Abstract
Cloning of cattle, sheep, and mice by somatic cell nuclear transfer (SCNT) can result in apparently healthy offspring, but the probability of a successful and complete pregnancy is less than 5%. Failures of SCNT pregnancy are associated with placental abnormalities, such as placentomegaly, reduced vascularisation, hypoplasia of trophoblastic epithelium, and altered basement membrane. The pathogenesis of these changes is poorly understood, but current evidence implicates aberrant reprogramming of donor nuclei by the recipient oocyte cytoplast, resulting in epigenetic modifications of key regulatory genes essential for normal placental development. The purpose of this review is to provide an overview of the anatomic pathology of abnormal placentae of SCNT clones and to summarize current knowledge concerning underlying pathogenetic mechanisms.
Introduction
In cloning by somatic cell nuclear transfer (SCNT), the nucleus of a donor somatic cell is transferred into an enucleated and unfertilized recipient oocyte. 102 Since the first success of cloning in sheep, 108 viable offspring have been obtained by SCNT in ungulates (cattle, 32 goat, 4 mule, 115 horse, 33 pig, 84 and mouflon 65 ), rodents (mouse 104 and rat 112 ), lagomorphs (rabbit 13 ), and carnivores (cat 92 and dog 59 ). Such cloning has many potential benefits and research applications, including study of genetic and epigenetic mechanisms underlying developmental biology, aging, and carcinogenesis 67 ; generation of organs for xenotransplantation; development of transgenic animals for production of valuable recombinant proteins; and preservation of endangered species. 103
The major impediment to the practical use of SCNT technology is that less than 5% of SCNT embryos develop normally to term. 41 In cattle and sheep, many established SCNT pregnancies are lost in the first trimester; 50 to 70% loss in that period is reported for cattle. 108 In contrast, pregnancy failure in ewes and cows with in-vitro fertilized embryos average 5 and 11%, respectively, compared with 1 to 3% among natural pregnancies. 29, 40, 64 In mice, only 50% of SCNT embryos develop to the blastocyst stage, with 97 to 99% of pregnancies failing at term, 76, 77, 104, 105 compared with 30% after in-vitro fertilization. 71 The reasons for such losses are not well understood, but abnormal placental development is a consistent feature of SCNT pregnancies.∗ This review presents current information on the pathology of SCNT placentae in cattle, sheep, and mice, the species in which SCNT cloning is best characterized.
Morphologic Pathology of SCNT Placentae
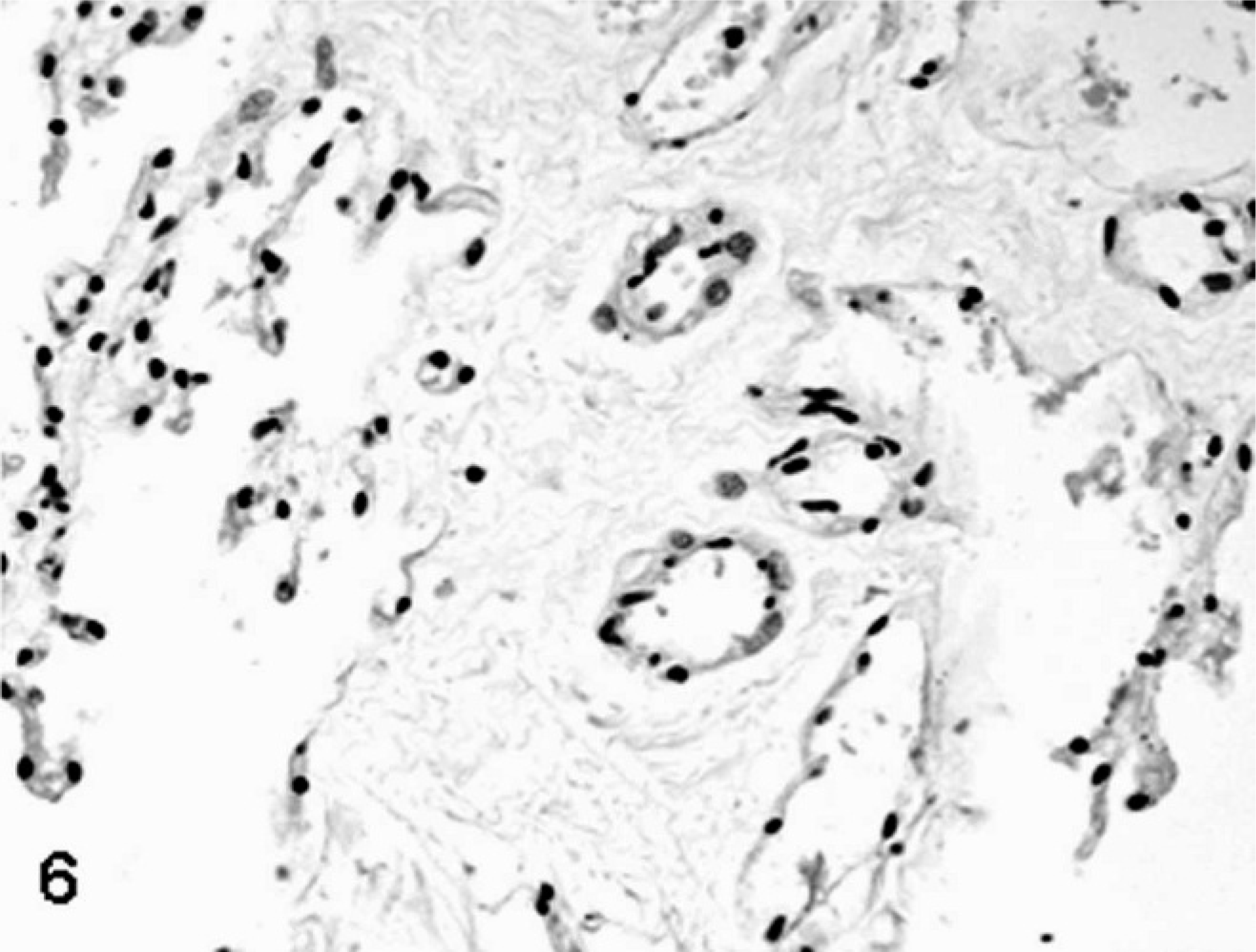
SCNT placentae have been examined at various stages of pregnancy in slaughtered, sacrificed, or C-sectioned animals (early pregnancy) and after delivering of SCNT full-term fetuses by natural route (late pregnancy). SCNT placentae collected from slaughtered ruminants are grossly characterized by reduced number and/or abnormal shape of caruncles and cotyledons. 22, 39, 43, 60 The major histologic finding, especially in sheep, is severely reduced placental vascular development evident in bovine placentae collected from sacrificed animals at 30 days of gestation and in ovine placentae at 35 days. 22 Similar lesions were also found at later stages of development. 22, 39, 43, 60, 82, 85, 86 In ovine SCNT placentae at term (147 days of gestation) with fetuses delivered alive and healthy, the number of capillaries per mm2 (50.7/mm2), evaluated by means of image analysis on semithin sections of resin-embedded samples, was lower than in control placentae (503/mm2), as was the capillary total area per villous area (0.01 versus 0.05). 81 Moreover, reduced numbers of vessels (Figs. 1, 2) were associated with relative immaturity of vessels of the placental villi, as determined by reduced or no immunohistochemical expression of von Willebrand factor and calponin (Figs. 3–6), 79 a marker of late differentiation in vascular smooth muscle cells. 56 Placental vasculogenesis was significantly reduced as indicated by lack of CD34 expression by individual foetal blood vessels, 79 similar to the abnormal placental vasculogenesis observed with spontaneous abortions in humans. 101
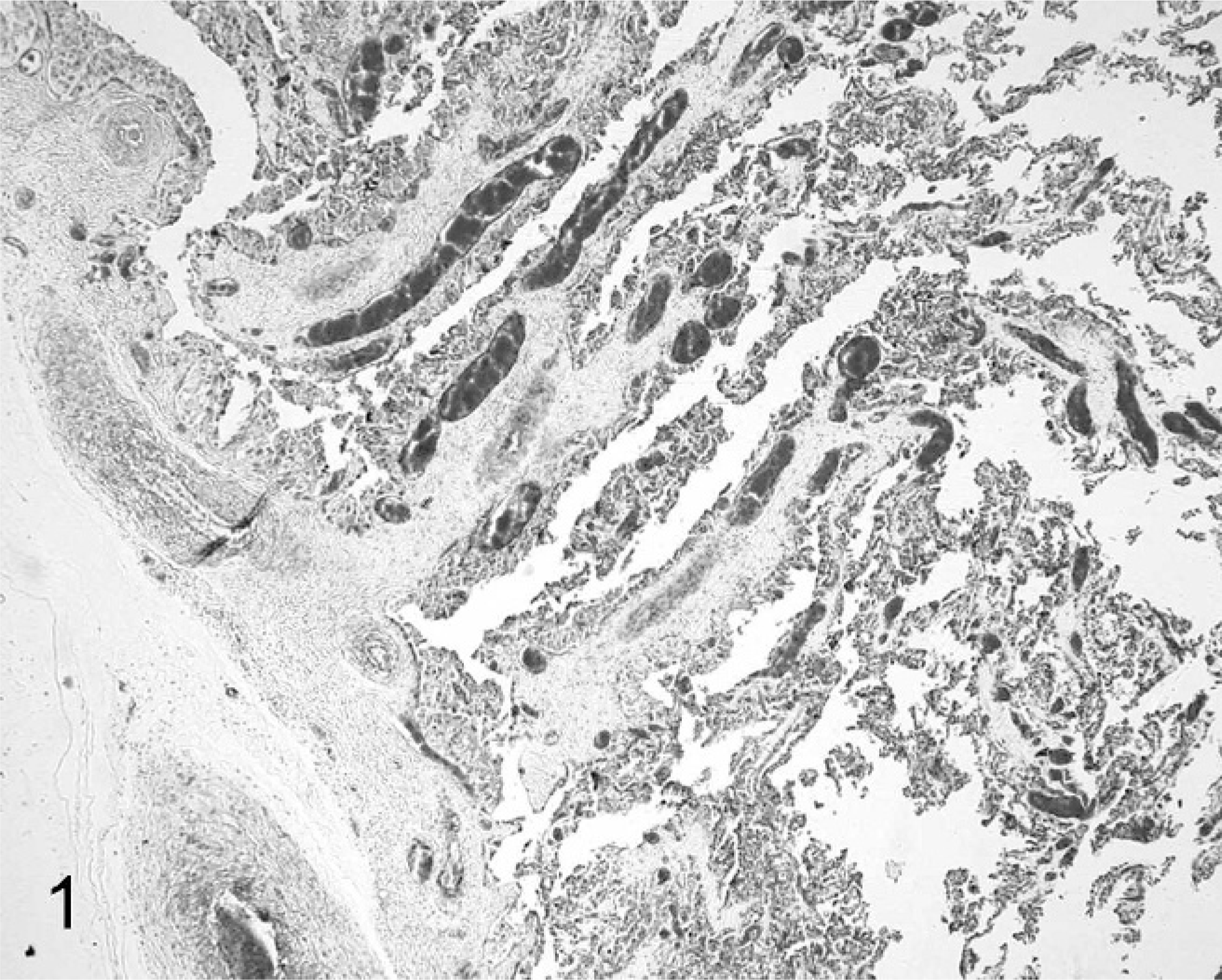
Placenta; sheep (normal pregnancy; day 147 of gestation). The cotyledonary area shows a high degree of vascularisation and integrity of trophoblastic layer. HE.
