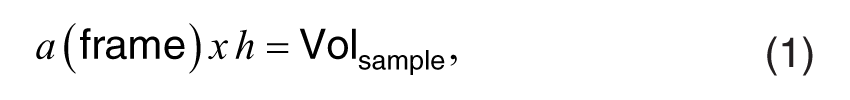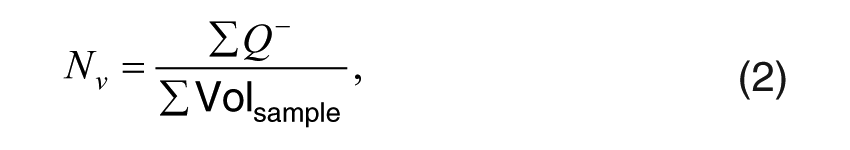Abstract
In response to toxic stressors, cancer cells defend themselves by mobilizing one or more epidermal growth factor receptor (EGFR) cascades that employ xeroderma pigmentosum–A (XPA) to repair damaged genes. Recent experiments discovered that neurons within the auditory nerve exhibit basal levels of EGFR+XPA co-expression. This finding implied that auditory neurons in particular or neurons in general have the capacity to mobilize an EGFR+XPA defense. Therefore, the current study tested the hypothesis that noise stress would alter the expression pattern of EGFR/XPA within the auditory nerve. Design-based stereology was used to quantify the proportion of neurons that expressed EGFR, XPA, and EGFR+XPA with and without noise stress. The results revealed an intricate neuronal response that is suggestive of alterations to both co-expression and individual expression of EGFR and XPA. In both the apical and middle cochlear coils, the noise stress depleted EGFR+XPA expression. Furthermore, there was a reduction in the proportion of neurons that expressed XPA-alone in the middle coils. However, the noise stress caused a significant increase in the proportion of neurons that expressed EGFR-alone in the middle coils. The basal cochlear coils failed to mobilize a significant response to the noise stress. These results suggest that EGFR and XPA might be part of the molecular defense repertoire of the auditory nerve.
Introduction
As a prosurvival response to toxic chemical (e.g., chemotherapy) and physical (e.g., radiation) stressors, some cells mobilize one or more epidermal growth factor receptor (EGFR) signaling cascades that employ xeroderma pigmentosum–A (XPA) to protect their genome. Patients with mutations in the xpa gene are highly susceptible to the development of cancers such as basal cell carcinomas and exhibit a shorter life span. Progressive neurological degeneration among such patients occurs early in life and includes truncal ataxia, inability to walk, dysphasia, diffused neural loss, myelin pallor, gliosis, global cerebral atrophy, and infantile brain size.1–7 The clinical audiology profile of these patients includes progressive bilateral sensorineural hearing loss with or without preservation of speech discrimination and the presence of vestibular hypofunction. 8 Totonchy et al 9 reported audiology data for patients with xpa mutations and revealed that hearing loss could occur within the second decade of life. This hearing loss is initially a sloping high-frequency loss but can eventually flatten out in the third decade of life. Interestingly, clinically significant hearing loss was associated with a 39-fold increase in neurological degeneration, and the degree of hearing loss paralleled neurological decline. At autopsy, the clinical audiology profiles are supported by histological findings. For instance, the temporal bones revealed unremarkable external auditory meatus, tympanic membranes, and ossicles, which suggest normal outer and middle ear functions. 8 However, the cochlea exhibits scattered loss of hair cells, atrophy of the stria vascularis, and severe degeneration of spiral and Scarpa’s ganglion neurons.8,9 Interestingly, spiral ganglion neurons degenerate in areas of the inner ear with normal hair cells, which has supported the conclusion that XPA is important for neurons. 8
Neurons appear to employ both EGFR and XPA. EGFR mediates a multiplicative signaling network in neurons which underlies its diverse roles in neuronal survival, development, proliferation, and migration; synaptic plasticity; and axonal sprouting.10,11 In addition, EGFR signaling is coupled to Ca2+ waves, and it has been suggested that this signaling employs XPA to maintain the integrity of neuronal DNA during glutamatergic neurotransmission.12–15 Indeed, human and animal loss-of-function mutations in the xpa gene result in neurodegeneration. 8 Nonetheless, only recently it was shown that neurons could co-express EGFR+XPA. 15 Here, the spiral ganglion neurons that comprise the auditory nerve were shown to co-express EGFR+XPA under normal conditions. However, it is not clear whether or not neurons in general or spiral ganglion neurons in particular employ both EGFR and XPA in response to stress. The current work is a preliminary attempt to address this issue.
Noise overexposure is known to stress spiral ganglion neurons via an increase in glutamate load. 16 Furthermore, noise stress induces DNA damage within these neurons, and the neurons are known to elicit DNA damage responses due to noise overexposure.17,18 Therefore, the hypothesis of the current work is that noise stress would force spiral ganglion neurons within the auditory nerve to alter their expression of EGFR/XPA. If noise stress is able to alter the expression pattern of EGFR/XPA, then this would provide the motivation to develop therapies that augment this endogenous defense to rescue spiral ganglion neurons and other types of neurons from acquired neuropathies.
It is important to explore the interaction of XPA and EGFR within inner ear spiral ganglion neurons for at least three reasons. First, XPA is important for the survival of spiral ganglion neurons. 8 Spiral ganglion neurons harbor high endogenous metabolic activity, and it is known that neurons with high intrinsic metabolism exhibit specific types of bulky helix-distorting DNA damage products that specifically require the repair function of XPA.19,20 Previous studies have revealed constitutive expression of XPA within spiral ganglion neurons, 17 an indication that endogenous XPA is important for the survival of spiral ganglion neurons under normal conditions. Interestingly, spiral ganglion neurons appear to mobilize XPA as a defensive response to ototoxic conditions such as cisplatin intoxication and exposure to loud noises.18,21 In addition, previous research has shown that small molecular weight carboxy alkyl esters can therapeutically enhance the expression of XPA within spiral ganglion neurons, and this increase is associated with the preservation of spiral ganglion functions following noise trauma. 17 Therefore, determining signaling mechanisms that regulate the expression of XPA could lead to new therapies that preserve the function of spiral ganglion neurons after ototoxic exposures. 22 Second, EGFR signaling could be one potent mechanism for regulating XPA expression within spiral ganglion neurons. EGFR signaling regulates the expression, translocation, and stability of XPA via the Ras/Raf/MEK/ERK and the PI3K/Akt pathways. 15 Indeed, EGFR signaling is known to be important for the survival of spiral ganglion neurons. 23 Third, by exploring the interaction between EGFR and XPA within spiral ganglion neurons, it might be possible to develop new therapies that enhance XPA expression via manipulation of the EGFR. For instance, ototoxic exposures such as noise exposure and chemical intoxications result in DNA damage within spiral ganglion neurons.21,24 In addition to disrupting the functions of active genes, DNA damage is a potent inducer of cell death. 25 Therefore, future therapies designed to rescue spiral ganglion functions after ototoxic exposure could target XPA activity by manipulating EGFR signaling. The current study provides the first experimental evidence that spiral ganglion neurons co-express EGFR and XPA in response to stress, an indication that EGFR/XPA signaling might be part of the molecular defense repertoire of the auditory nerve.
