Abstract
Twenty-five 22– to 46–day-old broilers with Newcastle disease (ND) were investigated pathologically and immunohistochemically in order to evaluate the mechanism of ND outbreak in vaccinated broilers. The broilers were vaccinated with ND live vaccine via drinking water. Clinical signs were neurologic and respiratory in nature. Macroscopically, bursal atrophy, white spots on the pancreas, and discoloration and enlargement of kidneys and spleen were observed in the broilers. Histologically, perivascular cuffing, neuronal degeneration and necrosis, and glial proliferation were present in the cerebrum, cerebellum, and medulla oblongata. There was extensive rarefaction and malacia in the parenchyma of severely affected brains. There were extensive degeneration, necrosis, and depletion of acinar cells in the pancreas. There was proliferation of macrophages in the lungs with congestion, tubulointerstitial nephritis, hepatocytic necrosis with thrombi in the sinusoids, and lymphocytic depletion in the cloacal bursa. Immunohistochemically, ND virus antigens were detected in the lesions. ND virus isolated from the present cases did not cause encephalitis or pancreatitis in specific-pathogen-free chickens, but it induced mortality with hepatocytic sinusoidal thrombi, splenic necrosis, lymphoid necrosis and depletion, and conjunctival hemorrhage. Severe nonpurulent encephalitis with extensive rarefaction and malacia, and necrotizing pancreatitis in the present case may suggest a close possibly causal relation with vaccination.
Newcastle disease (ND) is caused by ND virus, which is synonymous with avian paramyxovirus type 1. 1 ND is one of the most harmful diseases in the poultry industry in the world. Along with highly pathogenic avian influenza (HPAI), ND belongs to one of the important World Organization for Animal Health (OIE) listed diseases in avian diseases. There are 5 pathologic types of ND in chickens: viscerotropic velogenic ND, neurotropic velogenic ND, mesogenic, lentogenic and asymptomatic-enteric form. 1
Since the development of ND vaccines, poultry have been protected from outbreaks. Recently, there have been some outbreaks in unvaccinated poultry, for example, small flocks of backyard chickens or chickens kept without the use of synthetic chemicals, including vaccines, in organic husbandry (data from press releases from the Ministry of Agriculture, Forestry, and Fishery of Japan [MAFF]). In addition, occasional outbreaks of ND are reported in vaccinated chicken flocks in Japan (data from press releases, MAFF). The vaccinated chicken flocks were mainly broilers and rarely layers. Various factors (more velogenic ND virus overwhelming the vaccination, improper vaccination, inhibited vaccine takes by maternal antibody, and immunosuppression by infectious or noninfectious factors) have been discussed. Virologic, pathologic, and immunohistochemical examinations of chickens with ND in the field have been useful to investigate the mechanism of ND outbreaks in vaccinated chickens.
The main histologic lesions of ND are nonpurulent encephalomyelitis, vasculitis, lymphoid necrosis (cloacal bursa, spleen, thymus and intestinal mucosal lymphoid tissue), tracheitis and pneumonia, salpingitis, liver necrosis, cellular infiltration of pancreas, and conjunctivitis. 1 However, there have been few reports on the formation of encephalomalacia and necrotizing pancreatitis in chickens affected with ND, except the report of Bhaiyat et al. 2 concerning broilers infected with mesogenic ND virus and infectious bursal disease (IBD) virus. In their report, immunohistochemical investigation was unsuccessful. The detailed information about epidemiologic and virologic findings of more field cases, including the present case, will be reported elsewhere.
We found velogenic ND virus infections in vaccinated broilers in Japan. The broilers had characteristic lesions: nonpurulent encephalitis with malacia and necrotizing pancreatitis. We investigate the pathologic and immunohistochemical analyses of them in order to evaluate the pathogenesis of nonpurulent encephalitis with malacia and necrotizing pancreatitis observed in the broilers that suffered from ND. Moreover, we try to reproduce the lesions by inoculating specific-pathogen-free chickens with ND virus isolate.
First outbreak
In December 2004, ND occurred first in 1 chicken flock (11,950 birds) of a commercial broiler farm (Farm 1) (rearing 100,000 birds) in Fukuoka Prefecture in Japan. Ten flocks were kept in 10 broiler houses on Farm 1. Of the affected flock, 2,551 birds (21.5%) died. Clinical symptoms were neurologic signs such as depression, somnolence, and leg paralysis.
