Abstract
An 11-week-old, male, Staffordshire Bull Terrier had a history of generalized ataxia and falling since birth. The neurologic findings suggested a localization in the cerebellum. Magnetic resonance imaging of the brain was performed. In all sequences the area of the cerebellum was almost replaced by fluid isointense to cerebrospinal fluid. A complete necropsy was performed after euthanasia. Histologically, the lesion was characterized by extensive loss of cerebellar tissue in both hemispheres and vermis. Toward the surface of the cerebellar defect, the cavity was confined by ruptured and folded membranes consisting of a layer of glial fibrillary acidic (GFAP)-positive glial cells covered multifocally by epithelial cells. Some of these cells bore apical cilia and were cytokeratin and GFAP negative, supporting their ependymal origin. The histopathologic features of our case are consistent with the diagnosis of an ependymal cyst. Its glial and ependymal nature as demonstrated by histopathologic and immunohistochemical examination differs from arachnoid cysts, which have also been reported in dogs. The origin of these cysts remains controversial, but it has been suggested that they develop during embryogenesis subsequent to sequestration of developing neuroectoderm. We speculate that the cyst could have been the result of a pre- or perinatal, possibly traumatic, insult because hemorrhage, and tissue destruction had occurred. To our knowledge, this is the first description of an ependymal cyst in the veterinary literature.
