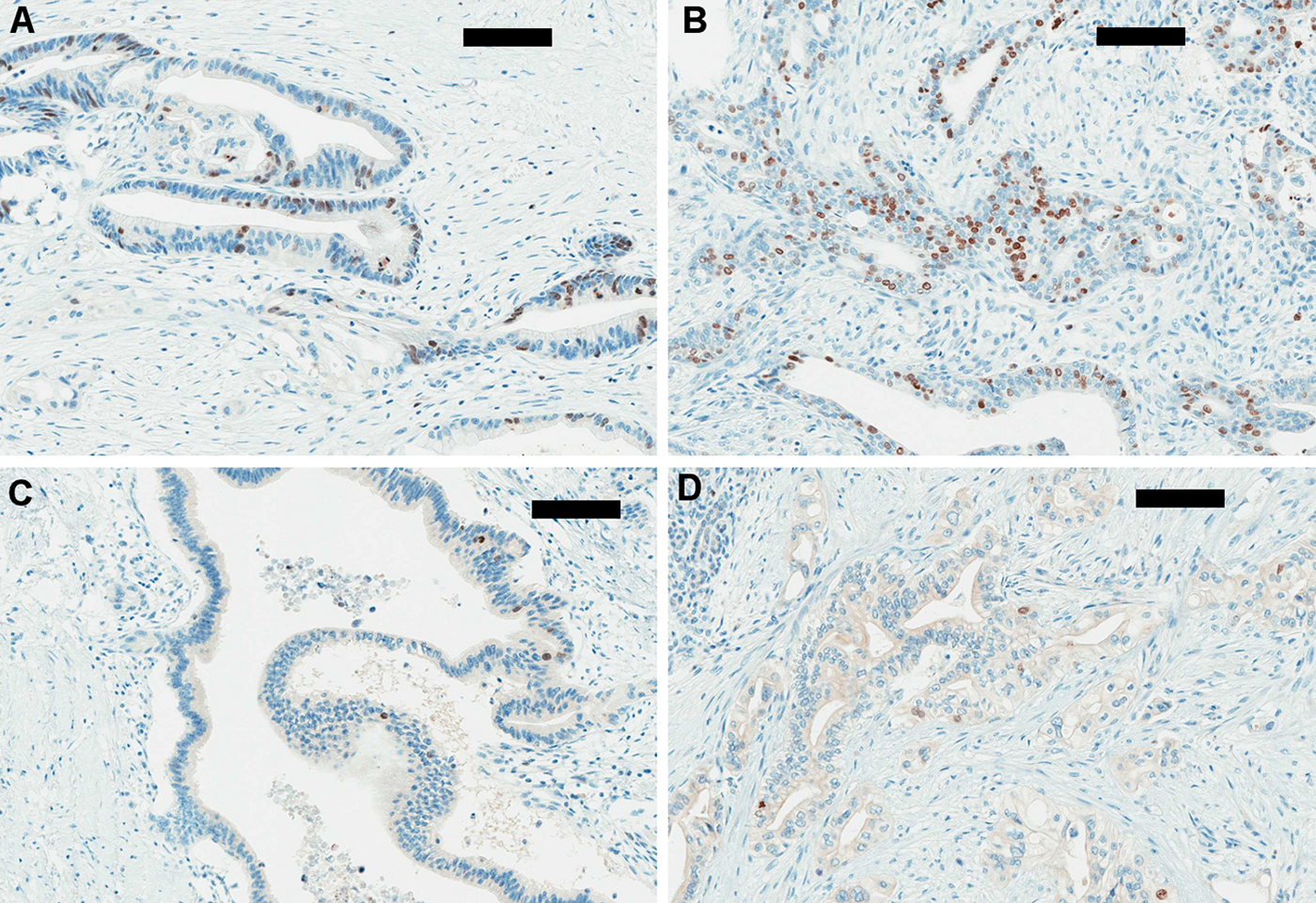
(A, B) High labeling index (>10%) of survivin in two pancreatic adenocarcinomas (immunoperoxidase stain, ×200, scale bar represents 100 µm). (C, D) Low labeling index of survivin (<10%) in two pancreatic adenocarcinomas (immunoperoxidase stain, ×200, scale bar represents 100 µm).