Materials and Methods
Research Design
Twenty-four rats were used in this study. They were randomly assigned to either a control group (n=12) that received no noise exposure or a noise-exposed group (n=12). After noise exposure, the animals from both groups were sacrificed and their inner ears were processed for IHC. Then, design-based stereology was used to quantify the number of immunoreactive spiral ganglion neurons in inner ears from both groups. Statistical analyses were then performed to determine differences between the groups.
The structure of the IHC studies was as follows: Negative control (received no antibody) tissue sections mounted on microscope slides and slides with tissues from both the control and noise-exposed groups were all processed simultaneously. The slides were processed via two rounds of IHC. In the first round, the slides were stained to detect one protein (e.g., XPA). Then, the same slides received a second round of IHC to detect the second protein (e.g., EGFR). After IHC, the slides were imaged and cell counts were conducted via design-based stereology. Details of each of these steps are presented below.
Animals
Male Long–Evans rats (250–300 g) were acquired from Harlan Laboratories, Inc. (Livermore, CA) and served as subjects. The animals were given 1 week to acclimatize to the rat vivarium after they were delivered from Livermore, CA. Each animal was then randomized into control and noise exposure groups, with 12 animals per group. All experimental manipulations of the animals were approved by the Institutional Animal Care and Use Committee at the Loma Linda Veterans Affairs Medical Center, Loma Linda, CA, USA.
Noise Exposure
The animals were conditioned to the noise chamber before the actual noise exposure. Each day, the animals were removed from their home cages and staged in the noise exposure cage within the noise chamber, with the noise turned off. This started by staging the animals in the noise chamber for 1 hr the first day, 2 hr the second day, 3 hr the third day, and so on, until the day of the actual noise exposure. To avoid audiogenic seizures on the day of the noise exposure, the noise was raised to 90-dB sound pressure level (SPL); then, the animals were visually monitored for physical signs of stress (e.g., hyperactivity, excessive grooming, scrawling on the sides of the cage, etc.). After 1 or 2 min, the noise was slowly raised in 5-dB steps (at each step, the animals were visually monitored) until the desired noise level was reached. The noise dose used in the current study was an 8-kHz octave band noise at 105-dB SPL for 4 hr. This noise dose was chosen because in Long–Evans rats, this dose results in DNA damage within spiral ganglion neurons leading to permanent sensorineural loss.26,27 Furthermore, this noise dose does not kill spiral ganglion neurons within 24 hr of the noise. 17 This provided an opportunity to study XPA and EGFR expression within the neurons. The animals were unrestrained in individual cages within a reverberant 40-liter noise chamber. Within 24 hr of the noise exposure, the animals were euthanized for IHC.
Immunohistochemistry
Following gravity-assisted intravascular perfusion, the head of each animal was removed, skinned, and postfixed overnight. The temporal bones were decalcified, processed, and paraffin-embedded according to published procedures. 28 Microtomy consisted of serial sectioning in the midmodiolar plane. Cochlear sections (8 µm per section) were then deparaffinized and hydrated. IHC was then performed on these sections, and the procedure has been described in detail previously. 29 Antigen retrieval was accomplished with 20-min incubation in a low-pH (0.80–3.06), high-temperature (90C–98C) sodium citrate–citric acid buffer. The primary antibodies were diluted in blocking solution at a 1:100 concentration. The primary antibodies are commercially available and included goat anti-EGFR (sc-31155) and rabbit anti-XPA (sc-853) (Santa Cruz Biotechnology, Inc.; Dallas, TX). We and others have previously characterized the validity of these antibodies in preabsorption experiments, immunofluorescent assays, IHC, and Western blots, and the results are as follows15,21: (1) Western blots revealed that the EGFR antibody selectively detects the 170-kDa protein product and the XPA antibody selectively detects the 40-kDa protein product, (2) incubating the antibody with the immunogen yields no immunoreactivity in tissue sections, and (3) negative control sections where the antibody is omitted from the IHC procedure do not result in immunoreactivity. Chromogen consisted of 3,3′-diaminobenzidine tetrahydrochloride (DAB) to produce a brown reaction product or DAB+1% cobalt (Co) to produce a blue reaction product.30,31
Double Immunohistochemistry
The procedure for double IHC has been reported previously.15,32,33 Briefly, the double IHC procedure produces three reaction products (brown, blue, and black to blackish-blue) in the same tissue sections. For instance, a section that was initially treated with the XPA antibody and developed with DAB exhibited a brown reaction product. This same section is then microwaved, blocked for peroxidase activity, processed with the EGFR antibody, and treated with DAB+Co to produce a blue reaction product. In areas where both EGFR and XPA are approximated, the reaction product is black to blackish-blue.