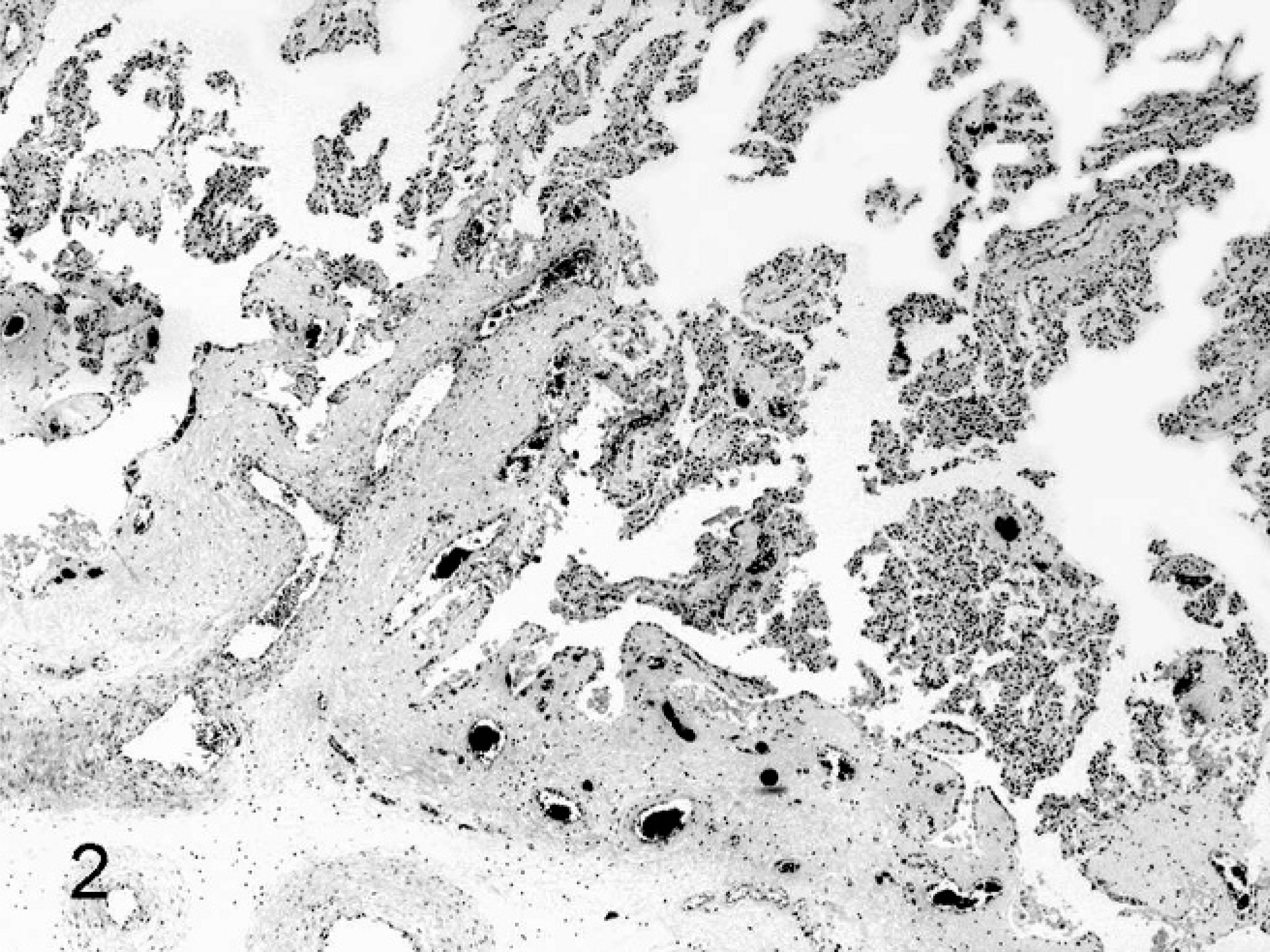
Placenta; sheep (somatic cell nuclear transfer pregnancy; day 147 of gestation). Notice the reduced number of cotyledonary vessels. HE.
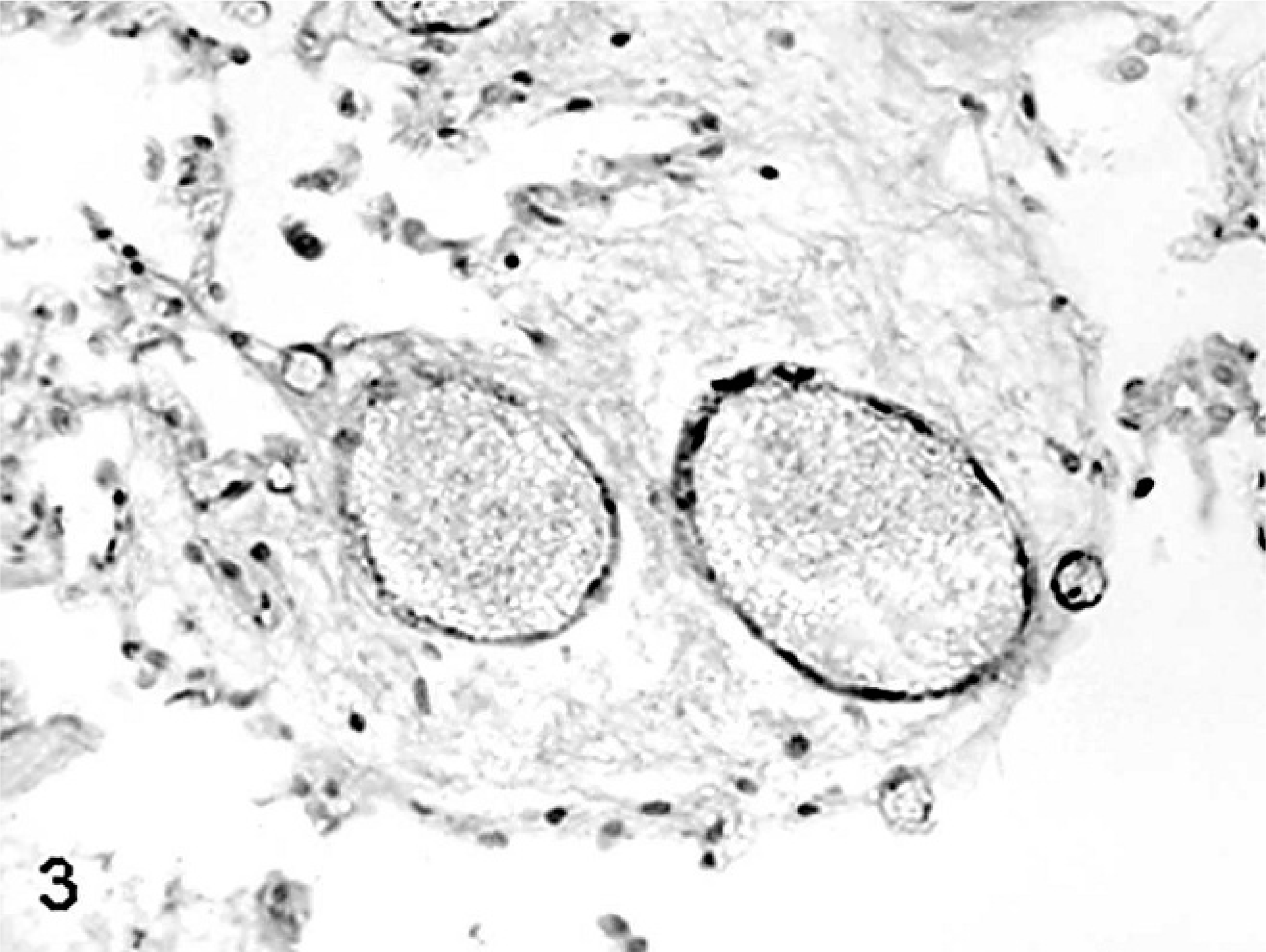
Placenta; sheep (normal pregnancy; day 147 of gestation). Positive immunohistochemical labeling for von Willebrand factor in endothelial cells of venules and capillaries. Streptavidin-biotin immunoperoxidase method, Papanicolau's hematoxylin counterstain.
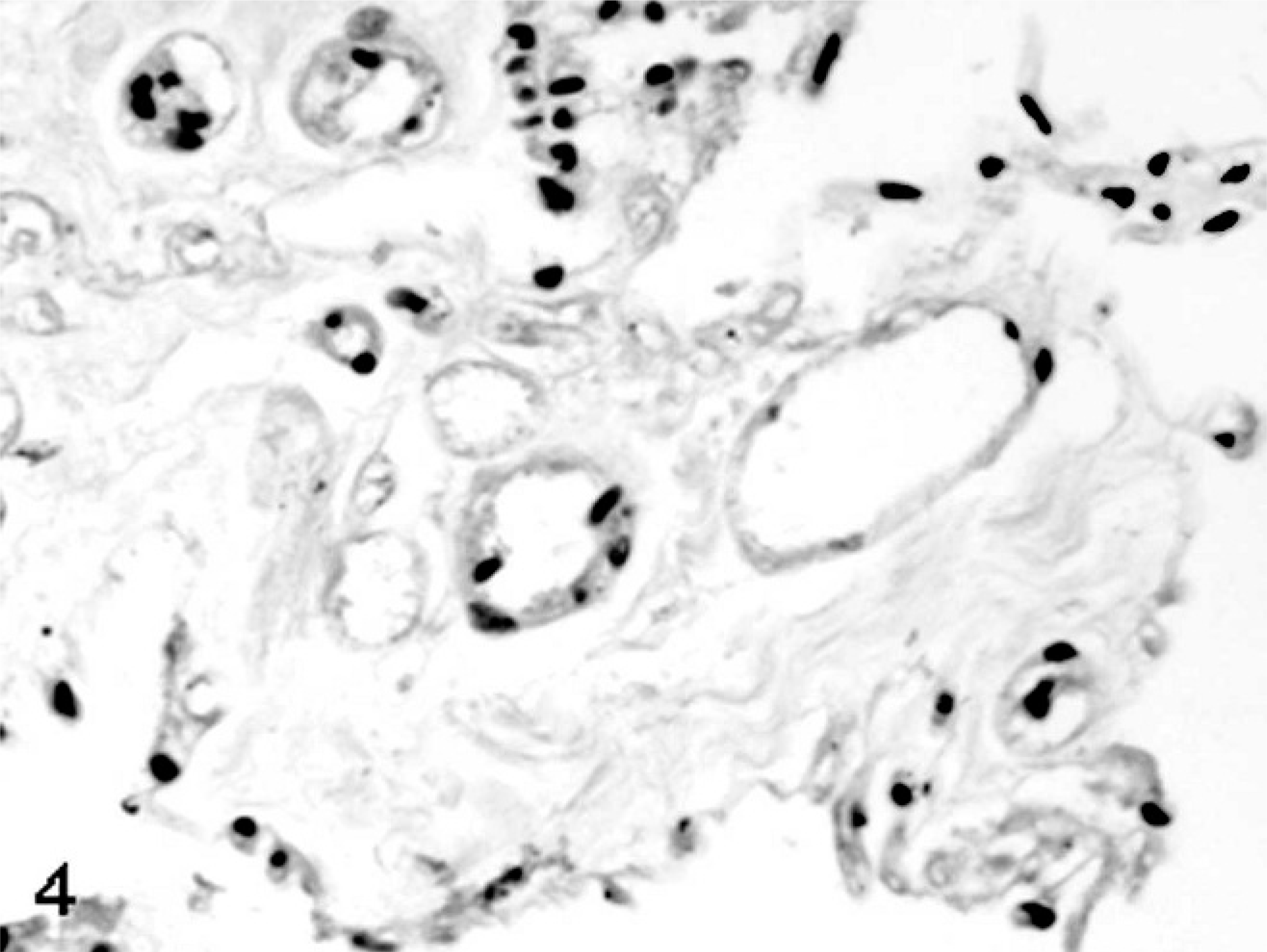
Placenta; sheep (somatic cell nuclear transfer pregnancy; day 147 of gestation). Lack of von Willebrand factor expression in endothelial cells of cloned placental tissue. Streptavidin-biotin immunoperoxidase method, Papanicolau's hematoxylin counterstain.
