Abstract
A cerebral tumor was identified at necropsy in a mature female hooded crane (Grus monacha). On gross examination, the cut surface of the tumor revealed a soft gelatinous mass. On histologic examination, the tumor was mainly composed of 2 discrete components that resembled oligodendroglioma and astrocytoma. Both components had anaplastic changes, such as pleomorphism, high proliferative activity, microvascular proliferation, and necrosis. The oligodendrogliomatous component showed a honeycomb appearance formed by the accumulation of variably sized neoplastic cells with perinuclear halos and central nuclei. The astrogliomatous component consisted of remarkably pleomorphic cells, including bizarre giant cells. Immunohistochemistry revealed that the oligodendrogliomatous component cells were partially immunoreactive for vimentin and myelin basic protein, and the astrogliomatous component cells were immunoreactive for vimentin, S-100, and glial fibrillary acidic protein. Based on these findings, the tumor was diagnosed as an oligoastrocytoma.
Gliomas of the avian central nervous system are very uncommon. 9 In domestic poultry, a so-called fowl glioma that is characterized by multiple nodular gliomatous growths associated with disseminated nonsuppurative encephalitis has occasionally been described. 9, 10 Since 1983, we have performed more than 300 necropsies of overwintering cranes, with the main causes of death from bone fractures and other trauma-associated lesions, disseminated visceral coccidiosis, pseudomembranous enterocolitis, and aspergillosis. The purpose of this report is to describe the histopathologic and immunohistochemical characteristics of the first case of a spontaneously occurring oligoastrocytoma in an overwintering hooded crane (Grus monacha).
An exhausted mature female hooded crane had been kept in the Crane Preservation Center but had died of debilitation without specific clinical signs. On gross examination, the right cerebrum was slightly enlarged because of a 1-cm-diameter mass, which, on transverse section, was round to oval, poorly demarcated, dark brown, and gelatinous in texture. There were no other gross lesions in the crane.
Tissue samples were fixed in 10% phosphate-buffered formalin, embedded in paraffin, and sectioned at 3–4 microns. Sections were routinely stained with hematoxylin and eosin (HE), as well as histochemically with periodic acid–Schiff, and Alcian blue. The Envision Polymer Method (Dako Japan Co. Ltd., Kyoto, Japan) was used for immunohistochemical staining of the replicate brain sections. Primary antibodies and same-species positive control tissues are listed in Table 1. An antibody for Rous-associated virus 2 (RAV-2) was provided by Dr. K. Tsukamoto, National Institute of Animal Health, along with positive control tissue from a chicken with “fowl glioma” induced by RAV-2 was used as positive control tissue. 4, 11 Endogenous peroxidase was inactivated with 3% H2O2–methanol. Autoclaving (121°C for 15 minutes) was used for antigen retrieval. Detected antigens were visualized with 3-3′-diamino-benzidine-tetrahydrochloride reagent. Tris/HCl buffer (pH 7.5), with sections then counterstained with Meyer's hematoxylin.
Primary antibodies and control tissues used from immunohistochemical evaluation of a cerebral oligoastrocytoma in a hooded crane.*
Mab = monoclonal antibody, Pab = polyclonal antibody.
Tissue derived from the hooded crane presented in this report, except seminoma was from another hooded crane.
