Abstract
We have observed a renal toxicity consistent with an obstructive protein cast nephropathy in cynomolgus macaques but not in other species treated with different therapeutic candidates having a common carboxylic acid moiety, suggesting a species-specific sensitivity. Here, we present renal toxicity findings consistent with a protein cast nephropathy in a 2–week safety study in cynomolgus monkeys. Light microscopic changes consisted of intratubular cast formation, tubular dilatation, interstitial inflammation, and expansion of the medullary interstitium. Tubular cast material was identified as Tamm-Horsfall protein (THP) and, on ultrastructure, crystalloid material was present in vacuoles of tubular epithelium. It is hypothesized that microcrystal formation in the urinary tubular spaces induces aggregation of THP protein and cast formation in monkeys. Drug-induced obstructive nephropathy is not identified as a major problem in humans; thus, the clinical relevance of the above findings in monkeys is not clear.
The kidney is a common target of xenobiotic-induced toxicity because of its high blood flow and ability to concentrate, metabolize, and excrete solutes. Injury to the kidney may be induced by a direct cytotoxic effect of parent compound/metabolite to tubular epithelial cells. 3 Another mechanism of injury involves accumulation or precipitation of drug material, causing tubular obstruction within the nephron. 5 Obstruction of the nephron may be exacerbated by the formation of tubular casts from endogenous urinary proteins such as Tamm-Horsfall protein (THP). 12, 13 This form of injury, commonly referred in the literature as obstructive nephropathy, has been reported in laboratory animal species treated with a variety of compounds, including biologics, 2, 4 nonsteroidal anti-inflammatories (NSAIDS), 7 and antithrombotics (fibans).
The carboxylic acid moiety is an important molecular component of major experimental as well as marketed drugs. The kidney plays an important role in the excretion and elimination of carboxylic acid drugs, as these are metabolized into more watersoluble compounds usually by glucuronidation. 10 Here, we describe a renal toxicity consistent with an obstructive protein cast nephropathy observed in a 2-week safety assessment study in cynomolgus monkeys. We had previously observed a similar renal toxicity in monkeys treated with other small molecule drug candidates. Although the test compounds were of different molecular structure and therapeutic indication, all shared a common carboxylic acid moiety.
Adult cynomolgus monkeys (Charles River) were orally dosed with a small organic carboxylate molecule (MW = 444 grams/mol) being developed for the treatment of osteoarthritis for 2 weeks at daily doses of 100, 500, and 2,000 mg/kg. Additionally, Sprague-Dawley rats were treated for 2 weeks up to 2,000 mg/kg/day. The following parameters were evaluated: life clinical observations, hematology, clinical biochemistry, urinalysis, organ weights, gross pathology, and histopathology. In addition, samples of renal cortex were collected from selected animals, trimmed into sections approximately 1–2 mm3, rinsed in phosphate buffered saline, postfixed in osmium tetroxide, processed, and embedded in blocks of epoxy resin. Selected blocks were “thin” sectioned, stained with uranyl acetate and lead citrate, and examined with a FEI/Philips BioTWIN 100 transmission electron microscope (Hillsboro, OR, USA). For immunohistochemical analysis, kidneys from selected animals were sectioned at 3 µm, placed on charged slides, and air dried. Deparaffinized slides were loaded onto the Nexes IHC Automated stainer (Ventana Medical Systems, Tucson, AZ, USA) for staining using a heat protocol. Slides were enzymatically digested with Protease 2 (Ventana Medical Systems) and then incubated with sheep anti-Tamm-Horsfall Glycoprotein antibody (Chemicon, Cat#AB733, Billerica, MA, USA) at a dilution of 1:20 for 32 minutes. The Ventana Basic DAB Kit (Ventana Medical Systems) was used to detect the antibody, with the exception of the biotinylated secondary antibody. Anti-sheep biotinylated secondary antibody (Vector Labs, Burlingame, CA, USA) was used at a dilution of 1:50 for 8 minutes in place of manufacturer's secondary antibody. Slides were counterstained with hematoxylin, washed, cleared in xylene and mounted with Permount.
