Abstract
A tumor behind the left eye in a female Crj:CD(SD)IGS rat was investigated histopathologically, immunohistopathologically, and electron microscopically. The tumor invaded and destroyed orbital tissues and bones. It consisted of various tumor cells; namely, spindle-shaped, epithelioid, anaplastic melanoma cells, and had prominent eosinophilic cytoplasm and nuclei with a greater variation in size. Immunohistochemically, almost all of the tumor cells were positive for antimelanoma, PNL2 antibody. Ultrastructurally, the tumor cells were rich in small vesicles containing fine granules and filamentous structures. This is the first report describing an amelanotic melanoma in the head of an albino rat.
Keywords
In albino rats, the predilection sites of spontaneous amelanotic melanomas have been reported to be the uveal tract and the skin of the pinna, eyelid, scrotum, and perianal/tail. 5, 6, 8– 10 However, to the author's knowledge, no spontaneous amelanotic melanoma originating from sites other than the above-mentioned ones has been reported yet.
We confirmed that antimelanoma, clone PNL2 (PNL2), has high specificity for normal and neoplastic melanocytes in albino rats and demonstrated that it is a useful reagent for diagnosis of amelanotic melanomas in the strains examined. 3 In the present study, we investigated a tumor that was observed behind the left eye of an albino rat using PNL2. Consequently, the tumor was diagnosed as an amelanotic melanoma. Although the predilection sites of melanomas have been reported in the past, 5, 6, 8– 10 this is the first report of an amelanotic melanoma in this region of an albino rat.
The tumor arose in a female Crj:CD(SD)IGS rat that was used as a control, in which the vehicle was administered by gavage in a 104-week toxicity study. This animal was supplied by Charles River Japan, Inc. (Kanagawa, Japan). The animal was housed and cared for according to the principles outlined in the guidelines for the care and use of laboratory animals prepared by the Japanese Association for Laboratory Animal Science and our laboratory.
The rat presented progressive proptosis of the left eye at 42 weeks of age. Turbidity was recognized in the vitreous body at 45 weeks of age. Tachypnea was observed at 60 weeks of age, and this animal died at 67 weeks of age. An autopsy was performed a few hours after death.
All tissues were fixed in 10% neutral-buffered formalin and were embedded in paraffin. Deparaffinized sections were stained with hematoxylin and eosin, and the Schmorl method and the Masson-Fontana's technique for melanin detection were applied. The tumor was stained with anti-human melanoma, clone PNL2 (PNL2, Dako Japan Ltd., Kyoto, Japan), anti-Melan A, clone A103 (Melan A, Dako Japan Ltd.), and anti-S100 (Dako Japan Ltd.) according to previously reported methods. 3 The canine cutaneous benign melanoma observed in the previous study 3 was used as the positive control for this study, as it contained numerous melanin granules positive for PNL2. For the negative controls, the above-mentioned primary antibodies were omitted. For the electron microscopic examination, blocks (1 mm3) of the formalin-fixed samples were postfixed with 1% phosphate-buffered osmium tetroxide. Then, the samples were routinely processed and embedded in resin. Ultrathin sections were stained and examined by a Hitachi H-7600 transmission electron microscope (Hitachi, Ltd., Tokyo, Japan).
Macroscopically, the tumor was observed behind the left eye; it filled the orbital cavity and aggressively invaded the bones circumferentially. It invaded and destroyed the left orbital tissues and bones (the frontal and sphenoid bones), infiltrated into the nasal cavity, and penetrated to the right orbital cavity (Figs. 1, 2). In addition, it extended to the intracranial cavity through the optic canal and then proliferated at the anterior basal region of the brain. However, the brain could easily be taken from the intracranial cavity, and we did not recognize any adhesion of the tumor to the brain. Though the cornea of the left eye could be observed, the eyeball was buried in the mass and could not be recognized (Fig. 3). The tumor was white to tan colored and friable. Metastatic lesions in the lung, which were approximately 2 to 5 mm in diameter, were recognized as white multiple nodules. As for the other findings, atrophy of the thymus and spleen and symptosis were observed.
