Abstract
Twenty-four border disease virus-seronegative, pregnant, mixed breed goats were experimentally comingled with 3 heifers persistently infected with bovine viral diarrhea virus type 2a (BVDV-2a). Twelve of the 24 exposed does aborted. Twenty-nine fetuses and 16 placentas from affected does were submitted to the Oklahoma Animal Disease Diagnostic Laboratory for a necropsy examination. Infection with BVDV was confirmed with a combination of immunohistochemistry, BVDV-2 polymerase chain reaction, and virus isolation in 19 of the 29 fetuses. On gross examination of the 19 fetuses and placentas in which BVDV-2a infection was confirmed, a mild placentitis (3/19), fetal mummification (1/19), and facial deformities (4/19) were noted. Histologically, placentitis (2/19), myocarditis (4/19), thymic depletion (5/19), choroid plexitis (3/19), encephalitis (2/19), and cerebral gliosis (1/19) were noted. Other causes of abortion in goats, including common bacterial and viral infections, were ruled out with histology, virus isolation, polymerase chain reaction, and aerobic bacteriologic cultures. As supported by the findings in this case, BVDV-2a should be included as a differential for abortion in goats. This is the first report of abortion in goats after exposure to persistently infected cattle.
Bovine viral diarrhea virus type 2 (BVDV-2) is a member of the pestivirus genus, along with BVDV-1, border disease virus (BDV), and classical swine fever virus (CSFV). Each of these pestiviruses may affect multiple species, although their natural host species are cattle (BVDV-1 and BVDV-2), sheep (BDV), and swine (CSFV). 1 Historically, any pestivirus infection affecting sheep and goats was considered to be BDV without supportive testing. It is therefore unclear in the older literature whether the pestivirus isolated was BVDV or BDV. 12
Bovine viral diarrhea virus is responsible for significant reproductive losses in the cattle industry. 5 Reproductive dysfunction may be associated with infection at any time during gestation, and the specific outcome of infection depends in part on the stage of gestation in which the dam is infected. Typically, cows that are infected between approximately 30 and 120 days of gestation may produce persistently infected calves, because this window of time is the period before immunocompetence is established. 3 It is the existence of these persistently infected (PI) calves that is the most damaging aspect of BVDV infection, because these calves shed large amounts of BVDV throughout their life. 10 After BVDV infection of pregnant heifers at day 85/86 of gestation, BVDV antigen can be detected in the fetuses as early as 14 days post inoculation, with cells in the liver and lungs testing positive by immunofluorescence. Bovine viral diarrhea virus antigen in these cases was not detected in the placenta until 18 to 22 days post inoculation. 6 These findings indicate that fetal infection with BVDV can occur without concurrent high levels of BVDV antigen in the placenta.
Infection of pregnant cows with BVDV may cause abortion at any time during gestation. After the development of immunocompetence of the fetus (at approximately 125 to 175 days of gestation) and during organogenesis, infection also commonly results in congenital defects. 14 Congenital defects in bovine fetuses include cerebellar hypoplasia, microencephaly, hydrocephalus, hydranencephaly, porencephaly, and hypomyelination. 3 Many other congenital defects associated with BVDV infection in the bovine have been observed, including osteochondrosis, growth retardation, optic neuritis, retinal degeneration, thymic hypoplasia, hypotrichosis/alopecia, curly haircoat, osteosclerosis, microopthalmia, cataracts, and mandibular brachygnathism. 3 If infection of the dam occurs after approximately 175 days of gestation, fetuses are usually able to mount an effective immune response against the virus and are usually normal at birth. 3
Similar to the effects of BVDV in cattle, BDV has traditionally been considered a cause of reproductive dysfunction in sheep, with abortions and congenital defects being the primary manifestation of disease. 17 The diagnosis of this disease in all affected ruminant species has been problematic in the past, because many of the tests conducted did not differentiate BDV from BVDV. Historically, if a pestivirus infection was documented in sheep, the disease condition was termed “border disease,” and it was assumed to be caused by BDV. Because of this difficulty in diagnosing BDV, the prevalence of the disease is currently unknown in North America. Border disease confirmed by polymerase chain reaction (PCR) is not known to have been reported in North America. Like with BVD, the most serious consequences of BDV infection arise when the virus infects susceptible ewes during pregnancy. In this case, the fetus is transplacentally infected, and the infection is manifested in the fetus while the dam's immune response typically clears the virus from the maternal system. In sheep, if the dam is infected before 60 days of gestation, fetal death is likely. Between 60 and 80 days, the fetus may become persistently infected with the virus, resulting in congenital defects. Infection after 80 days of gestation (when the fetal immune system is mature) usually results in normal lambs. 17
The reproductive effects of BVDV-2 infection in sheep inoculated at 3 stages of gestation have been previously investigated. 20 In this study, BVDV-2 antigen was detected in placentomes from 7 to 36 days post inoculation and in fetal fluids and tissues from 10 to 28 days post inoculation. Lesions observed in the fetuses examined at 21, 28, and 36 days post inoculation included cardiac petechial hemorrhages and hemoperitoneum accompanied by a severe ulcerative placentitis. Ewes inoculated at days 55 to 60 and 65 to 70 experienced a 77 and 67% abortion/perinatal death rate, respectively. Ewes inoculated at days 120 to 125 of gestation all delivered healthy, antibody-positive and virus-negative lambs. 20 These findings suggest that the pathogenesis of BVDV-2 infection in pregnant sheep is similar to that of pregnant cattle.
Several studies have shown that goats are susceptible to pestivirus infection under experimental conditions, and there are reports of natural infections of pestiviruses causing abortions in goats. 4, 15, 17 Intramuscular injection of goats with BVDV results in abortion without gross lesions but with extensive histologic changes within the brain, including encephalitis, necrosis, and cerebellar dysplasia. 15 A recent study demonstrated that nonpregnant goats seroconvert after natural exposure to BVD PI cattle. 2 In addition, there have been 2 recent reports of natural BVDV-2 infection of goats as detected and confirmed by PCR and phylogenetic analysis. 11, 15 The present report is the first documentation of infection and abortion in serologically negative, pregnant goats exposed to BVDV-2a PI cattle. This report describes novel gross and histologic changes within aborted fetuses naturally infected with BVDV-2a.
