Abstract
Manserin is a 40–amino acid neuropeptide derived from rat brain. Manserin has been shown to distribute in the neuroendocrine system, such as the pituitary and adrenal glands, but it has been little studied in other organs. In this study, the authors examined localization of manserin in the inner ear of the adult Wistar rat using immunohistochemical analyses. Manserin immunoreactivity was detected in the neuronal terminals of the organ of Corti and type II spiral ganglion cells. In addition to being identified in the auditory system, manserin was detected at the synapses of the vestibular system, such as saccule, utricle, and semicircular canal. These results suggest that inner ear manserin may be involved in the function of peripheral auditory and vestibular systems.
Keywords
The inner ear is sensory organ comprising the auditory and vestibular systems. The auditory system is the sensory system for the sense of hearing. Sound is detected by the sensory hair cells located within the organ of Corti, in the cochlea of the inner ear, producing neural impulses that are transmitted first to the spiral ganglion, where the synapses are formed with the hair cells, and then to the cochlear nucleus, in the brainstem (for reviews, see Raphael and Altschuler 2003; Patuzzi and Robertson 1988). The vestibular system is composed of otolith organs and semicircular canals. The saccule and utricle are the two otolith organs. A macula is a sheet of sensory hair cells located inside the utricle and saccule, which allows detection of changes in linear acceleration and gravitational forces. The three semicircular canals are perpendicular to one another, and one end of each canal has an ampulla, containing hair cells for perception of rotation. Similar to the auditory system, the hair cells in macula act as the sensory receptors in the vestibular system. Input signals from the hair cells in macula project to the vestibular nucleus of the brainstem via the vestibular ganglion (for reviews, see Minor 1998; Schubert and Minor 2004; Rutka 2004; Hain and Helminski 2007).
Similar to the processing of the central nervous system, the neural transmission of the auditory and vestibular system in the inner ear is mediated in part by neurotransmitters (Raphael and Altschuler 2003; Meza 2008). Neuropeptides, which act as neurotransmitters, might be attractive targets for drug discovery (Hökfelt et al. 2003). However, the role of neuropeptides in the inner ear remains unclear.
Manserin is a recently identified 40–amino acid peptide derived from precursor protein secretogranin II (Yajima et al. 2004). Manserin is distributed in the neuroendocrine system in the brain, such as hypothalamus and median eminence, as well as in the duodenum (Yajima et al. 2008) and pancreas (Tano et al. 2010). Several peptides have also been reported to be cleaved from secretogranin II, such as secretoneurin and EM66 (Anouar et al. 1998; Marksteiner et al. 1993). Information of these peptides being localized in hypothalamic nuclei and pituitary and adrenal glands led us to speculate that these peptides may function as a neurotransmitter in the endocrine and neuronal system, although their receptors have not been identified yet. We recently reported that manserin’s expression in the adrenal is stress dependent (Kamada et al. 2010), indicating that manserin might be involved in stress response. There are many stress-related diseases in modern society. It has been claimed that inner ear diseases such as sudden deafness and Ménière disease may be caused by stress (Horner 2003); however, the mechanisms behind these associations are not well understood.
In this study, we investigated the neuropeptide manserin with the aim of identifying the function of neuropeptides in the inner ear. This study is the first to identify the localization of manserin in the inner ear.
Materials and Methods
Animals and Tissue Preparation
Wistar rats were purchased from CLEA Japan, Inc (Tokyo, Japan). All animal experiments were approved by Animal Research Committee in the Mie University. Twelve- to 15-week-old male Wistar rats were anesthetized with pentobarbital and perfused transcardially with 4% paraformaldehyde in 10 mM PBS, pH 7.4, and inner ears were dissected out. The samples were immersed in the same fixative overnight at 4C. For peripherin immunostaining, samples were immersed in the same fixative for 1 hr. After being washed with PBS, the samples were decalcified with 0.25 M EDTA for 1 week at 4C and then rinsed several times with PBS. The inner ears were cryoprotected in sucrose overnight at 4C, embedded in Tissue-Tek OTC compound (Sakura Finetek, Torrance, CA), and stored at −80C. The inner ears were cryosectioned at 10-µm thickness, mounted on glass slides, dried for 30 min, and stored at −80C before use.