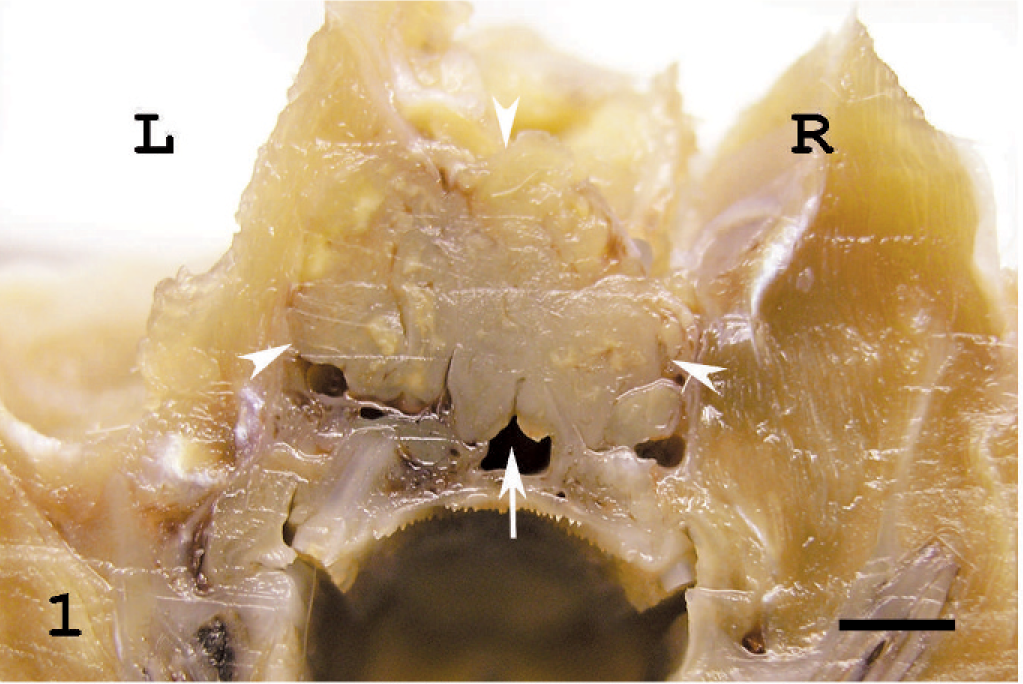
The head of a rat showing melanoma in transverse section. The tumor is white to tan-colored and friable and is not encapsulated (arrow heads). It invaded and destroyed the left orbital tissues and bones (frontal and sphenoid bones), infiltrated into the nasal cavity and nasopharynx (arrow), and penetrated to the right orbital cavity. A part of tumor mass containing the left eyeball and the orbital tissues is removed. L: left. R: right. Bar = 2 mm.
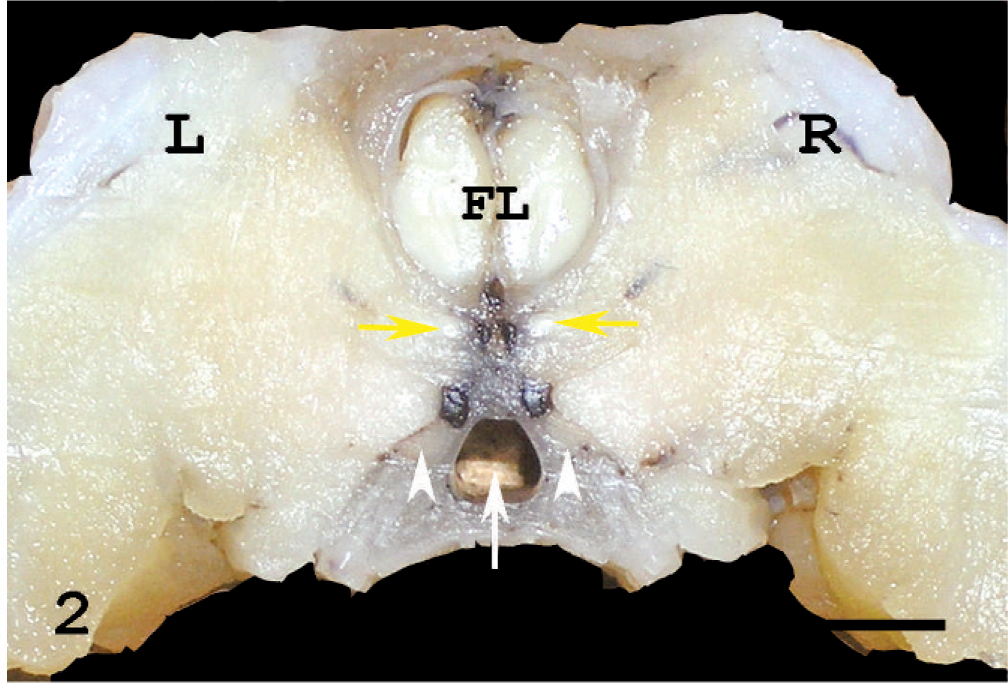
The head of a normal rat in transverse section, corresponding to that in Fig. 1. The optic nerve (yellow arrows), harderian glands (asterisks), maxillary nerves (arrowheads), and nasopharynx (arrow) are visible. L: left. R: right. FL: frontal lobe of the brain. Bar = 2 mm.
