Abstract

Over the past 10 years, pharmaceutical companies have found compelling reasons to use human tissue in drug discovery and development. In preclinical work, human tissue enables researchers to select and validate drug targets based on human-disease phenotypes, find inherited gene variation that contribute to disease, validate animal disease models, and reclassify disease based on molecular signature. Human tissue allows researchers to overcome some of the inherent disadvantages of animal disease models, which are often poor predictors of similar disease states in man. Also, therapeutic interventions in these animal models often do not have the same pharmacologic results seen in human clinical trials.
In clinical research, use of human tissue helps medical teams segment patient populations based on biomarker profiles and thereby better target new medicines for greater patient safety and efficacy. Patients with similar clinical profiles can have different tissue pathology and disease etiology, therefore, combining standard clinical evaluations with assessment of variations in genetic expression or sequence provides clinicians with critical tools to improve patient care.
Given the benefits of human tissue, many pharmaceutical companies and academic research centers are motivated to increase its use in target validation and early drug development but encounter a multitude of hurdles in the process. Use of human tissue requires rigorous patient confidentiality and data-security measures, because tissue could be used to reconstruct a patient's genome, with potential implications for compromising privacy, insurability, and job security. 2, 3, 18, 25 This article discusses the regulatory, pathology, information technology, and administrative issues involved in establishing, managing, and using pathologic samples from surgical specimens in a tissue bank. 22
Understanding Governmental and Professional Requirements
In 1993, the US Food and Drug Administration (FDA) began to regulate tissue banking with the publication of “Interim Rule for Human Tissue Intended for Transplantation.” In responding to the growing risk of transmission of hepatitis and human immunodeficiency virus related disease through handling of human tissue, the publication focuses on testing for infectious diseases, screening donors, and maintaining written records. The document also requires written procedures for all steps in the testing process, as well as assessment and documentation of the medical history of the donor. After the FDA began to regulate tissue banking, many states wrote additional regulations. 2, 9
In the mid 1990s, the National Institutes of Health and Centers for Disease Control convened workshops to examine consent for genetic research on both DNA and tissue. The resulting report recommends restriction of access to archival clinical specimens for molecular genetic research, including frozen blood, frozen tissue samples, stored DNA, and paraffin-embedded tissue blocks. The report states that research on stored samples should not proceed without first obtaining informed consent from the patient or, if deceased, then the patient's relatives. For newly collected samples, the requirements are even more stringent. Patients must be given a multitiered consent form with numerous options and permutations. Opposing responses by the College of American Pathologists, the American Society of Human Genetics, and the Association for Molecular Pathology are still being circulated. 3, 5, 7, 10, 14, 18, 20
The College of American Pathologists met in January 1996 to discuss the use of human tissue in research and produced a draft document that defined genetic testing, genetic research, confidentiality, and research uses. This document was revised in August 1997 and concludes that pathologists, as recipients of tissue and medical specimens, should consider themselves stewards of patient tissue whose duty is to protect the interests of the individual patient. This stewardship of frozen tissue, paraffin blocks, glass slides, and other material includes providing tissue only to qualified researchers who intend to use the tissue to affect advances in medical sciences. The decision to provide human tissue should be based on the specific and general interests of the patient and of society. 1, 4, 8, 10, 14, 16, 17, 19, 20
Tissue banking requirements of patient confidentiality, informed consent, and local institutional review board (IRB) review find their beginnings in the Holocaust, which led to the creation of the Nuremberg Code and the Declaration of Helsinki. The Health Insurance Portability and Accountability Act's (HIPAA) final modifications to the Privacy Rules went into effect in April 2003, and they have added to the statutory and regulatory requirements in the United States. By reiterating that patients have a right to informed consent whenever a medical treatment or surgery puts the patient at risk, HIPAA also stresses that patients have a right to privacy with respect to information related to their health and medical care.