Materials and Methods
Animals and facilities
Thirty-six mature female mixed Spanish and Boer meat goats were initially tested for BVDV-1 and BVDV-2 by serum neutralization test and immunohistochemistry (IHC) for BVDV antigen performed on full-thickness ear biopsies. All goats tested were confirmed to be seronegative for antibodies to BVDV-1 and BVDV-2 and were negative for BVDV antigen on ear notches. During the natural fall breeding season, all 36 does had their estrous cycles synchronized using norgestomet implants followed by prostaglandin at the time of implant removal. All does cycled within several days of implant removal and were bred by either transcervical artificial insemination with frozen semen or natural cover by a fertile buck. Bovine viral diarrhea virus testing was not conducted on the buck. All breedings were recorded, ensuring a known breeding date for all does. All does were examined via transabdominal or transrectal ultrasonography for pregnancy approximately 35 days post breeding, and 24 does that were confirmed pregnant were selected for inclusion in this study. The goats were maintained on pasture prior to exposure to PI heifers. In accordance with the goats being bred in the fall, most of the goats were exposed to the heifers in January through March, and most of them kidded in March and April.
Three approximately 18-month-old black angus heifers persistently infected (PI) with BVDV were obtained from a private herd. The heifers appeared to be healthy on physical examination and remained clinically healthy throughout the duration of the study (6 months). Bovine viral diarrhea virus type 2a was isolated from the skin and blood of the heifers (3/3) over a period of 3 months, confirming PI status of these animals. The heifers were housed in a square pen of 100 ft × 100 ft (10,000 sq ft) with a 200-sq-ft shelter in one corner. Hay, grain, and water were each provided from a single source within the pen. All animals were handled in accordance with ethical measures, and experimental protocols were approved by the Oklahoma State University (OSU) Institutional Animal Care and Use Committee (IACUC).
Experimental design
The 24 pregnant goats were divided into 6 groups of 4 goats each; each group was exposed to the PI heifers at a different stage of gestation. Each group of pregnant goats was introduced to the heifers at days 41, 60, 82, 97, 118, and 139. Each group was added to the PI heifers in succession at approximate 3-week intervals, that is, 19 days after the initial group was introduced at 41 days of gestation, another group of 4 goats was introduced (at 60 days of gestation). In this manner, at the end of gestation, there were 24 goats cohabitating with the PI heifers in the single pen.
Serology, virus isolation, and polymerase chain reaction in exposed does
Blood samples and nasal swabs were collected from each doe on days 6, 7, and 8 after exposure to the PI heifers and every 3 weeks thereafter. Pooled blood samples collected on days 6, 7, and 8 from each doe were submitted and analyzed by PCR for the presence of pestiviral DNA. Two different PCR protocols were employed to detect and genotype the virus. First, genotyping was initially performed on the detected virus using a nested multiplex PCR. 9 This multiplex PCR technique was used on the initial samples submitted to the Oklahoma Animal Disease Diagnostic Laboratory and then intermittently throughout the project to confirm the presence of BVDV-2. Second, pan-pestivirus reverse transcriptase-PCR (rt-PCR) was performed on RNA extracted from serum using the Invitrogen One Step rt-PCR with Platinum Taq as previously described. 2 Two universal BVDV primers and the TaqMan probe were used under standard reaction conditions as previously described. 2 Bovine viral diarrhea virus type 1 and BVDV-2 from a collection of field samples served as the PCR positive controls. This real-time PCR procedure was less time intensive and allowed for high throughput detection of the virus.
Nasal swabs were also obtained and placed in a prepared viral transport media. The fluid bathing the nasal swab was filtered using a syringe filter (0.45 μm), and the samples from days 6, 7, and 8 were pooled for each doe. Virus isolation for BVDV was performed on the pooled media samples for each doe as previously described using Madin-Darby bovine kidney cells and a 5-day incubation period for cytopathogenic effect. 8 The virus isolation control was the uninoculated cell line, which was validated to be free of BVDV.
Blood for serology was collected from each doe at 3-week intervals (until parturition) after introduction to the pen containing the PI heifers. Serum samples were tested for BVDV-2a antibodies using a serum neutralization test. A virus neutralization test in Madin-Darby bovine kidney cells in 96-well microtiter plates was used to quantitate virus-neutralizing antibodies to BVDV. The virus used in this test was cytopathic BVDV-2a (125-C strain). The 1 : 4 dilution was the lowest dilution tested, and titers less than 1 : 4 are represented as negative titers. 7 Hyperimmune serum of known BVDV titre from National Veterinary Services Laboratories served as the positive control. The negative control for serum neutralization was fetal bovine serum, known to be negative for BVDV antibodies.
Pregnancy monitoring
All does were examined twice daily for gross evidence of abortion. Because does in early pregnancy do not always expel the fetus or may expel a small fetus with minimal signs, all does were examined for pregnancy at 3-week intervals via transabdominal ultrasonography. Visualization of a fetus with a beating heart, fetal movement, and clear allantoic/amniotic fluid and structurally intact placentomes was used as a positive sign of continued pregnancy. Statistical analysis was performed using Fisher's exact test to compare abortion rates between each of the 6 groups.