Design-Based Stereology
The disector principle was used in first-order stereology quantification of neurons that expressed XPA, EGFR, or EGFR+XPA. The theoretical aspects of the principle and the protocol have been described in detail.15,34–36 Briefly, the animals were randomly assigned to either the control or the noise-exposed group. Their cochleae were serial sectioned in a midmodiolar plane beginning before the opening of the bulla then passing through the cochlea. This serial sectioning made it possible to access all the neurons which are contained in Rosenthal’s canal (Rc). Therefore, Rc served as the reference space. Rc was an appropriate reference space because its borders are well defined and the entire canal was available for each animal. Slides from both the control and noise groups were run together through the IHC procedure by a technician who was blinded to the experimental conditions/groupings. After IHC, the slides remained masked so that the second technician who conducted the cell counts was completely blinded to the experimental design, grouping, and treatment of the sections. A modiolar plane of sectioning through the cochlea produced radial cross-sections through Rc. In these radial cross-sections, Rc appeared as a crude triangle with three borders. Therefore, three straight lines were used to trace the three borders of Rc. Then, two median lines (plumb lines) were drawn through the centroid of the canal. The three straight lines that formed the triangle shape of Rc along with their median plumb lines through the centroid of the canal divided the canal into four quadrants where each quadrant was delimited by straight lines. These delimiting lines served as unbiased disector counting frames that prevented multiple counting of a neuron that straddles more than one quadrant. 15 Only cells with a clearly defined soma (Q¯) were counted, and the sum of Q¯ [∑Q¯] was determined. To prevent counting the same cell more than once (e.g., edge effects), the counting rule was to only count cells within each quadrant of Rc but exclude cells that touch the virtual lines that divided the quadrants and demarcate the canal. These virtual lines outlined the area of the disector frame [a(frame)] and defined x and y dimensions. The thickness between adjacent serial sections provided the z-axis distance (h). Therefore,
where (Volsample) is the sample volume. Cell counts were collected from the Volsample of adjacent serial sections called a disector pair. In stereological equation,
where Nv is the numerical density or the total number of cells per unit of volume. Systematic random sampling was used to select sections from the reference space, and all immunoreactive cells were counted. By superimposing the unbiased disector counting frame on each section of the disector pair, the number of neurons of a given type (XPA, EGFR, or EGFR+XPA) could be counted within each quadrant. The number of neurons of a given type was then divided by the total number of immunoreactive neurons to obtain the proportion of neurons of a certain type within a given quadrant. The total number of immunoreactive neurons within each quadrant across groups was as follows: superior medial (SM) = 380, superior lateral (SL) = 215, inferior medial (IM) = 305, and inferior lateral (IL) = 382. The coefficient of error (CE) for the control group was 0.008 (0.8%), and the CE for the noise group was 0.022 (2.2%), indicating high counting precision for both groups. The proportion of neurons that showed positive staining is displayed in Figs. 4 to 6.
Statistical Analysis
Prism5 v5.03 (GraphPad Software, Inc., La Jolla, CA) was used to perform all statistical computations. Statistical computations consisted of two-way ANOVAs. These computations were conducted to determine significant differences in cell counts. A p value <0.05 was considered statistically significant.
Results
Heterogeneous Expression Patterns Within Rosenthal’s Canal
Double IHC identified three expression patterns within Rc. These expression patterns are consistent with that of previous experiments. 15 Figure 1A shows a cross-section of Rc, where the neurons expressed XPA but there is some expression of the EGFR. Figure 1B and C are high-resolution views of fields of neurons within Rc, where the expression of EGFR, XPA, and EGFR+XPA could be detected within the neurons with (Fig. 1C) or without noise exposure (Fig. 1B). Figure 2A and B shows three neighboring cells. One cell expresses EGFR and is therefore labeled blue; another cell expresses XPA and it is labeled brown. The third cell expresses both XPA and EGFR (EGFR+XPA) along its basolateral border; therefore, this area is labeled black to blackish-blue. However, brown chromogen representing XPA expression alone can be seen in other areas of the same cell. Figure 3A and B shows two cells that express EGFR+XPA. Here, the black to blackish-blue reaction products are distributed throughout the two cells. It is important to note that not all cells were immunoreactive. Therefore, Rc contains cells that express XPA, EGFR, and EGFR+XPA as well as cells that show no such expression. For stereology quantification, proportion was determined by dividing the number of cells immunoreactive for a particular protein within a specific area by the total number of immunoreactive cells contained in that same area.
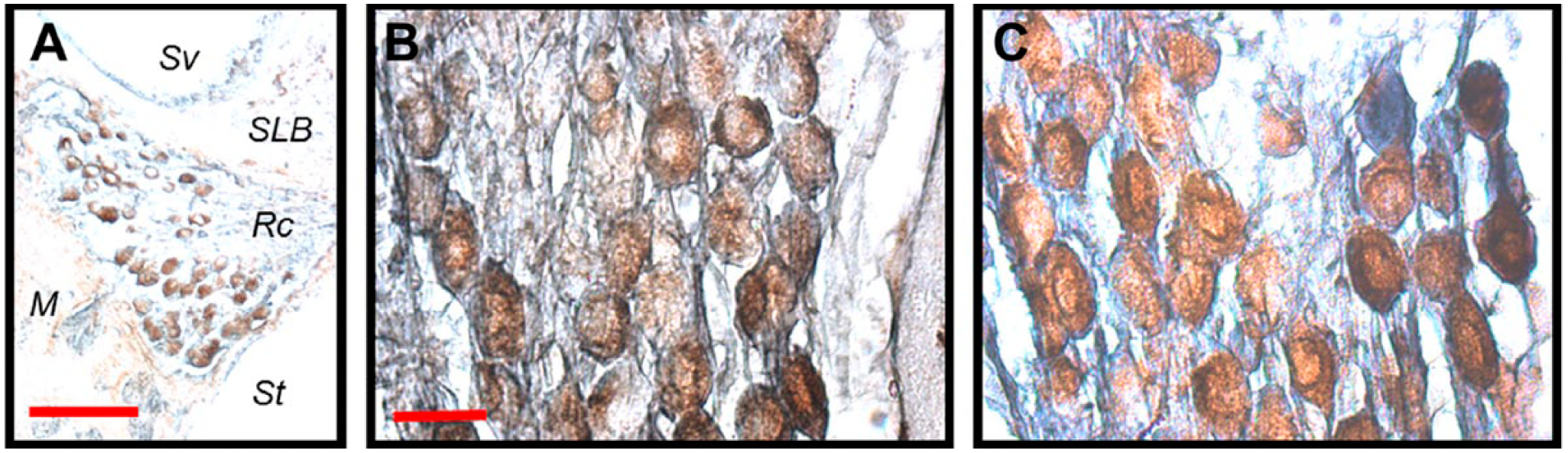
Immunoreactivity for xeroderma pigmentosum–A (XPA) and epidermal growth factor receptor (EGFR) within the spiral ganglion. Double IHC for XPA and EGFR was performed on the same spiral ganglion sections. 3,3′-Diaminobenzidine tetrahydrochloride (DAB) was used as a substrate to produce brown chromogen for XPA, and DAB+Co was used as a substrate to produce blue chromogen for EGFR. The co-reaction of brown (DAB) and blue (DAB+Co) resulted in a black to blackish-blue reaction product. A, Double staining of a radial cross-section through Rosenthal’s canal. This low-power micrograph of Rosenthal’s canal shows cells that were immunoreactive for both XPA and EGFR. B, High-power micrograph of a field of spiral ganglion cells without noise exposure. C, High-power micrograph of a field of spiral ganglion cells after noise exposure. Cells expressing EGFR, XPA, and EGFR+XPA were found in the spiral ganglion with and without noise exposure. Abbreviations: Sv, scala vestibuli; SLB, spiral limbus; Rc, Rosenthal’s canal; M, modiolus; St, scala tympani. Scale bars: A = 100 µm; B = 20 µm; scale bar in panel B applies to panel C.