Sourcing human tissue
Pharmaceutical companies should begin the process of human tissue use in early discovery research by selecting and coordinating with a tissue source. Companies may purchase tissue from a vendor that specializes in tissue (e.g., Asterand, Cooperative Human Tissue Network, National Disease Research Interchange, and/or Ontario Tissue Bank) or enter into a customized agreement with a hospital or consortium of hospitals. 12, 15 Although there are advantages and disadvantages to each route, this article focused on the latter.
If the research community chooses the customized route, one of the first tasks after signing an agreement is to secure the trust and cooperation of scores of people who are not employed by the project but who are critical to its overall operation. These include hospital surgeons, admitting nurses, surgical nurses, and administration. Crucial to building trust with these constituencies is assuring them that 1) tissue will be used for legitimate research purposes and 2) patient confidentiality will be handled with the utmost rigor. Offering research seminars on the pharmacogenomic uses and potential of the tissue bank and explaining how cooperation can help develop new therapies for patients are valuable factors to building and sustaining the confidence of these groups.
Finding the right people to develop, gain approval, coordinate, and administer patient consent is paramount to a successful effort. The hospital's attorneys, the scientific review committee, local HIPAA compliance committee, and local IRB must review and approve the consent form. The consent document is then reviewed annually by each committee and reapproved whenever changes are required. 2, 9, 18
Ensuring patient privacy in an informatics system
To protect patient privacy and tissue data, appropriate technology must either be purchased or developed. In either case, tissue specimens and clinical data are uniquely recorded, and no personal identifying information that pertains to the consenting tissue donor is stored by the informatics system. 24, 25 Our approach to this conundrum was an informatics system that required the tissue-sourcing organization to establish an intermediate patient identifier that bore no relationship either to the patient or to associated hospital records. This intermediate identifier used when enrolling the patient is not stored in its original form. Instead, it is unidirectionally encrypted to yield a unique result that is consistently reproducible. This ensures that every patient is anonymized but still uniquely identified within the system. The unidirectional encryption process dictates much of the design of the informatics system. For example, patients who have given consent can be enrolled and follow-up information appended only as long as the unidirectional encryption process is maintained as part of the system. 26
A modern cryptography-engineered solution was designed to provide sufficient security to counter escalating computing power, mathematical advances, and genetic computing developments, as well as ensuring one-way encryption. Message digests can provide the nonreversible nature, but standard configurations of related algorithms provide inadequate security. There are high security matrix techniques, 5 but these are expensive and not readily available. Lilly's Clinically Annotated Tissue Databank (CATD) system, used for several tissue banks, uses a proprietary Patient Encryption Service that achieves security levels millions of times more secure than typical applications and theoretically would take millions of years of supercomputer time to break. Such extreme security is appropriate to assure long-term patient confidentiality in the face of advancing technologies. 26
Designing other data-security features
An informatics system for leveraging human-tissue research can store tissue descriptors, experimental results, and clinical annotations about the patient, disease pathology, and therapeutic outcomes. The system can facilitate data mining to show correlations between pathology profiles, target profiles, and outcomes. Access to the data can be restricted to qualified, approved researchers. Experimental results collected in the information system may include expression data generated from specimens, pathology images, and conventional alphanumeric data. CATD is one of the databases attached to the tissue bank accessible by Indiana University (IU) (see Fig. 1).