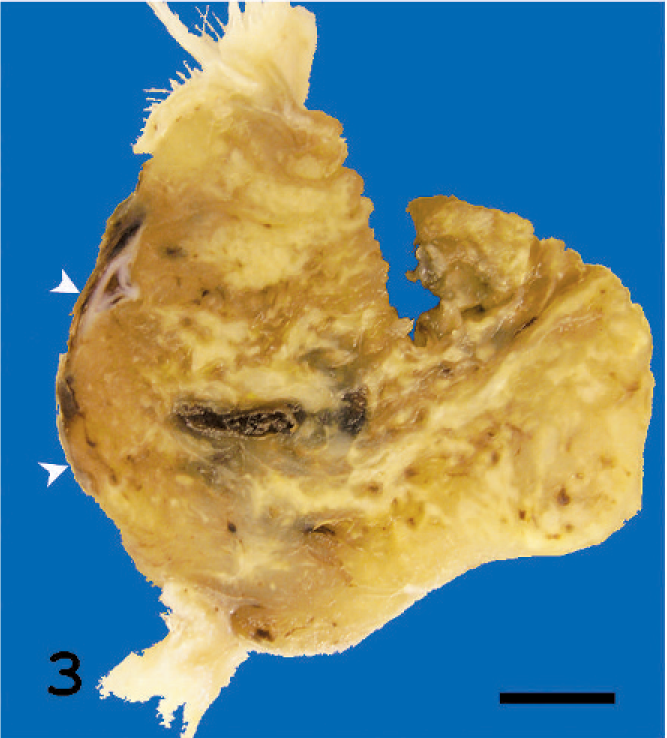
Sagittal section of the tumor tissue behind the left eye. It is a tan-colored tumor with a necrotic area. Though the cornea of the left eye is visible (arrow heads), the eyeball is buried in the mass and cannot be recognized. Bar = 3 mm.
In the superficial areas of the tumor, the cornea, retina, and sclera atrophied considerably due to the pressure of the mass (Fig. 4). Areas of necrosis were scattered in the tumor (Fig. 4). The tumor consisted of variable tumor cells that were mainly composed of eosinophilic cytoplasm and chromatin-rich nuclei with a large variation in size. In the spindle-shaped melanoma cell area, the tumor cells were arranged in an interlacing fascicular pattern. Moreover, they showed mild nuclear pleomorphism (Fig. 5). In the epithelioid melanoma cell area, the tumor cells were composed of round to oval, loosely adherent large cells with pale staining. Their cytoplasm was often vacuolated and nuclei were pleomorphic (Fig. 6). In the anaplastic melanoma cell area, the tumor cells revealed large bizarre nuclei and highly pleomorphic cytoplasm (Fig. 7). No pigment was seen in the cytoplasm of any tumor cells. Moreover, no melanin was observed according to the Schmorl method or Masson-Fontana's technique. Mitotic figures were observed but were extremely rare (1 to 2 mitotic figures in 10 high-powered fields [HPF]). While the boundaries with the tissues other than the brain were not clear, the tumor was sharply demarcated from the brain. Based on that fact, we confirmed that the brain was not infiltrated by the tumor. Immunohistochemically, the cytoplasm of almost all tumor cells was positive for PNL2 with clear granular immunoreactivity (Fig. 8) and for S100, but negative for Melan A. In the positive control canine melanoma cells, 3 not only melanin granules but also spindle tumor cells with no melanin granules were stained. None of the negative controls were stained. Ultrastructurally, the tumor cells observed in this study were rich in small vesicles. These vesicles were characteristically round to ellipsoid and had a single membrane-bound structure containing fine granules and filamentous structures (Fig. 9). Neither prominent tumor cell process nor desmosomes was observed, only desmosome-like structures were observed (Fig. 9). The nodules in the lung were also examined histologically and immunohistochemically. Since the spindle-shaped tumor cells in the nodules were positive for PNL2 as in the primary tumor behind the left eye, the nodules were concluded to be metastases from the primary tumor.
