Abstract
Apoptosis has gained central importance in the study of many biological processes, including neoplasia, neurodegenerative diseases, and development. One of the limitations of many studies is the difficulty of specifically identifying individual apoptotic cells. Of the many specific methods developed to detect apoptotic cells, most are not applicable to histological sections of archival paraffin-embedded tissues. Recently, advances in the understanding of the molecular events in apoptosis have led to the realization that caspase activation is by far the most specific indicator of this cell suicide mechanism. Several publications have reported the development of antibodies directed at neoepitopes that are generated in various substrates through the action of caspases. One of these is that present on activated caspase 3, a ubiquitously distributed caspase that is a main effector caspase of the apoptotic cascade within cells. This study demonstrates the utility of using a recently commercially available antibody to cleaved caspase 3 in archival paraffin sections, suggesting that this may be a highly specific and sensitive method generally applicable to many studies of archival material.
A
Because archival material represents a potential wealth of information that could be employed to study many disease processes, there has been considerable interest in developing methods that could specifically demonstrate apoptotic cells in paraffin sections. Through the study of the molecular events in apoptosis systems, it is now clear that cleavage of protein substrates by caspases is a pivotal cascade that is unique to apoptotic cells (Green 2000). When caspases cleave proteins, they generate novel peptide ends that could represent neoepitopes. Generation of antibodies to such neoepitopes has been reported for several caspase protein substrates, including caspases themselves (Srinivasan et al. 1998; Knaapen et al. 1999; Tanaka et al. 2000), and for other substrates, such as PARP (O'Brien et al. 2001) and cytokeratin 18 (Caulin et al. 1997; Leers et al. 1999). The use of such antibodies for immunohistochemistry could represent a specific method for detection of apoptotic cells in archival materials if the substrates are present in many cells and if the neoepitopes are accessible and preserved with routine formaldehyde fixation and paraffin embedding. With the advent of commercially available antibodies to these neoepitopes, we and others have evaluated the utility for apoptosis detection in routinely processed cells and tissues. In this article we demonstrate the utility of one of these recently available antibodies, anti-cleaved caspase 3, both in a model system in cell culture (Barrett et al. 2001) subsequently processed routinely into paraffin and in archival tissues. We conclude that this particular neoepitope is uniquely accessible and that these antibodies may represent valuable reagents for the study of apoptosis in archival tissues.
Materials and Methods
Cell Culture and Reagents
KB human carcinoma cells were obtained from American Type Culture Collection (Rockville, MD) and propagated in RPMI-1640 medium supplemented with 10% fetal calf serum, penicillin, and streptomycin at 37C in a 5% CO2/95% air atmosphere. Ricin, TNF, cycloheximide, and culture media were obtained from Sigma Chemicals (St Louis, MO). Rabbit polyclonal antibody to cleaved caspase 3 was obtained from Cell Signaling Technologies (Beverly, MA; catalog #9661). Affinity-purified goat anti-rabbit IgG labeled with rhodamine was obtained from Jackson ImmunoResearch (West Grove, PA).
Tissues
Blocks of paraffin-embedded tissues that had been formalin-fixed were obtained from the archives of PhenoPath Laboratories, corresponding to normal human tissues, including lung, liver, breast, lymph node, skin, brain, kidney, pancreas, intestine, and skeletal muscle.
Apoptosis Experiments
For long-term apoptosis experiments, KB cells in T-75 flasks were treated with either ricin (1 ng/ml) or a mixture of tumor necrosis factor (TNF; 10 ng/ml) together with cycloheximide (5 μg/ml) for 2 days. The floating cells from treated flasks were then harvested at room temperature (RT) using a Shandon Cytospin centrifuge, fixed in 3.7% formaldehyde for 15 min, and permeabilized using 0.1% Triton X-100 for 5 min. The cytospins were then incubated in rabbit anti-cleaved caspase 3 antibody (1:100 dilution of manufacturer's stock) in 1% bovine serum albumin with PBS for 30 min. After a PBS wash, the cells were incubated with 25 μg/ml affinity-purified goat anti-rabbit IgG conjugated with rhodamine for 30 min. After washing and postfixation in formaldehyde, the preparations were incubated with 0.1 μg/ml DAPI in methanol and mounted under a coverslip in glycerol.