All monkeys and rats treated for 2 weeks survived to study termination. There were no drug-related gross or clinical pathology findings. Histologically, kidneys of treated monkeys had minimal to mild multifocal mononuclear tubulointerstitial inflammation and epithelial cell degeneration/regeneration associated with the presence of intratubular, pale basophilic homogeneous material in the form of casts (Fig. 1). Tubular dilatation occurred with or without the presence of tubular casts in proximal/distal convoluted tubules and Bowman's capsule of the renal cortex and sometimes in tubules along medullary rays (Fig. 2). There was mild expansion of the interstitium in the corticomedullary junction and medulla consistent with edema. Lesions were dose dependent in incidence and were observed down to the lowest dose. No renal pathology was seen in Sprague-Dawley rats given this experimental drug for 2 weeks at doses as high as 2,000 mg/kg/day. Tubular cast material, distal convoluted tubules and thick ascending limbs were strongly positive for THP by immunohistochemistry (Fig. 3). Distal convoluted tubules and thick ascending limbs are known to produce THP and were used as an internal positive control to validate the specificity of the antibody. In addition, tubular cast material was periodic acid–Schiff positive. At the ultrastructural level, tubular cast material consisted of interlacing bundles of fine fibrillar material mixed with cellular debris. Clusters of slender, acicular, electron dense crystalloid material were identified within membrane bound intracytoplasmic vacuoles in proximal tubular epithelial cells (Fig. 4).
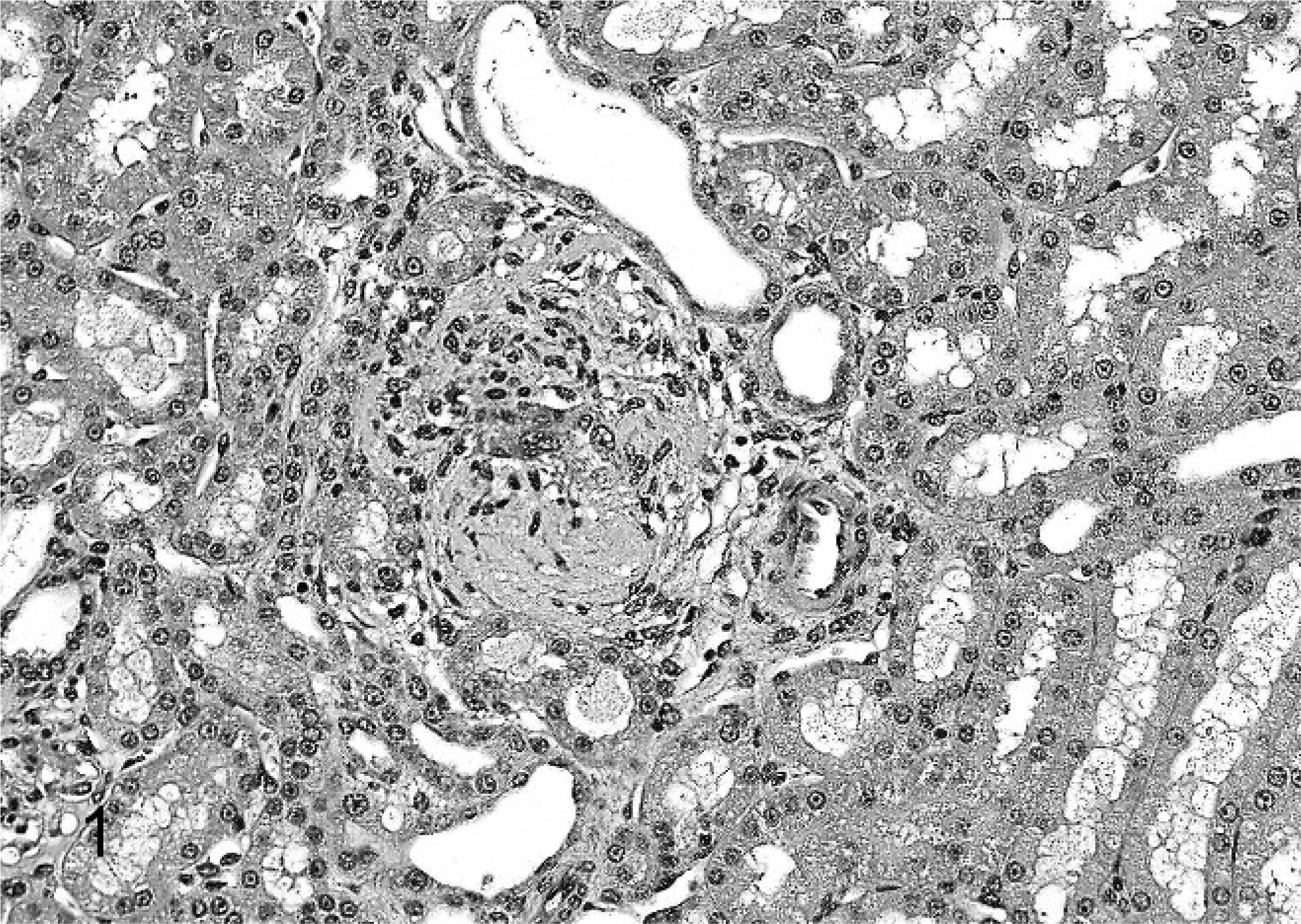
Renal cortex; monkey. Note tubular casts surrounded by a mononuclear inflammatory infiltrate. HE.
