Abstract
Ganglioneuroma involving the brachial plexus, paraspinal ganglia, and cervical-thoracic spinal cord was diagnosed in 2 adult cockatiels (Nymphicus hollandicus). Both birds had a chronic 1-year history of ataxia and perching difficulty. At necropsy, each bird had a unilateral, firm, gelatinous white to tan multilobular mass at the thoracic inlet expanding and partially obliterating the brachial plexus and cervical spinal cord. Histologically, the masses were characterized by a locally infiltrative neoplasm comprised of spindloid cells forming streams and sheets with interspersed distinct neuron cell bodies consistent with ganglion cells. The spindloid cell population was immunohistochemically positive for neurofilament protein in one of the birds.
Ganglioneuroma is a rare neuroectodermal tumor of the peripheral nervous system. Ganglioneuromas generally are considered to be benign neoplasms composed of both mature ganglion cells and nerve fascicles with axons, Schwann cells, fibroblasts, and other connective tissue elements. 4 Ganglioneuromas originate from the cranial and spinal ganglia, adrenal medulla, and enteric nervous system. 5 These tumors have been described in humans, 7 rodents, 9 dogs, 4, 8, 9 cats, 6 pigs, 11 horses, 1 and cattle. 10 In humans, ganglioneuromas often are diagnosed by careful cervical palpation and frequently are associated with elevated serum catecholamine levels proportional to tumor size. 2 Ganglioneuromas can be solitary or multifocal in occurrence and tend to be slow growing and minimally invasive tumors. 9 In the intestinal tract of dogs, these tumors typically have a benign clinical course, with little tendency for recurrence or metastasis following complete surgical excision. 8 Histologically, ganglioneuromas are characterized by a biphasic cell population of closely packed streams of spindloid cells (Schwann cell cords) and tangled nerve fascicles with variable numbers of interspersed, well-differentiated neuron cell bodies (ganglion cells) occurring singly or in clusters. The nerve fascicles generally are positive for neurofilament expression. 5