For shorter-term apoptosis experiments, two T-75 flasks of KB cells were incubated with 1 μg/ml ricin for 7 hr, and the floating cells were removed by shaking and pelleted by centrifugation at 5000 × g in 1% BSA-PBS. Similarly, untreated control cells were removed from flasks by trypsinization and pelleted in 1% BSA-PBS. Small aliquots of these preparations before centrifugation were also used to prepare cytopreps that were fixed in acetone and stained with hematoxylin. The pellets were fixed by slowly dripping 3.7% formaldehyde in PBS on top of the solid pellets and incubating overnight at RT. The pellets were then gently removed from the centrifuge tubes, placed in filter paper and into a cassette, and routinely processed for paraffin embedding.
Immunohistochemistry using the rabbit anti-cleaved caspase 3 antibody was performed on deparaffinized tissue sections using a routine avidin-biotin-immunoperoxidase technique [Vectastain Elite avidin-biotin-immunoperoxidase kit reagents (Vector Labs, Burlingame, CA)]. Before incubation with the primary antibody (1:200 dilution as supplied by the manufacturer), tissue sections were subjected to heat-induced epitope retrieval by incubation in a pH 8.0 0.01 M EDTA solution for 10 min in a vegetable steamer, followed by 20-min cool-down and treatment with 3% hydrogen peroxide before antibody application.
Microscopy
Fluorescence images were prepared using a Zeiss Axioplan 2 microscope equipped with phase-contrast optics and with rhodamine and UV channel filters. Images were recorded using either a Dage 300 monochrome CCD camera or a Zeiss Axiocam CCD camera, and composite figures were prepared using Adobe Photoshop.
Results
A polyclonal affinity-purified rabbit antibody to the neoepitope peptide at the end of cleaved caspase 3 (17-20-kD fragments) was obtained from Cell Signaling Technologies (catalog #9661). This antibody reacts with this neoepitope in mouse, rat, and human caspase 3. To evaluate its utility in human cells, we employed a model apoptosis sytem in KB human carcinoma cells that we have previously described, using either TNF or ricin to induce apoptosis. Cells in culture exposed to either of these agents for 2 days were then fixed in formaldehyde, permeabilized with Triton X-100, and reacted with rabbit anti-cleaved caspase 3, followed by anti-rabbit IgG conjugated to rhodamine. These cells were further incubated with DAPI to visualize nuclear DNA. Figure 1 demonstrates the appearance of apoptotic and normal cells using this method. Cells with normal nuclear morphology showed no detectable reaction with this antibody, but cells with apoptotic nuclear and surface morphology showed various amounts of reactivity in a diffuse or punctate cytoplasmic pattern. The strength of this signal suggested that there was sufficient caspase 3 in such cells to allow easy detection of its catalysis during apoptosis.