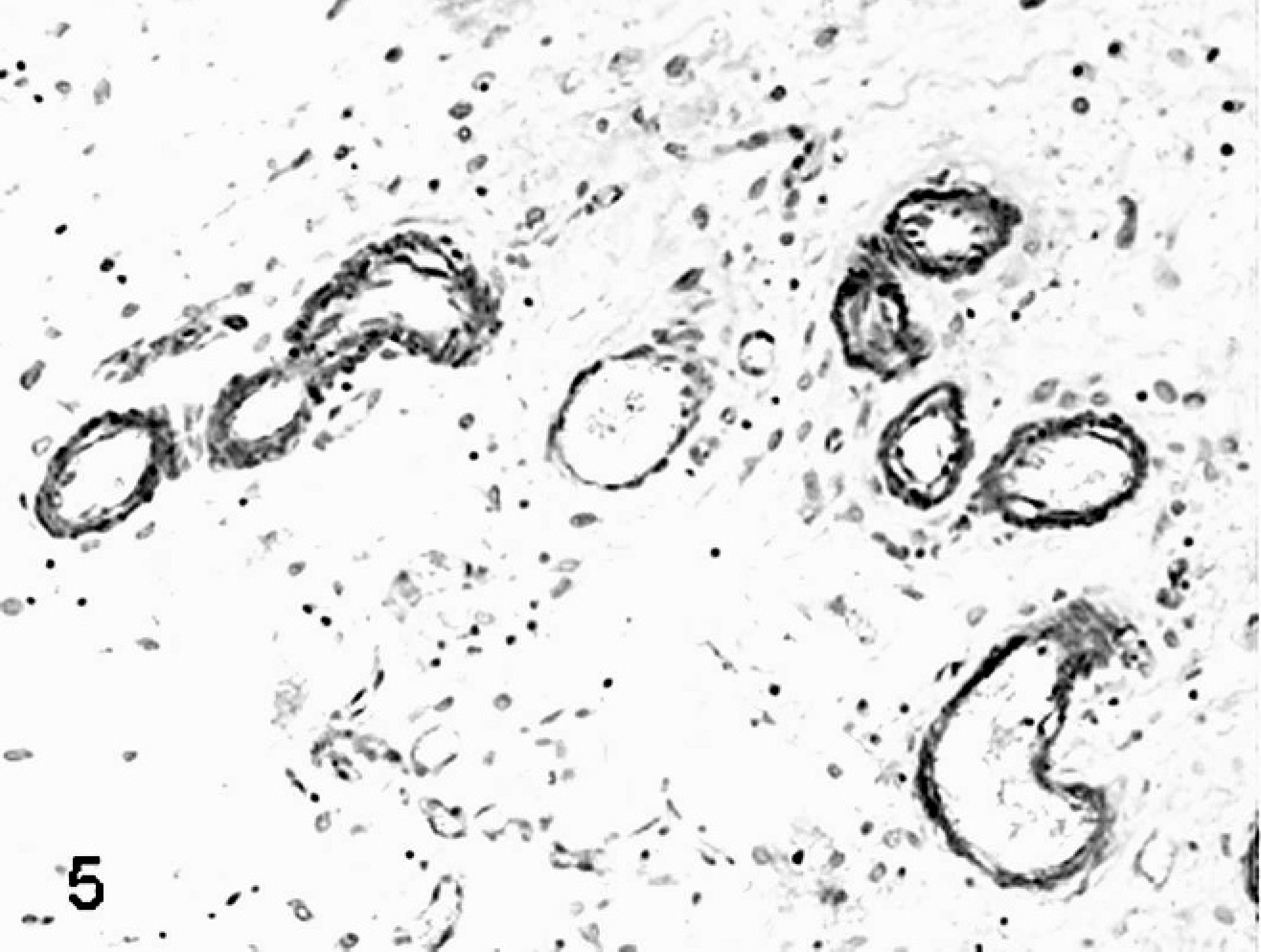
Placenta; sheep (normal pregnancy; day 147 of gestation). Vascular smooth muscle cells have strong cytoplasmic staining for calponin antigen. Streptavidin-biotin immunoperoxidase method, Papanicolau's hematoxylin counterstain.
Placenta; sheep (somatic cell nuclear transfer pregnancy; day 147 of gestation). Vascular smooth muscle cells show weak if any positive cytoplasmic staining for calponin immunohistochemistry. Streptavidin-biotin immunoperoxidase method, Papanicolau's hematoxylin counterstain.
In both ovine and bovine SCNT pregnancies, some investigators report that reduction in villous vascularization is accompanied by hypoplasia of trophoblastic epithelial cells, apoptosis and shedding of trophoblastic epithelial cells from foetal villi, 28 and/or reduced numbers of the trophoblast-derived binucleate cells. 28, 39, 64, 79 In these studies, caruncular and cotyledonary tissues were collected at 60 days of gestation from slaughtered cattle carrying SCNT motile and immotile embryos 39 and in sheep pregnancies at term with alive and healthy conceptus 28, 64, 79 or within 2 to 24 hours of fetal death. 28 Binucleate cells, which are homologues of the human extravillous trophoblast and rodent giant cells, normally migrate through the chorionic tight junctions to the uterine epithelium throughout gestation and produce several pregnancy-related proteins. 111 In contrast to the described hypoplastic phenotype, other investigators reported a threefold increase in SCNT placental binucleate cells in pregnant cattle slaughtered at gestation days 50 to 150 and carrying a viable fetus. 85, 86 The increased numbers of binucleate cells were associated with increased numbers of fetal trophectodermal mononucleate cells and maternal uterine epithelial cells, 87 resulting in so-called “large placentomes.” These are observed in surviving bovine SCNT pregnancies at 100 and 150 days of gestation and are associated with progressive pregnancy failure compared with the unchanged success rates after 50 days of gestation in artificially inseminated and in-vitro–fertilized animals. 60 Large placentomes are fist-like shaped, heavier, and thicker compared with flat, discoid placentomes generated from in-vitro fertilization or by artificial insemination. Mean caruncle weights (±SD) were 7.37 ± 0.92 g, 7.48 ± 0.90 g, and 11.52 ± 1.52 g and total caruncle weights were 802 ± 28 g, 811 ± 109 g, and 1,133 ± 86 g for artificially inseminated, in-vitro–fertilized, and SCNT pregnancies, respectively, at 150 days of gestation. 60
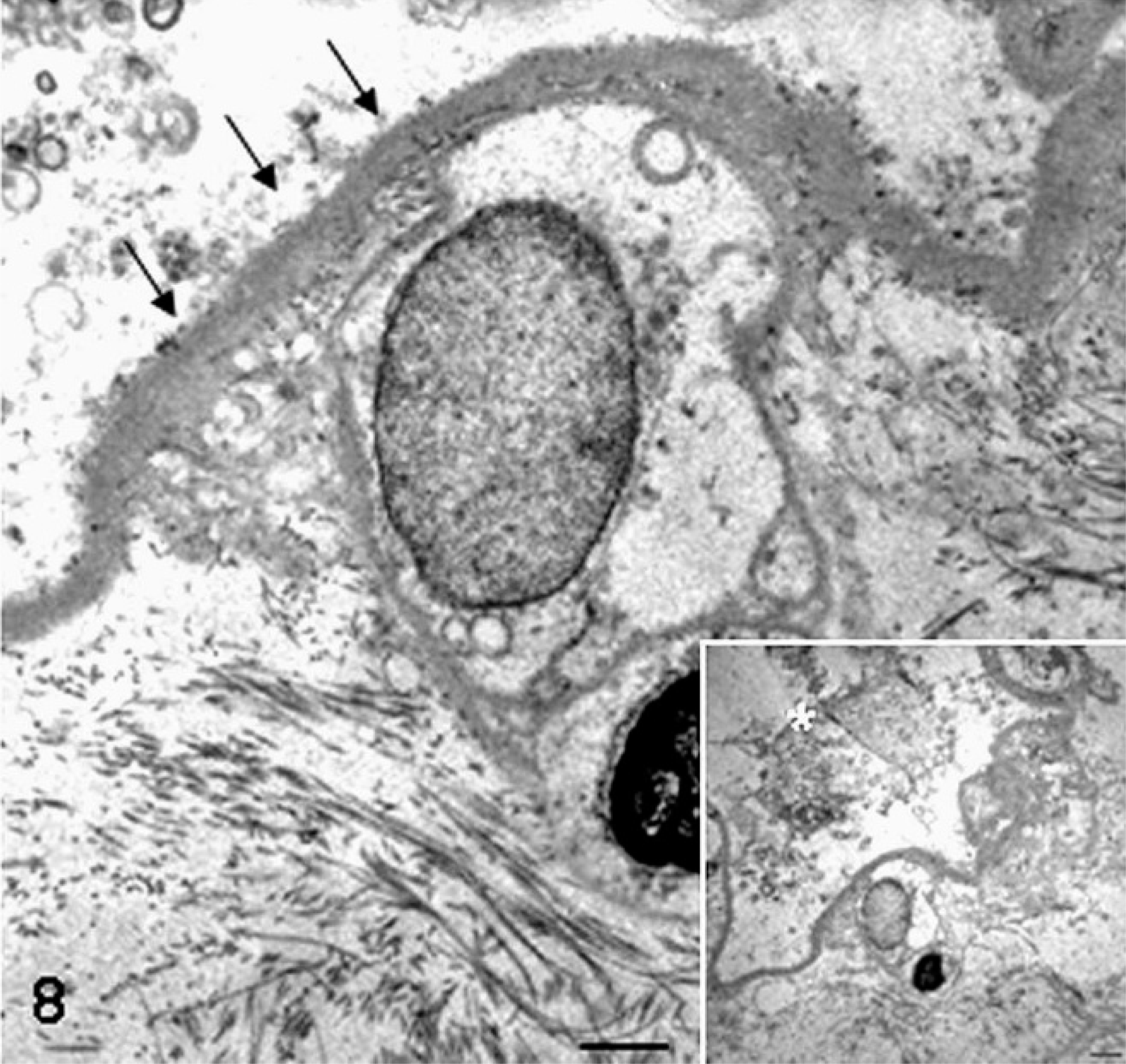
