Abstract
A 10-year-old, intact, female yellow-naped Amazon parrot (Amazona ochrocephala auropalliata) was examined because of anemia, lymphocytic leukocytosis, regurgitation, and weight loss. A positive fecal occult blood and monoclonal globulinopathy were present. A distended proventriculus and diffusely thickened loops of small intestine with irregular luminal surfaces were identified with contrast radiography and contrast computed tomography. A micro positron emission tomography scan was performed with 18F-fluorodeoxyglucose. Diffuse intestinal T-cell lymphosarcoma was diagnosed based on histopathology and immunohistochemistry of full thickness small intestinal biopsies. The patient was treated with a multidrug chemotherapy protocol with little to no effect. Euthanasia was elected, and intestinal lymphosarcoma was confirmed on histopathology of necropsy intestinal samples; no other organs demonstrated neoplastic infiltration. To the authors' knowledge, no reports are currently available detailing the clinical presentation or diagnosis of diffuse intestinal T-cell lymphosarcoma in any avian species.
Lymphosarcoma (LSA) is the most commonly reported lymphoid neoplasm in parrots and has been reported in many species. Onset typically occurs from 5 months to 30 years, with an average of 8 years. 14 Clinical signs associated with LSA in parrots can include any of the following: periorbital or cutaneous swelling, depression, anorexia, weight loss, paresis, lameness, abdominal swelling, diarrhea, blindness, scant droppings, dyspnea, polydipsia, feather loss, and folliculitis. 14 Lymphosarcoma can be either multicentric or localized, and most reports in parrots detail cutaneous or periorbital disease (France M, Gilson S: 1993, Chemotherapy treatment of lymphosarcoma in a Moluccan cockatoo. Proceedings of the Annual Conference of the Association of Avian Veterinarians, pp. 15–19; Rivera S, Reavill DR, McClearen JR: 2002, Treatment of cutaneous lymphosarcoma in an Umbrella cockatoo (Cacatua alba). Proceedings of the Annual Conference of the Association of Avian Veterinarians, pp. 99–100). 6,13,15 Anemia is a common finding, but a leukemic blood profile is uncommon in parrots with LSA. 14 Changes in biochemical values can be nonspecific and are typically related to the organs affected. Lymphoid neoplasia in poultry can be caused by either a herpesvirus (Marek's disease) or a retrovirus (lymphoid leukosis), 11 but no viral etiology has been associated with lymphoid neoplasia in parrots (Campbell TW: 1984, Lymphoid leukosis in an Amazon parrot–a case report. Proceedings of the International Conference of Avian Medicine, pp. 229–234). 3,13
A 10-year-old, intact, female yellow-naped Amazon parrot (Amazona ochrocephala auropalliata) was referred to the Avian and Zoological Medicine Service at the University of Tennessee, College of Veterinary Medicine (UT-CVM; Knoxville, TN) for evaluation of anemia, lymphocytic leukocytosis, weight loss, and lethargy of approximately 2 weeks' duration. The referring veterinarian diagnosed anemia, an enlarged hepatic silhouette based on radiographs, and an increased population of yeast on fecal Gram stain. The parrot was transfused and prescribed ketoconazole for the gastrointestinal yeast overgrowth.
On presentation, the bird was bright and alert, weighed 481 g (body condition score: 4/5), and no abnormalities were found on physical examination. Anemia (packed cell volume [PCV]: 23%, reference [ref.] interval: 44–56%),17 heterophilia (10.1 × 103/μl, ref. interval: 1.6–3.8 × 103/μl),17 lymphocytosis (11.7 × 103/μl, ref. interval: 0.6–2.8 × 103/μl),17 and a monocytosis (5.4 × 103/μl, ref. interval: 0.0–0.1 × 103/μl)17 were present. Hypoalbuminemia (< 1.0 g/dl, ref. interval: 1.9–3.52 g/dl), 8 hypophosphatemia (2.4 mg/dl, ref. interval: 3.1–5.5 mg/dl),17 elevated creatine kinase activity (409 U/l, ref. interval: 55–345 U/l),17 hypocholesterolemia (176 mg/dl, ref. interval: 180–305 mg/dl),17 and decreased lactate dehydrogenase (LDH) activity (44 U/l, ref. interval: 155–245 U/l)17 were also present. A DNA probe and fluorescent antibody test for Chlamydophila psittaci were negative. A fecal Gram stain showed a mix of Grampositive rods and cocci; no yeast was present.
