Abstract
In this study, we investigated the pathogenesis of Newcastle disease virus (NDV) in the chicken kidney. Twenty-six 32-day-old specific pathogen-free chickens were intranasally inoculated with the 9a5b NDV mutant isolate. Kidney tissue samples, collected at 6 and 12 hours postinoculation (hpi) and 1, 2, 3, 5, and 10 days postinoculation (dpi), were analyzed by histopathology, immunohistochemistry (IHC), reverse transcription polymerase chain reaction (RT-PCR), and virus titration. Histopathologically, tubulointerstitial nephritis was detected in the renal cortex and predominantly in the medulla. Nephrotropism of 9a5b NDV was confirmed by IHC, RT-PCR, and virus isolation. Massive degenerative changes and infiltration of CD3-immunopositive cells accompanied replication of the 9a5b NDV isolate in chicken kidneys. In conclusion, pathological changes that were caused by NDV in chicken kidneys were similar to those caused by avian influenza virus, infectious bronchitis virus, and avian nephritis virus, and this highlights the importance of including NDV in the differential diagnosis of kidney disease in chickens.
Newcastle disease virus (NDV) is the causative agent of Newcastle disease. NDV is a single-stranded, nonsegmented, negative-sense enveloped RNA virus. 4,5 The genome of NDV encodes 6 structural proteins: nucleoprotein (NP), fusion protein (F), RNA polymerase protein (L), matrix protein (M), hemagglutinin-neuraminidase protein (HN), and phosphoprotein (P). 4,5 Virulence of NDV is multigenic and the F, HN, and P genes are key players in its virulence. 5,16 Based on the severity of clinical signs, NDVs can be categorized into strains of high virulence (velogenic), moderate virulence (mesogenic), and low virulence (lentogenic); 11 also based on the intracerebral pathogenicity index (ICPI) in 1-day-old chicks, NDVs can be categorized into virulent and avirulent strains. 11
The fusion (F) protein cleavage site amino acid sequence of avirulent strains is 112(G/E)(K/R)Q(G/E) RL117 and cleaved by trypsin-like enzymes found in certain tissues, while that of virulent strains is 112(R/K)-R-Q-(R/K) RF117 and cleaved by ubiquitous intracellular host proteases, causing systemic infection. 4
Viral tropism for chicken kidneys has been shown by certain viruses such as avian infectious bronchitis virus (IBV), 1 avian nephritis virus (ANV), 10 and some strains of avian influenza virus (AIV). 17 Renal pathological manifestations of these viruses include renal tubular necrosis, degeneration, and nephritis. 1,10,17 Renal lesions due to NDV have been reported in several avian species, for example, chickens, turkeys, and double-crested cormorants. 2,3,9,12,16 However, little is known about renal lesions in NDV infections, and no previous studies have addressed the relationship between histopathological alterations in chicken kidney tissues and NDV replication.
Avirulent wild viruses, having the typical avirulent fusion protein cleavage site sequence, have the potential to become velogenic after passage in chickens. 15,18 The virulent 9a5b NDV mutant isolate was generated from the lentogenic Goose/Alaska/415/91 strain by 9 consecutive passages in chicken air sacs, followed by 5 passages in chicken brain. 15 The original strain has an ICPI of zero and avirulent F protein cleavage site compared with the 9a5b NDV isolate, which has an ICPI equal to 1.88 and a virulent F protein cleavage site. 15 Previously, a comparative study found that renal lesions were more prominent in chickens than ducks after experimental infection with 9a5b NDV. 2 Therefore, we aimed in this study to investigate the renal lesions in chicken kidneys using the same isolate, from an early to a late phase of infection, as well as the renal pathogenesis of NDV strains that may spontaneously mutate from avirulent to virulent ones.
The 9a5b NDV isolate was propagated in specific pathogen-free (SPF) eggs, and 0.1 ml of the viral suspension containing 108.75 50% embryo infectious dose (EID50) was inoculated intranasally in each chicken. 15
Twenty-six 32-day-old male SPF white Leghorn chickens (Nippon Institute for Biological Science, Tokyo, Japan) were assigned into 2 groups: the control group (n = 5) and the infected group (n = 21). Chickens were reared under biosafety level 2 and acclimatized for 1 week prior to virus inoculation with free access to food and water. Three chickens from the infected group were euthanized each at 6 and 12 hours postinoculation (hpi) and 1, 2, 3, 5, and 10 days postinoculation (dpi). Chickens in the control group were euthanized on the final experimental day. After euthanasia, necropsy was performed and kidneys were examined for gross abnormalities. Kidney tissues were further processed by either fixation in 10% neutral buffered formalin for hematoxylin and eosin (HE) and immunohistochemical (IHC) staining or preserved at –80°C for viral RNA isolation and virus titration. The Tottori University ethics committee approved all experimental procedures.