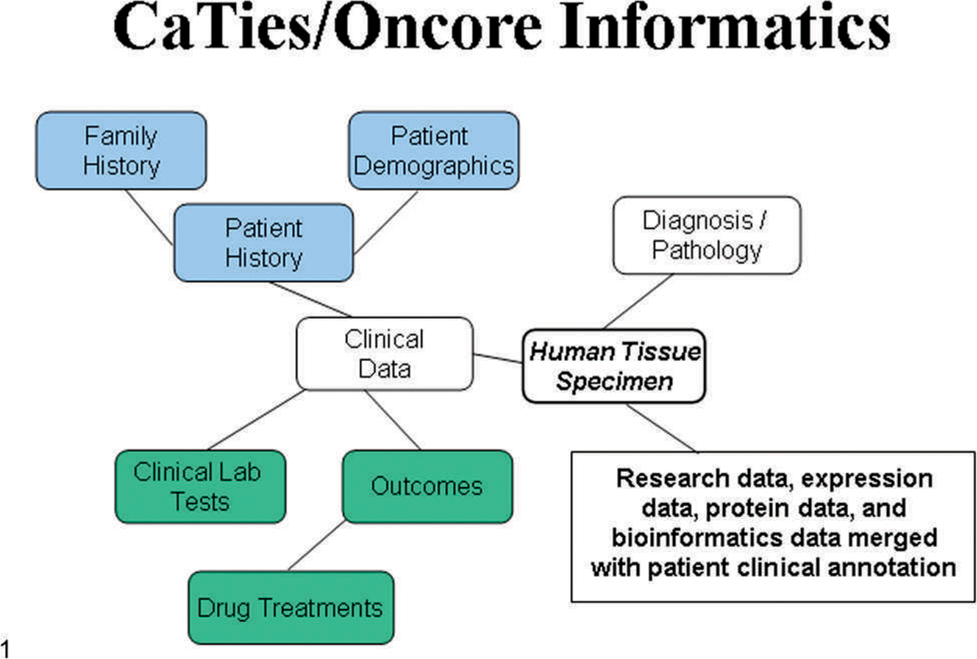
Illustration of the informatics database surrounding the IU tissue bank. The white coded boxes are Oncore/Biospecimen Management and/or CATD databases, which are currently operational. The blue and green boxes are tied to the Oncore/Biospecimen Management via CaTIES, an NCI CaBig module, which is part of CaBig (the NCI Cancer Biomedical Informatics Grid). The research data will be stored in and tied to the rest by a CaBig module called ca Tissue Experimental Annotation Module.
We found that a good informatics system can provide a flexible means of controlled data sharing. Scientists select an experiment definition from predefined types and attach 4 different levels of sharing security. The security levels range from viewing to the originating scientist only, with a description of the experiment's existence but not its content, to sharing an experiment and results with other IU and Lilly scientists. In some cases, the sourcing organization adds experimental data, which can be similarly restricted to viewing by the researcher only to sharing between Lilly and the outside scientist. A scientist can change the security level of any experiment he or she owns at any time, and the resulting setting with be handled by the security algorithm controlling the display of experimental results. 26
Handling and storing tissue specimens
The success of molecular research and its applications in both the clinical and research arenas depend on appropriate handling and storage of tissue samples.∗ The flow scheme currently used by IU for collection and handling of tissue samples is outlined in Fig. 2. This protocol enables the laboratory to collect and store both snap-frozen and formalin-fixed tissue in an organized and efficient manner.
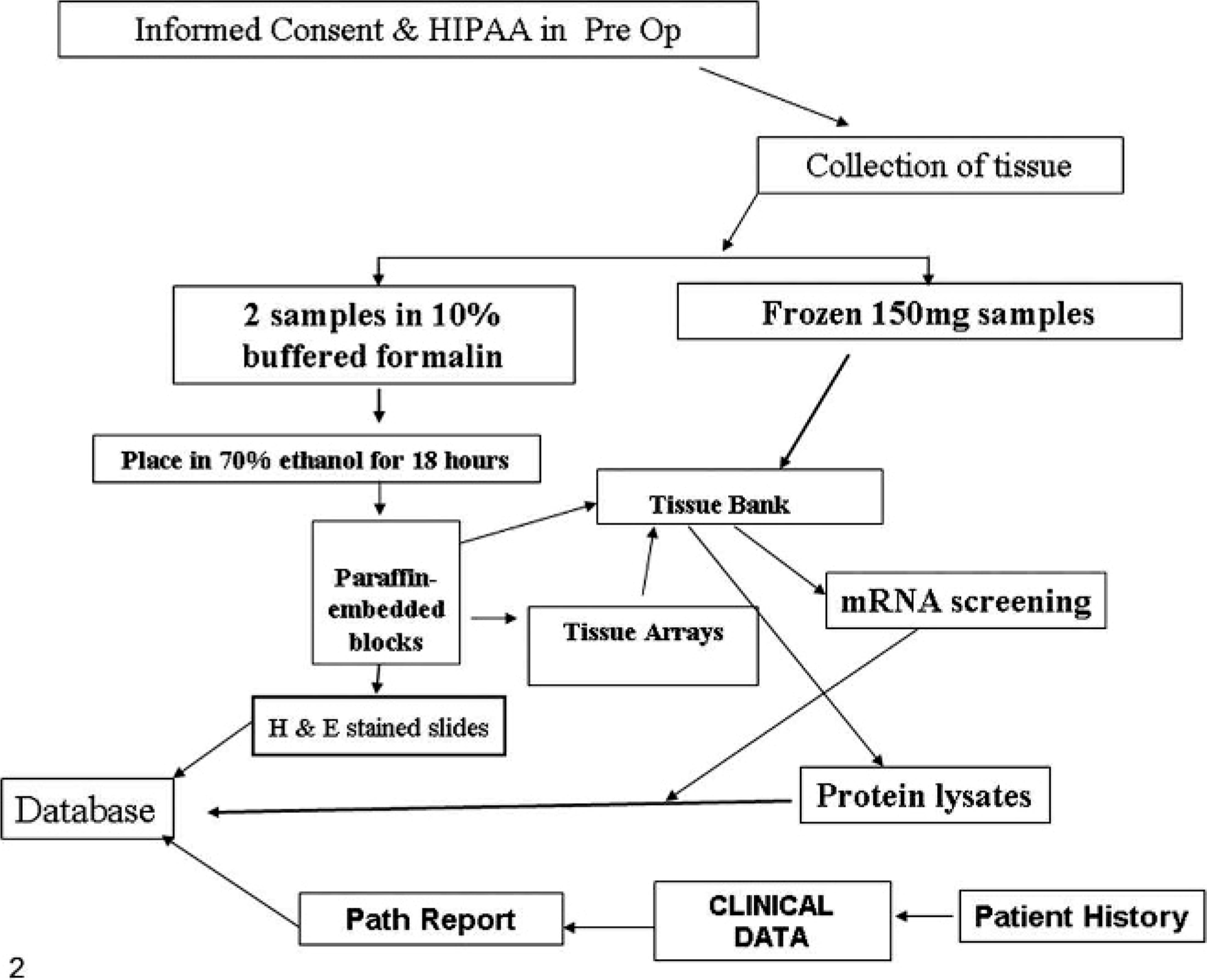