Numerous diagnostic tests were performed over the next 6 weeks, and the most significant findings are described. The hepatic silhouette again appeared enlarged on survey radiographs. The proventriculus was distended and gastrointestinal transit time was rapid on barium contrast radiographs. Barium reached the ventriculus by the 5-min time point, and slightly irregular filling of the intestines was present at numerous time points. A fecal occult blood test a was positive, and fecal culture was within normal limits. Two bone marrow samples were collected but were nondiagnostic. A serum protein electrophoresis was performed, and a monoclonal spike was present in the beta (β) region (1.5 g/dl, ref. interval: 0.38–0.76 g/dl). 8
Because of the persistent anemia and apparent loss of blood into the gastrointestinal tract, upper gastrointestinal and cloacal endoscopy was performed. No gross abnormalities were seen; a biopsy of the ventricular mucosal wall was collected with spoon-shaped, oval biopsy forceps and showed no evidence of lesions on histopathology. In the 2 days following endoscopy, the patient became progressively anemic (PCV = 14%) and was transfused; no transfusion reactions occurred. Because of a high index of suspicion of infiltrative intestinal disease based on the barium contrast radiographs, contrast computed tomography (CT), micro positron emission tomography (microPET), and an intestinal biopsy were performed.
Thirty minutes following oral iohexol b administration, the CT scan showed that the mucosal surface of the small intestinal bowel was irregular and indistinct. Additionally, a segment of the small intestine had thickened walls with a decreased diameter of the lumen. Findings on CT imaging were consistent with a diffuse, infiltrative intestinal disease such as LSA, inflammatory bowel disease, or mycobacteriosis.
A whole body microPET c scan was performed to determine the extent of disease and to provide a baseline scan that could be compared with posttherapy scans. The patient was injected with 1 mCi (millicurie) of 18F-fluorodeoxyglucose (18F-FDG) into the right jugular vein and then allowed to rest quietly for 60 min. The microPET scan showed diffuse areas of increased radioactivity within segments of the intestines, but no distinct masses were present (Fig. 1A). However, when compared with microPET scans of healthy Hispaniolan Amazon parrots (Fig. 1B), considerable variation in intestinal radioactivity was noted. The intestinal uptake in the clinical patient may represent increased metabolism due to an intestinal infiltrate, but the intestinal radioactivity representing normal variation could not be excluded.
Four days after the microPET scan, the patient was prepared for surgery. Two full-thickness biopsies, 1 from the duodenum and 1 from the jejunum, were taken and submitted for histopathology. Both sections of sampled bowel were markedly thickened with a corrugated appearance to the luminal surface; other areas of bowel that were not sampled were also thickened. Histopathology of both duodenal and jejunal biopsy samples revealed distortion of villar architecture and expansion predominantly of the lamina propria, and to a lesser extent the submucosa, by closely packed neoplastic round cells arranged in sheets (Fig. 2A, 2B). Neoplastic cells did not infiltrate into the epithelium or transmurally into muscle layers. The cells had distinct borders, moderate amounts of eosinophilic cytoplasm, and eccentrically placed vesicular nuclei. The mitotic index was 0–1 per high power field. Special stains (Ziehl-Neelsen and Fites) were negative for acid-fast organisms. Immunohistochemistry (IHC) was performed on biopsy samples and showed approximately 95% of neoplastic cells to be cluster of differentiation 3 (CD3)-positive T-lymphocytes (Fig. 2C, 2D); a psittacine spleen sample used as a control had strong specific CD3 d staining of cells in a pattern consistent with expected distribution of T cells (paracortical expansion around follicular white pulp follicles). CD79a and B-lymphocyte antigen 36 (BLA36) d expression were not obtained in control psittacine splenic tissue or in the neoplastic cells, which made an assessment of B-cell immunophenotype difficult. Based on these findings, the histologic diagnosis was diffuse T-cell LSA. The patient was treated with a multidrug chemotherapy protocol with little to no effect clinically. After approximately 6 weeks of chemotherapy and supportive care, the owner elected euthanasia.