Cockatiel No. 1 was an 8-year-old female with a history of multiple episodes of difficulty with egg laying (“egg binding”) and perching for 12 months and difficulty defecating for 1 week prior to presentation. At presentation, the bird had marked hypercalcemia and died under anesthesia during a radiographic procedure. A full necropsy examination was performed. The bird was in good body condition with excellent pectoral muscling. An 8 × 5 × 5 mm, firm white multilobular mass was identified adjacent to the spine and right first rib, expanding and distorting the brachial plexus at the thoracic inlet (Fig. 1). Additionally, the oviduct was greatly enlarged, thick walled, and convoluted (hypertrophy and hyperplasia). Gross metastatic foci were not identified.
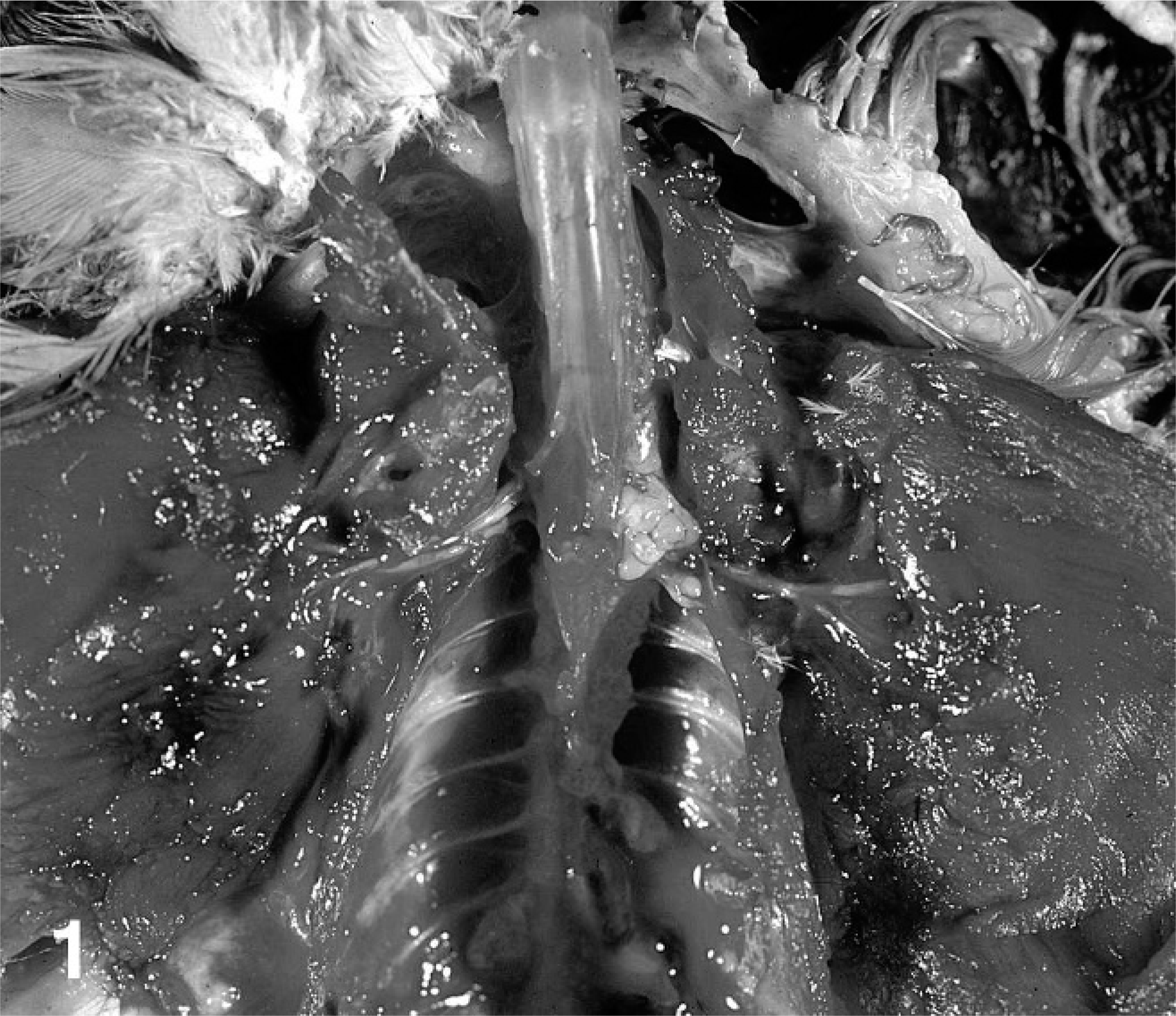
Gross photograph. Unilateral brachial plexus mass; cockatiel No. 1.
The cervical region was fixed en block in 10% neutral buffered formalin for 48 hours prior to decalcification for 12 hours. Transverse sections were embedded in paraffin and 5-µm sections were cut, mounted, and stained with hematoxylin and eosin (HE). Immunohistochemistry was performed for neurofilament protein. Formalin-fixed tissue specimens were cut from paraffin blocks and placed on glass slides. Sections were deparaffinized in xylene, placed in absolute alcohol, and processed in a Dako Autostainer Universal Staining System (Dako Diagnostics, Carpinteria, CA). Endogenous peroxidase was inhibited by 5-minute incubation in 3% hydrogen peroxide in water. Antigen retrieval was accomplished by heating for 30 minutes at 37°C in 0.1% trypsin and 0.1% CaCl2 buffer. Sections were blocked in 5% normal goat sera for 10 minutes at room temperature, and mouse monoclonal IgG1 anti-human neurofilament protein was applied at a 1 : 200 dilution (Dako Diagnostics). The detection system utilized a murine streptavidin–horseradish–peroxidase secondary antibody and 3-amino-9-ethylcarbazole (AEC) chromogen (Dako Diagnostics). Positive control slides consisted of known positive mammalian tissue incubated with the relevant monoclonal antibody. Negative control slides consisted of the test tissue incubated with an isotype and concentration matched, irrelevant monoclonal antibody.