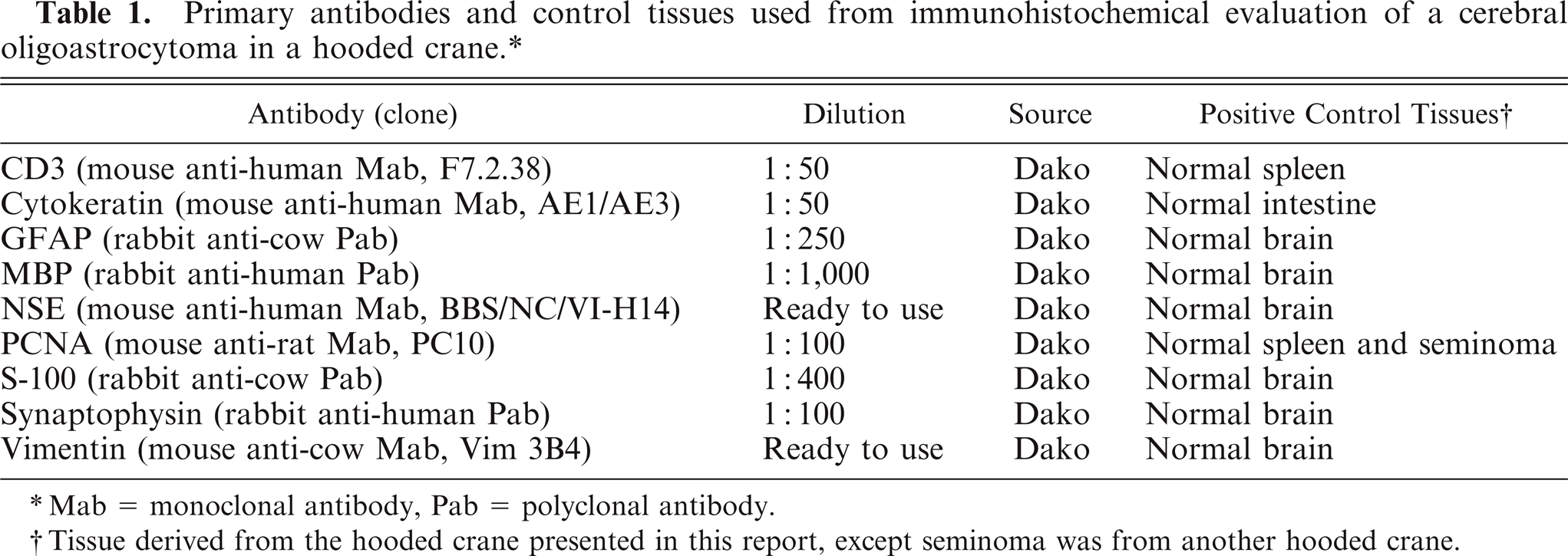
Histopathologically, the mass was nonencapsulated, infiltrated the surrounding normal tissue, and was composed of 2 discrete components resembling ether an oligodendroglioma and an astrocytoma, between which there was some intermingling. The oligodendrogliomatous component consisted of dense sheets of small, round neoplastic cells with a honeycomb structure (Fig. 1). These tumor cells usually had round to slightly ovoid, hyperchromatic central nuclei of various sizes and abundant clear cytoplasm with perinuclear halos. They also had prominent cytoplasmic membranes but did not have cytoplasmic processes.
Cerebrum; Hooded crane. Sheets of neoplastic oligodendrocytes with central nuclei surrounded by perinuclear halos. HE. Bar = 50 µm.