Stromal components of the placenta also are altered in bovine and ovine SCNT pregnancies. In cattle at day 60 of gestation, there is disruption of chorionic villi and caruncular septa, being irregularly and sparsely arranged, associated with reduced procollagen type I and collagen type III fibers. 39 These changes are linked to increased expression of heparanase, an extracellular matrix-degrading enzyme, 55 in intercotyledonary areas. 39 In sheep at term with alive and healthy fetuses, the subtrophoblastic basement membrane and capillary basal lamina include increased thickness, lamellation, and deposition of collagen fibrils (Figs. 7–10). 79 Such changes are attributed to increased deposition of basement membrane components associated with hypoxia. 31 SCNT placentae do not have histologic features typical of intrauterine embryonic and fetal death, stillbirth, and placental retention, such as stromal fibrosis of terminal villi, 25 subendothelial proliferation of undifferentiated or fibromuscular tissue, division of vascular lumina by septa, 1 coagulation necrosis, and/or infarction. 14 Moreover, the presence of activated caspase-3–positive apoptotic shed trophoblasts, normally absent in normal conditions, in ovine SCNT placentomes near (105–134 days of gestation) or at term (135–154 days) associated with alive conceptus or within 2 to 24 hours of fetal death suggest that histopathologic lesions occur prior to fetal loss. 28
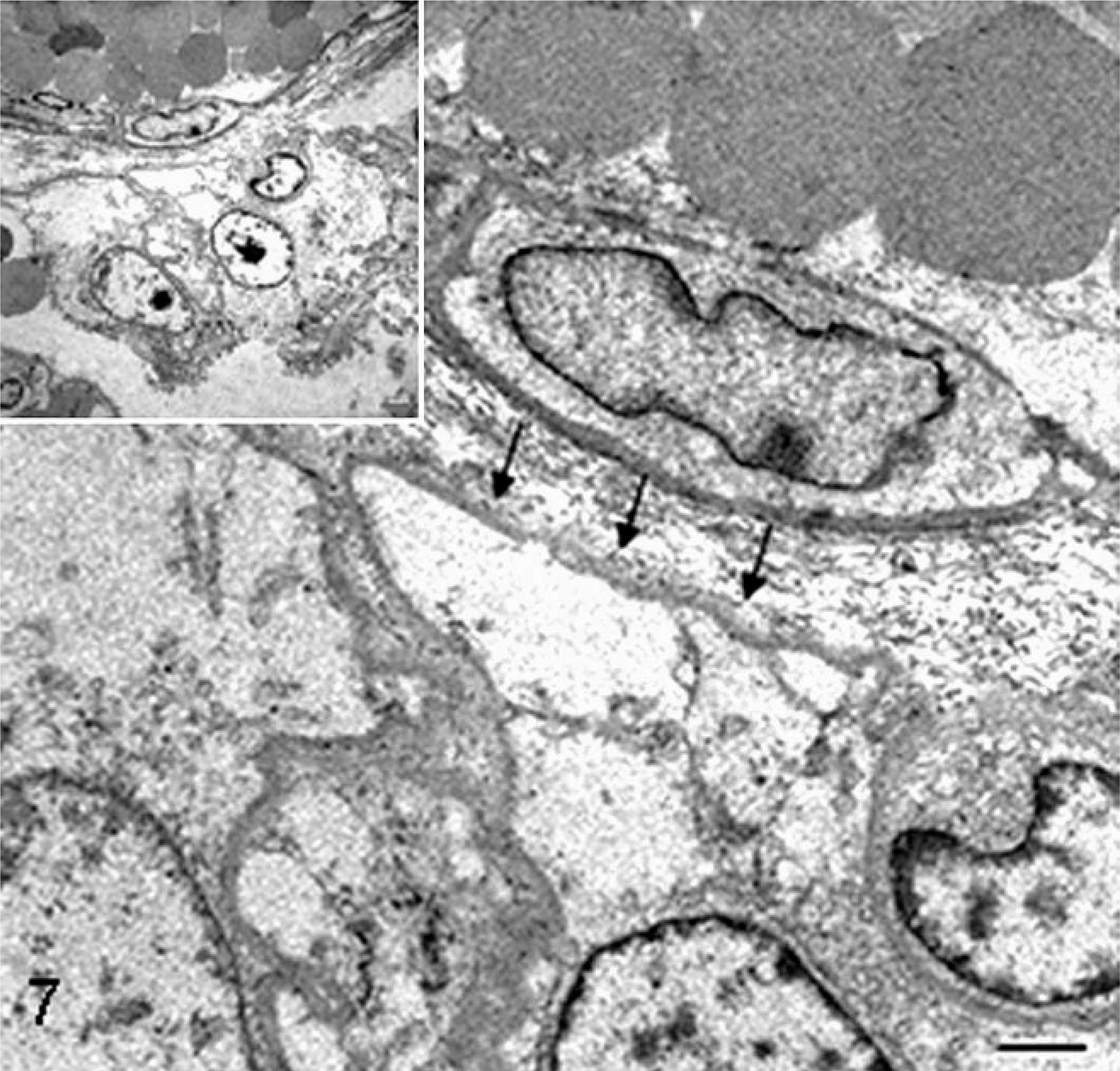
Placenta; sheep (normal pregnancy; day 147 of gestation). Electron microscopy reveals a thin subtrophoblastic basement membrane (arrows) between the trophoblastic layer (lower) and stromal collagen fibers and blood vessel (upper). Bar = 1.3 µm. Inset shows the same image at low magnification. Bar = 2.5 µm. Glutaraldehyde fixative, uranyl acetate, and lead citrate.
Placenta; sheep (somatic cell nuclear transfer pregnancy; day 147 of gestation). Transmission electron micrograph of the thickened subtrophoblastic basement membrane (arrows). Bar = 1.3 µm. In the inset showing the same image at low magnification, the focal lack and detachment of trophoblastic cells admixed with cellular debris are evident (asterisk). Bar = 2.5 µm. Glutaraldehyde fixative, uranyl acetate, and lead citrate.
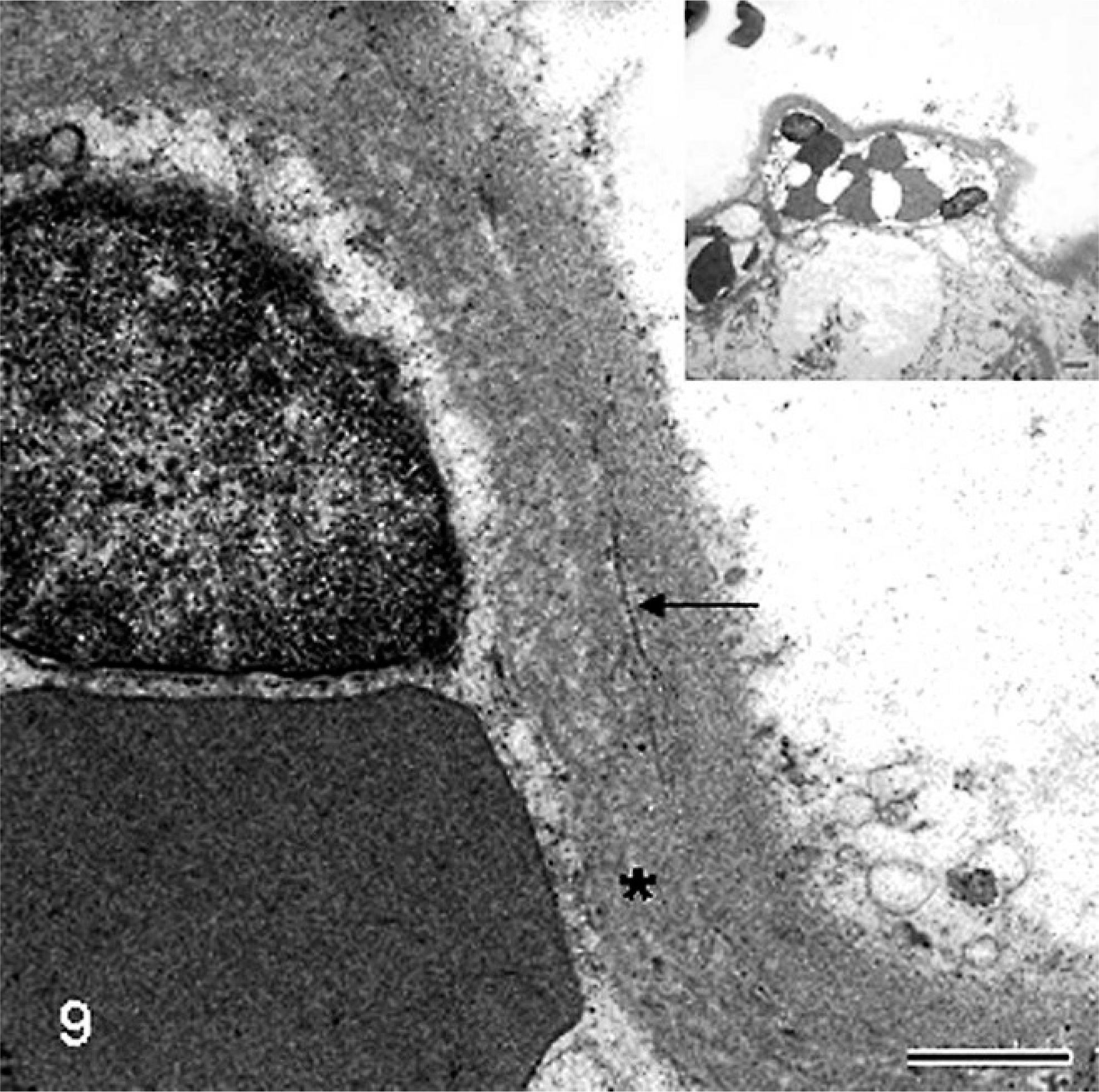
Placenta; sheep (somatic cell nuclear transfer pregnancy; day 147 of gestation). At the ultrastructural level, the thickened subtrophoblastic basement membrane is characterized by lamellation (asterisk) and focal deposition of scattered collagen fibers (arrow). Bar = 0.45 µm. Inset shows the same image at low magnification. Bar = 2.5 µm. Glutaraldehyde fixative, uranyl acetate, and lead citrate.