An 11-week-old male, Staffordshire Bull Terrier with a history of intermittent generalized ataxia since birth was presented for examination. The dog was alert and clinically normal except for a gait characterized by generalized hypermetria and head bobbing with a tendency to fall. There was an obvious intention tremor and decreased proprioception in all limbs without lateralization. Cranial nerve function and spinal reflexes were normal. The neurologic findings suggested a lesion localized in the cerebellum. Magnetic resonance imaging (MRI) of the brain was performed with a 0.3 Tesla MRI unit (Hitachi Airis II; Hitachi Medical Systems, Düsseldorf, Germany). Sequences included a FSE T2 in sagittal and transverse orientation, a FE 3D T1 in transverse plane, a FE 3D MPR (high-resolution T1w gradient echo), a FLAIR (cerebrospinal fluid-suppression), and a BASG-sequence in dorsal plane. In all sequences the area where the cerebellum is normally located was almost completely filled with fluid isointense to cerebrospinal fluid (CSF) (Figs. 1, 2). The left part of the vermis and the left ventral hemisphere of the cerebellum were reduced to approximately one fifth of their normal volume. These cerebellar remnants were deformed and irregularly shaped with concave and convex surfaces. The fourth ventricle seemed to be separated from the fluid-filled cavity by a thin membrane and appeared normal in size. There were no signs of increased intracranial pressure. The dog was euthanized on the owner's request.