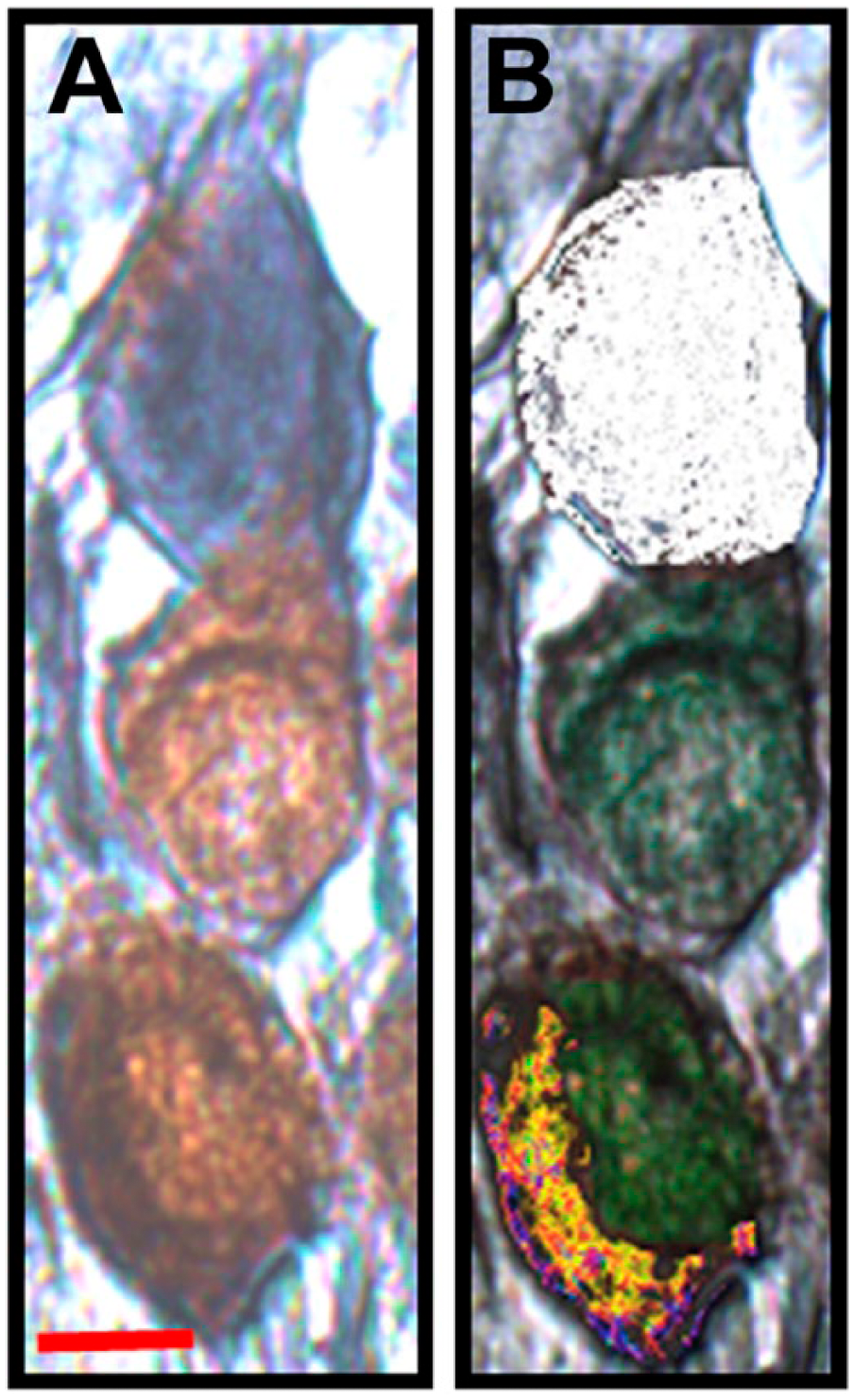
Heterogeneity among spiral ganglion neurons. A, Three adjacent cells, where one cell is expressing epidermal growth factor receptor (EGFR; blue), another cell is expressing xeroderma pigmentosum–A (XPA; brown) while a third cell is expressing EGFR+XPA (black and brown). B, Color masks are applied to further reveal the expression pattern among the three cells in panel A. The white color mask identifies the cell expressing EGFR, whereas the green color mask identifies XPA expression among the cells. The multicolor color mask identifies the cell that is expressing EGFR+XPA. Scale bar: A = 10 µm; scale bar in panel A applies to panel B.
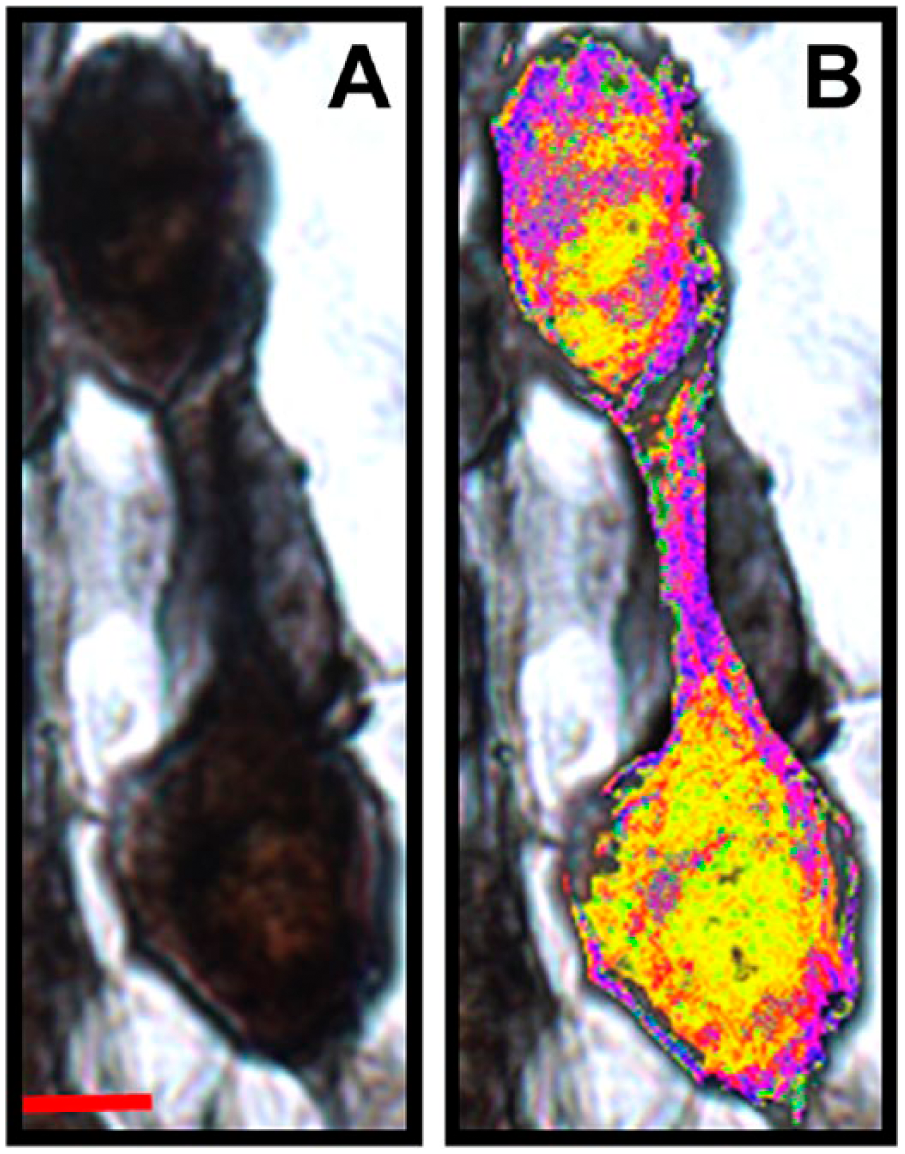
Co-expression of xeroderma pigmentosum–A (XPA) and epidermal growth factor receptor (EGFR) within spiral ganglion neurons. A, Two adjacent cells that seem to equally express both XPA and EGFR. In this expression pattern, co-expression (black to blackish-blue) is distributed throughout the soma. B, Multicolor color masks are applied to further reveal the expression pattern among the two cells in panel A. Scale bar in panel A = 10 µm; scale bar in panel A applies to panel B.