Histologically, the right seventh spinal nerve was greatly enlarged and effaced by a unilateral, 5 × 3 mm, multinodular neoplastic mass. The mass was unencapsulated and invaded, compressed, and focally obliterated the gray and white matter of the spinal cord at the cervical-thoracic junction. In the most severely affected sections, only a thin rim of compressed spinal cord was apparent within the spinal canal (Fig. 2). The examined spinal cord segments cranial and caudal to the neoplastic lesion were histologically normal.
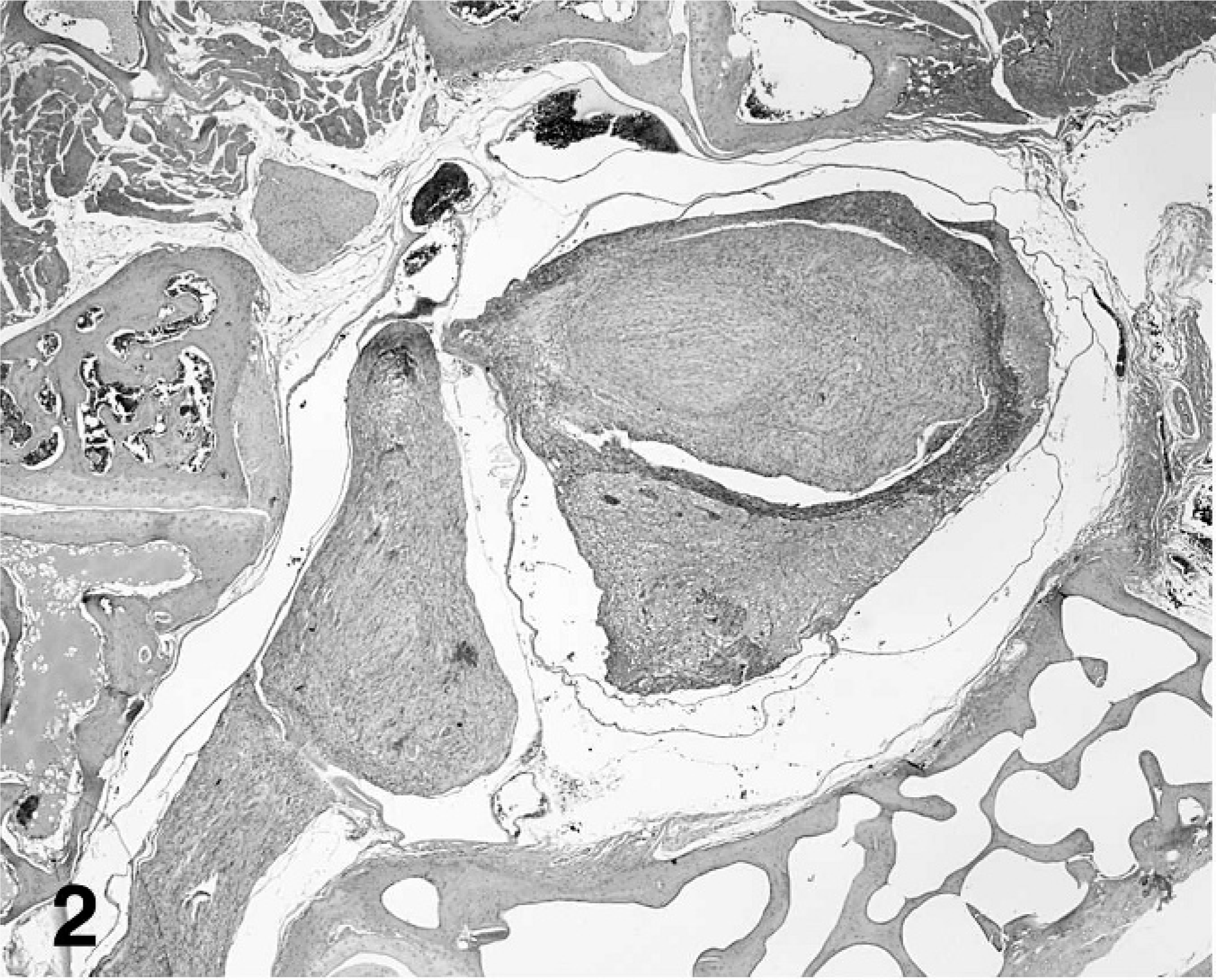
Ganglioneuroma replacing the spinal nerve, brachial plexus and effacing the spinal cord; cockatiel No. 1. HE stain.