Flow scheme of the IU Simon Cancer Center Tissue Bank from informed consent, tissue collection and storage, and clinical data capture.
For decades the process of formalin-fixing, paraffin embedding (FFPE) has been the method of choice for tissue preservation, because this technique best maintains the morphology of the original tissue. Hence, FFPE tissues represent the most abundant supply of archival material for clinical and molecular analyses. However, with the advent of new molecular techniques that require relatively unaltered nucleic acids and proteins, particularly those that rely on oligo-dT priming for cDNA synthesis, frozen specimens are generally preferred.
Prefixation time (the time between tissue dissection and freezing or fixation) is a major factor that influences the integrity of banked samples. To avoid tissue autolysis and digestion of nucleic acids by endogenous nucleases, it is useful to establish guidelines that will help keep the prefixation times as short as possible. For example, our guidelines for the IU tissue bank suggest a maximum of 45 minutes ischemic time (the time between cessation of blood flow and preservation of the specimen). The database contains a check box to indicate whether samples were collected within this time frame. On average, approximately 80 to 85% of samples in the bank are harvested within 30 minutes, whereas 90% of the samples are collected within the 45-minute time window. Also, to discourage nucleic-acid degradation, archived frozen tissue should be homogenized in a denaturing solution, such as TRIzol, immediately upon removal from storage. 32, 33 No matter the type of specimen used (FFPE or fresh frozen), it is important to implement both histologic and molecular quality-control processes for examination of sample integrity. This will help ensure the quality and reproducibility of the data generated from banked samples.