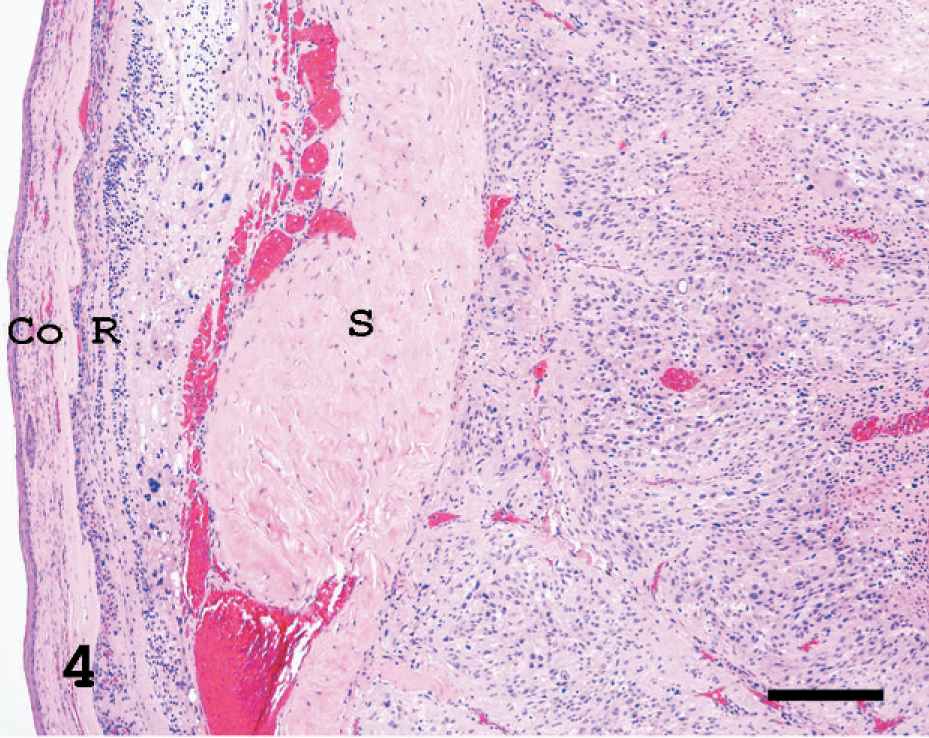
The tumor behind the left eye. In the superficial areas of the tumor, the cornea (Co), retina (R), and sclera (S) are pushed aside by the mass. HE. Bar = 28 µm.
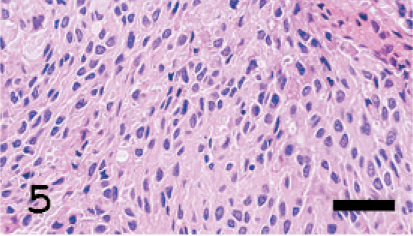
The tumor cells are arranged in an interlacing fascicular pattern, showing mild nuclear pleomorphism. HE. Bar = 2 µm.
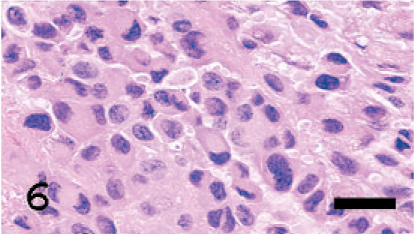
In the epithelioid melanoma cell area, the tumor cells are composed of round to oval, loosely adherent large cells with pale staining cytoplasm. HE. Bar = 3.2 µm.
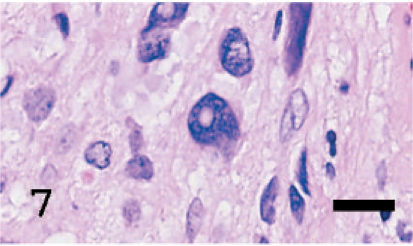
In the anaplastic melanoma cell area, the tumor cells reveal large bizarre nuclei and are highly pleomorphic. HE. Bar = 5 µm.