Distribution of EGFR+XPA Co-Expression Within Rc Under Control Conditions
To evaluate the distribution of neurons within Rc, the approach developed by Guthrie 15 was used. Here, three straight lines were traced along the crude triangle shape of Rc. These lines formed the borders of the canal. Then, median plumb lines were drawn through the centroid of the canal. This effort allowed the canal to be divided into four quadrants: SM, SL, IM, and IL. Superior means oriented toward scala vestibuli, whereas inferior means oriented toward scala tympani. A medial orientation refers to the cochlear modiolus, whereas a lateral orientation refers to the cochlear lateral wall. Therefore, the precise location of neurons within Rc could be determined for the purpose of stereological quantification.
The expression patterns within Rc were initially conceptualized as expression patterns from two related population of neurons. One population of neurons constitutively expressed XPA with and without EGFR co-expression and was thus referred to as the XPA population. The second population of neurons constitutively expressed EGFR with and without XPA co-expression and was therefore referred to as the EGFR population. Therefore, a determination of whether or not the noise exposure would alter EGFR+XPA co-expression within each of the two populations of neurons was pursued. Figure 4A to C reveals the mean proportion of EGFR+XPA co-expressing neurons among the XPA population of neurons. Here, the number of neurons that co-express EGFR+XPA was divided by the total number of neurons that expressed XPA-alone plus EGFR+XPA. These EGFR+XPA co-expressing neurons were distributed across the four quadrants under normal conditions. Figure 5A to C reveals the proportion of neurons that co-express EGFR+XPA among the EGFR population. Here, the number of neurons that co-express EGFR+XPA was divided by the total number of neurons that expressed EGFR-alone plus EGFR+XPA. Among the EGFR population, neurons expressing ® were also distributed across the four quadrants under normal conditions.
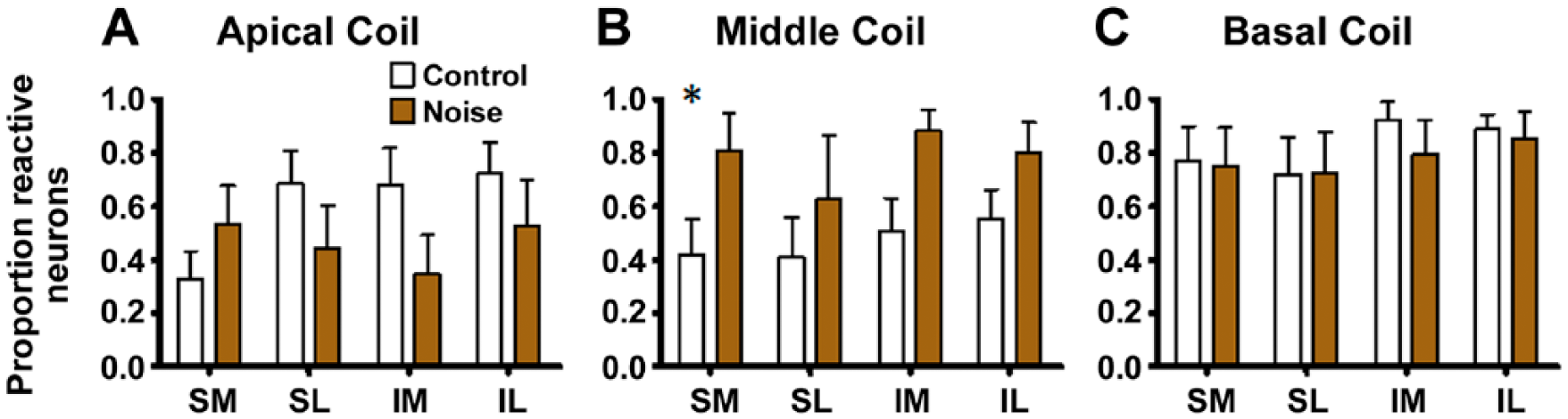
Noise-induced modifications to xeroderma pigmentosum–A + epidermal growth factor receptor (EGFR+XPA) co-expression among XPA neurons. Panels A to C reveal the distribution of cells that co-express EGFR+XPA within the population of neurons that were immunoreactive for XPA. Noise exposure induced a marked increase in the co-expression of EGFR+XPA within the middle cochlear coil with little or no effects on the apical and basal cochlear coils. Panels where comparison of control with noise exposed is statically significant are indicated with an *. Bars represent mean ± standard error. Abbreviations: SM, superior medial; SL, superior lateral; IM, inferior medial; IL, inferior lateral.