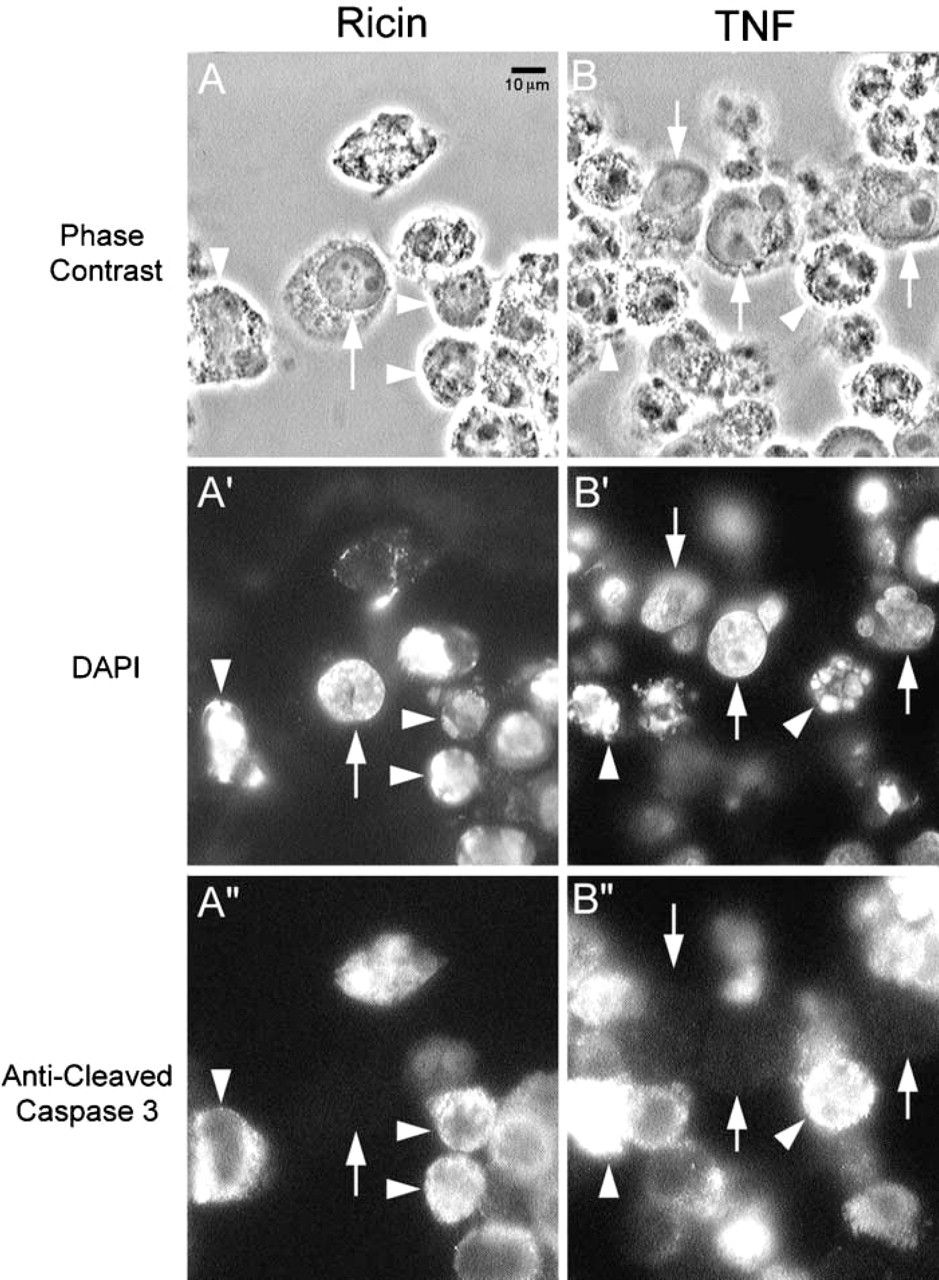
Detection of cleaved caspase 3 in apoptotic cells in culture. KB human carcinoma cells were incubated with either ricin (1 ng/ml) (
To determine if this signal would be detectable after fixation and routine processing into paraffin, cell pellets were prepared from such cells treated with 1 μg/ml ricin for 7 hr in culture. These pellets were then fixed in formaldehyde, dehydrated through ethanol and xylene, and embedded in paraffin by routine methods. Sections from these blocks were then subjected to immunohistochemistry using anti-cleaved caspase 3 and labeling with anti-rabbit IgG conjugated to horseradish peroxidase, followed by development using diaminobenzidine. Figure 2 shows images from these sections, which clearly display a strong signal in the cytoplasm of such apoptotic cells and no such reaction in cells with normal morphology. Therefore, this antibody could clearly specifically distinguish cells in apoptosis from normal cells.
To further evaluate the utility of this approach, we employed a variety of archival blocks of normal human tissues. Most tissues showed no significant immunostaining, although prominent immunostaining was noted corresponding to the apoptotic cells present within the germinal centers of normal lymph nodes. In many tissues, cytoplasmic signal was also noted in polymorphonuclear leukocytes. Apoptosis of neutrophils has been previously described (reviewed in Webb et al. 2000), and it is possible that the staining observed represents specific detection of neutrophil apoptosis. Representative tissues immunostained with antibodies to cleaved caspase 3 are shown in Figure 3.