Antibodies
Affinity-purified rabbit anti-manserin antibody was prepared as described previously (Yajima et al. 2004; Kamada et al. 2010; Tano et al. 2010). The anti-synaptophysin antibody (Chemicon, Temecula, CA), anti-peripherin antibody (Santa Cruz biotechnology, Santa Cruz, CA), anti-myosin VIIa antibody (Santa Cruz biotechnology), and anti-neuronal class III β-tubulin antibody TUJ1 (Covance, Richmond, CA) were purchased. The secondary antibodies were as follows: biotinylated goat anti-rabbit IgG (Chemicon), Alexa Fluor 568-conjugated goat anti-rabbit IgG, and Alexa Fluor 488-conjugated goat anti-mouse IgG (Invitrogen, Carlsbad, CA).
Immunohistochemistry
Frozen sections were washed with PBS and treated with 3% hydrogen peroxide for quenching intrinsic peroxidase activity. After being washed with PBS, sections were blocked with 3% skim milk in PBS and incubated with anti-manserin antibody. For antibody absorption experiment, anti-manserin antibody was preincubated with 1 µM recombinant peptide overnight at 4C and used as primary antibody. After being washed with PBS, sections were treated sequentially with biotinylated goat anti-rabbit IgG. After being washed with PBS, sections were immunostained by the ABC method (Vectastain ABC Elite kit, Vector Laboratories, Burlingame, CA) and visualized using DAB.
For immunofluorescent double staining, sections were incubated with manserin antibody and other antibodies, such as anti-myosin VIIa antibody, anti-synaptophysin antibody, anti-peripherin antibody, and TUJ1. Sections were then incubated with fluorescent-labeled secondary antibodies. After being mounted with Prolong Gold Antifade reagent (Invitrogen), the sections were examined with a laser-scanning confocal microscope (FV1000, Olympus, Japan).
Results
Manserin Localization in the Organ of Corti
Inner and outer hair cells are connected to afferent terminals from spiral ganglion cells, regulated by the efferent connection from olivocochlear nucleus as a feedback system (Fig. 1A). To speculate the physiological roles of manserin from its localization in the inner ear, immunohistochemical examination of manserin in the inner ear was performed. Frozen sections of the rat inner ear were prepared, and immunohistochemical staining using anti-manserin antibody was performed. Positive signals of manserin were detected near the inner (arrow) and outer (arrowhead) hair cells of the organ of Corti in the cochlea (Fig. 1B). No signal was detected in the absorption experiment, which was performed to verify the specificity of the signal (Fig. 1C). Next, the localization of manserin near both hair cells of the organ of Corti was examined in detail by performing fluorescent double staining using the hair cell marker myosin VIIa (Hasson et al. 1997). Positive signals for manserin were observed (Fig. 1D), which are localized beneath the inner and outer hair cells stained by myosin VIIa (Fig. 1E, F). No colocalization was found with manserin and myosin VIIa inside the hair cells.
To investigate whether manserin occurs at synaptic sites connected to hair cells, we performed double staining with a presynapse marker, synaptophysin (Khalifa et al. 2003) and manserin (Fig. 1G–I). Manserin was colocalized with synaptophysin at the bases of only inner hair cells (Fig. 1I, arrow, white signals), indicating localization of manserin in efferent synapses, but not colocalized at the bases of outer hair cells indicating afferent synapse (Fig. 1I, arrowhead). Pearl chain–like localization of manserin was also detected in the fibers of outer hair cell bases (Fig. 1I, inset)—that is the afferent fibers of type II spiral ganglion neurons (Waka et al. 2003; Thiers et al. 2008). These results demonstrate that manserin localizes at the synapse of the inner hair cells and, to some extent, at the synapse of the outer hair cells.