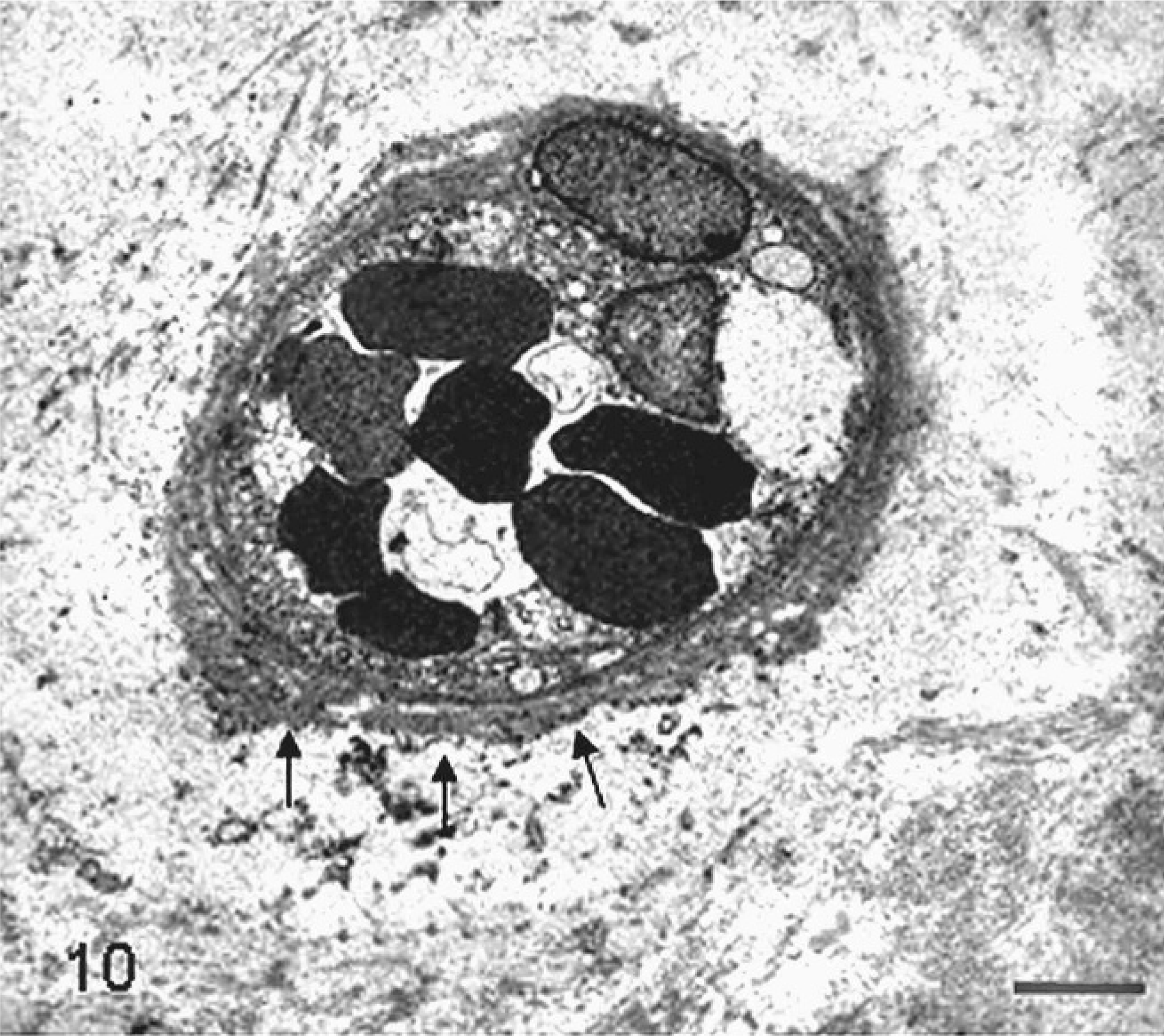
Placenta; sheep (somatic cell nuclear transfer pregnancy; day 147 of gestation). The basal lamina encircling capillary of the cotyledonary area is thickened and multistratified (arrows). Bar = 3.3 µm. Glutaraldehyde fixative, uranyl acetate, and lead citrate.
Even among the few bovine SCNT pregnancies that reach full term, many have abnormal placentae and peripartum neonates are malformed and/or die. 18, 44, 107 Abnormalities include hydroallantois, enlarged umbilical vessels, edematous placental membranes, placentomegaly, and increased fetal or birth weight. 11, 18, 44, 107 Increased fetal or birth weight occurs in 13.3% of SCNT births 3 and is collectively referred to as “large offspring syndrome” 109 or “abnormal offspring syndrome.” 26 Hepatomegaly, dermal hemorrhages, hydrocephalus due to fourth ventricle swelling in nuclear transfer (NT) ovine viable embryos recovered at 35 days of pregnancy, 22 foci of hepatic fatty accumulation, or hepatic congestion in bovine surviving NT fetuses at 100 and 150 days of gestation 60 were described. The basis of the fetal abnormalities is not clear. In view of the high prevalence of placental vascular and other changes, SCNT fetuses could suffer from impaired feto-maternal circulation.
In mice, placentomegaly with expansion of the junctional or basal layer, extensive proliferation of glycogen cells, and enlargement of trophoblast giant cells is a distinctive alteration in SCNT placentae (Figs. 11, 12). 71, 76, 77, 90, 91, 100 Structural modifications have been reported in mouse placental tissues collected by cesarean section at 18.5 days post coitum (dpc) 100 and after natural delivery at 19.5 dpc 71, 76 from SCNT pregnancies with live pups 71, 76, 100 and, in few cases, with fetuses that died in utero 76 or from respiratory distress after cesarean delivery. 100 Specifically, disorganization of the labyrinthine layer, characterized by irregular branching and dilation of foetal capillaries, and disruption of placental zonation with interdigitations of the boundary between the labyrinthine and basal layers have been described (Figs. 11, 12) 71, 76, 100 but also are associated with other oocyte micromanipulation techniques, such as microinsemination (sperm or spermatid injection) and pronuclear exchange. 71
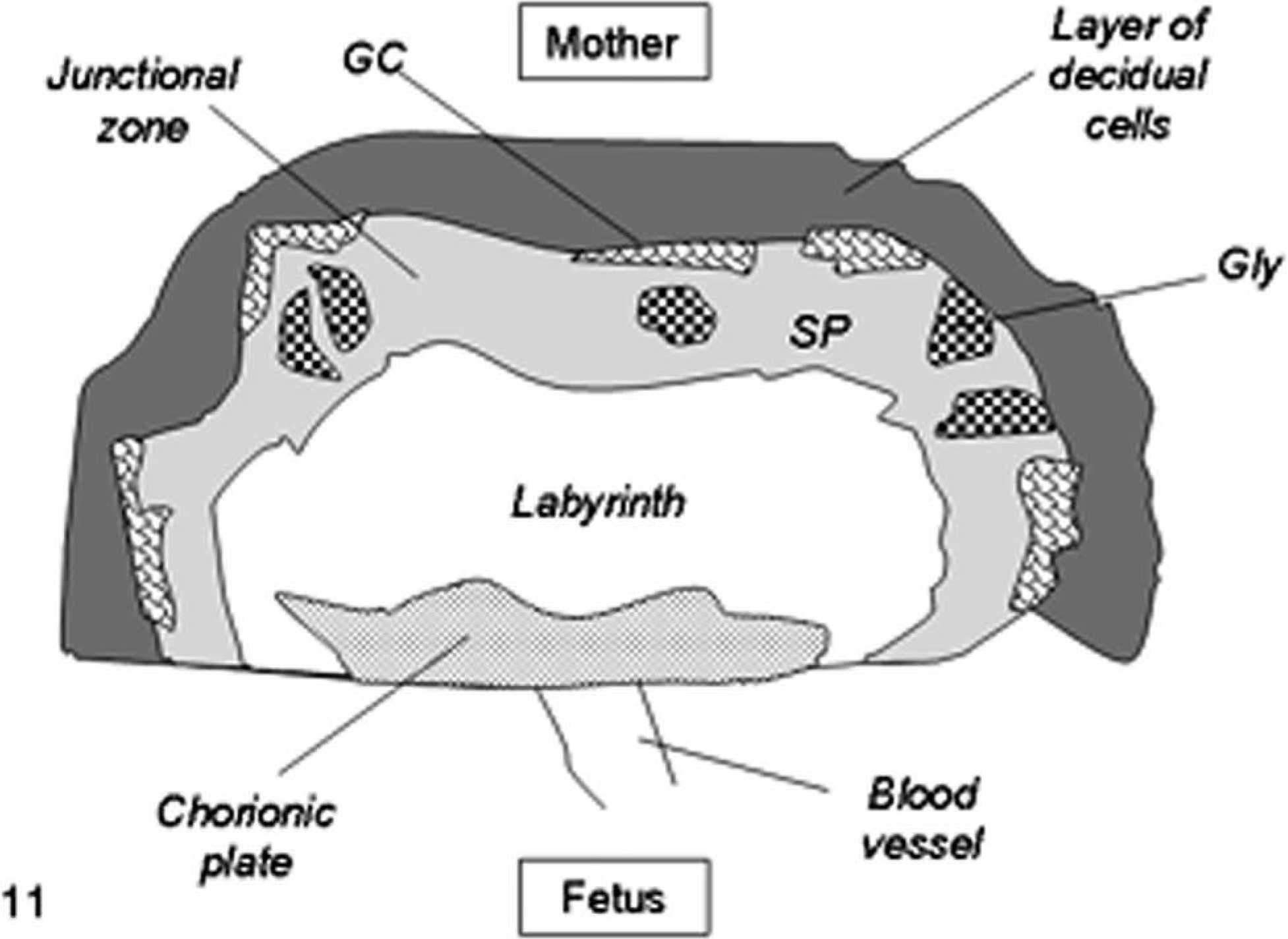
Schematic diagram of mouse placenta (normal pregnancy). The labyrinth or labyrinthine zone is closest to the fetus and consists of a complex interconnecting system of maternal blood spaces separated by trabecular cords of foetal tissue containing foetal capillaries. The junctional zone is composed of 2 cell types: spongiotrophoblast (SP) and glycogen cells (Gly). Trophoblast giant cells (GC) border the junctional zone and the maternal decidua.