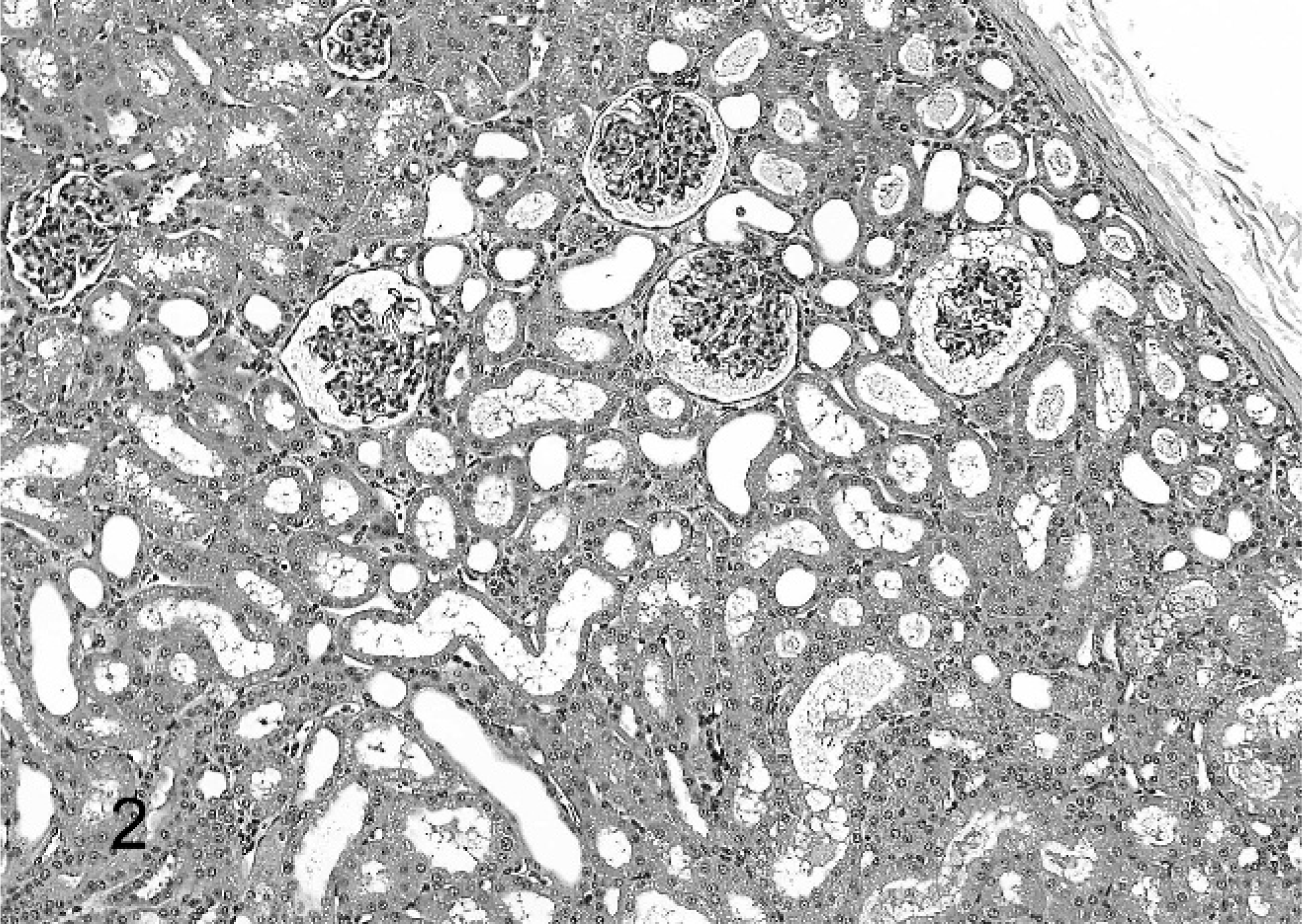
Renal cortex; monkey. There is dilatation of convoluted tubules, Bowman's capsule, and tubules along medullary rays. HE.