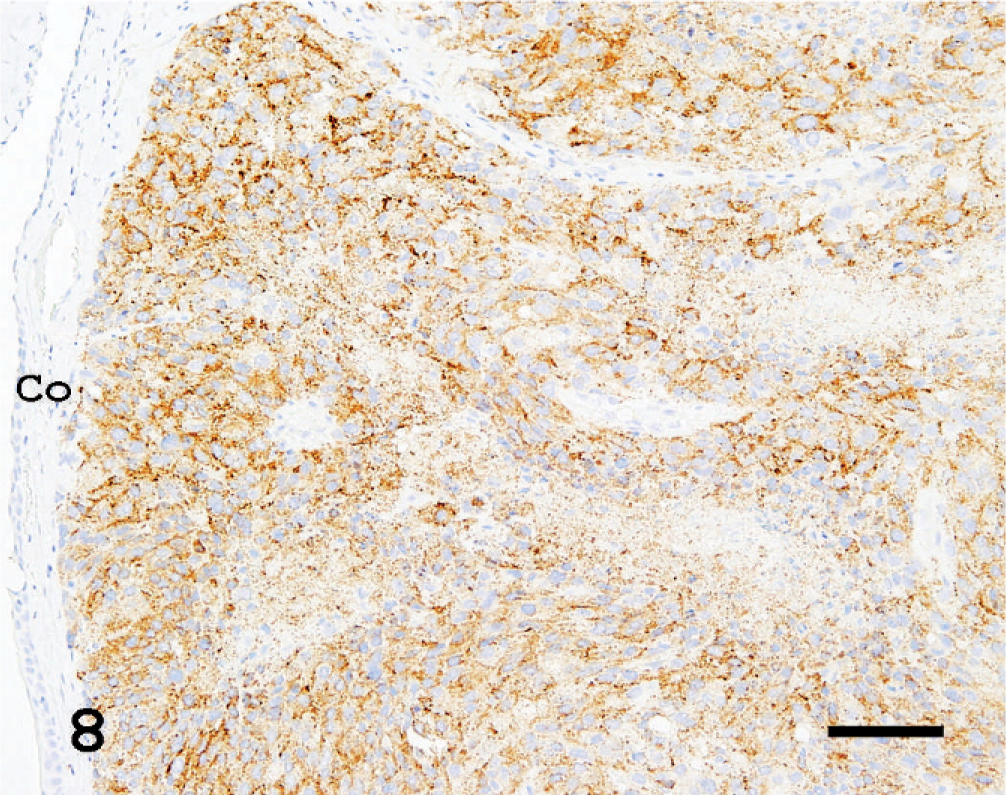
The tumor behind the left eye. PNL2 consistently showed moderate finely granular cytoplasmic staining in most of the tumor cells with no background staining. Co: cornea. LSB method, counterstained with Mayer's hematoxylin. Bar = 20 µm.
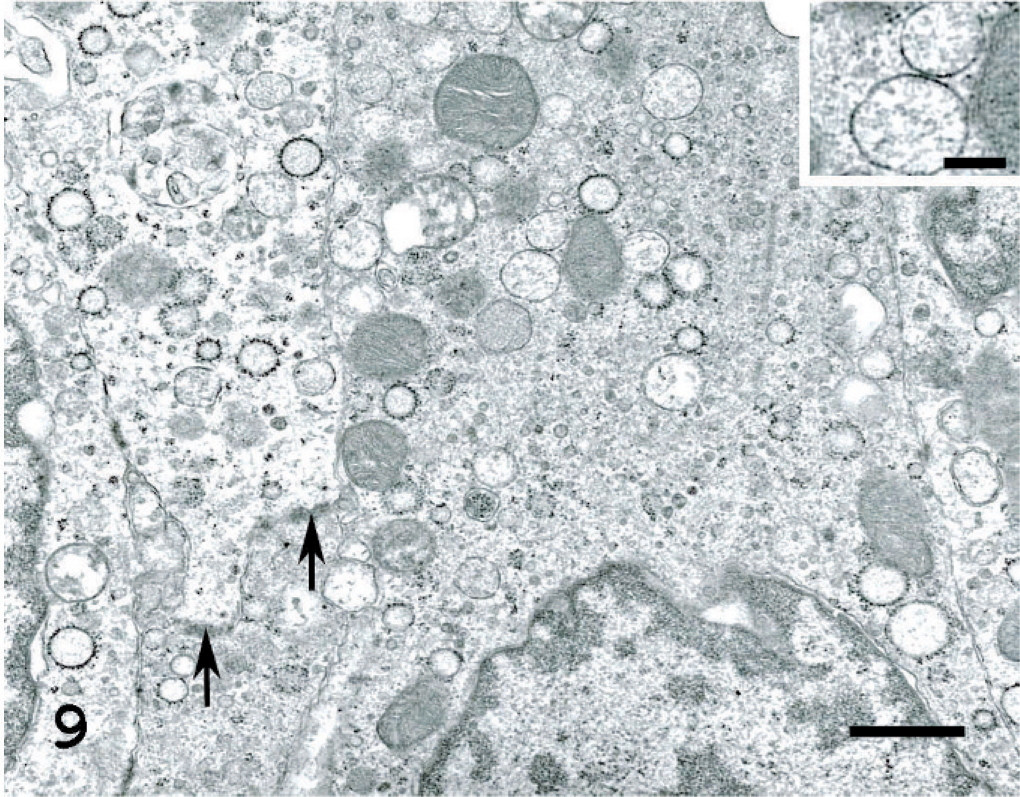
The tumor behind the left eye. The tumor cells are rich in small vesicles. These vesicles are characteristically round to ellipsoidal and have a single membrane-bound structure containing fine granules and filamentous structures inside (Inset: Bar = 31 nm). Neither prominent tumor cell invasion process nor desmosomes was observed, only desmosome-like structures were observed (arrows). Uranyl acetate and lead citrate. Bar = 125 nm.