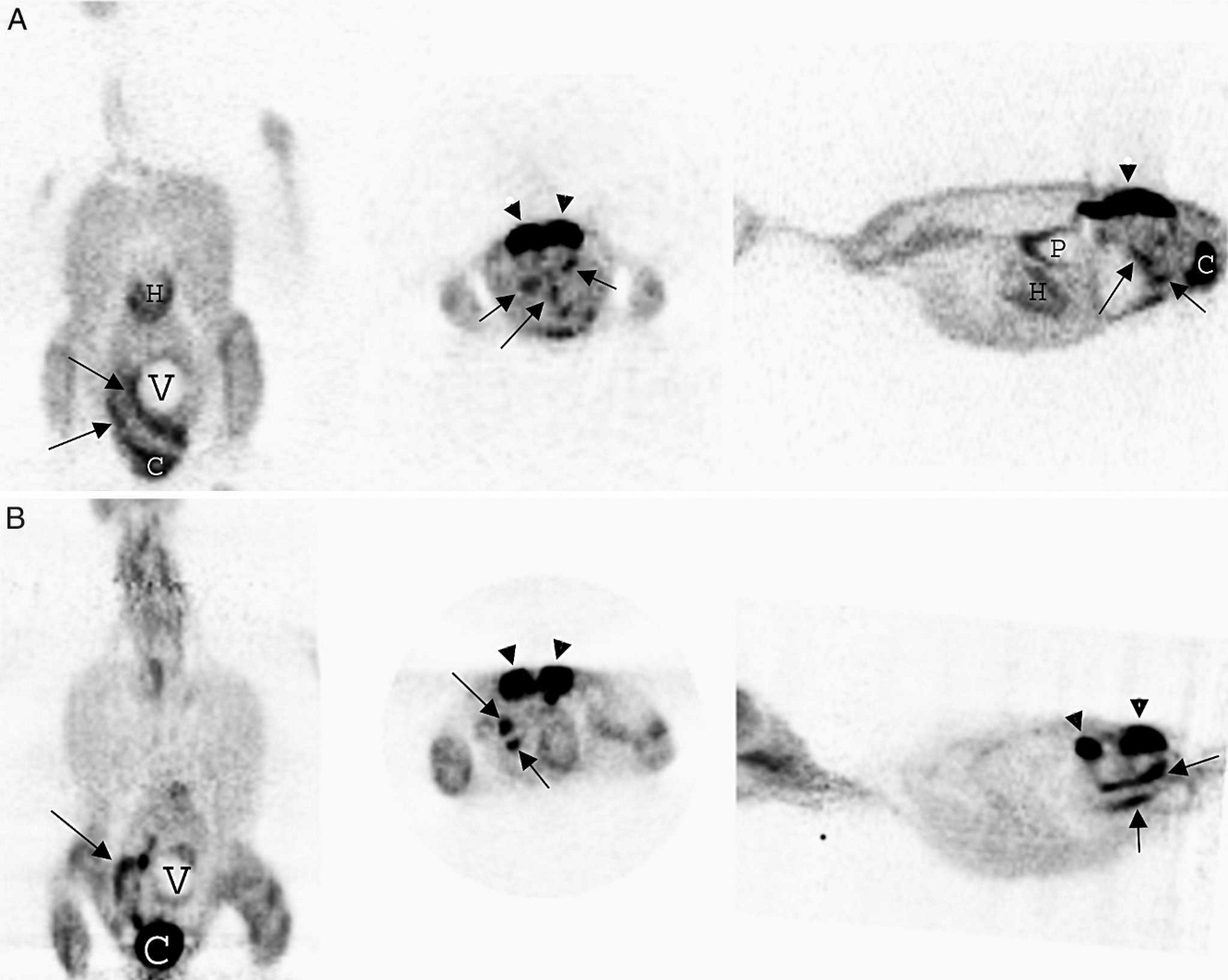
A, micro positron emission tomography (microPET) scan of the yellow-naped Amazon parrot with diffuse intestinal T-cell lymphosarcoma. From left to right, dorsal, transverse, and parasaggital plane images of the parrot show increased radioactivity (dark areas) in numerous segments of the intestine (arrows). Increased radioactivity is also present in the cloaca (C). Other organs present are heart (H), kidneys (arrowheads), and ventriculus (V).
Necropsy confirmed diffuse intestinal thickening that involved the entire length of the small intestine. Histopathology revealed that the intestinal mucosa was diffusely and markedly distended by neoplastic round cells that resembled those in the biopsy sections. The neoplastic cells had not infiltrated into the submucosa or muscular tunic but were confined to the intestinal mucosa. The large intestine was not affected, and no evidence of neoplasia was present in any other organs. The bone marrow showed marked erythropoiesis and myelopoiesis. Immunohistochemistry for CD3 in the bone marrow showed occasional cells with strong staining but no evidence of neoplastic infiltration. Additionally, the intestinal biopsy sites had healed well and showed no evidence of dehiscence. The final diagnosis was diffuse intestinal T-cell LSA.