Second outbreak
Nineteen days later, the other 2 flocks in Farm 1 suffered from ND secondly in January 2005. Clinical signs were neurologic signs and watery diarrhea. Eventually, all 10 broiler flocks in Farm 1 were depopulated. The broilers were inoculated with live ND vaccine (B1 strain) and live IBD vaccine via drinking water. ND vaccine and IBD vaccine were mixed to reduce labor and chickens were inoculated with mixture. On the farm, chickens were not supplied drinking water before the vaccination.
Third outbreak
In April 2005, there was an outbreak of ND in ND-vaccinated broilers on another broiler farm (Farm 2; rearing 27,500 birds) near Farm 1, which had the first and second outbreaks in Fukuoka. The broilers of Farm 2 had been vaccinated via drinking water with ND live vaccine at 7 days old. Clinical signs were neurologic and respiratory in nature.
Twenty-five 22- to 46-day-old broiler chickens (broiler Nos. 1 to 25; Table 1) were collected from broiler houses A to C of the broiler farm and examined for pathology, immunohistochemistry, and microbiology. Broiler Nos. 1 to 10 were obtained from house A of Farm 1 that experienced the first outbreak of ND. Broiler Nos. 11 to 20 were from house B of Farm 1 that had the second outbreak of ND. Broiler Nos. 21 to 25 were from house C of Farm 2, which had the third outbreak of ND. Broiler Nos. 16 to 20 died. Other broilers were euthanatized for examination using CO2.
Histologic lesions and immunohistochemical detection of ND virus antigen in the broiler chickens affected with ND.
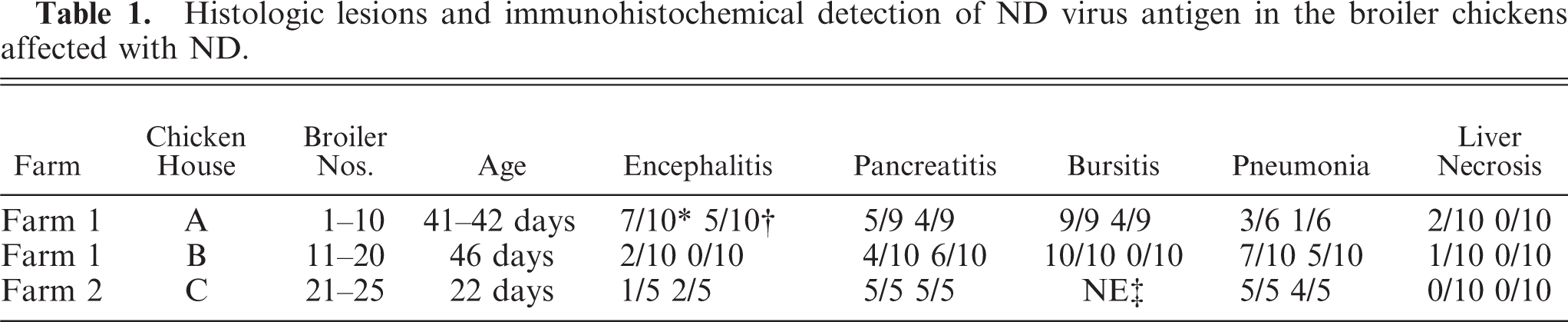
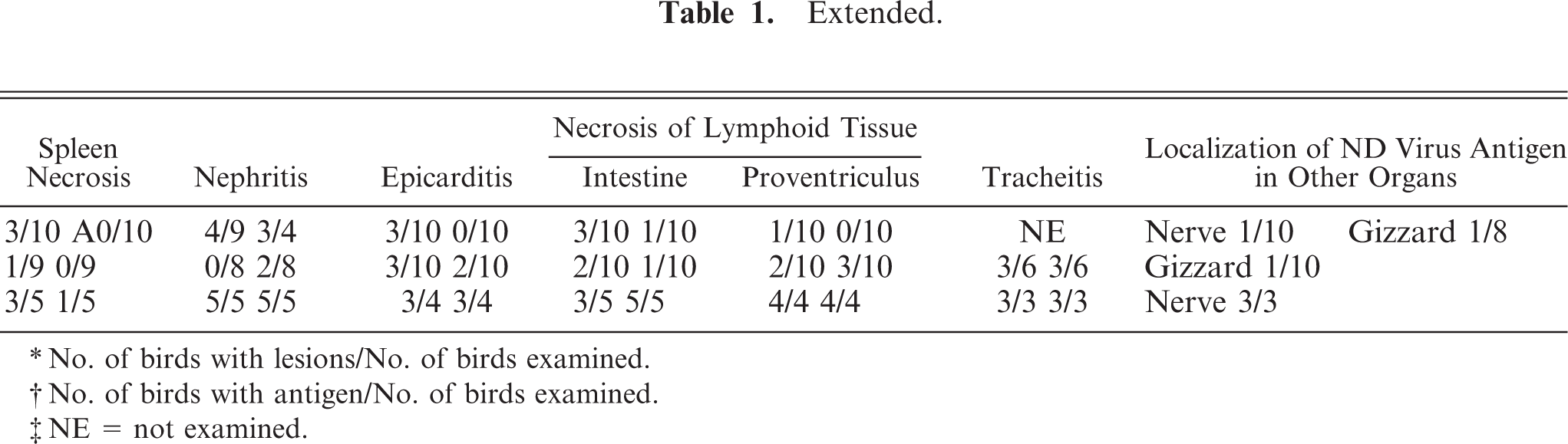
No. of birds with lesions/No. of birds examined.
No. of birds with antigen/No. of birds examined.
NE = not examined.
After a postmortem examination, the liver, spleen, kidney, heart, lung, trachea, air sacs, proventriculus, gizzard, duodenum, pancreas, ileum, cecum, cloacal bursa, cerebrum, cerebellum, optic lobes, medulla oblongata, peripheral nerve, testis, ovary, and skeletal muscles were removed and fixed in 10% buffered formalin. All tissue samples were then embedded in paraffin, sectioned at 4 µm, and stained with HE.