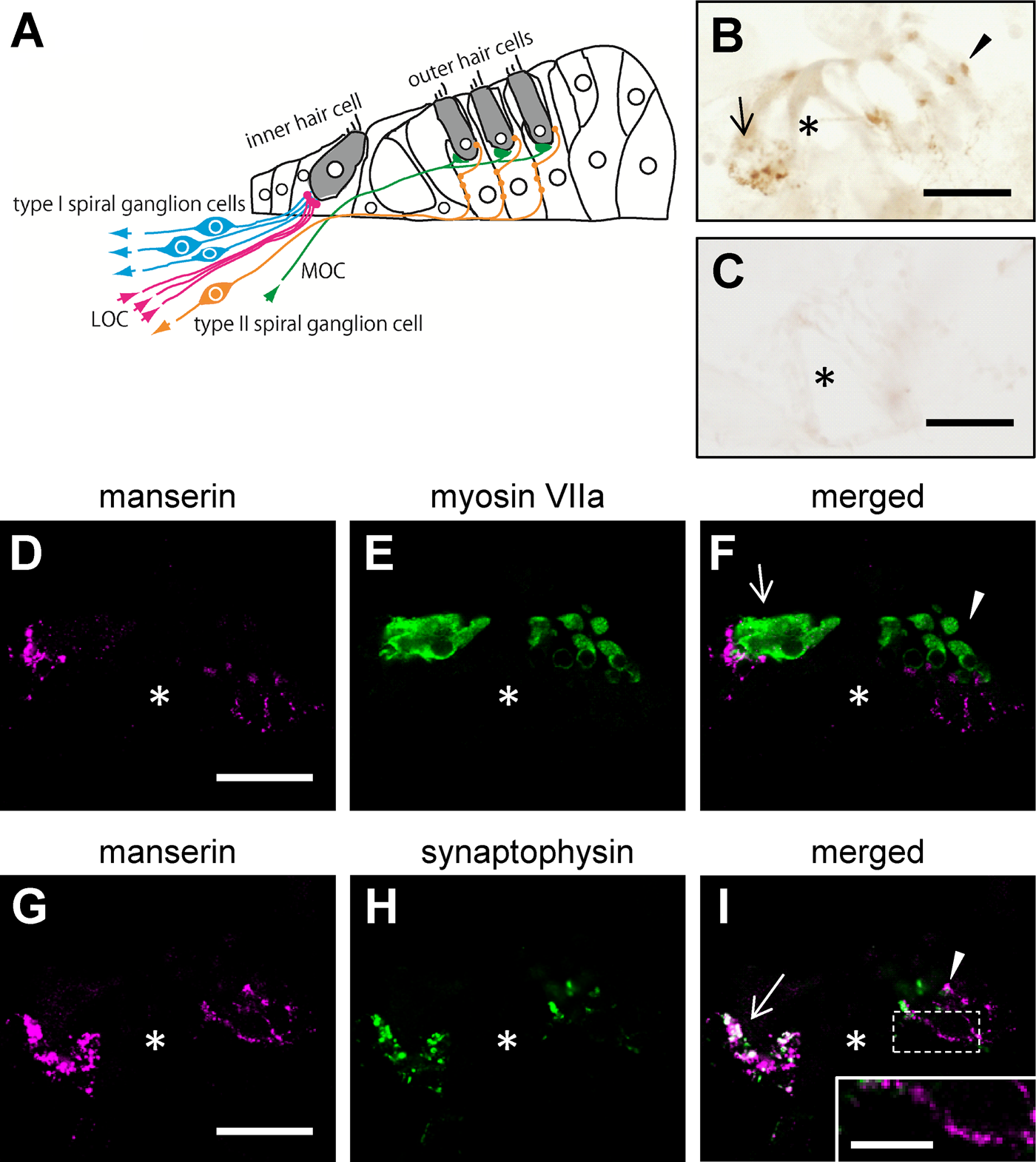
Manserin immunoreactivity in the organ of Corti. Schematic drawing of the anatomical structure in the organ of Corti are shown (A). Colored arrows indicate directions of neural transmission. Sections were incubated with anti-manserin antibody (B) or absorbed antibody (C), and antigen was detected using DAB staining. Sections are also immunofluorescently labeled with antibody against manserin (D and G; magenta), myosin VIIa (E; green), or synaptophysin (H; green). (F, I) Merged images (D + E, G + H, respectively). (I, inset) Enlarged views of the dashed frame (I). Manserin staining showed a pearl chain–like staining pattern below the outer hair cells (inset). Asterisk indicates tunnel of Corti. Arrow and arrowhead indicate inner and outer hair cells, respectively. LOC (MOC), lateral (medial) olivocochlear projections. Scale bars, 30 µm (inset, 10 µm).