Univariate Analysis for Overall Survival and Progression-Free Survival in the Entire Cohort
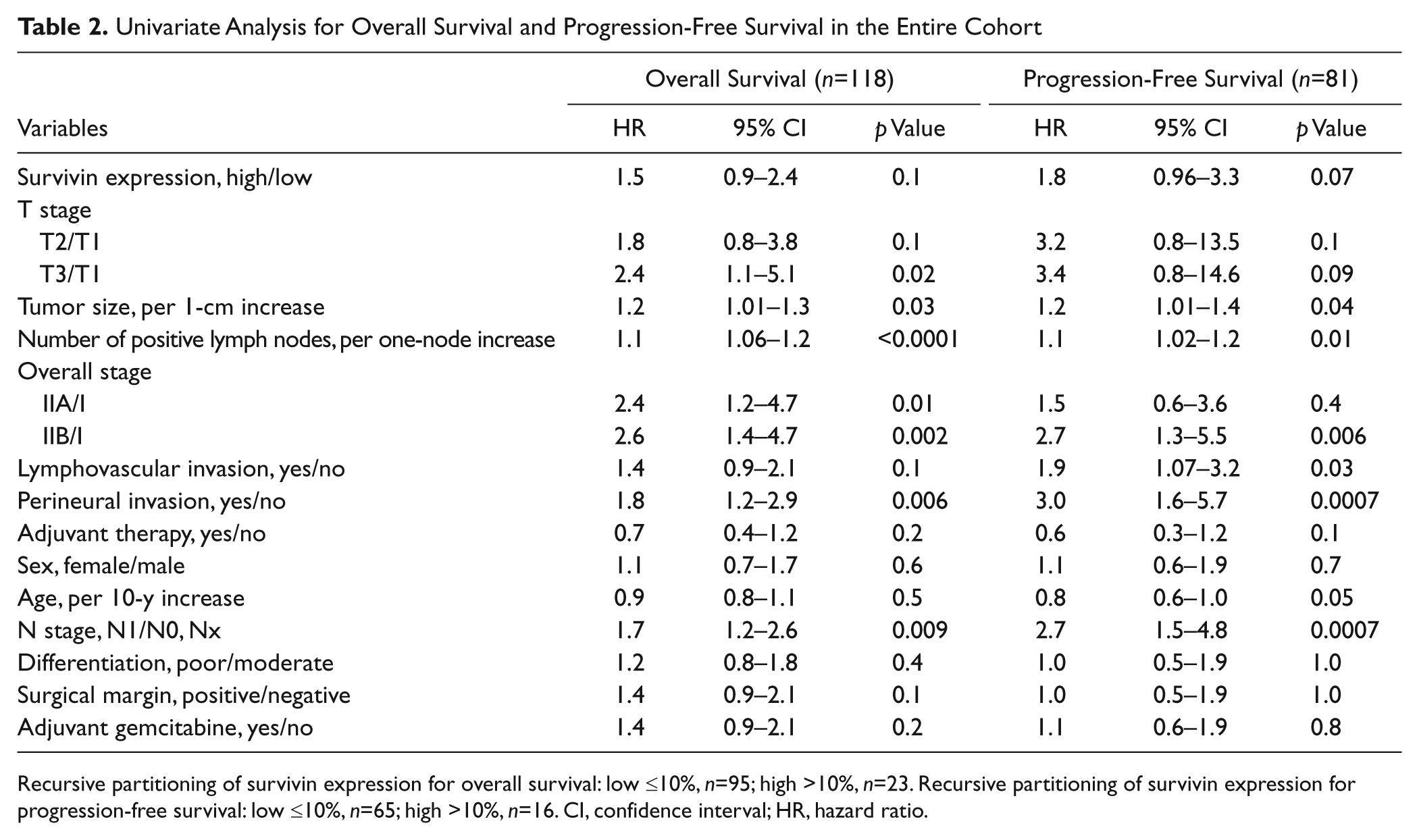
Recursive partitioning of survivin expression for overall survival: low ≤10%, n=95; high >10%, n=23. Recursive partitioning of survivin expression for progression-free survival: low ≤10%, n=65; high >10%, n=16. CI, confidence interval; HR, hazard ratio.