Kidney tissues were fixed in formalin for approximately 60 hours and were processed routinely for HE and IHC staining. For NDV detection, the primary antibody was prepared in rabbits against the NDV nucleoprotein (NDV-NP), and the IHC protocol was performed as previously described 2 using antigen retrieval with citrate buffer (pH 5.4) for 10 minutes, overnight incubation with primary antibody (1:8000 dilution) at 4°C, detection with a labeled polymer (ChemMate DAKO EnVision/HRP [DAP]; Dako, Carpinteria, CA), and staining with 3, 3′-diaminobenzidine tetrahydrochloride (DAB) and a hematoxylin counterstain. Positive control tissues were spleen and kidney of experimental infected NDV in chickens. For negative control, the primary antibody was replaced by phosphate-buffered saline.
For CD3 IHC, we performed the previously mentioned protocol except that the primary antibody was rabbit polyclonal anti-human CD3 (1:300 dilution; Dako), and the antigen retrieval time was 20 minutes.
Virus titers were measured using 10-day-old embryonated SPF chicken eggs. Briefly, kidney tissue from each chicken was homogenized in PBS (1:10 [wt:vol]), and a 10-fold serial dilution of the clear homogenate (100 μl) was inoculated into the allantoic cavity of the eggs. The eggs were incubated at 37°C for 3 days followed by incubation at 4°C for 1 day. The hemagglutination assay was performed on all eggs, and viral titers were expressed as EID50 per gram. 13
Total RNA was extracted from the kidney tissue using TRIzol reagent (Invitrogen, Carlsbad, CA), and then the extracted RNA (500 ng) from each sample was reverse transcribed to complementary DNA (cDNA) using PrimeScript II first strand cDNA synthesis kit (Takara, Otsu, Japan). Primer sets from the F gene were used to amplify a target sequence of 168 bp. Primer sequences were as follows: 5′-CGCAGATCACAGCGGCTTCTG-3′ (forward) and 5′-GGTCGT TTA CAAACTGCTGC- 3′ (reverse). 18 The polymerase chain reaction (PCR) conditions comprised 40 amplification cycles of 94°C for 2 minutes, followed by denaturation at 94°C for 30 seconds, annealing at 56°C for 30 seconds, extension at 72°C for 20 seconds, and a final extension at 72°C for 5 minutes. In total, 10 μl of the PCR product was detected by 2% agarose gel electrophoresis. The gel was precast with Gel Red Nucleic Acid Stain (Biotium, Hayward, CA).
The clinical signs were mild and transient. Chickens showed depression, lack of appetite, lethargy, green diarrhea, and closed eyes at 3–5 dpi. After 5 dpi, the chickens returned to an almost normal state and recommenced food and water consumption.
The findings are summarized in Table 1. Grossly, the kidneys were pale with mild lesions. Multifocal small whitish areas were observed in cross sections of kidneys in 2 and 3 chickens at 5 and 10 dpi, respectively.