The paraffin sections were stained using an immunoperoxidase kit, Histofine Simple Stain PO (M) (Nichirei Inc., Tokyo, Japan). A Histofine simple stain PO (M) kit was used according to the manufacturer's instructions. 10 This method is called the “Universal Immuno-enzyme Polymer method”; it is a highly sensitive method and reduces time of staining. The labeled polymer was prepared by combining amino acid polymers with peroxidase and goat anti-mouse Ig that are reduced to Fab'. The sections were pretreated with 10 mmol of citrate buffer at pH 6.0 in a microwave oven at 500 W for 10 minutes for antigen retrieval. The primary antibody was a mouse monoclonal antibody against the nucleoprotein of ND virus 12 and was used at 1:100,000 dilutions. After staining, the sections were counterstained with hematoxylin.
In order to evaluate whether the lymphoid necrosis of cloacal bursa was caused by IBD virus or not, immunohistochemical staining of IBD virus antigen was also performed on cloacal bursa of broilers affected with ND as mentioned above. Rabbit anti-IBD virus serum was used as the primary antibody. 14
The homogenates of the trachea and rectum were inoculated into 10-day-old embryonating chicken eggs via the allantonic cavity 8 and into chicken kidney (CK) cell culture 6 for viral isolation. The allantonic fluid was collected from eggs 48 hours after inoculation or when the embryos died. In hemagglutination (HA)-positive samples, ND virus was confirmed by a hemagglutination inhibition (HI) test 5 with ND-virus-specific antiserum.
Viral RNA was extracted from infected allantonic fluid using a commercial kit (ISOGEN-LS; Nippon Gene, Tokyo, Japan). The regions of the fusion (F) protein gene, including the F protein cleavage site, were amplified by reverse transcription polymerase chain reaction (RT-PCR). 8 RT-PCR amplification and sequencing were performed as described previously. 8
Assays for serum HI antibodies against ND virus in the broilers of the affected and nonaffected flocks on the farms were done.
Macroscopically, bursal atrophy, white foci in the pancreas, discoloration and enlargement of kidneys and spleen were observed in the chickens.
Histologic lesions are summarized in Table 1. Brain lesions (severe nonpurulent encephalitis with malacia and rarefaction) and pancreatic lesions (extensive pancreatic necrosis) were characterized in the present cases.