Postmortem examination of aborted fetuses
All aborted fetuses were collected and submitted for necropsy examination to the Oklahoma Animal Disease Diagnostic Laboratory. Severely debilitated kids that did not attempt to rise from lateral recumbency within 30 minutes after birth were killed by intracardiac injection of pentobarbital. A postmortem examination on aborted fetuses and killed kids was performed. When available, the placenta was also examined. Representative sections were collected and placed in 10% neutral-buffered formalin. Fixed tissues were paraffin embedded, sectioned at 4 to 5 μm, and stained with hematoxylin and eosin. Horseradish peroxidase strepavidin staining with a mouse monoclonal antibody for BVDV (BVDV Mab 3.12F1) was performed on 4- to 5-μm tissue sections. Virus isolation and PCR were performed as described above on placenta (when available), as well as pooled tissue samples containing liver, kidney, spleen, thymus, and lung.
Diagnostic testing was also performed to rule out other potential causes of caprine abortion. Bacterial aerobic culture and culture favoring the conditions necessary for Campylobacter fetus and Campylobacter jejuni growth were conducted on fetal tissues, including placenta (when available), lung, and liver. Polymerase chain reaction was performed on the placenta for chlamydophila, on the placenta and spleen for bluetongue virus, and on the placenta and liver for caprine herpesvirus. Fluorescent antibody testing was conducted on the liver and lung for caprine herpesvirus using a heterologous infectious bovine rhinotracheitis fluorescein isothiocyanate conjugate. Horseradish peroxidase streptavidin staining with a mouse monoclonal antibody for Toxoplasma (Vision Biosystems) and mouse monoclonal antibody for Neospora (VMRD) was routinely applied to 4- to 5-um sections of placenta. Histologic examination was conducted on all tissues to evaluate the potential for abortion by causes other than BVDV. All methods were performed following commonly accepted techniques as listed in the standard operating protocols of the Oklahoma Animal Disease Diagnostic Laboratory. This extensive collection of protocols is derived from published papers, and all protocols have undergone quality assurance/quality control before adoption in the diagnostic laboratory.
Blood and tissue samples from live kids
Blood samples and ear notches were obtained from all live-born kids at the time of birth. Samples were taken before kids had an opportunity to suckle colostrum, ensuring active antibody neutralizing titer. Serum samples were tested for BVDV-2a antibodies using serum neutralization as described above. Whole blood samples were tested for the presence of BVDV-2a antigen using PCR as described above.
Immunohistochemical staining to detect the presence of BVDV antigen was performed on ear notch skin biopsies obtained from the kids at the time of birth. Paraffin-embedded skin samples were serially sectioned and mounted on poly-
Results
Animal interaction
Upon introduction of each new group of goats into the pen, the cattle and goats used the same feeders and waterers, and fighting was not observed. The brief interactions between new goats and existing goats and heifers in the pen may be considered to be marginally stressful events that could potentially precipitate abortion, but no overt stress was seen to be experienced by the pregnant goats.
Abortion and neonatal death rates
Overall, 50% (12/24) of the goats aborted or delivered 1 or more weak kids that were unable to stand and died within 1 to 2 hours after birth or were killed. An additional 13% (3/24) delivered 1 or more kids that failed to thrive and died 24 to 36 hours after birth. Depending on the time of introduction to the PI heifers, each group of goats experienced varying abortion rates (Table 1).
Stratification of does by time of bovine viral diarrhea virus (BVDV) initial exposure to persistently infected (PI) heifers. Does that were exposed to PI heifers at different days of gestation experienced differing prevalence and timing of abortion or neonatal mortality. Differing superscripts denote significant differences (P < .05) in the prevalence of abortion between groups.
Of the group exposed to the PI heifers at day 41 of gestation, all does aborted (100%). On day 80 of gestation, 1 goat aborted a small fetus that was found in the pen, and on day 83 of gestation, this doe and 1 other were confirmed to be nonpregnant by ultrasound evaluation. On day 104 of gestation, ultrasound examination revealed that the remaining 2 does in this group had lost their pregnancies as well (Table 1). A second group of goats was introduced to the heifers at day 60 of gestation and remained pregnant until late in gestation. Three of the 4 goats aborted or delivered weak, nonviable kids between 144 and 149 days of gestation (Table 1).
Of the goats exposed to the PI heifers at 82 days of gestation, 2 of the 4 goats experienced abortion or weak kids that died soon after birth. The fourth group of goats was introduced to the PI heifers at 97 days of gestation. One doe aborted at day 131 of gestation, and a second doe delivered a weak kid that was killed soon after birth (Table 1). Another goat in this group accounted for the only doe mortality in this study; she died of hepatic lipidosis at day 120 of gestation.
Each of the goats introduced at 118 days of gestation experienced abortions or weak nonviable kids. The final group was introduced at 139 days of gestation, and none of the does in this group aborted (Table 1).
The statistical analysis of these abortion rates using Fisher's exact test revealed a significant difference (P < .05) between the group exposed at 41 days (4/4 abortions) and the group exposed at 139 days (0/4 abortions). A second significant difference (P < .05) was found between the group exposed at 118 days (4/4 abortions) and the group exposed at 139 days. There were no significant differences between any of the other groups in the number of does experiencing abortions (Table 1).
Serology, polymerase chain reaction, and virus isolation on doe samples
Neither virus nor viral antigen was detected in the blood (using PCR) or nasal secretions (using virus isolation) obtained on days 6 to 8 post exposure to PI heifers from any of the does in this study. Despite the inability to detect virus in the acute period after exposure, all of the does seroconverted with a significant fourfold rise in serum BVDV titers on BVDV-2a serum neutralization by 42 days post exposure. However, because of the variability in the time of seroconversion after exposure and the range of titers observed, it is not possible to determine the exact time of infection within this 42-day window.
Immunohistochemistry, virus isolation, and serologic findings on kid samples
Bovine viral diarrhea virus was detected using IHC performed on ear notch skin samples in 1 of the 13 live-born kids. This IHC-positive kid was weak at birth and subsequently died at 24 hours of age. The dam of this ear notch–positive kid was exposed to the PI heifers at 82 days of gestation. Virus isolation for BVDV was negative for blood samples obtained from all 13 live-born kids.