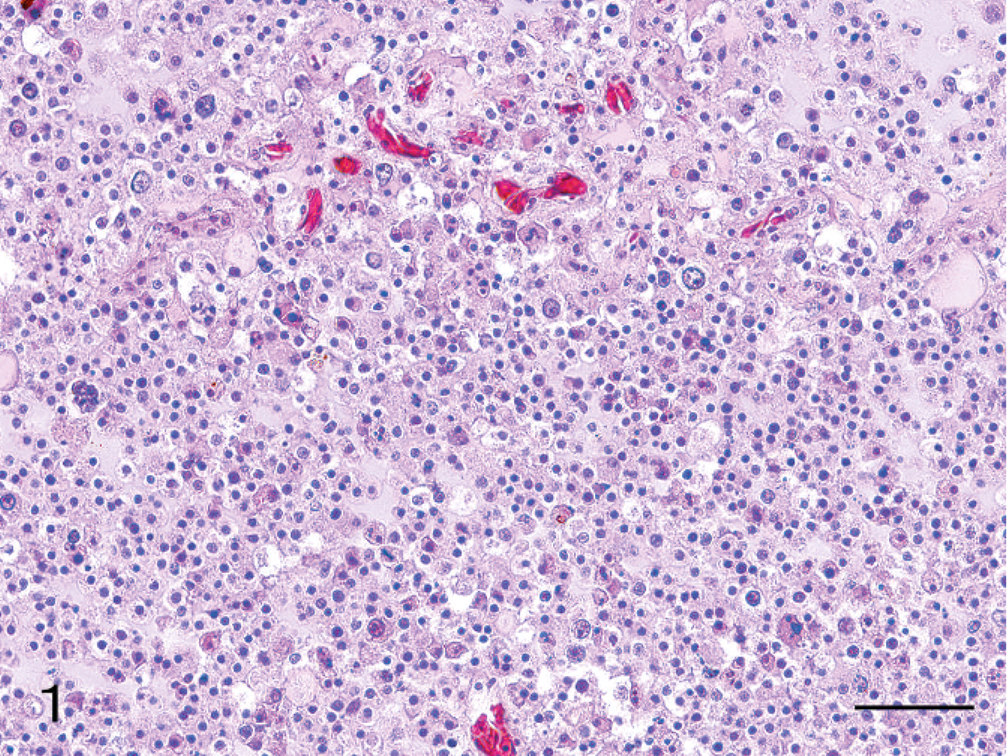
In the areas of astrocytoma predominance, cells were remarkably pleomorphic, with some bizarre giant cells present (Fig. 2). Nuclei were spherical to polygonal, occasionally hyperchromatic, and there was moderate anisokaryosis, with karyomegalic nuclei present. These tumor cells had abundant eosinophilic cytoplasm and indistinct margins. Some cells were poorly differentiated, whereas, other cells had the appearance of well-differentiated gemistocytic astrocytes.
Cerebrum; Hooded crane. Clusters of pleomorphic neoplastic astrocytes showing both small cells and bizarre giant cells. HE. Bar = 50 µm.
Within both components, there were interstitial accumulations of a pale basophilic mucinous-like substance that stained positively with Alcian blue. There were also minute foci of necrosis, hemorrhage, and microvascular proliferation. A small number of atypical mitotic figures were in the areas of both of the tumor-cell types. There was no metastasis of the tumor, and there were no significant histologic findings in the remaining brain parenchyma or in any of the other organs.
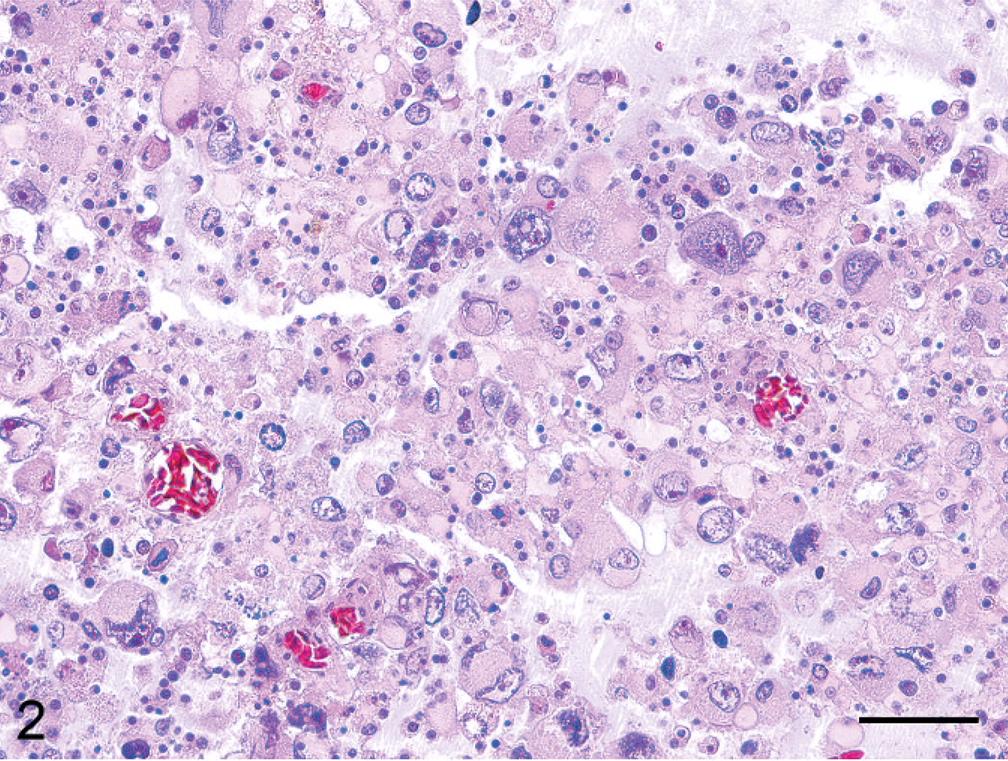