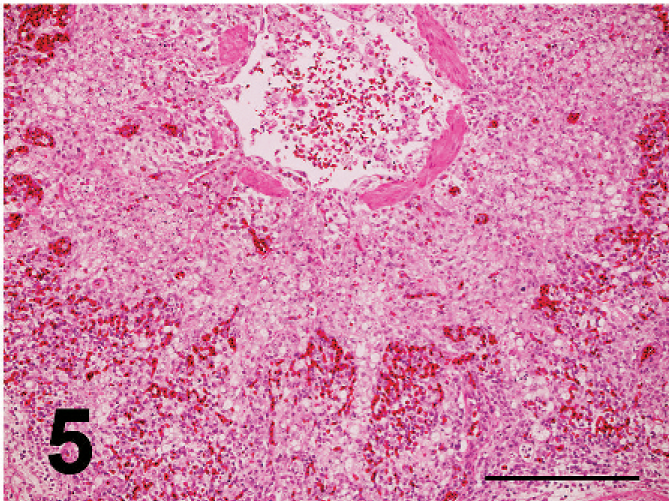
Nonpurulent encephalitis was present in the cerebrum, cerebellum, and medulla oblongata, but not in the optic lobe. There was massive rarefaction and malacia (Fig. 1) in the parenchyma of severely affected brain. There were lattice cells within malacic lesions (Fig. 2). Perivascular cuffing, glial proliferation, cellular infiltration, and neuronal degeneration and necrosis were observed (Fig. 3). There was extensive degeneration, necrosis, and depletion of acinar cells in the pancreas (Fig. 4). There was marked proliferation of macrophages in the lungs with congestion (Fig. 5). In addition, there was tubulointerstitial nephritis, hepatocytic necrosis with thrombi in the sinusoids, focal necrosis of myocardium, lymphocytic depletion in the cloacal bursa, degeneration and necrosis of glandular epithelium of gizzard, tracheitis, fibrinoid necrosis of blood vessels, necrosis of lymphoid tissue in the proventriculus, proliferation of macrophage in lamina propria of intestines, and epicarditis (Fig. 6).
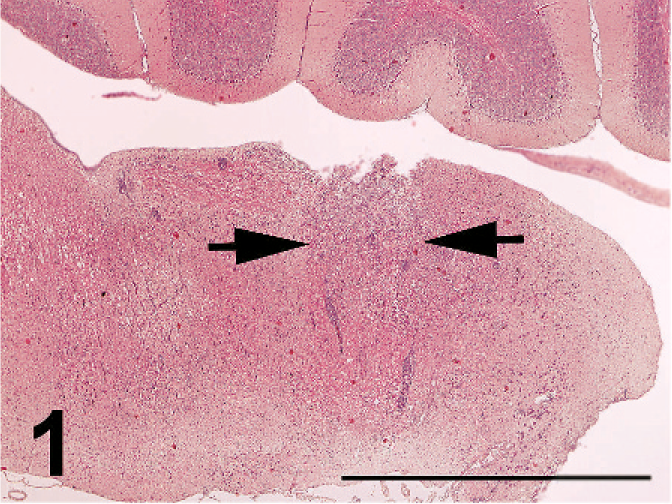
Malacia (arrows) in the medulla oblongata; broiler No. 5. HE. Bar = 2 mm.
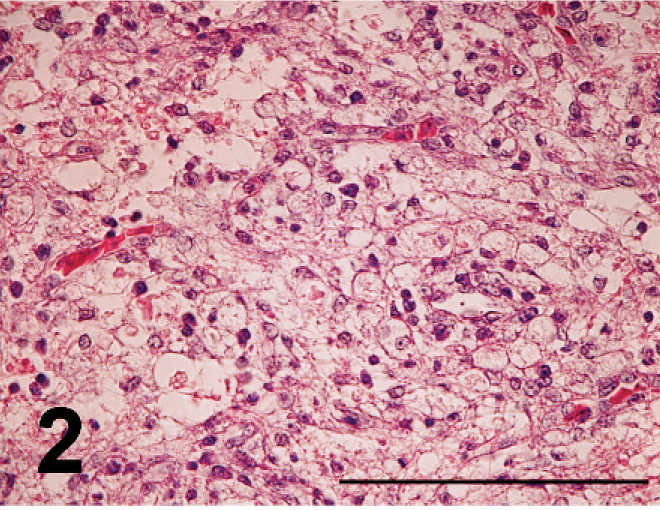
Lattice cells within malacia in the medulla oblongata; broiler No. 5. HE. Bar = 100 µm.