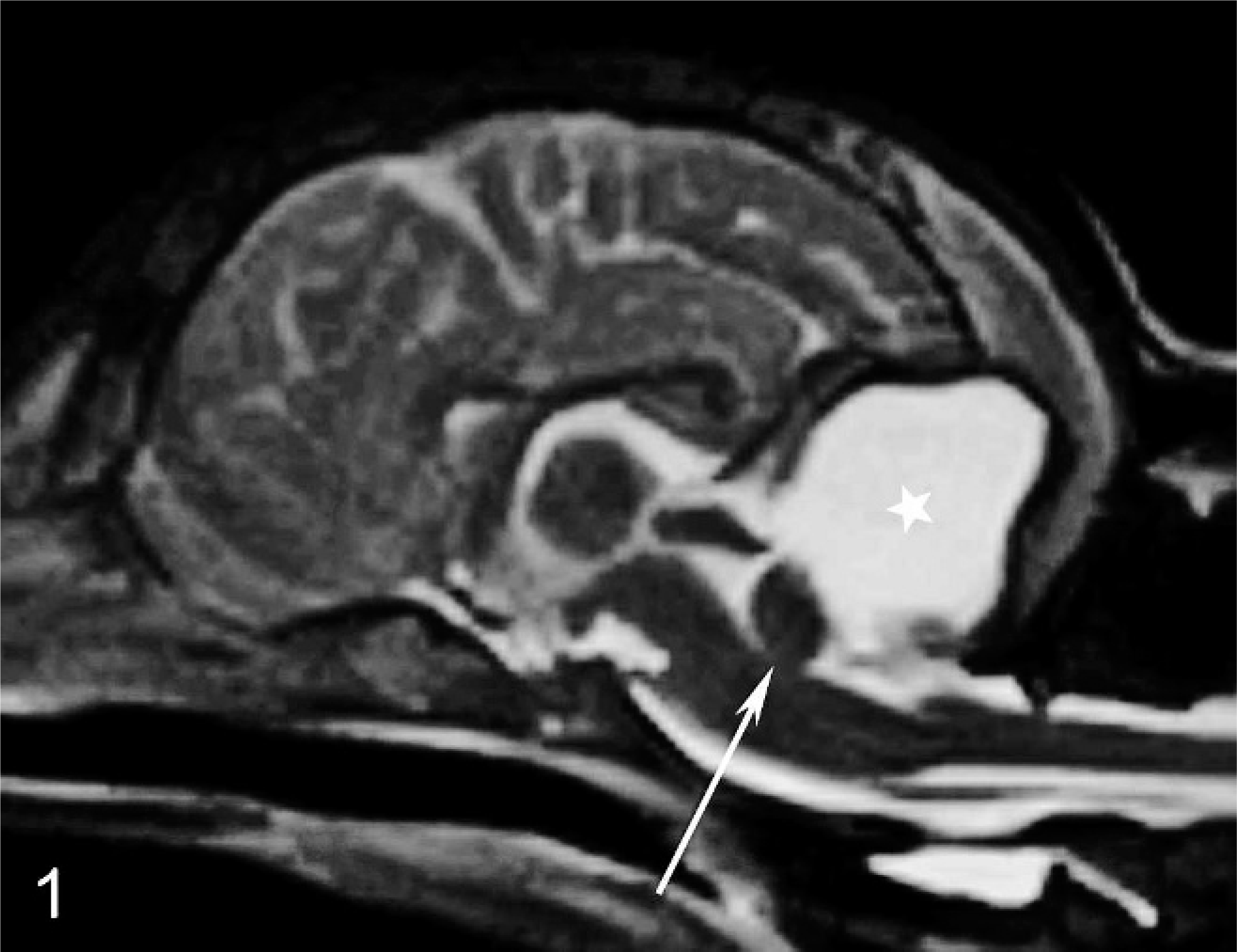
Midsagittal T2-weighted (FSE T2) magnetic resonance image. There is a hyperintense lesion (star) filling more than two thirds of the caudal fossa. Only small rests of the cerebellum (arrows) can be delineated. Neither the residual cerebellum nor the medulla oblongata shows signs of compression.
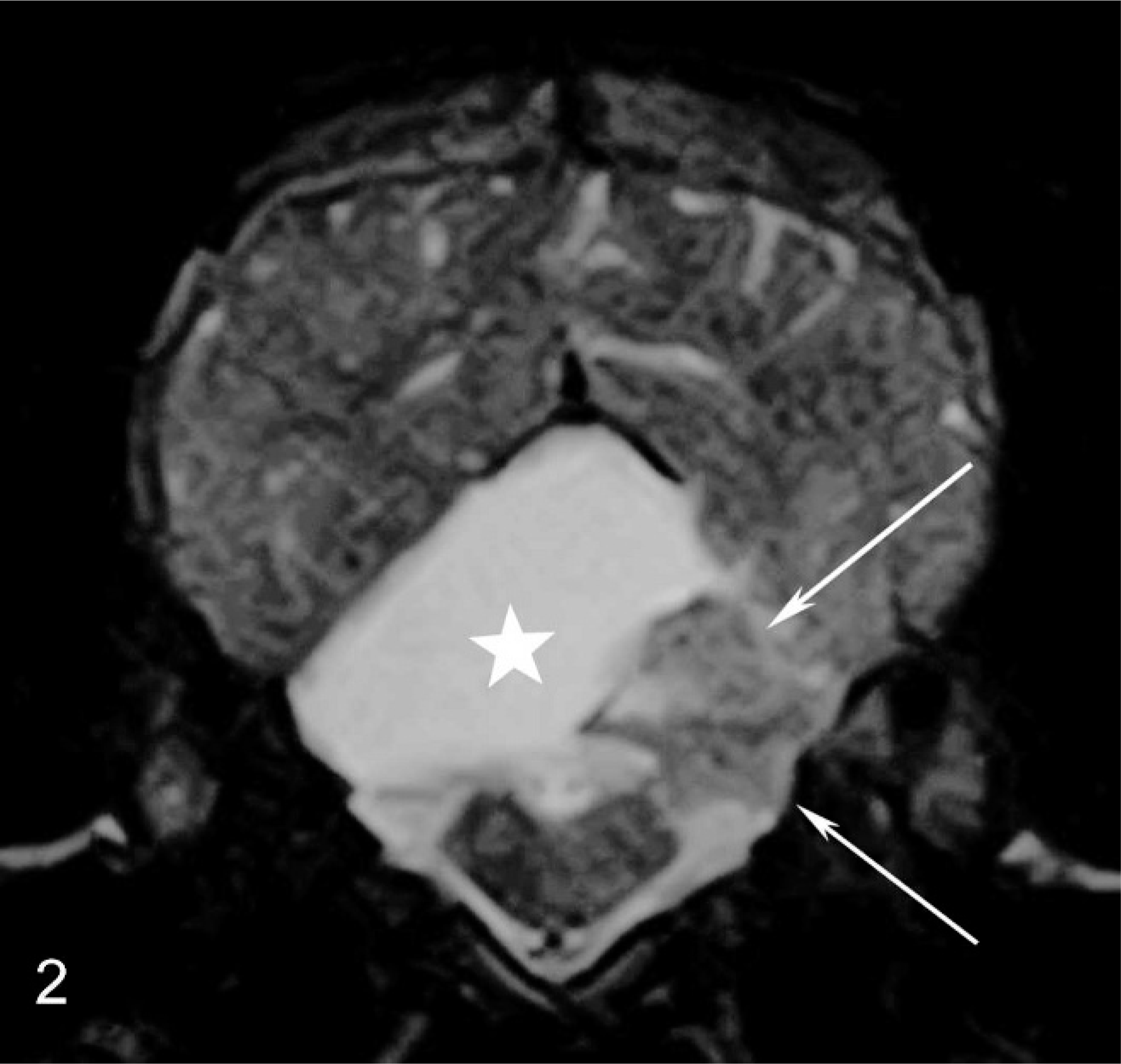
Transverse T2-weighted (FSE T2) magnetic resonance image at the level of the midcerebellum. Hyperintense lesion (star) and small rests of the cerebellum (arrows) can be delineated. Neither the residual cerebellum nor the medulla oblongata show signs of compression.
A complete necropsy performed shortly after euthanasia did not reveal any significant lesions outside the central nervous system. The cyst observed on the MRI examination was found to be collapsed following removal of the head and opening of the calvarium. Macroscopic lesions of the brain were confined to the cerebellum and consisted of an asymmetric and irregular reduction in size of both hemispheres and the vermis (Fig. 3). The right hemisphere was particularly affected, being almost completely absent. Within the left hemisphere the caudal portion of the paramedian lobe was absent. The defects were covered by collapsed membranes.