Manserin Localization in Type II Spiral Ganglion Cells
As described above, manserin localized at the synapse of the hair cells of the organ of Corti. Since signals from the organ of Corti are transmitted to the central nervous system via the spiral ganglion (Raphael and Altschuler 2003), we examined whether manserin localizes in the spiral ganglion. As shown in Figure 2A, DAB staining showed positive signals for manserin in the spiral ganglion cells, but the positive signals were limited to only some of the cells. There are two apparent subtypes of spiral ganglion cells: type I spiral ganglion cells comprise the majority (more than 90%), and type II spiral ganglion cells make up the remainder (less than 10%). The nerve fibers of type I spiral ganglion cells are large and myelinated, whereas the nerve fibers of type II spiral ganglion cells are small and non-myelinated (Rusznak and Szucs 2009). Fluorescent double staining with type I marker TUJ1 antibody (Sekerkova et al. 2008) and type II marker peripherin antibody (Hafidi 1998) was employed to clarify the type of spiral ganglion cells in which manserin localizes. Positive signals for manserin were observed in the form of dotlike patterns in the cells (Fig. 2E, H) and fibers (Fig. 2K). These positive signals matched with that of peripherin-labeled cells (Fig. 2H–M) but not with that of TUJ1-labeled cells (Fig. 2B–G). These results indicate that manserin exclusively localizes in type II spiral ganglion cells.
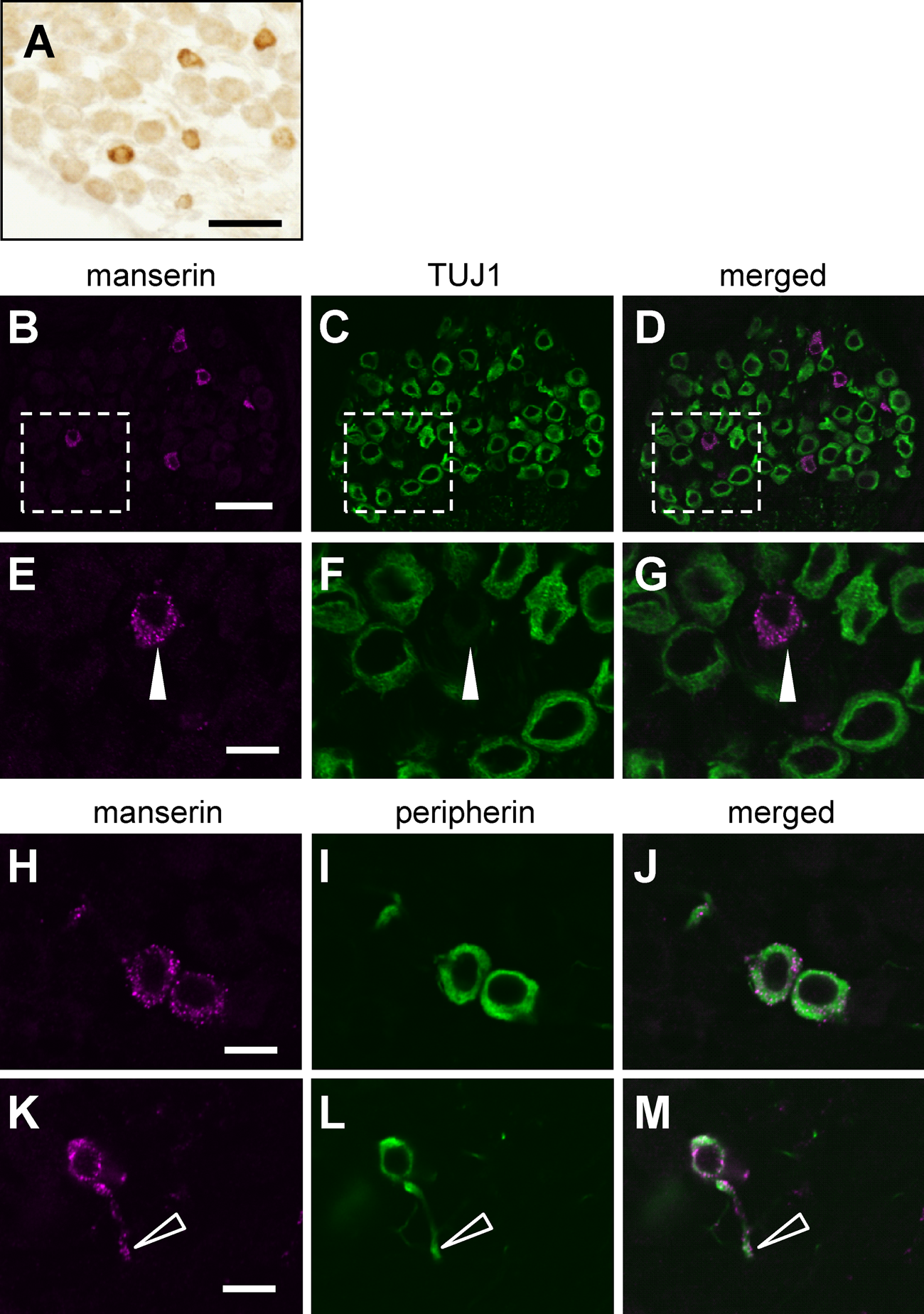