Perivascular cuffing of lymphocytes and plasma cells and diffuse infiltration of plasma cells and lymphocytes with glial proliferation in the parencyma in the cerebrum; broiler No. 5. HE. Bar = 200 µm.
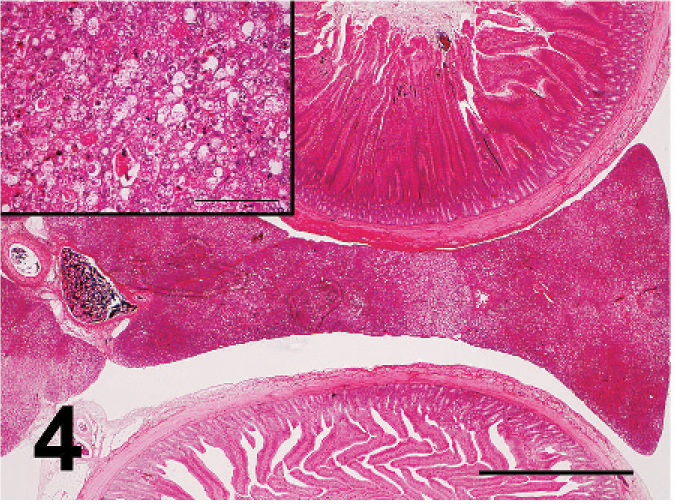
Extensive necrosis of pancreas showing pale areas. Bar = 2 mm. Inset: Necrosis of acinar cells. Bar = 100 µm. Broiler No. 5. HE.
Marked proliferation of macrophages in the lining and wall of atrium of parabronchus in the lung. There is thickening of area of atrium that compressing the congestive area of air capillaries and blood capillaries. Macrophages are seen in the lumen of parabronchus; broiler No. 17. HE. Bar = 200 µm.
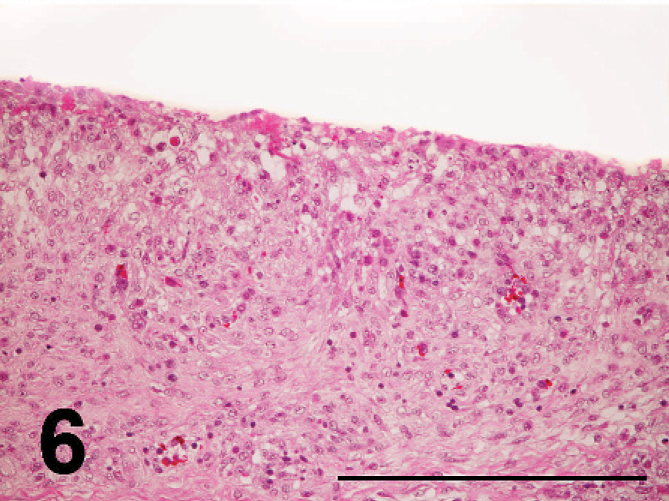
Fibrinous exudation in the surface mesothelium and proliferation of macrophages in the pericardium; broiler No. 17. HE. Bar = 200 µm.
ND virus antigens were detected in the lesions of various organs. They were primarily found in the cytoplasm, but rarely in the nucleus of affected cells. ND virus cells within malacia areas (Fig. 7) in the medulla and necrotic nerve cells in the cerebrum, necrotic acinar cells of pancreas (Fig. 8), necrotic follicles of cloacal bursa, bronchiolar epithelial cells and macrophages of lining and walls of atria (Fig. 8) in the lungs, tracheal epithelial cells, epithelial cells of air sac, necrotic renal epithelial cells, glandular epithelial cells of proventiculus and gizzard, and necrotic lymphoid tissue of the lamina propria of intestines. Occasionally, mesothelial cells of the epicardium were positive for ND antigen (Fig. 10), as well as peripheral nerves (Fig. 11) the muscular layer of intestines and proventriculus, or in the connective tissue around the trachea without apparent histologic lesions in these organs. ND virus antigens were seen also in the squamous epithelial cells of esophagus (Fig. 12) adjacent to the proventriculus.
