Abstract
The present article describes an occurrence of eosinophilic airway inflammation of a 4-year-old female cynomolgus monkey in a vehicle control group of a routine toxicology study. Histologically, the airway lesion was characterized by prominent eosinophilic infiltrates, accompanied by mast cells, lymphocytes, and plasmacytes. The eosinophilic infiltrates were distributed throughout the airway: from trachea through respiratory bronchioles in the lung. The morphological feature of the lesion was indicative of an allergic airway disorder that can occur in humans with asthma. The present case is remarkable in that there is a paucity of reports on naturally occurring allergic airway disorders in nonhuman primates.
Human allergic disorders, such as anaphylaxis, hay fever, eczema, and asthma, have been increasing in the developed world, with roughly 25% of the population currently being affected. 4 For many asthma patients, the disease has its roots in infancy, and factors both genetic and environmental contribute to its inception and evolution. 2 Allergic asthma can be referred to as abnormal adaptive immune responses in the airway. Briefly, this begins with presentation of an allergen to naive CD4+ helper T cells by dendritic cells. Then, T-helper-2 (TH2) cells are generated and cytokines (IL-4, IL-5, IL-13) are released from TH2 cells. These cytokines induce activation of eosinophils (IL-5) and immunoglobulin E–producing (IgE-producing) B cells (IL-4 and IL-13) and mast cells (IL-4); subsequently, IgE binds to mast cells. After that, exposure of IgE-coated mast cells to the allergen leads to the release of proinflammatory mediators (including those stored in the cytoplasmic granules), lipid-derived mediators, and newly synthesized cytokines, chemokines, and growth factors, as well as other mediators. These induce the recruitment and activation of TH2 cells, eosinophils, and other leucocytes and the persistent mediator production by resident cells (such as mast cells). Prolonged or repetitive exposure to specific allergens ultimately results in airway remodeling, characterized by goblet cell hyperplasia and increases in submucosal tissue, the adventitia, and smooth muscle. 4,13
The animal was a captive-bred 4-year-old female cynomolgus monkey (
Reportedly, the monkey had a history of exhibiting intermittent dry cough 3 months and 6 months before the study. No remarkable respiratory signs were noted during the study. The monkey gained 0.7 kg during the last 8 months (from 2.9 kg to 3.6 kg). Hematological examination revealed increased circulating eosinophil counts in this animal. Eosinophil counts on day –2 (561/μl) slightly exceeded the range (51 to 289/μl) obtained from samples at multiple points of the other 2 female monkeys in the same control group and furthermore increased to 1,516/μl and 1,400/μl on day 7 and day 30, respectively. Other hematological parameters and serum chemistry parameters remained within the historical normal range. No gross pathology observations were recorded. Microscopic examination revealed mild to moderate diffuse infiltration of eosinophils in the trachea, intrapulmonary bronchi, bronchioles, and terminal and respiratory bronchioles of the left and right lobes of the lung. In the trachea, moderate diffuse infiltration of eosinophils was observed in the epithelium, accompanied by slight infiltration of lymphocytes, plasmacytes, and eosinophils in the lamina propria (Fig. 1 ). A considerable number of mast cells, as identified with toluidine blue stain, was also found in the lamina propria (Fig. 2). Pulmonary changes consisted of infiltration of eosinophils and associated epithelial degeneration of bronchial mucosa. When compared to normal lung (Fig. 3), there was an accumulation of eosinophils and mucus within peribronchial glandular ducts, a slight increase in the size of peribronchial lymphatic tissue, and a slight degeneration/hypertrophy of smooth muscle cells in the bronchi (Figs. 4, 5). The terminal and respiratory bronchioles were also infiltrated with eosinophils along their walls (Fig. 6).
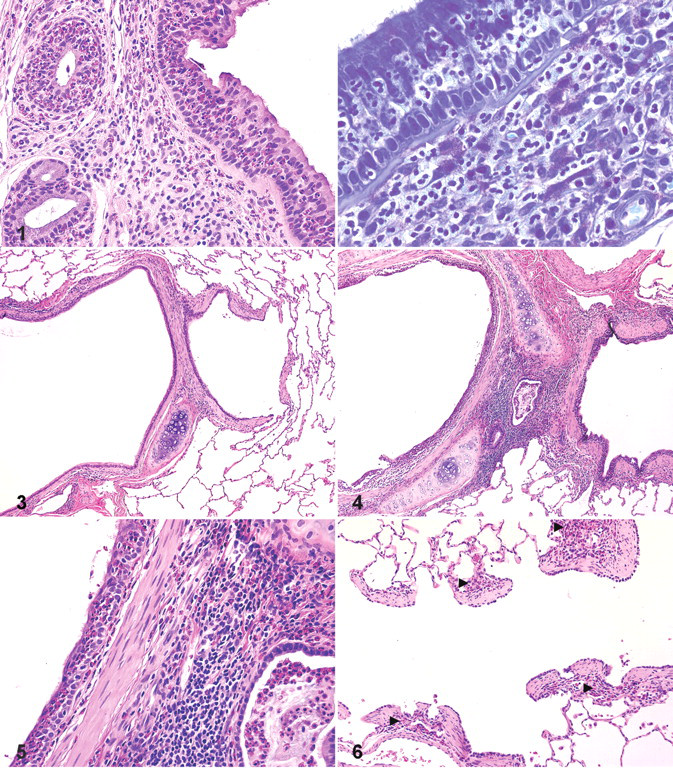
In humans, morphologic changes in asthma have been principally described in patients with fatal asthma. Those include occlusion of the bronchial lumen by mucus, an inflammatory infiltrate in the bronchial walls with a prominence of eosinophils and mast cells, hypertrophy of the bronchial smooth muscle, an increase in size of the submucosal glands, and a thickening of the basement membrane of the bronchial epithelium. 7 In patients with asthma with mild to moderate disease, substantial inflammation has been seen in bronchial-biopsy specimens. Although not observed uniformly, denudation of the airway epithelium, deposition of collagen beneath the basement membrane, mast cell degranulation, and infiltration of the airway by lymphocytes and eosinophils have been found. 2
The present case exhibited prominent airway eosinophilia and peripheral blood eosinophilia, which are characteristic features of allergic inflammation and components of asthma. An allergen presentation may have generated TH2 lymphocytes and contributed to the airway eosinophilia, given that the TH2 cytokine IL-5 is known as a central factor mediating eosinophil expansion, priming, recruitment, and prolonged tissue survival in response to allergic stimuli.
10
A formerly recorded dry cough, which was noted 3 and 6 months before the study, may have implied the persistent nature of the disorder. Many investigators have used monkeys to model allergic asthma or airway responses. A majority of nonhuman primates caught in the wild demonstrate skin test sensitivity to
Spontaneous allergic airway inflammation seems to be rare in monkeys, but a natural occurrence of Japanese cedar (
Although immunologic examinations supporting the evidence of allergy (eg, measurement of specific IgE) were not performed in the present case, the distribution and features of the microscopic lesions were consistent with allergic airway responses and could correspond to an early or mild phase of the asthmatic disease process. Possible allergens include dusts, foods, pollens, cage-cleaning solvents, and so on, but potentially any environmental antigen is implicated.
Pulmonary acariasis (
Footnotes
The authors declared that they had no conflicts of interest with respect to their authorship or the publication of this article.
The authors declared that they received no financial support for their research and/or authorship of this article.