Histology Quality Control of the Samples
The routine diagnosis and quality of the samples in a tissue bank are typically supervised by a pathologist. Through histologic review of banked specimens, the pathologist can determine that the tissue collected and stored represents the disease that the researchers are studying and that normal human tissue being stored is, in fact, normal. IU uses the following procedures to ensure adequate histologic quality control of each of the specimens in its sample bank. Before use, each tissue sample requested from the bank is evaluated by hematoxylin and eosin. Tumor samples that contain at least 65% tumor are released to the tissue requestor for molecular work, whereas tumor samples with less than 50% tumor are released only for immunohistochemistry (IHC) and/or fluorescence in situ hybridization (FISH). Normal adjacent tissue samples that contain tumor are also released only for IHC or FISH applications, in which evaluation (and distinction) of both normal and tumor tissue can be performed in the same section.
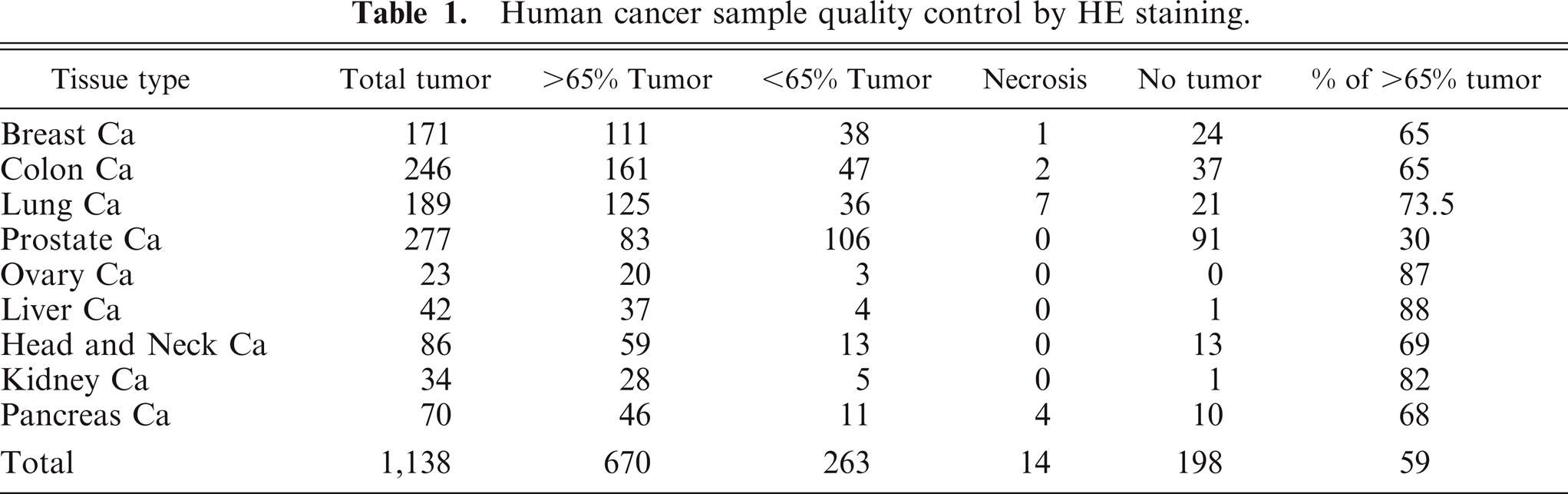
Of 1,138 total samples in the IU tissue bank (Table 1), only 59% were found to contain 65% or more of tumor, whereas the remaining specimens contained less than 65% (23% of samples), were entirely negative for tumor (17% of samples), or were completely necrotic (1% of samples). Prostate samples, in particular, had a small percentage of samples suitable to release for molecular analyses. These samples were collected as sequential punches taken from the whole prostate gland by using the Baylor protocol. However, because prostate cancer is detected very early and the tumors tend to be localized to one area of the gland, 8 of 9 prostate cores are often negative for tumor. These findings underscore the importance of instituting adequate measures for histologic sample quality control before the release of banked samples for downstream testing.