Serologic titers on the kids at birth ranged from <1 : 4 to 1 : 96. Of the 4 kids that survived 1 day or greater from the goats exposed at 82 days, 3 of the 4 had titers of <1 : 4. The fourth kid had a titer of 1 : 96. The titers of the 3 kids from the group exposed at 97 days were <1 : 4, 1 : 32, and 1 : 96. Six healthy kids were produced from the group exposed at 139 days; each had a titer of <1 : 4.
Necropsy findings
Twenty aborted or stillborn fetuses and 9 killed kids from 21 does were subjected to complete necropsy examination over a 2-month period. All were in good body condition with adequate body fat stores. The aborted kids ranged in weight from 0.9 to 2.7 kg and had a crown rump length ranging from 28 to 41 cm (Table 2). Nineteen kids/fetuses were male, and 10 were female. Of the 29 cases submitted for necropsy, 19 were confirmed infected with BVDV-2a with IHC, PCR, virus isolation, and/or serum neutralization.
A summary of the postmortem body condition and gross and histopathologic findings in goat fetuses and kids from 12 does exposed to bovine viral diarrhea virus persistently infected heifers.
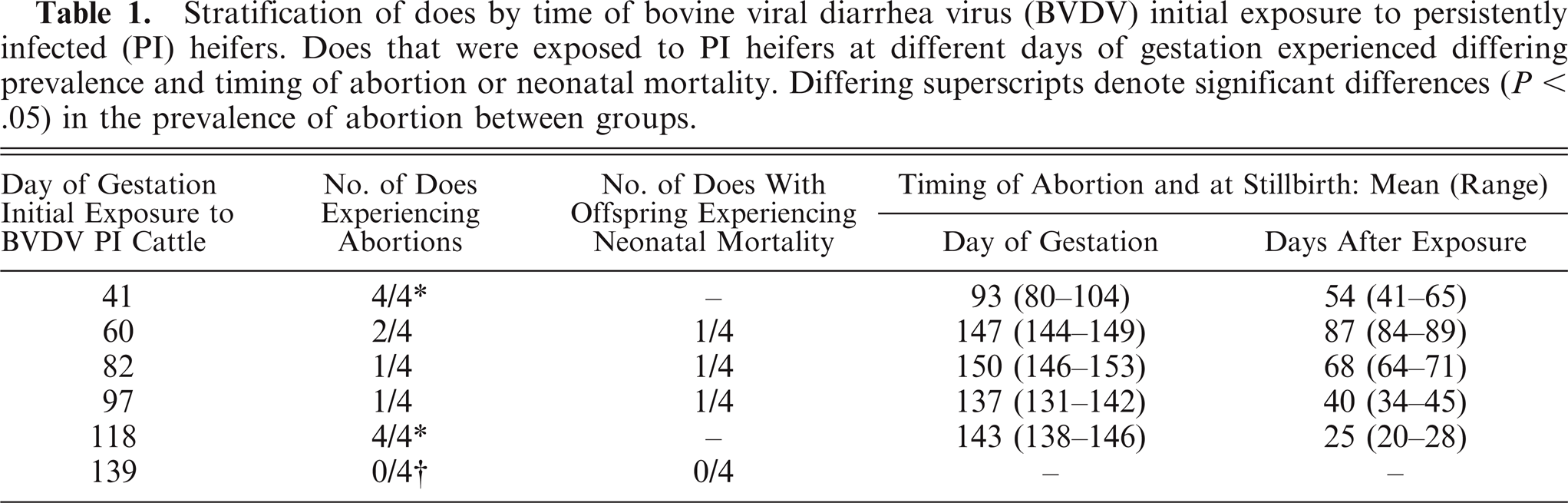
Gross lesions were minimal in the 19 cases of confirmed BVDV-2a infection. In 9 of 19 cases, gross lesions were not observed. Placentitis (1/19), fetal mummification (1/19), and facial deformities (4/19) were noted. Facial deformities included brachygnathia inferior (2/19) and prognathia (2/19) (Fig. 1). Other lesions observed included fibrinous peritonitis (1/19), hydrothorax and ascites (1/19), pulmonary hemorrhages (1/19), unilateral keratitis (1/19), and bronchopneumonia (1/19). The gross and histologic changes by organ system for each animal submitted are summarized in Table 2.

Head, goat. There is marked brachygnathia inferior.