Intensity and Distribution of Renal Gross Lesions, Histopathology, and Immunohistochemistry of NDV-Nucleoprotein and CD3 Staining in Chickens After Experimental Infection With 9a5b NDV Mutant Isolate.a
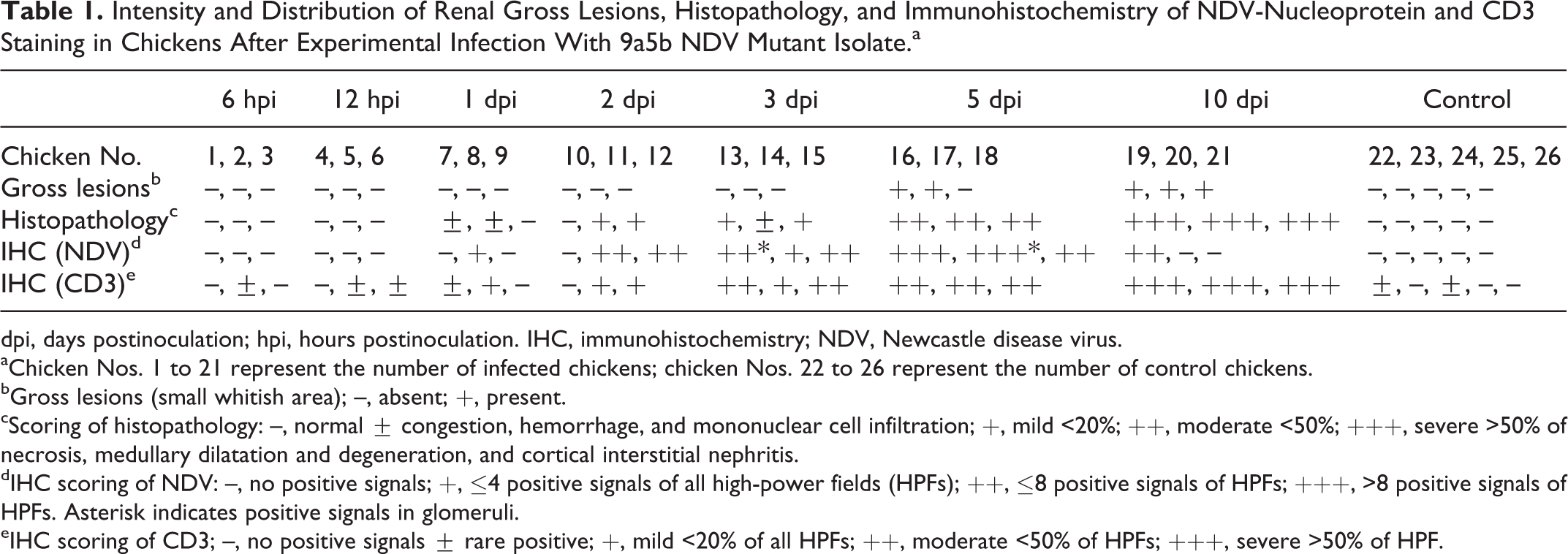
dpi, days postinoculation; hpi, hours postinoculation. IHC, immunohistochemistry; NDV, Newcastle disease virus.
aChicken Nos. 1 to 21 represent the number of infected chickens; chicken Nos. 22 to 26 represent the number of control chickens.
bGross lesions (small whitish area); –, absent; +, present.
cScoring of histopathology: –, normal ± congestion, hemorrhage, and mononuclear cell infiltration; +, mild <20%; ++, moderate <50%; +++, severe >50% of necrosis, medullary dilatation and degeneration, and cortical interstitial nephritis.
dIHC scoring of NDV: –, no positive signals; +, ≤4 positive signals of all high-power fields (HPFs); ++, ≤8 positive signals of HPFs; +++, >8 positive signals of HPFs. Asterisk indicates positive signals in glomeruli.
eIHC scoring of CD3; –, no positive signals ± rare positive; +, mild <20% of all HPFs; ++, moderate <50% of HPFs; +++, severe >50% of HPF.
Histologically, around 1 to 2 small lymphoid foci were observed in the interstitium of the kidney in 2 of the 5 control chickens and in the ureteral mucosa of all examined birds. Areas of hematopoiesis composed of red blood cells and granulocytes were observed in both control and infected birds. No histopathological changes were observed in infected kidneys at 6 and 12 hpi. At 1 dpi, 2 chickens had congestion, hemorrhage, and marked mononuclear cell infiltration. At 2 and 3 dpi, 2 chickens in each had mild multifocal lymphohistiocytic tubulointerstitial nephritis within the cortex, and lymphoid nodules were observed and gradually increased. The medullary cones were mildly dilated and infiltrated with heterophils and other mononuclear cells (Fig. 1). At 5 dpi, 3 chickens had moderate multifocal lesions, and occasionally these lesions coalesced to form diffuse cortical tubulointerstitial nephritis. Marked degeneration, necrosis, and apoptosis of the renal tubular epithelium in the cortical region were identified (Suppl. Fig. S1). The medullary cone showed moderate dilatation and degeneration of its components (collecting tubules and ducts, medullary loops, and ureteral branches) associated with calcification and inflammatory reaction. At 10 dpi, 3 chickens had severe cortical tubulointerstitial nephritis. The medullary cones were markedly dilated and more severely damaged than the cortex. Mild to moderate interstitial fibrosis and protein casts were observed in the renal tubular lumen. Severe dystrophic calcification and inflammatory response were observed (Suppl. Fig. S2). Clinical signs and gross and histopathologic lesions were absent in control chickens.