The neoplasm was comprised of a majority stromal cell population of interwoven fascicles of spindloid cells and a second population of interspersed neuron cell bodies that had morphology consistent with ganglion cells and among 4 cross sections appeared to occupy 10% of the tumor volume (Fig. 3). The spindloid cell population formed interlacing bundles, had poorly defined margins, minimal eosinophilic cytoplasm, and long, tapered nuclei without mitotic figures. Small islands of neoplastic spindloid cells were present within the spinal cord parenchyma. The second population of well-differentiated ganglion cells was arranged singly or in small often closely packed clusters. Cells were variable in diameter up to 30 µm and without mitotic figures. Occasional regions of the neoplasm had a third pleomorphic cell population characterized by often a high nuclear to cytoplasmic ratio and nuclei with vesicular chromatin, 1 to 2 nucleoli, 1 to 3 mitotic figures per 400× field, binucleation, and anisokaryosis (Fig. 4). By immunohistochemistry for neurofilament protein, numerous linearly arranged processes were immunoreactive, as demonstrated by densely packed, dark red–brown granules (Fig. 5). The ganglion cells were not immunoreactive.
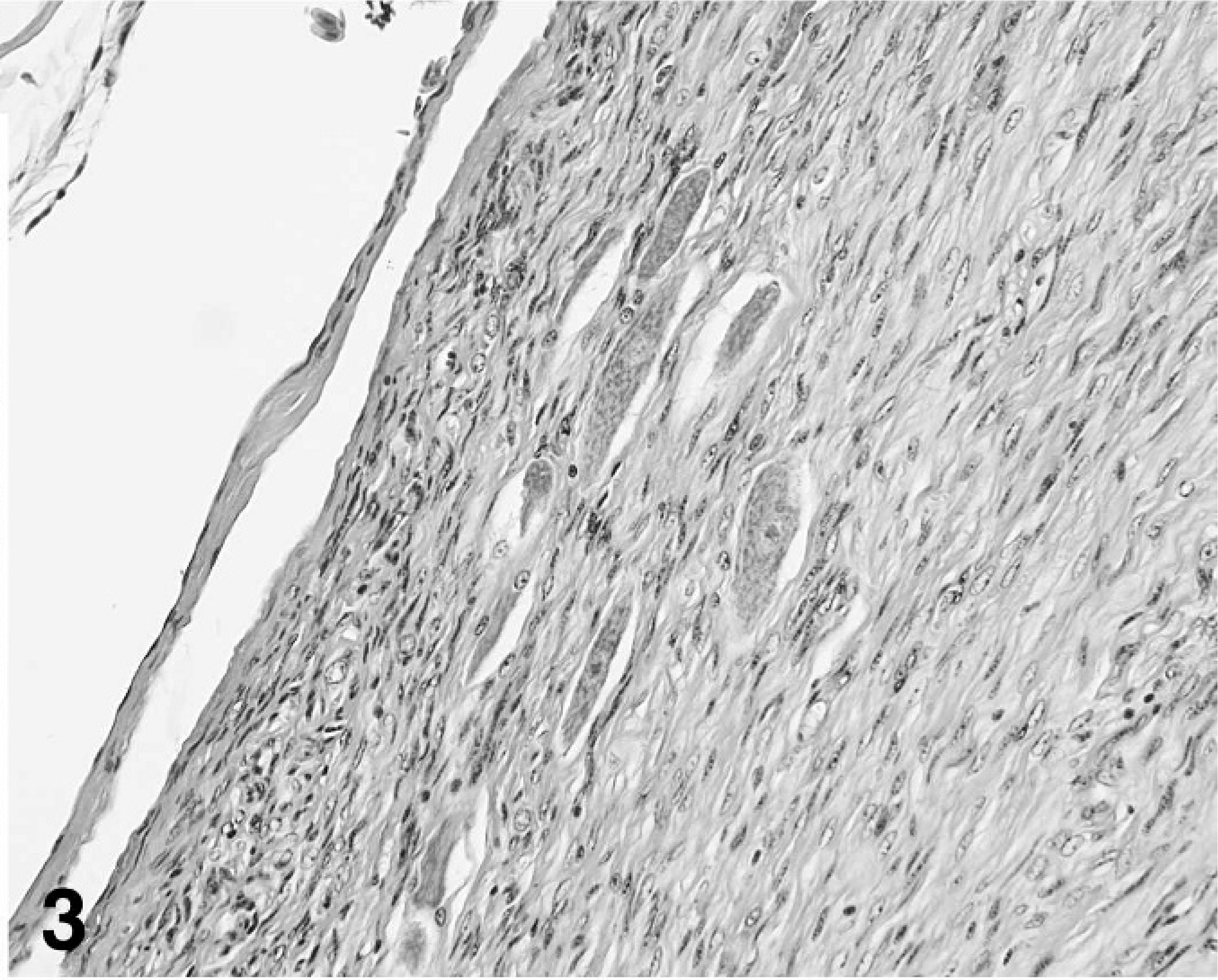
Well-differentiated ganglion cells amidst streams and bundles of spindloid cells, ganglioneuroma; cockatiel No. 1. HE stain.
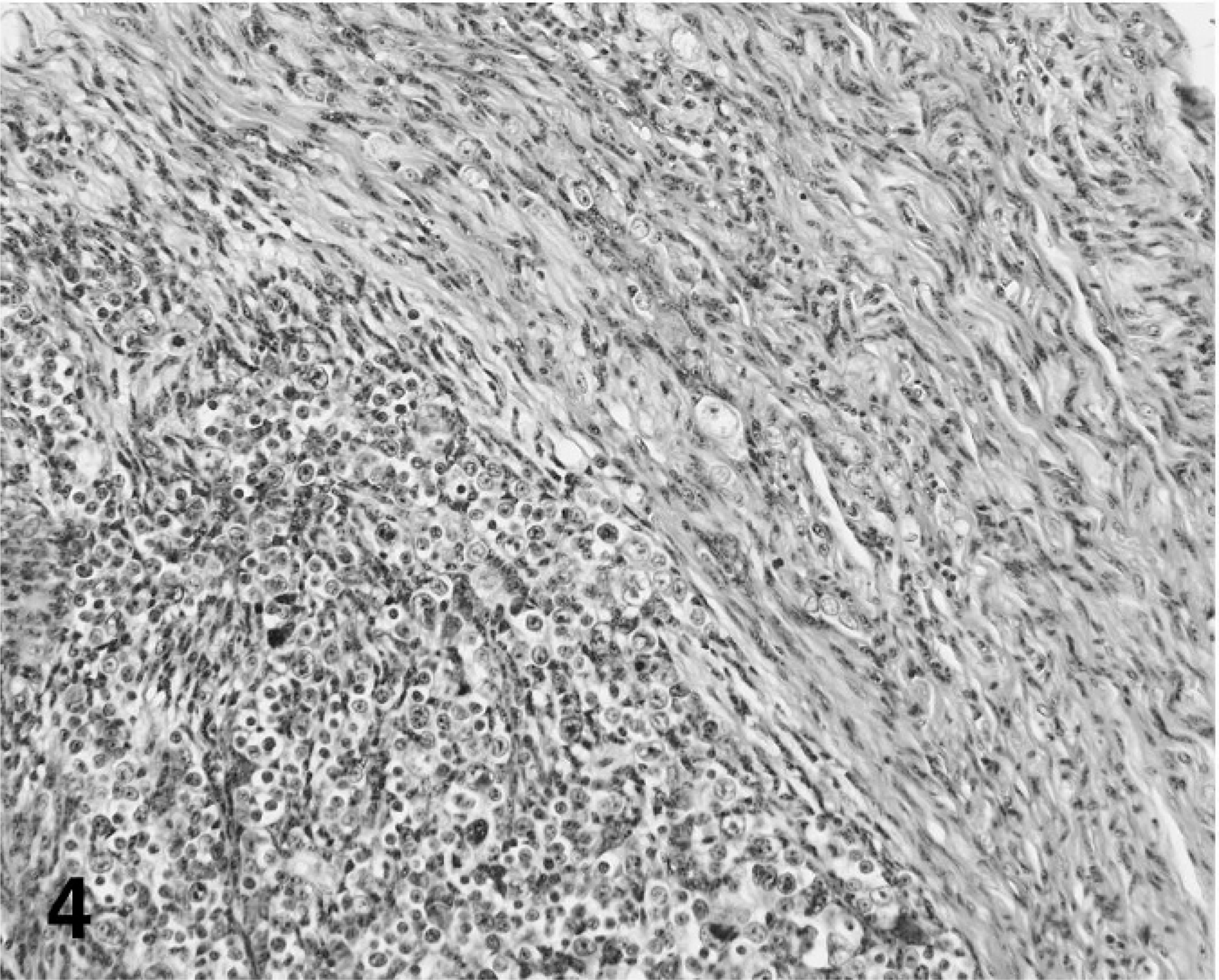
Pleomorphic ganglion cells and adjacent streams and bundles of spindloid cells, ganglioneuroma; cockatiel No. 1. HE stain.
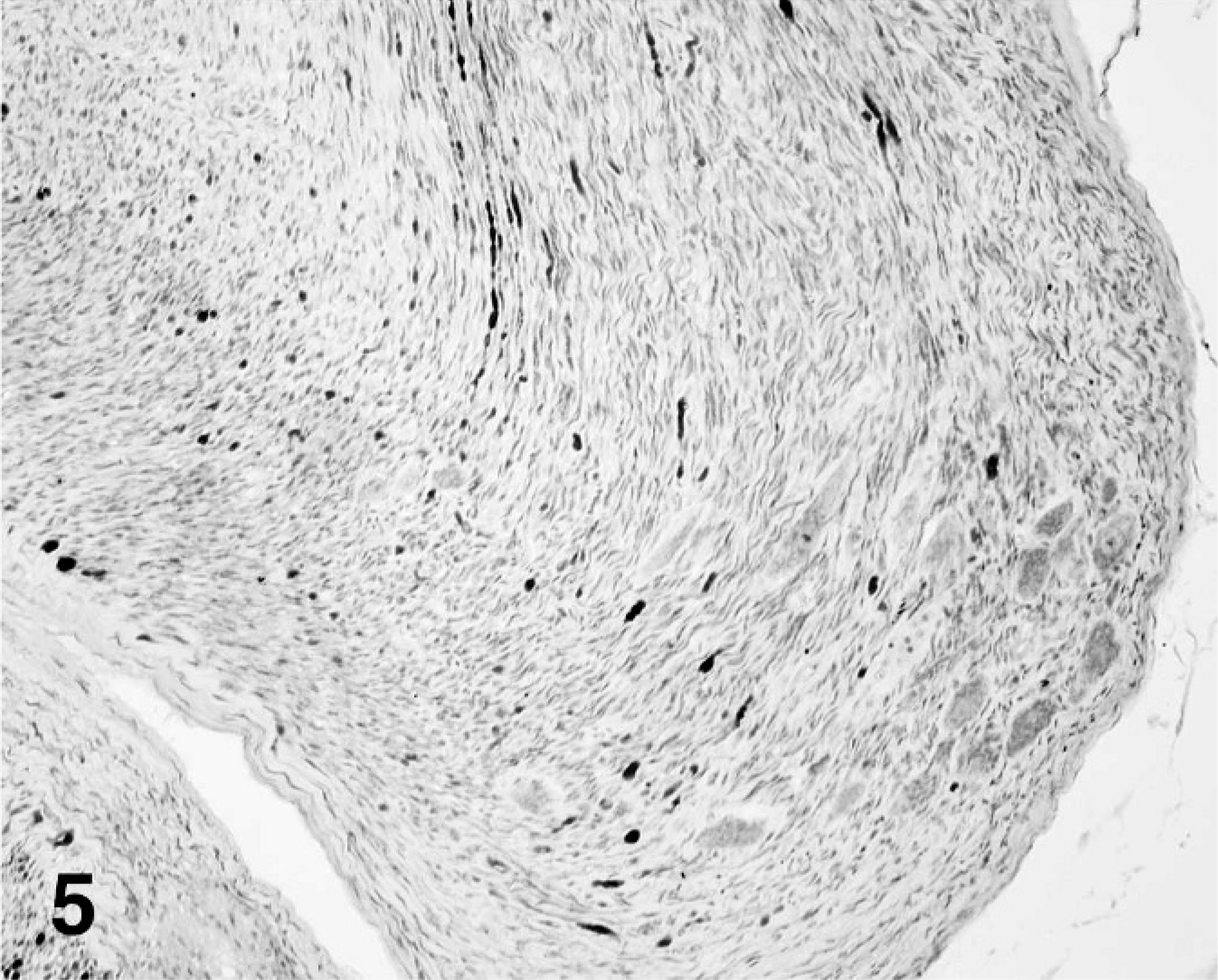
Ganglioneuroma, neurofilament immunohistochemistry stain; cockatiel No. 1.