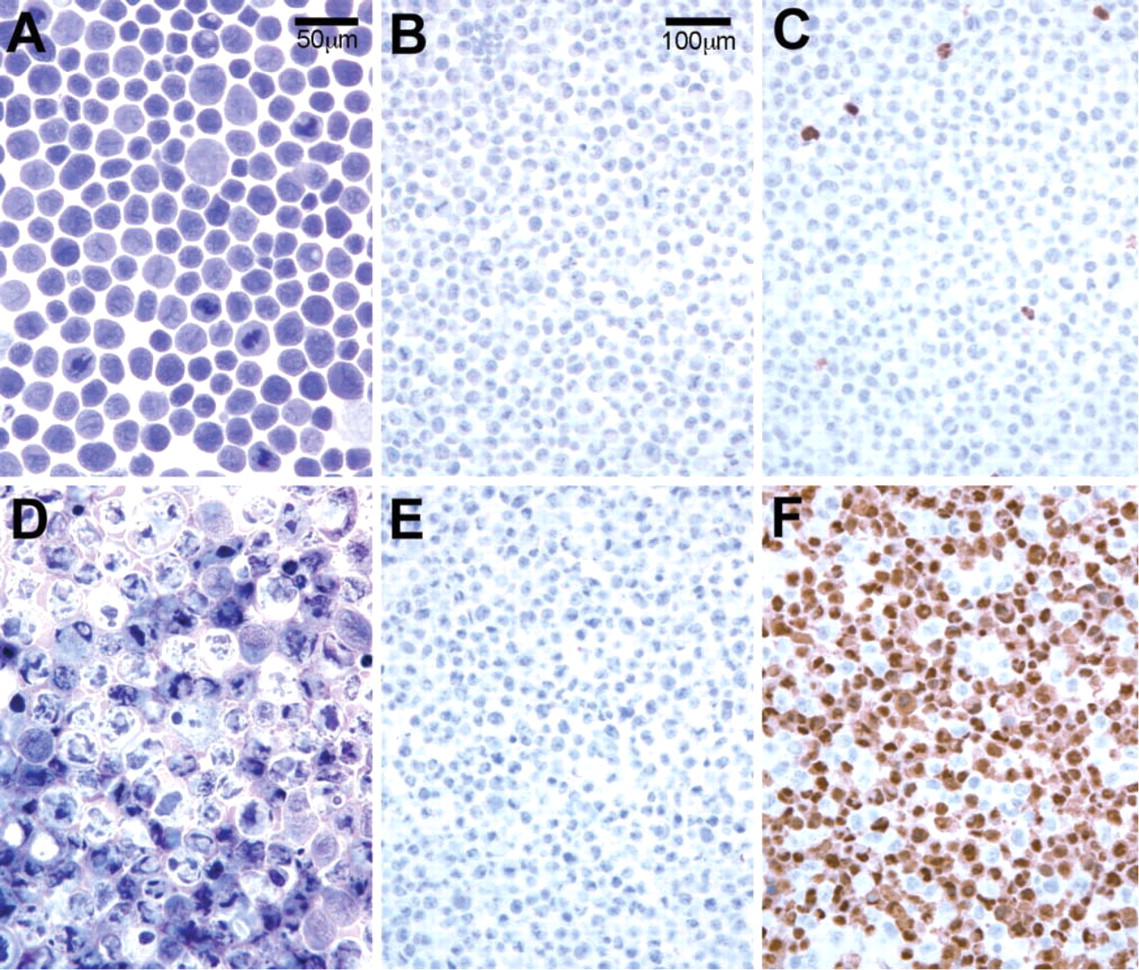
Detection of cleaved caspase 3 in apoptotic cells in paraffin sections. KB cells either untreated (
Discussion
We and others have previously presented reviews of the various methods available for detection of apoptotic cells (Collins et al. 1997; Sanders 1997; Huppertz et al. 1999; Willingham 1999; Barrett et al. 2001). Many of these methods are specific for apoptosis but are applicable only to isolated cells, such as in cell culture. Of the methods available for archival paraffin sections, TUNEL and ISNT detection of DNA ends has been extensively employed (Negoescu et al. 1996). However, positive TUNEL signals are found in necrotic cells and in some cells in which DNA fragmentation is later repairable and not indicative of apoptotic cell death (deBoer et al. 2000).