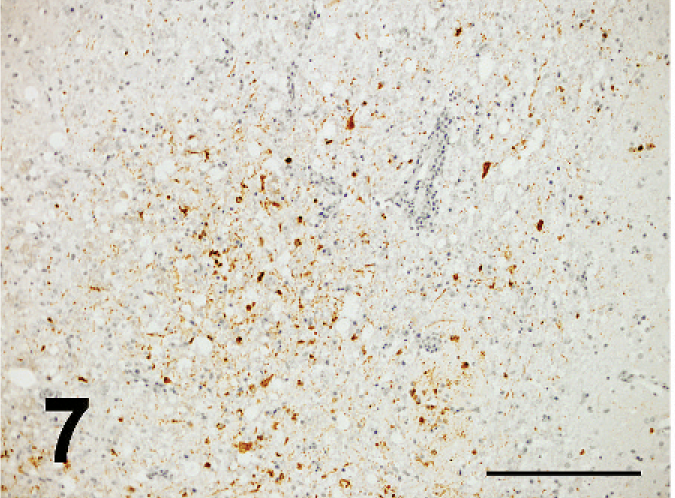
ND virus antigens were observed in the malacic lesion of medulla oblongata; broiler No. 5. Immunoperoxidase staining. Counterstain with hematoxylin. Bar = 200 µm.
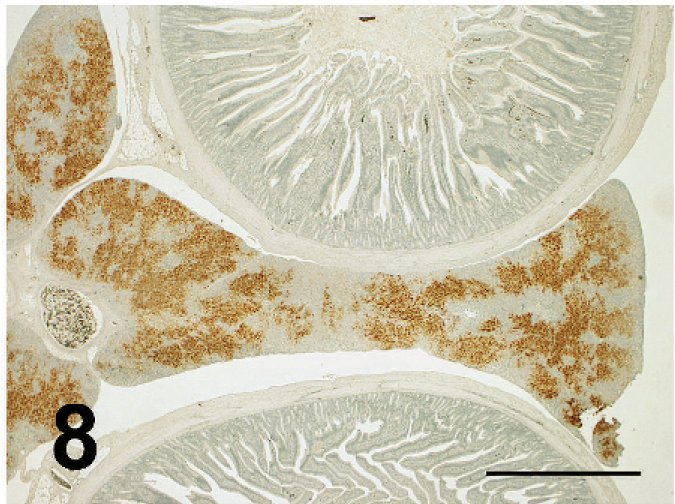
Prominent ND virus antigens in the necrotic areas of pancreas. Immunoperoxidase staining. Counterstain with hematoxylin; broiler No. 5. Bar = 2 mm.

ND virus antigens are seen in the macrophages in the lining and walls of atrium; broiler No. 17. Immunoperoxidase staining. Counterstain with hematoxylin. Bar = 200 µm.
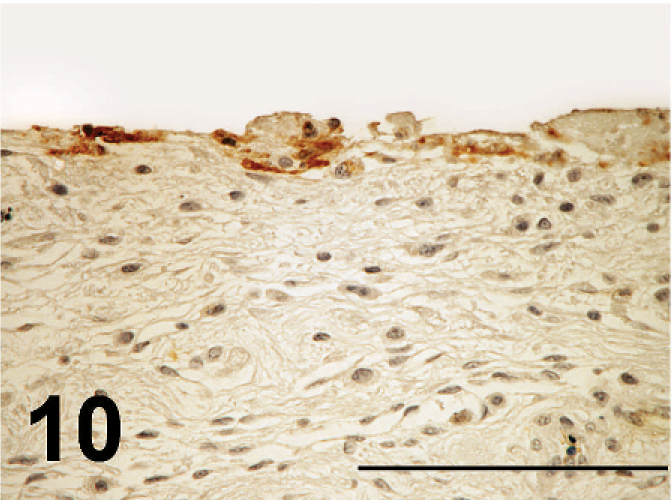
ND virus antigens are seen in the lining mesothelial cells of epicardium; broiler No. 17. Immunoperoxidase staining. Counterstain with hematoxylin. Bar = 200 µm.