Immunohistochemically, cells that morphologically resembled astrocytes were immunoreactive for vimentin and partly positive for cytokeratin (Fig. 3). Pleomorphic giant cells were devoid of glial fibrillary acidic protein (GFAP) and S-100 expressions, whereas the smaller gemistocytic tumor cells that surrounded them were well stained (Fig. 4). Cells that morphologically resembled oligodendrocytes surrounded by perinuclear halos were partly positive for vimentin and myelin basic protein (MBP), and negative for cytokeratin AE1/AE3, GFAP, and S-100. The number of tumor cells with positive proliferating cell nuclear antigen (PCNA) immunostaining included approximately 64% of the nuclei of all cell types, but the pleomorphic giant cells had less proliferative activity (approximately 35%). All of the tumor cells were negative for neuron-specific enolase, synaptophysin, CD3, and RAV-2.
Cerebrum; Hooded crane. Positive immunoreactivity to cytokeratin in neoplastic astrocytes. Envision polymer method, hematoxylin counterstain. Bar = 20 µm.
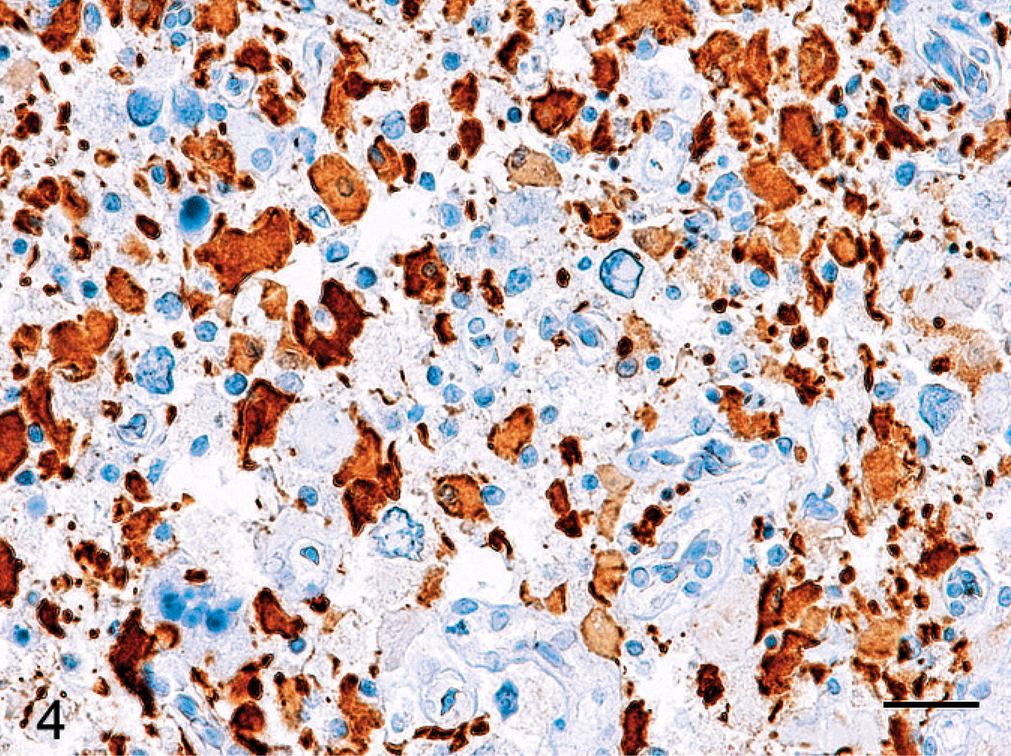
Cerebrum; Hooded crane. Expression of GFAP shows variation within the neoplastic astrocytes, with many of the pleomorphic giant cells immunonegative for GFAP. Envision Polymer Method, hematoxylin counterstain. Bar = 20 µm.