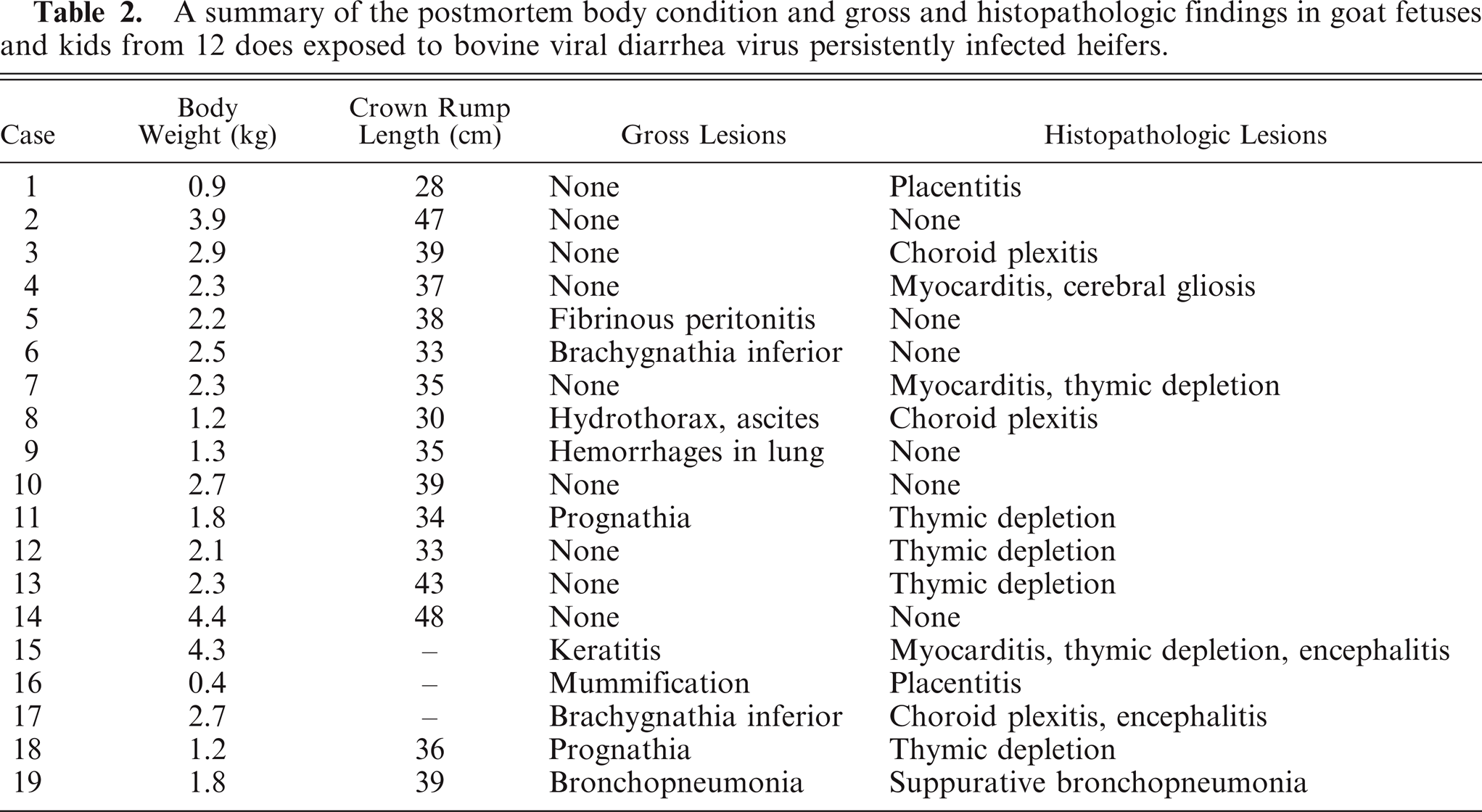
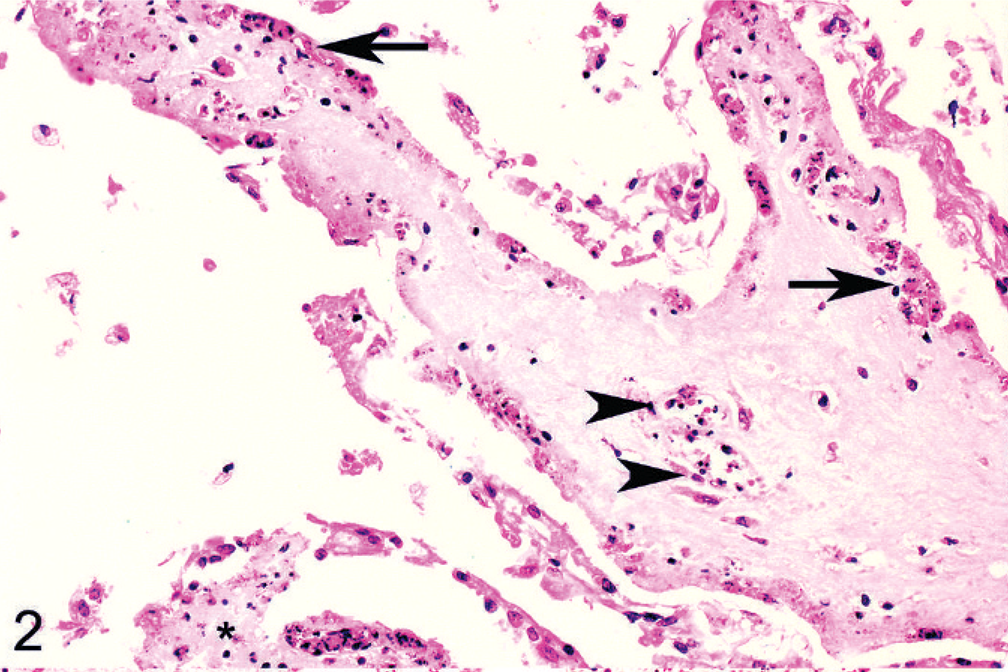
Histologic lesions were restricted to 4 organ systems in fetuses with confirmed BVDV-2a infection. They included placentitis (2/19), thymic depletion (5/19), myocarditis (4/10), and a variety of brain lesions (5/19). Six out of 19 animals had no histologic lesions. The placentitis was characterized by necrosis of the trophoblastic epithelium, a mononuclear stromal infiltrate, and necrosis of the deep stromal vessels (Fig. 2). There was diffuse thymic depletion within affected animals, which varied in severity. The myocarditis was characterized by a multifocal lymphohistiocytic infiltrate with scattered small areas of myocardial necrosis (Fig. 3). The brain lesions concentrated within the cerebrum and spared the cerebellum, and there were 3 distinct lesions: choroid plexitis (3/19); encephalitis characterized by perivascular infiltrate (2/19); and multifocal glial nodules (1/19). In the stroma of the choroid plexus, there was a multifocal infiltrate of lymphocytes, macrophages, and plasma cells admixed with nuclear debris (Fig. 4). Inflammatory cells were also present free within the ventricular cavity. The epithelium was often thin. Multifocal glial nodules were also present (Fig. 5). There was a multifocal mononuclear perivascular infiltrate of lymphocytes, plasma cells, and macrophages within meninges or gray matter within the cerebral parenchyma. Within 1 animal, there was a bile duct hamartoma within the liver, which was considered incidental. The other fetal tissues examined, including lung, liver, kidney, spleen, thyroid, trachea, and eye, were within normal limits. Bone and bone marrow were not examined in these cases.