Spontaneous melanomas in the head of pigmented rats (Brown Norwegian Rat) have been reported. 4 In these cases, the tumor cells were filled with melanin pigment, but areas of cells without cytoplasmic pigment were also observed. The morphologic characteristics of the tumor cells of the present case are similar to those of pigmented cells observed in the above-mentioned cases. However, the occurrence of melanoma behind the eye in an albino rat has not been previously reported. This is the first report describing an amelanotic melanoma behind the eye of an albino rat.
In this case, no premelanosomes in the tumor cells could be confirmed by the electron microscopic examination. However, a large number of small vesicles containing fine granules and filamentous structures were recognized. It has been reported that typical premelanosomes in melanomas were observed less frequently in albino rats, whereas granular bodies similar to primary lysosomes, but not lysosomes, were often noted in the cytoplasm. 6 Furthermore, in amelanotic melanomas of hamsters, such primary lysosome-like structures were found in the cytoplasm of tumor cells. After these tumor cells were serially transplanted in hamsters, many melanosomes could be induced in the cytoplasm by an appropriate treatment of primary culture cells. 1 The small vesicles observed in this case were strikingly similar to these vacuoles (granular and primary lysosome-like bodies). Accordingly, the small vesicles observed in this case were estimated to be premelanosomes (stage I melanosomes).
When spindle cell tumors occur in the predilection sites (uveal tract and the skin of the pinna, eyelid, scrotum, and perianal/tail) in albino rats, 5, 6, 8– 10 pathologists must consider the possibility that the tumors represent amelanotic melanomas. To make an exact diagnosis, further ultrastructural investigation is necessary. Namely, it is difficult to make a diagnosis of amelanotic melanomas in albino rats without an electron microscopic examination, since there is no effective antibody for diagnosis at present. Moreover, spindle cell tumors in the above-mentioned sites may be diagnosed as sarcoma NOS or undifferentiated sarcomas, if no premelanosomes are observed in an electron microscopic examination. The previous study evaluated the immunoreactivity for anti-PNL2 antibody in 11 amelanotic melanomas of albino rats. 3 At that time, we did not show the results of the electron microscopic examination for case No. 11, because we could not find any premelanosomes and, therefore, were unable to diagnose this case as amelanotic melanoma. Afterwards, a more detailed investigation was performed for case No. 11. Consequently, it was confirmed that premelanosomes were not observed in any tumor cells. The case in this study and case No. 11 were diagnosed as amelanotic melanomas because they were stained positive for PNL2, even though we could not observe premelanosomes in electron microscopic examination. If the cases that were tentatively diagnosed as sarcoma NOS or undifferentiated sarcomas are reexamined with PNL2 immunostaining, some of them may be diagnosed as amelanotic melanomas.
It is obvious that the tumor observed in this study arose from behind the left eye. Primary melanomas at this site are commonly observed to protrude into the eyeball. 7 However, in this case it proliferated from behind and pushed the eyeball to the front. In humans, the occurrence of this type of melanoma is extremely rare. 2 As for this study, it was impossible to identify whether the primary lesion was the optic nerve because it could not be recognized in the orbital cavity.
Footnotes
Acknowledgements
We thank Takayoshi Ito, Masayo Onozawa, Chie Shiromaru, Misako Morino, Tomomi Sakuma, Kiyoe Hosoya, Yuuki Ikeda, and Miki Kozasa for excellent technical support and Steve Yamakami and Ai Ishii for language editing. We thank Yuki Kamihara from Dako Japan, Inc., for her advice and support.