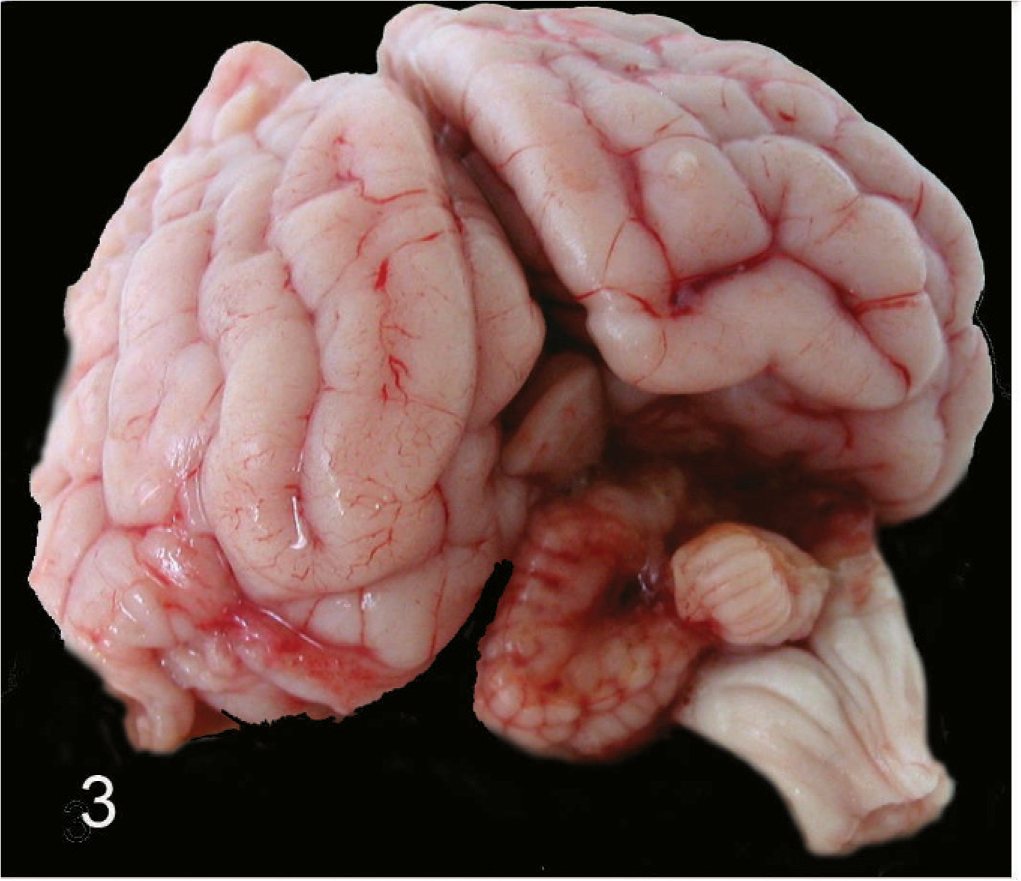
Brain of an 11-week-old Staffordshire Bull Terrier. Gross specimen, caudal view. Absence of most of the right cerebellar hemisphere, dorsal portion of the vermis, and paramedian lobe on the left.
The brain was fixed in 4% buffered formalin for 7 days. Representative specimens were embedded in paraffin and cut at 3 µm for histopathologic and immunohistochemical examination. Sections for histopathologic examination were stained with HE. Immunohistochemistry was performed with specific antisera against cytokeratin (1:500, DAKO, Glostrup, Denmark) and glial fibrillary acidic protein (GFAP; 1:1000, DAKO, Glostrup, Denmark). Sections were dewaxed, rehydrated, and treated with hydrogen peroxide 3% in methanol for 5 minutes to eliminate endogenous peroxidase activity. For antigen retrieval (cytokeratin), sections were treated by microwaving in citric acid buffer, pH 6.0 for 5 times at 100°C (780 W) for 5 minutes. To prevent unspecific reactions, tissue sections were blocked with normal serum (5%) in PBS for 20 minutes. Sections were then incubated with the primary antibodies either at 37°C for 1 hour (GFAP) or at room temperature for 45 minutes (cytokeratin). The avidin–biotin–complex (ABC) method was used for detection of a positive reaction (DAKO ChemMate detection kit) producing a brown color at the site of the reaction. In negative control sections, the primary antibody was substituted with an irrelevant antibody (rabbit IgG). Positive control sections for cytokeratin included epidermis. For GFAP, the adjacent brain tissue was used as an internal positive control.
Histologically, the lesion was characterized by extensive loss of cerebellar parenchyma affecting the gray and white matter of both hemispheres and the adjacent areas of the vermis (Fig. 4). The parenchyma was replaced by a collapsed cavity containing remnants of pale eosinophilic fluid, suggesting its high protein content (Figs. 4, 5). Toward the surface of the cerebellar defect, the cavity was confined by ruptured and folded membranes (Fig. 5) consisting of a layer of GFAP-positive glial cells (Fig. 6) covered multifocally by epithelial cells. Some of these cells bore apical cilia (Fig. 7) and were cytokeratin negative, supporting their ependymal origin. The cyst was overlaid by arachnoid membrane. Attenuated ependymal cells were also observed ventrally and medially, covering the remaining cerebellar parenchyma. Within the cerebellar folia bordering the cyst, the layered architecture was blurred because of a loss of Purkinje cells and a marked decreased density of the granule layer associated with gliosis and accumulation of hemosiderin-laden macrophages (Fig. 7). A clear communication between the cyst and the fourth ventricle was not observed. The medullary velum was thinned on the right side as compared with the left and was markedly deviated dorsally, suggesting distention of the fourth ventricle.