Clinical signs and diagnostic findings, likely associated with LSA, in this patient included anemia, dark stool (melena), weight loss, regurgitation, enlarged proventriculus, and an irregular surface to the lumen of the intestines. Anemia and blood loss into the gastrointestinal tract have been reported in cats with diffuse intestinal LSA. 12 ,18 Plasma biochemical abnormalities consistent with intestinal LSA, caused by malabsorption of nutrients and possibly a protein-losing enteropathy, included hypoproteinemia, hypoalbuminemia, hypophosphatemia, and hypocholesterolemia.
Fecal occult blood testing was positive in this patient; however, it has not been validated in Amazon parrots, and dietary myoglobin, hemoglobin, and plant peroxidases may give false positives. 5 One study did validate the use of fecal occult blood testing in cockatiels (Nymphicus hollandicus). 7 Additionally, a monoclonal globulinopathy was present, which is still unexplained by the diagnosis of T-cell LSA, as T cells do not typically produce immunoglobulins; the exact protein(s) present in the globulinopathy is still being investigated. Monoclonal gammopathies in mammals are typically associated with a monoclonal line of plasma cells or lymphocytes that produce immunoglobulin or immunoglobulin fragments. 2 Monoclonal globulinopathies have been reported in 2 Amazon parrots with malignant T-cell lymphoma; however, the α1 and α2 proteins were elevated. The nature of the proteins in those 2 parrots was not definitively determined. 4
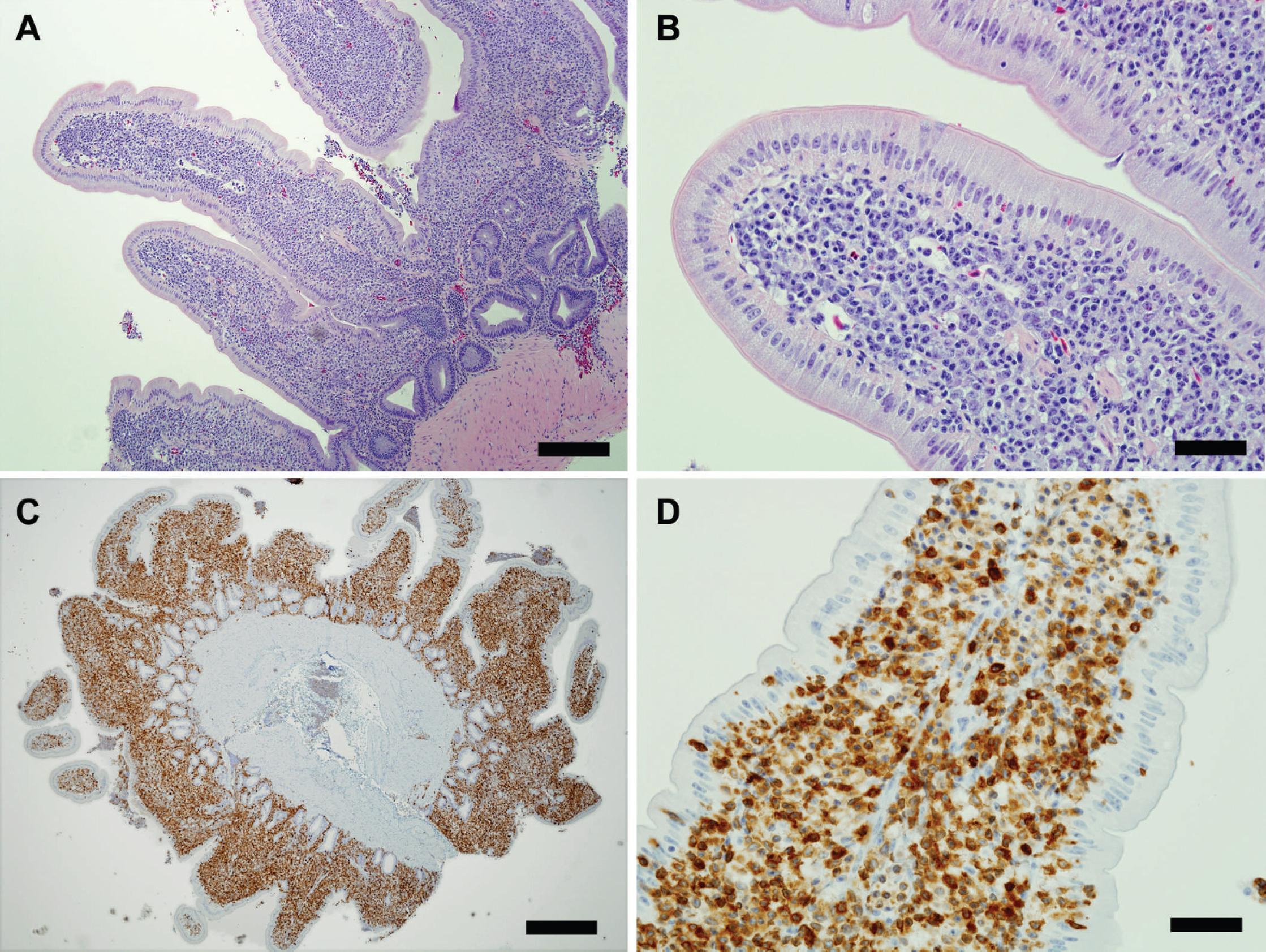
Jejunum; yellow-naped Amazon parrot.