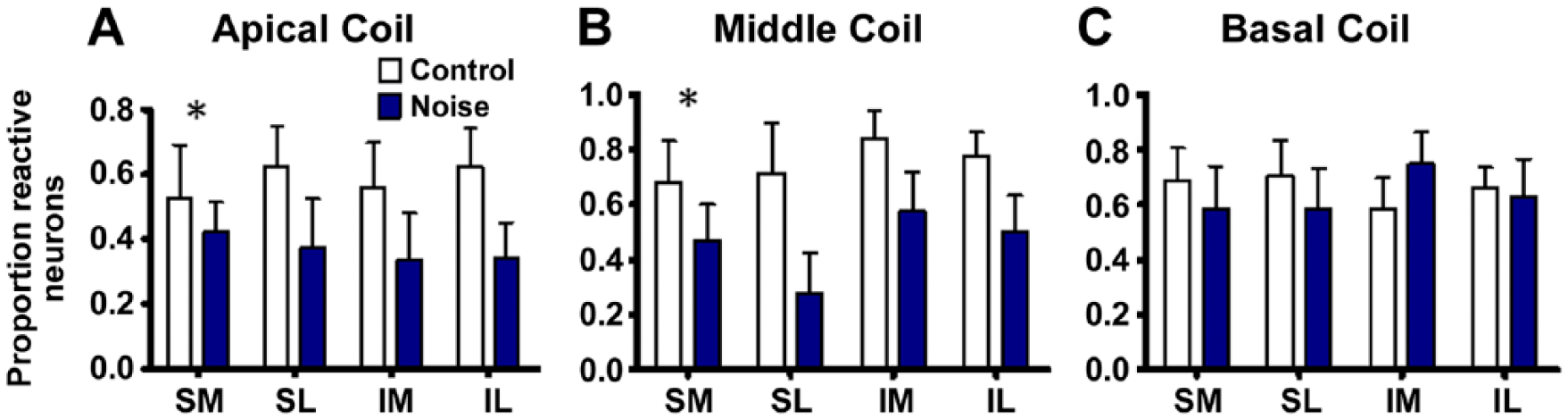
Noise-induced modifications to xeroderma pigmentosum–A + epidermal growth factor receptor (EGFR+XPA) co-expression among EGFR neurons. Stereology quantification of cells that co-express EGFR+XPA was conducted within each quadrant to more precisely reveal cellular distribution patterns across the canal. Under normal (control) conditions, EGFR+XPA co-expression seemed to be fairly distributed across the four quadrants within the apical, middle, and basal cochlear coils. However, noise exposure caused a marked suppression of EGFR+XPA co-expression in the apical and middle coils with little or no effect in the basal coil. Panels where comparison of control with noise exposed is statically significant are indicated with an *. Bars represent mean ± standard error. Abbreviations: SM, superior medial; SL, superior lateral; IM, inferior medial; IL, inferior lateral.
The three distinct expression patterns (XPA-brown, EGFR-blue, and EGFR+XPA-black) were then conceptualized as three distinct populations of neurons within Rc (Fig. 6). Therefore, the proportion of neurons that expressed XPA-alone, EGFR-alone, and EGFR+XPA with and without noise exposure was quantified in each of the four quadrants (SL, SM, IM, and IL). Figure 6A and B illustrates the results for the apical cochlear coil. Under control conditions, there seems to be more neurons that express XPA-alone and EGFR+XPA, but this was not statistically significant, F(2, 108) = 1.47. Furthermore, there were no statistically significant differences between the proportion of neurons within the quadrants, F(3, 108) = 0.00, p>0.05. Figure 6C and D illustrates the results for the middle cochlear coil. Under control conditions, the middle coil seems to be dominated with neurons that expressed XPA-alone and EGFR+XPA. Indeed, there were over 2-fold more neurons expressing XPA-alone and EGFR+XPA compared with neurons that expressed EGFR-alone, and this difference was statistically significant, F(2, 132) = 14.76, p<0.05. However, there were no statistically significant differences between the proportion of neurons within the quadrants, F(3, 132) = 0.00, p>0.05. Figure 6E and F illustrates the results for the basal cochlear coil. Under control conditions, there seems to be a difference in the proportion of neurons that express XPA-alone, EGFR-alone, and EGFR+XPA. This difference was found to be statistically significant, F(2, 132) = 16.48, p<0.05. However, there were no statistically significant differences between the proportion of neurons within the quadrants, F(3, 132) = 0.00, p>0.05.
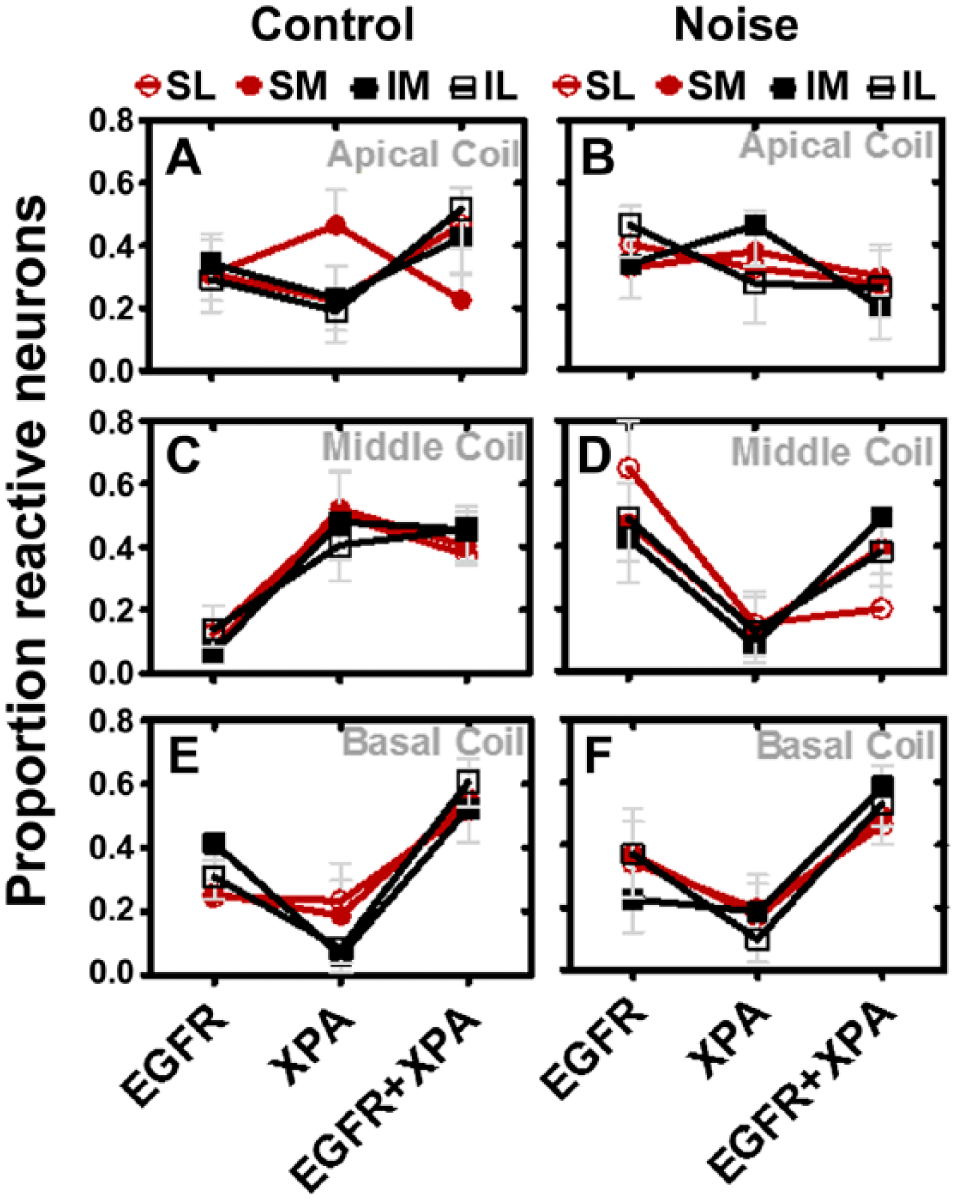
Heterogeneous response to noise exposure within the middle cochlear coil. Apical coils (A, B): Under normal conditions, EGFR expression is fairly distributed within the four quadrants, whereas XPA is most abundant in the SM quadrant. The expression of EGFR+XPA is fairly distributed across the quadrants except there is a downregulation in the SM quadrant. Noise exposure caused a suppression of XPA expression in the SM quadrant while increasing XPA expression in the IM quadrant. EGFR+XPA expression was suppressed in the SL, IM, and IL quadrants due to noise exposure. However, these noise-induced alterations were not statistically significant (p>0.05). Middle coils (C, D): Under normal conditions, XPA and EGFR+XPA expressions were the most abundant, whereas EGFR expression was the least abundant. However, after noise exposure, EGFR exhibited a dominant form of expression, whereas XPA expression was suppressed and the EGFR+XPA expression became unevenly distributed within the quadrants. These noise-induced alterations were statistically significant (p<0.05). Basal coils (E, F): Under normal conditions, EGFR+XPA expression was the most abundant form of expression, whereas XPA was the least. This expression pattern was unchanged after the noise exposure (p>0.05). The data represent mean ± standard error. Abbreviations: EGFR, epidermal growth factor receptor; XPA, xeroderma pigmentosum–A; SM, superior medial; SL, superior lateral; IM, inferior medial; IL, inferior lateral.
Noise-Induced Heterogeneity Within Rc