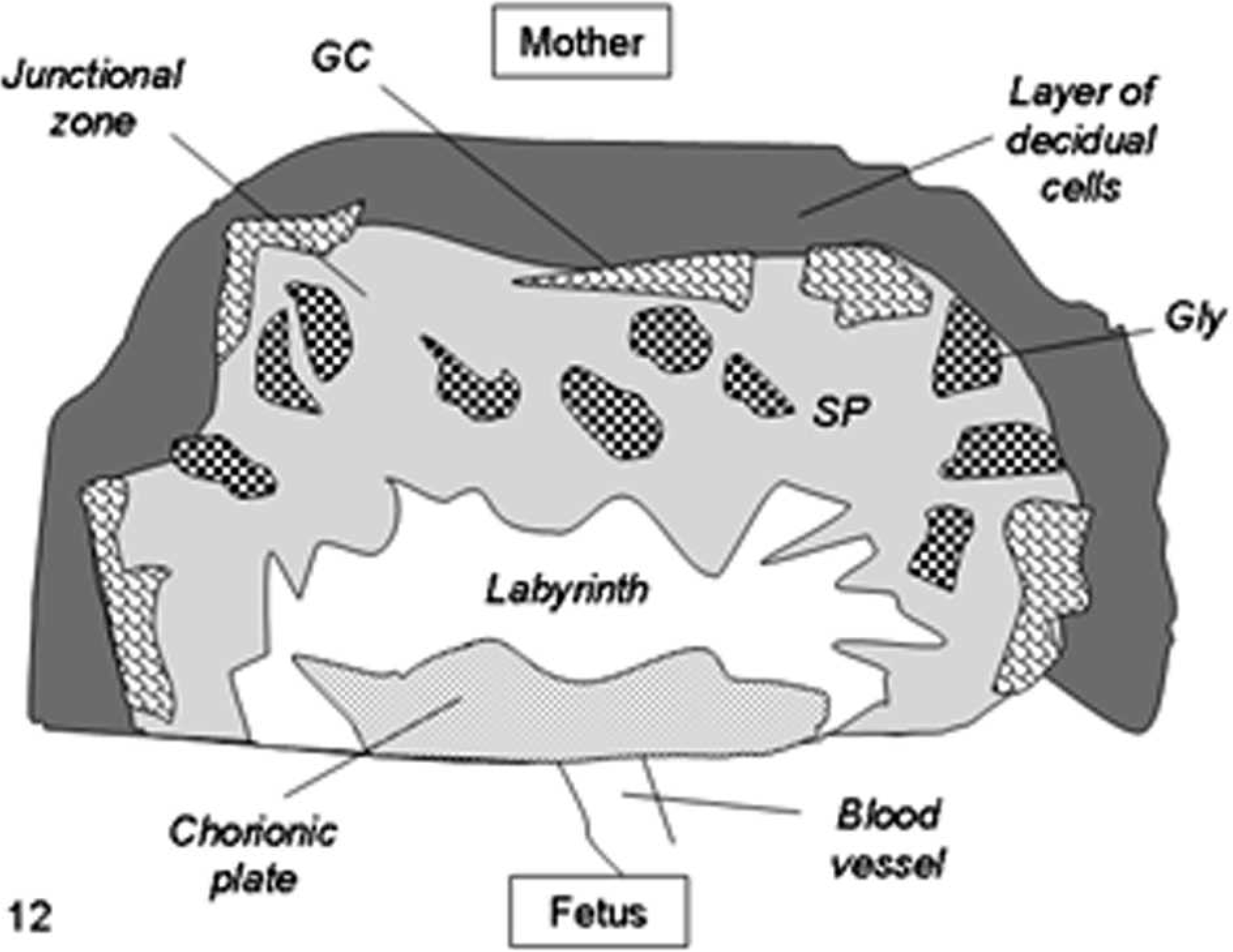
Schematic diagram of mouse placental modifications after somatic cell nuclear transfer, showing expansion of the junctional zone with proliferation of glycogen cells (Gly) and enlargement of trophoblast giant cells (GC) and the interdigitations of the boundary between the labyrinth and the junctional zone.
Temporal development of SCNT placental lesions is not well characterized. The earliest defective extraembryonic cell lineage detected is the epiblast. 52 At 7 dpc most of the NT embryos are developmentally delayed compared with controls and reveal 2 abnormal phenotypes: 1 with an abnormal rounded rather than elongated shape, resulting from an aberrant elongation of epiblast, and 1 called “large Exe” phenotype, characterized by an abnormal ratio between extraembryonic and embryonic regions, with only 14% of epiblast cells compared with 30% of control embryos. 52 The defective epiblast is at least partly responsible for early embryonic loss, because the described abnormal phenotypes are present also when normal embryonic stem cells are injected into NT blastocysts. The normal morphology of chimerae obtained from the aggregation of 8-cell–stage NT embryos with normal trophoblast cells reveals, on the contrary, that the defective tissue is the extraembryonic lineage. 52 Placentomegaly occurs between 12.5 and 19.5 dpc. 72, 91 Underdevelopment of the labyrinth and spongiotrophoblast is characterized by reduced expression of the spongiotrophoblast marker Tpbpa, observed at 10.5 dpc in somatic cloned concepti. 106
Pathogenesis
In the last years, several genetic and molecular studies have been carried out to identify key factors, molecular patterns, gene sequences, hormone-like proteins, and cytokines whose altered expression is linked with placental abnormalities in SCNT pregnancies (Table 1, Table 2, Figs. 13–15). Candidates are encoded mainly by pregnancy-related genes and include glycoproteins, growth factors, hormones, and hormone-like factors. 81 Proteins involved in the regulation of placentogenesis, fetogenesis, mammogenesis, steroidogenesis, and immune system activity, 2 such as chorionic somatomammotropin hormone 1, bovine pregnancy-associated glycoproteins 1 and 9, and bovine prolactin-related protein 1, are deregulated throughout the pregnancy in SCNT placentae. 39, 82 The reduced expression of these proteins might be responsible for inadequate preparation of the birth canal at parturition and poor mammary gland development in cows carrying SCNT embryos. 107 In SCNT placentae characterized by large placentomes with increased proliferation of feto-maternal cells, the expression of leptin in trophectoderm and uterine epithelial cells at days 50, 100, and 150 of pregnancy is evident, whereas placentomes after artificial insemination are immunonegative at all stages of gestation. 86 Leptin regulates different processes during fetal and placental development, including nutrient partitioning, invasion and proliferation of extravillous trophoblastic cells, metabolic processes, angiogenesis and hematopoiesis, and local immunomodulation. 86 In humans, low placental and cord blood levels of leptin are observed in fetal growth retardation cases. 5
Genes involved in cloned animal molecular placental pathology.


Official symbols of genes for each species; the numbers in parentheses refer to the species listed in the third column of the table.
Species in which the gene has been characterized and sequenced.
Summary of morphologic lesions of somatic cell nuclear transfer placentae in cattle, sheep, and mice and abnormal expression of related key factors, gene sequences, hormone-like proteins, and cytokines.
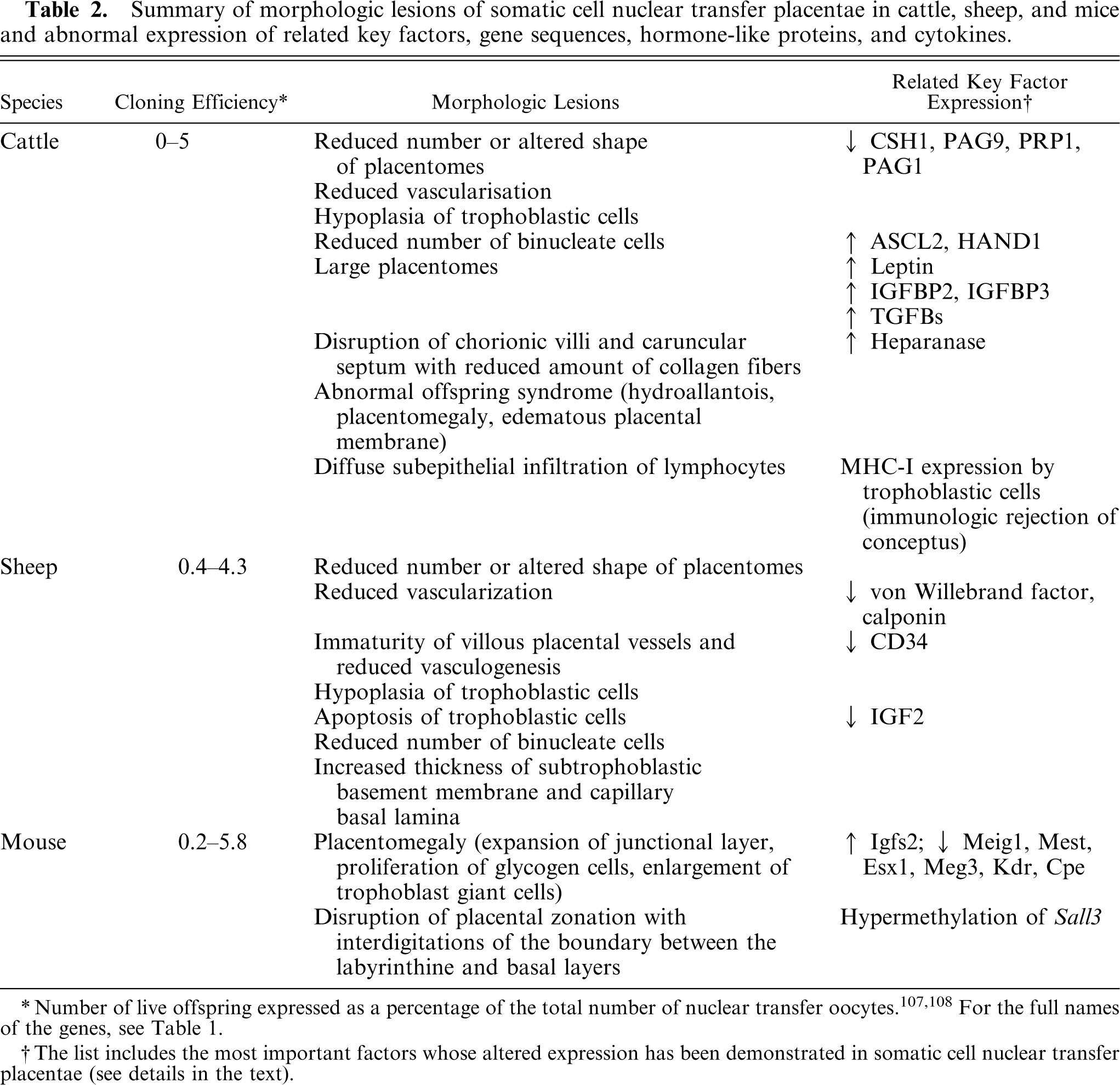
Number of live offspring expressed as a percentage of the total number of nuclear transfer oocytes.107,108 For the full names of the genes, see Table 1.
The list includes the most important factors whose altered expression has been demonstrated in somatic cell nuclear transfer placentae (see details in the text).