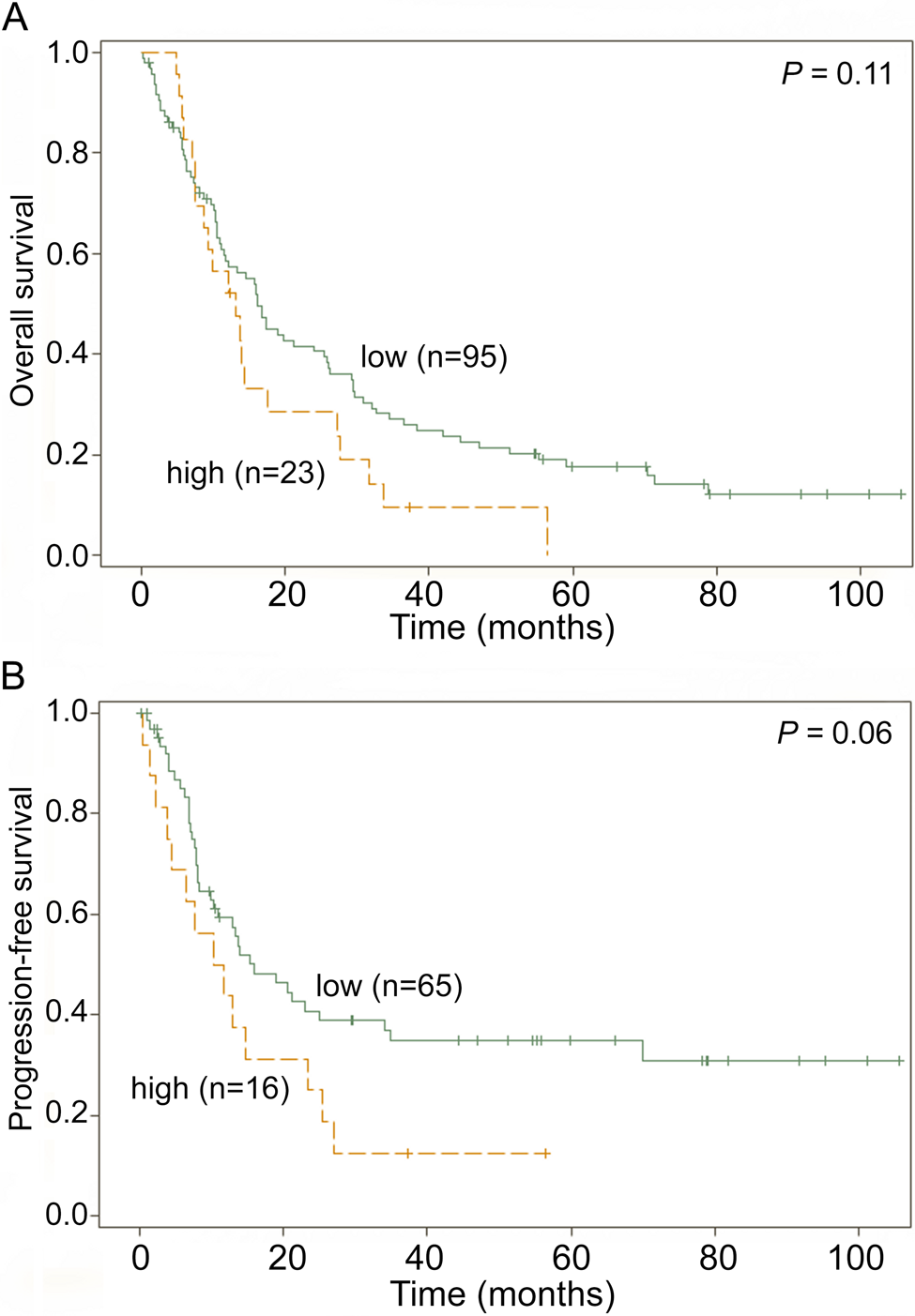
(A) Overall survival (OS) by survivin expression in patients with resectable pancreatic adenocarcinoma (survivin-low: median OS 16.1 months; survivin-high: median OS 13.1 months). (B) Progression-free survival (PFS) by survivin expression (survivin-low: median PFS 15.9 months; survivin-high: median PFS 11.1 months).