Cockatiel No. 2 was a 3-year-old male that presented with a 1-year history of ataxia, inability to fly, and feet dragging. Gross lesions were confined to the brachial plexus and cervical-thoracic spinal cord. A mass, 15-mm diameter, which was of similar gross appearance to that in the first bird, expanded and effaced the left brachial plexus. On section, the mass was pale white to yellow and extended partially into the spinal cord, obliterating the cord parenchyma. The spinal nerve proximal to the lesion and 2 spinal nerves distal to the brachial plexus had similar 5-mm diameter nodules. Histomorphology was similar to that in bird No. 1, except that it did not have the pleomorphic cell population.
A diagnosis of ganglioneuroma was based on the long clinical history, gross and histologic features, and results from immunohistochemistry. Although regions of pleomorphic cells were identified in the tumor from cockatiel No. 1, the presence of differentiated ganglion cells within the tumor suggested the diagnosis of ganglioneuroma rather than ganglioneuroblastoma. In dogs and humans, ganglioneuroma has the biphasic cell population with or without the pleomorphic cell population. 3, 4 Despite a usually benign clinical course, the extended duration of clinical signs and the presence of the tumor likely explains the invasion and partial effacement of the spinal cord in both birds. The clinical signs of chronic egg binding, ataxia, and difficulty perching are believed to be the result of partial obliteration and compression of the cervical-thoracic spinal cord. The cause of the hypercalcemia identified at presentation in cockatiel No. 1 was not determined but may have been the result of a paraneoplastic syndrome.
Footnotes
Acknowledgements
We would like to thank Drs. Charles Leathers, Gary Haldorson, and Peter Moore for assistance with the figures.