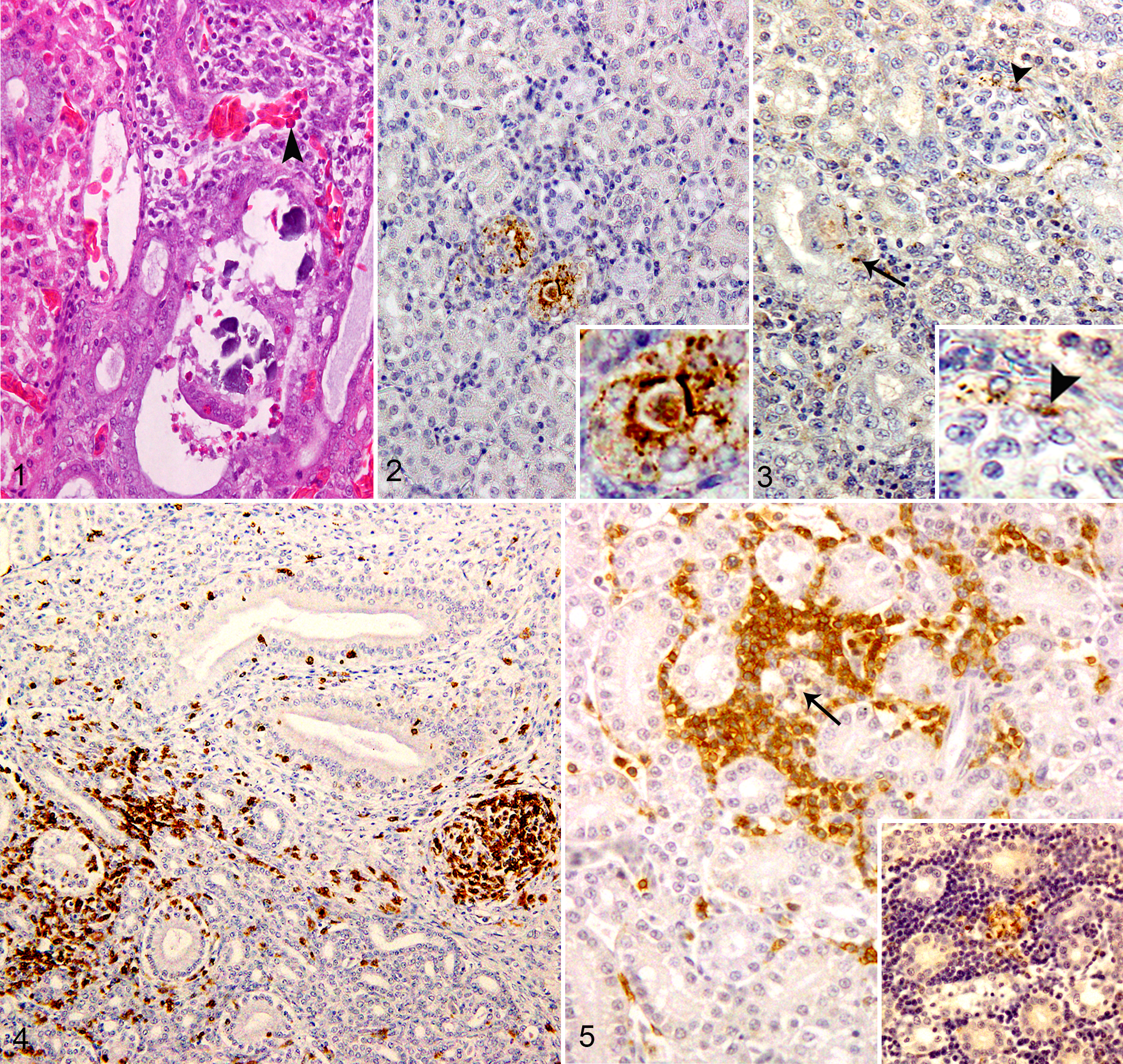
Immunohistochemically, NDV-NP was detected in infected renal tissues at 1 dpi, followed by an increase in intensity and distribution at 2 to 5 dpi and then decreased or became absent by the final experimental day. Most immunostaining for NDV-NP was distributed in a multifocal pattern in the intact (Fig. 2) and vacuolated epithelium of proximal and distal tubules (Suppl. Fig. S1), in infiltrating macrophages, and occasionally in some cells of reptilian-type glomeruli in the cortex (Fig. 3). In the renal medulla, NDV-NP immunostaining was mainly present in the tubular epithelium and the infiltrating mononuclear cells. Kidney of control chickens was negative to NDV-NP IHC (Suppl. Fig. S3).