Multivariate analyses identified prognostic factors in the entire cohort for survival based on the predefined entry and retention criteria (Table 3). The presence of perineural invasion was associated with both decreased OS and decreased PFS. In addition, the number of positive lymph nodes was associated with decreased OS. After adjusting for these prognostic factors, high survivin expression was associated with significantly shorter OS and PFS in the entire cohort.
Multivariable Analysis for Overall Survival and Progression-Free Survival in the Entire Cohort
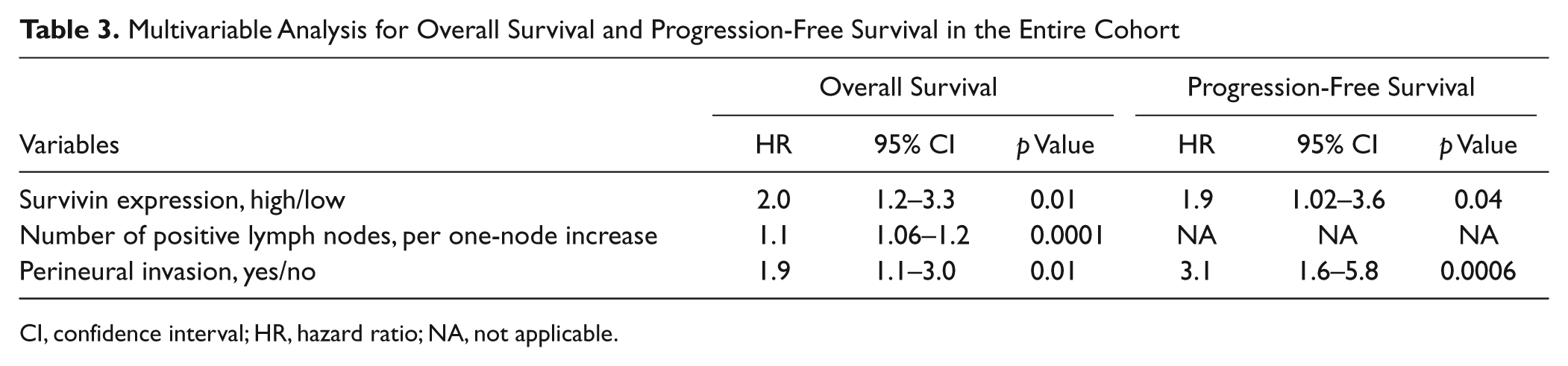
CI, confidence interval; HR, hazard ratio; NA, not applicable.