Numerous imaging modalities, including contrast radiography, contrast CT, and microPET, were used to define better the extent of disease and plan further diagnostics in this patient. Contrast CT defined the extensive and infiltrative nature of intestinal disease. Positron emission tomography scans are often used in human oncology cases to stage cancers prior to treatment and to evaluate the response to therapy. To the authors' knowledge, the current case represents the first attempt to use this unique imaging modality clinically in a parrot. The most commonly used radiopharmaceutical is 18F-FDG, which is concentrated and trapped within cells that utilize glucose as their energy substrate. Tissues with higher metabolic rates such as brain, liver, heart, neoplastic lesions, or areas of inflammation will have increased 18F-FDG uptake compared with surrounding tissues. This increased metabolic activity is then detected by the PET scanner as areas of increased radioactivity. 10 The microPET scan revealed increased radioactivity throughout numerous sections of intestine; however, considerable variability in intestinal accumulation of 18F-FDG in healthy Hispaniolan Amazon parrots that is likely caused by reflux from the cloaca into the colon (Souza, Daniel, and Avenell, unpublished data) has been found. Further research is needed to determine the amount of radioactivity present in intestines of normal parrots and the effect of reflux on this radioactive tracer. The information gathered from different imaging modalities allowed for better surgical planning to obtain diagnostic biopsy samples and to rule out the possibility of a single resectable mass.
Definitive diagnosis of intestinal LSA is best achieved with full-thickness intestinal biopsies. Histopathology of affected tissue reveals intestinal villi that are distended with lymphocytes and that may progress to transmural spread. Neoplastic lymphocytes can vary from mature, well-differentiated cells to immature blastic cells; diagnosis is often made by the presence of a monomorphic population of lymphocytes. Inflammatory bowel disease is rare in birds but is characterized by a more mixed population of lymphocytes and plasma cells that do not distort tissue architecture.
Immunohistochemistry can be performed to immunophenotype neoplastic lymphocytes, differentiating
Regurgitation is a common presenting complaint in avian medicine and can be caused by numerous factors, including proventricular dilatation disease, gastrointestinal yeast infections, inappropriate feeding, and foreign bodies.16 In the current study, hematology, plasma biochemistry, fecal occult blood, and imaging results were consistent with diffuse infiltrative intestinal disease, and confirmation of LSA was achieved with histopathology and IHC of fullthickness duodenal and jejunal intestinal biopsies. Cutaneous or periorbital LSA is the most common presentation in parrots, and intestinal LSA, with no other organ involvement, is unusual. When common causes of regurgitation are ruled out and other signs associated with intestinal disease are present, intestinal LSA should be considered a differential diagnosis.
Acknowledgements. The authors thank David W. Townsend, PhD, of the Preclinical Imaging Laboratory at the University of Tennessee Health Science Center; Jennifer Stafford, DVM; Michael Lutz, DVM; Tarah L. Hadley, DVM, DABVP (Avian); Carolyn Cray, PhD; and Linden Craig, DVM, PhD, DACVP.
Footnotes
a.
Quik-Cult, Laboratory Diagnostics Company Inc., Morganville, NJ.
b.
Omnipaque®, Amersham Health Inc., Princeton, NJ.
c.
microPET Primate P4, Siemens Medical Solutions USA Inc., Malvern, PA.
d.
Dako North America Inc., Carpinteria, CA.