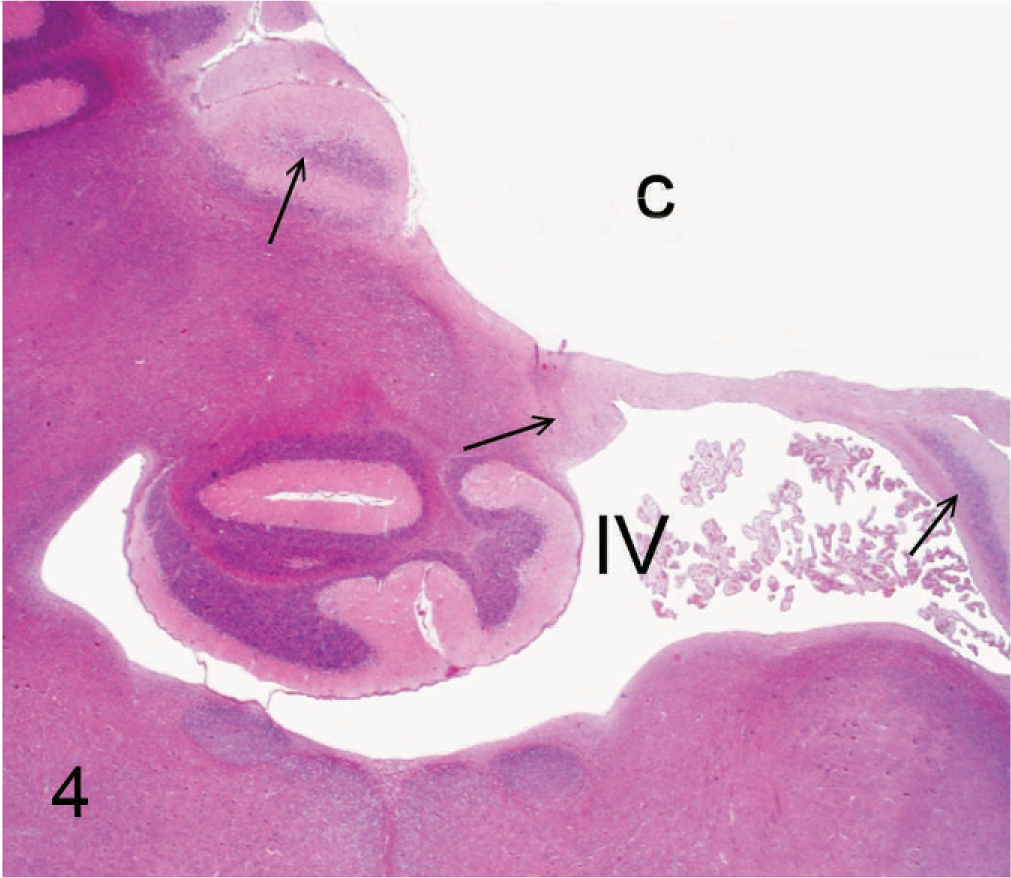
Cerebellar remnants and cyst; 11-week-old Staffordshire Bull Terrier. Histologic cross-section at the level of the middle part of the fourth ventricle (IV) and cerebellum. Cystic defect (C) in the right cerebellar hemisphere surrounded by remnants of atrophic cerebellar tissue (arrows). HE. Bar = 450 µm.