Human cancer sample quality control by HE staining.
Molecular Quality Control of the Tissue-Bank Samples
Unfortunately, histologic quality of a sample does not guarantee the quality of the nucleic acids in that sample. Therefore, it is also important to evaluate the molecular quality of each sample before its release for subsequent molecular testing. In particular, archival FFPE samples are notorious for yielding poor-quality nucleic acids, largely because of the handling practices used for collection of these specimens before the era of whole transcriptome analysis. 33 Because the quality of the input nucleic acids can greatly influence the reliability and reproducibility of a molecular test, the assessment of RNA and/or DNA quality and integrity should be a prerequisite for predicting the potential usefulness of existing FFPE material for downstream molecular studies. The Agilent Bioanalyzer is a useful tool for evaluation of nucleic-acid quality. This microfluidics-based platform allows, for example, total RNA integrity to be assessed through visualization of the behavior of the 28S and 18S ribosomal RNAs on either an electropherogram or a virtual gel. A 28S : 18S ratio of 2.0 is ideal and denotes high-quality total RNA, whereas a ratio of 1.4 to 1.8 is generally acceptable. 6, 13
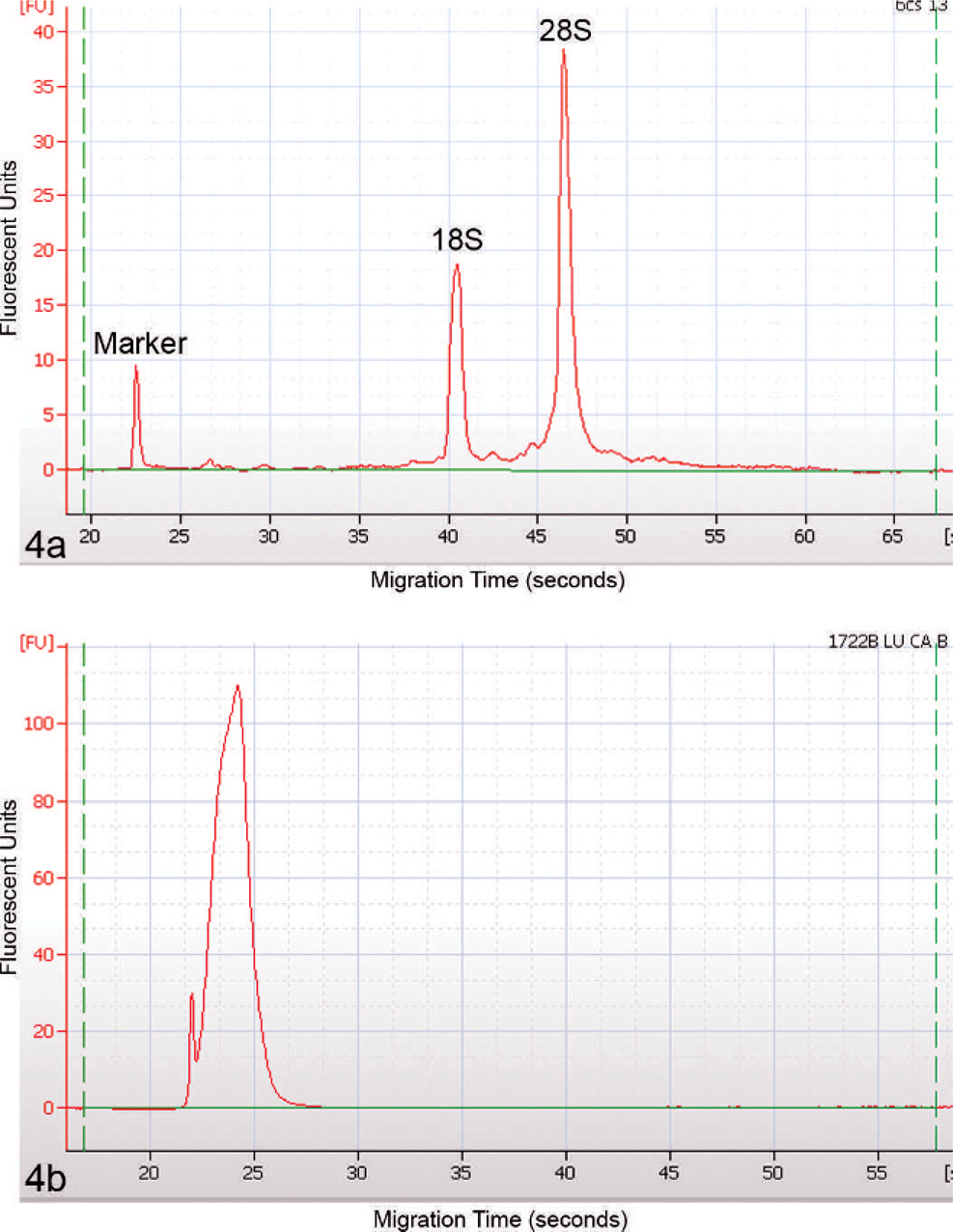
To demonstrate the importance of implementing processes for molecular quality control, particularly in archival specimens, 2 subsets of tumor and normal adjacent tissue from the IU tissue bank were evaluated for molecular quality. First, we compared the integrity of RNA derived from tumor tissues that had been stored frozen for less than 18 months to that of tumor tissue that had been stored frozen since the inception of the bank (8–9 years prior). Both sets of samples had been stored in a −80°C freezer for the indicated amounts of time. The Agilent Bioanalyzer was used to examine the quality of the RNA derived from a total of 60 samples from the shorter storage time (<18 months) and 122 samples from the longer storage period (>8 years). High-quality RNA was found in all clinical samples examined that had been stored for less than 18 months. However, for the long-term storage samples, only 59 of 122 samples (48%) were defined as being of high enough quality for subsequent molecular work (see examples in Fig. 3).