Figure 4A to C shows that after the noise exposure, EGFR+XPA co-expressing neurons were distributed across the four quadrants. The noise exposure failed to induce a significant change in the proportion of neurons in the apical, F(3, 64) = 2.06, p>0.05, and basal, F(3,65) = 0.28, p>0.05, cochlear coils. However, the noise induced a statistically significant increase in the proportion of neurons in the middle cochlear coil, F(3, 64) = 8.79, p<0.05. In Fig. 5A to C, note that the noise exposure induces a significant change in the proportion of neurons in the apical, F(3, 58) = 5.20, p<0.05, and middle, F(3, 66) = 9.79, p<0.05, coils. However, the noise failed to induce a statistically significant, F(3, 74) = 0.07, p>0.05, increase in the proportion of neurons that co-express EGFR+XPA in the basal coil. Figure 6A and B illustrates that after the noise exposure, there were no statistically significant differences between the three types of immunoreactive neurons, F(2, 108) = 1.35, p>0.05, or between neurons within the quadrants, F(3, 108) = 0.00, p>0.05, at the apical cochlear coil. Figure 6C and D illustrates the results for the middle cochlear coil. After noise exposure, there was still no difference between the quadrants, F(3, 108) = 0.00, p>0.05, but there were statistically significant, F(2, 108) = 11.14, p<0.05, changes in the proportion of immunoreactive neurons. For instance, the noise caused an increase in the proportion of neurons that expressed EGFR-alone, and this increase was 2- to 3-fold more than that under control conditions. Furthermore, the noise caused a reduction in the proportion of neurons that expressed XPA-alone and EGFR+XPA. These reductions were about 2- to 3-fold lower when compared with control levels. Figure 6E and F illustrates the results for the basal cochlear coil. After noise exposure, there was a difference between the three types of immunoreactive neurons, F(2, 108) = 8.12, p<0.05, with no significant differences between the quadrants, F(3, 108) = 0.01, p>0.05.
The data in Fig. 6 provide further insight into the results displayed in Fig. 4B and Fig. 5A and B. In Fig. 4B, it appears that the noise exposure caused a dramatic increase in the co-expression of EGFR+XPA within the population of neurons that constitutively express XPA. However, part of this noise-induced increase in co-expression of EGFR+XPA (Fig. 4B) is due to a large noise-induced decrease in the expression of XPA-alone (Fig. 6C and D). In Fig. 5A and B, it appears that the noise exposure caused a dramatic decrease in the co-expression of EGFR+XPA within the population of neurons that constitutively express EGFR. However, part of this noise-induced decrease in co-expression of EGFR+XPA (Fig. 5A and B) is due to a dramatic noise-induced increase in the expression of EGFR-alone (Fig. 6C and D). In addition, there was a decrease in the co-expression of EGFR+XPA, and this decrease occurred in both the apical (Fig. 6A and B) and middle (Fig. 6C and D) coils.
Discussion
Noise exposure has been shown to induce DNA damage within spiral ganglion neurons. 26 XPA is a prosurvival protein that can maintain genomic integrity by repairing damaged DNA. Recent research has shown that DNA repair adjuvant therapy can increase the expression of XPA among spiral ganglion neurons and this effect is associated with significant preservation of auditory nerve function after noise stress.17,22 EGFR is also a prosurvival factor that can increase cellular survival by triggering XPA-dependent and XPA-independent mechanisms. 15 Furthermore, it is known that EGFR is necessary for the survival of mature spiral ganglion neurons. 23 Recent experiments discovered that spiral ganglion neurons could co-express XPA and EGFR under normal/non-stress conditions. 15 However, no previous study has revealed whether or not this population of neurons is responsive to noise stress. In addition to DNA damage, noise exposure kills spiral ganglion neurons with or without damage to preneural hair cells.37,38 Therefore, the current study investigated whether or not noise exposure could alter the expression of XPA/EGFR among spiral ganglion neurons.