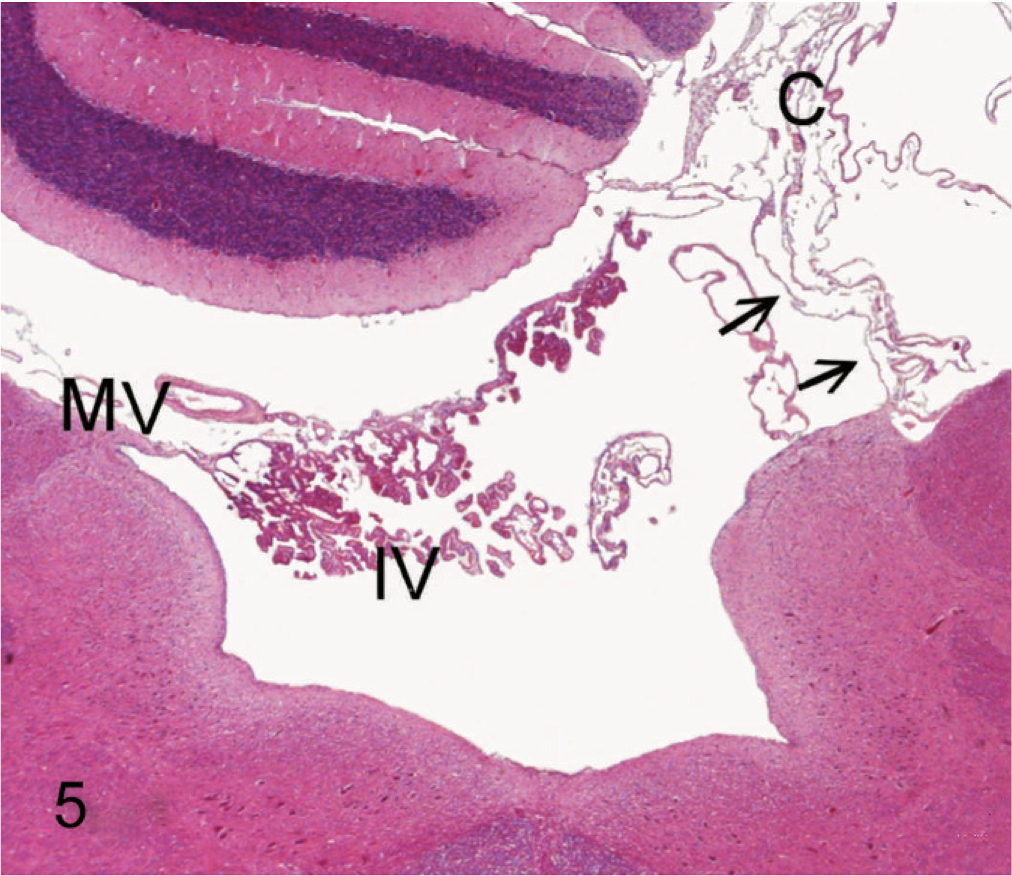
Fourth ventricle and cyst; 11-week-old Staffordshire Bull Terrier. Histologic cross-section through the caudal part of the fourth ventricle and cerebellum. Roof of the fourth ventricle is formed by the medullary velum (MV) with attached choroid plexus. The right medullary velum (arrows) is bulged dorsally with enlargement of the fourth ventricle. At the level of the right cerebellar hemisphere there is a stack of folded membranes derived from the collapsed cyst (C). HE. Bar = 250 µm.
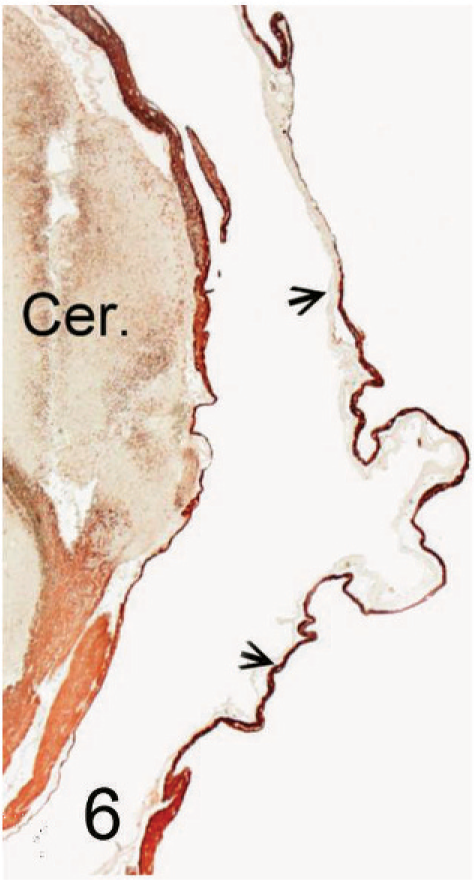
Ependymal cyst; 11-week-old Staffordshire Bull Terrier. Cross-section of a portion of the cyst wall overlying the cerebellum (Cer) showing intense positive staining for astroglial fibers (arrows). GFAP immunostain. Bar = 100 µm.