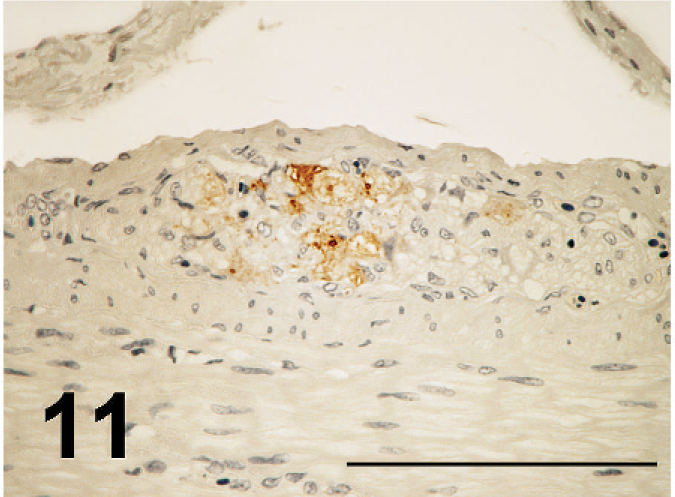
ND virus antigens in the nerve of the muscular layer of the duodenum; broiler No. 29. Immunoperoxidase staining. Counterstain with hematoxylin. Bar = 200 µm.
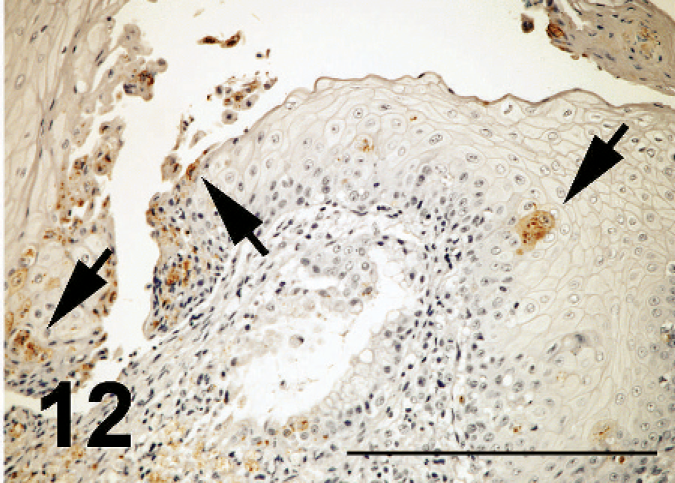
ND virus antigens in squamous epithelial cells (arrows), some that showed necrosis, desquamation and erosion in the esophagus; broiler No. 29. Immunoperoxidase staining. Counterstain with hematoxylin. Bar = 200 µm.
ND viruses were isolated from the trachea and rectum of affected chickens. In embryonating chicken eggs, the embryos died within 48 hours after inoculation with samples. HA units/50 µl of allantonic fluids were 32 to 512, and those of chicken kidney cell culture were 4 to 8. No other viruses were detected with chick embryo inoculation.
The genotype of isolated virus was VII. The amino acid sequence at F protein cleavage site was virulent type (-R-R-Q-K-R-F-). More detailed information about the molecular characterization of the isolated strains will be reported elsewhere.
The serum HI antibodies against ND virus were low in the chickens of affected flocks, while those in nonaffected flocks were high.
Eight 6-week-old specific-pathogen-free chickens were inoculated intranasally with 107 plaque-forming units (PFU) of the ND virus isolate (APMV1/chicken/Japan/Fukuoka-1/2004) to examine the mortality rate and pathology of chickens by ND virus. Throughout the experiment period, the chickens were kept in isolators under negative pressure and supplied with filtered air. The chickens were housed in stainless-steel isolation cabinets that were ventilated under negative pressure with HEPA-filtered air, and care was provided as required by the Institutional Animal Care Committee. The chickens were observed for 10 days after inoculation. Chickens that died during the observation period were necropsied as soon as possible.
All SPF chickens inoculated with ND virus died at 3 days after inoculation. Clinically, the chickens exhibited no apparent clinical signs except depression. Macroscopically, the chickens exhibited hemorrhages in the conjunctiva. Histologically, the chickens had splenic necrosis, hepatocytic sinusoidal thrombi, and lymphocytic necrosis and depletion in the lymphoid tissues (cloacal bursa, thymus, and cecal tonsil), and hemorrhages and vascular necrosis of the conjunctiva. There were no lesions in the central nervous system or pancreas.