The identification of spiral ganglion neurons that express XPA, EGFR, or EGFR+XPA would support the hypothesis that these neurons are related but heterogeneous populations, where each population exhibits its own prosurvival response. Indeed, the notion of heterogeneous populations within the spiral ganglion has been supported by decades of electrophysiology and morphology research.39,40 More importantly, this heterogeneity hypothesis may provide a basis to interpret selective neuronal vulnerability within the spiral ganglion. For instance, both human and animal temporal bones have shown that some spiral ganglion neurons consistently survive ototoxic drug treatment (e.g., cisplatin and aminoglycoside) and noise stress, whereas other, neighboring, neurons die.41–43 This selective vulnerability has been attributed to differences in molecular defense repertoires. 18 In the current study, noise exposure suppressed the expression of XPA in the middle cochlear coils but elevated the expression of EGFR in the same middle coils. Although more modest, the noise also suppressed EGFR+XPA co-expression in the SL quadrant of the middle coil and in the SL, IM, and IL quadrants of the apical coil. The biological rationale for such a heterogeneous response within the spiral ganglion could be based on a drive to preserve the integrity of the nerve as a whole. For instance, different/heterogeneous responses to a given stress increase the probability that at least one response might be the right one needed to maintain the survival of some neurons and thus preserve residual functions of the nerve. In this case, some neurons will eventually die because they perpetuated the wrong response, whereas others will ultimately survive because the right response was achieved. Therefore, this line of thinking could help to explain why ototoxic stress whether from noise or chemicals typically results in the death of some spiral ganglion neurons while their neighboring (even touching) neurons survive. Although this line of thinking is novel when applied to spiral ganglion neurons, it is not without precedent. For instance, in the biodiversity–ecosystem field, it is known that the survival of species within a given ecosystem is driven by the diversity within the species, such that the more diverse (e.g., genetic and/or intraspecies diversity) the species, the more resilient the population to ecosystem stressors. 44 In the field of microbiology, it is also known that heterogeneity within a bacterial colony increases the probability that the colony will survive various types of stressors. 45 Nonetheless, additional research is needed to further support or refute the heterogeneity hypothesis as it relates to spiral ganglion neurons.
An alternative to the heterogeneity hypothesis is a homogeneity hypothesis. Here, all spiral ganglion neurons have the same defensive response. For instance, it is possible that all spiral ganglion neurons have the capacity to express EGFR+XPA, XPA-alone, or EGFR-alone. In the current study, the noise-induced decrease in XPA that accompanied the noise-induced increase in EGFR in the middle cochlear coil may represent the response of the same neurons. As such, the neurons are responding to the noise by suppressing XPA in favor of EGFR. Furthermore, the fact that spiral ganglion neurons can co-express EGFR+XPA is indicative of their inherent plasticity, and what seems to be different neuronal populations might simply be one population, where some neurons express different proteins at different times, but all the neurons can express each or both proteins when needed. A biological rationale to explain this scenario is more elusive, because if the neurons had the same capacity, then one would expect them to express the same proteins at the same time in response to a common stressor. For instance, after noise exposure, one would expect similar expression of XPA and EGFR among neighboring/touching neurons. However, the data show that there can be statistically significant differences in the expression of XPA, EGFR, and EGFR+XPA, even within the same cochlear coil. Instead of refuting the homogeneity hypothesis, these data could actually support the homogeneity hypothesis when conceptualized from the perspective of microenvironments.
The microenvironment conceptual framework suggests that Rc is composed of microenvironments that shape the local and ultimately the aggregate protein expression profile of the entire spiral ganglion. Microenvironments consist of two or more cells (e.g., a neuron and a satellite cell or two neurons, etc.) and their excretions (e.g., reactive metabolites, cytokines, neurotransmitters, etc.) along with insoluble intermediaries (e.g., extracellular matrix). 46 A given microenvironment will drive the genomic, transcriptomic, proteomic, and metabolomic profile of constituent cells. For instance, in a hypothetical two-cell microenvironment that contains a relatively electrophilic neuron and a nucleophilic neuron, the electrophilic neuron may employ EGFR to monitor the redox microenvironment produced by the nucleophilic neuron and thus trigger the expression of XPA as a defense (e.g., EGFR+XPA co-expression). This “paracrine situation” reflects the need of one cell to mobilize defenses due to the activity of a neighboring cell. 47 Another situation may occur where the internal metabolic drive of one neuron increases the redox load within its own cytoplasm and the neuron must constitutively mobilize XPA (with or without the need for EGFR) to survive. This “autocrine situation” reflects the need of some cells to persistently protect themselves from their own caustic internal cytoplasm, whereas other, even neighboring cells with lower metabolic rates may not need persistent protection. 48 Under these and other microenvironments, a given neuronal population may appear to exhibit vastly different defense responses, but in fact, all the neurons have the same capacity. What drives the differences in expression is the basal resting state of a given cell. Gross and indirect evidence of this can be observed in radial cross-sections through Rc, where some spiral ganglion neurons constitutively upregulate antioxidant defenses while adjacent (even touching) neurons do not.49–51
In this context of a microenvironment, each neuron within the ganglion has the capacity to produce the repertoire of molecular defenses that are endogenous to all neurons, but individual neurons within a given microenvironment differ in their decision on when and how much to express such molecular defenses. Therefore, all the neurons may have the capacity to express EGFR+XPA, XPA-only, or EGFR-only but display this capacity based on the demands within the microenvironment. Thus, a given cross-section through Rc that contains different microenvironments would be expected to exhibit a mosaic of expression patterns. Indeed, in the current study, a mosaic of XPA, EGFR, and EGFR+XPA expressing cells could be found in all cochlear coils. It is conceivable that multiple independent microenvironments may then integrate to produce a macroenvironment that is favorable to specific expression patterns. For instance, under normal and noise exposure conditions, cells in the basal coil that co-express EGFR+XPA were enriched, whereas cells that express XPA-alone were sparse. This notion of micro- and macroenvironments within Rc provides a basis to understand why two or more neurons of the same type may be equipped with what appears to be different defense strategies. The advantage of micro- and macroenvironments that produce mosaic patterns of defense strategies might be to ensure the survival of the ganglion as a whole. For instance, if all the neurons used one particular defense strategy, then they would share similar susceptibilities and likely die at the same rate. However, several studies have shown that even neighboring (e.g., touching) spiral ganglion neurons exhibit different susceptibilities to cell death.38,43,52 Micro- and macroenvironments that produce mosaic patterns of defensive strategies would likely increase the probability that some cells may survive a given stressor and thus maintain the survival of the ganglion. Ultimately, additional research is needed in this area, and the current study may serve as the basis to further interrogate the validity of the homogeneity and heterogeneity hypotheses.
Footnotes
Acknowledgements
The author would like to acknowledge the technical contributions of Xiao-Ping Xu, MD, and Mohamed Elgendy.
Competing Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
The author conceived the hypothesis and design of the experiment, supervised the data collection and analysis, and then wrote the manuscript.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by a Rehabilitation Research and Development Service award (C7600-W) from the U.S. Department of Veterans Affairs.