Prognostic Value of Survivin in the Subgroup of Resectable Pancreatic Adenocarcinoma Patients Treated with Adjuvant Gemcitabine
Similar analyses were performed for a subpopulation of 45 patients who received adjuvant gemcitabine treatment. RPA identified a cutoff value of 10% for the association of survivin expression with both OS and PFS. For OS, 27 patients were segregated into the survivin low-expression group and 18 into the survivin high-expression group. For PFS, 21 patients were assigned to the survivin low-expression group and 13 to the survivin high-expression group. Univariate analysis revealed that a higher number of positive lymph nodes (hazard ratio [HR], 1.2 per one-node increase; p=0.004) was associated with decreased OS. Only perineural invasion (HR, 5.7; p=0.006) was associated with decreased PFS (Table 4). Survivin expression did not predict the treatment benefit of adjuvant gemcitabine in terms of OS or PFS (Table 4). As depicted in Figure 3, patients with low survivin expression in tumors did not have a significantly longer OS than patients with high survivin expression (median OS 15.9 vs 16.8 months; p=0.28). The same conclusion was reached for PFS (median PFS 13.8 vs 14.7 months; p=0.91). Multivariable analyses shown in Table 5 were consistent with these findings. Number of positive lymph nodes and perineural invasion were predictors of shorter OS (HR, 1.2 per 1 more; p=0.004) and shorter PFS (HR, 5.7; p=0.006), respectively.
Univariate Analysis for Overall Survival and Progression-Free Survival in Patients Treated with Adjuvant Gemcitabine
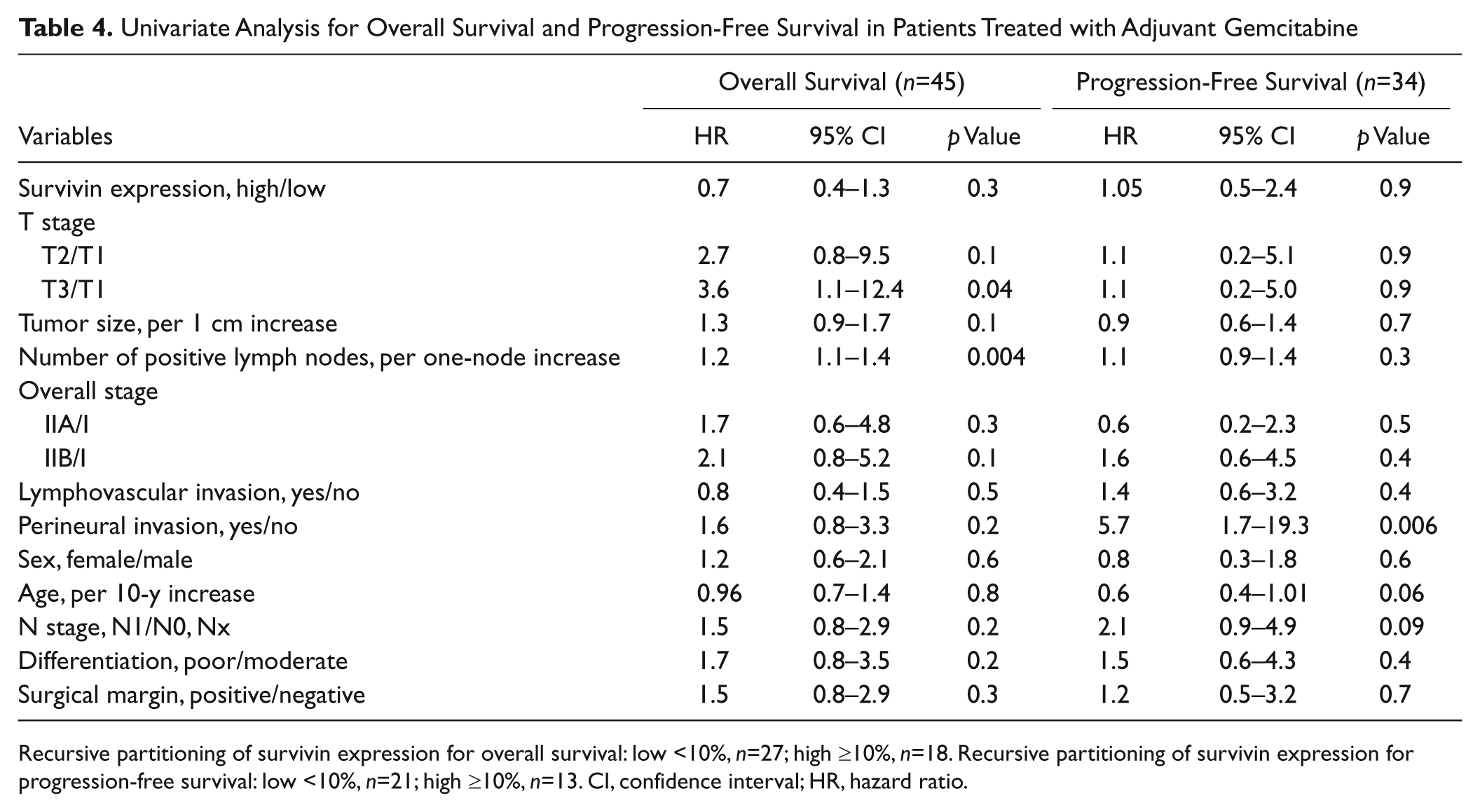
Recursive partitioning of survivin expression for overall survival: low <10%, n=27; high ≥10%, n=18. Recursive partitioning of survivin expression for progression-free survival: low <10%, n=21; high ≥10%, n=13. CI, confidence interval; HR, hazard ratio.