Total RNA derived from prostate cancer samples that had been stored at −80°C for either fewer than 18 months (left) or 8–9 years (right) was run on the Agilent Bioanalyzer. The presence of intact 28S and 18S ribosomal bands illustrates the high quality of RNA derived from each of the short-term storage samples, whereas the samples stored over a longer period of time displayed varied levels of degradation.
In addition to examining archival frozen tissues, we also evaluated total RNA quality in 70 FFPE blocks that had been stored for between 8 and 9 years. Samples consisted of 37 tumor and 33 normal adjacent tissues of various tissue origin. Although spectrophotometric analysis determined the RNA yields of most samples to be sufficient for downstream testing, Agilent Bioanalyzer analysis revealed that each of the RNA samples were highly degraded and consisted solely of low-molecular-weight species of fewer than 200 base pairs (bp). Fragment size was determined by overlaying the sample electropherogram on that of an RNA 6000 ladder (Applied Biosystems/Ambion, Austin, TX, USA) (see Fig. 4b). In a similar experiment, von Ahlfen et al. 33 found that RNA isolated from FFPE specimens within 1 to 3 days after paraffin embedding was largely intact, whereas RNA isolated from paraffin blocks stored for even just 1 year at room temperature lacked distinct RNA bands.
The fragmentation state of total RNA derived from archival FFPE tissue blocks was evaluated by using the Agilent Bioanalyzer. The electropherogram in
Because even moderate RNA degradation was shown to decrease data quality on microarrays and severely degraded RNA demonstrated low overall data quality and poor correlation with intact sample counterparts, 28, 32 stringent procedures for sample quality management should be in place and specific acceptance criteria should be met before banked samples are released for subsequent testing.