CD3-positive cells were observed rarely in the interstitium of the kidneys in 2 chickens of the control group and in the ureteral mucosa of all control chickens. In the infected renal tissues, infiltration of CD3-immunopositive cells commenced at 1 dpi and became marked at 10 dpi. CD3-positive cells had focal and diffuse infiltration throughout the entire kidney (Fig. 4) and in areas with immunostaining signals of 9a5b NDV (Fig. 5).
Virus titration results are summarized in Figure 6. At 6 and 12 hpi, 9a5b NDV was not detected in kidneys. Virus was detected in 1 chicken at 1 dpi, in 2 chickens at 2 dpi, in all examined chickens at 3 and 5 dpi with a replication peak at 5 dpi, and in only 1 chicken at 10 dpi. Reverse transcription (RT)–PCR results in infected chickens kidneys coincided with IHC and virus titration results (data not shown). Kidneys of control chickens were negative by virus titration and RT-PCR.
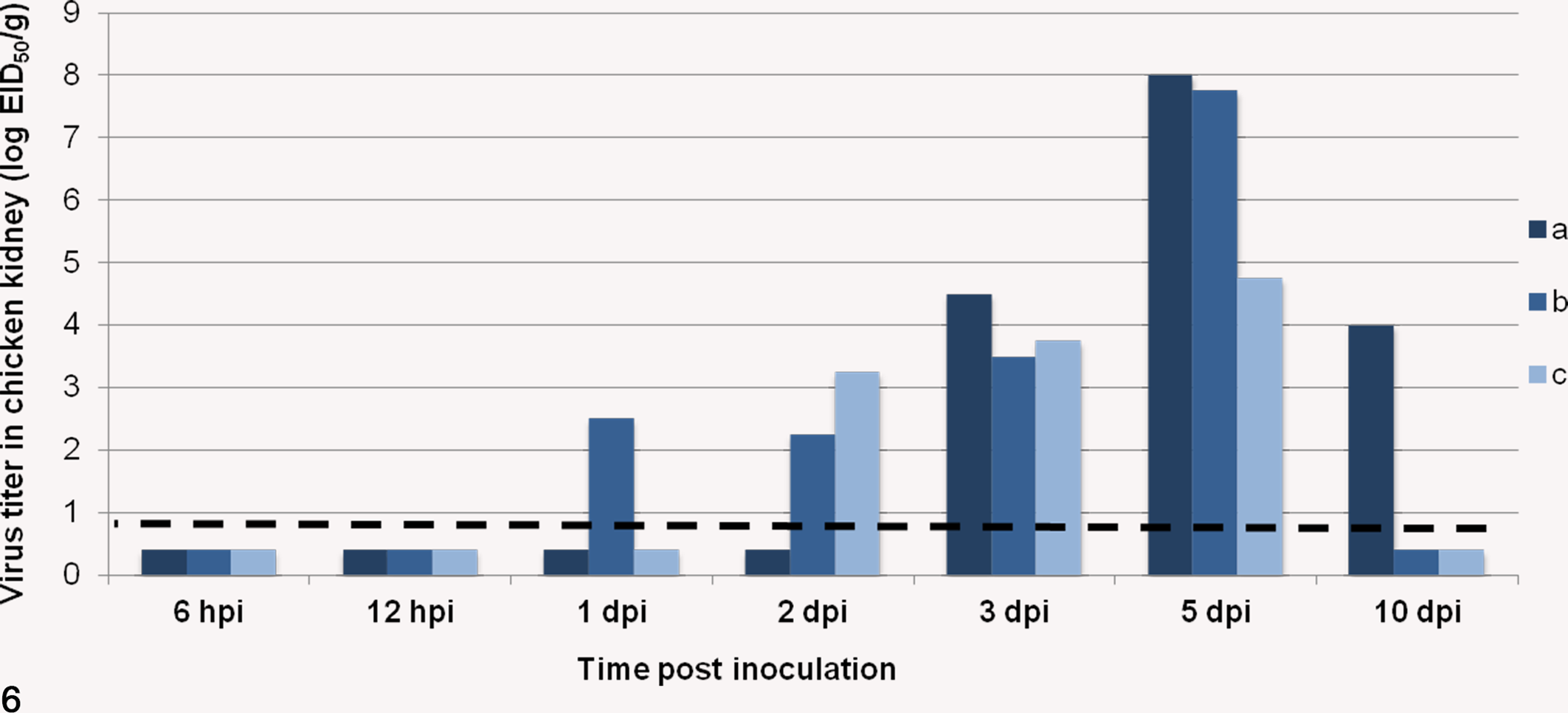
Individual viral titers in chicken kidneys at indicated times postinoculation. Kidneys from 3 individual chickens evaluated at each time point are represented as a, b, and c. The dashed line represents the virus detection limit. Undetectable samples were given a half of detection limit value. Virus titers were determined in 10-day-old embryonated specific pathogen-free chicken eggs and are presented as EID50 per gram. dpi, days postinoculation; hpi, hours postinoculation.