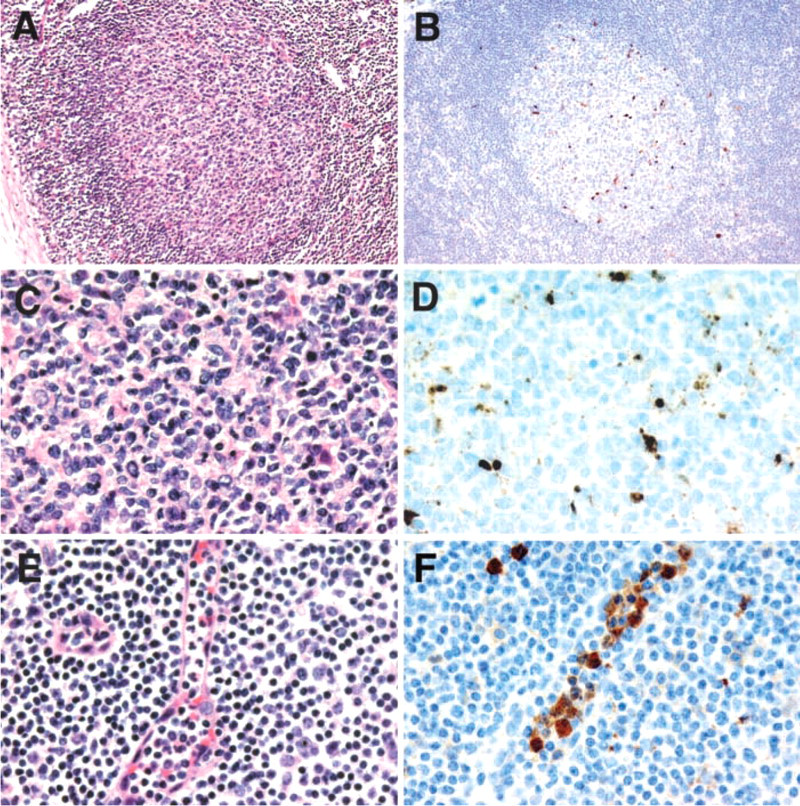
Localization of cleaved caspase 3 in deparaffinized sections of formalin-fixed tissues. Sections of germinal centers of normal lymph node seen at low (
It is now clear that caspase activation (cleavage of procaspase to active caspase) is a hallmark of almost all apoptotic systems (Green 2000). Caspase 3, for example, is a central effector caspase in many cells and mediates the cleavage of itself, other downstream caspases, and other caspase substrates. In normal cells, caspase 3 should exist as a procaspase in which the potential cleavage site is intact. Once cleaved through the activation of the apoptotic cascade, the peptide end of this active caspase represents a novel epitope not present in normal cells. Therefore, the detection of this novel epitope should be a unique and sensitive indicator of apoptosis. As described in the Introduction, several groups have prepared antibodies against novel neoepitopes generated through caspase action, including epitopes in caspases, cleaved PARP, cleaved cytokeratin 18, and other proteins. Of these new antibodies, some have become commercially available, such as the antibody employed in this study.
Not all such antibodies may work well in archival material because of the nature of the peptide or the levels of the specific caspase or other substrate present in cells. The neoepitope of cleaved caspase 3 should, however, be fairly ubiquitous and of sufficient levels to be useful in most cell types. The crossreactivity of this antibody with this neoepitope in major mammalian species used clinically and in experimental studies makes this antibody very useful. Evaluation of this and other caspase cleavage epitopes should provide a generally useful tool to identify apoptotic cells in archival material, expanding the range of experimental and disease settings in which apoptosis can be accurately evaluated.