Managing requests for tissue
In working with tissue banking, IU has discovered that establishing a rigorous process for managing tissue requests both promotes good science and facilitates the operation. The following list illustrates what a pharmaceutical company sponsor can require of those working with human tissue. For example, the pharmaceutical company can
Ask scientists to demonstrate knowledge of human subjects' protection regulations and practices 8
Establish a peer review of the scientific goals and methods for research uses of the tissue
Ensure that scientists complete biohazard safety training for handling human tissue or biologic fluid
Require that everyone in the laboratory who is working with tissue or biologic fluids gets an up-to-date vaccination against hepatitis B
A jointly managed tissue bank may institute a standing governing board, which represents the pharmaceutical company and the sourcing organization that approves tissue requests and ensures a fair balance of tissue allocation. 25, 31
Motivating tissue use
Once tissue collection starts, the pharmaceutical company needs to find ways to motivate tissue use so that the organization incorporates pharmacogenomics data and tissue tools appropriately and in a way that offers the greatest potential for developing new patient therapies. 4, 16, 21 Peer education, which is critical to initial acceptance, depends on recruiting early adopters to present to staff meetings on how to use specimens in actual experimental situations, by emphasizing the results obtained and ways that tissue use afforded them results difficult or impossible to obtain in more conventional experimentation. It is important at this stage to stress that tissue procurement and handling follow rigorous regulatory practices and that using tissue is both reasonably streamlined and productive.
As an organization gains more access to clinically annotated tissue, management and governance committees can motivate use by setting expectations. For example, a key discovery committee might require pharmacogenomic experiments as part of target validation. Management at all stages of pharmaceutical development can work with scientists and clinicians to incorporate pharmacogenomic objectives into annual performance management plans and drug-development life-cycle plans.
Also, any successful tissue-banking effort depends on 5 key roles at the pharmaceutical company. An executive sponsor acts as an advocate of the potential of pharmacogenomics in all phases of research and works cross functionally and cross organizationally to support and implement the project. A scientific leader educates peer scientists about the potential of the tissue bank, mentors other scientists in its application, and organizes and chairs meetings between the pharmaceutical and sourcing organizations. A project manager takes the lead on developing timelines, designing governance structures, and helping the organizations involved in the effort work together effectively. Functional leads, including systems experts, help to develop and implement the project plans. Regulatory scientists help to guide the project so that it meets all regulations and guidances. Some of these roles, depending on the scientist's expertise, may be combined in one individual.
Conclusions
Today's convergence of pharmaceutical research with genetic research and biomarkers to categorize disease states provides a unique opportunity to both understand and directly engineer interventions into human disease. The use of human tissue in early pharmaceutical research, humanizing drug discovery, can help select and validate drug targets, find genetic variation contributing to disease, accelerate progress in designing clinical trials for biomarkers, and focus patient selection in clinical trials. Knowing and implementing effective regulatory, pathology, informatics, and management systems can together make possible even greater progress in alleviating human disease and improving the quality of life.
Extra resources
U.S. Patent 6,219,421 B1 “Virtual Matrix Encryption (VME) and Virtual Key Cryptographic Method and Apparatus,” Shaul O. Backal, Apr 17, 2001.
One of the best sources for cryptology is Schneier B. Applied Cryptography: Protocols, Algorithms, and Source Code in C, 2nd ed. John Wiley & Sons, Hoboken, NJ; 1996.
Many institutions use the computer-based training on human subjects found on the National Institutes of Health Web site, http://nihtraining.com/ohsrsite/cbt/cbt.html.
Footnotes
Acknowledgements
We thank the IU School of Medicine Simon Cancer Center tissue banking staff for their work; Christina Beard for her contribution with gathering patient data and the informed consent process. We thank Carol S. Boyd, Dana Borneo, and Mary James for their continuing efforts to collect, process, and prepare the tissues for long-term storage and extraction for medical research studies; the following Lilly staff for the informatics work, Jeff C. Hanson and Rebecca Lynch, who helped to streamline this tissue banking process to enable it to flow smoothly.