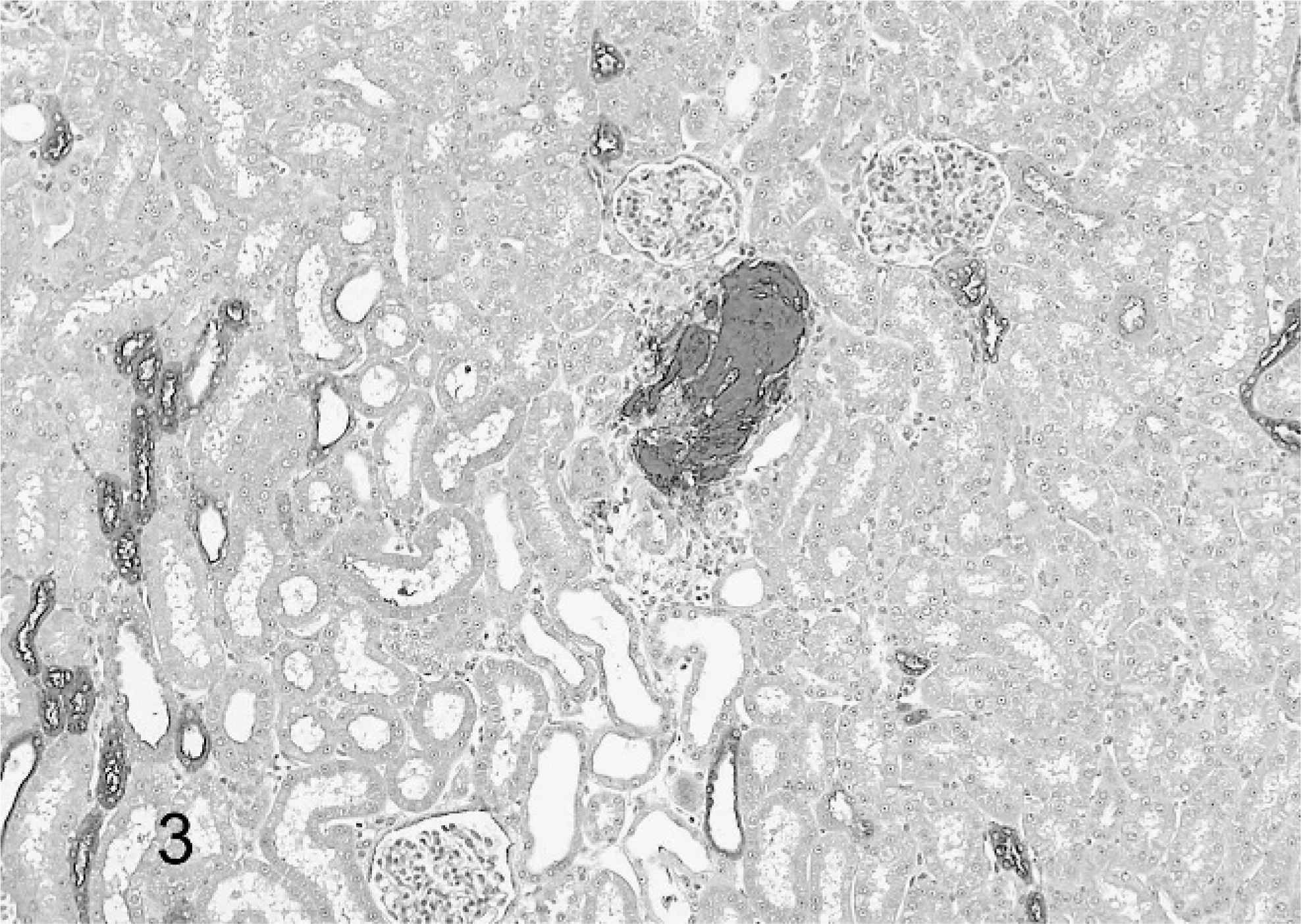
Renal cortex; monkey. Tubular cast material is strongly positive for THP by immunohistochemistry. Note that distal convoluted tubules and thick ascending limbs, which are known to express THP, also show strong staining and serve as an internal positive control.