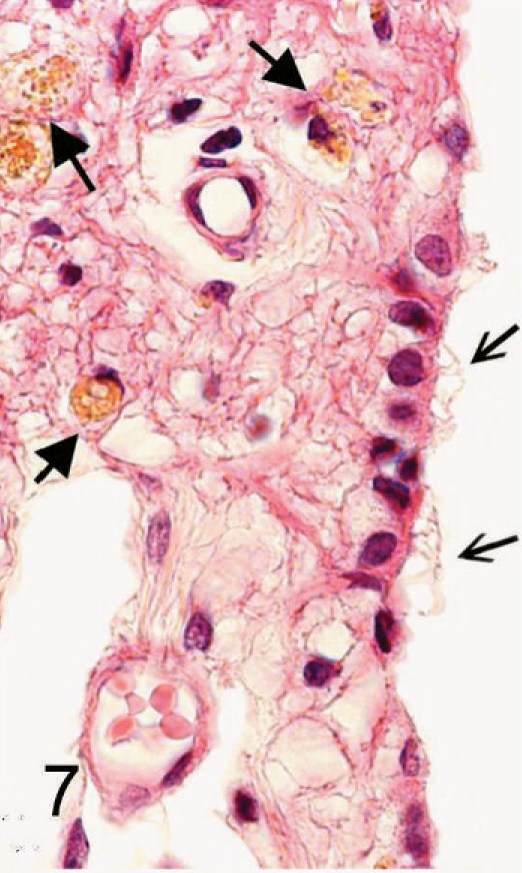
Ependymal cyst; 11-week-old Staffordshire Bull Terrier. Portion of the wall of the cyst covered with ciliated epithelium (open arrows) on the luminal side. Underlying gliotic tissue contains several hemosiderin laden macrophages (closed arrows). HE. Bar = 30 µm.
The classification of intracranial cysts in literature is not uniform. 2– 5, 7, 9, 10, 12, 13 However, according to the current World Health Organization classification of human brain tumors, benign cystic lesions in the nervous system are classified into Rathke cleft cysts, epidermoid cysts, dermoid cysts, colloid cysts of the third ventricle, enterogenous cysts, and neuroglial cysts. 8 Ependymal cyst is a more specific term for neuroglial cysts that are lined by ciliated epithelial cells resting directly upon brain parenchyma or a layer of astroglia rather than a basement membrane or connective tissue wall. 3 Some authors also use the term glioependymal cyst for these benign lesions. The histopathologic features of our case are consistent with the diagnosis of an ependymal cyst. Its ependymal nature demonstrated by histopathologic and immunohistochemical examination differs from arachnoidal cysts, which have been reported in dogs. 14 The latter are entirely surrounded by an arachnoid membrane and lack a glial-limiting membrane or an ependymal lining. In humans ependymal cysts are rare, benign lesions that typically occur intraparenchymally and often paraventricularly in the central white matter of the temporoparietal and frontal lobes but also in the subarachnoid spaces, brainstem, and cerebellum. 12 In the posterior fossa, ependymal cysts in humans usually present with symptoms of raised intracranial pressure, such as headache and, less commonly, ataxia. 4 The origin of these cysts remains controversial, but it has been suggested that ependymal cysts develop during embryogenesis subsequent to sequestration of developing neuroectoderm. 12 These cysts may originate from diverticula isolated during development of the neural tube, or from subarachnoidal glial heterotopias that contain an ependymal element. 1, 5 A cyst can also arise in any area of brain that has been damaged by hemorrhage, infarction, infection, trauma, or surgery. 11 Abnormally developed areas of the brain may be conducive to cyst formation. Chick embryos experimentally exposed to lead are documented to form cerebral cysts after toxin-induced hemorrhages. 6 The presence of marked gliosis and many hemosiderophages in the adjacent cerebellar tissue in our case clearly suggests that hemorrhage and tissue destruction had occurred. Those could be explained by vascular disruption and compression caused by the expansion of the cyst during brain development. Alternatively, the cyst could have been the result of a pre- or perinatal, possibly traumatic insult leading to tissue necrosis and sequestration of neuroectoderm with subsequent cyst formation.
To our knowledge, this is the first description of an ependymal cyst in the veterinary literature.