Grossly, this tumor formed a soft gelatinous mass, and, on histologic examination, it had mucinous accumulation and a honeycomb appearance formed by the sheets of tumor cells with artifactually swollen clear cytoplasm and central nuclei. These features represent the typical appearance of oligodendroglioma. 6, 7, 13 Several immunohistochemical markers, such as MBP, galactocerebroside, and Leu7 (CD57), have been detected in human oligodendrogliomas. 6 Although some tumor cells have expressed these markers, their expression is not specific for oligodendrogliomas, and, thus, it appears that there are no specific markers for the detection of neoplastic oligodendrocytes. 12 In the sections examined for MBP, the myelin sheathes were well detected, but, the cell bodies of normal oligodendrocytes, which were less frequently encountered, showed faint positive immunoreactivity for MBP in a portion of cells that morphologically resembled oligodendrocytes. Recently, it was reported that some brain tumors, including astrocytoma, ependymoma, and central neurocytoma, can occasionally exhibit a honeycomb-like appearance. 2, 12 However, the negative immunoreactivity of the tumor for GFAP, cytokeratin, NSE, and synaptophysin supports an oligodendrocytes origin for the tumor cells with perinuclear halos. In this case, however, there were also morphologically variable cells that resembled astrocytes, which ranged from small and well differentiated to large and bizarre. Immunohistochemical staining detected a variation in expression of GFAP within the astrogliomatous component, with many of the large pleomorphic cells without expression of GFAP. Such variation in expression of GFAP is common in mammalian high-grade astrocytomas. 3 There have been reports of glial neoplasms expressing cytokeratin markers, 1, 5, 8 and, for this reason, coexpression of GFAP and cytokeratin is a fairly common phenomenon. 1 We also recognized the expression of cytokeratin in tumor cells of astrocytic origin. Based in the result of staining for GFAP and cytokeratin, both the gemistocytic and the bizarre giant cells were identified as being astrocytic in origin.
Based on the morphologic features noted in this case, in conjunction with the criteria that are normally used for domestic animals, we made a diagnosis of anaplastic oligoastrocytoma. The most current World Health Organization histologic classification guidelines in domestic animal tumors define oligoastrocytomas as mixed gliomas with neoplastic oligodendrocytes and astrocytes that are either intermingled or separated into distinct clusters. 5 When 2 components are geographically distinct, the diagnosis is quite clear. In the current case, the neoplasm had distinct regions of classical anaplastic oligodendroglioma and classical glioblastoma multiforme (high-grade astrocytoma), and occasionally showed a diffuse admixture of the 2 components. The histologic features of anaplasia, such as nuclear atypia, cellular pleomorphism, high PCNA immunoreactivity, microvascular proliferation, and necrosis, are present both in the astrocytic and oligodendroglial components. In most human anaplastic oligoastrocytomas, these histologic features of a more anaplastic tumor are also present in both components. 3
Footnotes
Acknowledgements
The authors thank Dr. Kenji Tsukamoto, National Institute of Animal Health, Tsukuba, Japan, for providing the Rous-associated virus 2 polyclonal antibody, and would like to thank Dr. Hitoshi Hatai for his technical assistance.