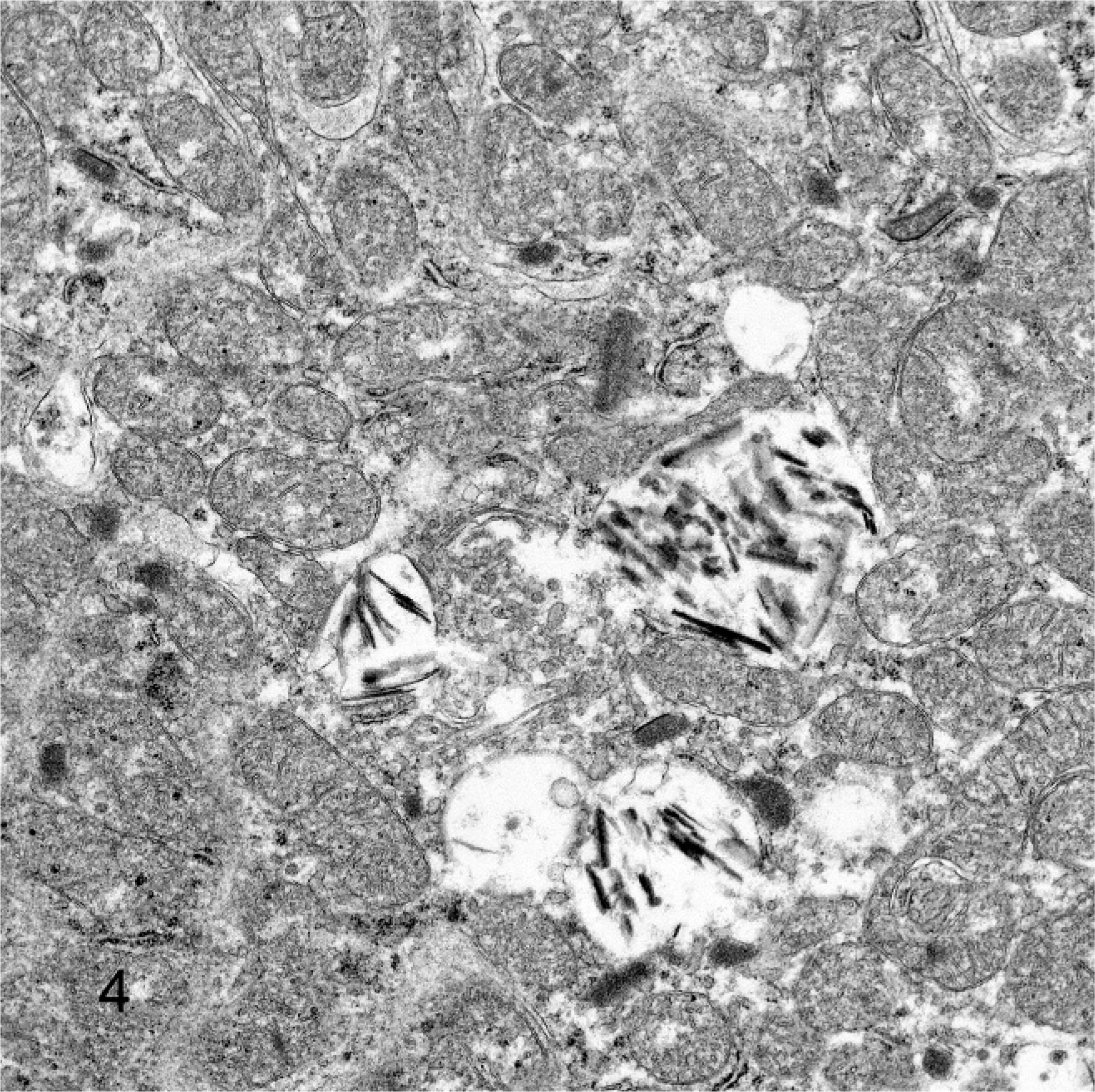
Proximal tubular epithelial cell. Note the presence of electron dense crystalloid material within membrane-bound intracytoplasmic vacuoles. Transmission electron microscopy.