Manserin immunoreactivity in spiral ganglion. DAB staining of manserin in spiral ganglion (A). Double immunostaining for colocalization of manserin (B, E, H, K; magenta) and TUJ1 as type I spiral ganglion cell marker (C, F; green) or peripherin as type II spiral ganglion cell marker (I, L; green). (D, G, J, M) Merged images (B + C, E + F, H + I, K + L, respectively). (E–G) Enlarged views of the dashed frame (B–D, respectively). Manserin was not localized in TUJ1-positive type I spiral ganglion cells (closed arrowheads). (H–J) Manserin (magenta) was colocalized in peripherin-positive type II spiral ganglion cells (green). (K–M) Manserin immunoreactivity present in not only cytoplasm but also fibers (open arrowheads) of type II spiral ganglion cells. Scale bars, 30 µm (A–D) and 10 µm (E–M).
Manserin Localization in the Macula of Utricle and Saccule and Crista Ampullaris
The inner ear has a vestibular system composed of semicircular canals and otolith organs such as utricle and saccule. The macula of the utricle and saccule contain hair cells that detect linear acceleration. Crista ampullaris localizes at the end of the semicircular canals and contain hair cells that detect rotation (Rutka 2004). Manserin’s localization was also examined in the vestibular system. Positive signals with DAB staining for manserin were observed in the form of dotlike patterns in macula of saccule (Fig. 3A), utricle (Fig. 3B), and crista ampullaris (Fig. 3C). To investigate the detail localization of manserin in this area, we performed double staining using an antibody against synaptophysin and found that localization of synaptophysin matched, in part, with that of manserin (Fig. 3D–L, arrowheads). These results indicate that manserin localized at some synapse of vestibular organs and semicircular canals.