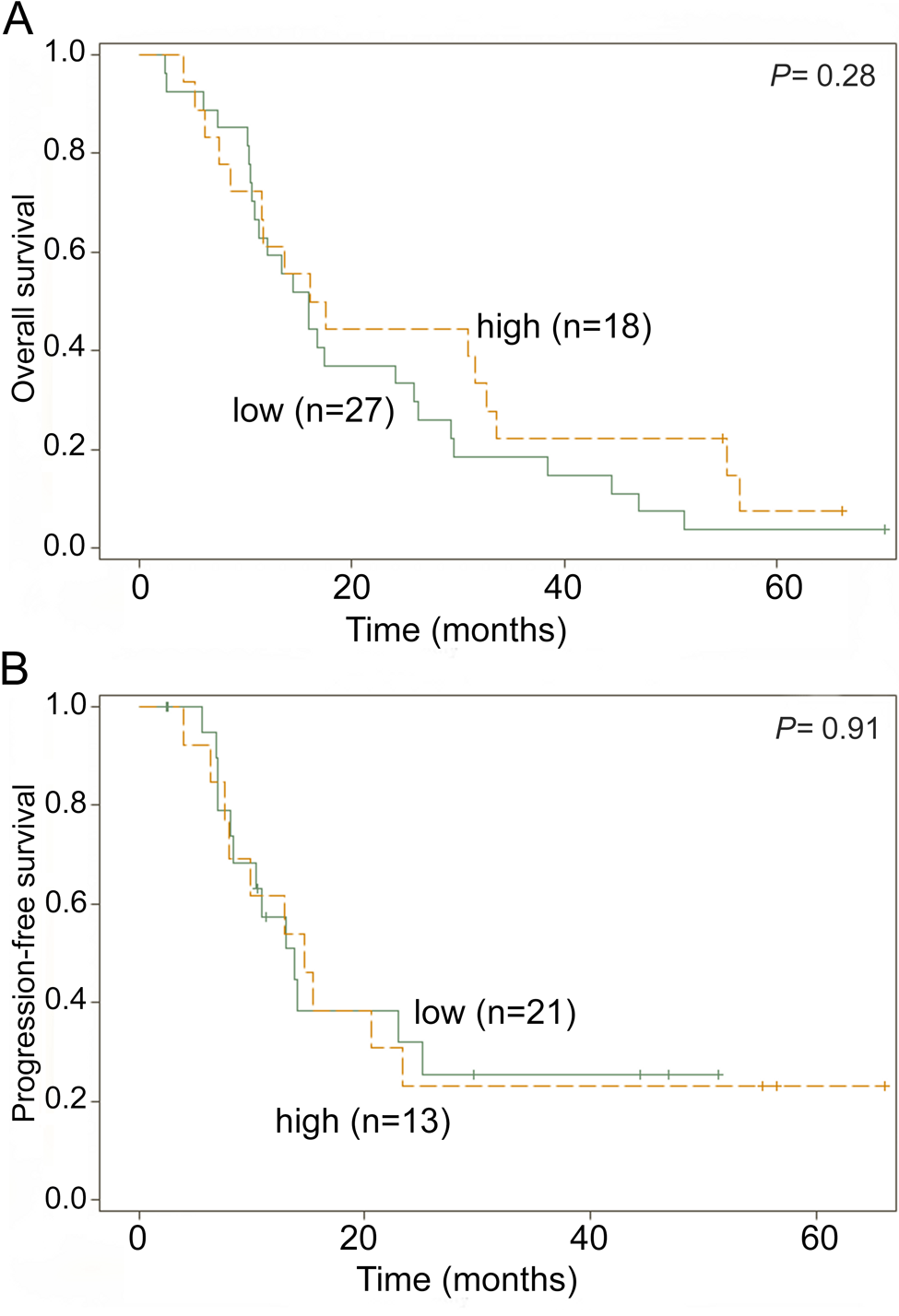
(A) Overall survival (OS) by survivin expression in patients with resectable pancreatic adenocarcinoma and treated with gemcitabine therapy (survivin-low: median OS 15.9 months; survivin-high: median OS 16.8 months). (B) Progression-free survival (PFS) by survivin expression (survivin-low: median PFS 13.8 months; survivin-high: median PFS 14.7 months).
Multivariable Analysis for Overall Survival and Progression-Free Survival in Patients Treated with Adjuvant Gemcitabine

CI, confidence interval; HR, hazard ratio; NA, not applicable.
Discussion
We presented a retrospective study on the prognostic and predictive roles of survivin in 118 patients with resectable pancreatic adenocarcinoma. To our knowledge, this study has the largest cohort among similar studies from the literature. The median age, sex, treatment regimens, and the clinical outcomes were not different from those in previous studies (Giovannetti et al. 2006; Fujita et al. 2010). Previous univariate analyses had revealed common histopathological prognostic factors for resectable pancreatic adenocarcinoma, including tumor size, stage, number of positive lymph nodes, the presence of lymphovascular invasion or perineural invasion, and the absence of adjuvant therapy (Sohn et al. 2000; Brennan et al. 2004). In this study, univariate analyses showed a trend toward shorter OS and PFS for patients with high survivin expression (although short of statistical significance). However, after adjusting for the number of positive lymph nodes and perineural invasion, there was a significant association between survivin expression and both OS and PFS. These results confirmed the findings from the Tonini et al. (2005) study and the Kami et al. (2004) study. Our findings, however, are contradictory to some other earlier studies. Sarela et al. (2002) measured survivin expression in 64 patients with resectable pancreatic adenocarcinoma. Univariate analysis from their study did not reveal a value of survivin in predicting the prognosis. Similarly, studies from three other groups (Lee et al. 2005; Sagol et al. 2005; Sun et al. 2007) evaluated pancreatic adenocarcinoma tumor samples at all stages. None of the univariate analyses from these studies demonstrated a significant association between survivin expression and survival.