This study investigated the pathogenesis of NDV in the kidney of chickens after experimental infection. To our knowledge, no previous studies have addressed the relationship between histopathological alterations in chicken kidney tissues and NDV replication. The clinical signs were mild and compatible with our previous report. 2 Also, similar findings were observed for virulent NDV strains that do not produce much clinical disease, 6,16 although those previously studied viruses were classified as virulent strains based on their ICPI and F protein cleavage site. 16 Dortmans et al 5 mentioned that intracerebral inoculation, not being the natural way of infection, may lead to a difference in ICPI value compared with the natural route of infection. In addition, NDV virulence is multigenic, and the F gene is not the only key player in NDV virulence. 5,16
Nephrotropism of the 9a5b NDV isolate in the chicken kidneys was confirmed by IHC, RT-PCR, and virus isolation from an early time after infection (1 dpi). Virus detection was consistent among these 3 methods and started at 1 dpi, peaked at 5 dpi, and diminished at 10 dpi. The histopathological changes in the kidneys of infected chickens were consistent with the viral replication. The 9a5b NDV isolate caused severe degeneration and necrosis in the renal epithelium with positive viral immunostaining in both the tubular epithelium and mononuclear cells as previously reported for other birds. 2,3,9,12,16 The virus distribution was in the cortex and medulla of kidney as previously described. 9 Moreover, in this study, positive viral immunostaining was observed occasionally in some glomerular cells.
In this study, NDV tubulointerstitial nephritis can be classified into cortical tubulointerstitial nephritis and intratubular medullary cone nephritis as previously described in the case of AIV infection in chickens. 17 The 9a5b NDV isolate caused more severe tubulointerstitial nephritis in the renal medulla than in the cortex, similar to findings with IBV infection in chickens. 1 In humans, the medulla is more susceptible to infection than the cortex due to the low pH, high osmolality, and high concentration of ammonia in the medulla. 14 In birds, the susceptibility of the renal cortex and the medulla to infection is unknown as the pH is variable. 8 Tubular injury in the medulla more likely results from plugging of the lumina by cell debris, calcified materials, or both, subsequently increasing inflammation as proposed by others in the case of AIV infection in chickens. 17 Loss of renal function combined with water deprivation during the peak of clinical signs may enhance the effect.
CD3-positive cells were the main inflammatory component in chicken kidneys as previously reported in chicken brain during NDV infection. 7 Infiltration of CD3-positive cells in the infected kidneys was associated with a decrease or complete absence of viral replication at 10 dpi.
In conclusion, the 9a5b NDV mutant isolate showed a nephrotropism to chicken kidneys early after infection (1 dpi). Tubulointerstitial nephritis was more severe in the renal medulla than in the cortex, and the local inflammatory response may play a role in suppressing viral replication. Lesions that were caused by NDV in the kidneys of chickens were similar to those caused by AIV, IB, and ANV, and this highlights the importance of including NDV in the differential diagnosis of kidney disease in chickens.
Footnotes
Acknowledgments
We thank the Egyptian and Education Culture Office for providing a doctoral fellowship to the first author.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Egyptian government and the Laboratory of Veterinary Pathology, Tottori University, Japan.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