Chorioallantois, goat. Necrotic cellular debris is scattered within the superficial stroma (arrows), and the trophoblast epithelium is lifted away. The stroma is infiltrated with mononuclear inflammatory cells admixed with necrotic cellular debris (asterisk). There is necrosis of the deep stromal vessels (arrowheads).
Heart, goat. Lymphocytes and macrophages (arrow) surround degenerate and necrotic myofibers (arrowheads). HE stain.
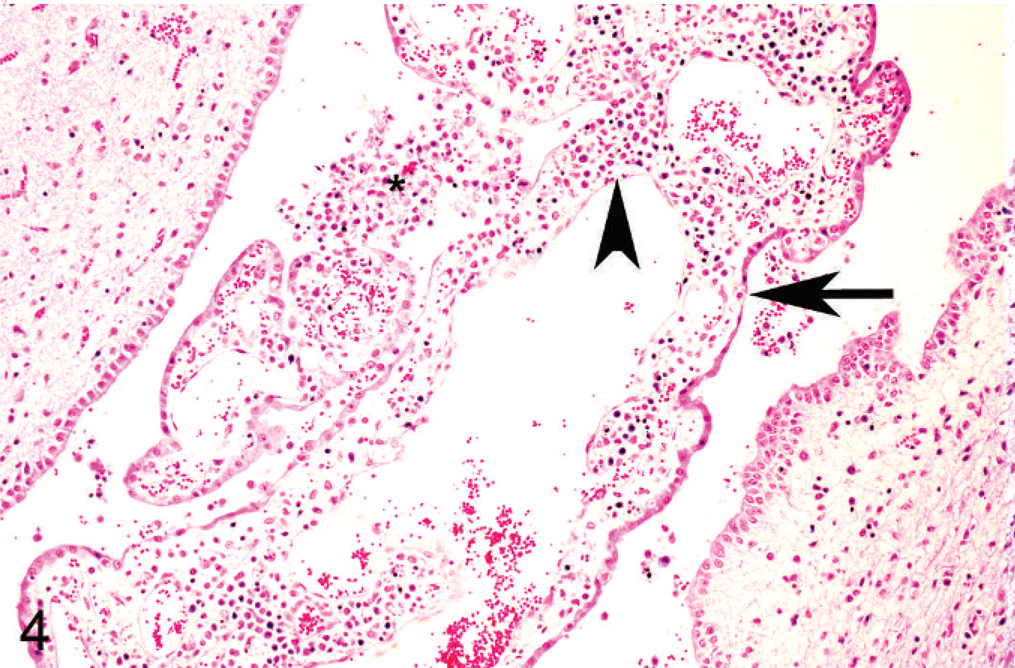
Cerebrum, goat. The stroma of the choroid plexus is diffusely infiltrated with lymphocytes, macrophages, and plasma cells (arrowhead) admixed with necrotic cellular debris. Inflammatory cells are also present within the ventricle (asterisk). The epithelial lining is multifocally attenuated (arrow). HE stain.
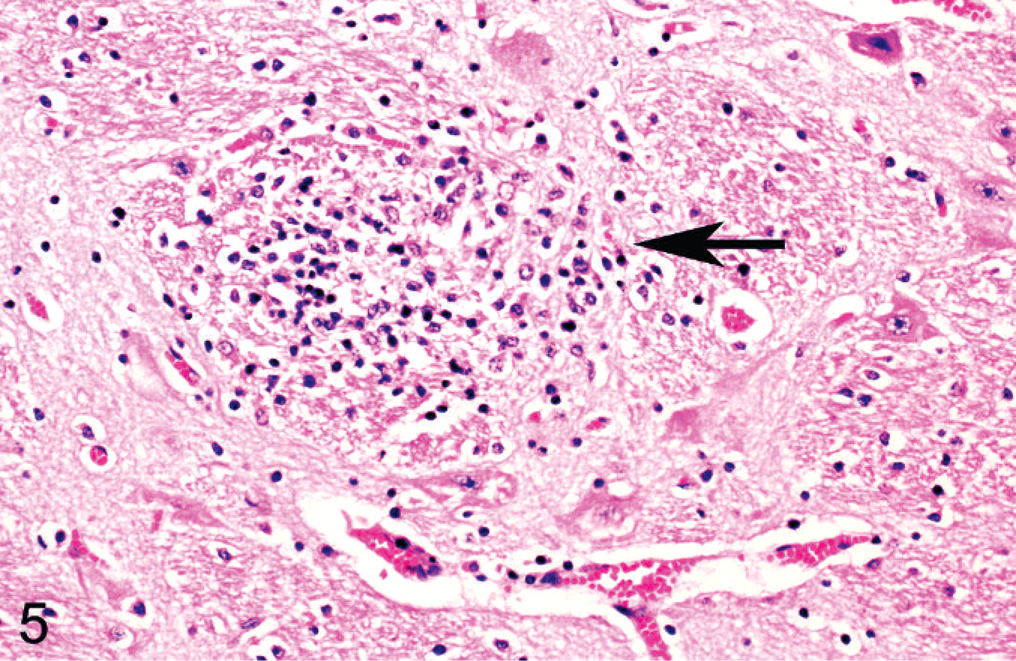
Cerebral cortex, goat. There is a focal glial nodule (arrow). HE stain.
Results of immunologic procedures, PCR, and virus isolation are provided in Table 3. Immunohistochemistry in the placenta, heart, brain, or thymus was positive in 12 of 19 of the animals examined. Immunohistochemistry findings are further described in the companion paper to this manuscript by Lamm, et al. 13 Virus isolation was positive in 2 cases, and each isolation occurred in kids that were born alive but were very weak and died within 1 to 2 days. Bovine viral diarrhea virus type 2 PCR of fetal tissues was positive in 12 of the 29 cases in kids whose dams were exposed at 60, 82, 118, and 139 days of gestation. The tissues that tested positive for BVDV-2 by PCR included the placenta, heart, brain, and thymus. The PCR results confirmed the presence of the same BVDV-2 in the infected fetal tissues as in the PI heifers.