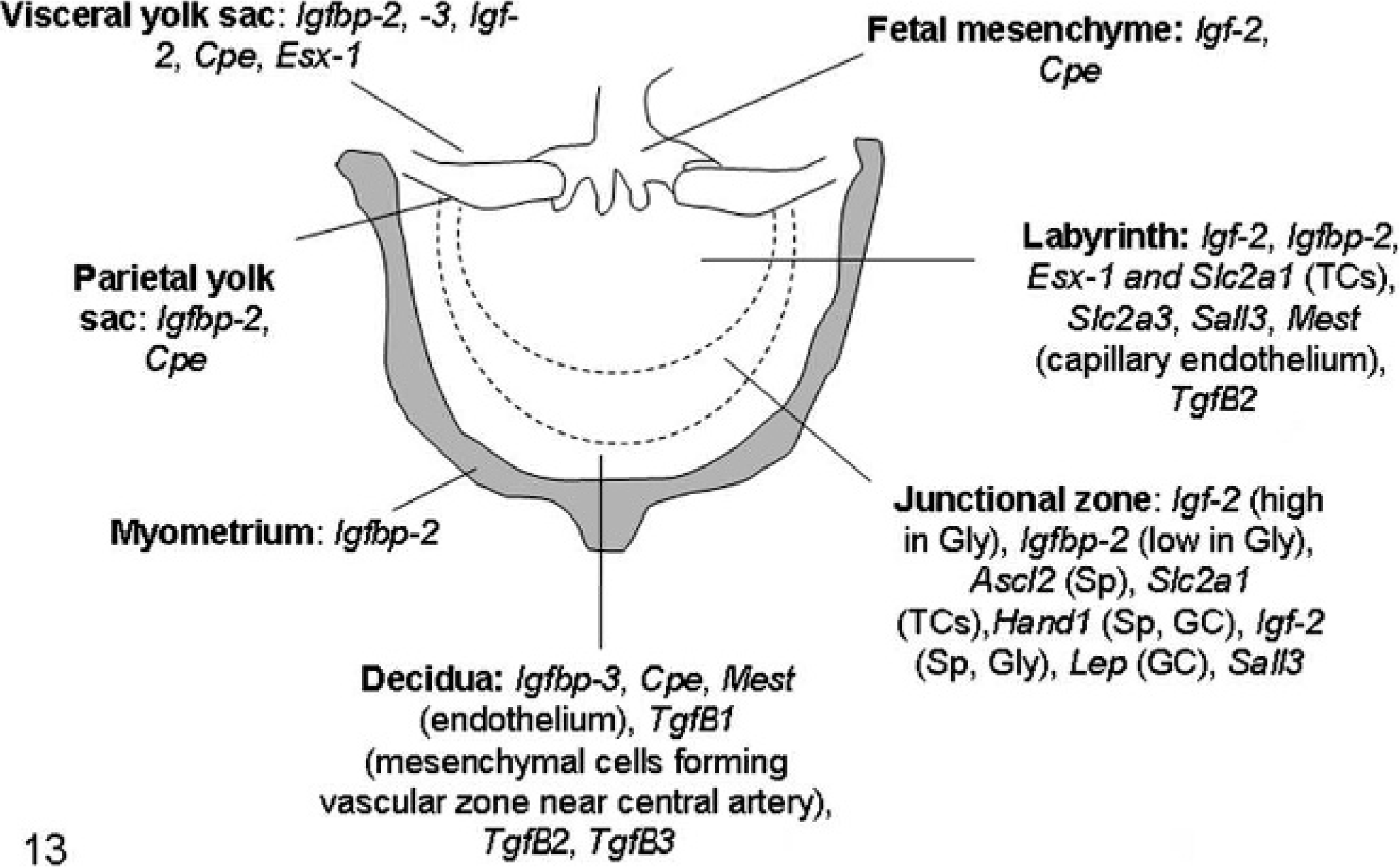
Schematic diagram of mouse placenta illustrating the expression of the most important genes involved in placental pathology of somatic cell nuclear transfer clone pregnancies. For the full names of the genes, see Table 1. GCs: trophoblastic giant cells; Gly: glycogen cells; Sp: spongiotrophoblast; TCs: trophoblastic cells.
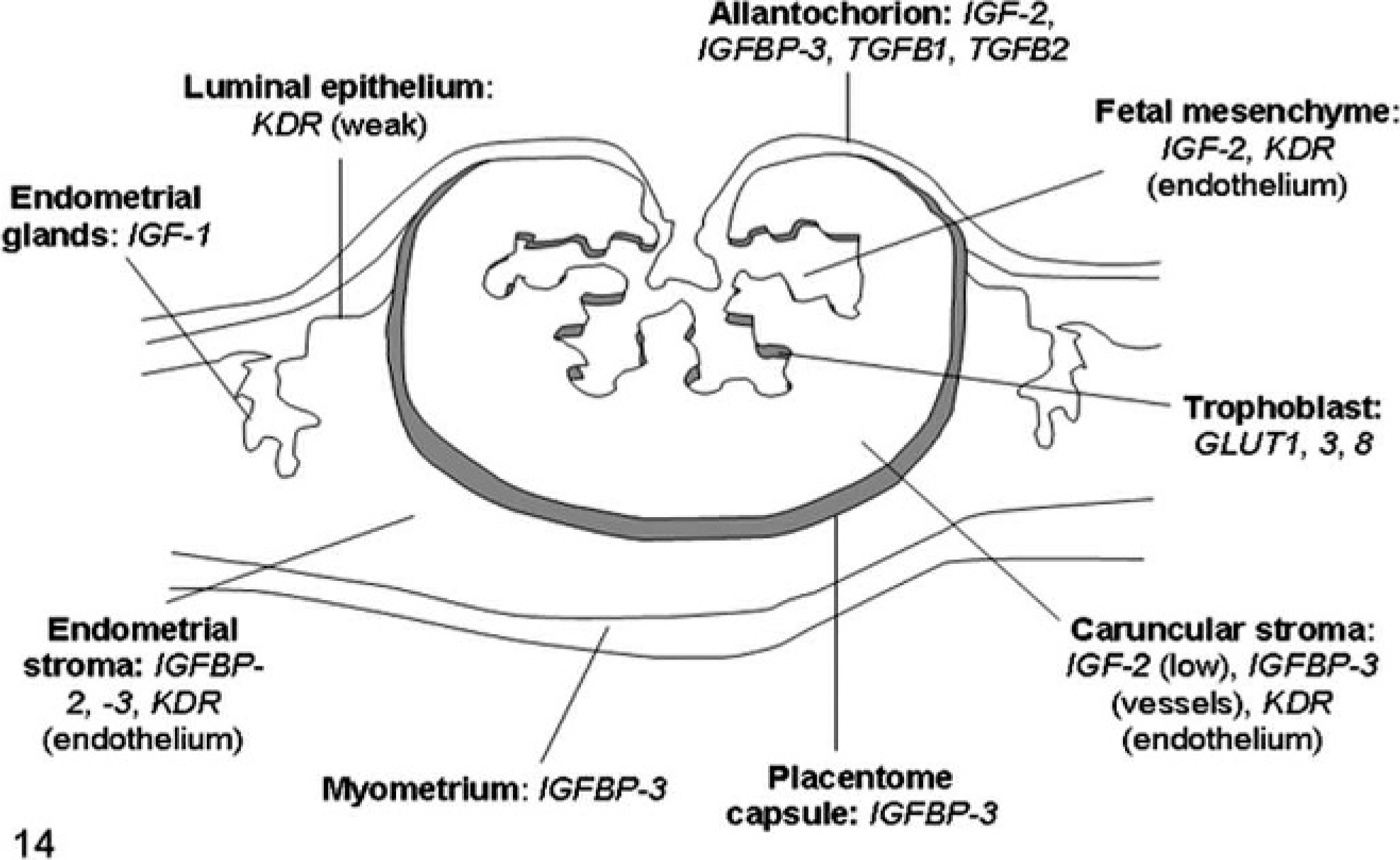
Schematic diagram of sheep placenta illustrating the expression of the most important genes involved in placental pathology of somatic cell nuclear transfer clone pregnancies. For the full names of the genes, see Table 1.
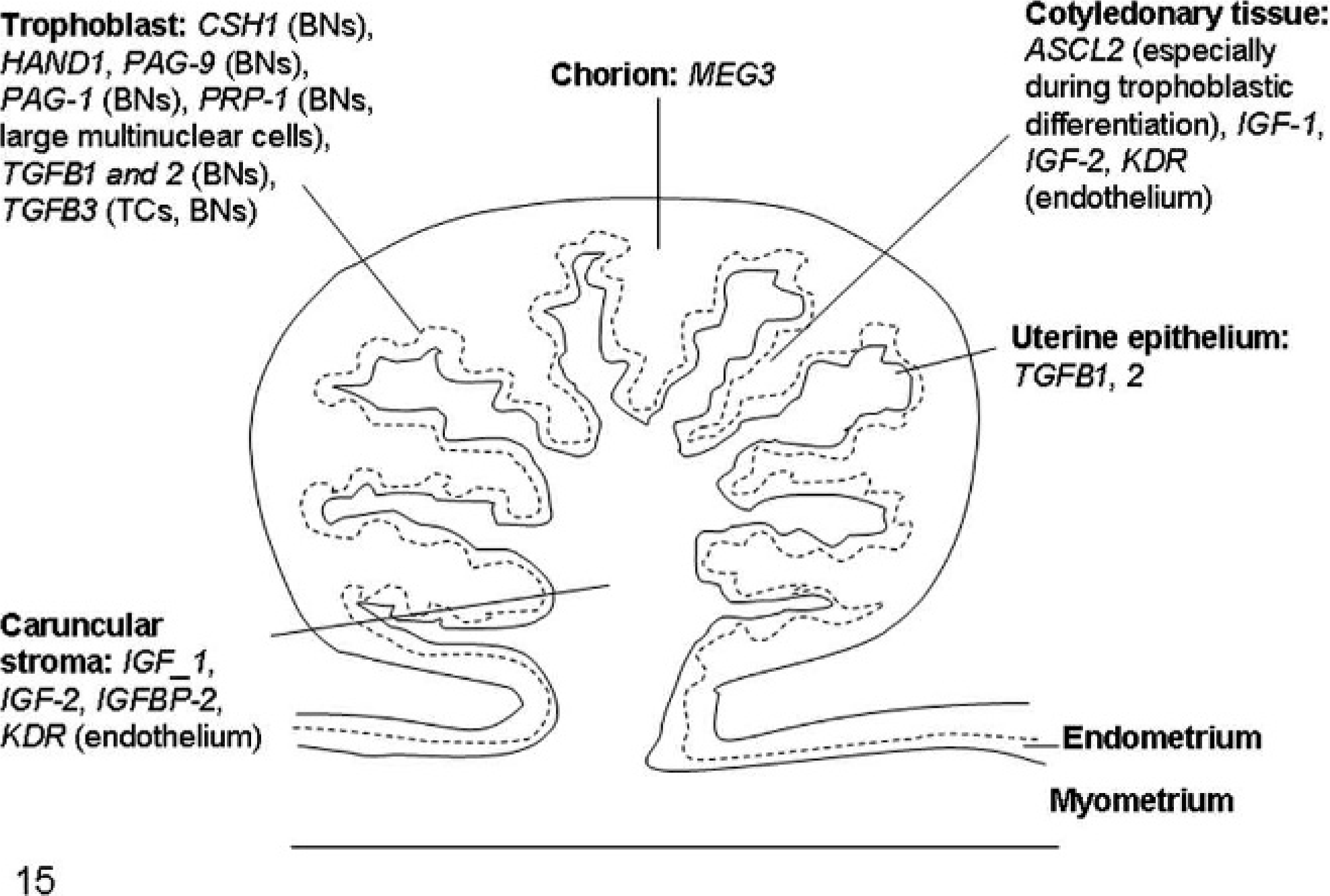
Schematic diagram of bovine placenta illustrating the expression of the most important genes involved in placental pathology of somatic cell nuclear transfer clone pregnancies. For the full names of the genes, see Table 1. BNs: binucleate cells; TCs: trophoblastic cells.