Renal toxicity consistent with an obstructive nephropathy occurred in cynomolgus macaques but not in rats treated with a small molecule drugs. The toxicity was characterized by mild multifocal tubular degeneration/regeneration with interstitial inflammation associated with the presence of intratubular material identified as THP. We have observed similar morphologic changes in cynomolgus macaques but not in rats treated with a different therapeutic candidate. Both agents had in common a carboxylic acid moiety (data not presented).
Accumulation of parent drug and/or metabolites in the form of crystals is usually the most common form of renal tubular obstruction, especially in compounds that are poorly soluble. Renal changes in monkeys associated with administration of NSAIDs containing carboxylic acid, such as naproxen, are also shown to induce formation of casts with crystalline outlines that were presumably drug precipitate. 7 These changes were associated with increased kidney weights, tubular dilatation, tubulointerstitial inflammation, and expansion of the medullary interstitium. Fibans are a group of antithrombotic agents that are metabolized into the active drug that contains a carboxylic acid group. The compound induces an obstructive nephropathy in rats because of accumulation of intratubular crystal precipitates. 8 Conditions of high ionic strength and hyperosmolarity could promote precipitation of drug/metabolite, uric acid, or oxalate crystals as well as aggregation of THP.
The spectrum of lesions noted in our study has also been reported to occur in monkeys treated with a human recombinant IL-18 4 , CD4 2 receptor proteins, and in immunoglobulin-induced Bence-Jones cast nephropathy in humans. 11 In these cases, it was shown that lesions were caused by coprecipitation of drug material with THP, the most abundant urinary protein produced exclusively at the thick ascending loop of Henle and distal convoluted tubules. 12 Cytokines, immunoglobulins (Bence-Jones protein), and urinary crystals bind with great affinity to THP and intensify its gelification/aggregation. In fact, THP has been shown to be an important defense factor against the formation of renal stones by inhibiting the adhesion of crystals to the cell surface. 1, 6, 9 Electron microscopy revealed the presence of crystaloid material within intracytoplasmic vacuoles in proximal tubular epithelial cells of treated monkeys. Although the composition of these crystals is uncertain, it could represent precipitating drug material or its metabolites. It has been shown that, once formed in the tubular lumen, crystals such as calcium oxalate rapidly bind to the apical surface of renal epithelial cells, where further crystal aggregation may occur. In addition, these crystals may be internalized into membrane-bound vesicles where they may eventually dissolve. We hypothesize that urinary THP is aggregating in response to the accumulation of crystalloid material, causing consequent tubular obstruction, epithelial cell degeneration, and tubulointerstitial inflammation. A similar hypothesis has been proposed for the protein cast nephropathy induced by plasma cell dyscrasias in humans. 11
Although we have observed several representative cases of obstructive nephropathy in monkeys after treatment with small organic carboxylates, a cause and effect relationship cannot be clearly established. Carboxylic acids represent a common chemotype used in medicinal chemistry, and a similar pattern of renal pathology in monkeys treated with this class of molecules has not been described by others to this date. However, it is plausible that under certain conditions, compounds containing a carboxylic acid moiety may promote alterations in ionic concentration, osmolarity, and pH within the urinary microenvironment of the nephron. Such physicochemical alterations can facilitate the formation of crystals and THP casts, causing obstruction and tubulointerstitial inflammation. Other factors such as hydration status of the animal, compound bioavailability, solubility, degree of protein binding, and chemical pKa (dissociation equilibrium constant) likely play a role in the development of these renal lesions. Drug-induced obstructive nephropathy is not identified as a significant adverse event in humans, thus the clinical relevance of the above findings in monkeys is not clear.