Immunohistochemistry (IHC), fluorescent antibody (FA), polymerase chain reaction (PCR), and virus isolation (VI) findings in fetuses and kids from 12 does exposed to bovine viral diarrhea virus (BVDV) persistently infected (PI) heifers.
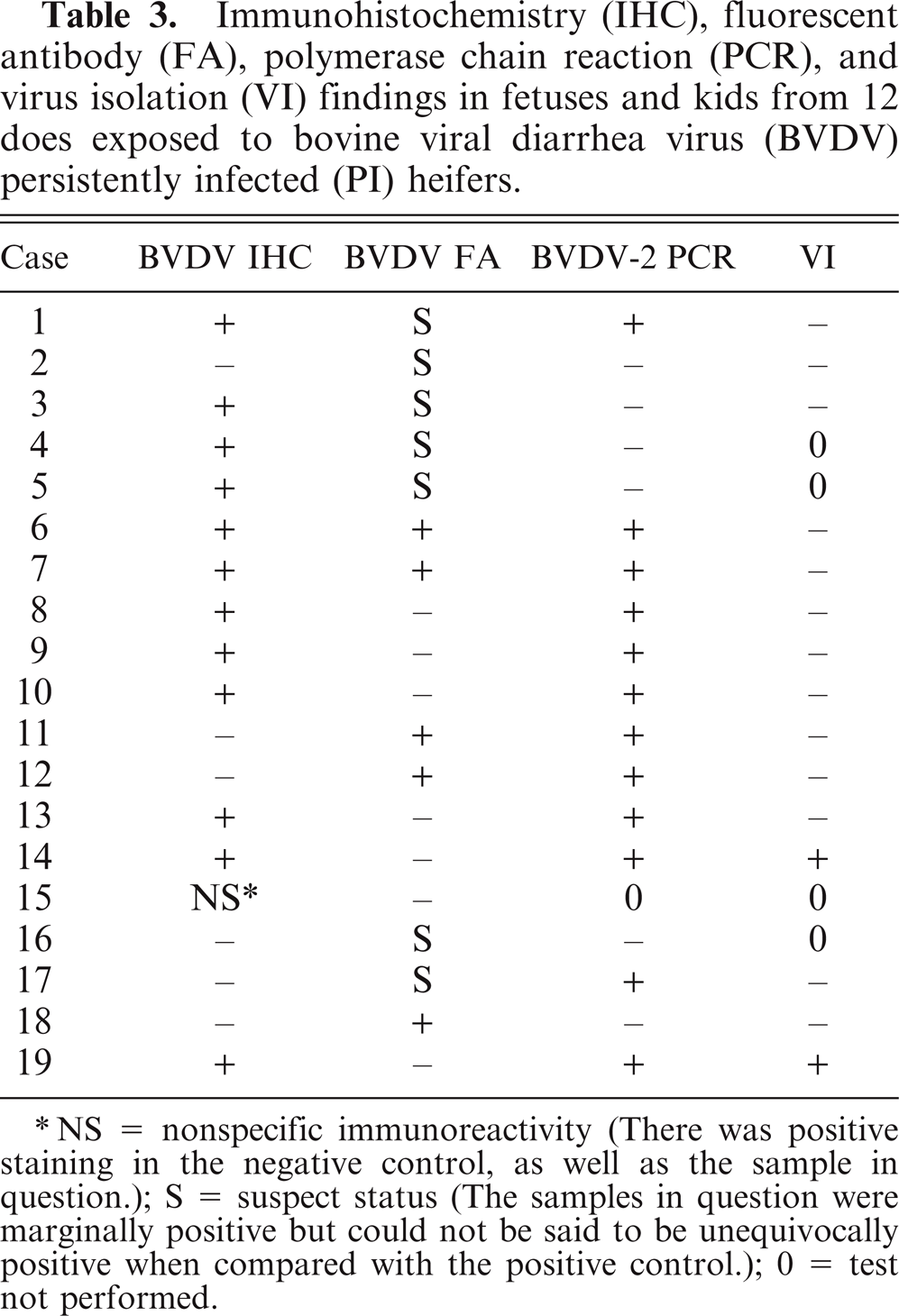
NS = nonspecific immunoreactivity (There was positive staining in the negative control, as well as the sample in question.); S = suspect status (The samples in question were marginally positive but could not be said to be unequivocally positive when compared with the positive control.); 0 = test not performed.
All fetal tissues were negative for C. fetus, C. jejuni, bluetongue virus, caprine herpesvirus, and chlamydia. Neither Toxoplasma nor Neospora were detected by IHC of appropriate tissues. Coxiella burnetti was ruled out based on the lack of intratrophoblastic organisms on histologic examination. Leptospirosis was not consistent with the lesions seen in the fetal tissues. Border disease virus was ruled out based on the viral isolation negative status of all animals at the beginning and end of the study.
Discussion
The overall abortion rate of 50% found in this study is especially significant because it is the result of natural exposure of pregnant goats to PI heifers. Several studies have demonstrated that pregnant goats will experience widespread abortions after inoculation with BVDV, and abortions, stillbirths, and persistently infected lambs result from experimental inoculation of pregnant sheep. 4, 15, 20, 21 A recent report documented the establishment of a PI white-tailed deer after intranasal inoculation of BVDV-1 and BVDV-2. 18 Although the inoculation and subsequent fetal lesions of these animals demonstrate the pathogenicity of BVDV to the goat, sheep, and deer fetus, it had been previously unclear if natural transmission of the virus from cattle to goats can result in similar fetal lesions. The present study has shown that a naturally occurring strain of BVDV-2a in PI heifers may be transmitted under field conditions to pregnant goats, resulting in abortions.