In mice, placentomegaly also has been related to an increased placental concentration of insulin-like growth factor 2 (IGF2), a cytokine promoting cellular mitosis, differentiation, and migration. 30 Strong support for the importance of IGF2 is provided by the findings of small placentae in Igf2 knockout mice and enlarged placentae in IGF2-overexpressing mice. 35, 97 Furthermore, lack of expression of IGF2 receptor (IGF2R), which binds and removes IGF2 from circulation, leads to placental hypertrophy. 58 In cattle, no differences were found in amounts of IGF1 or IGF2 in SCNT placentomes compared with those of controls. 85 However, expression of IGF-binding proteins IGFBP2 and IGFBP3 is significantly increased in placental tissue samples collected from SCNT clone recipient pregnant animals at 50 to 150 days of gestation and characterized by an increase in fetal, maternal, and binucleate cell numbers. 85 In sheep, reduced amounts of IGF2 mRNA were observed from 105 to 154 days of gestation in SCNT placentae, 28 which could promote trophoblast cell apoptosis, inasmuch as IGF2 has high antiapoptotic activity in the pancreatic islet cells of neonatal rats. 83
Transforming growth factor-betas (TGF-Bs) are highly conserved growth factors controlling cell growth and differentiation and tissue remodeling during menstruation, proliferation, decidualization, and establishment of pregnancy through opposite promotory and inhibitory effects. 51 Expression of TGFB1, TGFB2, and TGFB3 mRNAs is significantly increased in the feto-maternal interface of SCNT bovine placentae from 50 to 150 days of gestation compared with samples derived from artificial insemination pregnancies. 87 TGF-Bs can also affect placental structure, resulting in uneven deposition of extracellular matrix (ECM) components, because it stimulates fibroblasts and other cells to produce ECM proteins and cell adhesion proteins (collagen, integrins, and fibronectin), reduces the production of ECM-degrading enzyme (collagenase and heparanase), and upregulates endogenous tissue inhibitors of metalloproteases. 51, 87
Most of the genes deregulated in SCNT pregnancies are imprinted genes involved in growth regulation. Genes down-regulated in placentae of SCNT mice include meiosis-expressed gene 1; mesoderm-specific transcript; and extraembryonic, spermatogenesis, homebox 1 genes in mid-gestation; and maternally expressed gene 3 and kinase insert domain protein receptor in late gestation. 47, 49, 71, 94, 100 Results of gene-targeting studies suggest that the decreased expression of these genes contribute cumulatively to the hyperplastic changes in SCNT placentae. 62, 94
Moreover, down-regulation of carboxypeptidase E is associated with placentomegaly in SCNT mice. 94 Mice homozygous for mutant carboxypeptidase E, which encodes a serine-to-proline mutation at amino acid 202 that abolishes enzymatic activity, show a striking placental phenotype characterized by increased giant and glycogen cells number and giant cell hypertrophy. 95 An additional potential contributor to placental hyperplasia is Esx1, an X-chromosome–imprinted regulator of placental growth and differentiation. 62 Placentae of fetal heterozygous female mice inheriting a null mutant Esx1 allele from their mother (Esx1tm1Bhr /Esx1+), as well as those of hemizygous mutant males (Esx1tm1Bhr /Y) and homozygous mutant females (Esx1tm1Bhr /Esx1tm1Bhr ), have features similar to those of SCNT placentae, including hyperplastic spongiotrophoblast, excessive differentiation of spongiotrophoblast into glycogen cells, and significantly increased placental weight compared with placentae of wild-type littermates on day 15 of gestation. 62, 94, 114
In early bovine SCNT pregnancies, aberrant expression of genes necessary for trophoblast proliferation and differentiation, such as achaetescute complex homologue 2 (ASCL2) and heart and neural crest cell derivatives expressed 1 (HAND1), occurs. 3 ASCL2, an imprinted gene normally expressed by both alleles prior to implantation but maternally expressed after implantation, stimulates trophoblast proliferation and inhibits binucleate cell formation, 36 whereas HAND1, which is expressed at low levels or not expressed in the chorion after implantation, stimulates binucleate cell development. 88 In postimplantantion bovine SCNT pregnancies at 40 days of gestation, fetal cotyledons display higher ASCL2 and HAND1 mRNA than controls, due to abnormal epigenetic reprogramming of these genes or genes regulating their expression. 3 In Rcho-1 trophoblast cell lines coexpressing both Hand1 and Ascl2, giant cell development is inhibited. 19 Thus, although the 2 gene products exert somewhat opposing effects, their simultaneous overexpression leads to failure to stimulate binucleate cell differentiation by trophoblastic cells and reduced binucleate cell numbers. 3
In placentae of nonviable bovine SCNT embryos at 60 days of gestation, cDNA macroarray analysis revealed a twofold increased expression of IGF2, 75 overexpression of which in transgenic mice is embryonic lethal, 98 and less than 50% reduction of hemoglobin alpha 1, hemoglobin alpha 2, and the bovine homologues of spectrin, beta, erythrocytic and spectrin, beta, nonerythrocytic 1. 75
Genetic studies of the underlying mechanisms of placental defects in SCNT sheep have not been as extensive as those in cattle or mice because of the limited knowledge of sheep genetics. Placental gene expression, which is reduced in ovine SCNT placentae at 105 to 154 days of gestation, includes the glucose transporters GLUT1, GLUT3, and GLUT8, with subsequent reduced fetal plasma glucose concentration after 135 days of pregnancy. 28
The underlying cases of SCNT placental pathology are uncertain but usually appear to involve inefficient epigenetic reprogramming of the somatic cell genome by the oocyte cytoplasm. 53, 73 Such reprogramming is necessary for a somatic cell nucleus to reacquire totipotency and become functionally equivalent to that of a zygote, which requires reestablishment of the coordinated pattern of gene expression necessary for embryonic and fetal development. 99 Acquisition of this state by a somatic cell nucleus and subsequent development of the fetus and placenta require global epigenetic changes, including erasure of the somatic pattern of DNA methylation and its progressive reestablishment throughout development.
Epigenetic modification of DNA by methylation in mammals occurs predominantly at CpG dinucleotides and is involved in genome functions, including imprinting, X chromosome inactivation, and genome stability, as well as embryonic development. 9 DNA methylation occurs first during germline development and later during fertilization, with paternal DNA demethylation, particularly dramatic in mice, rats, pigs, and humans, 66 followed by a passive demethylation in preimplantation embryos and de-novo methylation at the blastocyst stage, corresponding to early differentiation events. 99 Species differences in methylation patterns occur: bovine and ovine embryos undergo a much more limited global demethylation during the first cleavage stages after fertilization. 7 In somatic cell cloning, the donor cell genome is compelled to bypass gametogenesis and thus has a different chromatin structure from that of sperm. Aberrant DNA methylation patterns in SCNT embryos and fetuses 11 suggest that most somatic nuclei fail to be reprogrammed, resulting in aberrant gene expression and abnormal phenotypes, particularly in extraembryonic tissues.
Epigenetic perturbations influencing expression of genes involved in embryonic development and fetal growth regulation are responsible for “abnormal offspring syndrome” of SCNT bovine fetuses. 113 Such SCNT fetuses are disproportionately large at 80 days of gestation. Their DNA is globally hypermethylated, 42 suggesting that active demethylation of the embryonic genome is inhibited, perhaps as a consequence of improper chromatin remodeling by factors contained in the oocyte cytoplasm 23 or aberrations in DNA methyltransferase 1 (DNMT1) activity, specifically an abnormal regulation of the 2 DNMT1 forms (oocyte-derived and somatic). Somatic DNMT1 protein is expressed in mouse embryos at the 8-cell stage, whereas this protein is normally not expressed until much later in development. Moreover, abundant cytoplasmic oocyte-derived DNMT1, the only form expressed in the embryo before implantation, is associated in clones with little or no nuclear uptake. 15
In sheep, trophectoderm cells of SCNT blastocysts show abnormally distributed DNA methylation. 7 Because trophectoderm contributes to the trophoblast and chorion components of the placenta and to the inner cell mass-derived allantois, abnormal gene expression associated with perturbed methylation in trophectoderm might contribute to embryo loss and placental defects.
In mice, SCNT placental weight positively correlates with hypermethylation (68 ± 8%) of placental Sal-like 3 locus at 19.5 dpc, compared with low patterns of methylation after natural mating (54 ± 7%), intracytoplasmic sperm injection (53 ± 4%), and in-vitro fertilization (52 ± 6%). 73 The Sal-like 3 locus has a CpG island containing a tissue-dependent differentially methylated region specific to the trophoblast cell lineage. 93 This locus represents the area in which frequent epigenetic errors occur that are believed to be responsible for alterations of placental gene expression resulting in placental overgrowth in SCNT mice.
Aberrant development of SCNT conceptus might also result from chromosomal instability and aneuploidies occurring in in-vitro manipulations. 10, 38, 54 In humans, aneuploidies, such as triploidy, monosomy X, and trisomies of chromosomes 8, 13, or 21, account for 50% of all early-pregnancy failure, and are associated with reduced placental vascularisation, 50 one of the main features of SCNT pregnancies. Epigenetic abnormalities associated with SCNT cloning might also increase the risk of endogenous retrovirus activation, carcinogenesis, and genomic instability. 99 A final possibility is that abnormalities associated with SCNT cloning might lead to immunologic rejection of the conceptus. 21, 45 In a normal bovine pregnancy, major histocompatibility complex class I is expressed by the fifth month in the interplacentomal regions. 21 However, major histocompatibility complex class I was expressed in trophoblasts of bovine SCNT placentae at 35 days of gestation with normal trophoblastic and vascular architecture but 50% embryo mortality. Moreover, the endometrium contained a higher number of diffusely spread subepithelial CD3+ T lymphocytes and larger lymphocyte aggregates in the intercotyledonary and cotyledonary stroma than controls, suggesting a detrimental maternal immune response. 45
Summary
Cloning by SCNT is associated with high rates of fetal loss and abnormal development of the fetus and placenta. SCNT placental pathology has been described in all species studied thus far, although morphologic and molecular features are not consistent across species. The underlying pathogenesis for SCNT placental pathology has not been determined but may result from aberrant expression of the SCNT genome.
Footnotes
∗
References 11, 18, 22, 28, 39, 43, 44, 60, 64, 71, 76–78, 82, 85, 86, 90, 91, 100, 107
Acknowledgements
The authors thank Professor Francesca Rosati, Dr. Aida Corvelli, and Dr. Sabrina Mazzara (University of Teramo) for their help in reviewing the English version of the manuscript and Stylianos Kontos (Department of Comparative Biomedical Sciences, Faculty of Veterinary Medicine, University of Teramo) for helpful assistance. The research of GP and PL is partly covered by European Science Foundation EUROCORES Programme EuroSTELLS, supported by funds from the European Commission Sixth Framework Programme under contract no. ERAS-CT-2003-980409, and by MIUR PRIN 2006. CP, LP, and GP acknowledge funds received from MIUR PRIN 2006 and 60% from the University of Teramo.