Nonpurulent encephalomyelitis with neuronal degeneration, foci of glial cells, perivascular cuffing of lymphocytes, and hypertrophy of endothelial cells of the brain are seen in ND in chickens. 1 These brain lesions are invariably observed in the chickens infected with the neurotropic velogenic pathotype 3 and commonly with viscerotropic 3, 4 and mesogenic pathotypes. 2 Generally, histologic lesions of the central nervous system are found in the cerebellum, medulla, midbrain, and spinal cord but rarely found in the cerebrum. 1 However, Bhaiyat et al. 2 reported that the lesions were most frequently observed in the cerebrum in the broilers naturally infected with ND. The lesions of the present case were seen in the medulla, cerebrum, and cerebellum in decreasing order of frequency.
There are few reports on extensive malacia in chickens infected naturally with viscerotropic velogenic ND virus. Bhaiyat et al. 2 reported nonpurulent encephalitis with malacia and pancreatic necrosis in the broilers naturally infected with mesogenic ND virus and IBD virus. The broilers had been vaccinated with ND vaccine. They failed to detect ND virus antigen in any lesions by immunohistochemistry using monoclonal antibody against hemagglutinin-neuramidase protein of the ND virus. The present case also had extensive focal malacia with nonpurulent encephalitis in the brain. Localization of ND virus antigens in necrotic nerve cells within the malacia suggests that the malacia may be caused by direct virus infection of nerve cells. In addition, malacia may be also induced as blood circulation disturbance by severe vascular damage in the brains of ND virus–infected chickens, as generally ND virus may induce vascular damage. 1
Immunohistochemical study of IBD virus antigen suggested the lymphoid necrosis in the cloacal bursa was caused by IBD virus. 2 IBD virus may enhance the virulence of mesogenic ND virus. In the present case, the necrotic lymphoid follicles of the bursa showed positive for ND virus antigen but not for IBD virus antigen. Therefore, the virulent ND virus alone produced lymphoid necrosis in the present cases.
Immunohistochemical detection of ND virus antigen in the peripheral nerves is a unique finding and has never been reported. Some workers observed nonpurulent neuritis in the chickens affected with ND. 7, 9 However, it is not clear whether this lesion was caused by ND virus or not. Localization of ND virus antigen in the nerves was correlated with the lesions and antigens in the lymphoid tissues of the intestine and proventriculus and in the tracheal epithelial cells. Therefore, ND virus antigens in the nerves may be from these mucosal lymphoid tissues or epithelial cells. In addition, ND virus antigens were demonstrated in the mesothelial cells of areas affected from epicarditis and in the squamous epithelial cells of esophagus with erosion. There is no report about the localization of ND virus antigens in such tissues. It is an interesting finding and must be studied in detail in the future.
ND and highly pathogenic avian influenza (HPAI) exhibit similar pathologic conditions. Focal necrosis of the central nerve system with glial nodules and mild necrotizing pancreatitis are observed in HPAI of chickens. 13 However, severe nonpurulent encephalitis and necrotizing pancreatitis are observed more frequently in HPAI of the more resistant call ducks 16 and crows 15 than in chickens. The chickens of the present case became resistant to velogenic ND virus because of vaccination, and then experienced severe encephalitis and pancreatitis. There were neither brain lesions nor pancreatic lesions in specific-pathogen-free chickens infected experimentally with velogenic ND viruses. 11 Severe nonpurulent encephalitis with extensive rarefaction and malacia and necrotizing pancreatitis may be closely related with vaccination.
In this study, intranasal inoculation of SPF chicken with virus isolated from field outbreaks resulted in a disease syndrome that differed pathologically from the field disease specifically, experimentally inoculated chickens did not have central nervous system or pancreatic lesions, both of which were prominent in the field cases. The presence of pre-existing partial immunity in the field cases, due to vaccination, may be responsible for the differing presentations.
Footnotes
Acknowledgements
We thank Mr. M. Kobayashi and Miss Megumi Shimada for histologic and immunohistochemical assistance.