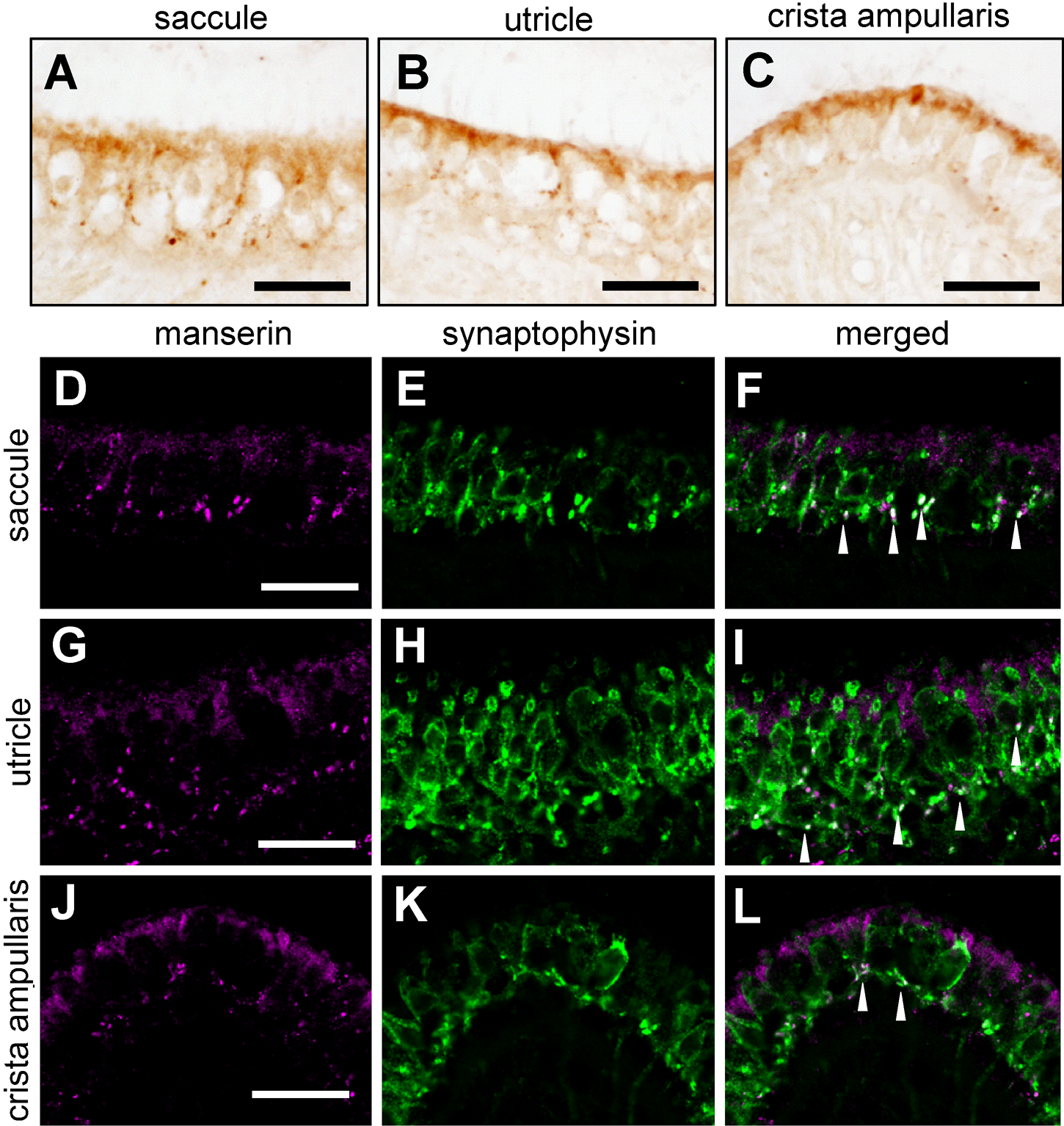
Manserin immunoreactivity in the macula of utricle, saccule, and crista ampullaris. Manserin was detected by DAB at saccule (A), utricle (B), and crista ampullaris (C). Colocalization of manserin (D, G, J; magenta) with synaptophysin (E, H, K; green) was immunofluorescently examined in saccule (D–F), utricle (G–I), and crista ampullaris (J–L). (F, I, L) Merged images (D + E, G + H, J + K, respectively). Arrowheads indicate colocalization of manserin and synaptophysin. Scale bar, 30 µm.