The isolate from the aborted fetuses was identified as a pestivirus by rt-PCR and confirmed as BVDV and not BDV using the nested multiplex PCR assay. Additional factors supporting the assertion that the pestivirus detected by PCR was BVDV and not BDV included the fact that BDV has not been isolated in Oklahoma, abortions in goats due to BDV have not been reported in North America, and the virus detected was the same genotype as that identified in the PI heifers.
Presuckle serum samples were obtained from kids when possible to determine if the fetus mounted an immune response to BVDV in utero. Immune competence in the goat against pestivirus is thought to develop between 80 and 100 days of gestation. 15 The serologic findings described in this study indicate that the kids born to does exposed at 82 days were not born with antibodies to BVDV in 3 of 4 cases, thus suggesting that infection occurred before the development of immunocompetence in some cases. The positive titers of 2 of 3 kids born to does exposed at 97 days of gestation indicate that these kids were immunocompetent at the time of infection. As noted above, however, without a known time of infection, we cannot correlate the development of immunocompetence with the presuckle BVDV antibody titers.
One very interesting aspect of BVDV infection in goats is the potential development of a PI kid. Based on the results in this study, the development of a PI kid seems to be much less likely than fetal death and subsequent abortion. However, in 1 kid, IHC testing was positive for BVDV antigen on ear notch skin obtained at birth.
The tissue most commonly supporting a positive diagnosis of BVDV in the fetuses was the placenta. In several cases, the placenta was positive for BVDV while the fetal tissues were negative for the presence of BVDV antigen. This finding supports earlier experimental work that demonstrated that the chorioallantois and amnion (but not the fetus) becomes infected as soon as 72 hours after intranasal inoculation of sheep with pestivirus, whereas the fetus may not become infected until 10 days post inoculation.
Infection with BVDV is rarely reported in goats. In a previous study, 25 goats were experimentally inoculated (either intranasally or subcutaneously) with BVDV (unspecified genotype) isolates at different stages of gestation. Of the 25 goats inoculated, 22 aborted or gave birth to stillborn kids. 4 The most prevalent lesions associated with these cases was leucoencephalomalacia.22 In a second study, 50 pregnant goats were experimentally inoculated intramuscularly with a noncytopathic ovine pestivirus or a cytopathic bovine pestivirus (both of unspecified species). 15 In 23 progeny, histologic changes were observed mainly in the central nervous system and consisted of “cerebral white matter necrosis, cerebellar dysplasia, hypercellular areas in white matter, and lymphocytic perivascular cuffing.” 15 A case report of natural pestiviral infections (of unspecified type) of 2 lambs and 2 kids described findings of areas of hypomyelination in the brain and a slight depletion of thymic medullary lymphocytes associated with an increase in reticular cells. 19
In this study, encephalitis, choroid plexitis, and gliosis were noted, as have been reported previously. On the other hand, we did not observe cerebellar dysplasia and malacia, as noted in other studies. In addition to the cerebral lesions, myocarditis, thymic depletion, and placentitis were also noted histologically in this study, and facial deformities were observed grossly. These changes have not been reported before in goats infected with BVDV. These gross and histopatholog differences could be related to the route of infection, the strain of BVDV, the dose of BVDV inoculated, or differences in susceptibility of different breeds of goats.
Viral isolation on pooled samples of nasal secretion and PCR testing of pooled sera were negative at days 6 to 8, indicating that either the virus was not present in the samples or pooling limited the test sensitivity. Nevertheless, all does did seroconvert by 42 days after exposure to the PI heifers, indicating that each doe did become infected. Because the exact time of the virus exposure is not known, it is not possible to correlate the pathologic findings with infection at a specific time of gestation. However, it is important to note that abortions resulted when does were introduced at all periods of gestation except for those introduced at day 139. These results indicate that exposure of goats at less than 139 days of gestation to BVDV PI calves can result in abortion rates of approximately 50%.
Because of cost and facilities considerations, a negative control group of pregnant goats housed with non-BVDV PI infected heifers was not utilized. Although a control group demonstrating that the presence of heifers with pregnant goats in a similarly sized pen does not cause spontaneous abortions would strengthen the conviction that the abortions in this study were due to transmission of BVDV-2 from the heifers to the goats, widespread copasturing of goats and cattle in many parts of the world without abortions along with the lack of other pathogens detected in this study suggests that abortions in these goats are not caused by stresses associated with the presence of cattle or by infectious agents other than BVDV.
Based on the findings in this study, BVDV infection should be a differential diagnosis for reproductive losses in goats cohabitating with cattle. This report is the first to describe natural transmission of BVDV-2a from PI cattle to seronegative pregnant goats with subsequent abortion. The PCR findings indicate that the same BVDV-2a virus that was present in the PI heifers was present in 12 of the 29 fetal tissues examined. In conjunction with the BVDV-2 serology data indicating BVDV-2 infection in the pregnant does, finding the same viral genotype in the aborted fetal tissues and the PI heifers provides strong support that the abortions were caused by BVDV-2a infection of the pregnant goats. In addition, many other potential causes of abortion in goats were ruled out. In particular, BDV is ruled out because the PCR and serology findings were specific for BVDV-2a and BDV was not isolated. However, because of the lack of controls in this study, the results cannot totally exclude the possibility that unmeasured or unknown factors, such as stress and unidentified infectious agents, may have caused or contributed to the abortions.
Footnotes
Acknowledgements
This work was supported by grants from the Department of Clinical Sciences, Center for Veterinary Health Sciences, Oklahoma State University, Stillwater, Oklahoma; Oklahoma Animal Disease Diagnostic Laboratory; and the Noble Foundation, Oklahoma State University. The authors thank Mr. Erick Loetz and the American Institute for Goat Research, Langston University, Langston, Oklahoma, for providing the goats used in this study and Mr. A. C. Box of the Oklahoma State University Center for Veterinary Health Sciences Ranch for providing daily care of the animals. Drs. Tony Confer, Bill Johnson, and Charles MacAllister were instrumental in facilitating financial support for this project. Dr. Mark Payton kindly assisted with statistical analysis.