We propose several possible explanations to address these discrepancies. First, in this study, we only included patients with stage I and stage II disease. No patients with metastatic disease were included. No patients included had undergone neoadjuvant therapy, which might potentially influence the expression of survivin in tumor cells (Tamm et al. 1998). These patient characteristics were closest to those in the Tonini et al. (2005) study. In contrast, a significant proportion of the patients in four of the previous studies (Kami et al. 2004; Lee et al. 2005; Sagol et al. 2005; Sun et al. 2007) had metastatic disease or had been extensively treated. The biology of the resectable pancreatic adenocarcinoma differs from that of the unresectable type, as manifested by the drastically distinct response of patients to treatments and their prognosis (Hidalgo 2010). Thereby, studies on patients with heterogeneous diseases tend to yield diluted clinical effects. In this study, we evaluated only patients with resectable pancreatic adenocarcinoma (stages I and II) to avoid this potential issue. Second, as one of the advantages of our study, the relatively large sample size allows for a thorough multivariable analysis of the relationship between survivin and clinical outcomes with the control of potential histopathological confounders. Third, survivin expression assessed by immunohistochemistry was graded into positive and negative groups using different criteria. Four previous studies (Sarela et al. 2002; Sagol et al. 2005; Tonini et al. 2005; Sun et al. 2007) generated weighted indices from both intensity and percentage of positive tumor cells. However, all four sets of weighted indices were generated using different methods, which rendered a direct comparison almost impossible. The other two previous studies (Kami et al. 2004; Lee et al. 2005) graded survivin expression into tiers based on the percentage of positive tumor cells and arbitrarily chose 10% as the cutoff. In this study, we adopted recursive partitioning analysis for the optimal cutoff. Although this type of data-mining method tends to provide thresholds that are only native to the corresponding data sets, the cutoff value identified in our study turned out to be quite close to that in the previously reported studies (Kami et al. 2004; Lee et al. 2005).
Survivin, a member of the apoptosis inhibitor family, plays important roles in multiple cellular processes; it inhibits caspase-dependent or caspase-independent apoptosis, stabilizes microtubules during mitosis, and regulates cell cycle progression (Altieri 2003, 2008), two key biological processes in cancer cells targeted by gemcitabine. Because preclinical studies demonstrated that survivin overexpression renders pancreatic cancer cell chemoresistance to gemcitabine (Liu et al. 2009), we evaluated the predictive value of survivin in adjuvant gemcitabine benefit in these patients. We did not detect any role of survivin in predicting adjuvant gemcitabine benefit for either OS or PFS in the subgroup of 45 patients who received such treatment. This suggests that survivin may not play a significant role in mediating gemcitabine resistance in resectable pancreatic adenocarcinoma. Several recent studies have shown that either human equilibrative nucleoside transporter 1 (hENT-1) proteins and/or ribonucleotide reductase subunit M1 (RRM1) are predictive of gemcitabine benefit in patients with resectable pancreatic cancer (Akita et al. 2009; Farrell et al. 2009; Xie et al. 2012), supporting that molecules directly involved in gemcitabine transport, metabolism, and targeting are more important in predicting gemcitabine benefit. However, survivin may still play a role in gemcitabine resistance in pancreatic cancer because not all pancreatic cancer patients with a low expression of RRM1 or a high expression of hENT-1 will benefit from gemcitabine therapy (Farrell et al. 2009; Xie et al. 2012). Given the current in vitro data that suppression of survivin resulted in gemcitabine sensitivity in pancreatic cancer cells (Liu et al. 2009) and the continuing investigation focusing on the development of a small-molecule inhibitor of survivin (Giaccone et al. 2009) and vaccine-based immunotherapy targeting survivin selectively in tumor cells, the potential role of survivin in gemcitabine resistance in a subgroup of pancreatic cancer warrants further study. However, testing this hypothesis requires a large cohort of pancreatic cancer patients treated with gemcitabine and a simultaneous evaluation of these markers.
As a retrospective study, it is not possible to completely eliminate all potential information bias, especially considering the fact that patient demographics, histopathological data, therapeutic details, and outcomes were collected over a time span of 8 years. Another limitation of this study is the interobserver variability as an intrinsic problem associated with the interpretation of immunostaining results. In the current study, a central review process was adopted to reduce this type of variation. With this limitation, we have considered the alternative of measuring survivin in a more quantitative manner, such as computer-aided image analysis, to have a better potential of being used in practice. However, further studies are needed to address this issue.
In summary, this study demonstrated that survivin expression level determined by immunohistochemistry on paraffin-embedded pancreatic adenocarcinoma tissue is a prognostic factor for OS and PFS in the entire cohort of patients with resectable pancreatic adenocarcinoma. However, survivin expression level does not predict adjuvant gemcitabine benefit in patients with resectable pancreatic adenocarcinoma.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