Discussion
In this study, we demonstrated that manserin in the rat inner ear localizes at the following sites: (1) neuronal terminals of the organ of Corti, (2) type II spiral ganglion cells, and (3) the synapses in the vestibular system. These results suggest that, in the inner ear, manserin might be involved in the regulation of auditory and vestibular system.
The following reports on the localization of neuropeptides and their physiological roles in the inner ear: neuropeptide Y in the organ of Corti and spiral ganglion (Gomide et al. 2009); pituitary adenylate cyclase–activating polypeptide in the organ of Corti and spiral ganglion (Drescher et al. 2006); neurotensin in the inner hair cells, Deiters cells, and spiral ganglion (Gomide et al. 2005); and substance P, enkephalin, and calcitonin gene-related peptide in the organ of Corti (Ylikoski et al. 1989). Furthermore, it has been reported that dynorphin, calcitonin gene-related peptide, and enkephalin are distributed in the efferent fibers of the lateral olivocochlear nucleus and are involved in the synaptic transmission of type I spiral ganglion cells that form a synapse with the inner hair cells (Raphael and Altschuler 2003). This suggests the role of neuropeptides in the auditory nervous transmission. In our study, since manserin was localized in the synapses of the inner and outer hair cells of the organ of Corti, it is suggested that manserin plays a role in the auditory nervous transmission in the same manner as the other neuropeptides, such as dynorphin and enkephalin. We have also found that the upregulation of manserin in response to physical stress (Kamada et al. 2010), indicating that manserin is involved in stress response. Therefore, manserin might be involved in the occurrence of stress-related inner ear diseases, such as sudden deafness and Ménière disease.
Manserin localized in the type II spiral ganglion cells. The spiral ganglion cells are the first in the auditory pathway to fire action potentials from the hair cells of the organ of Corti (Rusznak, and Szucs 2009). Type I spiral ganglion cells, which comprise the majority of spiral ganglion cells, innervate with the inner hair cells, while type II spiral ganglion cells innervate with the outer hair cells. Inner hair cells are responsible for the transmission of sound information through the afferent fibers of spiral ganglion cells. Conversely, outer hair cells play a role in the amplification of sound due to their ability to contract and elongate via efferent fibers of the medial olivocochlear nuclei. Thus, the primary role of outer hair cells is to amplify the movement of the basilar membrane by changing the cell length, to amplify the frequency-specific signal, and to transmit the tuned signal to type II spiral ganglion cells (for reviews, see Raphael and Altschuler 2003; Ashmore 2008). The fact that manserin localized specifically in type II spiral ganglion cells suggests that manserin is involved in the signal transmission from outer hair cells and might subsequently lead to regulation of the amplification of sound in the inner ear. This is indeed interesting, since unlocking the mechanism of how manserin is involved in the amplification of sound information may help to elucidate pathophysiology of diseases related to acoustic hyperesthesia and obtundation.
Manserin was localized in the synapses of macula and crista ampullaris in vestibular system as well. Macula is found inside the two otolith organs, such as utricle and saccule, where they sense the acceleration and tilt of the head by monitoring movements of macula hair cells, such as vibration and inclination. Crista ampullaris is found inside the semicircular canals, detecting angular acceleration (Minor 1998; Schubert and Minor 2004). We have found in this research that manserin was localized at the synapses of the utricle, saccule, and crista ampullaris, suggesting that manserin acts as a transmitter for vestibular systems. This finding may open up the possibility to elucidate the pathogenesis of vestibular disorders.
The limitations of this study are that the information obtained in the present study was from morphological experiments. Although manserin in the inner ear shows characteristic localization and thus shows indication to the role of manserin in auditory and vestibular system, the physiological role of manserin in auditory and vestibular system is still unclear. In the future, further experimental investigations are required, such as electrophysiological analysis.
Footnotes
The author(s) declared no potential conflicts of interest with respect to the authorship and publication of this article.
This study was supported by the Grant-in-Aid for Young Scientists (B) from the Japan Society for the Promotion of Science (27391891 to MI-E).
